Introduction
Fifty years ago, in 1973, V. Anisimov and coauthors have demonstrated for the first time an inhibitory effect of the pineal gland hormone melatonin on cancer in vivo, namely on transplantable mammary tumors in mice [1]. Subsequently, it was shown in a number of studies that melatonin administration with drinking water at night inhibits chemically induced mammary carcinogenesis in mice and rats (reviewed in [2, 3]).
On the contrary, maintaining female mice and rats under round-the-clock lighting conditions, which suppresses the nighttime production of melatonin, stimulates spontaneous and chemical carcinogen-induced mammary tumors development [4].
As of today, the query “cancer AND melatonin AND mice” in Pubmed returns about 550 entries. In Figure 1 the time course of the annual numbers of such publications is compared with the time courses of publications on melatonin and cancer and on cancer in general. All trends are traced after 1958, the year of the discovery of melatonin. The reports on experiments with mice are obviously much less numerous than on melatonin and cancer in general (NB: Y-axis is logarithmic). Both trends related to melatonin are parallel to the trend of publications on cancer, and all three trends are almost exponential (linear in a semilogarithmic plot), as usual. However, the “cancer, melatonin and mice” trend is peculiar in that the interest in melatonin-and-cancer-related work with mice seems to be going down in the period from about 2005 to 2015; however, the interest rejuvenated thereafter. No reasons for this zigzag are apparent.
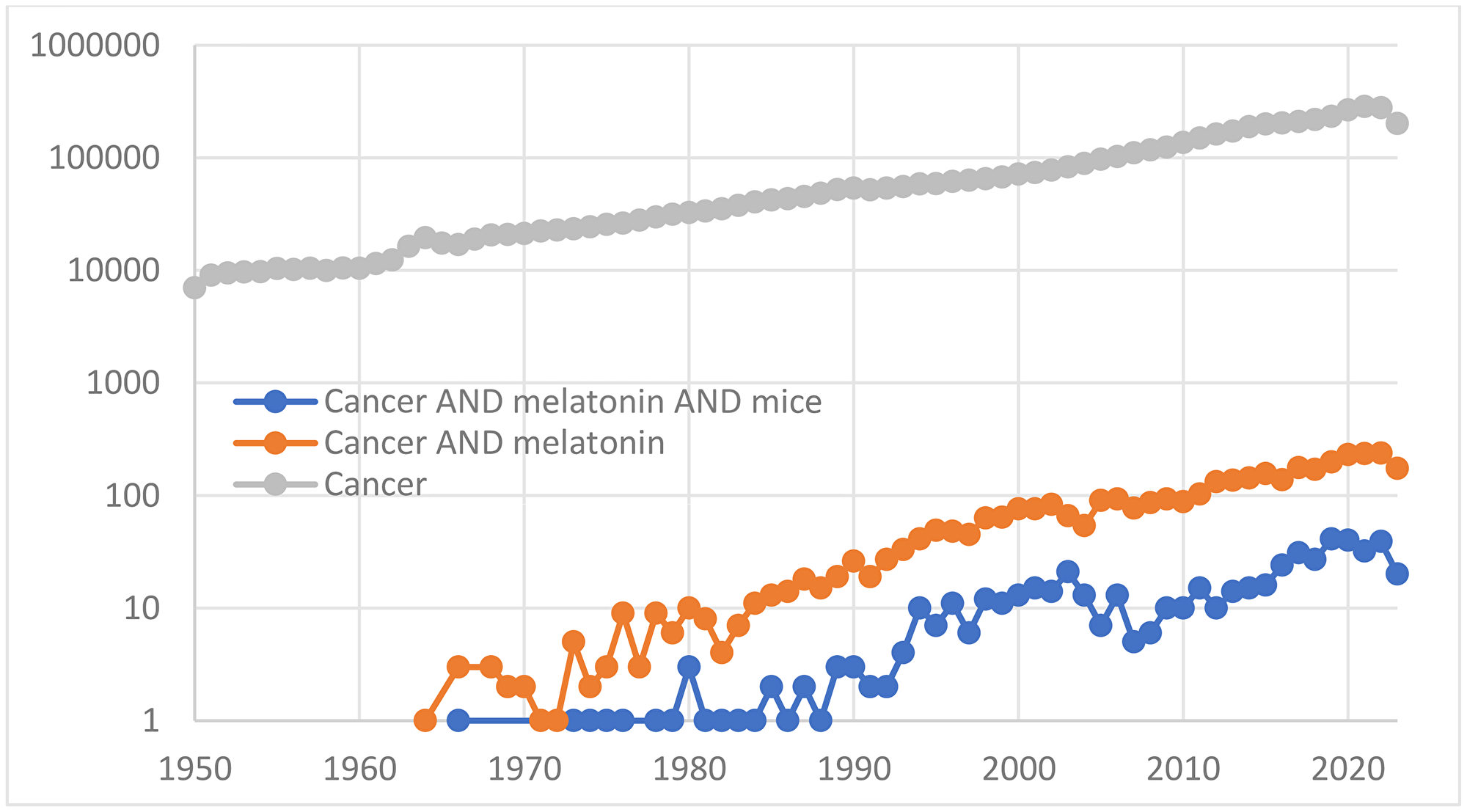
Figure 1: Trends in the annual numbers of publications.
Early studies
Other controversies, which were recognized as early as in 1970-ies, include the lack of consistency in the antitumor effects of melatonin observed in in vivo models, including murine ones, as has been analyzed by Bartsch and Bartsch in 1981 [5], see Table 1.
Table 1: Controversies in the early studies of the effects of melatonin on cancer in rodents (modified from ref. [5])
| References | Experimental system | Results |
|---|---|---|
| Tumor inhibition in intact animals | ||
| [1] | Transplantable mammary tumor in mice | Inhibition of tumor development |
| [6] | Solid leukemia in mice (i.m.) | Statistically significant inhibition of tumor size |
| [7] | Methylcholanthrene-induced sarcoma in mice | Slight delay in occurrence of tumors |
| [8] | Transplantable mammary tumor in rats | Statistically significant decrease in tumor size |
| [9] | DMBA-induced mammary tumors in rats | Decreased tumor incidence and tumor growth (melatonin given at 5 p.m.) |
| Tumor inhibition in pinealectomized animals | ||
| [9] | DMBA-induced mammary tumors in rats | Decreased tumor incidence |
| [10] | Transplantable melanoma in hamsters | Inhibition of tumor development |
| [7] | Yoshida sarcoma in Wistar rats | Prolonged survival time |
| No effect in intact animals | ||
| [7] | Yoshida sarcoma in Wistar rats | No effect |
| [11] | Yoshida sarcoma in rats | No effect |
| [12] | Walker carcinosarcoma in rats | No effect |
| [9] | DMBA-induced mammary tumors in rats | No effect under constant light |
| No effect in pinealectomized animals | ||
| [9] | DMBA-induced mammary tumors in rats | No effect under constant light |
| Tumor stimulation in intact animals | ||
| [13] | DMBA-induced mammary tumors in rats | Increased tumor incidence and higher degree of malignancy |
| [7] | Lewis lung carcinoma in mice | Slight reduction of survival time |
To find the cause of the apparent discrepancies, mice were used by Bartsch and Bartsch [5]. These authors came to the important conclusion that the effects of melatonin on cancer crucially depend on the time of treatment: “It appears that under long photoperiods melatonin shows opposite effects on fibrosarcoma ascites and Ehrlich solid tumors depending on the time of the day at which the compound was administered. Tumors are stimulated by melatonin injections in the morning and inhibited by late afternoon injections.”
In more general terms, this conclusion means that the effects of melatonin depend on animal conditions and therefore, since melatonin is a hormone, which is able to change these conditions, melatonin effects on cancer must depend to a significant extent on the systemic effects of melatonin, which may be seen only in animal models, not in cell cultures, ex vivo explants etc.
Murine models for melatonin-vs-cancer studies
Murine models for studying melatonin effects on cancer comprise, in the time order of their introduction, mice grafted with murine tumors [1], chemically induced tumors [7], spontaneous carcinogenesis in mice [14, 15], transgenic HER2/neu oncogene-bearing mice [16, 17], and nude mice grafted with human prostate tumors [18]. Besides mammary and prostate carcinomas, other cancers, such as tumors of the colon [19], lung [20], muscle [21], and stomach [22], were studied in mice under the influence of melatonin. An important aspect of such studies is that the effects of melatonin not only on cancer itself, but also on the consequences of anticancer therapy, including its side-effects, may be evaluated.
Each line of experimentation has provided for coming to important results and conclusions. For example, lifelong treatment of mice with melatonin decreased the incidence of spontaneous tumors, mostly mammary carcinomas, only at a low content of melatonin in drinking water (2 mg/L), but not at a high content (20 mg/L) [23]. The potentiating effect of melatonin on cytotoxic therapy against mammary tumors in HER2/neu transgenic mice was shown to depend on the timing of melatonin administration relative to cytotoxic drug administration [24]. Melatonin was shown to alleviate the depression syndrome in mice treated with the alkylating agent temozolomide used in therapy for brain cancer [25]. The ability of melatonin to alleviate the side effects of cytotoxic drugs and radiation has been demonstrated in several murine models, e.g., [26–28].
In nude mice grafted with human gastric cancer, melatonin inhibited lung metastases development, importantly, by suppressing the epithelial to mesenchymal transition (EMT) [22]. EMT was inhibited by melatonin in a murine model of low-grade inflammation [29]. Although no antitumor effects of melatonin were evaluated in this model, the increasing awareness of the importance of inflammation and EMT for primary cancer and metastases development [30] makes this finding highly relevant for oncology.
Conclusion
Thus, having been used for fifty years, murine models proved to be valuable and, in some cases, indispensable for advancing melatonin applications in oncology by suggesting novel facets of melatonin effects and utility. Because the employment of melatonin beyond its established clinical fields needs no licensing, such suggestions may be tested in practice more readily than in the cases of other drugs. In fact, such trials have already yielded promising clinical results, e.g., [31].
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
References
1. Anisimov VN, Morozov VG, Khavinson VKh, Dil’man VM. Sopostavlenie protivoopukholevoĭ aktivnosti ékstraktov épifiza, gipotalamusa, melatonina i sigetina u mysheĭ s perevivnym rakom molochnoĭ zhelezy [Comparison of the anti-tumor activity of extracts of the epiphysis and hypothalamus, melatonin and sigetin in mice with transplanted mammary gland cancer]. Vopr Onkol. 1973; 19:99–101. [PubMed].
2. Anisimov VN, Popovich IG, Zabezhinski MA, Anisimov SV, Vesnushkin GM, Vinogradova IA. Melatonin as antioxidant, geroprotector and anticarcinogen. Biochim Biophys Acta. 2006; 1757:573–89. https://doi.org/10.1016/j.bbabio.2006.03.012. [PubMed].
3. Mihanfar A, Yousefi B, Azizzadeh B, Majidinia M. Interactions of melatonin with various signaling pathways: implications for cancer therapy. Cancer Cell Int. 2022; 22:420. https://doi.org/10.1186/s12935-022-02825-2. [PubMed].
4. Blask DE, Sauer LA, Dauchy RT. Melatonin as a chronobiotic/anticancer agent: cellular, biochemical, and molecular mechanisms of action and their implications for circadian-based cancer therapy. Curr Top Med Chem. 2002; 2:113–32. https://doi.org/10.2174/1568026023394407. [PubMed].
5. Bartsch H, Bartsch C. Effect of melatonin on experimental tumors under different photoperiods and times of administration. J Neural Transm. 1981; 52:269–79. https://doi.org/10.1007/BF01256752. [PubMed].
6. Buswell RS. Letter: The pineal and neoplasia. Lancet. 1975; 1:34–5. https://doi.org/10.1016/s0140-6736(75)92390-9. [PubMed].
7. Lapin V, Ebels I. Effects of some low molecular weight sheep pineal fractions and melatonin on different tumors in rats and mice. Oncology. 1976; 33:110–3. https://doi.org/10.1159/000225117. [PubMed].
8. Karmali RA, Horrobin DF, Ghayur T, Manku MS, Cunnane SC, Morgan RO, Ally AI, Karmazyn M, Oka M. Influence of agents which modulate thromboxane A2 synthesis or action on R3230AC mammary carcinoma. Cancer Lett. 1978; 5:205–8. https://doi.org/10.1016/s0304-3835(78)80040-8. [PubMed].
9. Aubert C, Janiaud P, Lecalvez J. Effect of pinealectomy and melatonin on mammary tumor growth in Sprague-Dawley rats under different conditions of lighting. J Neural Transm. 1980; 47:121–30. https://doi.org/10.1007/BF01670163. [PubMed].
10. El-Domeiri AA, Das Gupta TK. The influence of pineal ablation and administration of melatonin on growth and spread of hamster melanoma. J Surg Oncol. 1976; 8:197–205. https://doi.org/10.1002/jso.2930080303. [PubMed].
11. Huxley M, Tapp E. Effects of biogenic amines on the growth of rat tumours. Life Sci. 1972; 11:19–23. https://doi.org/10.1016/0024-3205(72)90147-6.
12. Bostelmann W, Göcke H, Ernst B, Tessmann D. Der Einfluss einer Melatoninbehandlung auf das Wachstum des Walker-Carcinosarkoms der Ratte [Effect of melatonin treatment on growth of Walker carcinosarcoma in rats]. Zentralbl Allg Pathol. 1971; 114:289–91. [PubMed].
13. Hamilton T. Influence of environmental light and melatonin upon mammary tumour induction. Br J Surg. 1969; 56:764–6. https://doi.org/10.1002/bjs.1800561018. [PubMed].
14. Subramanian A, Kothari L. Melatonin, a suppressor of spontaneous murine mammary tumors. J Pineal Res. 1991; 10:136–40. https://doi.org/10.1111/j.1600-079x.1991.tb00830.x. [PubMed].
15. Anisimov VN, Zavarzina NY, Zabezhinski MA, Popovich IG, Zimina OA, Shtylick AV, Arutjunyan AV, Oparina TI, Prokopenko VM, Mikhalski AI, Yashin AI. Melatonin increases both life span and tumor incidence in female CBA mice. J Gerontol A Biol Sci Med Sci. 2001; 56:B311–23. https://doi.org/10.1093/gerona/56.7.b311. [PubMed].
16. Rao GN, Ney E, Herbert RA. Effect of melatonin and linolenic acid on mammary cancer in transgenic mice with c-neu breast cancer oncogene. Breast Cancer Res Treat. 2000; 64:287–96. https://doi.org/10.1023/a:1026552405042. [PubMed].
17. Baturin DA, Alimova IN, Anisimov VN, Popovich IG, Zabezhinski MA, Provinciali M, Mancini R, Franceschi C. The effect of light regimen and melatonin on the development of spontaneous mammary tumors in HER-2/neu transgenic mice is related to a downregulation of HER-2/neu gene expression. Neuro Endocrinol Lett. 2001; 22:441–7. [PubMed].
18. Xi SC, Siu SW, Fong SW, Shiu SY. Inhibition of androgen-sensitive LNCaP prostate cancer growth in vivo by melatonin: association of antiproliferative action of the pineal hormone with mt1 receptor protein expression. Prostate. 2001; 46:52–61. https://doi.org/10.1002/1097-0045(200101)46:1<52::aid-pros1008>3.0.co;2-z. [PubMed].
19. Winczyk K, Pawlikowski M, Guerrero JM, Karasek M. Possible involvement of the nuclear RZR/ROR-alpha receptor in the antitumor action of melatonin on murine Colon 38 cancer. Tumour Biol. 2002; 23:298–302. https://doi.org/10.1159/000068569. [PubMed].
20. Rapozzi V, Perissin L, Zorzet S, Giraldi T. Effects of melatonin administration on tumor spread in mice bearing Lewis lung carcinoma. Pharmacol Res. 1992 (Suppl 1); 25:71–2. https://doi.org/10.1016/1043-6618(92)90545-m. [PubMed].
21. Dauchy RT, Blask DE, Dauchy EM, Davidson LK, Tirrell PC, Greene MW, Tirrell RP, Hill CR, Sauer LA. Antineoplastic effects of melatonin on a rare malignancy of mesenchymal origin: melatonin receptor-mediated inhibition of signal transduction, linoleic acid metabolism and growth in tissue-isolated human leiomyosarcoma xenografts. J Pineal Res. 2009; 47:32–42. https://doi.org/10.1111/j.1600-079X.2009.00686.x. [PubMed].
22. Wang X, Wang B, Zhan W, Kang L, Zhang S, Chen C, Hou D, You R, Huang H. Melatonin inhibits lung metastasis of gastric cancer in vivo. Biomed Pharmacother. 2019; 117:109018. https://doi.org/10.1016/j.biopha.2019.109018. [PubMed].
23. Anisimov VN, Alimova IN, Baturin DA, Popovich IG, Zabezhinski MA, Rosenfeld SV, Manton KG, Semenchenko AV, Yashin AI. Dose-dependent effect of melatonin on life span and spontaneous tumor incidence in female SHR mice. Exp Gerontol. 2003; 38:449–61. https://doi.org/10.1016/s0531-5565(02)00240-1. [PubMed].
24. Panchenko AV, Tyndyk ML, Maydin MA, Baldueva IA, Artemyeva AS, Kruglov SS, Kireeva GS, Golubev AG, Belyaev AM, Anisimov VN. Melatonin Administered before or after a Cytotoxic Drug Increases Mammary Cancer Stabilization Rates in HER2/Neu Mice. Chemotherapy. 2020; 65:42–50. https://doi.org/10.1159/000509238. [PubMed].
25. Cabrera-Muñoz EA, Ramírez-Rodríguez GB, Díaz-Yañez L, Reyes-Galindo V, Meneses-San Juan D, Vega-Rivera NM. Melatonin Prevents Depression but Not Anxiety-like Behavior Produced by the Chemotherapeutic Agent Temozolomide: Implication of Doublecortin Cells and Hilar Oligodendrocytes. Int J Mol Sci. 2023; 24:13376. https://doi.org/10.3390/ijms241713376. [PubMed].
26. Rodrigues TD, Lima KR, Uggioni MLR, Ferraz SD, Cardoso HS, Colonetti T, da Rosa MI. Effectiveness of melatonin adjuvant treatment in cisplatin to prevent depletion of ovarian follicles in mice: systematic review†. Biol Reprod. 2022; 107:1386–94. https://doi.org/10.1093/biolre/ioac164. [PubMed].
27. Zhang X, Xia Q, Wei R, Song H, Mi J, Lin Z, Yang Y, Sun Z, Zou K. Melatonin protects spermatogonia from the stress of chemotherapy and oxidation via eliminating reactive oxidative species. Free Radic Biol Med. 2019; 137:74–86. https://doi.org/10.1016/j.freeradbiomed.2019.04.009. [PubMed].
28. Abadi SHMH, Shirazi A, Alizadeh AM, Changizi V, Najafi M, Khalighfard S, Nosrati H. The Effect of Melatonin on Superoxide Dismutase and Glutathione Peroxidase Activity, and Malondialdehyde Levels in the Targeted and the Non-targeted Lung and Heart Tissues after Irradiation in Xenograft Mice Colon Cancer. Curr Mol Pharmacol. 2018; 11:326–35. https://doi.org/10.2174/1874467211666180830150154. [PubMed].
29. Chowdhury O, Ghosh S, Das A, Liu H, Shang P, Stepicheva NA, Hose S, Sinha D, Chattopadhyay S. Sustained systemic inflammation increases autophagy and induces EMT/fibrotic changes in mouse liver cells: Protection by melatonin. Cell Signal. 2023; 101:110521. https://doi.org/10.1016/j.cellsig.2022.110521. [PubMed].
30. Ribatti D, Tamma R, Annese T. Epithelial-Mesenchymal Transition in Cancer: A Historical Overview. Transl Oncol. 2020; 13:100773. https://doi.org/10.1016/j.tranon.2020.100773. [PubMed].
31. Zharinov GM, Bogomolov OA, Chepurnaya IV, Neklasova NY, Anisimov VN. Melatonin increases overall survival of prostate cancer patients with poor prognosis after combined hormone radiation treatment. Oncotarget. 2020; 11:3723–29. https://doi.org/10.18632/oncotarget.27757. [PubMed].

