Introduction
Osimertinib is a highly effective third generation epidermal growth factor receptor tyrosine kinase inhibitor (EGFR TKI) that is the current first line standard of care for patients with metastatic non-small cell lung cancer (NSCLC) with EGFR activating mutations. Importantly, osimertinib provides anti-tumor efficacy for patients with central nervous system (CNS) involvement, either intracranial metastases or leptomeningeal disease (LMD) [1].
Clinically, the early identification of LMD is difficult, and patients often present with diffuse, clinically debilitating LMD. Furthermore, isolation of sufficient tissue for molecular analysis in patients with LMD is a barrier to allocating patients to optimal therapy. Herein, we present the case of a patient who presented with life threatening LMD with insufficient tissue for molecular analysis, who was urgently started on empiric osimertinib with successful disease improvement for over 18 months to date on osimertinib monotherapy.
CASE REPORT
A 64-year-old woman with no smoking history and of East Asian descent presented to the emergency department with four months of headaches, one month of right eye vision loss, right lower extremity weakness, and syncope. Upon admission, she was also noted to have a spontaneous pneumothorax with an 18 mm spiculated lesion in her left upper lobe (LUL).
A non-contrast MRI of the brain and spine showed no intracranial abnormalities and no LMD was noted. Cerebrospinal fluid (CSF) cytology raised concern for malignancy with large, atypical cells. Bronchoscopy with biopsy of the LUL lesion revealed NSCLC (adenocarcinoma), and carcinomatous meningitis from NSCLC was suspected given the CSF findings. The lung tissue and CSF cytology were insufficient for molecular analysis. Peripheral blood circulating tumor DNA using a Tempus xF® assay was assessed, and there were no pathogenic mutations identified.
Because of the high pre-test probability of this cancer being driven by an EGFR activating mutation and the patient’s ongoing clinical decline from carcinomatous meningitis, osimertinib 80 mg daily was empirically started. Within two days headaches improved, and within 2 weeks she had a dramatic improvement in her functional status. Imaging after approximately one month on therapy showed slight improvement in her lung nodule (Figure 1). Repeat bronchoscopy with biopsy of the LUL lesion was completed and Tempus xT® molecular assessment revealed an EGFR L858R activating mutation.
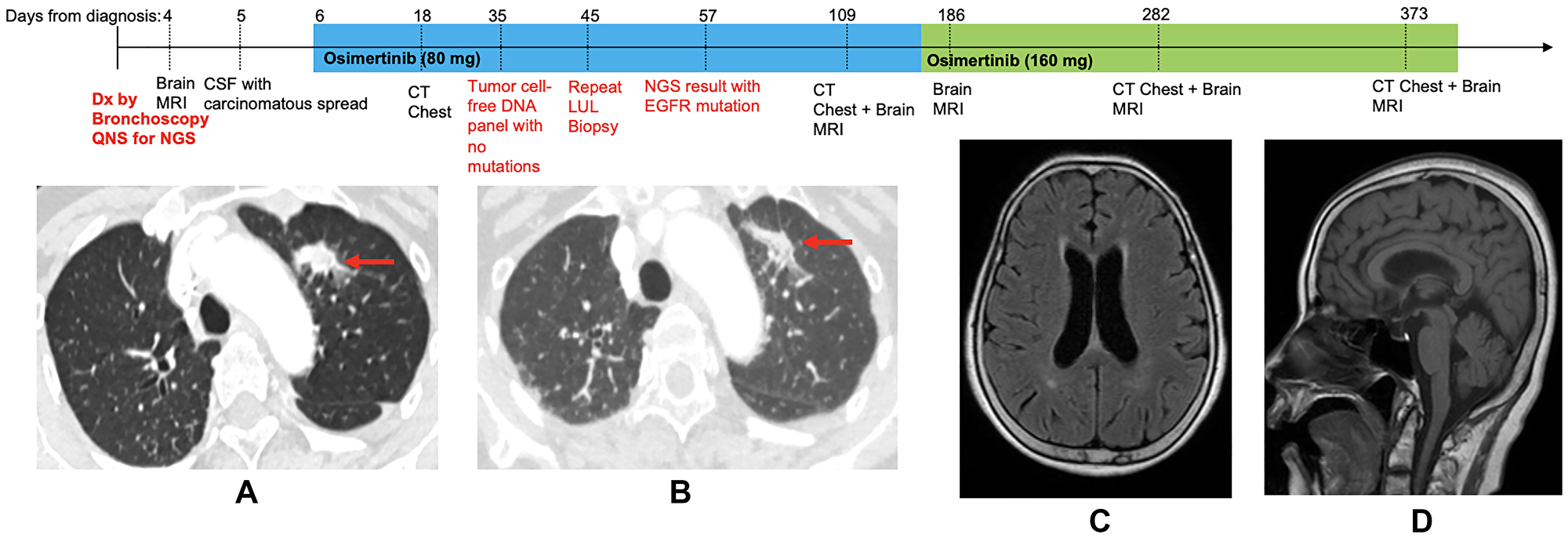
Figure 1: Treatment timeline with chest computed tomography (CT) and brain magnetic resonance images (MRI) on osimertinib.
(A) Pretreatment CT chest, demonstrating 18 mm spiculated mass in the left upper lobe (LUL). (B) CT chest following 12 days of Osimertinib treatment, showing decreased size of LUL mass. (C) Pretreatment brain MRI showing no acute abnormalities (axial). (D) Pretreatment brain MRI showing no acute abnormalities (sagittal).
The patient required escalated dosing of osimertinib to 160 mg daily for progressive cranial nerve deficits from LMD six months into therapy, and she has since continued osimertinib at 160 mg daily for a total of 18 months to date with ongoing excellent disease control in the lung and CNS.
DISCUSSION
Leptomeningeal disease is characterized by CNS metastases with spread to the CSF or leptomeninges. In patients with NSCLC with EGFR activating mutations, there is an increased occurrence of LMD [2], which occurs in 9% of patients [3].
Osimertinib is an EGFR TKI that has been shown to be safe and efficacious for patients with metastatic EGFR mutant NSCLC complicated by LMD [1]. Notably in the BLOOM trial, patients who had previously progressed on other TKI therapies showed clinical improvement in neurological function on osimertinib 160 mg daily [1]. Current data on the use of osimertinib supports its application in previously treated or untreated patients with metastatic EGFR mutant NSCLC with LMD.
Our case demonstrates the nuances of decision-making in starting osimertinib in urgent clinical settings. Given our patient’s progressively worsening functional status and spread of disease to her CNS upon presentation, there was a need to begin treatment imminently. Time constraints, financial constraints, and lack of sufficient tissue for analysis ultimately led to the empiric use of osimertinib. Through the urgent initiation of appropriate anti-cancer therapy, she experienced both a life saving improvement in functional status and improvement in her LUL primary tumor one month into treatment.
Conclusion
Overall, this case supports a body of literature noting potentially dramatic clinical benefits of administering appropriate oncogene directed targeted therapy for critically ill patients with NSCLC (Table 1), and in this specific case it is noted that epidemiology can help guide empiric treatment in patients with prohibitive challenges to adequate and timely molecular assessment, an approach that has also been report on two previous occasions (Table 2).
Table 1: Lazarus responses with targeted therapies in non-small cell lung cancer
| Study | Oncogenic mutation | Clinical status | Treatment | Disease response |
|---|---|---|---|---|
| Ninomaru 2021 [4] (Kobe, Japan) | MET Exon 14 | Leptomeningeal disease | Tepotinib | ECOG PS 3 to 1 PFS 5 months |
| Facchinetti 2021 [5] (Villejuif, France) | ROS1 | Leptomeningeal disease | Lorlatinib | PFS 9 months |
| Beninato 2020 [6] (Milan, Italy) | ROS1 | Respiratory failure | Crizotinib | PFS >2 months |
| Ahn 2013 [7] (Seoul, South Korea) | ALK | Respiratory failure | Crizotinib | PFS 3 months (2 patients) PFS 8 months (1 patient) |
| Kerrigan 2016 [8] (Columbus, USA) | ROS1 | Respiratory failure | Crizotinib | PFS 16 months |
| EGFR | Respiratory failure | Erlotinib | PFS 8 months | |
| Adam 2015 [9] (Leuven, Belgium) | ALK | Respiratory failure | Ceritinib | PFS >1 year |
| Van Geffen 2013 [10] (Groningen, The Netherlands) | ALK | Bilateral LE paresis and blindness | Crizotinib | PFS 12 months |
Table 2: Empiric treatment with an EGFR tyrosine kinase inhibitor
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
Ethical statement
The highest ethical standards were maintained in protecting patient data and maintaining the integrity of all data presented in this manuscript. This case report was written with obtained consent from the patient’s next of kin, as the patient is deceased. No further ethical review was required for this case report.
FUNDING
No funding was used for this paper.
References
1. Yang JCH, Kim SW, Kim DW, Lee JS, Cho BC, Ahn JS, Lee DH, Kim TM, Goldman JW, Natale RB, Brown AP, Collins B, Chmielecki J, et al. Osimertinib in Patients With Epidermal Growth Factor Receptor Mutation-Positive Non-Small-Cell Lung Cancer and Leptomeningeal Metastases: The BLOOM Study. J Clin Oncol. 2020; 38:538–47. https://doi.org/10.1200/JCO.19.00457. [PubMed].
2. Li YS, Jiang BY, Yang JJ, Tu HY, Zhou Q, Guo WB, Yan HH, Wu YL. Leptomeningeal Metastases in Patients with NSCLC with EGFR Mutations. J Thorac Oncol. 2016; 11:1962–69. https://doi.org/10.1016/j.jtho.2016.06.029. [PubMed].
3. Hegde A, Velcheti V. Osimertinib for Leptomeningeal Disease in EGFR-Mutated NSCLC. J Thorac Oncol. 2020; 15:1705–8. https://doi.org/10.1016/j.jtho.2020.08.016. [PubMed].
4. Ninomaru T, Hata A, Kokan C, Okada H, Tomimatsu H, Ishida J. Higher osimertinib introduction rate achieved by multiple repeated rebiopsy after acquired resistance to first/second generation EGFR-TKIs. Thorac Cancer. 2021; 12:746–51. https://doi.org/10.1111/1759-7714.13822. [PubMed].
5. Facchinetti F, Di Maio M, Perrone F, Tiseo M. First-line immunotherapy in non-small cell lung cancer patients with poor performance status: a systematic review and meta-analysis. Transl Lung Cancer Res. 2021; 10:2917–36. https://doi.org/10.21037/tlcr-21-15. [PubMed].
6. Beninato T, Lo Russo G, Garassino MC, De Braud F, Platania M. Recurrent thrombosis followed by Lazarus response in ROS1 rearranged NSCLC treated with crizotinib: a case report. Tumori. 2020; 106:NP41–45. https://doi.org/10.1177/0300891620905665. [PubMed].
7. Ahn HK, Jeon K, Yoo H, Han B, Lee SJ, Park H, Lee MJ, Ha SY, Han JH, Sun JM, Ahn JS, Ahn MJ, Park K. Successful treatment with crizotinib in mechanically ventilated patients with ALK positive non-small-cell lung cancer. J Thorac Oncol. 2013; 8:250–53. https://doi.org/10.1097/JTO.0b013e3182746772. [PubMed].
8. Kerrigan K, Shoben A, Otterson G. Treatment of Lung Cancer Patients With Actionable Mutations in the Intensive Care Unit. Clin Lung Cancer. 2016; 17:523–27. https://doi.org/10.1016/j.cllc.2016.04.004. [PubMed].
9. Adam V, Dooms C, Vansteenkiste J. Lung cancer at the intensive care unit: The era of targeted therapy. Lung Cancer. 2015; 89:218–21. https://doi.org/10.1016/j.lungcan.2015.05.009. [PubMed].
10. van Geffen WH, Hiltermann TJ, Groen HJ. Surviving respiratory insufficiency with intensive care support in a pretreated, extensively metastasized patient with an EML4-ALK translocation. J Thorac Oncol. 2013; 8:e1–2. https://doi.org/10.1097/JTO.0b013e3182762812. [PubMed].
11. Bosch-Barrera J, Sais E, Lorencio C, Porta R, Izquierdo A, Menéndez JA, Brunet J, Sirvent JM, Rosell R. Successful empirical erlotinib treatment of a mechanically ventilated patient newly diagnosed with metastatic lung adenocarcinoma. Lung Cancer. 2014; 86:102–4. https://doi.org/10.1016/j.lungcan.2014.07.010. [PubMed].
12. Jeong S, Um SW, Lee H, Jeon K, Lee K, Suh G, Chung M, Kim H, Kwon O, Choi Y. Successful Treatment with Empirical Erlotinib in a Patient with Respiratory Failure Caused by Extensive Lung Adenocarcinoma. Korean J Crit Care Med. 2016; 31:44–48. https://doi.org/10.4266/kjccm.2016.31.1.44.

