Introduction
Central nervous system (CNS) tumors, including primary tumors and metastatic or leptomeningeal disease (LMD), are associated with high morbidity and mortality in both adults and children [1–4]. Immunotherapies, including oncolytic virotherapy, offer novel, targeted approaches to treat brain tumors with increased efficacy and reduced toxicity [5]. Oncolytic herpes simplex virus type-1 (oHSV) has shown promise in clinical trials in both pediatric and adult brain tumors [6–9]. These completed trials all utilized intratumoral inoculation of virus via needles or catheters with convection-enhanced delivery or slow-rate infusion, allowing the virus to bypass the blood-brain barrier and facilitating maximum intratumoral concentrations with low systemic toxicity. However, direct inoculation requires an invasive neurosurgical procedure and may disqualify tumors in surgically inaccessible or eloquent brain regions as well as metastatic and/or LMD. Additionally, while repeated administration of virus has demonstrated encouraging effects on survival, it requires repeated neurosurgical procedures [6]. Direct injection into cerebral ventricles and/or intrathecal delivery has the potential to overcome the limitations of intratumoral delivery, but has since been avoided due to concerns of toxicity, even excluding patients where intratumoral administration would lead to ventricular breech [6–9].
With the goal of maximizing the therapeutic potential of oHSV, Kang and Bernstock et al., inoculated ventricles of mice with γ134.5-deleted oHSV (G207) to identify mechanisms of damage with intraventricular therapy (IVT) and potential steps to mitigate toxicity [10]. They found that toxicity was due to damage to the ependymal lining, in part due to viral replication and induction of CD8+ T cells (Figure 1). They therefore hypothesized that the interferon (IFN)-induced protein kinase R phosphorylation of eukaryotic initiation factor-2α (eIF2α) is not initiated effectively in ependymal cells leading to toxicity [11]. As such the research team decided to employ a pretreatment paradigm with the potent IFN inducer polyinosinic-polycytidylic acid (poly I:C) or with IVT low-dose HSV in order to “prime” the ependymal cells. This approach with clinically-relevant oHSV G207 enabled safe delivery of multiple therapeutic doses and in so doing prolonged survival in human and murine metastatic medulloblastoma models.
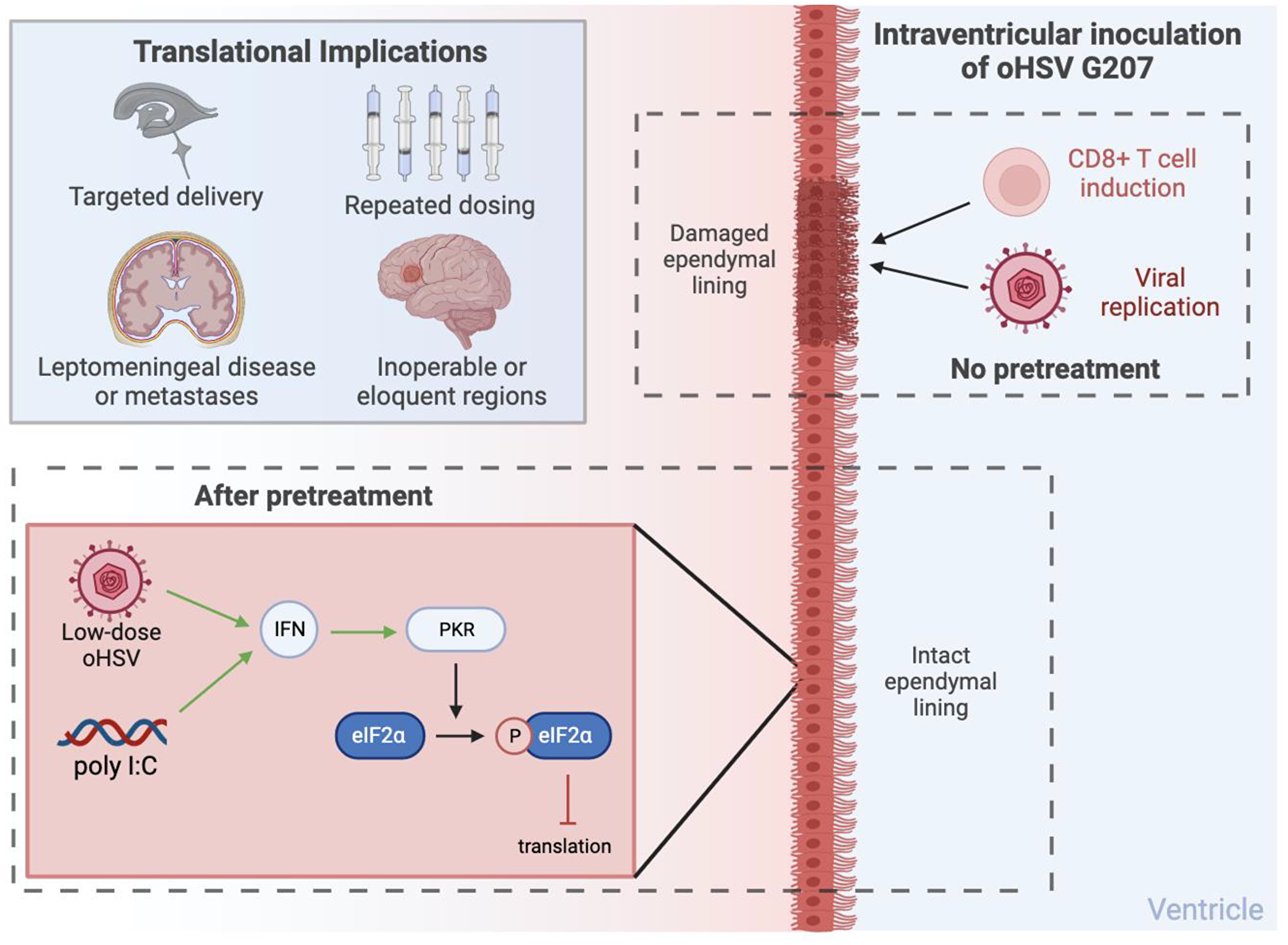
Figure 1: Putative mechanisms of toxicity. Damage of the ependymal lining can be mitigated via pretreatment with IVT low-dose oHSV or poly I:C. Translational implications of such an approach are also highlighted. Made with https://BioRender.com. Abbreviations: oHSV: oncolytic herpes simplex virus; poly I:C: polyinosinic-polycytidylic acid; IFN: interferon; PKR: protein kinase R; eIF2α: eukaryotic initiation factor-2α.
The work has significant translational implications. G207 is the most extensively studied oHSV in preclinical and clinical brain tumor studies and has been proven safe in the CNS of both adults and children [6–9, 12–15]. In a completed phase I trial in children with recurrent/progressive supratentorial high-grade glioma, G207 was safe with promising efficacy results including prolonged responses in some patients and a median overall survival of 12.2 months, which compares favorably with historical survival of approximately 6 months for children at first relapse [9, 16]. This promising efficacy data will need to be confirmed in larger studies and has led to the development of a multi-institutional phase II trial (NCT04482933) in pediatric high-grade glioma at first relapse/progression, which is expected to open in early 2023. Additionally, there is an ongoing first-in-human phase I trial studying intratumoral G207 for recurrent malignant pediatric cerebellar brain tumors [17].
Critically, this preclinical study demonstrated the putative safety/efficacy of IVT G207 and in so doing highlighted a new route for inoculation; such work may allow for the targeting of previously unreachable diseases such as disseminated LMD or metastases as well as tumors in inoperable and/or eloquent regions. This has the potential to expand the variety of tumor types and locations targetable by G207 and increase opportunities for patients that traditionally have had very limited options. Addressing metastases may also lead to lower and more focused doses of radiation thereby reducing treatment-related toxicity, as radiation and oHSV appear to have a synergistic role [18, 19]. Accordingly, we feel that such studies support future clinical trials of G207 through this approach.
IVT would also pave the way for an accessible route for repeated doses of virus as compared to multiple invasive neurosurgical procedures. Increased dosing may increase the efficacy of virotherapy or extend the clinical benefit for longer time periods. A preclinical trial of G207 demonstrated superior efficacy of six intratumoral doses over one with a tenfold higher dose [20]. A recent phase II clinical trial of G47Δ in adults with residual/recurrent supratentorial glioblastoma used a maximum of six intratumoral doses, and patients had a median survival of 20.2 months after virotherapy initiation, which exceeds historical survival in this patient population [6]. Although intravenous delivery of oHSV was safe with some evidence of viral replication in extracranial solid tumors [21], hurdles such as neutralizing antibodies, the blood-brain barrier, and first-pass metabolism prevent adequate virus from reaching intracranial tumors to generate sufficient response. Preclinical trials have attempted to overcome these challenges through a variety of techniques, but have not yet been tested in patients [22–25]. Intraventricular or intrathecal inoculation bypasses many of these barriers allowing for more targeted administration.
Overall, demonstrating the safety and efficacy of IVT with G207 is a significant step towards expanding the capabilities of oHSV, paving the way for new clinical trials, and increasing the potential of an already promising therapy.
CONFLICTS OF INTEREST
JDB has an equity position in Treovir Inc., an oHSV clinical stage company and is a member of the POCKiT Diagnostics and NeuroX1 Board of Scientific Advisors. JDB, KK, and GKF have a Provisional Patent App No. 63/273,577 entitled, “METHODS AND FORMULATIONS RELATED TO THE INTRATHECAL DELIVERY OF ONCOLYTIC VIRUS”. SB has declared that no conflict(s) of interest exists.
References
1. Akmal S, Ginalis EE, Patel NV, Aiken R, Dicpinigaitis AJ, Hanft SJ. Leptomeningeal disease in glioblastoma: endgame or opportunity? J Neurooncol. 2021; 155:107–15. https://doi.org/10.1007/s11060-021-03864-x. [PubMed].
2. Ostrom QT, Cioffi G, Gittleman H, Patil N, Waite K, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Neuro Oncol. 2019; 21:v1–100. https://doi.org/10.1093/neuonc/noz150. [PubMed].
3. Wang N, Bertalan MS, Brastianos PK. Leptomeningeal metastasis from systemic cancer: Review and update on management. Cancer. 2018; 124:21–35. https://doi.org/10.1002/cncr.30911. [PubMed].
4. Lamba N, Wen PY, Aizer AA. Epidemiology of brain metastases and leptomeningeal disease. Neuro Oncol. 2021; 23:1447–56. https://doi.org/10.1093/neuonc/noab101. [PubMed].
5. Suryawanshi YR, Schulze AJ. Oncolytic Viruses for Malignant Glioma: On the Verge of Success? Viruses. 2021; 13:1294. https://doi.org/10.3390/v13071294. [PubMed].
6. Todo T, Ito H, Ino Y, Ohtsu H, Ota Y, Shibahara J, Tanaka M. Intratumoral oncolytic herpes virus G47Δ for residual or recurrent glioblastoma: a phase 2 trial. Nat Med. 2022; 28:1630–39. https://doi.org/10.1038/s41591-022-01897-x. [PubMed].
7. Markert JM, Liechty PG, Wang W, Gaston S, Braz E, Karrasch M, Nabors LB, Markiewicz M, Lakeman AD, Palmer CA, Parker JN, Whitley RJ, Gillespie GY. Phase Ib trial of mutant herpes simplex virus G207 inoculated pre-and post-tumor resection for recurrent GBM. Mol Ther. 2009; 17:199–207. https://doi.org/10.1038/mt.2008.228. [PubMed].
8. Markert JM, Razdan SN, Kuo HC, Cantor A, Knoll A, Karrasch M, Nabors LB, Markiewicz M, Agee BS, Coleman JM, Lakeman AD, Palmer CA, Parker JN, et al. A phase 1 trial of oncolytic HSV-1, G207, given in combination with radiation for recurrent GBM demonstrates safety and radiographic responses. Mol Ther. 2014; 22:1048–55. https://doi.org/10.1038/mt.2014.22. [PubMed].
9. Friedman GK, Johnston JM, Bag AK, Bernstock JD, Li R, Aban I, Kachurak K, Nan L, Kang KD, Totsch S, Schlappi C, Martin AM, Pastakia D, et al. Oncolytic HSV-1 G207 Immunovirotherapy for Pediatric High-Grade Gliomas. N Engl J Med. 2021; 384:1613–22. https://doi.org/10.1056/NEJMoa2024947. [PubMed].
10. Kang KD, Bernstock JD, Totsch SK, Gary SE, Rocco A, Nan L, Li R, Etminan T, Han X, Beierle EA, Eisemann T, Wechsler-Reya RJ, Bae S, et al. Safety and Efficacy of Intraventricular Immunovirotherapy with Oncolytic HSV-1 for CNS Cancers. Clin Cancer Res. 2022; 28:5419–30. https://doi.org/10.1158/1078-0432.CCR-22-1382. [PubMed].
11. Chou J, Chen JJ, Gross M, Roizman B. Association of a M(r) 90,000 phosphoprotein with protein kinase PKR in cells exhibiting enhanced phosphorylation of translation initiation factor eIF-2 alpha and premature shutoff of protein synthesis after infection with gamma 134.5- mutants of herpes simplex virus 1. Proc Natl Acad Sci U S A. 1995; 92:10516–20. https://doi.org/10.1073/pnas.92.23.10516. [PubMed].
12. Friedman GK, Bernstock JD, Chen D, Nan L, Moore BP, Kelly VM, Youngblood SL, Langford CP, Han X, Ring EK, Beierle EA, Gillespie GY, Markert JM. Enhanced Sensitivity of Patient-Derived Pediatric High-Grade Brain Tumor Xenografts to Oncolytic HSV-1 Virotherapy Correlates with Nectin-1 Expression. Sci Rep. 2018; 8:13930. https://doi.org/10.1038/s41598-018-32353-x. [PubMed].
13. Markert JM, Medlock MD, Rabkin SD, Gillespie GY, Todo T, Hunter WD, Palmer CA, Feigenbaum F, Tornatore C, Tufaro F, Martuza RL. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther. 2000; 7:867–74. https://doi.org/10.1038/sj.gt.3301205. [PubMed].
14. Bernstock JD, Vicario N, Li R, Nan L, Totsch SK, Schlappi C, Gessler F, Han X, Parenti R, Beierle EA, Whitley RJ, Aban I, Gillespie GY, et al. Safety and efficacy of oncolytic HSV-1 G207 inoculated into the cerebellum of mice. Cancer Gene Ther. 2020; 27:246–55. https://doi.org/10.1038/s41417-019-0091-0. [PubMed].
15. Ghajar-Rahimi G, Kang KD, Totsch SK, Gary S, Rocco A, Blitz S, Kachurak K, Chambers MR, Li R, Beierle EA, Bag A, Johnston JM, Markert JM, et al. Clinical advances in oncolytic virotherapy for pediatric brain tumors. Pharmacol Ther. 2022; 239:108193. https://doi.org/10.1016/j.pharmthera.2022.108193. [PubMed].
16. Kline C, Felton E, Allen IE, Tahir P, Mueller S. Survival outcomes in pediatric recurrent high-grade glioma: results of a 20-year systematic review and meta-analysis. J Neurooncol. 2018; 137:103–10. https://doi.org/10.1007/s11060-017-2701-8. [PubMed].
17. Bernstock JD, Bag AK, Fiveash J, Kachurak K, Elsayed G, Chagoya G, Gessler F, Valdes PA, Madan-Swain A, Whitley R, Markert JM, Gillespie GY, Johnston JM, Friedman GK. Design and Rationale for First-in-Human Phase 1 Immunovirotherapy Clinical Trial of Oncolytic HSV G207 to Treat Malignant Pediatric Cerebellar Brain Tumors. Hum Gene Ther. 2020; 31:1132–39. https://doi.org/10.1089/hum.2020.101. [PubMed].
18. Bradley JD, Kataoka Y, Advani S, Chung SM, Arani RB, Gillespie GY, Whitley RJ, Markert JM, Roizman B, Weichselbaum RR. Ionizing radiation improves survival in mice bearing intracranial high-grade gliomas injected with genetically modified herpes simplex virus. Clin Cancer Res. 1999; 5:1517–22. [PubMed].
19. Advani SJ, Sibley GS, Song PY, Hallahan DE, Kataoka Y, Roizman B, Weichselbaum RR. Enhancement of replication of genetically engineered herpes simplex viruses by ionizing radiation: a new paradigm for destruction of therapeutically intractable tumors. Gene Ther. 1998; 5:160–65. https://doi.org/10.1038/sj.gt.3300546. [PubMed].
20. Chahlavi A, Rabkin S, Todo T, Sundaresan P, Martuza R. Effect of prior exposure to herpes simplex virus 1 on viral vector-mediated tumor therapy in immunocompetent mice. Gene Ther. 1999; 6:1751–58. https://doi.org/10.1038/sj.gt.3301003. [PubMed].
21. Streby KA, Currier MA, Triplet M, Ott K, Dishman DJ, Vaughan MR, Ranalli MA, Setty B, Skeens MA, Whiteside S, Yeager ND, Haworth KB, Simpson K, et al. First-in-Human Intravenous Seprehvir in Young Cancer Patients: A Phase 1 Clinical Trial. Mol Ther. 2019; 27:1930–38. https://doi.org/10.1016/j.ymthe.2019.08.020. [PubMed].
22. Ikeda K, Ichikawa T, Wakimoto H, Silver JS, Deisboeck TS, Finkelstein D, Harsh GR 4th, Louis DN, Bartus RT, Hochberg FH, Chiocca EA. Oncolytic virus therapy of multiple tumors in the brain requires suppression of innate and elicited antiviral responses. Nat Med. 1999; 5:881–87. https://doi.org/10.1038/11320. [PubMed].
23. Liu R, Martuza RL, Rabkin SD. Intracarotid delivery of oncolytic HSV vector G47Delta to metastatic breast cancer in the brain. Gene Ther. 2005; 12:647–54. https://doi.org/10.1038/sj.gt.3302445. [PubMed].
24. Yoo JY, Pradarelli J, Haseley A, Wojton J, Kaka A, Bratasz A, Alvarez-Breckenridge CA, Yu JG, Powell K, Mazar AP, Teknos TN, Chiocca EA, Glorioso JC, et al. Copper chelation enhances antitumor efficacy and systemic delivery of oncolytic HSV. Clin Cancer Res. 2012; 18:4931–41. https://doi.org/10.1158/1078-0432.CCR-12-0697. [PubMed].
25. Leoni V, Gatta V, Palladini A, Nicoletti G, Ranieri D, Dall’Ora M, Grosso V, Rossi M, Alviano F, Bonsi L, Nanni P, Lollini PL, Campadelli-Fiume G. Systemic delivery of HER2-retargeted oncolytic-HSV by mesenchymal stromal cells protects from lung and brain metastases. Oncotarget. 2015; 6:34774–87. https://doi.org/10.18632/oncotarget.5793. [PubMed].

