INTRODUCTION
Activation of monocytes and differentiation into macrophages are major steps in vascular proliferative diseases like atherosclerosis and restenosis [1-3]. Monocytes/macrophages produce growth factors that stimulate vascular smooth muscle cell (VSMC) differentiation and growth [4, 5], while contributing to the deposition of extracellular matrix (ECM) to facilitate immune cell infiltration [6, 7] and myofibroblastic migration from the adventitia [8]. It is believed that aged-related changes in monocyte numbers and phenotypes contribute to the development of vascular diseases in the elderly [9-12].
Most recently, we and others demonstrated that aging exacerbates neointimal hyperplasia (NIH) after vascular injury, likely as a consequence of increased infiltration of monocytes [6, 13] and higher sensitivity of VSMC to proliferation stimuli [8]. Interestingly, we also found that monocytes/macrophages in aged rats are more pro-inflammatory and adhesive than in younger animals, which in part explains the increased number of macrophages after vascular injury in aged vasculature [6]. Not surprisingly, pharmacological depletion of monocytes compromises neointima development after arterial injury [6].
This study aims at demonstrating the restenotic properties of aged monocytes in the rat model of vascular injury. We controled for additional age-related interacting factors and demonstrate for the first time that changes in monocytes from aged rats are sufficient to exacerbate NIH after arterial injury. Gene expression profiles of monocytes from young and aged rats revealed significant differences in the expression of adhesion molecules, cytotoxic factors, as well as genes associated with inflammation, cellular differentiation and migration. Altogether, our work identifies potential molecular players that may be involved in the differential contribution of monocytes to post-injury restenosis with aging.
RESULTS
Aged rats have higher counts of blood monocytes than younger animals
In order to investigate possible age-related changes in monocytes that may explain a differential contribution to NIH after vascular injury, monocytic cells from Fischer rats aged 2 and 22 months old (n = 3 per age group) were isolated and purified by negative immunomagnetic separation. Purified live cells were 90% monocytes, of which over 75% stained positive for both CD11b/c and CD18 (Figure 1A). The isolated monocytes were essentially negative for the CD4 and CD8 markers (Figure 1A). The total percentage of monocytes in peripheral blood mononuclear cells (PBMC) was significantly higher in aged rats compared to young animals (14.4 ± 2.0 vs. 6.6 ± 0.2%, p = 0.02; Figure 1B), as previously reported [13], while the opposite was observed for peripheral lymphocytes, although not statistically significant (43.1 ± 12.3 vs. 49.4 ± 21.2%, p = 0.8).
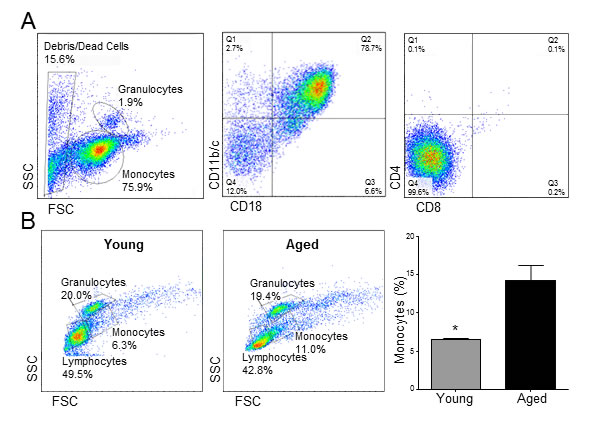
Figure 1: Aging causes monocytosis in rats. A. Representative flow cytometry analysis of purified monocytes. B. Cell counts ± SEM of monocytes, granulocytes and lymphocytes in young and aged rats (n = 3 per group). Numbers are expressed as percentages of peripheral blood mononuclear cells. *p < 0.05.
Monocytes from aged rats exacerbate NIH in injured arteries of young animals
Age-related phenotypic differences in human monocytes have been associated with an increased risk for atherosclerosis and restenosis [12, 14, 15]. We investigated whether, in the absence of confounding age-related factors, monocytes from aged rats were sufficient to exacerbate NIH in injured arteries of young animals. To this end, approximately 2x106 monocytes isolated from young and aged animals were delivered perivascularly around injured arteries of young recipients (n = 6 per age group). A control group received an equivalent volume of vehicle under the same conditions (n = 6).
Arteries that received monocytes from aged animals developed thicker neointimas than those that received cells from young rats (N/M ratio: 0.63 ± 0.09 vs. 0.40 ± 0.05, p = 0.049; Figure 2). In contrast, monocytes from young animals did not modify neointima thickness with respect to rats receiving vehicle alone (N/M ratio: 0.37 ± 0.05, p = 0.7). The number of CD68+ macrophages increased significantly in the adventitia of arteries that received monocytes compared to the vehicle control (24.50 ± 5.07 in control vs. 47.25 ± 6.29 cells per section in aged, p = 0.03; Figure 3A-3B). However, despite the differences observed in neointima thickness measurements between arteries that received cells from young or aged donors, no significant differences in the number of infiltrated macrophages were detected between both experimental conditions (52.00 ± 5.20 vs. 47.25 ± 6.29 cells per section, p = 0.6; Figure 3B).
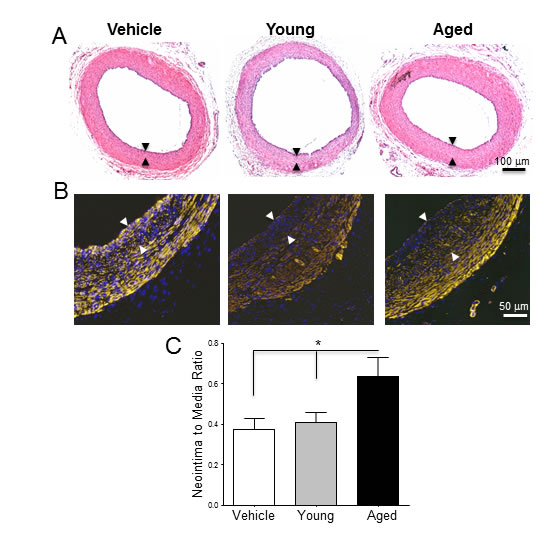
Figure 2: Monocytes from aged rats exacerbate post-injury neointimal hyperplasia in young arteries. A-B. Equal numbers of monocytes from young and aged rats were suspended in Matrigel and exogenously seeded around arteries prior to balloon injury. A. Hematoxylin and Eosin and B. SMA stained cross-sections of injured arteries treated with Matrigel alone, or monocytes from young and aged donors, and harvested 21 days after injury. The neointima layer is delineated by arrows. C. Neointima-to-media ratios of the three experimental groups expressed as mean ratio ± SEM (n = 6 per group). *p < 0.05 .
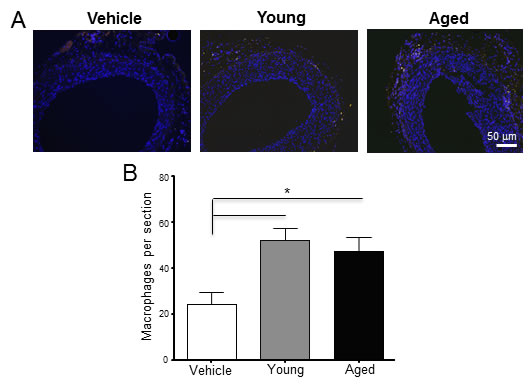
Figure 3: Perivascular delivery of monocytes from young and aged rats increased the number of CD68+ macrophages in the adventitia. A. Immunofluorescent staining of injured arteries 21 days after surgery using an anti-CD68 antibody. CD68+ macrophages are shown in yellow, while nuclei were counter-stained with DAPI (blue). B. Number of macrophages per section in the three experimental groups. Bars represent the mean ± SEM (n = 4 per group). *p < 0.05 .
Adhesion molecules and pro-inflammatory factors are differentially expressed in monocytes from aged and young animals
We then looked for differentially expressed genes that could account for the increased restenotic effect of monocytes from aged animals. Table 1 lists significantly up-regulated genes in monocytes from aged rats compared to those from young donors (n = 3 per group). Interestingly, this list includes a number of transcripts involved in differentiation (Csf1, Egr1, Gfi1, Gprc5a, Jun, Rasl11b), cell survival (Dapk2, Nol3), migration (Lpar3), cell adhesion (Asam, Cd24, Cdh13, Chn2, Cldn23, Esam, Mcam, Mpp7), cytotoxicity (Gzmbl3, Gzmf), ECM remodeling (Cela1), and inflammation (Cxcl1, Dusp2, Ifng, Klrb1, Lyzl4, Nr4a2, Nr4a3, Plk2, Plk3, Rgs16). Numerous genes coding for cytoskeletal proteins (Actn2, Anxa8, Calb2, Dnah8, Gels, Mark1, Myh2, Scin, Tnni1), cell cycle modulators (Btg3, Cdkn2a, Cenpt, Cks1b, Cks2, Gmnn, Kif20b, Plk3, Prc1, Spag5, Spc25), as well as lipid degradation enzymes (Pla2g2f, Pnliprp2) were also up-regulated.
Table 1: Significantly up-regulated genes in monocytes from aged vs. young rats. |
|||||
Gene |
Description |
Fold change |
p |
FDR |
|
Pnliprp2 |
Pancreatic lipase-related protein 2 |
372.49 |
0.00E+00 |
1.00E-05 |
|
Ispd |
Isoprenoid synthase domain-containing protein |
366.83 |
0.00E+00 |
1.13E-03 |
|
Anxa8 |
Annexin A8 |
210.29 |
0.00E+00 |
1.70E-04 |
|
Gzmf |
Granzyme F |
208.57 |
0.00E+00 |
3.24E-03 |
|
Sfrp1 |
Secreted frizzled-related protein 1 |
195.25 |
1.00E-05 |
3.24E-03 |
|
Cdh13 |
Cadherin 13 |
61.88 |
0.00E+00 |
3.10E-04 |
|
Lama3 |
Laminin alpha 3 |
57.29 |
3.00E-05 |
5.99E-03 |
|
Calb2 |
Calbindin 2 |
52.87 |
5.00E-05 |
7.68E-03 |
|
Thumpd1 |
THUMP domain-containing protein 1 |
40.75 |
0.00E+00 |
1.00E-05 |
|
Klrb1 |
Killer cell lectin-like receptor subfamily B member 1 |
40.66 |
3.00E-05 |
6.22E-03 |
|
Ppic |
Peptidyl-prolyl cis-trans isomerase C |
34.04 |
1.80E-04 |
1.73E-02 |
|
Asam |
Adipocyte adhesion molecule |
24.46 |
1.00E-05 |
3.24E-03 |
|
Lyzl4 |
Lysozyme A |
22.60 |
9.00E-05 |
1.10E-02 |
|
Cdkn2a |
Cyclin-dependent kinase inhibitor 2A |
21.33 |
2.00E-05 |
5.55E-03 |
|
Cela1 |
Chymotrypsin-like elastase family member 1 |
15.50 |
6.00E-05 |
8.58E-03 |
|
Lrrc72 |
Leucine-rich repeat-containing protein 72 |
12.78 |
1.00E-05 |
3.24E-03 |
|
Tnni1 |
Troponin I, slow skeletal muscle |
12.60 |
4.80E-04 |
3.50E-02 |
|
Resp18 |
Regulated endocrine-specific protein 18 |
12.48 |
3.00E-04 |
2.63E-02 |
|
Nr4a3 |
Nuclear receptor subfamily 4 group A member 3 |
12.13 |
1.00E-05 |
4.56E-03 |
|
Dnah8 |
Dynein, axonemal heavy chain 8 |
11.74 |
1.30E-04 |
1.45E-02 |
|
Ephb3 |
Ephrin type-B receptor 3 |
11.41 |
2.00E-05 |
5.55E-03 |
|
Atp1a2 |
Sodium/potassium-transporting ATPase subunit alpha-2 |
10.16 |
2.00E-05 |
5.55E-03 |
|
Mcam |
Cell surface glycoprotein MUC18 |
9.85 |
2.00E-05 |
5.35E-03 |
|
Mpp7 |
MAGUK p55 subfamily member 7 |
8.67 |
3.00E-05 |
6.33E-03 |
|
Actn2 |
Actinin alpha 2 |
8.53 |
7.80E-04 |
4.89E-02 |
|
Slc4a4 |
Electrogenic sodium bicarbonate cotransporter 1 |
8.42 |
1.00E-05 |
3.24E-03 |
|
Dusp2 |
Dual specificity protein phosphatase 2 |
8.19 |
3.00E-05 |
6.33E-03 |
|
Ifng |
Interferon gamma |
6.45 |
2.00E-05 |
5.43E-03 |
|
Spag5 |
Sperm associated antigen 5 |
5.99 |
7.00E-04 |
4.52E-02 |
|
Pask |
PAS domain-containing serine/threonine protein kinase |
5.93 |
7.00E-04 |
4.87E-02 |
|
Cldn23 |
Claudin 23 |
5.86 |
7.00E-05 |
9.12E-03 |
|
Gstt3 |
Glutathione S-transferase theta 3 |
5.75 |
5.40E-04 |
3.79E-02 |
|
Gfi1 |
Zinc finger protein Gfi-1 |
5.73 |
4.00E-05 |
6.77E-03 |
|
Lpar3 |
Lysophosphatidic acid receptor 3 |
5.65 |
1.00E-04 |
1.18E-02 |
|
Nol3 |
Nucleolar protein 3 |
5.62 |
1.30E-04 |
1.43E-02 |
|
Gzmbl3 |
Granzyme B-like 3 |
5.56 |
0.00E+00 |
2.46E-03 |
|
Copz2 |
Coatomer protein complex, subunit zeta 2 |
5.14 |
3.20E-04 |
2.68E-02 |
|
Kif20b |
M-phase phosphoprotein 1 |
5.04 |
6.20E-04 |
4.15E-02 |
|
Nr4a2 |
Nuclear receptor subfamily 4 group A member 2 |
5.00 |
5.00E-05 |
7.84E-03 |
|
Myh2 |
Myosin, heavy chain 2 |
4.95 |
4.50E-04 |
3.35E-02 |
|
Cks1b |
Cyclin-dependent kinases regulatory subunit 1 |
4.83 |
4.10E-04 |
3.16E-02 |
|
Spc25 |
Kinetochore protein Spc25 |
4.72 |
6.30E-04 |
4.22E-02 |
|
Arhgef39 |
Rho guanine nucleotide exchange factor 39 |
4.51 |
4.00E-04 |
3.13E-02 |
|
Gfra2 |
GDNF family receptor alpha-2 |
4.47 |
5.20E-04 |
3.71E-02 |
|
Plk3 |
Serine/threonine-protein kinase PLK3 |
4.47 |
2.00E-05 |
5.36E-03 |
|
Plk2 |
Serine/threonine-protein kinase PLK2 |
4.43 |
2.00E-05 |
5.35E-03 |
|
Dapk2 |
Death-associated kinase 2 |
4.35 |
3.40E-04 |
2.78E-02 |
|
Agpat3 |
1-acyl-sn-glycerol-3-phosphate acyltransferase gamma |
4.34 |
2.20E-04 |
2.00E-02 |
|
Tmem246 |
Transmembrane protein 246 |
4.33 |
1.00E-05 |
4.56E-03 |
|
Scin |
Adseverin |
4.32 |
1.70E-04 |
1.69E-02 |
|
Esam |
Endothelial cell-selective adhesion molecule |
4.21 |
3.60E-04 |
2.93E-02 |
|
Cks2 |
Cyclin-dependent kinases regulatory subunit 2 |
4.18 |
8.00E-05 |
1.07E-02 |
|
Sgca |
Alpha sarcoglycan |
4.18 |
3.10E-04 |
2.66E-02 |
|
Rasl11b |
Ras-like protein family member 11B |
4.15 |
7.00E-05 |
9.12E-03 |
|
Csf1 |
Macrophage colony-stimulating factor 1 |
4.11 |
4.50E-04 |
3.35E-02 |
|
Fv1 |
Friend virus susceptibility protein 1 |
4.07 |
4.70E-04 |
3.46E-02 |
|
Chn2 |
Beta-chimaerin |
4.01 |
4.40E-04 |
3.34E-02 |
|
Ube2t |
Ubiquitin-conjugating enzyme E2T |
3.95 |
5.10E-04 |
3.76E-02 |
|
Xxylt1 |
Xyloside xylosyltransferase 1 |
3.95 |
6.00E-05 |
8.91E-03 |
|
Srpk3 |
Serine/arginine-rich splicing factor protein kinase 3 |
3.90 |
4.00E-05 |
6.56E-03 |
|
Prc1 |
Protein regulator of cytokinesis 1 |
3.82 |
7.40E-04 |
4.72E-02 |
|
Cxcl1 |
Chemokine (C-X-C motif) ligand 1 |
3.76 |
7.60E-04 |
4.81E-02 |
|
Mfsd4 |
Major facilitator superfamily domain-containing protein 4 |
3.74 |
7.00E-05 |
9.23E-03 |
|
Cd24 |
Signal transducer CD24 |
3.68 |
5.80E-04 |
3.98E-02 |
|
Gprc5a |
Retinoic acid-induced protein 3 |
3.63 |
3.40E-04 |
2.80E-02 |
|
Mark1 |
Serine/threonine-protein kinase MARK1 |
3.61 |
1.00E-04 |
1.22E-02 |
|
Rgs16 |
Regulator of G-protein signaling 16 |
3.61 |
1.00E-04 |
1.18E-02 |
|
Gmnn |
Geminin |
3.54 |
2.00E-04 |
1.90E-02 |
|
Chrne |
Acetylcholine receptor subunit epsilon |
3.45 |
1.90E-04 |
1.80E-02 |
|
Calcr |
Calcitonin receptor |
3.41 |
7.30E-04 |
4.66E-02 |
|
Sccpdh |
Saccharopine dehydrogenase-like oxidoreductase |
3.37 |
4.00E-04 |
3.14E-02 |
|
Pik3r1 |
Phosphoinositide-3-kinase, regulatory subunit 1 alpha |
3.36 |
5.30E-04 |
3.77E-02 |
|
Ly49i4 |
Ly49 inhibitory receptor 4 |
3.35 |
4.50E-04 |
3.35E-02 |
|
Slco4a1 |
Solute carrier organic anion transporter family member 4A1 |
3.23 |
1.30E-04 |
1.43E-02 |
|
Ppp1r36 |
Protein phosphatase 1, regulatory subunit 36 |
3.04 |
7.00E-04 |
4.52E-02 |
|
Pla2g2f |
Group IIF secretory phospholipase A2 |
3.03 |
3.20E-04 |
2.70E-02 |
|
Syne1 |
Spectrin repeat-containing, nuclear envelope protein 1 |
2.97 |
6.60E-04 |
4.34E-02 |
|
Egr1 |
Early growth response protein 1 |
2.95 |
7.00E-04 |
4.52E-02 |
|
Gels |
Gelsolin |
2.92 |
1.30E-04 |
1.43E-02 |
|
Wdr25 |
WD repeat-containing protein 25 |
2.90 |
1.80E-04 |
1.73E-02 |
|
Prdm1 |
PR domain zinc finger protein 1 |
2.82 |
4.80E-04 |
3.50E-02 |
|
Jun |
Transcription factor AP-1 |
2.73 |
1.40E-04 |
1.51E-02 |
|
Grk4 |
G protein-coupled receptor kinase 4 |
2.42 |
6.20E-04 |
4.15E-02 |
|
Lmnb1 |
Lamin B1 |
2.39 |
6.20E-04 |
4.15E-02 |
|
Btg3 |
Protein BTG3 |
2.19 |
7.30E-04 |
4.66E-02 |
|
On the other hand, the group of down-regulated genes (or up-regulated in monocytes from young rats compared to aged animals) included transcripts involved in immune regulation (Tnfrsf17), chemotaxis (CCr10, S1pr1), TLR signaling (Ticam1), and antigen presentation (RT1; Table 2). Numerous B cell and red blood cell (RBC) related transcripts are also up-regulated in monocytes from young rats. While the latter may be suggestive of contamination with other cell types, other studies have reported similar findings in the past, including the expression of IgM proteins [16].
The mRNA and protein expressions of two of the most up-regulated transcripts (Cdh13 and Sfrp1) were confirmed by qRT-PCR and immunofluorescence (Figure 4). Both analyses demonstrate higher expressions of both genes in monocytes from aged rats compared to young animals. Nevertheless, the immunofluorescence staining clearly shows that monocytes isolated from animals of both age groups are heterogeneous in nature, since not all cells stained uniformly for SFRP1 and different levels of fluorescence are evident for CADH13 (Figure 4B).
Table 2: Significantly down-regulated genes in monocytes from aged vs. young rats.
Gene |
Description |
Fold change |
p |
FDR |
S1pr1 |
Sphingosine 1-phosphate receptor 1 |
0.03 |
3.20E-04 |
2.68E-02 |
Hbe1 |
Hemoglobin, epsilon 1 |
0.04 |
7.40E-04 |
4.72E-02 |
Slc4a1 |
Solute carrier organic anion transporter family member 4A1 |
0.05 |
1.70E-04 |
1.69E-02 |
Eraf |
Alpha hemoglobin stabilizing protein |
0.08 |
5.50E-04 |
3.83E-02 |
IGKV |
Immunoglobulin kappa light chain V region |
0.08 |
4.00E-05 |
6.79E-03 |
IGG2A |
Immunoglobulin gamma-2A chain C region |
0.10 |
1.20E-04 |
1.38E-02 |
Ccr10 |
C-C chemokine receptor type 10 |
0.11 |
3.00E-05 |
6.05E-03 |
Zcchc14 |
Zinc finger, CCHC domain-containing protein 14 |
0.12 |
4.00E-05 |
6.53E-03 |
Mzb1 |
Marginal zone B and B1 cell-specific protein |
0.14 |
2.00E-05 |
5.35E-03 |
Tnfrsf17 |
Tumor necrosis factor receptor superfamily member 17 |
0.14 |
1.00E-05 |
3.24E-03 |
IGLC2 |
Immunoglobulin lambda-2 chain C region |
0.16 |
3.00E-05 |
6.19E-03 |
Cd79b |
B-cell antigen receptor complex-associated protein, beta chain |
0.17 |
1.00E-05 |
4.64E-03 |
Pxdc1 |
PX domain-containing protein 1 |
0.17 |
5.00E-05 |
7.84E-03 |
Vpreb3 |
Pre-B lymphocyte protein 3 |
0.17 |
3.00E-05 |
5.99E-03 |
Olr686 |
Olfactory receptor Olr686 |
0.19 |
1.30E-04 |
1.43E-02 |
Itih4 |
Inter-alpha-trypsin inhibitor heavy chain family, member 4 |
0.20 |
8.00E-05 |
1.01E-02 |
Slc22a23 |
Solute carrier family 22 member 23 |
0.20 |
2.00E-05 |
5.55E-03 |
Derl3 |
Derlin-3 |
0.23 |
1.00E-05 |
4.56E-03 |
Rhbdl1 |
Rhomboid, veinlet-like protein 1 |
0.23 |
3.00E-05 |
6.33E-03 |
Ms4a1 |
Membrane-spanning 4-domains, subfamily A, member 1 |
0.23 |
1.40E-04 |
1.46E-02 |
Cd79a |
B-cell antigen receptor complex-associated protein, alpha chain |
0.23 |
1.00E-04 |
1.20E-02 |
Eaf2 |
ELL-associated factor 2 |
0.24 |
1.80E-04 |
1.75E-02 |
Sspn |
Sarcospan |
0.24 |
4.90E-04 |
3.55E-02 |
Pde6h |
Cone-like cGMP-phosphodiesterase 6 gamma subunit |
0.24 |
2.90E-04 |
2.58E-02 |
Try10 |
Trypsin 10 precursor |
0.25 |
2.10E-04 |
1.92E-02 |
Fcer2 |
IgE Fc receptor, low affinity II, alpha polypeptide isoform b |
0.26 |
1.70E-04 |
1.72E-02 |
RT1-CE10 |
RT1 class I, locus CE10 |
0.28 |
8.00E-05 |
1.07E-02 |
Myf6 |
Myogenic factor 6 |
0.30 |
1.70E-04 |
1.72E-02 |
Gas6 |
Growth arrest-specific protein 6 |
0.30 |
1.40E-04 |
1.47E-02 |
Jakmip1 |
Janus kinase and microtubule-interacting protein 1 |
0.31 |
9.00E-05 |
1.13E-02 |
Pon1 |
Serum paraoxonase/arylesterase 1 |
0.32 |
6.90E-04 |
4.50E-02 |
Dgkg |
Diacylglycerol kinase, theta |
0.32 |
4.50E-04 |
3.35E-02 |
Txndc5 |
Thioredoxin domain-containing protein 5, endoplasmic reticulum |
0.33 |
4.00E-05 |
6.49E-03 |
RT1-CE3 |
RT1 class I, locus CE3 |
0.33 |
1.80E-04 |
1.75E-02 |
RT1-A2 |
RT1 class I, locus A2 |
0.33 |
3.90E-04 |
3.09E-02 |
Idh1 |
Isocitrate dehydrogenase [NADP], cytoplasmic |
0.35 |
5.80E-04 |
3.98E-02 |
Ticam1 |
Toll-like receptor adaptor molecule 1 |
0.36 |
3.90E-04 |
3.09E-02 |
Hvcn1 |
Hydrogen voltage-gated channel 1 |
0.37 |
3.60E-04 |
2.95E-02 |
Sec11c |
Signal peptidase complex catalytic subunit SEC11C |
0.37 |
6.00E-05 |
8.26E-03 |
Musk |
Muscle, skeletal, receptor tyrosine protein kinase |
0.38 |
6.40E-04 |
4.26E-02 |
Dgka |
Diacylglycerol kinase, alpha |
0.39 |
5.90E-04 |
4.02E-02 |
Nol4l |
Nucleolar protein 4-like |
0.42 |
6.80E-04 |
4.45E-02 |
Mkrn1 |
Makorin ring finger protein 1 |
0.43 |
4.10E-04 |
3.20E-02 |
Pdia4 |
Protein disulfide isomerase-associated 4 |
0.43 |
4.30E-04 |
3.31E-02 |
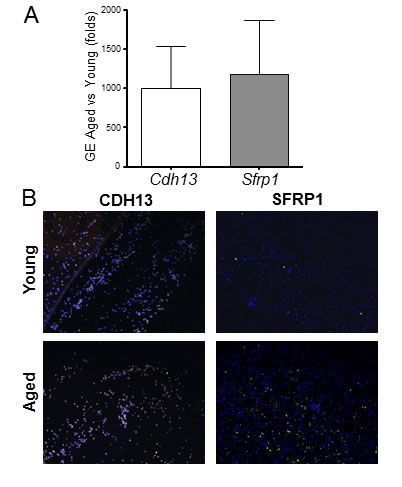
Figure 4: Aging induces significant gene expression differences in rat monocytes. A. Confirmatory qRT-PCR for cadherin 13 (Cdh13) and secreted frizzled-related protein 1 (Sfrp1) in monocytes. Gene expression (GE) is expressed in fold change of aged vs. young as calculated using the ddCT method. B. Immunofluorescent staining of young and aged monocytes using antibodies against CDH13 and SFRP1.
DISCUSSION
Numerous studies indicate that aging favors pro-inflammatory phenotypes on monocytes, which predispose for cardiovascular disease and chronic subclinical inflammation [9-12]. In this work we demonstrated that 1) aging causes monocytosis in rats, 2) monocytes from aged animals exacerbate NIH, and 3) aging induces profound differences in monocyte gene expression.
Our results underscore the pro-inflammatory potential of monocytes from aged animals in the vascular response to balloon injury. Perivascular delivery of equal numbers of monocytes from either young or aged donors to young injured arteries showed that cells from aged rats have restenotic properties, in contrast to those from young animals. In addition, the different effects of monocytes from both age groups on neointima development occurred in the absence of other age-related aggravating factors, including monocytosis. In agreement with previous reports [13], we confirmed that a higher percentage of monocytes in PBMC is associated with aging in rats. But more importantly, we showed that, although higher cell counts may contribute to NIH, it is the phenotypic changes in monocytes from aged rats that are truly responsible for inducing constrictive vascular remodeling. The number of infiltrated macrophages in injured arteries was the same at 21 days with monocytes from both groups. This suggests that the different effects on neointima formation were mostly caused by secretable factors and/or that detectable differences in macrophage infiltration occurred at an earlier time point after the procedure. It is also likely that other age-related factors in the vascular wall further stimulate infiltration, differentiation and macrophage survival in injured arteries, as previously demonstrated by our group [6].
The gene expression profile of monocytes from aged rats reveals possible mediators of their restenotic and pro-inflammatory properties. In addition to the numerous up-regulated adhesion molecules (Table 1 and Results), monocytes from aged animals have higher expression of Csf1, Cxcl1, and Ifng. This combination of cytokines is highly synergistic. As the primary growth factor for the differentiation of monocytes into macrophages, CSF1 is more potent in combination with IFNγ [17]. The latter is a major inflammatory cytokine and contributes to the development of NIH after balloon angioplasty by inducing VSMC proliferation [5]. CXCL1, in turn, amplifies the inflammatory response and stimulates the recruitment of additional monocytes [7]. As an antagonist of the Wnt/β-catenin pathway, SFRP1 is expected to reduce chemotaxis and decrease VSMC proliferation [18]. However, overexpression of this protein is also known to improve cell-to-cell contacts with endothelial cells (EC) and VSMC [19], and it may influence ECM remodeling by up-regulating metalloproteinases [20]. Other up-regulated genes such as Dusp2, Egr1, Lpar3, Plk3, and Rasl11b are also stimulators of monocyte differentiation and inflammatory responses [21-25].
Monocytes from aged rats also demonstrate up-regulated gene expression of the secretable cytotoxic factors GZMBL3, GZMF, and LYZL4. Granzymes are normally not regarded as monocyte-derived. However, several groups have shown that granzyme B is also produced by monocytes [26, 27]. Vascular research has been limited to this enzyme, which was found in the intima and media of advanced atherosclerotic lesions [28]. In addition, a recent work demonstrated that granzyme B increased vascular permeability by releasing vascular endothelial growth factor (VEFG) from the ECM [29]. In the setting of restenosis, it is possible that granzymes contribute to excessive VSMC cytotoxicity, which compromises the healing response of the wall and exacerbates inflammation [30]. Similarly, lysozyme M-positive monocytes are known to amplify oxidative stress in the vascular wall [31], suggesting that other lysozymes may also increase inflammation.
In contrast to the high number of adhesion molecules, chemotactic factors and inflammatory mediators overexpressed in monocytes from aged rats, up-regulated genes in cells isolated from young rats do not suggest a pro-inflammatory profile (Table 2 and Results). Instead, up-regulation of MHC class I receptor genes (RT1) and S1p1r indicate increased involvement in antigen presentation processes and trafficking to and from lymphoid organs [32]. Likewise, monocytes from young rats demonstrate up-regulated expression of the TLR3 and TLR4 intracellular adaptor TICAM-1, which likely relates to the increased response to bacterial LPS that is normally observed in young individuals [11].
Of note, a number of similarities in gene expression can be observed between monocytes from aged rats and pro-inflammatory CD16+ subpopulations in humans. These include up-regulation of granzymes, Dusp2, Egr1, Gfra2, and Klrb1 [26, 33, 34]. Interestingly, higher numbers of these subpopulations are found in the elderly and in individuals with chronic inflammatory conditions [9, 12], where they have been associated with atherosclerosis and restenosis [12, 14, 15]. In addition, multiple pro-atherogenic factors are overexpressed in monocytes from aged animals, including Cxcl1, Lpar3, and the secretable lipases Pla2g2f and Pnliprp2. Macrophage-derived lipases are known to induce foam cell formation [35]. CXCL1 is also involved in atherosclerosis [7], and LPAR3 is required for monocyte recruitment to atherosclerotic lesions by oxidized LDL [36].
The overexpression of inflammatory mediators, cytotoxic factors, and adhesion molecules in monocytes from aged rats fits two different but complementary theories of aging: the hyperfunction theory [37] and inflammaging [38]. In fact, the former theory explains the latter and postulates that increased functions in both immune and non-immune cells lead to a loss of homeostasis, chronic inflammation and age-related diseases.
The limitations of this study include the possibilities that cellular contaminants were introduced as part of our monocyte isolation procedures, and that the identified mRNA expression profiles do not correspond to protein levels as a result of post-translational regulation. In addition, it is conceivable that perivascular delivery of high numbers of monocytes using a Matrigel system might over-promote their migration into the wall, compared to physiological conditions. Despite these limitations, our study demonstrates that monocytes from aged rats, but not young animals, induce restenosis after arterial injury. Furthermore, we show that their gene expression profile not only explains this observed inflammatory and restenotic character, but also predicts a highly pro-atherogenic potential.
MATERIALS AND METHODS
Isolation and purification of monocytes
Aged Fischer (>22-month-old, F344) rats were purchased from the National Institute of Aging (Bethesda, MD), while young (2-month-old) rats were obtained from Harlan Laboratories (Indianapolis, IN). Blood monocytes were isolated using density gradient followed by negative immunomagnetic separation [39]. Briefly, non-monocytic cells from Histopaque-1087 gradients were labeled with specific monoclonal antibodies (20 µg of Ox-8, Ox-19, Ox-33, Ox-52, anti-CD4, and anti-granulocytes from AbD Serotec) and discarded using anti-mouse magnetic beads. Monocytes purity was determined by FACS analysis using fluorescently labeled anti-CD4, -CD8, -CD11b, and -CD18 antibodies (AbD Serotec).
Balloon injury and perivascular cell delivery
The Institutional Committee for Use and Care of Laboratory Animals at the University of Miami previously approved all animal procedures. Balloon injury in the right iliac artery was inflicted with a 2F Fogarty catheter (Baxter Corp., Irvine, CA, USA) adapted to a custom angiographic kit (Boston Scientific, Scimed) [40]. Approximately 2x106 monocytes from either aged or young rats were re-suspended in 0.2 ml of BD Matrigel™ Matrix (BD Biosciences, San Jose, CA) and seeded around iliac arteries prior to balloon injury. Control animals received a similar volume of Matrigel alone. Arterial specimens were collected 21 days after injury and fixed in 4% formalin-PBS (Sigma-Aldrich). The areas of the neointima and media layers were measured in H&E stained slides using the ImagePro software (Media Cybernetics, Bethesda, MD).
Immunohistochemistry
Paraffin sections were rehydrated and treated with 3% hydrogen peroxide to block endogenous peroxidase activity. Sections were boiled in citrate buffer to allow epitope retrieval. Non-specific binding sites were blocked with 0.5% blocking solution (DAKO, Carpinteria, CA) before adding primary antibodies against rat CD68 (AbD Serotec) and smooth muscle cell actin (DAKO) for 1 h at room temperature. Biotinylated secondary antibodies (DAKO Universal Link) were applied for 30 min, followed by a 15-min incubation with horseradish peroxidase-streptavidin solution (DAKO) at room temperature. Color was developed with a DAB chromogenic solution (DAKO). Nuclei were counterstained with Meyer’s hematoxylin and mounted as described above.
Fresh isolated monocytes were cytospun onto microscope slides and fixed with cold methanol for 10 min at -20°C. Non-specific binding sites were blocked with 10% goat serum (Chemicon International, Temecula, CA) in PBS for 1 h at room temperature. Primary rabbit polyclonal antibodies against cadherin 13 and secreted frizzled-related protein 1 (Santa Cruz Biotechnology) were diluted in blocking solution and applied onto sections overnight at 4°C. After washing twice with PBS for 3 min, tissue sections were incubated with Alexa Fluor 488 (or 546) goat anti-rabbit (Invitrogen, Carlsbad, CA) for 90 min at room temperature. Sections were mounted in Vectashield w/DAPI (Vector Laboratories, Burlingame, CA) and examined with an Olympus BX 40 microscope (Olympus America Inc., Center Valley, PA).
RNA microarray analysis
Total RNA from fresh isolated monocytes was extracted using TRI Reagent (Molecular Research Center, Cincinnati, OH), followed by an extra-cleaning step with the RNeasy kit (Qiagene, Valencia, CA). Gene expression profiling and data analyses were performed at Ocean Ridge Biosciences (Palm Beach Gardens, FL) using two-color Agilent Whole Rat Genome Arrays (Agilent Technologies). Differentially expressed genes were those with p-values and false discovery rates (FDR) under 0.05. Microarray results for selected genes were confirmed using TaqMan Gene Expression Assays (Applied Biosystems, Foster City, CA). Real-time PCR was performed on an ABI Prism 7500 Fast Real-Time PCR System and relative gene expression was determined using the ddCT method [41].
Statistical analyses
Results were expressed as means ± SE. Two-group comparisons were conducted using two-tailed t-tests for independent samples with unequal variances. Statistics were calculated with Prism 5 (GraphPad Software, La Jolla, CA).
FUNDING
This study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases [R01-DK-098511] and the National Heart, Lung and Blood Institute at the National Institutes of Health [R01-HL109582] to R.I.V.-P.
CONFLICTs OF INTERESTS
The authors indicate no potential conflicts of interests.
REFERENCES
1. Hokimoto S, Oike Y, Saito T, Kitaoka M, Oshima S, Noda K, Moriyama Y, Ishibashi F, and Ogawa H. Increased expression of monocyte chemoattractant protein-1 in atherectomy specimens from patients with restenosis after percutaneous transluminal coronary angioplasty. Circ J. 2002; 66:114-6.
2. Pietersma A, Kofflard M, de Wit LE, Stijnen T, Koster JF, Serruys PW, and Sluiter W. Late lumen loss after coronary angioplasty is associated with the activation status of circulating phagocytes before treatment. Circulation. 1995; 91:1320-5.
3. Fukuda D, Shimada K, Tanaka A, Kawarabayashi T, Yoshiyama M, and Yoshikawa J. Circulating monocytes and in-stent neointima after coronary stent implantation. J Am Coll Cardiol. 2004; 43:18-23.
4. Maddaluno M, Di Lauro M, Di Pascale A, Santamaria R, Guglielmotti A, Grassia G, and Ialenti A. Monocyte chemotactic protein-3 induces human coronary smooth muscle cell proliferation. Atherosclerosis. 2011; 217:113-9.
5. Kusaba K, Kai H, Koga M, Takayama N, Ikeda A, Yasukawa H, Seki Y, Egashira K, and Imaizumi T. Inhibition of intrinsic interferon-gamma function prevents neointima formation after balloon injury. Hypertension. 2007; 49:909-15.
6. Rodriguez-Menocal L, Faridi MH, Martinez L, Shehadeh LA, Duque JC, Wei Y, Mesa A, Pena A, Gupta V, Pham SM, and Vazquez-Padron RI. Macrophage-derived IL-18 and increased fibrinogen deposition are age-related inflammatory signatures of vascular remodeling. Am J Physiol Heart Circ Physiol. 2014; 306:H641-53.
7. Moore KJ, Sheedy FJ, and Fisher EA. Macrophages in atherosclerosis: a dynamic balance. Nat Rev Immunol. 2013; 13:709-21.
8. Si Y, Ren J, Wang P, Rateri DL, Daugherty A, Shi XD, Kent KC, and Liu B. Protein kinase C-delta mediates adventitial cell migration through regulation of monocyte chemoattractant protein-1 expression in a rat angioplasty model. Arterioscler Thromb Vasc Biol. 2012; 32:943-54.
9. Seidler S, Zimmermann HW, Bartneck M, Trautwein C, and Tacke F. Age-dependent alterations of monocyte subsets and monocyte-related chemokine pathways in healthy adults. BMC Immunol. 2010; 11:30.
10. Hearps AC, Martin GE, Angelovich TA, Cheng WJ, Maisa A, Landay AL, Jaworowski A, and Crowe SM. Aging is associated with chronic innate immune activation and dysregulation of monocyte phenotype and function. Aging Cell. 2012; 11:867-75.
11. Suchy D, Labuzek K, Buldak L, Szkudlapski D, and Okopien B. Comparison of chosen activation markers of human monocytes/macrophages isolated from the peripheral blood of young and elderly volunteers. Pharmacol Rep. 2014; 66:759-65.
12. Merino A, Buendia P, Martin-Malo A, Aljama P, Ramirez R, and Carracedo J. Senescent CD14+CD16+ monocytes exhibit proinflammatory and proatherosclerotic activity. J Immunol. 2011; 186:1809-15.
13. Eghbalieh SD, Chowdhary P, Muto A, Ziegler KR, Kudo FA, Pimiento JM, Mirmehdi I, Model LS, Kondo Y, Nishibe T, and Dardik A. Age-related neointimal hyperplasia is associated with monocyte infiltration after balloon angioplasty. J Gerontol A Biol Sci Med Sci. 2012; 67:109-17.
14. Liu Y, Imanishi T, Ikejima H, Tsujioka H, Ozaki Y, Kuroi A, Okochi K, Ishibashi K, Tanimoto T, Ino Y, Kitabata H, and Akasaka T. Association between circulating monocyte subsets and in-stent restenosis after coronary stent implantation in patients with ST-elevation myocardial infarction. Circ J. 2010; 74:2585-91.
15. Rogacev KS, Ulrich C, Blomer L, Hornof F, Oster K, Ziegelin M, Cremers B, Grenner Y, Geisel J, Schlitt A, Kohler H, Fliser D, Girndt M, et al. Monocyte heterogeneity in obesity and subclinical atherosclerosis. Eur Heart J. 2010; 31:369-76.
16. Huang J, Sun X, Gong X, He Z, Chen L, Qiu X, and Yin CC. Rearrangement and expression of the immunoglobulin mu-chain gene in human myeloid cells. Cell Mol Immunol. 2014; 11:94-104.
17. Hume DA and MacDonald KP. Therapeutic applications of macrophage colony-stimulating factor-1 (CSF-1) and antagonists of CSF-1 receptor (CSF-1R) signaling. Blood. 2012; 119:1810-20.
18. Mill C and George SJ. Wnt signalling in smooth muscle cells and its role in cardiovascular disorders. Cardiovasc Res. 2012; 95:233-40.
19. Dufourcq P, Descamps B, Tojais NF, Leroux L, Oses P, Daret D, Moreau C, Lamaziere JM, Couffinhal T, and Duplaa C. Secreted frizzled-related protein-1 enhances mesenchymal stem cell function in angiogenesis and contributes to neovessel maturation. Stem Cells. 2008; 26:2991-3001.
20. Foronjy R, Imai K, Shiomi T, Mercer B, Sklepkiewicz P, Thankachen J, Bodine P, and D’Armiento J. The divergent roles of secreted frizzled related protein-1 (SFRP1) in lung morphogenesis and emphysema. Am J Pathol. 2010; 177:598-607.
21. Friedman AD. Transcriptional control of granulocyte and monocyte development. Oncogene. 2007; 26:6816-28.
22. Stolle K, Schnoor M, Fuellen G, Spitzer M, Cullen P, and Lorkowski S. Cloning, genomic organization, and tissue-specific expression of the RASL11B gene. Biochim Biophys Acta. 2007; 1769:514-24.
23. Hu J, Wang G, Liu X, Zhou L, Jiang M, and Yang L. Polo-like kinase 1 (PLK1) is involved in toll-like receptor (TLR)-mediated TNF-alpha production in monocytic THP-1 cells. PLoS One. 2013; 8:e78832.
24. Lang R, Hammer M, and Mages J. DUSP meet immunology: dual specificity MAPK phosphatases in control of the inflammatory response. J Immunol. 2006; 177:7497-504.
25. Fueller M, Wang DA, Tigyi G, and Siess W. Activation of human monocytic cells by lysophosphatidic acid and sphingosine-1-phosphate. Cell Signal. 2003; 15:367-75.
26. Frankenberger M, Hofer TP, Marei A, Dayyani F, Schewe S, Strasser C, Aldraihim A, Stanzel F, Lang R, Hoffmann R, Prazeres da Costa O, Buch T, and Ziegler-Heitbrock L. Transcript profiling of CD16-positive monocytes reveals a unique molecular fingerprint. Eur J Immunol. 2012; 42:957-74.
27. Kim WD, Chi HS, Choe KH, Oh YM, Lee SD, Kim KR, Yoo KH, Ngan DA, Elliott WM, Granville DJ, Sin DD, and Hogg JC. A possible role for CD8+ and non-CD8+ cell granzyme B in early small airway wall remodelling in centrilobular emphysema. Respirology. 2013; 18:688-96.
28. Choy JC, McDonald PC, Suarez AC, Hung VH, Wilson JE, McManus BM, and Granville DJ. Granzyme B in atherosclerosis and transplant vascular disease: association with cell death and atherosclerotic disease severity. Mod Pathol. 2003; 16:460-70.
29. Hendel A, Hsu I, and Granville DJ. Granzyme B releases vascular endothelial growth factor from extracellular matrix and induces vascular permeability. Lab Invest. 2014; 94:716-25.
30. Bennett M, Yu H, and Clarke M. Signalling from dead cells drives inflammation and vessel remodelling. Vascul Pharmacol. 2012; 56:187-92.
31. Kossmann S, Hu H, Steven S, Schonfelder T, Fraccarollo D, Mikhed Y, Brahler M, Knorr M, Brandt M, Karbach SH, Becker C, Oelze M, Bauersachs J, et al. Inflammatory monocytes determine endothelial nitric-oxide synthase uncoupling and nitro-oxidative stress induced by angiotensin II. J Biol Chem. 2014; 289:27540-50.
32. Matloubian M, Lo CG, Cinamon G, Lesneski MJ, Xu Y, Brinkmann V, Allende ML, Proia RL, and Cyster JG. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature. 2004; 427:355-60.
33. Wong KL, Tai JJ, Wong WC, Han H, Sem X, Yeap WH, Kourilsky P, and Wong SC. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood. 2011; 118:e16-31.
34. Schmidl C, Renner K, Peter K, Eder R, Lassmann T, Balwierz PJ, Itoh M, Nagao-Sato S, Kawaji H, Carninci P, Suzuki H, Hayashizaki Y, Andreesen R, et al. Transcription and enhancer profiling in human monocyte subsets. Blood. 2014; 123:e90-9.
35. Pentikainen MO, Oksjoki R, Oorni K, and Kovanen PT. Lipoprotein lipase in the arterial wall: linking LDL to the arterial extracellular matrix and much more. Arterioscler Thromb Vasc Biol. 2002; 22:211-7.
36. Zhou Z, Subramanian P, Sevilmis G, Globke B, Soehnlein O, Karshovska E, Megens R, Heyll K, Chun J, Saulnier-Blache JS, Reinholz M, van Zandvoort M, Weber C, et al. Lipoprotein-derived lysophosphatidic acid promotes atherosclerosis by releasing CXCL1 from the endothelium. Cell Metab. 2011; 13:592-600.
37. Blagosklonny MV. Answering the ultimate question “what is the proximal cause of aging?”. Aging (Albany NY). 2012; 4:861-77.
38. Franceschi C, Bonafe M, Valensin S, Olivieri F, De Luca M, Ottaviani E, and De Benedictis G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci. 2000; 908:244-54.
39. Scriba A, Luciano L, and Steiniger B. High-yield purification of rat monocytes by combined density gradient and immunomagnetic separation. J Immunol Methods. 1996; 189:203-16.
40. Gabeler EE, van Hillegersberg R, Statius van Eps RG, Sluiter W, Gussenhoven EJ, Mulder P, and van Urk H. A comparison of balloon injury models of endovascular lesions in rat arteries. BMC Cardiovasc Disord. 2002; 2:16.
41. Livak KJ and Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25:402-8.

