Introduction
Multiple Myeloma (MM) is a malignant hematological neoplasm characterized by the uncontrolled proliferation of abnormal plasma cells within the bone marrow [1]. It represents the second most common hematological malignancy, accounting for a significant portion of cancer-related morbidity and 2% of cancer mortality worldwide [1–3]. Despite recent advances in the understanding and treatment of MM, it remains an incurable disease with a substantial impact on patient outcomes and healthcare systems [4].
MM is preceded by a premalignant condition known as monoclonal gammopathy of undetermined significance (MGUS) [5]. MGUS is characterized by the presence of abnormal monoclonal paraprotein in the blood without clinical signs of organ damage, and is often discovered incidentally when blood tests are done for other reasons [1]. MGUS has a prevalence of approximately 1–2% in the general population below 50 years of age, which increases with age reaching up to 3–8% in the population above 80 years [6–9]. While the majority of MGUS cases remain stable, approximately 1% of MGUS patients progress to symptomatic MM each year, presenting a significant clinical challenge in predicting and managing disease progression. The current approach that is used in the clinic for differentiating the stable and progressive myeloma precursor conditions is mainly based on surrogates’ measures of disease burden, such as bone marrow plasma cell percentage and quantity of serum monoclonal protein [10]. However, the utility of this approach is challenged by the diversity of MGUS patients and the fact that the behavior pattern of MGUS does not always correlate with the disease burden [10]. Therefore, development of a scalable, cost-effective approach for accurate risk stratification and identification of MGUS patients at high risk of developing MM could facilitate early detection of the disease progression, allowing for timely interventions and tailored therapeutic strategies before the onset of end-organ damage. This has the potential to revolutionize the clinical management of high-risk MGUS patients and significantly improve patient outcomes, enhance patients’ quality of life, and reduce the socioeconomic burden associated with MM.
Bone marrow adipose tissue (BMAT) is a major component of the bone marrow microenvironment [11–13]. Although traditionally perceived as a passive filler of the bone marrow cavity, BMAT has lately emerged as an active player with dynamic interactions that extend far beyond its previous characterization [12]. Bone marrow adipocytes (BMAds) not only coexist harmoniously alongside hematopoietic and stromal cell populations within the marrow, but also influence their behavior and function. BMAT has been recently implicated in various physiological and pathological processes [14], particularly in relation to hematological, endocrine, and skeletal disorders [15–23]. Obesity, a well-established risk factor for various types of cancers, including MM, is associated with increased bone marrow adiposity, together with altered hematopoiesis and immune regulation [24, 25]. Several studies have reported a correlation between increased bone marrow adiposity and increased risk of MGUS and progression to MM [21, 25]. These findings suggest that BMAds could play a role in the pathogenesis of MM. On the other hand, we have previously shown that BMAT is significantly decreased in non-treated MM patients compared to MGUS patients and healthy individuals [26], suggesting that BMAds could serve as a potential biomarker for MM progression. Therefore, in this study, we examined whether the composition of the BMAT is different in stable and progressing MGUS patients. We employed artificial intelligence (AI)-assisted histological analyses of unstained bone marrow biopsies and found that BMAd density, size, and roundness are significantly different between the two groups and could provide early signs for progression from MGUS to MM.
Results and discussion
We employed an AI-assisted approach to perform a systematic and objective histological characterization of the BMAT in progressing and non-progressing MGUS patients, with the aim of identifying possible BMAT features that were different between the two groups. To facilitate translation to high-throughput clinical screening, we utilized unstained bone marrow biopsies by taking advantage of the tissue’s autofluorescence in the FITC channel (Figure 1A–1C). We did not find a significant difference in the BMAT fraction within the bone marrow (% of total marrow) between the stable and progressing MGUS patients (Figure 2A). This suggests that the overall adipose tissue content in the bone marrow was comparable between the two groups. However, we found decreased BMAd density in MGUS patients who experienced progression to MM compared to the non-progressing MGUS patients (Figure 2B). To gain deeper insight into the characteristics of the BMAds associated with MM tumorigenesis, we performed a morphological characterization of the individual BMAds. Importantly, we found a significant shift towards increased BMAd size and roundness in progressing MGUS patients (Figure 2C, 2D). These observations suggest that during MM development, BMAds are subject to alterations which may be indicative of the disease progression. We and others have previously shown that BMAT is significantly reduced in non-treated MM patients [26, 27]. Lack of direct correlation between the bone marrow tumor burden and bone marrow adiposity in overt multiple myeloma, argues against the “space-constricted” mechanism causing decreased BMAT [26]. An alternative proposed mechanism is that cancer cells “hijack” the bone marrow metabolic programs to induce release of fatty acids from BMAds and fulfil their high metabolic demand [28]. It has recently been shown that induction of lipolysis and uptake of fatty acids by MM cells through fatty acid transporter proteins are involved in this process [28]. Therefore, it is possible that the shift towards increased BMAd size in progressing MGUS patients is an early sign that an altered metabolic program induced by MM cells has already started to take effect at this stage, leading to release of fatty acids from BMAds and disappearance of small BMAds, while exerting minor visual impact on the large adipocytes.
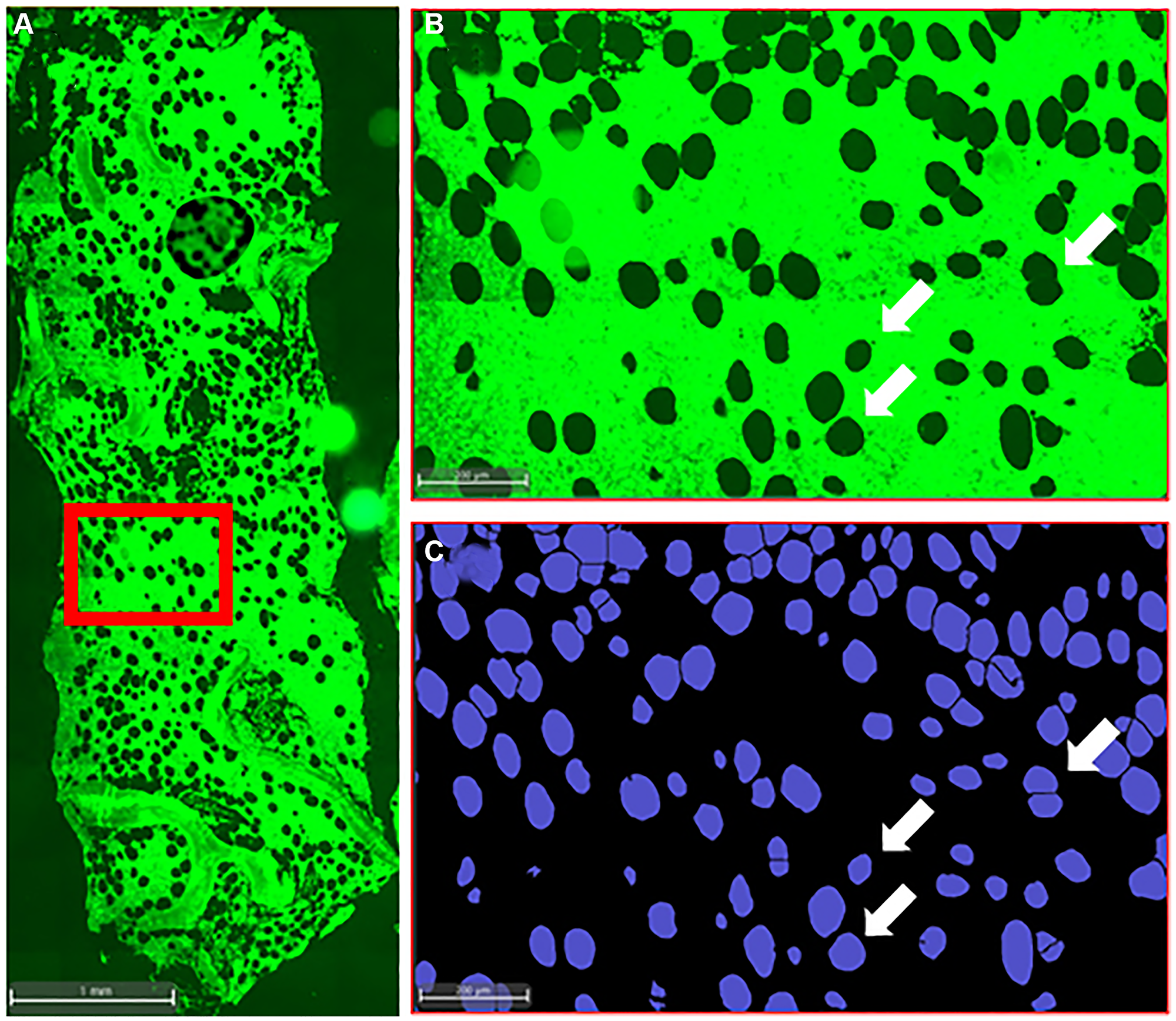
Figure 1: Histological analysis of iliac crest bone biopsy.
(A) Representative image of the iliac crest bone biopsy from a MGUS patient, illustrating the bone and bone marrow compartments. The autofluorescence highlights the structural arrangement of bone trabeculae and adipose tissue within the bone marrow. (B, C) Accurate identification of individual BMAds within the bone marrow by AI, indicating the heterogeneity in BMAd size and shape.
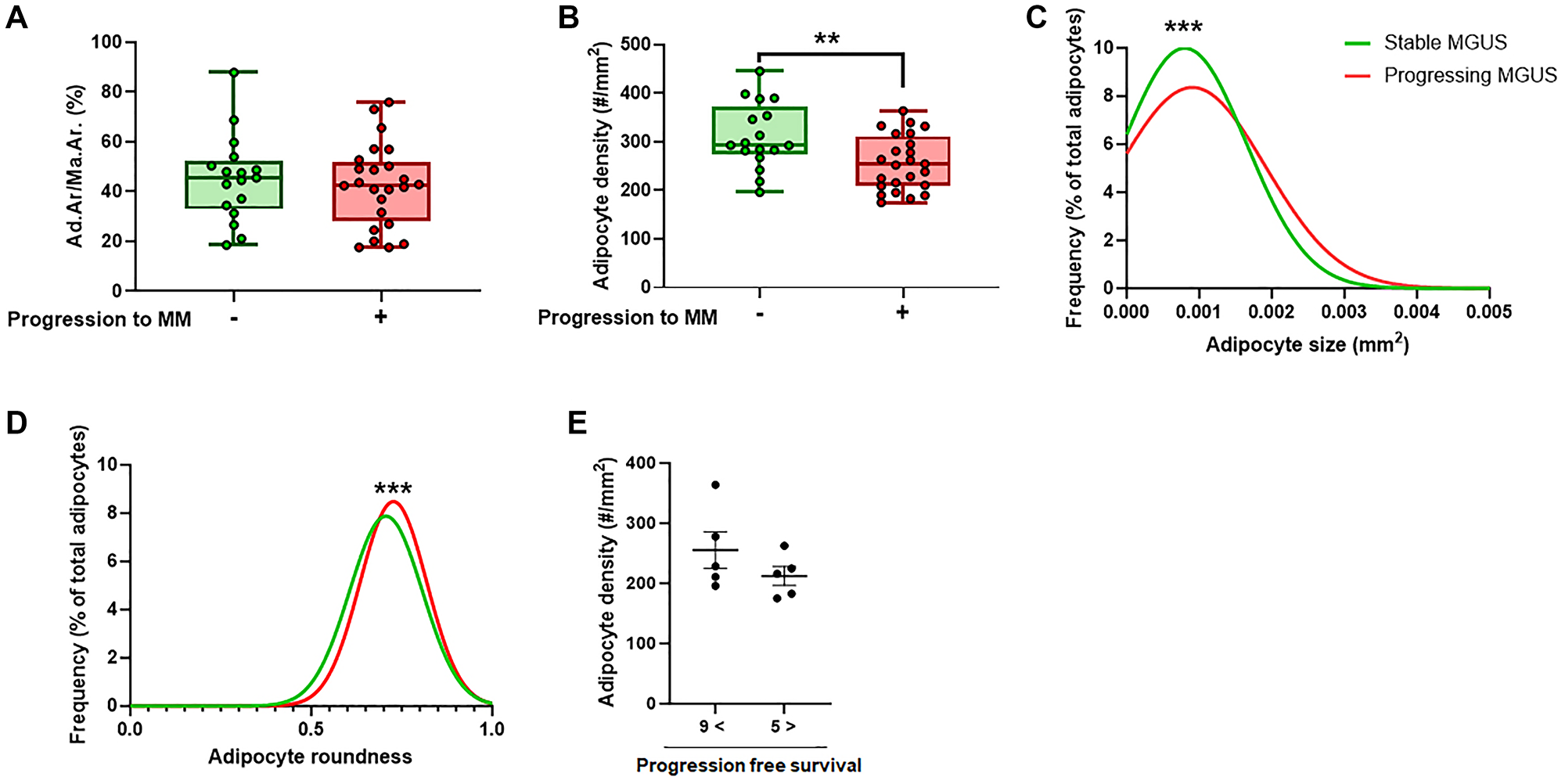
Figure 2: AI-assisted quantitative analysis of BMAT parameters in MGUS subjects with or without progression to MM.
(A) BMAT fraction within the marrow represented as adipocyte area/marrow area (Ad.Ar/Ma.Ar, %). (B) Density of adipocytes within the bone marrow (#/mm2). (C) Distribution profile of individual BMAd size in stable and progressing MGUS patients. (D) Distribution profile of individual BMAd roundness in stable and progressing MGUS patients. (E) Density of adipocytes within the bone marrow (#/mm2) in low risk MGUS patients who progressed to MM in more than 9 years after obtaining the biopsy compared to MGUS patients who had similar risk stratification score, but progressed to MM in less than 5 years after obtaining the biopsy. Data presented in A–D are obtained from 17 patients with stable MGUS and 24 patients with progressing MGUS. For C and D, number of analyzed BMAds was 26,601 for stable MGUS and 65,394 for progressing MGUS. Error bars on A and B represent standard error of the mean. **p < 0.01, ****p < 0.0001. Unpaired Student’s T-test was used for A and B, Kolmogorov-Smirnov test was used for C and D.
As expected, the risk stratification score was notably higher among progressing MGUS patients than their stable MGUS counterparts (Table 1). However, within progressing MGUS patients with comparable risk stratification scores, especially in the low-risk category, we observed a substantial variability in the time taken for progression to MM (progression free survival). Therefore, we aimed at discerning potential differences in BMAT parameters among progressing MGUS patients with similar risk scores but distinct timeframes for progression to MM. Interestingly, we found a discernible trend toward decreased BMAd density in low risk MGUS patients who developed MM within less than 5 years following biopsy collection, as compared to MGUS patients with similar risk score who developed MM in more than 9 years following biopsy collection (Figure 2E). We did not find notable changes in other BMAT parameters between these two groups (data not shown). Additional studies with larger number of patients are required to establish whether decreased BMAd density can be used as a novel risk factor for progression from MGUS to MM.
Table 1: Clinical characteristics of the patients
| Stable MGUS (n = 17) | Progressing MGUS (n = 24) | |
|---|---|---|
| Age (years; mean ± SD) | 42.53 ± 8.96 | 45.96 ± 11.70 |
| Sex (male n, %) | 10 (58.82) | 6 (25.00) |
| Serum paraprotein (g/L; mean ± SD) | 11.32 ± 8.28 | 11.62 ± 5.85 |
| Plasma cell burden (%; mean ± SD) | 5.04 ± 3.61 | 4.94 ± 3.22 |
| Risk stratification (low n, %) | 13 (76.47) | 21 (87.50) |
As the samples utilized in this study were obtained from biobanks, we did not have access to the data about patients’ weight or ongoing therapies that can affect bone marrow adiposity. Therefore, additional studies are warranted to corroborate the findings presented in this study.
To ensure the validity of AI-generated results, we performed head-to-head comparison between traditional (grid point) and AI-assisted histological analysis of BMAT parameters in a parallel cohort of trephine iliac crest human bone biopsies and observed comparable results (data not show). While one cannot rule out that our trained algorithm incorrectly recognizes empty lumen of blood vessel cross-sections as adipocytes, we estimate these uncommon occurrences to be unlikely and have a dismissible effect among the large adipocyte number detected in each biopsy.
We found a significant difference in the BMAd roundness coefficient between stable and progressing MGUS patients. In addition to the importance of this finding from the perspective of a potential biomarker for early detection of progressing MGUS patients, this finding indicates that the integration of advanced computational analysis and AI-driven algorithms in pathology holds the promise of revolutionizing the field by unraveling hidden patterns and subtle anomalies within the tissues that traditional methods might overlook. This paradigm shift towards computer/AI-augmented pathology could lead to enhanced diagnostic accuracy, personalized treatment strategies, and a deeper understanding of disease mechanisms across a spectrum of medical conditions. The additional advantage of applying this approach on unstained tissue sections highlights its potential for implementation as a cost-effective approach for routine pathological screening.
Taken together, our findings contribute to the growing understanding of the interplay between BMAT and hematological malignancies and the complex role of BMAT in the trajectory from MGUS to MM, urging further exploration and validation of these early indicators of the disease development to improve patient care and outcomes.
Materials and Methods
Patient cohorts
Iliac crest bone biopsies from 41 MGUS patients were used in this study, including 17 stable MGUS patients who did not progress to MM within 10 years after biopsy collection, and 24 MGUS patients with known progression to MM within 10 years following biopsy collection. The cohort of MGUS patients without progression to MM had a median age of 68 years (55–83 years, ~53% male) and the cohort of MGUS patients with progression to MM had a median age of 69 years (47–83 years, ~29% male). Patient characteristics are presented in Table 1. Patient risk stratification was performed based on myeloma type (IgA or not), M component (<15 g/L or not) and κ/λ ratio (normal or not) as described before [29]. Briefly, patients were stratified into a binary score of “yes” or “no” for each of the described risk factors and the sum of positive outcomes was defined as the overall risk stratification. Patients stratified into risk of progression ≤1 are defined as “low risk of progression”.
Histological analysis
Formalin-fixed, paraffin-embedded 3-mm trephine iliac crest bone biopsies were sliced into 3.5-μm sections and scanned at 82.6 ms exposure in the FITC immunofluorescent channel of an Olympus VS200 slide scanner. The artificial intelligence (AI) module from HALO (v.3.5, IndicaLabs, NM, USA) was used to detect and characterize bone marrow adipocytes, using the autofluorescence from unstained sections.
Statistical analysis
Data were analyzed in GraphPad Prism v9.3.1 (GraphPad Inc.) and are presented as mean ± standard error of the mean (SEM) or cumulative histograms. Data were analyzed by unpaired Student’s T-test or Kolmogorov-Smirnov test for cumulative distributions.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
Ethical statement
Biopsies were collected from Danish pathological biobanks in agreement with the Declaration of Helsinki and under approval from the Scientific Ethical Committee for the Region of Southern Denmark (S-2019110).
Consent
Patient consent was not requested, as historical biopsies retrieved from pathological biorepositories were used in this study.
FUNDING
Authors have received funding from the following institutions: NIH U01 grant AG075227, NEYE Foundation grant, Novo Nordisk Foundation grant (NNF22OC0075296), Danish Southern Region Research Grant, Odense University Hospital PhD grant, Gerda og Aage Haenschs Fond, Direktør Michael Hermann Nielsens mindelegat, Kirsten and Freddy Johansens Fond, Læge Sofus Carl Emil Friis og Hustru Olga Doris Friis’ Legat.
References
1. van de Donk NWC, Pawlyn C, Yong KL. Multiple myeloma. Lancet. 2021; 397:410–27. https://doi.org/10.1016/S0140-6736(21)00135-5. [PubMed].
2. Padala SA, Barsouk A, Barsouk A, Rawla P, Vakiti A, Kolhe R, Kota V, Ajebo GH. Epidemiology, Staging, and Management of Multiple Myeloma. Med Sci (Basel). 2021; 9:3. https://doi.org/10.3390/medsci9010003. [PubMed].
3. Kyle RA, Gertz MA, Witzig TE, Lust JA, Lacy MQ, Dispenzieri A, Fonseca R, Rajkumar SV, Offord JR, Larson DR, Plevak ME, Therneau TM, Greipp PR. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc. 2003; 78:21–33. https://doi.org/10.4065/78.1.21. [PubMed].
4. Schulman KL, Kohles J. Economic burden of metastatic bone disease in the U.S. Cancer. 2007; 109:2334–42. https://doi.org/10.1002/cncr.22678. [PubMed].
5. Landgren O, Kyle RA, Pfeiffer RM, Katzmann JA, Caporaso NE, Hayes RB, Dispenzieri A, Kumar S, Clark RJ, Baris D, Hoover R, Rajkumar SV. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: a prospective study. Blood. 2009; 113:5412–17. https://doi.org/10.1182/blood-2008-12-194241. [PubMed].
6. Han JH, Wang JN, Zhang YL, Cao XX, Zhou DB, Xu TD, Su W, Li J. Prevalence of monoclonal gammopathy of undetermined significance in a large population with annual medical check-ups in China. Blood Cancer J. 2020; 10:34. https://doi.org/10.1038/s41408-020-0303-8. [PubMed].
7. Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Offord JR, Dispenzieri A, Katzmann JA, Melton LJ 3rd. Prevalence of monoclonal gammopathy of undetermined significance. N Engl J Med. 2006; 354:1362–69. https://doi.org/10.1056/NEJMoa054494. [PubMed].
8. Wadhera RK, Rajkumar SV. Prevalence of monoclonal gammopathy of undetermined significance: a systematic review. Mayo Clin Proc. 2010; 85:933–42. https://doi.org/10.4065/mcp.2010.0337. [PubMed].
9. Anagnostopoulos A, Evangelopoulou A, Sotou D, Gika D, Mitsibounas D, Dimopoulos MA. Incidence and evolution of monoclonal gammopathy of undetermined significance (MGUS) in Greece. Ann Hematol. 2002; 81:357–61. https://doi.org/10.1007/s00277-002-0493-0. [PubMed].
10. Landgren O. Advances in MGUS diagnosis, risk stratification, and management: introducing myeloma-defining genomic events. Hematology Am Soc Hematol Educ Program. 2021; 2021:662–72. https://doi.org/10.1182/hematology.2021000303. [PubMed].
11. Cawthorn WP, Scheller EL, Learman BS, Parlee SD, Simon BR, Mori H, Ning X, Bree AJ, Schell B, Broome DT, Soliman SS, DelProposto JL, Lumeng CN, et al. Bone marrow adipose tissue is an endocrine organ that contributes to increased circulating adiponectin during caloric restriction. Cell Metab. 2014; 20:368–75. https://doi.org/10.1016/j.cmet.2014.06.003. [PubMed].
12. Fazeli PK, Horowitz MC, MacDougald OA, Scheller EL, Rodeheffer MS, Rosen CJ, Klibanski A. Marrow fat and bone--new perspectives. J Clin Endocrinol Metab. 2013; 98:935–45. https://doi.org/10.1210/jc.2012-3634. [PubMed].
13. Beekman KM, Regenboog M, Nederveen AJ, Bravenboer N, den Heijer M, Bisschop PH, Hollak CE, Akkerman EM, Maas M. Gender- and Age-Associated Differences in Bone Marrow Adipose Tissue and Bone Marrow Fat Unsaturation Throughout the Skeleton, Quantified Using Chemical Shift Encoding-Based Water-Fat MRI. Front Endocrinol (Lausanne). 2022; 13:815835. https://doi.org/10.3389/fendo.2022.815835. [PubMed].
14. Scheller EL, Doucette CR, Learman BS, Cawthorn WP, Khandaker S, Schell B, Wu B, Ding SY, Bredella MA, Fazeli PK, Khoury B, Jepsen KJ, Pilch PF, et al. Region-specific variation in the properties of skeletal adipocytes reveals regulated and constitutive marrow adipose tissues. Nat Commun. 2015; 6:7808. https://doi.org/10.1038/ncomms8808. [PubMed].
15. Ali D, Tencerova M, Figeac F, Kassem M, Jafari A. The pathophysiology of osteoporosis in obesity and type 2 diabetes in aging women and men: The mechanisms and roles of increased bone marrow adiposity. Front Endocrinol (Lausanne). 2022; 13:981487. https://doi.org/10.3389/fendo.2022.981487. [PubMed].
16. Diacinti D, Diacinti D, Iannacone A, Pepe J, Colangelo L, Nieddu L, Kripa E, Orlandi M, De Martino V, Minisola S, Cipriani C. Bone Marrow Adipose Tissue Is Increased in Postmenopausal Women With Postsurgical Hypoparathyroidism. J Clin Endocrinol Metab. 2023; 108:e807–15. https://doi.org/10.1210/clinem/dgad116. [PubMed].
17. Yeung DK, Griffith JF, Antonio GE, Lee FK, Woo J, Leung PC. Osteoporosis is associated with increased marrow fat content and decreased marrow fat unsaturation: a proton MR spectroscopy study. J Magn Reson Imaging. 2005; 22:279–85. https://doi.org/10.1002/jmri.20367. [PubMed].
18. Veldhuis-Vlug AG, Rosen CJ. Clinical implications of bone marrow adiposity. J Intern Med. 2018; 283:121–39. https://doi.org/10.1111/joim.12718. [PubMed].
19. Schwartz AV, Sigurdsson S, Hue TF, Lang TF, Harris TB, Rosen CJ, Vittinghoff E, Siggeirsdottir K, Sigurdsson G, Oskarsdottir D, Shet K, Palermo L, Gudnason V, Li X. Vertebral bone marrow fat associated with lower trabecular BMD and prevalent vertebral fracture in older adults. J Clin Endocrinol Metab. 2013; 98:2294–300. https://doi.org/10.1210/jc.2012-3949. [PubMed].
20. Dang T, Faje AT, Meenaghan E, Bredella MA, Bouxsein ML, Klibanski A, Fazeli PK. Bone marrow adipose tissue is associated with fracture history in anorexia nervosa. Osteoporos Int. 2022; 33:2619–27. https://doi.org/10.1007/s00198-022-06527-3. [PubMed].
21. Jafari A, Fairfield H, Andersen TL, Reagan MR. Myeloma-bone marrow adipocyte axis in tumour survival and treatment response. Br J Cancer. 2021; 125:775–77. https://doi.org/10.1038/s41416-021-01371-4. [PubMed].
22. Caers J, Deleu S, Belaid Z, De Raeve H, Van Valckenborgh E, De Bruyne E, Defresne MP, Van Riet I, Van Camp B, Vanderkerken K. Neighboring adipocytes participate in the bone marrow microenvironment of multiple myeloma cells. Leukemia. 2007; 21:1580–84. https://doi.org/10.1038/sj.leu.2404658. [PubMed].
23. Morris EV, Edwards CM. Bone marrow adiposity and multiple myeloma. Bone. 2019; 118:42–46. https://doi.org/10.1016/j.bone.2018.03.011. [PubMed].
24. Benova A, Tencerova M. Obesity-Induced Changes in Bone Marrow Homeostasis. Front Endocrinol (Lausanne). 2020; 11:294. https://doi.org/10.3389/fendo.2020.00294. [PubMed].
25. Choa R, Panaroni C, Bhatia R, Raje N. It is worth the weight: obesity and the transition from monoclonal gammopathy of undetermined significance to multiple myeloma. Blood Adv. 2023; 7:5510–23. https://doi.org/10.1182/bloodadvances.2023010822. [PubMed].
26. Fairfield H, Dudakovic A, Khatib CM, Farrell M, Costa S, Falank C, Hinge M, Murphy CS, DeMambro V, Pettitt JA, Lary CW, Driscoll HE, McDonald MM, et al. Myeloma-Modified Adipocytes Exhibit Metabolic Dysfunction and a Senescence-Associated Secretory Phenotype. Cancer Res. 2021; 81:634–47. https://doi.org/10.1158/0008-5472.CAN-20-1088. [PubMed].
27. Liu H, He J, Koh SP, Zhong Y, Liu Z, Wang Z, Zhang Y, Li Z, Tam BT, Lin P, Xiao M, Young KH, Amini B, et al. Reprogrammed marrow adipocytes contribute to myeloma-induced bone disease. Sci Transl Med. 2019; 11:eaau9087. https://doi.org/10.1126/scitranslmed.aau9087. [PubMed].
28. Panaroni C, Fulzele K, Mori T, Siu KT, Onyewadume C, Maebius A, Raje N. Multiple myeloma cells induce lipolysis in adipocytes and uptake fatty acids through fatty acid transporter proteins. Blood. 2022; 139:876–88. https://doi.org/10.1182/blood.2021013832. [PubMed].
29. Kyle RA, Larson DR, Therneau TM, Dispenzieri A, Kumar S, Cerhan JR, Rajkumar SV. Long-Term Follow-up of Monoclonal Gammopathy of Undetermined Significance. N Engl J Med. 2018; 378:241–49. https://doi.org/10.1056/NEJMoa1709974. [PubMed].

