Introduction
Tumor-related systemic inflammation based on host–tumor interaction between cancer loci and individuals is caused not only by local nutritional malabsorption but also by systemic metabolic disorders [1, 2]. Systemic inflammation must be evaluated objectively and dynamically as it changes constantly during multidisciplinary treatment. Against this background, the usefulness of biomarkers has been attracting attention in recent years, and it is desirable to devise biomarkers that can evaluate these dynamic changes more quickly, easily, accurately, and at a lower cost [3, 4]. Biomarkers are generally classified into three categories in cancer treatment: (1) having diagnostic significance for cancer, (2) serving as prognostic indicators, and (3) predicting therapeutic effects or risk of side effects. Although it may not be possible to develop significant markers that predict all three categories, tumor-related systemic inflammation and metabolic malnutrition in patients with cancer occur not only in advanced cancers but also in relatively early-stage cancers and are prognostic factors independent of pathological factors and induce treatment resistance [5, 6]. Therefore, it is reasonable to include systemic inflammation and metabolic nutritional status as indicators when devising biomarkers. If the prognosis can be predicted using the pre-treatment specimens, it will lead to the identification of a group of patients who require multimodal treatment, including aggressive chemotherapy and radiotherapy, which will lead to individualized treatment and improved prognosis [6, 7]. Occasionally, even stage 1 cases may recur after surgery. In this study, we focused on blood cell components that complementarily reflect systemic inflammation and metabolic status and devised a cancer-specific prognostic marker for all patients, not limited to patients in stages II and III.
In this study, we focused on blood cell components that complementarily reflect systemic inflammation and metabolic status and devised a cancer-specific prognostic marker.
Results
Association between the inflammatory biomarkers and clinicopathological features
The 480 patients were divided into the low and high groups based on the cutoff values of each inflammatory biomarker (Table 1); 207 patients (43.1% [male, 154; female, 53]) showed low LMR (median age, 74 [range, 38–91] years). Moreover, 273 patients (56.9% [male, 183; female, 90]) showed high LMR (median age, 69 [range, 36–89] years). Furthermore, 296 (61.7% [male, 204; female, 92]) patients showed low NLR (median age, 70 [range, 36–91] years), and 184 (38.3% [male, 133; female, 51]) patients showed high NLR (median age, 74 [range, 43–90] years). A total of 407 (84.8% [male, 286; female, 121]) patients showed low PLR (median age, 70 [range, 36–90] years), and 73 (15.2% [male, 51; female, 22]) showed high PLR (median age, 72 [range, 43–91] years).
Table 1: Association between the inflammatory biomarkers and clinicopathological features
| Characteristics | Total patients | LMR | NLR | PLR | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| <4.315 (n = 207) | ≥4.315 (n = 273) | p value | <2.344 (n = 296) | ≥2.344 (n = 184) | p value | <212.01 (n = 407) | ≥212.01 (n = 73) | p value | ||
| Age (years) | 74 (38–91) | 69 (36–89) | <0.001 | 70 (36–91) | 74 (43–90) | 0.008 | 70 (36–90) | 72 (43–91) | 0.062 | |
| Sex | 0.079 | 0.432 | 0.944 | |||||||
| Male | 337 | 154 | 183 | 204 | 133 | 286 | 51 | |||
| Female | 143 | 53 | 90 | 92 | 51 | 121 | 22 | |||
| ASA–PS | <0.001 | 0.002 | 0.007 | |||||||
| 1 | 25 | 5 | 20 | 19 | 6 | 22 | 3 | |||
| 2 | 409 | 171 | 238 | 259 | 150 | 354 | 55 | |||
| 3 | 46 | 31 | 15 | 18 | 28 | 31 | 15 | |||
| BMI | 21.9 (14.0–32.5) | 22.8 (14.8–40.4) | <0.001 | 22.3 (14.7–40.4) | 22.3 (14.0–32.7) | 0.754 | 22.4 (14.7–40.4) | 21.7 (14.0–32.5) | 0.063 | |
| WBC | 5730 (2870–13700) | 5630 (510–9830) | 0.292 | 5460 (510–9280) | 6115 (3510–13700) | <0.001 | 5710 (510–13700) | 5460 (1830–12730) | 0.133 | |
| Neutrophil | 3700 (1310–11460) | 3190 (250–6910) | <0.001 | 3010 (250–5100) | 4270 (2210–11460) | <0.001 | 3340 (250–8494) | 3850 (1100–11460) | 0.012 | |
| Lymphocyte | 1310 (230–3780) | 1850 (230–3780) | <0.001 | 1845 (230–3780) | 1255 (230–2270) | <0.001 | 1730 (230–3780) | 960 (230–2020) | <0.001 | |
| Monocyte | 408 (210–937) | 311 (3–727) | <0.001 | 339 (3–937) | 366 (85–829) | <0.001 | 348 (3–937) | 362 (37–829) | 0.192 | |
| Platelet | 216 (58–726) | 222 (39–460) | 0.432 | 220 (39–460) | 220 (58–726) | 0.267 | 215 (39–460) | 283 (119–726) | <0.001 | |
| Tumor location | 0.891 | 0.842 | 0.84 | |||||||
| EGJ | 15 | 7 | 8 | 8 | 7 | 13 | 2 | |||
| U | 93 | 43 | 50 | 58 | 35 | 76 | 17 | |||
| M | 204 | 85 | 119 | 129 | 75 | 174 | 30 | |||
| L | 168 | 72 | 96 | 101 | 67 | 144 | 24 | |||
| Tumor diameter (mm) | 44 (3–176) | 40 (4–180) | 0.01 | 40 (3–180) | 42 (5–176) | 0.059 | 40 (3–180) | 50 (16–150) | 0.001 | |
| Differentiation | 0.657 | 0.44 | 0.431 | |||||||
| Well | 94 | 37 | 57 | 63 | 31 | 83 | 11 | |||
| Moderate | 177 | 76 | 101 | 109 | 68 | 146 | 31 | |||
| Poor | 209 | 94 | 115 | 124 | 85 | 178 | 31 | |||
| Depth of tumor | 0.003 | 0.01 | <0.001 | |||||||
| T1a-1b | 252 | 89 | 163 | 171 | 81 | 226 | 26 | |||
| 2 | 62 | 29 | 33 | 39 | 23 | 55 | 7 | |||
| 3 | 71 | 38 | 33 | 39 | 32 | 59 | 12 | |||
| 4a-4b | 95 | 51 | 44 | 47 | 48 | 67 | 28 | |||
| Lymph node meta | 0.004 | 0.332 | 0.011 | |||||||
| N0 | 314 | 123 | 191 | 203 | 111 | 278 | 36 | |||
| N1 | 57 | 29 | 28 | 32 | 25 | 41 | 16 | |||
| N2 | 56 | 32 | 24 | 31 | 25 | 46 | 10 | |||
| N3 | 53 | 23 | 30 | 30 | 23 | 42 | 11 | |||
| pTNM stage | <0.001 | 0.004 | <0.001 | |||||||
| 1a-1b | 283 | 99 | 184 | 192 | 91 | 255 | 28 | |||
| 2a-2b | 87 | 50 | 37 | 47 | 40 | 67 | 20 | |||
| 3a-3c | 110 | 58 | 52 | 57 | 53 | 85 | 25 | |||
| Operative procedure | 0.001 | 0.209 | 0.034 | |||||||
| Total | 101 | 58 | 43 | 58 | 43 | 78 | 23 | |||
| Proximal | 50 | 14 | 36 | 36 | 14 | 46 | 4 | |||
| Distal | 329 | 135 | 194 | 202 | 127 | 283 | 46 | |||
| Operation time (min) | 390 (204–911) | 378 (70–808) | 0.51 | 381 (158–911) | 386 (70–703) | 0.5 | 383 (70–911) | 386 (231–692) | 0.288 | |
| Intraope. blood loss | 50 (0–3600) | 20 (0–5850) | 0.03 | 40 (0–5850) | 50 (0–2600) | 0.568 | 40 (0–5850) | 30 (0–1600) | 0.921 | |
| Postoperative complications | 0.036 | 0.486 | 0.419 | |||||||
| Present | 145 | 73 | 72 | 86 | 59 | 120 | 25 | |||
| Absent | 335 | 134 | 201 | 210 | 125 | 287 | 48 | |||
| CRP (mg/dl) | 0.11 (0.01–11.10) | 0.06 (0.01–4.26) | <0.001 | 0.07 (0.01–5.35) | 0.11 (0.01–11.1) | <0.001 | 0.07 (0.01–11.1) | 0.16 (0.01–7.09) | <0.001 | |
| CEA (ng/ml) | 3.4 (0.7–171.6) | 3.3 (0.7–86.4) | 0.23 | 3.2 (0.7–106.0) | 3.6 (0.7–171.6) | 0.064 | 3.3 (0.7–171.6) | 3.4 (0.8–163.3) | 0.566 | |
| Adjuvant chemotherapy | 0.919 | 0.558 | 0.557 | |||||||
| Yes | 131 | 56 | 75 | 78 | 53 | 109 | 22 | |||
| No | 349 | 151 | 198 | 218 | 131 | 298 | 51 | |||
Cox regression analysis of inflammatory biomarkers associated with cancer-specific survival (CSS)
In univariate analysis, tumor diameter (p < 0.001), histological differentiation (p = 0.029), pathological TNM (pTNM) stage (p < 0.001), LMR (hazard ratio [HR], 2.866; p < 0.001), NLR (HR, 2.778; p < 0.001), PLR (HR, 2.803; p = 0.001), C-reactive protein (CRP) level (p = 0.003), carcinoembryonic antigen (CEA) (p = 0.011), and postoperative chemotherapy (p < 0.001) were significantly associated with CSS. In multivariate analysis, pTNM stage (HR, 21.452; 95% confidence interval [CI], 2.268–8.151; p < 0.001) and CEA (HR, 2.000; 95% CI, 1.089–3.672; p = 0.025) were identified as independent risk factors for CSS, although LMR, NLR, and PLR were not found to be independent risk factors for CSS (Table 2).
Table 2: Cox regression analysis of inflammatory biomarkers associated with cancer-specific survival
| Variables | Category or characteristics | Patients (n = 480) | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p value | HR | 95% CI | p value | |||
| Age | (<70/≥70) | 225/255 | 1.373 | 1.134–1.662 | 0.258 | |||
| Sex | (female/male) | 143/337 | 1.534 | 0.782–3.009 | 0.196 | |||
| BMI | (≥18.5/<18.5) | 439/41 | 1.013 | 0.364–2.824 | 0.980 | |||
| Tumor diameter | (<5/≥5) | 285/195 | 4.299 | 2.268–8.151 | <0.001 | 1.713 | 0.875–3.354 | 0.116 |
| Differentiation | (well and mod/poor) | 271/209 | 1.895 | 1.067–3.364 | 0.029 | 0.822 | 0.447–1.511 | 0.528 |
| pTNM stage | (1/2,3) | 283 /197 | 40.385 | 9.799–166.437 | <0.001 | 21.452 | 4.549–101.176 | <0.001 |
| LMR | (≥4.315/<4.315) | 273/207 | 2.866 | 1.585–5.181 | <0.001 | 1.523 | 0.760–3.074 | 0.234 |
| NLR | (<2.344/≥2.344) | 296/184 | 2.778 | 1.557–4.956 | <0.001 | 1.563 | 0.782–3.123 | 0.206 |
| PLR | (<212.069/≥212.069) | 407/73 | 2.803 | 1.503–5.227 | 0.001 | 1.170 | 0.570–2.403 | 0.669 |
| CRP | (≦0.5/>0.5) | 413/67 | 2.664 | 1.409–5.037 | 0.003 | 0.954 | 0.472–1.929 | 0.896 |
| CEA | (<5.0/≥5.0) | 364/116 | 2.210 | 1.231–3.966 | 0.011 | 2.000 | 1.089–3.672 | 0.025 |
| Postope. Complications | (absent/present) | 335/145 | 1.280 | 0.695–2.358 | 0.428 | |||
| Adjuvant chemotherapy | (no/yes) | 349/131 | 6.206 | 3.329–11.570 | <0.001 | 1.371 | 0.675–2.787 | 0.383 |
CSS according to inflammatory biomarkers
The Kaplan–Meier survival curve revealed significantly worse CSS in the low LMR (p < 0.001) (Figure 1A), high NLR (p < 0.001) (Figure 1B), and high PLR (p < 0.001) groups (Figure 1C).

Figure 1: Cancer-specific survival curve based on the inflammatory biomarkers. (A) Lymphocyte-to-monocyte ratio, (B) neutrophil-to-lymphocyte ratio, and (C) platelet-to-lymphocyte ratio. Abbreviations: LMR: Lymphocyte-to-monocyte ratio, NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio.
Inflammation-based prognostic index formula
The inflammation-based prognostic index (ICPI) formula was constructed using HRs for three inflammation-based biomarkers with worse prognosis identified in the univariate analysis. LMR <4.315, NLR ≥2.344, and PLR ≥212.01, which were each scored as 1, with all remaining values pointed at 0.
The ICPI was calculated as follows: ICPI = 2.9 × LMR + 2.8 × NLR + 2.8 × PLR.
Using ROC analysis, the optimal cutoff value of ICPII was 2.9 based on CSS (sensitivity, 0.715%; specificity, 0.583%; area under the curve [AUC], 0.656).
Association between ICPI and clinicopathological features
A total of 329 (68.5% [male, 226; female, 103]) patients showed low ICPI (median age, 74 [range, 38–91] years), and 151 (31.5% [male, 111; female, 40]) patients showed high ICPI. The ICPI was significantly correlated with age, American Society of Anaesthesiologists Physical Status, white blood cell count, neutrophil count, lymphocyte count, monocyte count, platelet count, tumor diameter, tumor depth, lymph node metastasis, pTNM stage, operative procedure, and CRP (Table 3).
Table 3: Relationships between the inflammation-combined prognostic index and clinicopathological features
| Characteristics | Total patients | ICPI | p value | |
|---|---|---|---|---|
| ≤2.9 | >2.9 | |||
| (n = 329) | (n = 151) | |||
| Age (years) | 70 (36–90) | 74 (43–91) | 0.001 | |
| Sex | 0.281 | |||
| Male | 337 | 226 | 111 | |
| Female | 143 | 103 | 40 | |
| ASA-PS | <0.001 | |||
| 1 | 25 | 21 | 4 | |
| 2 | 409 | 288 | 121 | |
| 3 | 46 | 20 | 26 | |
| BMI | 22.4 (14.7–40.4) | 22.0 (14.0–32.5) | 0.102 | |
| WBC | 5580 (510–9830) | 5910 (2880–13700) | 0.001 | |
| Neutrophil | 3110 (250–6910) | 4070 (1310–11460) | <0.001 | |
| Lymphocyte | 1820 (230–3780) | 1190 (230–2100) | <0.001 | |
| Monocyte | 332 (3–937) | 382 (165–829) | <0.001 | |
| Platelet | 218 (39–460) | 230 (58–726) | 0.029 | |
| Tumor location | 0.592 | |||
| EGJ | 15 | 8 | 7 | |
| U | 93 | 62 | 31 | |
| M | 204 | 143 | 61 | |
| L | 168 | 116 | 52 | |
| Tumor diameter (mm) | 40 (3–180) | 45 (5–176) | 0.013 | |
| Differentiation | 0.644 | |||
| Well | 94 | 68 | 26 | |
| Moderate | 177 | 121 | 56 | |
| Poor | 209 | 140 | 69 | |
| Depth of tumor | <0.001 | |||
| T1a-1b | 252 | 189 | 63 | |
| 2 | 62 | 46 | 16 | |
| 3 | 71 | 43 | 28 | |
| 4a-4b | 95 | 51 | 44 | |
| Lymph node meta | 0.044 | |||
| N0 | 314 | 229 | 85 | |
| N1 | 57 | 34 | 23 | |
| N2 | 56 | 33 | 23 | |
| N3 | 53 | 33 | 20 | |
| pTNM stage | <0.001 | |||
| 1a-1b | 283 | 216 | 67 | |
| 2a-2b | 87 | 50 | 37 | |
| 3a-3c | 110 | 63 | 47 | |
| Operative procedure | 0.044 | |||
| Total | 101 | 60 | 41 | |
| Proximal | 50 | 39 | 11 | |
| Distal | 329 | 230 | 99 | |
| Operation time (min) | 380 (70–911) | 386 (204–703) | 0.641 | |
| Intraoperative blood loss | 40 (0–5850) | 50 (0–2600) | 0.314 | |
| Postoperative complications | 0.252 | |||
| Present | 145 | 94 | 51 | |
| Absent | 335 | 235 | 100 | |
| CRP (mg/dl) | 0.06 (0.01–5.35) | 0.12 (0.01–11.10) | <0.001 | |
| CEA (ng/ml) | 3.3 (0.7–106.0) | 3.4 (0.7–171.6) | 0.134 | |
| Adjuvant chemotherapy | 0.54 | |||
| Yes | 131 | 87 | 44 | |
| No | 349 | 242 | 107 | |
Comparison of predictive ability of inflammatory biomarkers for CSS
The AUC estimate method was used to compare the predictive ability of the inflammatory biomarkers. The AUCs of LMR, NLR, PLR, and ICPI were 0.594, 0.596, 0.585, and 0.656, respectively. The AUCs of ICPI were significantly higher than those of LMR (p = 0.029), NLR (p = 0.018), and PLR (p = 0.005) (Figure 2).
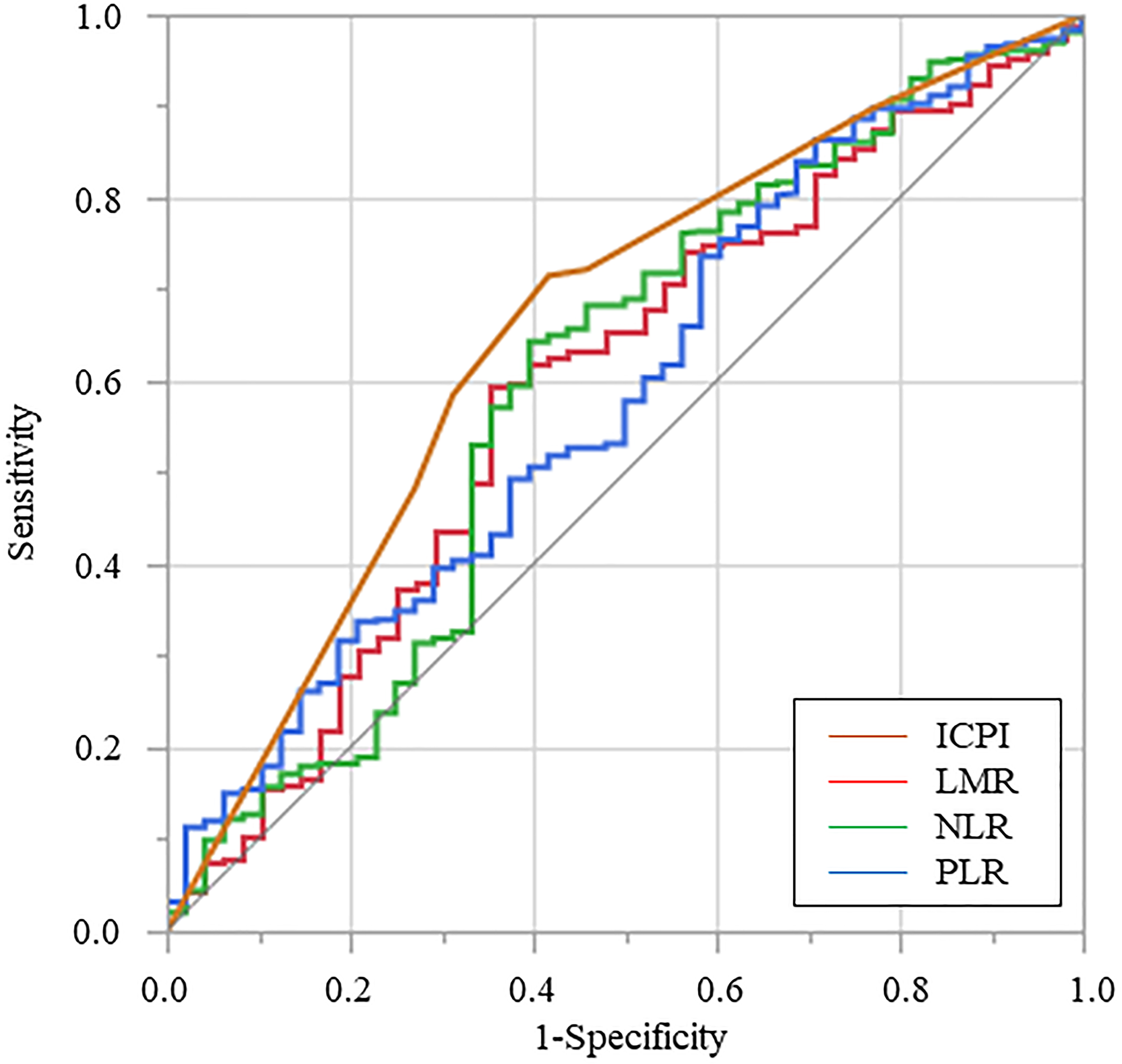
Figure 2: Receiver operating characteristic curve for cancer-specific survival was plotted to verify the optimum cutoff value of lymphocyte-to-monocyte ratio, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and inflammation-based prognostic index. Abbreviations: LMR: Lymphocyte-to-monocyte ratio, NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio, ICPI: Inflammation-combined prognostic index.
Cox regression analysis of ICPI associated with CSS
On multivariate analysis, pTNM stage (HR, 22.646; 95% CI, 4.826–106.277; p < 0.001), CEA (HR, 2.050; 95% CI, 1.120–2.570; p = 0.020), and ICPI (HR, 2.511; 95% CI, 1.383–4.562; p = 0.003) were confirmed as independent prognostic factors for CSS (Table 4).
Table 4: Cox regression analysis of the inflammation-combined prognostic index associated with cancer-specific survival
| Variables | Category or characteristics | Patients (n = 480) | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p value | HR | 95% CI | p value | |||
| Age | (<70/≥70) | 225/265 | 1.373 | 1.134–1.662 | 0.258 | |||
| Sex | (female/male) | 143/337 | 1.534 | 0.782–3.009 | 0.196 | |||
| BMI | (≥18.5/<18.5) | 439/41 | 1.013 | 0.364–2.824 | 0.980 | |||
| Tumor diameter | (<5/≥5) | 285/195 | 4.299 | 2.268–8.151 | <0.001 | 1.656 | 0.851–3.221 | 0.138 |
| Differentiation | (well and mod/poor) | 271/209 | 1.895 | 1.067–3.364 | 0.029 | 0.798 | 0.439–1.450 | 0.460 |
| pTNM stage | (1/2,3) | 283/197 | 40.385 | 9.799–166.437 | <0.001 | 22.646 | 4.826–106.277 | <0.001 |
| ICPI | (≦2.9/>2.9) | 329/151 | 3.757 | 2.115–6.674 | <0.001 | 2.511 | 1.383–4.562 | 0.003 |
| CRP | (≦0.5/>0.5) | 413/67 | 2.664 | 1.409–5.037 | 0.003 | 0.987 | 0.496–1.964 | 0.971 |
| CEA | (<5.0/≥5.0) | 364/116 | 2.210 | 1.231–3.966 | 0.011 | 2.050 | 1.120–3.753 | 0.020 |
| Postope. Complication | (absent/present) | 335/145 | 1.280 | 0.695–2.358 | 0.428 | |||
| Adjuvant chemotherapy | (no/yes) | 349/131 | 6.206 | 3.329–11.570 | <0.001 | 1.281 | 0.639–2.570 | 0.485 |
CSS according to ICPI
In the Kaplan–Meier survival analysis, CSS in the high ICPI group was significantly worse than that in the low ICPI group (Figure 3).
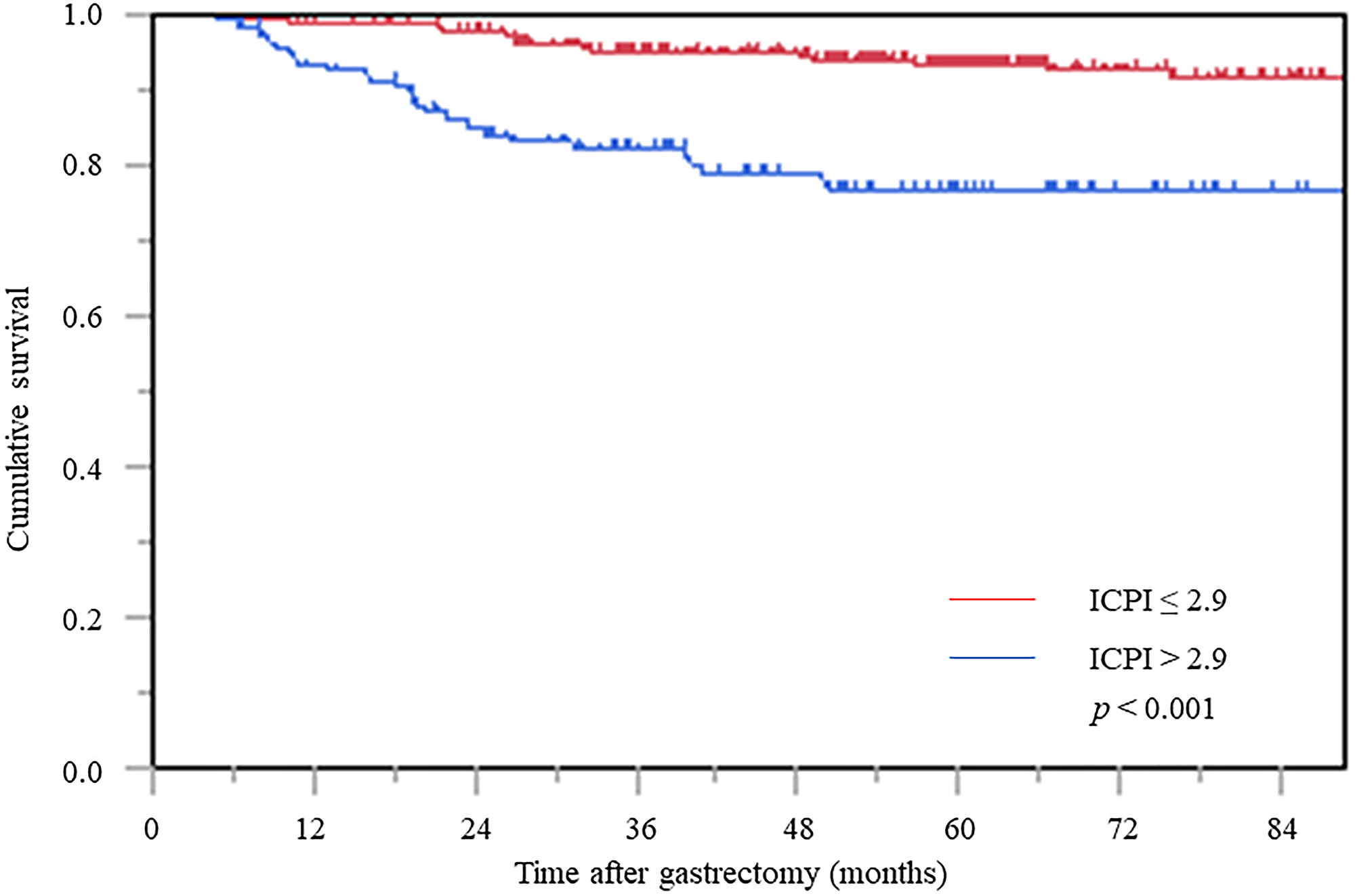
Figure 3: Cancer-specific survival curve based on the inflammation-based prognostic index. Abbreviation: ICPI: inflammation based prognostic index.
Furthermore, in stage stratification analysis, the high ICPI group was significantly associated with worse prognosis in stages II and III, whereas the prognosis of stage I patients did not reach statistical significance among the ICPI values (Figure 4A–4C).
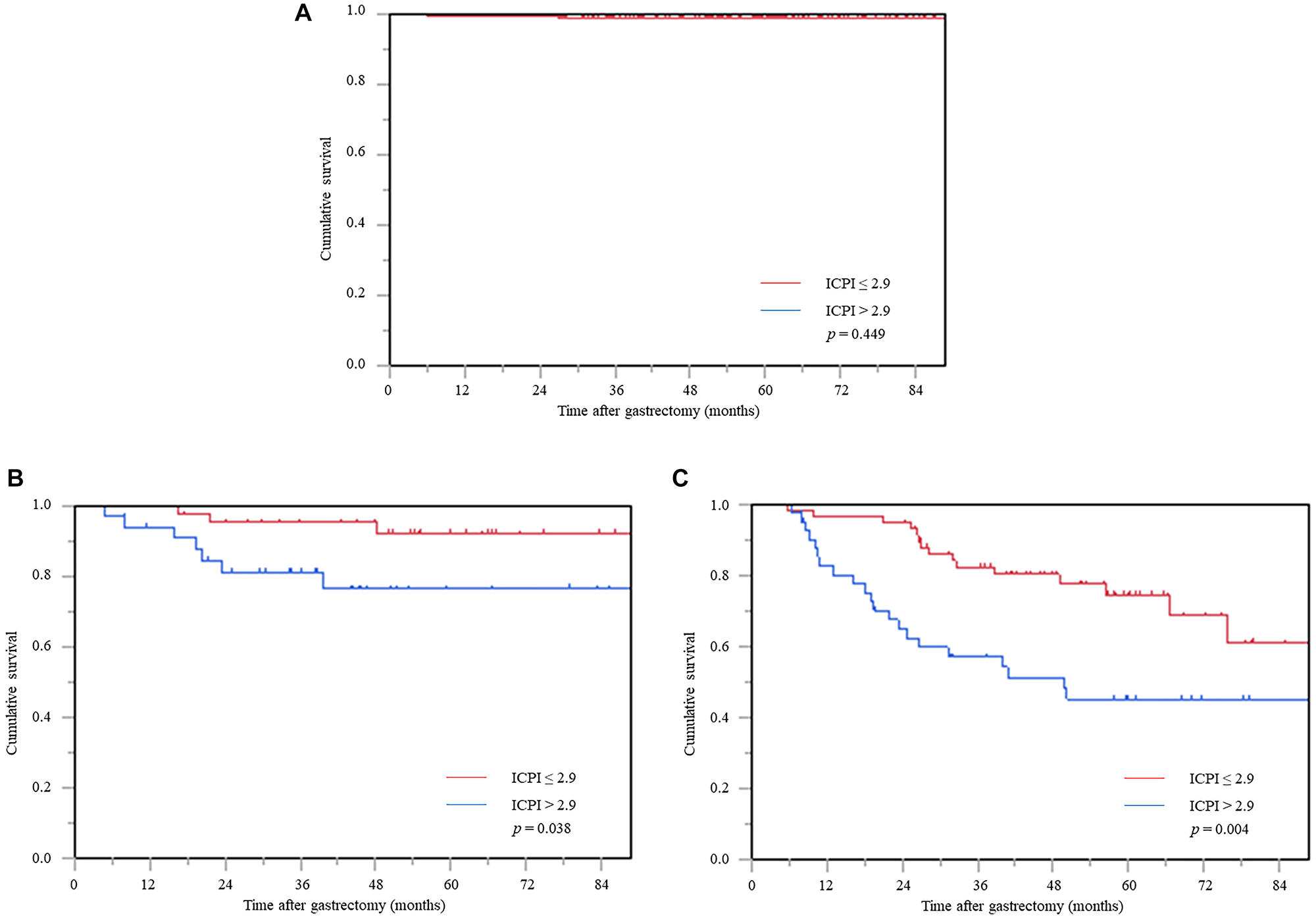
Figure 4: Cancer-specific survival curve based on the inflammation-based prognostic index in the stage stratification analysis. (A) Stage I, (B) stage II, and (C) stage III. Abbreviation: ICPI: inflammation based prognostic index.
DISCUSSION
In vivo inflammatory responses are involved in cancer growth, invasion, and metastasis, and the involvement of systemic inflammatory responses and the surrounding microenvironment is intricately intertwined [5, 8, 9]. Tumor necrosis factor-α (TNF-α), granulocyte colony-stimulating factor, interleukin-1 (IL-1), and IL-6 are produced by tumor cells and can induce a tumor-related systemic inflammatory reaction (SIR) [10, 11]. Among them, IL-6 is a multifunctional inflammatory cytokine that causes the proliferation and differentiation of various types of cells, such as immunocompetent and hematopoietic cells [12, 13]. Thus, dynamic changes in SIR resulting from tumor–host interaction can be accurately assessed by direct measurement of cytokines. However, routine measurement of cytokines in patients with cancer in clinical practice is expensive and impractical. In contrast, LMR, NLR, and PLR assessments using neutrophils, lymphocytes, monocytes, and platelets, which are regulated by cytokines, proliferate, and differentiate, are simple methods to evaluate the systemic inflammatory response using blood cell components and are complementary to each other [14, 15].
Neutrophils regulate the tumor microenvironment by producing pro-inflammatory cytokines/chemokines that promote proliferation, invasion, and metastasis of cancer cells, such as matrix metalloproteinase-9 and anti-apoptotic factor (nuclear factor kappa light chain enhancer of activated B cells) [16]. Furthermore, increased neutrophils produce large amounts of nitric oxide, arginase, and reactive oxygen species, which not only impair T-cell activation and reduce extracellular matrix adhesion but also promote angiogenesis and cellular DNA damage and inhibit tumor cell apoptosis [17]. As a result, a favorable microenvironment for tumor cells is established, which promotes tumor growth and metastasis. Lymphocytes function as an important component of the immune complex and serve as an antitumor immune response by inducing cytotoxic cell death and inhibiting tumor cell proliferation and migration. In addition, lymphocytes secrete cytokines, such as interferon-γ and TNF-α, which regulate cancer cell growth and metastasis through cellular and humoral immune mechanisms [18, 19]. It has also been shown to be a useful marker for screening nutritional status. Monocytes in the peripheral blood migrate to tissues, mature, and differentiate into macrophages. In patients with cancer, macrophages infiltrating the stroma of tumor tissues are called tumor-associated macrophages, which suppress tumor immunity and promote cancer cell proliferation by releasing angiogenic factors and inhibiting cytotoxic T cells [20]. Platelets allow circulating tumor cells to escape host immune surveillance via platelet-derived transforming growth factor-β and direct platelet-tumor cell contact to induce epithelial-mesenchymal transition, angiogenesis, and differentiation of cancer-associated fibroblasts and regulatory T cells. As a result, it induces microvascular permeability, which promotes the extravasation of cancer cells and induces distant metastasis [21].
Thus, LMR expresses the immune response in the tumor microenvironment and is an indicator of individual immunity [22, 23]. PLR serves as a marker for the balance between the inflammatory reaction and immune response of the host [24, 25]. NLR was initially reported as a predictor of outcome in critically ill patients admitted to intensive care units, but it has since been reported as an oncological prognostic marker and is the most evidence-accumulating biomarker [26, 27]. Since these biomarkers reflect different pathological conditions in patients with cancer, it is necessary to integrate and evaluate the three biomarkers to predict the prognosis of cancer more accurately. In this study, each inflammatory marker showed significant differences in univariate analysis but was not extracted as an independent prognostic factor in the multivariate analysis. Considering that these inflammatory markers calculated from two types of blood cell components are insufficient as prognostic predictors, we devised a novel biomarker reflecting systemic inflammation.
Since the HR is a numerical value that objectively compares the relative risk, we devised the ICPI, which is a novel prognostic marker calculated by adding the specific gravity provided to the prognosis of each inflammatory marker using the HR in univariate analysis. As a result, it was proven that the AUC value of ICPI was significantly higher than that of each inflammatory marker, demonstrating its high predictive and diagnostic ability. Furthermore, ICPI could be extracted as an independent prognostic factor in multivariate analysis.
We have previously reported the usefulness of an index calculated by adding the number of markers that recognized a significant difference in esophageal cancer, ignoring the specific gravity provided to the prognosis of each inflammatory marker. However, by considering the prognostic significance of each marker, the detection power of the index as a prognostic indicator increased.
Although ICPI is a new prognostic prediction index for cancer, sufficient attention is required for its interpretation. First, the number of cases was relatively small and included cases with a short postoperative follow-up period. Furthermore, some medicines, such as anticoagulants and anti-inflammatory agents, have not been evaluated. It is also necessary to measure inflammatory cytokines associated with tumors, and it is a future issue whether ICPI can be a prognostic index for other carcinomas. Second, we did not histologically examine leukocyte migration and infiltration into the cancer site. Third, the calculation formula is complicated, which impedes the generalization of this marker. Because the inflammatory biomarkers have similar hazard ratios, the ICBI formula may be simplified by unifying the coefficients to 2.8 or 2.9. Alternatively, removing the coefficient and adding the number of risk factor inflammation biomarkers will simplify the formula, but further examination is required in the future. In addition, further usefulness may be found by examining its association with the recurrence pattern.
In this study, the ICPI was devised as a novel predictive index of prognosis, and its usefulness was clarified. However, it is still unclear how active preoperative intervention using the ICPI as an indicator will contribute to improved oncological prognosis. In the future, it will be necessary to conduct a multicenter prospective study to examine the prognostic effect of preoperative interventions, including nutrition.
Materials and Methods
Patients
We conducted a retrospective study of patients with gastric cancer who underwent curative laparoscopic gastrectomy between January 2009 and December 2019 at our institution. The average follow-up period for survival was 1743.3 days, and the median follow-up period was 1709 days (interquartile range, 969–2304). Clinical patients’ clinicopathological data and laboratory records were collected using an electronic medical records platform. Blood biochemical examination was performed within 1 week prior to the surgery. All patients were eligible for laparoscopic surgery, but we excluded patients with severe adhesion in the abdominal cavity. Furthermore, laparoscopic surgery was not indicated for patients in whom gastrectomy could not be performed without grasping the cancer site with forceps.
Gastrectomy and lymphadenectomy were usually performed according to the guidelines of the Japanese Gastric Cancer Association [28]. The postoperative stage was based on the 7th edition of the tumor-node-metastasis (TNM) system [29]. The severity of postoperative complications was graded according to the Clavien–Dindo (CD) classification [30]. CD grade II or higher complications were defined as the occurrence of any complications. Postoperative adjuvant chemotherapy with tegafur/gimeracil/oteracil potassium (S-1) was recommended for patients with stage II or higher gastric cancer, usually for 1 year. Furthermore, 5-fluorouracil-based chemotherapy regimens (cisplatin plus S-1 or capecitabine) were recommended to the majority of patients with recurrent gastric cancer according to the Japanese Gastric Cancer Treatment Guidelines (Version 4) [28].
Inflammatory biomarkers
The lymphocyte-to-monocyte ratio (LMR) was calculated by dividing the absolute peripheral lymphocyte count by the absolute monocyte count, neutrophil-to-lymphocyte ratio (NLR) was calculated by dividing the absolute peripheral neutrophil count by the absolute lymphocyte count, and platelet-to-lymphocyte ratio (PLR) was calculated by dividing the absolute platelet count by the lymphocyte count.
The optimal cutoff values of the LMR, NLR, and PLR were determined via the receiver operating curve (ROC) analysis. The optimal cutoff values of LMR, NLR, and PLR for predicting cancer-specific survival (CSS) were 4.315, 2.344, and 212.1, respectively.
Statistical analyses
CSS was defined as the date of gastrectomy until death due to gastric cancer.
Student’s t-test was used when assessing continuous variables, and the chi-squared test or Fisher’s test was used when assessing categorical variables. The survival rate was calculated using Kaplan–Meier analysis, and statistical analysis was performed using the log-rank test. Significantly associated variables (p < 0.05) in univariate analysis were included in the multivariate analysis using the Cox proportional hazards model to identify the independent factors. Probability values less than 0.05 were defined as statistically significant factors. Statistical analyses were performed using JMP software (version 16.0; SAS Institute, Cary, NC, USA).
Abbreviations
AUC: area under the curve; CEA: carcinoembryonic antigen; CD: Clavien–Dindo; CI: confidence interval; CRP: C-reactive protein; CSS: cancer-specific survival; HR: hazard ratio; ICPI: inflammation-combined prognostic index; IL: interleukin; NLR: neutrophil-to-lymphocyte ratio; LMR: lymphocyte-to-monocyte ratio; PLR: platelet-to-lymphocyte ratio; pTNM: pathological tumor-node-metastasis; ROC: receiver operating curve; SIR: systemic inflammatory reaction; TNF-α: tumor necrosis factor-α.
CONFLICTS OF INTEREST
Noriyuki Hirahara, Takeshi Matsubara, Shunsuke Kaji, Hikota Hayashi, Yohei Sasaki, Koki Kawakami, Ryoji Hyakudomi, Tetsu Yamamoto, Wataru Tanaka, and Yoshitsugu Tajima have no conflicts of interest or financial ties to disclose.
Ethical statement and consent
All procedures performed in the study involving human participants were in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was registered with the University Hospital Medical Information Network Clinical Trials Registry (UMIN000030472). The protocol of this retrospective study was approved by the Ethical Review Board of Shimane University, Faculty of Medicine (Shimane, Japan). The protocol of this retrospective study was approved by the Ethical Review Board of Shimane University, Faculty of Medicine (Shimane, Japan).
References
1. Schrader H, Menge BA, Belyaev O, Uhl W, Schmidt WE, Meier JJ. Amino acid malnutrition in patients with chronic pancreatitis and pancreatic carcinoma. Pancreas. 2009; 38:416–21. https://doi.org/10.1097/MPA.0b013e318194fc7a. [PubMed].
2. Hanazaki K, Maeda H, Okabayashi T. Relationship between perioperative glycemic control and postoperative infections. World J Gastroenterol. 2009; 15:4122–25. https://doi.org/10.3748/wjg.15.4122. [PubMed].
3. Hirahara N, Tajima Y, Fujii Y, Kaji S, Kawabata Y, Hyakudomi R, Yamamoto T, Taniura T. Controlling Nutritional Status (CONUT) as a prognostic immunonutritional biomarker for gastric cancer after curative gastrectomy: a propensity score-matched analysis. Surg Endosc. 2019; 33:4143–52. https://doi.org/10.1007/s00464-019-06723-z. [PubMed].
4. Hirahara N, Matsubara T, Kaji S, Kawabata Y, Hyakudomi R, Yamamoto T, Uchida Y, Ishitobi K, Takai K, Tajima Y. Glasgow prognostic score is a better predictor of the long-term survival in patients with gastric cancer, compared to the modified Glasgow prognostic score or high-sensitivity modified Glasgow prognostic score. Oncotarget. 2020; 11:4169–77. https://doi.org/10.18632/oncotarget.27796. [PubMed].
5. Okugawa Y, Grady WM, Goel A. Epigenetic Alterations in Colorectal Cancer: Emerging Biomarkers. Gastroenterology. 2015; 149:1204–25.e12. https://doi.org/10.1053/j.gastro.2015.07.011. [PubMed].
6. Yamashita H, Katai H. Systemic inflammatory response in gastric cancer. World J Surg. 2010; 34:2399–400. https://doi.org/10.1007/s00268-010-0703-1. [PubMed].
7. Uhr A, Glick L, Gomella LG. An overview of biomarkers in the diagnosis and management of prostate cancer. Can J Urol. 2020 (Suppl 3); 27:24–27. [PubMed].
8. Gijsbers K, Gouwy M, Struyf S, Wuyts A, Proost P, Opdenakker G, Penninckx F, Ectors N, Geboes K, Van Damme J. GCP-2/CXCL6 synergizes with other endothelial cell-derived chemokines in neutrophil mobilization and is associated with angiogenesis in gastrointestinal tumors. Exp Cell Res. 2005; 303:331–42. https://doi.org/10.1016/j.yexcr.2004.09.027. [PubMed].
9. Ungefroren H, Witte D, Rauch BH, Settmacher U, Lehnert H, Gieseler F, Kaufmann R. Proteinase-Activated Receptor 2 May Drive Cancer Progression by Facilitating TGF-β Signaling. Int J Mol Sci. 2017; 18:2494. https://doi.org/10.3390/ijms18112494. [PubMed].
10. Ueda T, Shimada E, Urakawa T. Serum levels of cytokines in patients with colorectal cancer: possible involvement of interleukin-6 and interleukin-8 in hematogenous metastasis. J Gastroenterol. 1994; 29:423–29. https://doi.org/10.1007/BF02361238. [PubMed].
11. Wu JM, Yang HT, Ho TW, Shun SC, Lin MT. Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients. J Clin Med. 2019; 8:543. https://doi.org/10.3390/jcm8040543. [PubMed].
12. Zhang C, Li Y, Wu Y, Wang L, Wang X, Du J. Interleukin-6/signal transducer and activator of transcription 3 (STAT3) pathway is essential for macrophage infiltration and myoblast proliferation during muscle regeneration. J Biol Chem. 2013; 288:1489–99. https://doi.org/10.1074/jbc.M112.419788. [PubMed].
13. Chiang KC, Tsui KH, Chung LC, Yeh CN, Chen WT, Chang PL, Juang HH. Celastrol blocks interleukin-6 gene expression via downregulation of NF-κB in prostate carcinoma cells. PLoS One. 2014; 9:e93151. https://doi.org/10.1371/journal.pone.0093151. [PubMed].
14. Park JW, Chang HJ, Yeo HY, Han N, Kim BC, Kong SY, Kim J, Oh JH. The relationships between systemic cytokine profiles and inflammatory markers in colorectal cancer and the prognostic significance of these parameters. Br J Cancer. 2020; 123:610–18. https://doi.org/10.1038/s41416-020-0924-5. [PubMed].
15. Rodrigues ISS, Martins-Filho A, Micheli DC, Lima CA, Tavares-Murta BM, Murta EFC, Nomelini RS. IL-6 and IL-8 as Prognostic Factors in Peritoneal Fluid of Ovarian Cancer. Immunol Invest. 2020; 49:510–21. https://doi.org/10.1080/08820139.2019.1691222. [PubMed].
16. Su VY, Lin CS, Hung SC, Yang KY. Mesenchymal Stem Cell-Conditioned Medium Induces Neutrophil Apoptosis Associated with Inhibition of the NF-κB Pathway in Endotoxin-Induced Acute Lung Injury. Int J Mol Sci. 2019; 20:2208. https://doi.org/10.3390/ijms20092208. [PubMed].
17. Lelis FJN, Jaufmann J, Singh A, Fromm K, Teschner AC, Pöschel S, Schäfer I, Beer-Hammer S, Rieber N, Hartl D. Myeloid-derived suppressor cells modulate B-cell responses. Immunol Lett. 2017; 188:108–15. https://doi.org/10.1016/j.imlet.2017.07.003. [PubMed].
18. Hodge G, Barnawi J, Jurisevic C, Moffat D, Holmes M, Reynolds PN, Jersmann H, Hodge S. Lung cancer is associated with decreased expression of perforin, granzyme B and interferon (IFN)-γ by infiltrating lung tissue T cells, natural killer (NK) T-like and NK cells. Clin Exp Immunol. 2014; 178:79–85. https://doi.org/10.1111/cei.12392. [PubMed].
19. Young MR, McCloskey G, Wright MA, Pak AS. Increasing infiltration and activation of CD8+ tumor-infiltrating lymphocytes after eliminating immune suppressive granulocyte/macrophage progenitor cells with low doses of interferon gamma plus tumor necrosis factor alpha. Cancer Immunol Immunother. 1994; 38:9–15. https://doi.org/10.1007/BF01517164. [PubMed].
20. Li H, Huang N, Zhu W, Wu J, Yang X, Teng W, Tian J, Fang Z, Luo Y, Chen M, Li Y. Modulation the crosstalk between tumor-associated macrophages and non-small cell lung cancer to inhibit tumor migration and invasion by ginsenoside Rh2. BMC Cancer. 2018; 18:579. https://doi.org/10.1186/s12885-018-4299-4. [PubMed].
21. Fukagawa D, Sugai T, Osakabe M, Suga Y, Nagasawa T, Itamochi H, Sugiyama T. Protein expression patterns in cancer-associated fibroblasts and cells undergoing the epithelial-mesenchymal transition in ovarian cancers. Oncotarget. 2018; 9:27514–24. https://doi.org/10.18632/oncotarget.25518. [PubMed].
22. Feng X, Li L, Wu J, Zhang L, Sun Z, Li X, Wang X, Yu H, Chang Y, Wu X, Zhou Z, Wang G, Li W, et al. Complete Blood Count Score Model Integrating Reduced Lymphocyte-Monocyte Ratio, Elevated Neutrophil-Lymphocyte Ratio, and Elevated Platelet-Lymphocyte Ratio Predicts Inferior Clinical Outcomes in Adult T-Lymphoblastic Lymphoma. Oncologist. 2019; 24:e1123–31. https://doi.org/10.1634/theoncologist.2018-0789. [PubMed].
23. Ji H, Niu X, Yin L, Wang Y, Huang L, Xuan Q, Li L, Zhang H, Li J, Yang Y, An W, Zhang Q. Ratio of Immune Response to Tumor Burden Predicts Survival Via Regulating Functions of Lymphocytes and Monocytes in Diffuse Large B-Cell Lymphoma. Cell Physiol Biochem. 2018; 45:951–61. https://doi.org/10.1159/000487288. [PubMed].
24. Graziano V, Grassadonia A, Iezzi L, Vici P, Pizzuti L, Barba M, Quinzii A, Camplese A, Di Marino P, Peri M, Veschi S, Alberti S, Gamucci T, et al. Combination of peripheral neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio is predictive of pathological complete response after neoadjuvant chemotherapy in breast cancer patients. Breast. 2019; 44:33–38. https://doi.org/10.1016/j.breast.2018.12.014. [PubMed].
25. Ohe Y, Fushida S, Yamaguchi T, Kinoshita J, Saito H, Okamoto K, Nakamura K, Tajima H, Ninomiya I, Ohta T. Peripheral Blood Platelet-Lymphocyte Ratio Is Good Predictor of Chemosensitivity and Prognosis in Gastric Cancer Patients. Cancer Manag Res. 2020; 12:1303–11. https://doi.org/10.2147/CMAR.S241069. [PubMed].
26. Ha H, Nam AR, Bang JH, Park JE, Kim TY, Lee KH, Han SW, Im SA, Kim TY, Bang YJ, Oh DY. Soluble programmed death-ligand 1 (sPDL1) and neutrophil-to-lymphocyte ratio (NLR) predicts survival in advanced biliary tract cancer patients treated with palliative chemotherapy. Oncotarget. 2016; 7:76604–12. https://doi.org/10.18632/oncotarget.12810. [PubMed].
27. Ishibashi Y, Tsujimoto H, Hiraki S, Kouzu K, Tsuchiya S, Itazaki Y, Yaguchi Y, Horiguchi H, Nomura S, Ito N, Shinto E, Kishi Y, Ueno H. Predictive value of immuno-inflammatory and nutritional measures modulated by neoadjuvant chemotherapy on the response of neoadjuvant chemotherapy and long-term outcomes in patients with esophageal cancer. Oncol Lett. 2020; 19:487–97. https://doi.org/10.3892/ol.2019.11122. [PubMed].
28. Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer. 2017; 20:1–19. https://doi.org/10.1007/s10120-016-0622-4. [PubMed].
29. Sobin LH, Gospodarowicz MK, Wittekind C, and International Union against Cancer. TNM classification of malignant tumours, 7th ed. 2009. Hoboken, NJ: Wiley-Blackwell; 2010. https://www.ncbi.nlm.nih.gov/nlmcatalog/101511218.
30. Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibañes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009; 250:187–96. https://doi.org/10.1097/SLA.0b013e3181b13ca2. [PubMed].

