INTRODUCTION
It has long been appreciated that cancer is a disease of the whole organism, requiring not only the proliferation of malignant cells but also the participation of host cells and signaling factors [1, 2]. Cancer results in inappropriate host cell recruitment and activity, deregulated cell-signaling and regulatory mechanisms, and ultimately failure of the organism as a whole. This has striking parallels to the frameworks of ecological biology: multiple interacting ecosystems (organ systems) within a larger biosphere (body), alterations in species stoichiometry (host cell types), resource cycling (cellular metabolism and cell-cell signaling), and ecosystem collapse (organ failure and death) (Table 1).
In their landmark 2000 and 2011 articles, Hanahan and Weinburg proposed a theoretical framework of essential “hallmarks” or capabilities required by a tumor to become malignant [3, 4]. While the Hallmarks model provides a good framework and common nomenclature for the cancer research field, it does not describe the necessary external processes of selection that lead to the emergence of these traits. It is now well recognized that as cancer evolves and progresses, so too does the tumor microenvironment, thereby (as noted by Hanahan and Weinberg) “enabling primary, invasive, and then metastatic growth” [4–9]. Ecology principles describe the enabling characteristics of the tumor microenvironment, including the factors that promote selection of aggressive traits in malignant disease as well as the local and systemic impact of cancer cells on the native host.
Critical to the survival of an individual organism and the success of the species as a whole are its interactions with other species and with its habitat. The interactions among all the living organisms of a community and the associated abiotic environment form an ecosystem, a self-regulating unit that cooperates as a whole to maintain overall homeostasis [10, 11]. The structure of an ecosystem is governed by species stoichiometry, niche differentiation, environmental disturbances, and resource availability.
In humans, dozens of ecosystems cooperate to perpetuate the required functions necessary to support life. Each organ system requires a specific stoichiometry of cell types (“species”) that interact with each other and with the extracellular matrix (“abiotic factors”) to form a productive ecosystem. Like any ecosystem, the host microenvironment requires balanced nutrient cycling in the form of cellular signaling to respond dynamically to perturbations to the ecosystem. Typically, small disturbances result in a more robust ecosystem, such as immunity following viral infection. In contrast, the native ecosystem of the host microenvironment is unable to recover from the chronic acyclic nutrient cycling induced by an invading species: cancer.
Table 1: Ecological characteristics applied to cancer biology
Characteristic |
Ecology |
Cancer biology |
Biosphere |
Earth |
Patient |
Ecosystem |
Lake |
Organ system |
Species |
Animals and plants |
Cell types |
Abiotic factors |
Land or water |
Extracellular matrix |
Nutrient cycling |
Biogeochemical cycling |
Cell-to-cell signaling |
Invasive species |
Beavers, kudzu |
Cancer |
Biogas |
Swamp gas or firedamp |
Cytokine release |
Ecosystem collapse |
Mass extinction |
Organ failure and death |
Homeostatic nutrient cycling in ecosystems
Central to the success of an ecosystem is complete and efficient nutrient cycling. Nutrients, or resources, are substances required for healthy growth, viability, and function of a species [10]. Nutrient cycling is the exchange of these resources from the abiotic nutrient pool to biotic producers and consumers and ultimately back into the nutrient pool to repeat the cycle [10]. Nutrient loss must be countered by nutrient gain to maintain high ecosystem productivity [10, 12]. The classic example of closed resource cycling is the nitrogen cycle: atmospheric nitrogen (environmental nutrient sink) is fixed by plants (producers) that are then ingested by animals (consumers). Finally, animal waste and dead organic material decays and nitrogen is released back into the nutrient pool.
Nutrient cycles are self-regulated by dynamic feedback mechanisms to maintain ecosystem-wide homeostasis. Within this framework, each species occupies a unique niche, the functional role that describes its resource use and how it requires and supports other components of the ecosystem. A robust ecosystem with diverse niche occupants is able to rapidly recover from disturbances including altered resource availability, physical changes in the habitat, or other changes to the environment such as flooding or fire if they are separated in physical space and in time [13]. While minor disturbances to an ecosystem allow for increased resiliency, large disturbances may be catastrophic, resulting in community disassembly and ecosystem failure. Rapid or continuous alterations to any node of a nutrient cycle overwhelm the native feedback mechanisms that regulate complete resource recycling.
Acyclic nutrient cycling: ecosystem engineering by invasive species
One widespread instigator of unbalanced nutrient cycling is the introduction of a non-native species into an ecosystem. Successful invaders are characterized by rapid growth, high reproductive rates, and phenotypic plasticity [14, 15]. Concurrently, invasive species decrease the individual fitness of native species, altering community structure and nutrient cycling, and ultimately destroying the native ecosystem. Cancer cells act as an invasive species within a host organ, disrupting the homeostasis produced and controlled by the normal host cell species [2, 4, 16].
By nature of their expansion into a new ecosystem, invasive species are inherently ecosystem engineers. Ecosystem engineers change their abiotic habitat and modify resource availability to other species through direct mechanical alteration (allogenic engineering) or by modifying themselves (autogenic engineering) [17]. For example, kudzu is an invasive autogenic engineering species. As it grows and climbs native vegetation, it introduces new habitats for small animals, displacing the native vegetative species. In addition, kudzu competes with native plant species for sunlight and soil nutrients, thus altering local resource recycling. Perhaps the most recognizable allogenic engineer is the North American beaver. Beavers physically alter the native habitat by clear-cutting the terrestrial habitat and building dams that stop water flow on a river. The resulting beaver pond is a result of a destroyed habitat, displacing native species that require flowing water or tree stands for survival, thus breaking the consumer-producer relationship and altering species stoichiometry by altering resource cycling. Other species that favor open water or non-shaded wetlands colonize the beaver-engineered habitat. Ultimately, this results in complete community restructuring and the destruction of the native ecosystem even as a new one evolves to replace it.
Invasive species alter nutrient recycling through multiple different mechanisms. Any perturbation to an ecosystem’s community structure will influence nutrient cycling as critical nodes increase or decrease, thus inducing acyclic nutrient recycling. Discrete trait invaders may directly increase usable nitrogen levels for consumers by introducing a novel nitrogen-fixation ability or may decrease nitrogen fixation of native species by altering soil chemistry [18–21]. Invasive species with rapidly decomposed organic waste will inherently increase the levels of nutrients in the ecosystem, ultimately leading to eutrophication of the habitat [20, 22, 23]. This has long term effects on the species in the ecosystem as altered nutrient cycling influences the quality of subsequent generations.
Acyclic nutrient cycling: eutrophication
Eutrophication is the enrichment of an ecosystem with chemical or organic nutrients. Eutrophication is considered a healthy process when it occurs slowly on a geological time scale as part of the natural aging of a lake to a productive meadow [24]. When accelerated, however, eutrophication dismantles normal nutrient cycling and litter feedbacks, resulting in altered species stoichiometry and, if left unchecked, ecosystem failure [25]. Human acceleration of eutrophication (cultural eutrophication) of watersheds is one of the most apparent examples of forced acyclic nutrient cycling [10, 26]. Pollution in the form of fertilizers and sewage leads to local nutrient enrichment, specifically of phosphorus and nitrogen, two of the limiting growth factors necessary for photosynthesis [27]. The rapid accumulation of excess nutrients accelerates the creation of a swamp by inducing acyclic resource recycling that leads to the growth and expansion of photosynthetic organisms such as short-lived cyanobacteria that compose characteristic algal blooms. As the organic material of these algae accumulates, decomposition levels increase, consuming high levels of oxygen and leading to severe hypoxic conditions. The oxygen-poor environment is unable to support native consumer species such as fish or mollusks and is colonized by detritus-feeders (Figure 1). Nutrient cycling becomes weighted towards the activities of producers and lacks the negative feedback from consumer species. In addition, algae blooms may also directly poison consumer species, further exacerbating acyclic resource cycling [28, 29]. Such an ecosystem is unstable and susceptible to irreversible collapse.
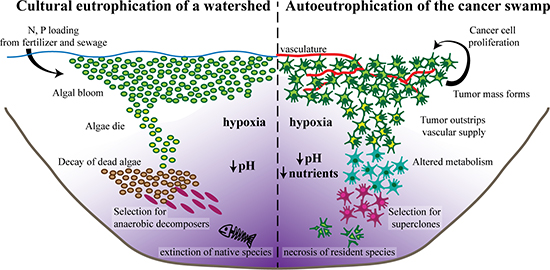
Figure 1: Autoeutrophication of the hypoxic, nutrient-poor, and acidic “cancer swamp”. (Left) Excess nitrogen and phosphorus deposits stimulate the growth of photosynthetic algae, resulting in a characteristic algal bloom. As the algae die off, organic material accumulates and decomposition levels increase, leading to severe hypoxia. These harsh conditions select for efficient anaerobic decomposers. The build-up of the waste product of anaerobic fermentation, carbon dioxide, results in an acidic environment. Ultimately, the severe conditions lead to the local extinction of native species and eventual irreversible ecosystem collapse. (Right) Even in the absence of external stimuli, cancer cells have a high proliferation rate, rapidly expanding to a tumor mass analogous to an algal bloom. As the tumor grows, it quickly outstrips its vascular supply, resulting in a hypoxic microenvironment. To survive, the cancer cells alter their metabolism to utilize relatively inefficient anaerobic glycolysis, exhausting available nutrient sources. The accumulation of lactic acid, the waste product of anaerobic glycolysis, results in an acidic microenvironment. Ultimately, the harsh “cancer swamp” selects for highly lethal cancer superclones. Simultaneously, the toxic conditions lead to increased rates of necrosis, extinction of native host cell types, and eventual organ failure.
The characteristic hypoxia of a eutrophic habitat requires anaerobic fermentation to decompose organic material. The byproduct of this process is typically a toxic combination of methane and hydrogen sulfide that bubbles to the surface as swamp gas. This phenomenon is largely harmless when gas is released into the atmosphere and diffuses, but in a closed habitat such as a peat bog or coal mine (“firedamp”) leads to dangerous accumulation of the flammable biogas, resulting in fires smoldering below ground or spontaneous explosions (Figure 2) [30, 31]. Thus, in a confined area, swamp gas can be rapidly catastrophic and may have far-reaching effects on neighboring ecosystems.
Invasion and autoeutrophication: the “cancer swamp”
Cancer cells act as an invasive species as they expand within the host ecosystem as a primary tumor and again later as they colonize a distant site as a metastasis [9, 16]. Many of the hallmarks of cancer including high proliferation rates, avoiding predation by the native species, and resisting death are analogous to the high trait values expressed by ecological invasive species [3, 4]. With the advantageous phenotypic traits of an invasive species, tumor cells rapidly become ecosystem engineers by dismantling the host species community structure leading to the destruction of the native ecosystem and by generating a pro-tumorigenic microenvironment. Tumor cells alter the host ecosystem through both allogenic and autogenic means. As allogenic engineers, the cells physically alter the native habitat by secreting factors to destroy the extracellular matrix (production of MMPs), to recruit pro-tumorigenic macrophages (production of cytokines and chemokines including IL-6, TNFα, etc.), and to induce angiogenesis (production of VEGF) [3, 4, 32–34]. Simultaneously, autogenic engineering occurs as the tumor physically grows in size: tumor cells exhaust local sinks of energy and oxygen and increase litter concentration, ultimately overwhelming native feedback mechanisms.
This process, which we define as autoeutrophication, is analogous to ecological cultural eutrophication, but with significant differences. Most notably, while eutrophic watersheds are the result of external pollution, the “cancer swamp” is self-instigated. The positive feedback of high cancer cell proliferation rates and rapid tumor growth mean that the cancer cells act as both the “pollution” stimulus and responding “algal bloom” in the tumor microenvironment. Autoeutrophication, therefore, is self-driven by the cancer itself and, though it has substantial effects on the host, it is not governed by external factors (Figure 1).
As the cancer cells proliferate, the tumor rapidly grows and physically displaces the native habitat through autogenic engineering processes. Importantly, even in a clinically undetectable mass of 1 mm3, the tumor has outgrown the available vasculature, overwhelming both the incoming tributaries that provide oxygen and energy sources and the distuaries that carry away cellular waste [35]. Therefore, the tumor rapidly exhausts the local nutrient and oxygen sources while simultaneously poisoning the habitat with waste products. Under the hypoxic conditions characteristic of a poorly vascularized tumor, cancer cells switch from using oxidative phosphorylation for energy production to utilizing relatively inefficient anaerobic glycolysis, a process known as the “Warburg effect.” The corresponding accumulation of lactic acid, a waste product of glycolysis, results in an acidic microenvironment [36, 37]. Because glycolysis is relatively inefficient (2 mol ATP for each mol glucose) as compared to mitochondrial oxidative phosphorylation, energy sources are rapidly exhausted, resulting in a metabolite-poor habitat. Ultimately, this autoeutrophication displaces the native healthy ecosystem with a nutrient-poor, acidic, and hypoxic “cancer swamp” (Figure 1).
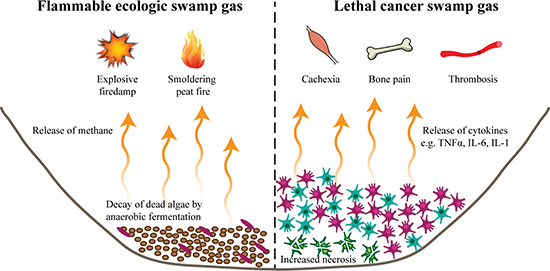
Figure 2: Release of toxic swamp gas. (Left) A byproduct of decomposition by anaerobic fermentation is methane gas that bubbles to the surface as swamp gas. When near a sufficient ignition source, accumulation of this gas can lead to smoldering underground fires in peat fields or explosions in coal mines (“firedamp”). (Right) The release of lysed cell products of necrotic cells combined with the pro-inflammatory cytokines secreted from the cancer cells produce the equivalent of swamp gas. At high and persistent levels, the release of this “swamp gas” from multiple metastatic sites leads to cytokine-mediated smoldering (e.g. cachexia or bone pain) or acute (e.g. thrombosis) lethal syndromes, the cause of death in the many of patients.
While a tumor mass directly promotes the destruction of the local ecosystem, the lethal clinical syndromes responsible for many cancer deaths are also the result of the toxic cancer swamp. Similar to algae blooms of a eutrophic lake, there are multiple simultaneous mechanisms of cancer cell-mediated toxicity. Analogous to the increased decomposition rates of algae, the hypoxic and glucose-depleted “cancer swamp” promotes cancer and host cell necrosis [38, 39]. In contrast with tightly regulated apoptotic cell death, necrotic cells expand, lose cell membrane integrity, and explode, releasing their intracellular contents into the environment. In addition, the deregulated “nutrient cycling” (cell-cell signaling) of the cancer cells within the “cancer swamp” leads to an accumulation of secreted cytokines and other factors. As with ecologic “swamp gas,” when low concentrations are released into open atmosphere, these factors are relatively harmless. However, at high levels within a closed system, such as the biosphere of a cancer patient, the release of “cancer swamp gas” is catastrophic, either inducing a smoldering lethal syndrome (e.g. cytokine-mediated cachexia or bone pain) or an acute lethal event (e.g. thrombosis) (Figure 2) [2, 40–45].
Ecological inheritance: selection of traits by the self-engineered ecosystem of the cancer swamp
While ecosystem engineering describes an organism’s impact on other species and communities of the ecosystem, the same actions also play a role in niche construction, i.e., how the environmental changes made by the species impact the selective pressure and subsequent adaptation of the species in its newly engineered niche [17, 46]. Inherent to the adaptive processes of niche construction is ecological inheritance. While genetic inheritance describes the genetic material inherited from an organism’s ancestors, ecological inheritance describes the selective pressure associated with the engineered environment inherited from an organism’s ancestors [47]. For example, when a beaver builds a dam, it not only is building a habitat, it also alters nutrient cycling and decomposition dynamics. Successful offspring, therefore, must be able to maintain and survive in the engineered environment by expressing high trait values for niche construction (dam building) and for traits favored in the niche (open-water pond and clear-cut wetland). Thus, there is a co-evolution of traits that generate the adaptive pressure (engineering) and traits that are dependent on the adaptive pressure (survival in the engineered ecosystem) [48].
As tumor cells engineer their environment to create the toxic “cancer swamp,” they simultaneously create a habitat that will exert selective pressure on subsequent generations of daughter cancer cells. Ecological inheritance of the cancer swamp may promote biodiversity within the cancer cell population, contributing to the high level of genetic and epigenetic heterogeneity within tumors [16]. However, the clonal architecture of cancer cells within a tumor suggests that most if not all cancer cells have the capacity to metastasize [49–51]. The stringent adaptive pressures of the malignant microenvironment of the “cancer swamp” may enrich for a small subset of highly lethal cancer cell clones that are phenotypically equipped to leave the primary ecosystem and migrate to and colonize a distant site (Figure 1). For example, hypoxia epigenetically induces the expression of the transcription factor HIF1α that in turn induces the expression of epithelial-to-mesenchymal transition (EMT) cellular programs that contribute to the production of the pro-metastatic mesenchymal phenotype [52–54]. Exposing murine sarcoma and melanoma cell lines to glucose starvation increases tumor foci in a forced lung metastasis model [55]. Moreover, culturing human cancer cell lines in acidic pH, the last characteristic of the “cancer swamp,” increases the mesenchymal phenotype in vitro, and increases metastatic potential in vivo [56–58].
The principals of ecology and ecological inheritance suggest that tumor cell heterogeneity can not only be generated by changes to the genome as a result of inherent genetic instability and altered DNA repair mechanisms but also by genetic trait selection as a result of environmental pressures in classic Darwinian fashion. Initiating events are likely the result of genomic alterations may be caused by many different events, including stochastic mutation and carcinogenic alterations to DNA damage repair mechanisms, among others [4]. Without the necessary selective pressures, however, the phenotypes that give rise to the additional necessary tumorigenic characteristics are unlikely to evolve. We hypothesize that the selective pressure of the “cancer swamp” provides the necessary selective pressure to direct natural selection to enrich for aggressive cancer cell clones with the phenotypic capacity to either survive in the harsh toxic “swamp” as an aggressive tumor cell or gain the ability to leave the tumor ecosystem and metastasize a distant site. Without the selective pressure of the engineered ecosystem, the cancer cells are much more likely to remain restrained to the primary tumor rather than metastasize to distant sites. This may explain, in part, why benign tumors exist. By growing extremely slowly, lipomas, for example, never outstrip their blood supply and therefore never feel the selective pressure to undergo the epigenetic changes that promote pro-metastatic cellular programs.
Using restoration ecology strategies as anti-cancer therapies
Restoration ecology arose as a practical field of study in response to the increasingly negative impact of human activity on ecosystems worldwide. The preferred scenario for preservation is conservation, i.e. protecting the ecosystem prior to invasion or pollution. In cancer biology, these strategies are parallel to patient recommendations such as diet and exercise modification or daily administration of low-dose aspirin [59] to reduce general cancer risk.
Conservation efforts, however, while preferable both in ecology and in cancer biology, are often not sufficient on a larger scale and further intervention is necessary to restore the native ecosystem. Restoration ecologists aim to actively restore damaged ecosystems through systematic intervention to remove invading species, reduce eutrophication, and improve habitat quality for native species. Taking advantage of the successes of restoration ecology, such as the restoration of Lake Erie, cleanup efforts following oil spills, and community watershed and beach management, points of therapeutic intervention to likewise restore the “cancer swamp” can be identified (Figure 3, Table 2).
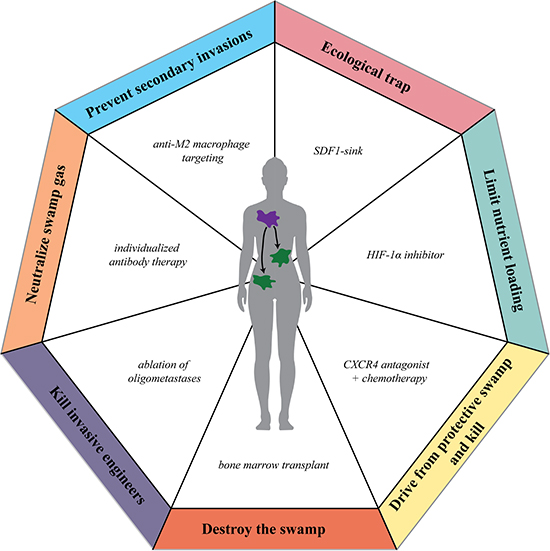
Figure 3: Using restoration ecology strategies as anti-cancer therapies. Strategies used to restore damaged ecologic ecosystems can be applied to develop therapeutics to restore the “cancer swamp.”
Table 2: Restoration ecology strategies applied to development of cancer treatments
Ecology |
Cancer |
|||
Problem |
Intervention |
Problem |
Intervention |
Clinical status |
Ecosystem collapse due to ecosystem engineering |
Draining the swamp |
Cancer microenvironment replaces native ecosystem |
Bone marrow transplant |
Standard of care (multiple myeloma, leukemia) |
Excessive nutrient loading |
Reduce pollution |
Cancer cell response to hypoxia |
HIF-1α inhibitor |
|
Invasive species |
Ecological trap |
Cancer cell homing to metastatic site |
SDF1-sink |
|
Repeated invasions |
Ecological trap and kill |
Cancer cell homing to metastatic site |
CXCR4 antagonist + chemotherapy |
Phase 1/2 clinical trial [67] |
Aggressive invasive ecosystem engineering |
Kill dispersing invasive engineers |
Oligometastases |
Ablation of oligometastases |
In use (radiation, surgery) |
Secondary invasions of an unstable ecosystem |
Physical barriers to prevent invasion |
Recruitment of pro-tumor M2 macrophages |
Anti-M2 macrophage targeting |
|
Release of toxic swamp gas |
Preventative burn-off |
Cytokine-mediated lethal syndromes |
Individualized antibody therapy |
In use as anti-inflammatory therapy |
One of the largest successes of active intervention in both ecology and oncology is draining swampland. In regions with low-lying geography and high waterfall, swamps are the ideal breeding ground for malaria-carrying mosquitos. Historically, to reduce malaria in these regions, rather than directly killing the mosquitos using insecticides, a more effective strategy is to drain the swamp water and eliminate the favorable ecosystem for the mosquitos. The parallel strategy in hematological malignancies is total body irradiation followed by bone marrow transplant, leading to effective cures in leukemia and multiple myeloma patients.
The most effective management strategy to restore eutrophic watersheds — especially striking in the recovery of Lake Erie — is to reduce nutrient loading by implementing anti-erosion protocols, altering fertilizer application timing, and improving sewage treatment. In the autoeutrophic “cancer swamp,” because the eutrophication is self-instigated and self-maintained, reducing the equivalent of nutrient loading in the cancer microenvironment is currently not possible. Unlike in ecology, however, we can modulate the response of cancer cells to the eutrophic microenvironment with strategies such as HIF-1α inhibitors [60–62].
Ecological traps are poor-quality habitats that have disproportionate attractiveness for the actual survival value of the region [63]. Ecological traps can be used to attract otherwise harmful invasive species from the primary tumor site to a specific engineered site that can be easily managed [8, 64]. For cancer types that home to bone following the SDF-1 gradient, an artificial SDF1-sink could be introduced to attract malignant cells to an engineered site and specifically ablated [65, 66]. Using a similar strategy, resident invasive species can be driven away from their protective engineered habitat and eliminated in the vulnerable transit state. This strategy using CXCR4 antagonists in combination with chemotherapy has already completed clinical trials in leukemia [67] and is entering clinical trial for treatment of solid tumors.
A major concern in a region with a series of susceptible ecosystems is the dispersal of aggressive invasive ecosystem engineers from one site to additional sites, eventually leading to widespread ecosystem destruction. Immediate and specific elimination of these invasive engineers and their adopted environments, prior to secondary ecosystem collapse, is essential to protecting the larger region, especially after the primary invaded site has already undergone substantial engineering. In cancer biology, the equivalent problem is oligometastases, an intermediate state between a localized primary tumor and systemic metastatic disease [68]. Ablation of these small metastatic sites is becoming more widespread in clinical practice and has shown a survival benefit in a variety of solid tumor types [69].
Another concern of an invaded ecosystem is its susceptibility to secondary invasion by another species that may accelerate native ecosystem collapse. In ecology, physical barriers, such as the electric barriers to restrict Asian Carp entry into Lake Michigan, can be established to eliminate or reduce migration of secondary invaders into an engineered ecosystem. In cancer biology, one of the most abundant and destructive secondary invaders to a tumor site is pro-tumorigenic M2-macrophages. Targeted anti-M2 macrophage therapy to specifically eliminate these tumor-associated-macrophages has entered clinical trial and developing novel M2-targeting modalities is an active area of research [70, 71].
Finally, when active management to restore a damaged ecosystem fails, ecologists turn to physically managing the destructive element. For instance, controlled burning eliminates accumulated biogas to prevent mine explosions. Following a large-scale oil spill, a combination of mechanical, chemical, and biological methods are used to minimize ecosystem damage of the toxic oil on native fauna and flora. Similarly, in advanced cancer patients, individualized antibody therapy using therapeutics already in use such as anti-inflammatory agents for arthritis, psoriasis, and asthma, could be used to specifically neutralize a patient’s specific cytokine “cancer swamp gas” to reduce cytokine-mediated syndromes (i.e. cachexia, bone pain, and thrombosis).
SUMMARY
As cancer cells generate their own niche within the tumor ecosystem, ecological engineering and autoeutrophication displace normal cell function and result in the creation of a hypoxic, acidic, nutrient-poor environment. This “cancer swamp” has effects at both the local ecosystem and systemic host levels through multiple genetic and epigenetic mechanisms. The microenvironment is a protective habitat for the tumor cells, helping them to avoid detection and destruction by host immune cells that avoid the harsh conditions. Simultaneously, the “cancer swamp” attracts pro-tumorigenic immune cells such as M2-tumor associated macrophages that further help engineer the environment (secretion of MMPs), further promoting tumor growth and expansion of the “cancer swamp.” Environmental hypoxia induces neo-angiogenesis to increase circulation and therefore oxygen and nutrient levels within the tumor, in essence “irrigating” the “cancer swamp,” ensuring that the tumor does not undergo immediate ecosystem collapse. Hypoxia and acidity also promote EMT in cancer cells, increasing the likelihood of successful metastasis by enhancing motility and invasive phenotypes. At the host level, the combination of the release of lysed cell products as well as pro-inflammatory chemokines and cytokines from multiple metastatic sites leads to the production of the cancer equivalent of swamp gas, ultimately leading to a cytokine mediated death in many cancer patients.
ACKNOWLEDGMENTS
This work was supported by NCI grant nos. U54CA143803, CA163124, CA093900 and CA143055. to K.J.P. The authors thank the members of the Brady Urological Institute, especially Dr. Donald S. Coffey and members of the Pienta laboratory for thoughtful discussion.
REFERENCES
1. Paget G. Remarks on a Case of Alternate Partial Anaesthesia. British medical journal. 1889; 1:1–3.
2. Weilbaecher KN, Guise TA, McCauley LK. Cancer to bone: a fatal attraction. Nature reviews Cancer. 2011; 11:411–425.
3. Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000; 100:57–70.
4. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144:646–674.
5. Merlo LM, Pepper JW, Reid BJ, Maley CC. Cancer as an evolutionary and ecological process. Nature reviews Cancer. 2006; 6:924–935.
6. Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012; 21:309–322.
7. Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nature medicine. 2013; 19:1423–1437.
8. Pienta KJ, Robertson BA, Coffey DS, Taichman RS. The cancer diaspora: Metastasis beyond the seed and soil hypothesis. Clinical cancer research : an official journal of the American Association for Cancer Research. 2013; 19:5849–5855.
9. Pienta KJ, McGregor N, Axelrod R, Axelrod DE. Ecological therapy for cancer: defining tumors using an ecosystem paradigm suggests new opportunities for novel cancer treatments. Translational oncology. 2008; 1:158–164.
10. Eugene P, Odum GWB. Fundamentals of Ecology: Thomson Brooks/Cole. 2005.
11. Tansley AG. The use and abuse of vegetational concepts and terms. Ecology. 1935; 16:284–307.
12. Krebs CJ. The message of ecology: Indo American Books. 2007.
13. McClain ME, Bilby RE, Triska FJ. Nutrient cycles and responses to disturbance. River ecology and management: Lessons from the Pacific coastal ecoregion. 1998; 347–372.
14. Davidson AM, Jennions M, Nicotra AB. Do invasive species show higher phenotypic plasticity than native species and, if so, is it adaptive? A meta-analysis. Ecology letters. 2011; 14:419–431.
15. Kolar CS, Lodge DM. Progress in invasion biology: predicting invaders. Trends in Ecology & Evolution. 2001; 16:199–204.
16. Yang KR, Mooney SM, Zarif JC, Coffey DS, Taichman RS, Pienta KJ. Niche inheritance: a cooperative pathway to enhance cancer cell fitness though ecosystem engineering. Journal of cellular biochemistry. 2014; 115:1478–1485.
17. Erwin DH. Macroevolution of ecosystem engineering, niche construction and diversity. Trends in Ecology & Evolution. 2008; 23:304–310.
18. Vitousek PM. Concepts of Ecosystem Ecology. A Comparative View. Pomeroy L.R, Alberts J.J. Springer-Verlag New York. 1988. xii, 384 pp., illus. $72. Ecological Studies, vol. 67 Based on a conference. Science. 1989; 243:825–826.
19. Vitousek PM, Walker LR, Whiteaker LD, Mueller-Dombois D, Matson PA. Biological Invasion by Myrica faya Alters Ecosystem Development in Hawaii. Science. 1987; 238:802–804.
20. Ehrenfeld JG. Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems. 2003; 6:503–523.
21. Rice E. Allelopathic effects on nitrogen cycling. Allelopathy: Springer. 1992; 31–58.
22. Van Vuuren M, Aerts E, Berendse F, De Visser W. Nitrogen mineralization in heathland ecosystems dominated by different plant species. Biogeochemistry. 1992; 16:151–166.
23. Van Vuuren MM, Berendse F. Changes in soil organic matter and net nitrogen mineralization in heathland soils, after removal, addition or replacement of litter from Erica tetralix or Molinia caerulea. Biology and Fertility of Soils. 1993; 15:268–274.
24. Nixon SW. Coastal marine eutrophication: a definition, social causes, and future concerns. Ophelia. 1995; 41:199–219.
25. Callisto M, Molozzi J, Barbosa JLE. Eutrophication of Lakes. Eutrophication: Causes, Consequences and Control: Springer. 2014; 55–71.
26. Nagar B, Datta M, Khakhar M, Indian J. Eutrophication and recovery in experimental lakes: implications for lake management. 1974.
27. Khan MN, Mohammad F. Eutrophication: challenges and solutions. Eutrophication: Causes, Consequences and Control: Springer. 2014; 1–15.
28. Francis G. Poisonous Australian Lake. Nature. 1878; 18.
29. Landsberg JH. The effects of harmful algal blooms on aquatic organisms. Reviews in Fisheries Science. 2002; 10:113–390.
30. Watts AC, Kobziar LN. Smoldering Combustion in Organic Soils: Peat and Muck Fires in the Southeastern US.
31. Singh AK, Singh R, Singh MP, Chandra H, Shukla N. Mine fire gas indices and their application to Indian underground coal mine fires. International Journal of coal geology. 2007; 69:192–204.
32. Deryugina EI, Quigley JP. Matrix metalloproteinases and tumor metastasis. Cancer metastasis reviews. 2006; 25:9–34.
33. Burkholder B, Huang RY, Burgess R, Luo S, Jones VS, Zhang W, Lv ZQ, Gao CY, Wang BL, Zhang YM, Huang RP. Tumor-induced perturbations of cytokines and immune cell networks. Biochimica et biophysica acta. 2014; 185:182–201.
34. Catalano V, Turdo A, Di Franco S, Dieli F, Todaro M, Stassi G. Tumor and its microenvironment: a synergistic interplay. Seminars in cancer biology. 2013; 23:522–532.
35. Folkman J. Toward an understanding of angiogenesis: search and discovery. Perspect Biol Med. 1985; 29:10–36.
36. Zheng J. Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (Review). Oncology letters. 2012; 4:1151–1157.
37. Fantin VR, St-Pierre J, Leder P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer cell. 2006; 9:425–434.
38. Kim CH, Ko AR, Lee SY, Jeon HM, Kim SM, Park HG, Han SI, Kang HS. Hypoxia switches glucose depletion-induced necrosis to phosphoinositide 3-kinase/Akt-dependent apoptosis in A549 lung adenocarcinoma cells. International journal of oncology. 2010; 36:117–124.
39. Steinbach J, Wolburg H, Klumpp A, Probst H, Weller M. Hypoxia-induced cell death in human malignant glioma cells: energy deprivation promotes decoupling of mitochondrial cytochrome c release from caspase processing and necrotic cell death. Cell Death & Differentiation. 2003; 10:823–832.
40. Loberg RD, Bradley DA, Tomlins SA, Chinnaiyan AM, Pienta KJ. The lethal phenotype of cancer: the molecular basis of death due to malignancy. CA: a cancer journal for clinicians. 2007; 57:225–241.
41. Narsale AA, Carson JA. Role of interleukin-6 in cachexia: therapeutic implications. Current opinion in supportive and palliative care. 2014; 8:321–327.
42. Cohen S, Nathan JA, Goldberg AL. Muscle wasting in disease: molecular mechanisms and promising therapies. Nature reviews Drug discovery. 2014; 14:58–74.
43. Lozano-Ondoua AN, Symons-Liguori AM, Vanderah TW. Cancer-induced bone pain: Mechanisms and models. Neuroscience letters. 2013; 557 Pt A:52–59.
44. Andrea N, Ansell J. Management of thrombosis in the cancer patient. The journal of supportive oncology. 2003; 1: 235–238, 240–232; discussion 239–240, 243–235.
45. Shelke AR, Khorana AA. Cancer-associated thrombosis: an update. Drug Discovery Today: Disease Mechanisms. 2011; 8:e39–e45.
46. Day RL, Laland KN, Odling-Smee FJ. Rethinking adaptation: the niche-construction perspective. Perspectives in biology and medicine. 2003; 46:80–95.
47. Odling-Smee J, Erwin DH, Palkovacs EP, Feldman MW, Laland KN. Niche construction theory: a practical guide for ecologists. The Quarterly review of biology. 2013; 88:3–28.
48. Laland KN, Boogert NJ. Niche construction, co-evolution and biodiversity. Ecological economics. 2010; 69:731–736.
49. Greaves M, Maley CC. Clonal evolution in cancer. Nature. 2012; 481:306–313.
50. Nowell PC. The clonal evolution of tumor cell populations. Science. 1976; 194:23–28.
51. Talmadge JE. Clonal selection of metastasis within the life history of a tumor. Cancer research. 2007; 67:11471–11475.
52. Semenza GL. Hypoxia-inducible factors: mediators of cancer progression and targets for cancer therapy. Trends in pharmacological sciences. 2012; 33:207–214.
53. Esteban MA, Tran MG, Harten SK, Hill P, Castellanos MC, Chandra A, Raval R, O’Brien TS, Maxwell PH. Regulation of E-cadherin expression by VHL and hypoxia-inducible factor. Cancer research. 2006; 66:3567–3575.
54. Krishnamachary B, Zagzag D, Nagasawa H, Rainey K, Okuyama H, Baek JH, Semenza GL. Hypoxia-inducible factor-1-dependent repression of E-cadherin in von Hippel-Lindau tumor suppressor-null renal cell carcinoma mediated by TCF3, ZFHX1A, and ZFHX1B. Cancer research. 2006; 66:2725–2731.
55. Schlappack OK, Zimmermann A, Hill RP. Glucose starvation and acidosis: effect on experimental metastatic potential, DNA content and MTX resistance of murine tumour cells. British journal of cancer. 1991; 64:663–670.
56. Suzuki A, Maeda T, Baba Y, Shimamura K, Kato Y. Acidic extracellular pH promotes epithelial mesenchymal transition in Lewis lung carcinoma model. Cancer cell international. 2014; 14:129.
57. Rofstad EK, Mathiesen B, Kindem K, Galappathi K. Acidic extracellular pH promotes experimental metastasis of human melanoma cells in athymic nude mice. Cancer research. 2006; 66:6699–6707.
58. Martinez-Zaguilan R, Seftor EA, Seftor RE, Chu YW, Gillies RJ, Hendrix MJ. Acidic pH enhances the invasive behavior of human melanoma cells. Clinical & experimental metastasis. 1996; 14:176–186.
59. Cuzick J, Thorat MA, Bosetti C, Brown PH, Burn J, Cook NR, Ford LG, Jacobs EJ, Jankowski JA, La Vecchia C, Law M, Meyskens F, Rothwell PM, Senn HJ, Umar A. Estimates of benefits and harms of prophylactic use of aspirin in the general population. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. 2015; 26:47–57.
60. Borsi E, Perrone G, Terragna C, Martello M, Zamagni E, Tacchetti P, Pantani L, Brioli A, Dico AF, Zannetti BA, Rocchi S, Cavo M. HIF-1alpha inhibition blocks the cross talk between multiple myeloma plasma cells and tumor microenvironment. Experimental cell research. 2014; 328:444–455.
61. Burroughs SK, Kaluz S, Wang D, Wang K, Van Meir EG, Wang B. Hypoxia inducible factor pathway inhibitors as anticancer therapeutics. Future medicinal chemistry. 2013; 5:553–572.
62. Palayoor ST, Mitchell JB, Cerna D, Degraff W, John-Aryankalayil M, Coleman CN. PX-478, an inhibitor of hypoxia-inducible factor-1alpha, enhances radiosensitivity of prostate carcinoma cells. International journal of cancer Journal international du cancer. 2008; 123:2430–2437.
63. Robertson BA, Rehage JS, Sih A. Ecological novelty and the emergence of evolutionary traps. Trends Ecol Evol. 2013; 28:552–560.
64. van der Sanden B, Appaix F, Berger F, Selek L, Issartel JP, Wion D. Translation of the ecological trap concept to glioma therapy: the cancer cell trap concept. Future oncology. 2013; 9:817–824.
65. Bladergroen BA, Siebum B, Siebers-Vermeulen KG, Van Kuppevelt TH, Poot AA, Feijen J, Figdor CG, Torensma R. In vivo recruitment of hematopoietic cells using stromal cell-derived factor 1 alpha-loaded heparinized three-dimensional collagen scaffolds. Tissue engineering Part A. 2009; 15:1591–1599.
66. Rabbany SY, Pastore J, Yamamoto M, Miller T, Rafii S, Aras R, Penn M. Continuous delivery of stromal cell-derived factor-1 from alginate scaffolds accelerates wound healing. Cell transplantation. 2010; 19:399–408.
67. Uy GL, Rettig MP, Motabi IH, McFarland K, Trinkaus KM, Hladnik LM, Kulkarni S, Abboud CN, Cashen AF, Stockerl-Goldstein KE, Vij R, Westervelt P, DiPersio JF. A phase 1/2 study of chemosensitization with the CXCR4 antagonist plerixafor in relapsed or refractory acute myeloid leukemia. Blood. 2012; 119:3917–3924.
68. Hellman S. Karnofsky Memorial Lecture. Natural history of small breast cancers. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 1994; 12:2229–2234.
69. Rastogi S, Gulia S, Bajpai J, Ghosh J, Gupta S. Oligometastatic breast cancer: A mini review. Indian journal of medical and paediatric oncology : official journal of Indian Society of Medical & Paediatric Oncology. 2014; 35:203–206.
70. Ries CH, Cannarile MA, Hoves S, Benz J, Wartha K, Runza V, Rey-Giraud F, Pradel LP, Feuerhake F, Klaman I, Jones T, Jucknischke U, Scheiblich S, Kaluza K, Gorr IH, Walz A, et al. Targeting tumor-associated macrophages with anti-CSF-1R antibody reveals a strategy for cancer therapy. Cancer Cell. 2014; 25:846–859.
71. Tang X, Mo C, Wang Y, Wei D, Xiao H. Anti-tumour strategies aiming to target tumour-associated macrophages. Immunology. 2013; 138:93–104.

