Introduction
Cyclin-dependent kinases (CDK) belong to a family of serine/threonine kinases that need to form heterodimeric complexes with cyclins to perform their functions. These kinases are involved in multiple processes within cells, including cell cycle, apoptosis, transcription and differentiation. These kinases are often overexpressed in different malignancies, making them potential targets for new drugs [1]. Among transcription-associated CDKs, CDK9, along with cyclin T, forms the main component of positive transcription elongation factor b (PTEFb). This complex phosphorylates RNA polymerase II (RNAPII) to stimulate transcription elongation of most protein coding genes [2, 3]. Inhibition of P-TEFb plays a key role in tumors that continuously produce short-lived proteins such as MYC, myeloid cell leukemia 1 protein (MCL1) and cyclin D1, which are well known to be involved in the development of most hematological malignancies [4]. The observed clinical activity of non-selective CDK inhibitors was attributed primarily to their CDK9 activity [5]. The first generation of pan-CDK inhibitors targeting CDK9, which included flavopiridol and dinaciclib, showed suboptimal efficacy and their development was therefore suspended [6, 7]. A second generation of inhibitors were further developed, and exhibited more specific affinity for CDK9. The first in this class of CDK9 inhibitors, atuveciclib, was evaluated in two clinical studies, involving patients with advanced solid tumors and in subjects with relapsed acute myeloid leukemia (AML) [8, 9]. Both studies demonstrated high incidences of neutropenia that could not be managed with granulocyte colony-stimulating factor across different dose schedules. This led to dose-limiting toxicity at sub-therapeutic doses and resulted in discontinuation of the drug. However, further optimization of atuveciclib led to the development of a highly potent and more selective CDK9 inhibitor, enitociclib (VIP152, formerly BAY 1251152) [10]. In human xenograft tumor models of AML, enitociclib showed promising in vivo antitumor efficacy as a single agent, with a favorable tolerability profile across various dosing schedules, including weekly intravenous dosing [10].
In the first in-human trial (NCT02635672), carried out by Diamond and collaborators [11], enitociclib showed evidence of clinical efficacy with a low toxicity profile in advanced malignancies. Remarkably, a subgroup of patients treated for lymphoma with MYC and BCL6 and/or BCL2 rearrangements, so-called high-grade B-cell lymphoma (HGBL), also showed clinical benefit [12]. The study enrolled 37 patients with solid tumors or lymphomas refractory to all available therapies. They received enitociclib monotherapy as a 30-minute intravenous infusion, given once-weekly, every 21 days. The escalating doses were 5, 10, 15, 22.5 and 30 mg until disease progression or unacceptable toxicity. The maximum tolerated dose of enitociclib was defined as 30 mg. The most common treatment-related adverse events were grade 1/2 in severity. Grade 3/4 events were infrequent and independent of dose, except for neutropenia (21.6%), which was satisfactorily managed with granulocyte colony-stimulating factor, G-CSF, but required dose reduction in 6 cases. The pharmacokinetic and pharmacodynamic profiles were consistent with the in vivo results. Comparison to baseline revealed downregulation of MYC, MCL1 and PCNA mRNA levels at all doses tested. This downregulation was dose- and time-dependent, with maximal pathway inhibition achieved at the two highest doses (22.5 and 30 mg).
With respect to its antitumor efficacy, enitociclib showed no objective activity in a cohort of solid tumors (not selected based on MYC expression) for 7 patients achieved stable disease (23%) 2 of those responses were durable over time (9.5 months in a patient with pancreatic cancer and 16.8 months in a patient with salivary gland cancer). The most remarkable responses were observed in the HGBL cohort. Of those 7 patients, 2 (29%) showed durable complete metabolic responses that lasted more than 2 and 3 years, respectively.
Taking a closer look at those 2 patients, both had localized disease at inclusion (Ann-Arbor stage II), and their responses to enitociclib progressively deepened over time. Partial responses were achieved after 5 and 6 months and complete metabolic responses (CMR) at 7 and 8 months from the first dose received (Figures 1 and 2). These patients stopped treatment (as a result of the COVID-19 pandemic during which follow-up became more difficult) after 20 and 36 months in complete remission, respectively. And after 6.4 and 4.8 years, their CMRs continue to be sustained as of today. These findings are indicative of the antitumor activity of this new CDK9 inhibitor against HGBL. However, it requires time to achieve this effect, likely because the drug’s mechanism of action requires time to fully inhibit the production of short-lived proteins and thus induce clinical responses.
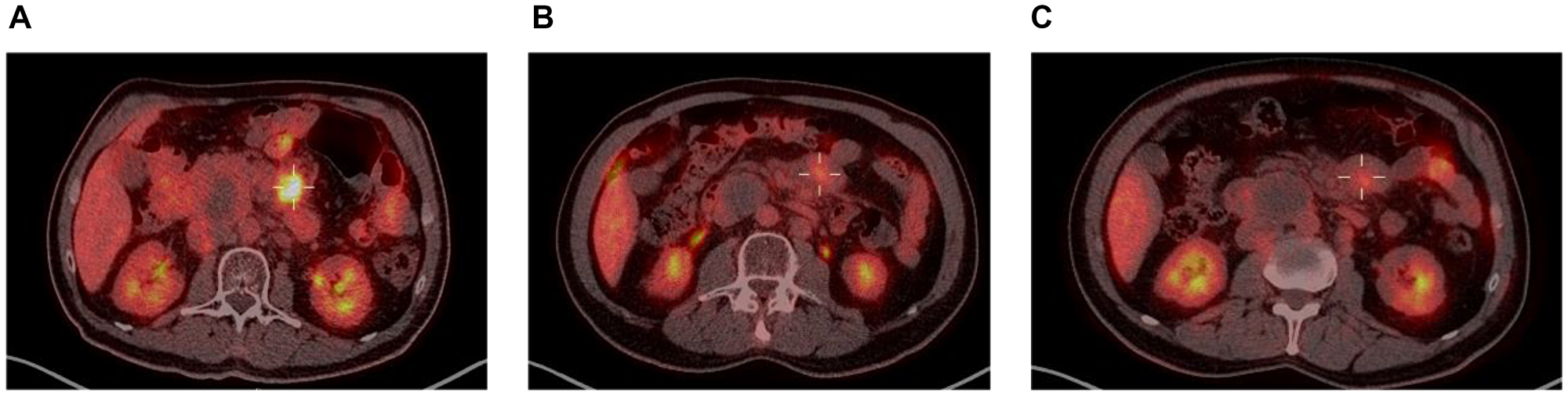
Figure 1: PET scan at screening (A), 5 months (B) and 8 months on treatment (C).
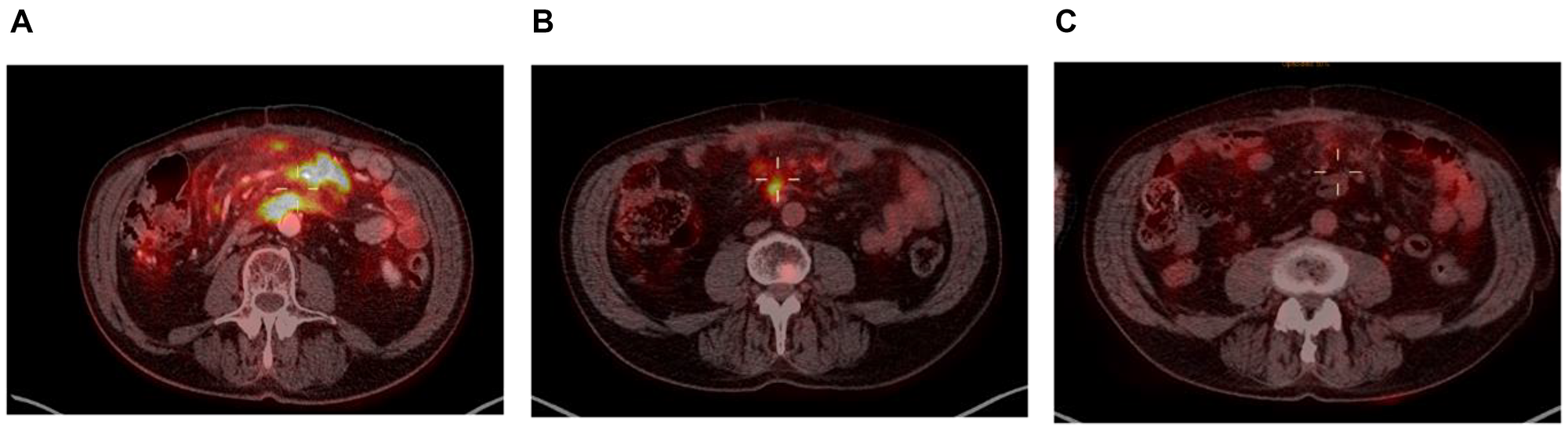
Figure 2: PET scan at screening (A), at 6 months (B) and at 7 months (C).
HGBLs are aggressive diseases with a high proliferation index and, unfortunately, most patients progress within the first weeks of treatment. Consequently, monotherapy with a CDK9 inhibitor would seems to be insufficient to overcome these highly proliferative tumors. For that reason, combined treatment strategies may be a reasonable approach to improve efficacy. Indeed, the rapid development of new drugs to treat DLBCL/HGBL in recent years offers a broad spectrum of treatment possibilities. For instance, the approval of CAR-T therapy has changed the present treatment paradigm in the relapse/refractory (R/R) setting, obtaining outstanding response rates. However, there are two main issues to be considered. First, long manufacturing processes lead to a higher risk of disease progression in patients awaiting therapy. Second, only about 40% of patients achieve durable remission [13]. Bispecific monoclonal antibodies are a great off-the-shelf alternative in patients with R/R DLBCL/HGBL, inducing complete responses in approximately 40% of patients, even after CAR-T [14], though long-term follow-up will be required to confirm whether the response rate is maintained over time. Several monoclonal antibodies and antibody drug conjugates, including tafasitamab [15] and loncastuximab tesirine [16], both targeting CD19, and polatuzumab, which targets antiCD79b, have been approved [17]. Zilovertamab vedotin is a new antibody-drug conjugate that targets ROR1 antigen and shows promising preliminary results [18]. Among the targeted small molecule therapies, the efficacy of cereblon E3 ligase modulators (CELMoDs) has been confirmed when used in monotherapy and in combination [15]. BTK inhibitors are being tested for ABC DLBCL [19] and MALT, and CARD11 inhibitors are under development. The new generation CDK9 inhibitors, especially enitociclib, could be a good combination partner with these drugs due to its low toxicity profile and different mechanism of action, which could result in synergistic effects. For instance, the second-generation CDK2/9 inhibitor fadraciclib (CYC065) was shown to reduce MCL-1 protein levels and to induce apoptosis in chronic lymphocytic leukemia cells. Moreover, preclinical work suggests fadraciclib overcomes microenvironment-mediated protection and exerts synergism between CDK2/CDK9 inhibitor and venetoclax [20].
Conclusion
In summary, most hematological malignancies are characterized by overexpression of certain cancer promoting genes, such as MYC and MCL1. CDK9 inhibitors are relatively new drugs that inhibit transcription of these anti-apoptotic and pro-survival proteins. Different preclinical studies have confirmed the activity of CDK9 inhibitors in animal models and their potential synergism with other drugs. In its first in-human trial, enitociclib demonstrated clinical activity in a small cohort of patients with HGBL harboring MYC and BCL2 and/or BCL6 rearrangements, and complete responses were achieved in 2 of 7 subjects (29%) in monotherapy. These data suggest CDK9 inhibitors could play a role in future treatments of hematological diseases and could be a great ally when combined with other therapeutic approaches.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
FUNDING
Instituto de investigación Sanitaria-Fundación Jiménez Díaz (IIS-FJD) and START.
References
1. Lim S, Kaldis P. Cdks, cyclins and CKIs: roles beyond cell cycle regulation. Development. 2013; 140:3079–93. https://doi.org/10.1242/dev.091744. [PubMed].
2. Huang Z, Wang T, Wang C, Fan Y. CDK9 inhibitors in cancer research. RSC Med Chem. 2022; 13:688–710. https://doi.org/10.1039/d2md00040g. [PubMed].
3. Mandal R, Becker S, Strebhardt K. Targeting CDK9 for Anti-Cancer Therapeutics. Cancers (Basel). 2021; 13:2181. https://doi.org/10.3390/cancers13092181. [PubMed].
4. Anshabo AT, Milne R, Wang S, Albrecht H. CDK9: A Comprehensive Review of Its Biology, and Its Role as a Potential Target for Anti-Cancer Agents. Front Oncol. 2021; 11:678559. https://doi.org/10.3389/fonc.2021.678559. [PubMed].
5. Gregory GP, Hogg SJ, Kats LM, Vidacs E, Baker AJ, Gilan O, Lefebure M, Martin BP, Dawson MA, Johnstone RW, Shortt J. CDK9 inhibition by dinaciclib potently suppresses Mcl-1 to induce durable apoptotic responses in aggressive MYC-driven B-cell lymphoma in vivo. Leukemia. 2015; 29:1437–41. https://doi.org/10.1038/leu.2015.10. [PubMed].
6. Zeidner JF, Karp JE. Clinical activity of alvocidib (flavopiridol) in acute myeloid leukemia. Leuk Res. 2015; 39:1312–18. https://doi.org/10.1016/j.leukres.2015.10.010. [PubMed].
7. Nemunaitis JJ, Small KA, Kirschmeier P, Zhang D, Zhu Y, Jou YM, Statkevich P, Yao SL, Bannerji R. A first-in-human, phase 1, dose-escalation study of dinaciclib, a novel cyclin-dependent kinase inhibitor, administered weekly in subjects with advanced malignancies. J Transl Med. 2013; 11:259. https://doi.org/10.1186/1479-5876-11-259. [PubMed].
8. Bayer Clinical Trials. 16520-phase I dose escalation of BAY1143572 in subjects with acute leukemia. https://clinicaltrials.bayer.com/.
9. Bayer Clinical Trials. 16519-open label phase I dose escalation study with BAY1143572 in patients with advanced cancer. https://clinicaltrials.bayer.com.
10. Lücking U, Kosemund D, Böhnke N, Lienau P, Siemeister G, Denner K, Bohlmann R, Briem H, Terebesi I, Bömer U, Schäfer M, Ince S, Mumberg D, et al. Changing for the Better: Discovery of the Highly Potent and Selective CDK9 Inhibitor VIP152 Suitable for Once Weekly Intravenous Dosing for the Treatment of Cancer. J Med Chem. 2021; 64:11651–74. https://doi.org/10.1021/acs.jmedchem.1c01000. [PubMed].
11. Diamond JR, Boni V, Lim E, Nowakowski G, Cordoba R, Morillo D, Valencia R, Genvresse I, Merz C, Boix O, Frigault MM, Greer JM, Hamdy AM, et al. First-in-Human Dose-Escalation Study of Cyclin-Dependent Kinase 9 Inhibitor VIP152 in Patients with Advanced Malignancies Shows Early Signs of Clinical Efficacy. Clin Cancer Res. 2022; 28:1285–93. https://doi.org/10.1158/1078-0432.CCR-21-3617. [PubMed].
12. Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M, Vardiman JW. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016; 127:2391–405. https://doi.org/10.1182/blood-2016-03-643544.
13. Kuhnl A, Roddie C, Tholouli E, Menne T, Linton KM, Lugthart S, Chaganti S, Sanderson R, O'Reilly M, Norman J, Osborne W, Radford JA, Besley C, et al. Real-world data of high-grade lymphoma patients treated with CD19 CAR-T in the UK. Br J Haematol. 2020; 189:30–31. http://hdl.handle.net/10541/623044.
14. Dickinson MJ, Carlo-Stella C, Morschhauser F, Bachy E, Corradini P, Iacoboni G, Khan C, Wróbel T, Offner F, Trněný M, Wu SJ, Cartron G, Hertzberg M, et al. Glofitamab for Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med. 2022; 387:2220–31. https://doi.org/10.1056/NEJMoa2206913. [PubMed].
15. Salles G, Duell J, González Barca E, Tournilhac O, Jurczak W, Liberati AM, Nagy Z, Obr A, Gaidano G, André M, Kalakonda N, Dreyling M, Weirather J, et al. Tafasitamab plus lenalidomide in relapsed or refractory diffuse large B-cell lymphoma (L-MIND): a multicentre, prospective, single-arm, phase 2 study. Lancet Oncol. 2020; 21:978–88. https://doi.org/10.1016/S1470-2045(20)30225-4. [PubMed].
16. Caimi PF, Ai W, Alderuccio JP, Ardeshna KM, Hamadani M, Hess B, Kahl BS, Radford J, Solh M, Stathis A, Zinzani PL, Havenith K, Feingold J, et al. Loncastuximab tesirine in relapsed or refractory diffuse large B-cell lymphoma (LOTIS-2): a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2021; 22:790–800. https://doi.org/10.1016/S1470-2045(21)00139-X. [PubMed].
17. Sehn LH, Herrera AF, Flowers CR, Kamdar MK, McMillan A, Hertzberg M, Assouline S, Kim TM, Kim WS, Ozcan M, Hirata J, Penuel E, Paulson JN, et al. Polatuzumab Vedotin in Relapsed or Refractory Diffuse Large B-Cell Lymphoma. J Clin Oncol. 2020; 38:155–65. https://doi.org/10.1200/JCO.19.00172. [PubMed].
18. Wang ML, Barrientos JC, Furman RR, Mei M, Barr PM, Choi MY, de Vos S, Kallam A, Patel K, Kipps TJ, Rule S, Flanders KB, Jessen KA, et al. Zilovertamab Vedotin Targeting of ROR1 as Therapy for Lymphoid Cancers. NEJM Evidence. 2022; 1. https://doi.org/10.1056/EVIDoa2100001.
19. Strati P, De Vos S, Ruan J, Maddocks KJ, Flowers CR, Rule S, Patel P, Xu Y, Wei H, Frigault MM, Calvo R, Dyer MJS. Acalabrutinib for treatment of diffuse large B-cell lymphoma: results from a phase Ib study. Haematologica. 2021; 106:2774–78. https://doi.org/10.3324/haematol.2021.278654. [PubMed].
20. Chen R, Chen Y, Xiong P, Zheleva D, Blake D, Keating MJ, Wierda WG, Plunkett W. Cyclin-dependent kinase inhibitor fadraciclib (CYC065) depletes anti-apoptotic protein and synergizes with venetoclax in primary chronic lymphocytic leukemia cells. Leukemia. 2022; 36:1596–608. https://doi.org/10.1038/s41375-022-01553-w. [PubMed].

