Introduction
Early detection and treatment of solid tumor malignancies has remained a major healthcare challenge. Identifying new tumor biomarkers, such as tumor associated antigens (TAAs), for diagnosis and as tumor targets for effective immunotherapies are critical needs. Antibody-based and cell-based immunotherapies (CAR-T cell therapy) targeting TAAs (e.g., CD19, Her-2, and CD52) have helped target blood cancers [1]. CD52 and Her-2 are both examples of TAAs that are often upregulated in breast cancer and can thus be targeted by antibody-drug conjugates, which help deliver cytotoxic drugs to cancer cells only [2]. Additional biomarkers on blood cancers and solid tumors will help expand this treatment repertoire to a variety of cancers arising from different tissues.
The LY6 genes on chromosome 8q24.3 are of growing interest as this LY6 locus is frequently amplified in human cancer [3]. Genes located at this locus include LY6E, LY6L, LY6D, LY6K, LY6H, SLURP1, LYPD2, LYNX1, GML, and GPIHBP1; these genes are syntenic to mouse chromosome 15. In total, the LY6 gene family is comprised of at least 26 members, which are located on chromosomes 6, 11, and 19, in addition to chromosome 8 [4, 5]. Transcriptome analysis of pancreatic tumors has revealed upregulated expression of many LY6 genes when compared to normal pancreatic tissue [3]. These findings are consistent with previous reports that show an increased expression of PSCA and other LY6 genes (e.g., SLURP1) on a variety of neoplasms arising from prostate, bladder, ovarian, urothelial, and skin tissues [6–11]. Regardless of their chromosomal location, the LY6 proteins are either glycosylphosphatidylinositol (GPI)-anchored to the membrane or are secreted [4]. A common feature present in all these proteins is the Ly-6/uPAR (LU) domain, which consists of 6–10 conserved cysteine residues [12]. These cysteine residues are arranged in specific spacing patterns that allow for disulfide bridge formation. The observed tertiary structure is a three-finger structural motif, which was first reported in the neurotoxin protein family [13]. Mouse LY6 proteins expressed on immune and non-immune cells are reported to possess cell adhesion roles [14–21]. The functions of mouse Ly-6 orthologs in humans are observed in neuronal and other tissues where LY6 proteins regulate nicotinic acetylcholine receptor [22]. Recent studies examining the function of LY6 genes on human chromosome 8 have shown these genes to serve as biomarkers of poor cancer prognosis, and other studies have found them to be involved in cancer progression and immune escape [23, 24]. We report bioinformatic observations concerning upregulated expression and amplification of many LY6 genes and their association with poor cancer patient survival in uterine corpus endometrial carcinoma (UCEC). Importantly, the expression of several LY6 genes is elevated in UCEC when compared to the expression in normal uterine tissue.
Results
Human LY6 gene expression in normal and tumor tissues
LY6 genes are expressed in a variety of normal, non-lymphoid tissues (Table 1). According to the GTEx Portal, tissues that normally express multiple LY6 genes include the brain, esophagus, skin, and testis. In human tumors, expression of LY6D, LY6E, LY6H, and LY6K genes, which share the Ly-6/uPAR domain (Supplementary Figure 1), is significantly upregulated compared to normal tissues, and this is true for ovarian, colorectal, gastric, breast, lung, bladder, brain, cervical, esophageal, head and neck, and pancreatic tumors. For ovarian, colorectal, gastric, and breast cancers, this elevated expression is associated with poor overall patient survival [23].
Table 1: Human LY6 Gene Family
| LY6 Member | UniProt ID | Chromosome | Cell surface (CS) or Secreted (S) | Normal tissue expression (From Gtex) | Function* |
|---|---|---|---|---|---|
| LY6E | Q16553 | 8 | CS | Widely expressed. Highest in cervix, lung, ovary, liver, breast, and uterus (150–200 TPM) | Regulates T-cell proliferation, differentiation, and activation. May be involved in cancer metastasis. Possible modulator of nicotinic acetylcholine receptors. Main receptor for syncytin-A during placenta formation [41–45]. |
| LY6L | H3BQJ8 | 8 | CS | Highest in kidney, testis, and prostate (<1 TPM) | Function is inferred from homology. An important paralog for LY6L is LY6H. |
| LY6D | Q14210 | 8 | CS | Highest in esophagus and skin (>1000 TPM), vagina (500 TPM) | Possible specification marker at earliest specification stage of lymphocytes between B- and T-cell development [46]. |
| LY6K | Q17RY6 | 8 | CS | Highest in testis, esophagus, and skin (<50 TPM) | Potential role in cell growth. Required for sperm migration into the oviduct and male fertility by controlling binding of sperm to zona pellucida [47, 48]. |
| LY6H | O94772 | 8 | CS | Primarily expressed in brain (>200 TPM) | Possible modulator of nicotinic acetylcholine receptors activity. Seems to inhibit alpha-7/CHRNA7 signaling in hippocampal neurons [41, 49]. |
| SLURP1 | P55000 | 8 | S | Primarily expressed in esophagus and skin (>500 TPM), vagina (>100 TPM) | Displays antitumor activity. Late differentiation marker in skin. Possible modulator of nicotinic acetylcholine receptors. Possible regulator of intracellular Ca2+ signaling in T cells [50–56]. |
| LYPD1 | Q8N2G4 | 2 | CS | Highest in brain (<50 TPM) | Possible modulator of nicotinic acetylcholine receptor activity [41, 49, 57]. |
| LYPD2 | Q6UXB3 | 8 | CS | Highest in esophagus (>250 TPM), skin and vagina (<50 TPM) | No known or proposed function available. |
| LYPD3 | O95274 | 19 | CS | Highest in esophagus and skin (>1000 TPM), vagina (>500 TPM) | Supports cell migration. May be involved in tumor progression [58–61]. |
| LYPD4 | Q6UWN0 | 19 | CS | Only in testis (>200 TPM) | No known or proposed function available. |
| LYPD5 | Q6UWN5 | 19 | CS | Highest in skin (<50 TPM), brain and esophagus (<10 TPM) | No known or proposed function available. |
| LYPD6 | Q86Y78 | 2 | Both | Highest in testis, brain, uterus, and bladder (<10 TPM) | Modulator of nicotinic acetylcholine receptor function in the brain [62]. |
| LYPD6B | Q8NI32 | 2 | CS | Highest in skin, testis, and stomach (<50 TPM) | Proposed modulator of nicotinic acetylcholine receptor activity [63]. |
| LYPD8 | Q6UX82 | 1 | Both | Highest in colon and small intestine (<50 TPM) | Secreted form prevents invasion of Gram-negative bacteria in the inner mucus layer of colon epithelium [64]. |
| LYPD9P | NA | 1 | NA | NA | Pseudogene |
| LYNX1 | P0DP58 | 8 | CS | Widely expressed. Highest in brain and heart (>50 TPM) | Interacts with nicotinic acetylcholine receptors [65]. |
| CD59 | P13987 | 11 | Both | Widely expressed. Highest in lung and breast (>500 TPM) | Involved in signal transduction for T-cell activation complexed to a protein tyrosine kinase [46]. |
| GML | Q99445 | 8 | CS | Expressed in testis and adrenal gland (<5 TPM) | Possible role in apoptosis or cell-cycle regulation. Induced by p53 after DNA damage [66]. |
| GPIHBP1 | Q8IV16 | 8 | CS | Widely expressed. Highest in breast and brain (>50 TPM) | Mediates transport of lipoprotein lipase from the basolateral to the apical surface of endothelial cells in capillaries [67–71]. |
| LY6G5B | Q8NDX9 | 6 | S | Widely expressed. Highest in brain, skin, ovary, cervix, uterus, and spleen (>50 TPM) | No known or proposed function available. |
| LY6G5C | Q5SRR4 | 6 | S | Highest in testis and brain (<50 TPM) | Possible role in hematopoietic cell differentiation [72]. |
| LY6G6C | O95867 | 6 | CS | Highest in skin (>500 TPM) | No known or proposed function available. |
| LY6G6D | O95868 | 6 | CS | Highest in testis and colon (<10 TPM) | Potential acetylcholine receptor inhibitor activity [46]. |
| LY6G6F | Q5SQ64 | 6 | CS | Highest in whole blood and testis (<10 TPM) | Potential role in downstream signal transduction pathways involving GRB2 and GRB7 [73]. |
| PLAUR | Q03405 | 19 | Isoform1: CS, Isoform2: S | Highest in whole blood and lung (>50 TPM) | Receptor for urokinase plasminogen activator and has role in localizing and promoting plasmin formation [74]. |
| PSCA | O43653 | 8 | CS | Highest in stomach (>1000 TPM) | Possibly involved in regulation of cell proliferation. Displays cell-proliferation inhibition activity in vitro [75, 76]. |
Analyzing LY6 gene expression in a tumor may provide insight into a patient’s probability of survival or potential to respond to a certain therapy. Many LY6 genes have recently garnered attention for their potential role as biomarkers of poor patient prognosis in pancreatic ductal adenocarcinoma [3]. LY6K is especially of interest as high mRNA expression of this gene is associated with poor patient survival in thyroid, kidney, uterine, and esophageal carcinomas [25]. Based on the results from these survival studies, we hypothesize that other LY6 genes may also serve as biomarkers of poor prognosis in different cancers. To explore this, we analyzed RNA-seq data from The Cancer Genome Atlas (TCGA) for 20 different cancers, separated patients into high and low expression groups for each LY6 gene, and compared overall survival between the two groups. We also compared LY6 gene family expression in normal tissue to expression in tumor tissue to determine if LY6 gene expression is upregulated in a given cancer. Highly upregulated LY6 genes in cancer may allow for their detection. Additionally, LY6 proteins on solid tumors may serve as targets for antibody and/or CAR-T cell therapies.
Human LY6 genes are biomarkers of poor prognosis and are upregulated in UCEC
Pan-cancer analysis of LY6 gene expression revealed a negative correlation between mRNA expression and overall survival for most LY6 genes in UCEC patients (Figure 1). In UCEC, there was a significant difference in survival between high and low expression groups for all LY6 genes, except for PSCA and LYPD1. Of the 23 LY6 genes for which there were significant differences in survival between high and low expression groups, 19 of these genes were associated with poor overall survival in high expression groups. CD59, LYPD5, PLAUR, and LY6G5C were all associated with increased survival in high expression groups. Negative and positive correlations between mRNA expression and overall survival were observed in other cancers as well. However, a focus was placed on UCEC since its LY6 gene expression pattern was most similar to that of pancreatic ductal adenocarcinoma, which has already been described [3].
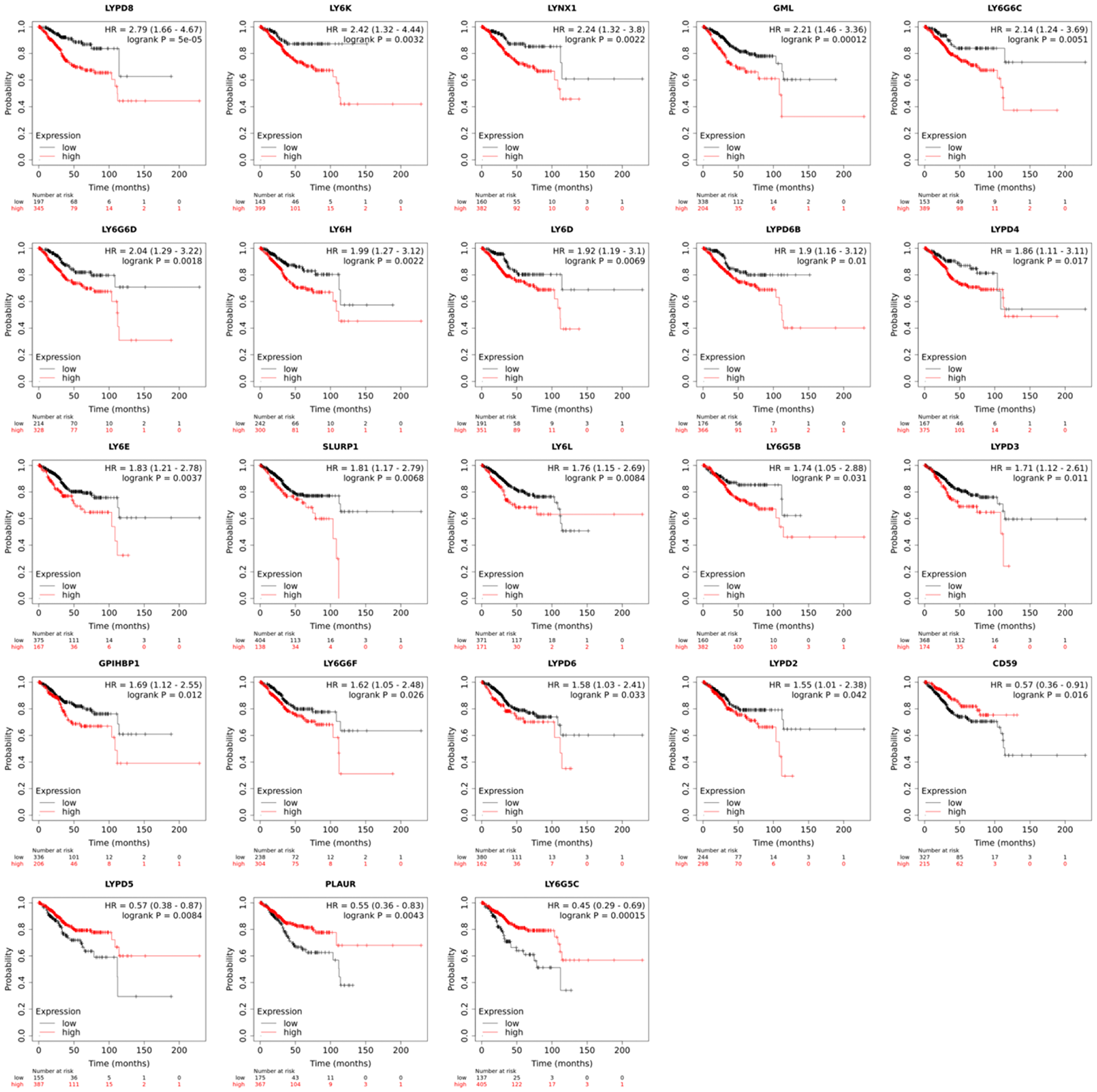
Figure 1: Overall patient survival in uterine corpus endometrial carcinoma (n = 543) based on high and low mRNA expression of a given human LY6 gene. The red line represents the overall survival of patients with high expression of that gene, and the black line represents the overall survival of patients with low expression of that gene. A Cox proportional hazards model was used to determine if differences in survival between high and low expression groups were significant. RNA-seq data for UCEC was downloaded from TCGA, and overall survival was plotted using KM plotter.
Analysis of LY6 gene expression in normal uterine tissue compared to UCEC revealed that mRNA expression of several LY6 genes is upregulated in UCEC (Figure 2). mRNA expression of LYPD1, LYPD6B, LY6K, PSCA, LY6D, LYPD3, PLAUR, LY6E, SLURP1, LYPD6, and LY6G5C is significantly elevated in UCEC. mRNA expression of CD59, LY6H, LYNX1 and LY6G5B is significantly reduced in UCEC. There is no significant change in mRNA expression for LYPD8, LY6G6D, LYPD4, LY6L, LYPD2, LYPD5, LY6G6F, LYPD4, GPIHBP1, and GML.
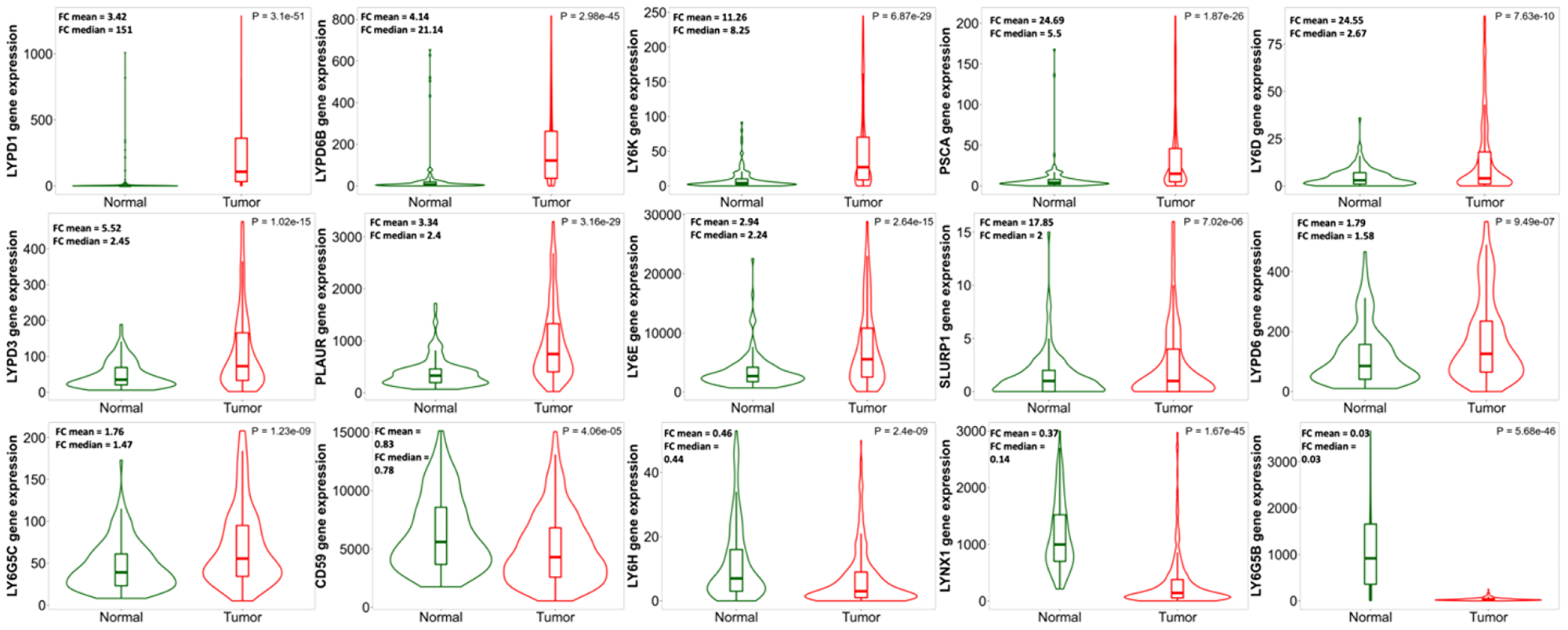
Figure 2: Comparison of human LY6 gene expression in normal uterine tissue (n = 146) to expression in uterine corpus endometrial carcinoma (n = 547). Violin plots were generated using TNMplot, and fold changes in mean and median expression values were calculated for each gene. Many LY6 genes show some degree of upregulation in UCEC with the exception of CD59, LY6H, LYNX1, and LY6G5B, which are downregulated. LY6 genes not differentially expressed between normal and UCEC tissues are not shown.
LY6 gene amplification in type I and II UCEC patients
The 8q24.3 locus contains several LY6 genes and is frequently amplified in cancer with the reason not being understood. We observed increased amplification of this locus in UCEC compared to normal tissue. However, it was not previously known if 8q24.33 amplification is associated with more severe subsets of UCEC. To explore this, we separated UCEC patients based on their cancer type: uterine endometroid carcinoma (type I) and uterine serous carcinoma (type II) and compared amplification frequencies for each LY6 gene. Type I tumors are estrogen driven and are associated with better prognosis whereas type II tumors are more aggressive and frequently carry genetic alterations in p53 and human epidermal growth factor-2 (HER-2) [26]. Of the UCEC patients represented in the TCGA pan cancer database, type I accounts for approximately 75% of cases, and type II accounts for approximately 20% of cases. A “mixed” type comprises the remaining 5% of cases [27, 28]. From our analyses, we found 8q24.3 amplification to be ~4× more prevalent in type II UCEC, and we also identified two additional LY6-containing loci that show increased amplification in this cancer type: 6p21.33 and 19q13.31 (Table 2).
Table 2: LY6 gene amplification frequencies in type I and II UCEC patients
| Gene | Locus | % Patients with gene amplification | P-value | ||
|---|---|---|---|---|---|
| Uterine endometroid carcinoma (n = 394) | Uterine serous carcinoma (n = 108) | Total (n = 502) | |||
| LY6L | 8q24.3 | 0 | 0 | 0.00 | >0.9999 |
| LYPD1 | 2q21.2 | 0 | 0.93 | 0.20 | 0.2151 |
| LYPD6B | 2q23.2 | 0 | 0.93 | 0.20 | 0.2151 |
| LYPD6 | 2q23.2 | 0 | 0.93 | 0.20 | 0.2151 |
| CD59 | 11p13 | 0.25 | 0.93 | 0.40 | 0.3843 |
| LYPD4 | 19q13.2 | 0 | 2.78 | 0.60 | 0.0097 |
| LYPD3 | 19q13.31 | 0 | 3.70 | 0.80 | 0.0020 |
| PLAUR | 19q13.31 | 0 | 3.70 | 0.80 | 0.0020 |
| LYPD5 | 19q13.31 | 0 | 3.70 | 0.80 | 0.0020 |
| LY6G6D | 6p21.33 | 0.76 | 3.70 | 1.39 | 0.0416 |
| LY6G5C | 6p21.33 | 0.76 | 3.70 | 1.39 | 0.0416 |
| LY6G6C | 6p21.33 | 0.76 | 3.70 | 1.39 | 0.0416 |
| LY6G5B | 6p21.33 | 0.76 | 3.70 | 1.39 | 0.0416 |
| LY6G6F | 6p21.33 | 0.76 | 3.70 | 1.39 | 0.0416 |
| LYPD8 | 1q44 | 1.78 | 3.70 | 2.19 | 0.2616 |
| GML | 8q24.3 | 2.03 | 6.48 | 2.99 | 0.0246 |
| LYPD2 | 8q24.3 | 2.03 | 6.48 | 2.99 | 0.0246 |
| LYNX1 | 8q24.3 | 2.03 | 6.48 | 2.99 | 0.0246 |
| LY6D | 8q24.3 | 2.03 | 6.48 | 2.99 | 0.0246 |
| LY6E | 8q24.3 | 1.78 | 7.41 | 2.99 | 0.0060 |
| PSCA | 8q24.3 | 1.78 | 7.41 | 2.99 | 0.0060 |
| SLURP1 | 8q24.3 | 2.03 | 6.48 | 2.99 | 0.0246 |
| LY6K | 8q24.3 | 2.03 | 7.41 | 3.19 | 0.0099 |
| GPIHBP1 | 8q24.3 | 2.03 | 8.33 | 3.39 | 0.0037 |
| LY6H | 8q24.3 | 2.03 | 8.33 | 3.39 | 0.0037 |
LY6 gene regulation in humans
Mouse Ly-6A/E protein expression is induced by type I (IFN-α/β) and type II (IFN-γ) interferons, which activate interferon regulatory factors (IRF) such as IRF9. IRFs activate expression by binding to cis-active interferon-sensitive response elements (ISRE) within distal enhancers of the mouse Ly-6A/E genes [29, 30]. To determine if human LY6 gene family expression is regulated by type I IFNs and mediated by IRF9, ChIP-seq data were analyzed to identify IRF9 binding sites within distal enhancer elements of LY6 genes. Bioinformatic tools were also used to infer ISRE-containing enhancers that possibly bind IRF9 and regulate LY6 genes. The results from the ChIP-seq and GeneHancer data analyses are shown in Table 3. The publicly available ChIP-seq data from Qiagen and SPP did not return any IRF9 binding sites within the promoters or enhancers that regulate LY6 gene family expression. However, GeneHancer was able to predict several distal enhancers that contain putative IRF9 binding sites and potentially regulate expression of the following LY6 genes: LY6E, LY6L, LYPD8, CD59, GPIHBP1, LY6G5B, LY6G5C, LY6G6C, and LY6G6D.
Table 3: Human LY6 gene regulation
| LY6 gene | Top TFs sites in gene promoter from Qiagen | Potential IRF9 binding site and distance from TSS (kb) from GeneHancer |
|---|---|---|
| LY6E | c-Rel, C/EBPα, En-1, IRF-1, LCR-F1, Lmo2, LUN-1, NF-κB, NF-κB1, TBP | Yes, +0.4 |
| LY6L | NA | Yes, −62.3 |
| LY6D | E2F, E2F-1, E2F-2, E2F-3a, E2F-4, E2f-5, HNF-4α1, HNF-4α2, LCR-F1, LUN-1 | No |
| LY6K | c-Myc, FOXD1, FOXO4, GR, GR-α, GR-β, Max, USF-1 | No |
| LY6H | HEN1, Olf-1, POU2F1, POU2F1a | No |
| SLURP1 | AP-2γ, C/EBPα, GR, GR-α, GR-β, ITF-2, Nkx2-5, p53, Tal-1β | No |
| LYPD1 | CUTL1, Evi-1, HTF, p53, SRF | No |
| LYPD2 | AP-2γ, c-Fos, c-Jun, C/EBPα, GR, GR-α, GR-β, NF-κB1, Nkx2-5, p53 | No |
| LYPD3 | AP-1, ATF-2, c-Jun, Sp1 | No |
| LYPD4 | GR, GR-α, GR-β, p53, PPAR-α | No |
| LYPD5 | AP-1, ATF-2, c-Jun, HOXA5, LUN-1, Meis-1b, p53, POU2F1, POU2F1a, SEF-1 | No |
| LYPD6 | AML1a, Egr-2, GATA-3, POU2F1, POU2F1a, YY1 | No |
| LYPD6B | AREB6, CUTL1, E2F-1, E47, FOXD3, FOXO3a, HOXA3, Pax-4a, Tal-1β, YY1 | No |
| LYPD8 | NA | Yes, −69.2 |
| LYPD9P | NA | NA |
| LYNX1 | E2F, E2F-1, E2F-2, E2F-5 | No |
| CD59 | GR | Yes, +14.2 |
| GML | ER-α, Nkx3-1, Nkx3-1 v1/2/2/4, p53, Roaz | No |
| GPIHBP1 | aMEF-2, C/EBPα, CHOP-10, GATA-2, Ik-3, Lmo2, MEF-2A, NF-1, NF-1/L, Pax-5 | Yes, +6.5 |
| LY6G5B | E47, Hand1, HNF-4α1, HNF-4α2, HTF, Pax-5, PPAR-γ1/2 | Yes, +156.1, +67.3, +58.4, −761.7, −949.7 |
| LY6G5C | AML1a, HSF2, LCR-F1, MRF-2, POU2F1a, PPAR-γ1/2, SRF, XBP-1 | Yes, −53.4 |
| LY6G6C | NF-κB1, p53, Sp1 | Yes, −104.5, −15.7 |
| LY6G6D | C/EBPα, CHOP-10, ITF-2, MRF-2, NF-κB1, PPAR-γ1, RFX1, Sp1, TaI-1β | Yes, +22.1, +110.9 |
| LY6G6F | ITF-2, MRF-2, NF-κB1, PPAR-γ1/2, RFX1, Sp1, TaI-1β | No |
| PLAUR | Sp1, STAT1, STAT3 | No |
| PSCA | AML1a, AREB6, c-Ets-1, FOXJ2, GATA-1/2/3, RREB-1, ZID | No |
DISCUSSION
We carried out in silico analyses of all reported LY6 genes, focused on their expression in different cancers, and analyzed patient survival by mining the TCGA database. We report that upregulated expression of many LY6 genes is associated with poor cancer patient survival in uterine corpus endometrial carcinoma (UCEC). The overall survival data confirms that many upregulated human LY6 gene products may serve as biomarkers for UCEC detection and may be useful in identifying high-risk UCEC patients. High expression of LY6 proteins on the surface of tumor cells also makes them potential targets for cell-based and antibody-based immunotherapies. However, the magnitude of tumor expression above normal expression is critical to avoid autoimmunity and prevent targeting of self-tissues. Our mined transcriptomic information indicates that LY6K mRNA is significantly upregulated (>8 fold) in UCEC patient tumor tissues. If this mRNA is being translated to yield high levels of LY6K on the surface of uterine tumor cells, then cell-based therapies against LY6K might be able to selectively target and kill these cancer cells. In addition to these findings, we also report that a patient’s LY6 gene amplification status may provide an alternative method for classifying type I and type II UCEC. Although rare, amplification of loci 8q24.3, 6p21.33, and 19q13.31 is more prevalent in type II UCEC and knowing a patient’s amplification status of these loci may help predict their likelihood of developing severe disease. Further analysis is needed, though, to determine if the LY6 proteins encoded within these loci are involved in the development of the severe disease and poor outcomes associated with type II UCEC.
Transcriptional regulation of LY6 genes is not well understood and has not been heavily investigated. Identifying the transcription factors that regulate LY6 gene expression will help uncover the signaling pathways used by cancer cells and T cells to upregulate surface expression of LY6 proteins. Expression of mouse Ly-6A/E is induced by type I (IFN-α/β) and type II (IFN-γ) interferons, but this has not been confirmed in human cell lines [29]. Interferon signaling is mediated by various IRFs, and many mouse Ly-6 gene enhancers contain cis-active ISREs [30]. IRF9 is a transcription factor that is activated by type I IFN signaling and binds to ISREs within distal enhancer elements of interferon-stimulated genes (ISG) [31]. A majority of LY6 genes contain regulatory sequences that can potentially bind IRF9 as well as an array of other transcription factors and activators (Table 3). The role of interferon responsive factors (e.g., IRF9), transcription factors, and other activators in upregulating the expression of LY6 genes appears complex. Their interdependence, cause and effect relationship, or lack of, will require considerable experimental work including ChIP-seq and RT-qPCR analyses. Our in-silico analyses did not discover any common potential transacting factor binding sites within the LY6 genes reported to be upregulated in UCEC patient tissues (Figure 2 and Table 3). Another future consideration is to understand the uterine tumor microenvironment, especially to delineate the expression of LY6 proteins on the surface of tumor subpopulations and/or tumor-infiltrating lymphocytes. Multiplex immunohistochemistry would be a good technique to analyze the expression of LY6 proteins on the surfaces of both cell types to assess their contributions to overall expression.
Further analysis of tumor-specific expression of the LY6 gene family is needed to uncover the function of LY6 proteins as well as the signaling pathways that these proteins trigger to endow tumor survival and poor prognosis in UCEC patients. A possible explanation, which would need to be tested, is that the ligands or receptors for LY6 proteins are expressed in the uterine tumor microenvironment and drive tumor progression through binding that specific upregulated member of the LY6 family. Further analysis of patient tumors is needed to uncover the functions of human LY6 proteins as well as the signaling pathways that these proteins trigger to endow tumor survival and poor patient prognosis in other cancers as well. While upregulated expression of some LY6 genes suggested poor patient prognosis, there are four LY6 genes that showed the opposite, which was unexpected. High expression of CD59, LYPD5, PLAUR, and LY6G5C is associated with better patient outcome (Figure 1). Further analysis of patient tumors is needed to uncover the signaling pathways that these proteins trigger to endow beneficial UCEC patient prognosis.
Materials and Methods
Analysis of human LY6 gene expression and amplification in cancer
RNA-seq data for 20 different cancers were obtained from TCGA [27, 28]. Kaplan-Meier Plotter was used to separate patients into high and low expression groups for each LY6 gene and then plot overall survival for each group [32, 33]. A proportional hazards model was used to calculate hazard ratios and p-values for each plot. GTEx Portal provided the top tissues in which LY6 genes are normally expressed [34]. TNMplot was used to generate violin plots and compare LY6 gene family expression in normal tissue to expression in tumor tissue [35]. Fold changes in expression were calculated using the median and mean expression values, and statistical significances were calculated using a Mann Whitney U test. cBioPortal was used to compare the amplification frequencies of LY6 genes in type I and type II UCEC patients [36, 37]. Differences in amplification frequency were compared using a Fisher’s Exact test. The mixed UCEC group (n = 21) was not included in this analysis.
Analysis of LY6 gene regulation in humans
Top TFs in LY6 gene promoters were provided by Qiagen and GeneCards [38]. Experimental ChIP-seq data for the LY6 gene family were downloaded from The Signaling Pathways Project (SPP) and GeneHancer was used to predict distal enhancers that regulate human LY6 genes [39, 40].
Abbreviations
LY6: Lymphocyte antigen-6; UCEC: uterine corpus endometrial carcinoma; TAA: tumor associated antigens; LU: Ly-6/uPAR domain; GPI: glycosylphosphatidyl-inositol; TCGA: The Cancer Genome Atlas; IFN: interferon; ISRE: interferon-sensitive response elements; ISG: interferon-stimulated gene; IRF: interferon regulatory factor.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
FUNDING
This work was supported by SRFG and SRG grants from Office of Research and Sponsored Projects (ORSP), Villanova University and Department of Biology, Villanova University to AKB. This work was also supported by Villanova University Department of Biology and Biochemistry Program to LAR.
References
1. Wang K, Wei G, Liu D. CD19: a biomarker for B cell development, lymphoma diagnosis and therapy. Exp Hematol Oncol. 2012; 1:36. https://doi.org/10.1186/2162-3619-1-36. [PubMed].
2. Ferraro E, Drago JZ, Modi S. Implementing antibody-drug conjugates (ADCs) in HER2-positive breast cancer: state of the art and future directions. Breast Cancer Res. 2021; 23:84. https://doi.org/10.1186/s13058-021-01459-y. [PubMed].
3. Russ E, Bhuvaneshwar K, Wang G, Jin B, Gage MM, Madhavan S, Gusev Y, Upadhyay G. High mRNA expression of LY6 gene family is associated with overall survival outcome in pancreatic ductal adenocarcinoma. Oncotarget. 2021; 12:145–59. https://doi.org/10.18632/oncotarget.27880. [PubMed].
4. Bamezai A. Mouse Ly-6 proteins and their extended family: markers of cell differentiation and regulators of cell signaling. Arch Immunol Ther Exp (Warsz). 2004; 52:255–66. [PubMed].
5. Loughner CL, Bruford EA, McAndrews MS, Delp EE, Swamynathan S, Swamynathan SK. Organization, evolution and functions of the human and mouse Ly6/uPAR family genes. Hum Genomics. 2016; 10:10. https://doi.org/10.1186/s40246-016-0074-2. [PubMed].
6. Amara N, Palapattu GS, Schrage M, Gu Z, Thomas GV, Dorey F, Said J, Reiter RE. Prostate stem cell antigen is overexpressed in human transitional cell carcinoma. Cancer Res. 2001; 61:4660–65. [PubMed].
7. Argani P, Rosty C, Reiter RE, Wilentz RE, Murugesan SR, Leach SD, Ryu B, Skinner HG, Goggins M, Jaffee EM, Yeo CJ, Cameron JL, Kern SE, Hruban RH. Discovery of new markers of cancer through serial analysis of gene expression: prostate stem cell antigen is overexpressed in pancreatic adenocarcinoma. Cancer Res. 2001; 61:4320–24. [PubMed].
8. Cao D, Ji H, Ronnett BM. Expression of mesothelin, fascin, and prostate stem cell antigen in primary ovarian mucinous tumors and their utility in differentiating primary ovarian mucinous tumors from metastatic pancreatic mucinous carcinomas in the ovary. Int J Gynecol Pathol. 2005; 24:67–72. [PubMed].
9. Cheng L, Reiter RE, Jin Y, Sharon H, Wieder J, Lane TF, Rao J. Immunocytochemical analysis of prostate stem cell antigen as adjunct marker for detection of urothelial transitional cell carcinoma in voided urine specimens. J Urol. 2003; 169:2094–100. https://doi.org/10.1097/01.ju.0000064929.43602.17. [PubMed].
10. Bergqvist C, Kadara H, Hamie L, Nemer G, Safi R, Karouni M, Marrouche N, Abbas O, Hasbani DJ, Kibbi AG, Nassar D, Shimomura Y, Kurban M. SLURP-1 is mutated in Mal de Meleda, a potential molecular signature for melanoma and a putative squamous lineage tumor suppressor gene. Int J Dermatol. 2018; 57:162–70. https://doi.org/10.1111/ijd.13850. [PubMed].
11. Arousse A, Mokni S, H’mida Ben Brahim D, Bdioui A, Aounallah A, Gammoudi R, Saidi W, Boussofara L, Ghariani N, Denguezli M, Belajouza C, Nouira R. Amelanotic melanoma arising in an area of SLURP-1 mutated Mal de Meleda. Int J Dermatol. 2019; 58:966–68. https://doi.org/10.1111/ijd.14231. [PubMed].
12. Bamezai AK, Miwa JM. Editorial: Biology of Ly-6 Supergene Family in Health and Disease. Front Cell Dev Biol. 2022; 10:949379. https://doi.org/10.3389/fcell.2022.949379. [PubMed].
13. Tsetlin V. Snake venom alpha-neurotoxins and other ‘three-finger’ proteins. Eur J Biochem. 1999; 264:281–86. https://doi.org/10.1046/j.1432-1327.1999.00623.x. [PubMed].
14. Bamezai A, Rock KL. Overexpressed Ly-6A.2 mediates cell-cell adhesion by binding a ligand expressed on lymphoid cells. Proc Natl Acad Sci U S A. 1995; 92:4294–98. https://doi.org/10.1073/pnas.92.10.4294. [PubMed].
15. Rock KL, Yeh ET, Gramm CF, Haber SI, Reiser H, Benacerraf B. TAP, a novel T cell-activating protein involved in the stimulation of MHC-restricted T lymphocytes. J Exp Med. 1986; 163:315–33. https://doi.org/10.1084/jem.163.2.315. [PubMed].
16. Malek TR, Ortega G, Chan C, Kroczek RA, Shevach EM. Role of Ly-6 in lymphocyte activation. II. Induction of T cell activation by monoclonal anti-Ly-6 antibodies. J Exp Med. 1986; 164:709–22. https://doi.org/10.1084/jem.164.3.709. [PubMed].
17. Stanford WL, Haque S, Alexander R, Liu X, Latour AM, Snodgrass HR, Koller BH, Flood PM. Altered proliferative response by T lymphocytes of Ly-6A (Sca-1) null mice. J Exp Med. 1997; 186:705–17. https://doi.org/10.1084/jem.186.5.705. [PubMed].
18. Henderson SC, Kamdar MM, Bamezai A. Ly-6A.2 expression regulates antigen-specific CD4+ T cell proliferation and cytokine production. J Immunol. 2002; 168:118–26. https://doi.org/10.4049/jimmunol.168.1.118. [PubMed].
19. Choi SJ, Devlin RD, Menaa C, Chung H, Roodman GD, Reddy SV. Cloning and identification of human Sca as a novel inhibitor of osteoclast formation and bone resorption. J Clin Invest. 1998; 102:1360–68. https://doi.org/10.1172/JCI2667. [PubMed].
20. Kitazawa H, Nishihara T, Nambu T, Nishizawa H, Iwaki M, Fukuhara A, Kitamura T, Matsuda M, Shimomura I. Intectin, a novel small intestine-specific glycosylphosphatidylinositol-anchored protein, accelerates apoptosis of intestinal epithelial cells. J Biol Chem. 2004; 279:42867–74. https://doi.org/10.1074/jbc.M408047200. [PubMed].
21. Miwa JM, Ibanez-Tallon I, Crabtree GW, Sánchez R, Sali A, Role LW, Heintz N. lynx1, an endogenous toxin-like modulator of nicotinic acetylcholine receptors in the mammalian CNS. Neuron. 1999; 23:105–14. https://doi.org/10.1016/s0896-6273(00)80757-6. [PubMed].
22. Bychkov ML, Shulepko MA, Shlepova OV, Kulbatskii DS, Chulina IA, Paramonov AS, Baidakova LK, Azev VN, Koshelev SG, Kirpichnikov MP, Shenkarev ZO, Lyukmanova EN. SLURP-1 Controls Growth and Migration of Lung Adenocarcinoma Cells, Forming a Complex With α7-nAChR and PDGFR/EGFR Heterodimer. Front Cell Dev Biol. 2021; 9:739391. https://doi.org/10.3389/fcell.2021.739391. [PubMed].
23. Upadhyay G. Emerging Role of Lymphocyte Antigen-6 Family of Genes in Cancer and Immune Cells. Front Immunol. 2019; 10:819. https://doi.org/10.3389/fimmu.2019.00819. [PubMed].
24. AlHossiny M, Luo L, Frazier WR, Steiner N, Gusev Y, Kallakury B, Glasgow E, Creswell K, Madhavan S, Kumar R, Upadhyay G. Ly6E/K Signaling to TGFβ Promotes Breast Cancer Progression, Immune Escape, and Drug Resistance. Cancer Res. 2016; 76:3376–86. https://doi.org/10.1158/0008-5472.CAN-15-2654. [PubMed].
25. Guo D, Liu Y, Jiang Y, Zheng S, Xu T, Zhu J, Chen P, Huang P, Zhang Y. A narrative review of the emerging role of lymphocyte antigen 6 complex locus K in cancer: from basic research to clinical practice. Ann Transl Med. 2022; 10:26. https://doi.org/10.21037/atm-21-5831. [PubMed].
26. Lobo FD, Thomas E. Type II endometrial cancers: A case series. J Midlife Health. 2016; 7:69–72. https://doi.org/10.4103/0976-7800.185335. [PubMed].
27. Hoadley KA, Yau C, Hinoue T, Wolf DM, Lazar AJ, Drill E, Shen R, Taylor AM, Cherniack AD, Thorsson V, Akbani R, Bowlby R, Wong CK, et al. Cell-of-Origin Patterns Dominate the Molecular Classification of 10,000 Tumors from 33 Types of Cancer. Cell. 2018; 173:291–304.e6. https://doi.org/10.1016/j.cell.2018.03.022. [PubMed].
28. Liu J, Lichtenberg T, Hoadley KA, Poisson LM, Lazar AJ, Cherniack AD, Kovatich AJ, Benz CC, Levine DA, Lee AV, Omberg L, Wolf DM, Shriver CD, et al. An Integrated TCGA Pan-Cancer Clinical Data Resource to Drive High-Quality Survival Outcome Analytics. Cell. 2018; 173:400–16.e11. https://doi.org/10.1016/j.cell.2018.02.052. [PubMed].
29. Khan KD, Shuai K, Lindwall G, Maher SE, Darnell JE Jr, Bothwell AL. Induction of the Ly-6A/E gene by interferon alpha/beta and gamma requires a DNA element to which a tyrosine-phosphorylated 91-kDa protein binds. Proc Natl Acad Sci U S A. 1993; 90:6806–10. https://doi.org/10.1073/pnas.90.14.6806. [PubMed].
30. Khodadoust MM, Khan KD, Bothwell AL. Complex regulation of Ly-6E gene transcription in T cells by IFNs. J Immunol. 1999; 163:811–19. https://doi.org/10.4049/jimmunol.163.2.811. [PubMed].
31. Paul A, Tang TH, Ng SK. Interferon Regulatory Factor 9 Structure and Regulation. Front Immunol. 2018; 9:1831. https://doi.org/10.3389/fimmu.2018.01831. [PubMed].
32. Lánczky A, Győrffy B. Web-Based Survival Analysis Tool Tailored for Medical Research (KMplot): Development and Implementation. J Med Internet Res. 2021; 23:e27633. https://doi.org/10.2196/27633. [PubMed].
33. Nagy Á, Munkácsy G, Győrffy B. Pancancer survival analysis of cancer hallmark genes. Sci Rep. 2021; 11:6047. https://doi.org/10.1038/s41598-021-84787-5. [PubMed].
34. GTEx Consortium. The Genotype-Tissue Expression (GTEx) project. Nat Genet. 2013; 45:580–85. https://doi.org/10.1038/ng.2653. [PubMed].
35. Bartha Á, Győrffy B. TNMplot.com: A Web Tool for the Comparison of Gene Expression in Normal, Tumor and Metastatic Tissues. Int J Mol Sci. 2021; 22:2622. https://doi.org/10.3390/ijms22052622. [PubMed].
36. Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012; 2:401–4. https://doi.org/10.1158/2159-8290.CD-12-0095. [PubMed].
37. Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013; 6:pl1. https://doi.org/10.1126/scisignal.2004088. [PubMed].
38. Stelzer G, Rosen N, Plaschkes I, Zimmerman S, Twik M, Fishilevich S, Stein TI, Nudel R, Lieder I, Mazor Y, Kaplan S, Dahary D, Warshawsky D, et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr Protoc Bioinformatics. 2016; 54:1.30.1–1.30.33. https://doi.org/10.1002/cpbi.5. [PubMed].
39. Ochsner SA, Abraham D, Martin K, Ding W, McOwiti A, Kankanamge W, Wang Z, Andreano K, Hamilton RA, Chen Y, Hamilton A, Gantner ML, Dehart M, et al. The Signaling Pathways Project, an integrated ‘omics knowledgebase for mammalian cellular signaling pathways. Sci Data. 2019; 6:252. https://doi.org/10.1038/s41597-019-0193-4. [PubMed].
40. Fishilevich S, Nudel R, Rappaport N, Hadar R, Plaschkes I, Iny Stein T, Rosen N, Kohn A, Twik M, Safran M, Lancet D, Cohen D. GeneHancer: genome-wide integration of enhancers and target genes in GeneCards. Database (Oxford). 2017; 2017:bax028. https://doi.org/10.1093/database/bax028. [PubMed].
41. Wu M, Puddifoot CA, Taylor P, Joiner WJ. Mechanisms of inhibition and potentiation of α4β2 nicotinic acetylcholine receptors by members of the Ly6 protein family. J Biol Chem. 2015; 290:24509–18. https://doi.org/10.1074/jbc.M115.647248. [PubMed].
42. Kosugi A, Saitoh S, Noda S, Miyake K, Yamashita Y, Kimoto M, Ogata M, Hamaoka T. Physical and functional association between thymic shared antigen-1/stem cell antigen-2 and the T cell receptor complex. J Biol Chem. 1998; 273:12301–6. https://doi.org/10.1074/jbc.273.20.12301. [PubMed].
43. Noda S, Kosugi A, Saitoh S, Narumiya S, Hamaoka T. Protection from anti-TCR/CD3-induced apoptosis in immature thymocytes by a signal through thymic shared antigen-1/stem cell antigen-2. J Exp Med. 1996; 183:2355–60. https://doi.org/10.1084/jem.183.5.2355. [PubMed].
44. Lv Y, Song Y, Ni C, Wang S, Chen Z, Shi X, Jiang Q, Cao C, Zuo Y. Overexpression of Lymphocyte Antigen 6 Complex, Locus E in Gastric Cancer Promotes Cancer Cell Growth and Metastasis. Cell Physiol Biochem. 2018; 45:1219–29. https://doi.org/10.1159/000487453. [PubMed].
45. Bacquin A, Bireau C, Tanguy M, Romanet C, Vernochet C, Dupressoir A, Heidmann T. A Cell Fusion-Based Screening Method Identifies Glycosylphosphatidylinositol-Anchored Protein Ly6e as the Receptor for Mouse Endogenous Retroviral Envelope Syncytin-A. J Virol. 2017; 91:e00832-17. https://doi.org/10.1128/JVI.00832-17. [PubMed].
46. Gaudet P, Livstone MS, Lewis SE, Thomas PD. Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief Bioinform. 2011; 12:449–62. https://doi.org/10.1093/bib/bbr042. [PubMed].
47. Fujihara Y, Okabe M, Ikawa M. GPI-anchored protein complex, LY6K/TEX101, is required for sperm migration into the oviduct and male fertility in mice. Biol Reprod. 2014; 90:60. https://doi.org/10.1095/biolreprod.113.112888. [PubMed].
48. Ishikawa N, Takano A, Yasui W, Inai K, Nishimura H, Ito H, Miyagi Y, Nakayama H, Fujita M, Hosokawa M, Tsuchiya E, Kohno N, Nakamura Y, Daigo Y. Cancer-testis antigen lymphocyte antigen 6 complex locus K is a serologic biomarker and a therapeutic target for lung and esophageal carcinomas. Cancer Res. 2007; 67:11601–11. https://doi.org/10.1158/0008-5472.CAN-07-3243. [PubMed].
49. Puddifoot CA, Wu M, Sung RJ, Joiner WJ. Ly6h regulates trafficking of alpha7 nicotinic acetylcholine receptors and nicotine-induced potentiation of glutamatergic signaling. J Neurosci. 2015; 35:3420–30. https://doi.org/10.1523/JNEUROSCI.3630-14.2015. [PubMed].
50. Ridge RJ, Sloane NH. Partial N-terminal amino acid sequence of the anti-neoplastic urinary protein (ANUP) and the anti-tumour effect of the N-terminal nonapeptide of the unique cytokine present in human granulocytes. Cytokine. 1996; 8:1–5. https://doi.org/10.1006/cyto.1996.0001. [PubMed].
51. Mastrangeli R, Donini S, Kelton CA, He C, Bressan A, Milazzo F, Ciolli V, Borrelli F, Martelli F, Biffoni M, Serlupi-Crescenzi O, Serani S, Micangeli E, et al. ARS Component B: structural characterization, tissue expression and regulation of the gene and protein (SLURP-1) associated with Mal de Meleda. Eur J Dermatol. 2003; 13:560–70. [PubMed].
52. Favre B, Plantard L, Aeschbach L, Brakch N, Christen-Zaech S, de Viragh PA, Sergeant A, Huber M, Hohl D. SLURP1 is a late marker of epidermal differentiation and is absent in Mal de Meleda. J Invest Dermatol. 2007; 127:301–8. https://doi.org/10.1038/sj.jid.5700551. [PubMed].
53. Chimienti F, Hogg RC, Plantard L, Lehmann C, Brakch N, Fischer J, Huber M, Bertrand D, Hohl D. Identification of SLURP-1 as an epidermal neuromodulator explains the clinical phenotype of Mal de Meleda. Hum Mol Genet. 2003; 12:3017–24. https://doi.org/10.1093/hmg/ddg320. [PubMed].
54. Lyukmanova EN, Shulepko MA, Kudryavtsev D, Bychkov ML, Kulbatskii DS, Kasheverov IE, Astapova MV, Feofanov AV, Thomsen MS, Mikkelsen JD, Shenkarev ZO, Tsetlin VI, Dolgikh DA, Kirpichnikov MP. Human Secreted Ly-6/uPAR Related Protein-1 (SLURP-1) Is a Selective Allosteric Antagonist of α7 Nicotinic Acetylcholine Receptor. PLoS One. 2016; 11:e0149733. https://doi.org/10.1371/journal.pone.0149733. [PubMed].
55. Moriwaki Y, Yoshikawa K, Fukuda H, Fujii YX, Misawa H, Kawashima K. Immune system expression of SLURP-1 and SLURP-2, two endogenous nicotinic acetylcholine receptor ligands. Life Sci. 2007; 80:2365–68. https://doi.org/10.1016/j.lfs.2006.12.028. [PubMed].
56. Swamynathan S, Swamynathan SK. SLURP-1 modulates corneal homeostasis by serving as a soluble scavenger of urokinase-type plasminogen activator. Invest Ophthalmol Vis Sci. 2014; 55:6251–61. https://doi.org/10.1167/iovs.14-15107. [PubMed].
57. Tekinay AB, Nong Y, Miwa JM, Lieberam I, Ibanez-Tallon I, Greengard P, Heintz N. A role for LYNX2 in anxiety-related behavior. Proc Natl Acad Sci U S A. 2009; 106:4477–82. https://doi.org/10.1073/pnas.0813109106. [PubMed].
58. Smith BA, Kennedy WJ, Harnden P, Selby PJ, Trejdosiewicz LK, Southgate J. Identification of genes involved in human urothelial cell-matrix interactions: implications for the progression pathways of malignant urothelium. Cancer Res. 2001; 61:1678–85. [PubMed].
59. Würfel J, Seiter S, Stassar M, Claas A, Kläs R, Rösel M, Marhaba R, Savelyeva L, Schwab M, Matzku S, Zöller M. Cloning of the human homologue of the metastasis-associated rat C4.4A. Gene. 2001; 262:35–41. https://doi.org/10.1016/s0378-1119(00)00515-1. [PubMed].
60. Hansen LV, Gårdsvoll H, Nielsen BS, Lund LR, Danø K, Jensen ON, Ploug M. Structural analysis and tissue localization of human C4.4A: a protein homologue of the urokinase receptor. Biochem J. 2004; 380:845–57. https://doi.org/10.1042/BJ20031478. [PubMed].
61. Fletcher GC, Patel S, Tyson K, Adam PJ, Schenker M, Loader JA, Daviet L, Legrain P, Parekh R, Harris AL, Terrett JA. hAG-2 and hAG-3, human homologues of genes involved in differentiation, are associated with oestrogen receptor-positive breast tumours and interact with metastasis gene C4.4a and dystroglycan. Br J Cancer. 2003; 88:579–85. https://doi.org/10.1038/sj.bjc.6600740. [PubMed].
62. Arvaniti M, Jensen MM, Soni N, Wang H, Klein AB, Thiriet N, Pinborg LH, Muldoon PP, Wienecke J, Imad Damaj M, Kohlmeier KA, Gondré-Lewis MC, Mikkelsen JD, Thomsen MS. Functional interaction between Lypd6 and nicotinic acetylcholine receptors. J Neurochem. 2016; 138:806–20. https://doi.org/10.1111/jnc.13718. [PubMed].
63. Ochoa V, George AA, Nishi R, Whiteaker P. The prototoxin LYPD6B modulates heteromeric α3β4-containing nicotinic acetylcholine receptors, but not α7 homomers. FASEB J. 2016; 30:1109–19. https://doi.org/10.1096/fj.15-274548. [PubMed].
64. Okumura R, Kurakawa T, Nakano T, Kayama H, Kinoshita M, Motooka D, Gotoh K, Kimura T, Kamiyama N, Kusu T, Ueda Y, Wu H, Iijima H, et al. Lypd8 promotes the segregation of flagellated microbiota and colonic epithelia. Nature. 2016; 532:117–21. https://doi.org/10.1038/nature17406. [PubMed].
65. Lyukmanova EN, Shenkarev ZO, Shulepko MA, Mineev KS, D’Hoedt D, Kasheverov IE, Filkin SY, Krivolapova AP, Janickova H, Dolezal V, Dolgikh DA, Arseniev AS, Bertrand D, et al. NMR structure and action on nicotinic acetylcholine receptors of water-soluble domain of human LYNX1. J Biol Chem. 2011; 286:10618–27. https://doi.org/10.1074/jbc.M110.189100. [PubMed].
66. Furuhata T, Tokino T, Urano T, Nakamura Y. Isolation of a novel GPI-anchored gene specifically regulated by p53; correlation between its expression and anti-cancer drug sensitivity. Oncogene. 1996; 13:1965–70. [PubMed].
67. Mysling S, Kristensen KK, Larsson M, Kovrov O, Bensadouen A, Jørgensen TJ, Olivecrona G, Young SG, Ploug M. The angiopoietin-like protein ANGPTL4 catalyzes unfolding of the hydrolase domain in lipoprotein lipase and the endothelial membrane protein GPIHBP1 counteracts this unfolding. Elife. 2016; 5:e20958. https://doi.org/10.7554/eLife.20958. [PubMed].
68. Kristensen KK, Midtgaard SR, Mysling S, Kovrov O, Hansen LB, Skar-Gislinge N, Beigneux AP, Kragelund BB, Olivecrona G, Young SG, Jørgensen TJD, Fong LG, Ploug M. A disordered acidic domain in GPIHBP1 harboring a sulfated tyrosine regulates lipoprotein lipase. Proc Natl Acad Sci U S A. 2018; 115:E6020–29. https://doi.org/10.1073/pnas.1806774115. [PubMed].
69. Beigneux AP, Franssen R, Bensadoun A, Gin P, Melford K, Peter J, Walzem RL, Weinstein MM, Davies BS, Kuivenhoven JA, Kastelein JJ, Fong LG, Dallinga-Thie GM, Young SG. Chylomicronemia with a mutant GPIHBP1 (Q115P) that cannot bind lipoprotein lipase. Arterioscler Thromb Vasc Biol. 2009; 29:956–62. https://doi.org/10.1161/ATVBAHA.109.186577. [PubMed].
70. Coca-Prieto I, Kroupa O, Gonzalez-Santos P, Magne J, Olivecrona G, Ehrenborg E, Valdivielso P. Childhood-onset chylomicronaemia with reduced plasma lipoprotein lipase activity and mass: identification of a novel GPIHBP1 mutation. J Intern Med. 2011; 270:224–28. https://doi.org/10.1111/j.1365-2796.2011.02361.x. [PubMed].
71. Gin P, Beigneux AP, Davies B, Young MF, Ryan RO, Bensadoun A, Fong LG, Young SG. Normal binding of lipoprotein lipase, chylomicrons, and apo-AV to GPIHBP1 containing a G56R amino acid substitution. Biochim Biophys Acta. 2007; 1771:1464–68. https://doi.org/10.1016/j.bbalip.2007.10.005. [PubMed].
72. Mallya M, Campbell RD, Aguado B. Characterization of the five novel Ly-6 superfamily members encoded in the MHC, and detection of cells expressing their potential ligands. Protein Sci. 2006; 15:2244–56. https://doi.org/10.1110/ps.062242606. [PubMed].
73. De Vet EC, Aguado B, Campbell RD. Adaptor signalling proteins Grb2 and Grb7 are recruited by human G6f, a novel member of the immunoglobulin superfamily encoded in the MHC. Biochem J. 2003; 375:207–13. https://doi.org/10.1042/BJ20030293. [PubMed].
74. Roldan AL, Cubellis MV, Masucci MT, Behrendt N, Lund LR, Danø K, Appella E, Blasi F. Cloning and expression of the receptor for human urokinase plasminogen activator, a central molecule in cell surface, plasmin dependent proteolysis. EMBO J. 1990; 9:467–74. https://doi.org/10.1002/j.1460-2075.1990.tb08132.x. [PubMed].
75. Sakamoto H, Yoshimura K, Saeki N, Katai H, Shimoda T, Matsuno Y, Saito D, Sugimura H, Tanioka F, Kato S, Matsukura N, Matsuda N, et al, and Study Group of Millennium Genome Project for Cancer. Genetic variation in PSCA is associated with susceptibility to diffuse-type gastric cancer. Nat Genet. 2008; 40:730–40. https://doi.org/10.1038/ng.152. [PubMed].
76. Jensen MM, Arvaniti M, Mikkelsen JD, Michalski D, Pinborg LH, Härtig W, Thomsen MS. Prostate stem cell antigen interacts with nicotinic acetylcholine receptors and is affected in Alzheimer’s disease. Neurobiol Aging. 2015; 36:1629–38. https://doi.org/10.1016/j.neurobiolaging.2015.01.001. [PubMed].

