Introduction
Penile cancer (PC) is a malignant neoplasm that frequently affects males in their sixth decade of life with low socioeconomic status and educational level [1]. This is an uncommon disease in developed countries, but highly incident in developing nations. Brazil holds the global record for PC incidence [2]. The etiology of PC is heterogeneous and remains under investigation; however, some risk factors, such as phimosis, poor hygiene, alcoholism and smoking, and human papillomavirus (HPV) infection, have been proposed [1–4].
Among these risk factors, HPV infection appears to play a role in approximately one-half of all cases. The mechanism of HPV action in PC appears to be similar to that in cervical cancer, in which the oncoproteins E6 and E7 act by inactivating the suppressor proteins p53 and retinoblastoma (pRb), respectively [5]. These oncoproteins directly impact control of the cell cycle, which leads to dysregulation in cell proliferation. When the E7 oncoprotein binds pRb, it triggers the release of the E2F transcription factor, which then induces cell cycle progression [6]. In several tumors, this mechanism triggers a cellular response frequently related to p16 overexpression and decrease in cyclin D1 expression [7, 8].
Cyclin D1 is a protein encoded by the CCND1 gene and acts in the G1/S phase of the cell cycle, positively regulating cell proliferation and differentiation [8]. This protein has been widely studied due to its important role in cell cycle regulation and biochemistry, both in normal and tumor cells [9]. Overexpression of cyclin D1 can lead to shortening of the G1 phase, reducing dependence on mitogenic factors and causing increased cell proliferation. Thus, it has been proposed that this mechanism may be involved in cell transformation and tumorigenesis [10].
Several studies have demonstrated changes in cyclin D1 expression, both in the early stage and along tumor progression, in different types of cancers, including breast [10], head and neck [11], prostate [12] and renal cell carcinoma [13] and PC [14]. These findings suggest that cyclin D1 may be a potential biomarker for cancer.
Regarding PC, however, few studies have assessed the role of cyclin D1, reinforcing the necessity for initiatives that aim to investigate its actual role in the pathophysiology of this disease. As such, the present study aimed to characterize the expression of cyclin D1 in patients with PC, and to determine possible correlations with the clinical and histopathological features of the disease.
Results
Clinical profile of patients with PC
The mean age of the study cohort was 60.1 years (range, 26 to 93 years), with the majority being ≤60 years of age (46.0%). These patients had a low educational level (99.0%), the majority were farmers (66.5%) with poor hygiene habits (72.5%) and phimosis (67.5%). Risky sexual behavior was also described, in which 70% of patients reported to have >6 sexual partners through life and 58% reported engaging in sex with animals (i.e., zoophilia). Clinical and epidemiological information is summarized in Table 1.
Table 1: Clinical-epidemiological features of 100 patients with penile cancer
| Variables | N (%) |
|---|---|
| Age group | |
| 18---| 40 years | 18 (18) |
| 41---| 60 years | 28 (28) |
| >60 years | 54 (54) |
| Educational level (N = 71) | |
| Uneducated or incomplete elementary school | 59 (83) |
| Elementary school or incomplete high school | 11 (16) |
| High school or complete/incomplete higher education | 1 (1) |
| Main occupation (N = 93) | |
| Farmer | 62 (66.5) |
| Retired | 12 (13) |
| Others | 19 (20.5) |
| Smoker (N = 81) | |
| No | 38 (47) |
| Yes | 43 (53) |
| Alcoholic (N = 54) | |
| No | 25 (46) |
| Yes | 29 (54) |
| Phimosis (N = 77) | |
| No | 25 (32.5) |
| Yes | 52 (67.5) |
| Zoophilia (N = 50) | |
| No | 21 (42) |
| Yes | 29 (58) |
| Genital hygiene (N = 58) | |
| Good hygiene | 16 (27.5) |
| Poor/moderated hygiene | 42 (72.5) |
| Number of sexual partners (N = 47) | |
| <6 partners | 14 (30) |
| 6–10 partners | 8 (17) |
| >10 partners | 25 (53) |
| Beginning of symptoms (N = 64) | |
| 0 to 12 months | 40 (62.5) |
| >12 months | 24 (37.5) |
Histopathological features
The primary surgery performed was partial amputation of the penis (73%), with lesions predominantly located on the glans (98%). All tumors were classified as squamous cell carcinoma with prevalence of the usual subtype (35.3%). When analyzing the subtypes according to HPV-associated or non-HPV-associated etiology, there was a prevalence of HPV-associated tumors (65%), especially warty, basaloid, warty-basaloid, and mixed. Most tumors were high-grade (G3 (57%)); pT3/pT4 (61%); and stage II (76%)). Approximately 44 patients underwent inguinal lymphadenectomy, of whom 28 (63.5%) had lymph node metastasis confirmed in the histopathological diagnosis. The presence of changes suggestive of HPV infection (i.e., koilocytosis) was observed in 87% of cases. A summary of the histopathological data is presented in Table 2.
Table 2: Clinical histopathological features of 100 patients with penile cancer
| Variables | N (%) |
|---|---|
| Topography | |
| Restricted to foreskin/balanopreputial groove | 2 (2) |
| Glans and foreskin/Glans and groove | 67 (67) |
| Glans and body/Body | 31 (31) |
| Size of lesion (N = 99) | |
| 0.6–2.0 cm | 7 (7.1) |
| 2.1–5.0 cm | 55 (55.5) |
| 5.1–10 cm | 37 (37.4) |
| Histological subtype (N = 99) | |
| Basaloid | 6 (6.1) |
| Warty | 29 (29.3) |
| Usual | 35 (35.3) |
| Wart-basaloid | 8 (8.1) |
| Mixed | 19 (19.2) |
| Others* (medullary and pseudo-hyperplastic) | 2 (2) |
| Differentiation grade | |
| G1 | 13 (13) |
| G2 | 30 (30) |
| G3 | 57 (57) |
| Perineural invasion | |
| No | 62 (62) |
| Yes | 38 (38) |
| Corpus cavernosum (N = 99) | |
| Free | 41 (41.4) |
| Compromised | 58 (58.6) |
| Corpus spongiosum | |
| Free | 16 (16) |
| Compromised | 84 (84) |
| Urethra | |
| Free | 25 (25) |
| Compromised by contiguity | 15 (15) |
| Compromised | 59 (59) |
| Tumoral focus | |
| Unifocal | 85 (85) |
| Multifocal | 15 (15) |
| Presence of carcinoma in situ | |
| No | 17 (17) |
| Yes | 83 (83) |
| Sarcomatoid transformation | |
| No | 79 (79) |
| Yes | 21 (21) |
| Lymphocytic infiltrate (N = 99) | |
| Absent | 6 (6.1) |
| Present | 93 (93.9) |
| Primary tumor | |
| pT1 | 14 (14) |
| pT2 | 25 (25) |
| pT3/pT4 | 61 (61) |
| Staging | |
| I | 13 (13) |
| II | 76 (76) |
| III–IV | 11 (11) |
| Surgery type | |
| Partial penectomy | 73 (73) |
| Total penectomy | 27 (27) |
| Lymph node metastasis (N = 44) | |
| Absent | 16 (36.4) |
| Present | 28 (63.6) |
| Extra node extension (N = 44) | |
| Absent | 20 (45.5) |
| Present | 24 (54.5) |
| Koilocytosis | |
| Absent | 13 (13) |
| Present | 87 (87) |
| Invasion pattern (N = 97) | |
| Expansive | 76 (78.5) |
| Infiltrative | 21 (21.5) |
| HPV DNA (N = 97) | |
| HPV-Positive | 63 (65) |
| HPV-Negative | 34 (35) |
Cyclin D1 expression
Analysis of the cyclin D1 expression profile revealed that most cases exhibited weak or no immunostaining (56%), which was considered to be negative for cyclin D1 expression (Figure 1). Statistical analysis revealed that the absence of cyclin D1 expression was associated with tumors with HPV-related histological etiology (p = 0.001), while its expression was associated with high-grade tumors (G3, p = 0.0014), sarcomatoid transformation (p = 0.04), perineural invasion (p = 0.023), and histological subtype (p = 0.001). Data regarding associations between cyclin D1 expression profile and clinical and histopathological features are presented in Table 3.
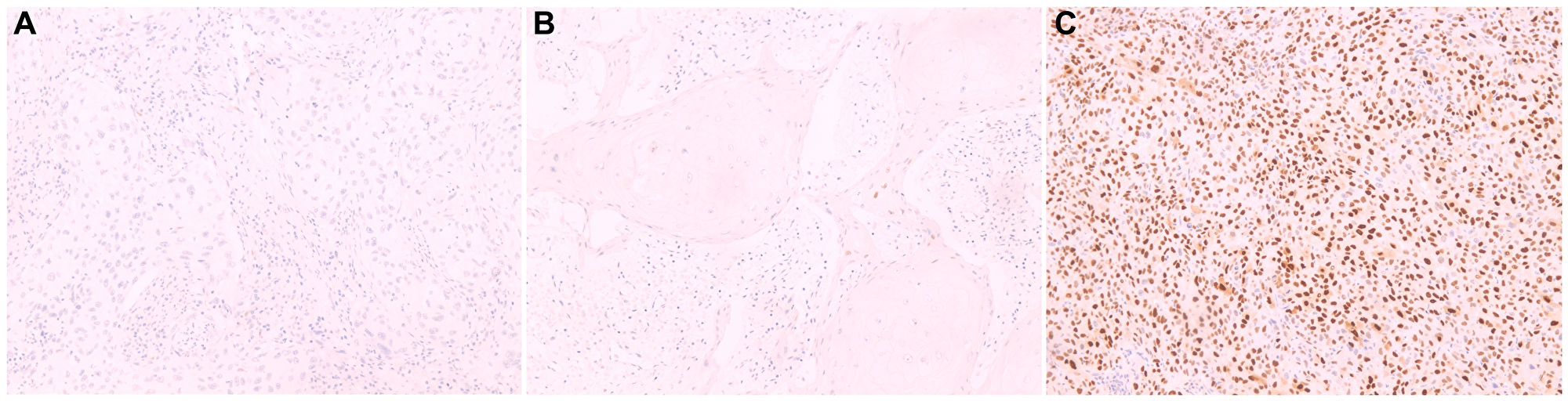
Figure 1: Cyclin D1 protein expression by immunohistochemistry in histological sections.
(A) Negative penile squamous cell carcinoma, showing absence of staining in the basal layer cells; (B) Negative penile squamous cell carcinoma showing weak staining in basal layer cells; (C) Positive penile squamous cell carcinoma, with staining in the suprabasal layers. Magnification 100x (A-O).
Table 3: Association of cyclin D1 regarding clinical-histopathological features
| Variables | Cyclin D1 | p-value* | |
|---|---|---|---|
| Negative (−) | Positive (+) | ||
| Phimosis (N = 77) | |||
| Absent | 11 | 14 | 0.6 |
| Present | 20 | 32 | |
| Surgery type | |||
| Glansectomy/partial penectomy | 29 | 44 | 0.17 |
| Total penectomy | 15 | 12 | |
| Size of lesion (N = 99) | |||
| 0.6–5.0 cm | 25 | 37 | 0.53 |
| 5.1–10 cm | 18 | 19 | |
| HPV-associated histological subtype (N = 81) | |||
| Non-associated | 4 | 33 | 0.001 |
| Associated | 23 | 21 | |
| Differentiation grade | |||
| G1/G2 | 18 | 25 | 0.014 |
| G3 | 11 | 46 | |
| Perineural invasion | |||
| No | 23 | 39 | 0.023 |
| Yes | 6 | 32 | |
| Tumoral focus | |||
| Unifocal | 35 | 50 | 0.25 |
| Multifocal | 9 | 6 | |
| Presence of carcinoma in situ | |||
| No | 6 | 11 | 0.59 |
| Yes | 38 | 45 | |
| Sarcomatoid transformation | |||
| No | 39 | 40 | 0.04 |
| Yes | 5 | 16 | |
| Lymph node infiltrate (N = 99) | |||
| Absent | 4 | 2 | 0.39 |
| Present | 39 | 54 | |
| Primary tumor | |||
| pT1 and pT2 | 18 | 21 | 0.83 |
| pT3 and pT4 | 26 | 35 | |
| Staging | |||
| I–II | 38 | 51 | 0.52 |
| III–IV | 6 | 5 | |
| Lymph node metastasis (N = 44) | |||
| Absent | 6 | 10 | 1 |
| Present | 11 | 17 | |
| Extra node extension (N = 43) | |||
| Absent | 7 | 10 | 0.76 |
| Present | 12 | 14 | |
| Koilocytosis | |||
| Absent | 1 | 12 | 0.10 |
| Present | 28 | 59 | |
| Invasion pattern (N = 97) | |||
| Expansive | 23 | 53 | 0.14 |
| Infiltrative | 3 | 18 | |
| Histological subtype (N = 98) | |||
| Basaloid | 5 | 1 | 0.001 |
| Warty | 14 | 15 | |
| Usual | 3 | 32 | |
| Warty-basaloid | 4 | 5 | |
| Mixed | 2 | 17 | |
| Others | 1 | 1 | |
Disease-free survival analysis
The data revealed that patients with PC and positive expression of cyclin D1 had a shorter disease-free survival (28.5 months) compared to those who did not express cyclin D1 (67.6 months), although the difference was not statistically significant (log-rank p = 0.669) (Figure 2).
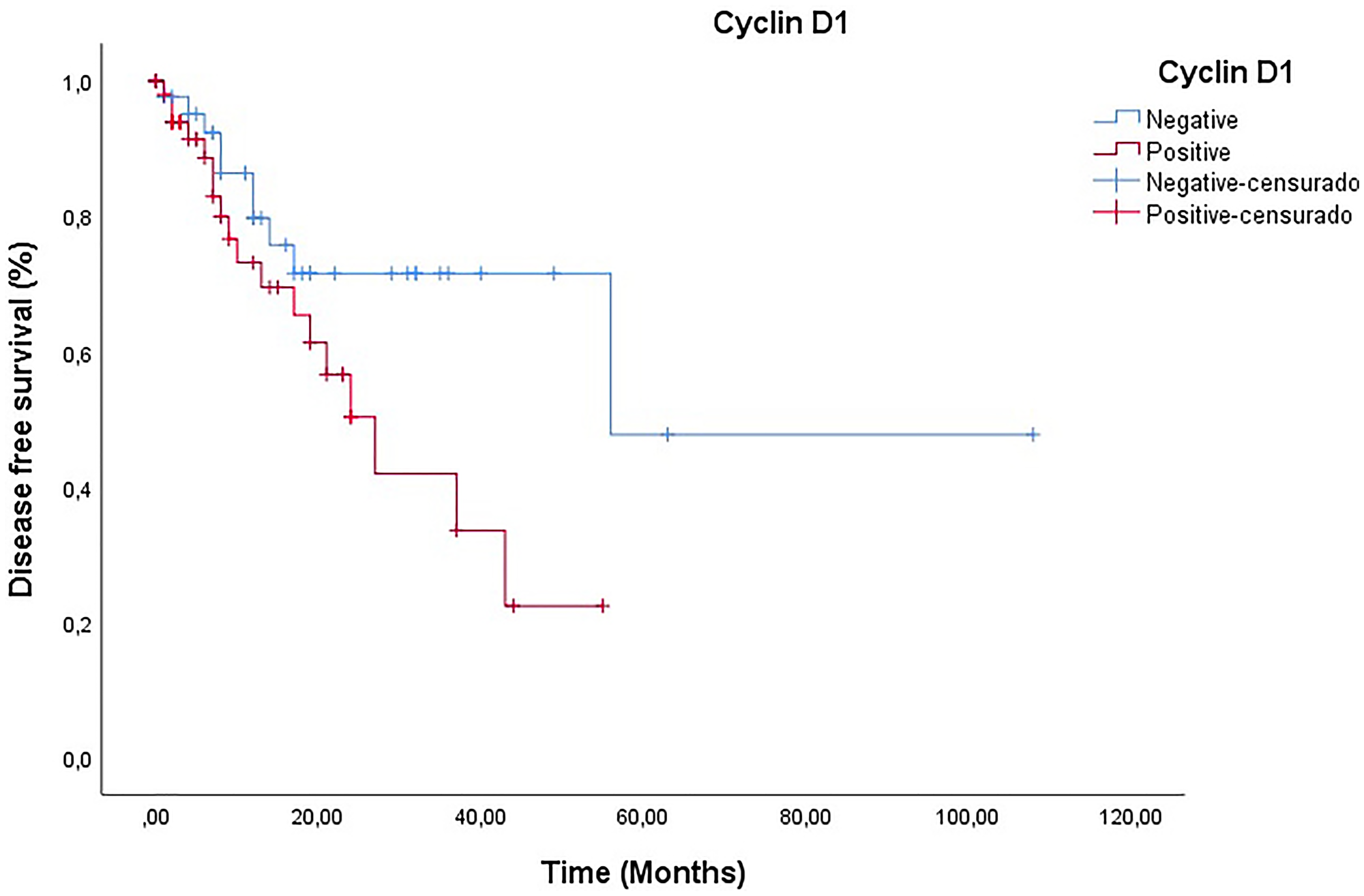
Figure 2: Analysis of disease-free survival of patients according to the presence and absence of cyclin D1 (Log-Rank p = 0.669)
DISCUSSION
The state of Maranhão, Brazil, registers the highest global incidence of PC, and has the worst Human Development Index (0.639) [1, 2, 4, 15]. In our sample, there was a predominance of patients younger than their seventh decade of life, with low educational level, poor hygienic conditions, and phimosis, as well as smokers and those who engaged in zoophilia. These data reinforce the relationship between PC and a low socioeconomic profile. Due to the advanced stage of disease in virtually all cases, the treatment was penile amputation, which has physical, social, and psychological implications for patients. As such, PC is considered to be a public health problem in many underdeveloped countries.
Therefore, one of the current targets of scientific research has been biomarkers that provide the possibility of aiding prognosis and therapy, while also being capable of identifying the main molecular agents involved in the tumorigenic process. Studies have shown that high expression of cyclin D1 in head and neck, colorectal, esophageal and breast tumors have been considered to be a characteristic of poorer prognosis, thus becoming a potential biomarker [11, 16, 17]. In our study, the expression of this protein was significantly associated with the following clinical features: histological subtype associated with HPV; degree of differentiation; sarcomatoid transformation; perineural invasion; and pattern of infiltrative invasion.
The absence of cyclin D1 expression was associated with the HPV-positive histological subtype. Corroborating our results, in head and neck, oropharyngeal and cervical cancers associated with HPV, cyclin D1 was down-regulated [11, 16, 18]. It has been well described that in HPV-associated tumors, the HPV oncoprotein E7 is responsible for the increase in p16 expression levels. It is also known that p16 is a tumor suppressor protein that acts by inhibiting the cyclin D1/CDK4 complex, favoring the survival of tumor cells [19]. Thus, overexpression of p16 leads to the absence of cyclin D1 expression and, consequently, of the p16INK4a/cyclinD/Rb pathway [20].
Cyclin D1 expression was more frequent in tumors with an infiltrative invasion pattern. This type of tumor is characterized by its rapid and disordered growth, with dissemination to nearby structures, presenting a greater risk for nodal involvement [21]. In basal cell and basosquamous carcinomas, cyclin D1 positivity was identified as a marker of aggressive behavior [22]. Furthermore, in prostate cancer, the expression of cyclin D1 appears to increase the capacity for tumor invasion, reinforcing the involvement of this protein in the more aggressive behavior of some tumors [12, 23].
Cyclin D1 expression was also associated with histological grade G3 and sarcomatoid transformation. Tumors with sarcomatoid areas must always be considered as G3, regardless of the ratio. Poorly differentiated tumors are characterized by greater aggressiveness, indicating faster growth compared to those that are well differentiated [24]. These data corroborate the findings in esophageal and breast cancer, in which the expression of cyclin D1 was associated with poorly differentiated tumors [10, 25]. Tumors with sarcomatoid alterations are more prone to lymph node metastasis, thus portending a poorer prognosis.
The presence of perineural invasion is associated with cyclin D1 expression. Studies have reported that the presence of this protein in cases of perineural invasion is considered to be a characteristic of poorer clinical prognosis because this condition is characterized by increased local recurrence and regional metastasis [26, 27]. It is believed that perineural invasion is factor-mediated by growth factors and that their receptors interact with cyclin D1.
In breast and oropharyngeal tumors, the positive expression of cyclin D1 was associated with lower disease-free survival [10, 28]. Thus, our results corroborate those described in the literature for cancer in general, in which patients who exhibited expression of this protein exhibited worse disease-free survival [27, 29]. The increased expression of this protein may be related to dysregulation in its degradation because this process is important for DNA replication.
To our knowledge, there are only two studies that have investigated the role of cyclin D1 in the genesis and behavior of PC. The first, by Papadopoulos et al. [30], reported the overexpression of cyclin D1 in 61.9% of assessed cases of PC, being more frequent in poorly differentiated tumors with a high proliferative index, although without statistical significance. The second, by Gunia et al. [14], evaluated the expression of cyclin D1 in 110 penile tumors. This study also found no association between the expression profile and the parameters studied. The results reported in these two studies are divergent from ours, as cyclin D1 appeared to demonstrate prognostic relevance. However, we emphasize the need for further studies to validate this protein as a biomarker in PC.
A limitation of our study was the lack of information in medical records, as well as the absence of follow-up for some patients. Furthermore, only 100 cases were selected due to the poor condition of the paraffin blocks, making it exceedingly difficult to perform immunohistochemical analysis with a larger sample. Nevertheless, due to the rarity of the disease, the number of cases in our study still makes a significant contribution to the understanding of this pathology.
Materials and Methods
Study design
The research was performed from 2013 to 2017, and included 100 patients with PC treated at two reference hospitals in São Luís, Maranhão, Brazil (University Hospital of the Federal University of Maranhão - HUUFMA; and Aldenora Bello Cancer Hospital - HCAB). Patients >18 years of age, who underwent surgery as the first treatment option and who provided informed written consent, approved by the Research Ethics Committee (CEP-HUUFMA), opinion No. 3.122.045 and CAAE No. 05918918.9.0000.5086, were included. Patients who refused to provide consent or who underwent radiotherapy or chemotherapy as primary therapy were excluded.
A survey of clinical and epidemiological data from patients with PC was performed through a review of medical records. Histological subtyping in all cases was reclassified according to the World Health Organization (2016), while the TNM system from the American Joint Committee on Cancer (AJCC), 8th Edition was used for staging. Histopathological parameters were also reviewed through re-analysis of hematoxylin and eosin (H&E) slides, which was performed independently by two pathologists (GEBS and SPCM). In cases of disagreement, the definition was established after consensus among pathologists. After reviewing the H&E slides, a formalin-fixed paraffin-embedded (FFPE) block containing at least 70% of the tumor tissue was selected for determining protein expression using immunohistochemical (IHC) methods and for polymerase chain reaction (PCR) analysis.
HPV histological identification
Histological diagnosis of HPV was performed by identifying koilocytosis, in accordance with three main criteria described by Abadi et al. (1998), more specifically: the presence of a perinuclear halo; nuclear atypia; and presence of multinucleation.
HPV molecular analysis
The presence or absence of HPV DNA in PC samples was performed according to a protocol described by Sambrook et al. (1989). Subsequently, nested PCR was performed in two stages. In the first, a set of generic primers, PGMY09/11, described by Gravitt et al. (2000), were used. These primers produce a 450 base pair (bp) fragment of the HPV L1 region. In the second, the primer GP5+/6+ was used, generating a 170 bp amplicon, which corresponds to the L1 region of the viral capsid. The amplicons were applied to a 1.5% agarose gel, subjected to a constant voltage of 90 V for 40 min, and visualized.
Cyclin D1 protein expression according to IHC analysis
For IHC analysis, a rabbit monoclonal antibody anti-human-cyclin D1 (Clone EP12, Dako), purchased from Agilent (Santa Clara, CA, USA) was used. A 3-μm thick section of FFPE block was used for each case, and placed on IHC-specific slides using a pre-warmed water bath at 50°C. The slides were then heated at 70°C for 30 min for drying and removing excess paraffin tissue. Briefly, for antigen retrieval, the slides were immersed in a high pH solution (Agilent) at a ratio of 1:50 with distilled water and subjected to temperature cycling in a rinse station (PT Link PT200, Dako, Agilent Technologies), according to manufacturer’s recommendations. After antigen retrieval, endogenous peroxidase activity was blocked, and monoclonal antibody (ready to use) was applied, followed by polymer application. Diaminobenzidine (i.e., “DAB”) was then applied for 10 min to develop immunostaining, followed by counter staining with hematoxylin for 5 min. Finally, the slides were analyzed under an optical microscope.
Protein expression analysis was performed semi-quantitatively in accordance with criteria proposed by Gunia et al., (2011), which characterizes cyclin D1 expression as negative when there is an absence or weak expression of this protein only in basal cells, and positive when expressed in both basal and suprabasal cells.
Data analysis
For statistical analysis, the chi-squared test was used to compare clinical/histopathological data for expression of cyclin D1. Survival analysis was performed using the Kaplan–Meier method to determine/estimate disease-free survival according to cyclin D1 expression, and the log-rank test was used to compare the survival curves. All analyses were performed using SPSS version 26.0 (IBM Corporation, Armonk, NY, USA). Differences with p ≤ 0.05 were considered to be statistically significant.
Conclusions
Our data demonstrated that cyclin D1 is a potential biomarker for PC and is associated with poorer prognosis, although no association was found with lymph node metastasis and disease-free survival. However, due to the limited number of studies in the literature, these data reinforce the importance of performing more studies investigating the actual role of cyclin D1 in PC.
Abbreviations
PC: Penile cancer; HPV: human papillomavirus; pRb: retinoblastoma protein; HUUFMA: University Hospital of the Federal University of Maranhão; HCAB: Aldenora Bello Cancer Hospital; HE: hematoxylin and eosin; FFPE: formalin-fixed paraffin-embedded; IHC: Immunohistochemistry.
ACKNOWLEDGMENTS
The authors are grateful for the collaboration of the Hospital do Câncer Aldenora Bello (HCAB) and University Hospital of the Federal University of Maranhão (HUUFMA) for the authorization to carry out this research. We also thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES), for supporting the training. We also thank the collaboration of Oncology Research Center, João de Barros Barreto University Hospital, Federal University of Pará, Belém, Brazil and PROPESP/UFPA.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
ETHICAL STATEMENT
This study was approved by the Research Ethics Committee (CEP-HUUFMA), opinion No. 3.122.045 and CAAE No. 05918918.9.0000.5086.
CONSENT
Patients provided informed written consent.
FUNDING
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001, and for Fundação de Amparo à Pesquisa e ao Desenvolvimento Científico e Tecnológico do Maranhão (FAPEMA) – Award Numbers 01339/17 and 713 01184/19.
References
1. Vieira CB, Feitoza L, Pinho J, Teixeira-Júnior A, Lages J, Calixto J, Coelho R, Nogueira L, Cunha I, Soares F, Silva GEB. Profile of patients with penile cancer in the region with the highest worldwide incidence. Sci Rep. 2020; 10:2965. https://doi.org/10.1038/s41598-020-59831-5. [PubMed].
2. Coelho RWP, Pinho JD, Moreno JS, Garbis DVE, do Nascimento AMT, Larges JS, Calixto JRR, Ramalho LNZ, da Silva AAM, Nogueira LR, de Moura Feitoza L, Silva GEB. Penile cancer in Maranhão, Northeast Brazil: the highest incidence globally? BMC Urol. 2018; 18:50. https://doi.org/10.1186/s12894-018-0365-0. [PubMed].
3. Douglawi A, Masterson TA. Penile cancer epidemiology and risk factors: a contemporary review. Curr Opin Urol. 2019; 29:145–49. https://doi.org/10.1097/MOU.0000000000000581. [PubMed].
4. Teixeira Júnior AAL, da Costa Melo SP, Pinho JD, Sobrinho TBM, Rocha TMS, Duarte DRD, de Oliveira Barbosa L, Duarte WE, de Castro Belfort MR, Duarte KG, da Silva Neto AL, de Ribamar Rodrigues Calixto J, Paiva Paiva LC, et al. A comprehensive analysis of penile cancer in the region with the highest worldwide incidence reveals new insights into the disease. BMC Cancer. 2022; 22:1063. https://doi.org/10.1186/s12885-022-10127-z. [PubMed].
5. Castle PE, Dockter J, Giachetti C, Garcia FA, McCormick MK, Mitchell AL, Holladay EB, Kolk DP. A cross-sectional study of a prototype carcinogenic human papillomavirus E6/E7 messenger RNA assay for detection of cervical precancer and cancer. Clin Cancer Res. 2007; 13:2599–605. https://doi.org/10.1158/1078-0432.CCR-06-2881. [PubMed].
6. Singh S, Johnson J, Chellappan S. Small molecule regulators of Rb-E2F pathway as modulators of transcription. Biochim Biophys Acta. 2010; 1799:788–94. https://doi.org/10.1016/j.bbagrm.2010.07.004. [PubMed].
7. Beasley MB, Lantuejoul S, Abbondanzo S, Chu WS, Hasleton PS, Travis WD, Brambilla E. The P16/cyclin D1/Rb pathway in neuroendocrine tumors of the lung. Hum Pathol. 2003; 34:136–42. https://doi.org/10.1053/hupa.2003.8. [PubMed].
8. Wang Q, He G, Hou M, Chen L, Chen S, Xu A, Fu Y. Cell Cycle Regulation by Alternative Polyadenylation of CCND1. Sci Rep. 2018; 8:6824. https://doi.org/10.1038/s41598-018-25141-0. [PubMed].
9. Otto T, Sicinski P. Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer. 2017; 17:93–115. https://doi.org/10.1038/nrc.2016.138. [PubMed].
10. Ortiz AB, Garcia D, Vicente Y, Palka M, Bellas C, Martin P. Prognostic significance of cyclin D1 protein expression and gene amplification in invasive breast carcinoma. PLoS One. 2017; 12:e0188068. https://doi.org/10.1371/journal.pone.0188068. [PubMed].
11. Novotný J, Bandúrová V, Strnad H, Chovanec M, Hradilová M, Šáchová J, Šteffl M, Grušanović J, Kodet R, Pačes V, Lacina L, Smetana K Jr, Plzák J, et al. Analysis of HPV-Positive and HPV-Negative Head and Neck Squamous Cell Carcinomas and Paired Normal Mucosae Reveals Cyclin D1 Deregulation and Compensatory Effect of Cyclin D2. Cancers (Basel). 2020; 12:792. https://doi.org/10.3390/cancers12040792. [PubMed].
12. Pereira RA, Ravinal RC, Costa RS, Lima MS, Tucci S, Muglia VF, Reis RB, Silva GE. Cyclin D1 expression in prostate carcinoma. Braz J Med Biol Res. 2014; 47:515–21. https://doi.org/10.1590/1414-431x20143240. [PubMed].
13. Lima MS, Pereira RA, Costa RS, Tucci S, Dantas M, Muglia VF, Ravinal RC, Barros-Silva GE. The prognostic value of cyclin D1 in renal cell carcinoma. Int Urol Nephrol. 2014; 46:905–13. https://doi.org/10.1007/s11255-013-0602-0. [PubMed].
14. Gunia S, Kakies C, Erbersdobler A, Hakenberg OW, Koch S, May M. Expression of p53, p21 and cyclin D1 in penile cancer: p53 predicts poor prognosis. J Clin Pathol. 2012; 65:232–36. https://doi.org/10.1136/jclinpath-2011-200429. [PubMed].
15. Instituto Brasileiro de Geografia e Estatística (IBGE). Censo demográfico: resultados preliminares - Maranhão. 8º Recenseamento Geral do Brasil; v. 1, t. 4. 2010. https://censo2010.ibge.gov.br/resultados.html.
16. Hou X, Liang RB, Wei JC, Xu Y, Fu JH, Luo RZ, He JH, Zhang LJ, Lin P, Yang HX. Cyclin D1 expression predicts postoperative distant metastasis and survival in resectable esophageal squamous cell carcinoma. Oncotarget. 2016; 7:31088–96. https://doi.org/10.18632/oncotarget.9078. [PubMed].
17. Jun SY, Kim J, Yoon N, Maeng LS, Byun JH. Prognostic Potential of Cyclin D1 Expression in Colorectal Cancer. J Clin Med. 2023; 12:572. https://doi.org/10.3390/jcm12020572. [PubMed].
18. Tripathi R, Rath G, Jawanjal P, Bharadwaj M, Mehrotra R. ≤ Cyclin D1 protein affecting global women’s health by regulating HPV mediated adenocarcinoma of the uterine cervix. Sci Rep. 2019; 9:5019. https://doi.org/10.1038/s41598-019-41394-9. [PubMed].
19. McLaughlin-Drubin ME, Park D, Munger K. Tumor suppressor p16INK4A is necessary for survival of cervical carcinoma cell lines. Proc Natl Acad Sci U S A. 2013; 110:16175–80. https://doi.org/10.1073/pnas.1310432110. [PubMed].
20. Knopp S, Bjørge T, Nesland JM, Tropé C, Holm R. Cyclins D1, D3, E, and A in vulvar carcinoma patients. Gynecol Oncol. 2005; 97:733–39. https://doi.org/10.1016/j.ygyno.2005.03.025. [PubMed].
21. Guimarães GC, Lopes A, Campos RS, Zequi Sde C, Leal ML, Carvalho AL, da Cunha IW, Soares FA. Front pattern of invasion in squamous cell carcinoma of the penis: new prognostic factor for predicting risk of lymph node metastases. Urology. 2006; 68:148–53. https://doi.org/10.1016/j.urology.2006.01.050. [PubMed].
22. N Sivrikoz O, Kandiloğlu G. The Effects of Cyclin D1 and Bcl-2 Expressıon on Aggressive Behavior in Basal Cell and Basosquamous Carcinoma. Iran J Pathol. 2015; 10:185–91. [PubMed].
23. Ding GX, Liu J, Feng CC, Jiang HW, Xu JF, Ding Q. Slug regulates Cyclin D1 expression by ubiquitin-proteasome pathway in prostate cancer cells. Panminerva Med. 2012; 54:219–23. [PubMed].
24. Velazquez EF, Ayala G, Liu H, Chaux A, Zanotti M, Torres J, Cho SI, Barreto JE, Soares F, Cubilla AL. Histologic grade and perineural invasion are more important than tumor thickness as predictor of nodal metastasis in penile squamous cell carcinoma invading 5 to 10 mm. Am J Surg Pathol. 2008; 32:974–79. https://doi.org/10.1097/PAS.0b013e3181641365. [PubMed].
25. Dey B, Raphael V, Khonglah Y, GiriLynrah K. Expression of Cyclin D1 and P16 in Esophageal Squamous Cell Carcinoma. Middle East J Dig Dis. 2015; 7:220–25. [PubMed].
26. Al-Rawi NH, Kawas SA, Ani MA, Alnuaimi AS, El-Sayed W, Alrashdan MS. Prediction of Lymphovascular and Perineural Invasion of Oral Squamous Cell Carcinoma by Combined Expression of p63 and Cyclin D1. Eur J Dent. 2023; 17:1170–78. https://doi.org/10.1055/s-0042-1760301. [PubMed].
27. Kornmann M, Danenberg KD, Arber N, Beger HG, Danenberg PV, Korc M. Inhibition of cyclin D1 expression in human pancreatic cancer cells is associated with increased chemosensitivity and decreased expression of multiple chemoresistance genes. Cancer Res. 1999; 59:3505–11. [PubMed].
28. Kumar B, Yadav A, Brown NV, Zhao S, Cipolla MJ, Wakely PE, Schmitt AC, Baiocchi RA, Teknos TN, Old M, Kumar P. Nuclear PRMT5, cyclin D1 and IL-6 are associated with poor outcome in oropharyngeal squamous cell carcinoma patients and is inversely associated with p16-status. Oncotarget. 2017; 8:14847–59. https://doi.org/10.18632/oncotarget.14682. [PubMed].
29. Chi Y, Huang S, Liu M, Guo L, Shen X, Wu J. Cyclin D3 predicts disease-free survival in breast cancer. Cancer Cell Int. 2015; 15:89. https://doi.org/10.1186/s12935-015-0245-6. [PubMed].
30. Papadopoulos O, Betsi E, Tsakistou G, Frangoulis M, Kouvatseas G, Anagnostakis D, Kouvidou Ch. Expression of cyclin D1 and Ki-67 in squamous cell carcinoma of the penis. Anticancer Res. 2007; 27:2167–74. [PubMed].

