Introduction
The oral mucositis (OM) is a mucosal alteration that usually arises from oncological treatments, such as chemotherapy, and it is characterized by an inflammatory process that promotes the development of painful ulcerations in the oral mucous membranes [1].
Among the most used chemotherapeutic drugs in antineoplasic therapies, the 5-Flourouracil (5-FU) can result in development of OM as a side-effect of its application [2]. Thus, the execution of a preventive and curative treatment to reduce OM incidence seems to be necessary. The regular medical interventions are palliative conducts related to releasing the OM symptomatology [3], such as the use of cryotherapy [4], topical anesthetics, antifungals, antiseptics, antivirals, antibiotics, anti-inflammatory drugs inhibitors of COX-2 and/or prostaglandin E2, protective agents of the mucosa, vitamins E and A [5], use of low-level laser therapy [6–9], and phytotherapy [10–12].
The use of phytotherapy shows satisfactory results in OM treatments, such as: chamomile [13–15], bilberry extract [16], Hydroalcoholic extract of Carumcarvi L [17], propolis [18] and eucalyptus hydroalcoholic extract [19]. Even though that herbal medicine is a very ancient practice, the studies related to the medical use of plants and its acceptance became more popular in the last years [20]. The plants are processed and their active substances are extracted and transformed into essential oils, which can be administered topically [21] or systemically [22]. The topical formulation presents higher acceptability by the patients, the prompt absorption and rapid onset of action [23]. However, the practice of phytotherapy is still hindered due to the lack of technical-scientific documentation and studies demonstrating clear clinical evidences and advantages of its use [20], as well as its pharmacological effects, genotoxicity and quality control, which must be overcome [24–29].
Andiroba (Carapa guianensis Aubl) is a plant that belongs to the Meliaceae family [30], and it will be found in Amazon region [31–34], southern Central America, Colombia, Venezuela, Suriname, French Guiana, Brazil, Peru, Paraguay and the Caribbean Islands [35, 36]. In Brazil, it’s found in North (states of Acre, Amazonas, Amapá, Pará) region [37] and Northeast (Maranhão state) regions [38]. The tree evidences good pharmacological properties considering its variety of components, such as flowers, leaves and stems extracts, but the oil demonstrated better medicinal effects when obtained by seeds pressing [39]. The seed oil presents yellowish and thick consistency and a very bitter taste (Meliacin presence in the composition), and are located into 6–8 cm fruits [40].
Andiroba’s oil is composed by saponifiable (95%) and unsaponifiable substances (2 a 5%), and within the saponifiable profile, it is important to highlight the presence of essential fatty acids, such as oleic, palmitic, stearic and linoleic acids, which demonstrate higher notoriety in medical applications [36, 37]. Studies have shown that essential fatty acids are the catalyzing agents of the healing process [38], stimulating cell proliferation, collagen production, antimicrobial, anti-inflammatory and antioxidant action [41–49]. The andiroba’s oil shows many therapeutic finalities, due its natural repellent [49], dermatological properties [50, 51], as well as its excellent anti-inflammatory, antibacterial performance, wound healing activity [52, 53], antiparasitic, insecticide [54, 55], anti-allergic effect [56], and antinociceptive responses [57, 58].
The andiroba extract in wounds provides acceleration in healing process, with increase of contraction rate and local re-epithelialization, resulting in the complete closure of wounds [52]. Cicatricial effect was evaluated in different concentrations of andiroba oil (Carapa guianensis Aubl) in wound healing (oral mucositis), demonstrating an excellent cicatricial effect and acceleration of this process [28]. A clinical research in cancer patients evaluating the potential of andiroba oil cicatricial compared to low-power laser showed a significant improvement in the clinical picture of oral mucositis lesions and the symptomatology [29].
The use of laser therapy is an alternative resource for treating OM due to its biostimulation potential which increases the ulcerated area healing process, promoting physiologic responses, modulating the inflammation, accelerating wound healing and relieving pain. As a non-invasive treatment, it’s a therapy method with large acceptance by most patients, also, because it works in lessen the pain symptomatology since the first use. Also, it potentializes the complete tissue local revascularization until the fifteenth day and an intense tissue healing already in the second to fourth day. The Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) published a clinical practice guidelines for mucositis recommended the use of laser therapy in patients undergoing anti-cancer therapy [59, 60].
The low-level laser therapy (LLLT) is the best standard treatment and the most efficient method in treating OM. Similarly, the andiroba oil presents great potential for the treatment of inflammatory diseases. Thus, this study aims to evaluate the healing and toxicological effects of andiroba oil, compared to the LLLT outcomes, observing if andiroba presents a similar/higher potential than the LLLT.
Results
GC-MS analysis of the oils
To verify the andiroba oil characterization, a layout of its chromatographic profile was made using gas chromatography, in which 12 saponifiable compounds were found, by the lipidic analysis. Among the compounds identified in the andiroba oil, the most expressive percentages were oleic acid (47.33%), palmitic acid (31.46%), linoleic acid (8.98%) and stearic acid (7.12%), as described in Table 1.
Table 1: Composition of the fatty acids present in andiroba oil (C. guianensis)
| Real Time | Compounds | Composition % |
|---|---|---|
| 7,199 | Heptanoic acid | 0,3989 |
| 13,674 | Lauric acid | 1,1572 |
| 15,883 | Myristic acid | 0,6394 |
| 17,982 | Palmitic acid | 31,4641 |
| 18,193 | Palmitoleic acid | 0,9184 |
| 18,862 | Heptadecanoic acid | 0,1154 |
| 19,845 | Stearic acid | 7,1279 |
| 20,064 | Oleic acid | 47,3356 |
| 20,428 | Linoleic acid | 8,9869 |
| 20,966 | Linolenic Acid | 0,2501 |
| 21,530 | Arachidic acid | 1,0897 |
| 23,562 | Behenic acid | 0,5164 |
To identify the most important lipidic compounds inside the andiroba oil, it was submitted to a chromatographic profile (Figure 1).
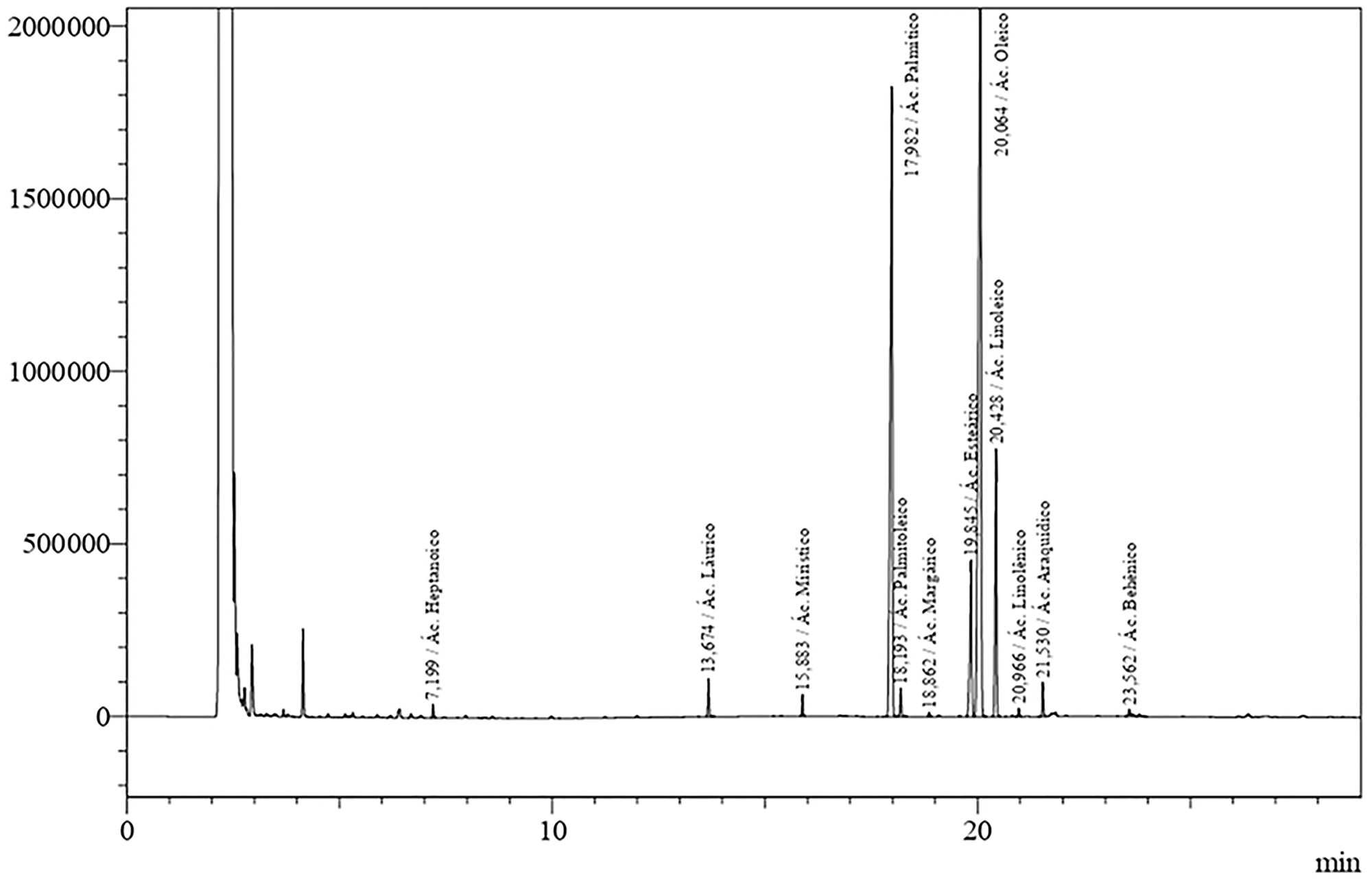
Figure 1: Chromatographic profile of C. Guianensis in natura oil. Presentation of the lipid composition based on Table 1.
Histopathological and clinical analysis
The clinical and histopathological evaluation of mucositis was performed by two previously trained examiners for each analysis. The scores used in research were based in “Lima et al. (2005) [61] modified” classification. To clarify clinical and histopathological results, we provide the Figures 2 and 3, corresponding to days 4, 8, 12 and 15 and their respective treatments.
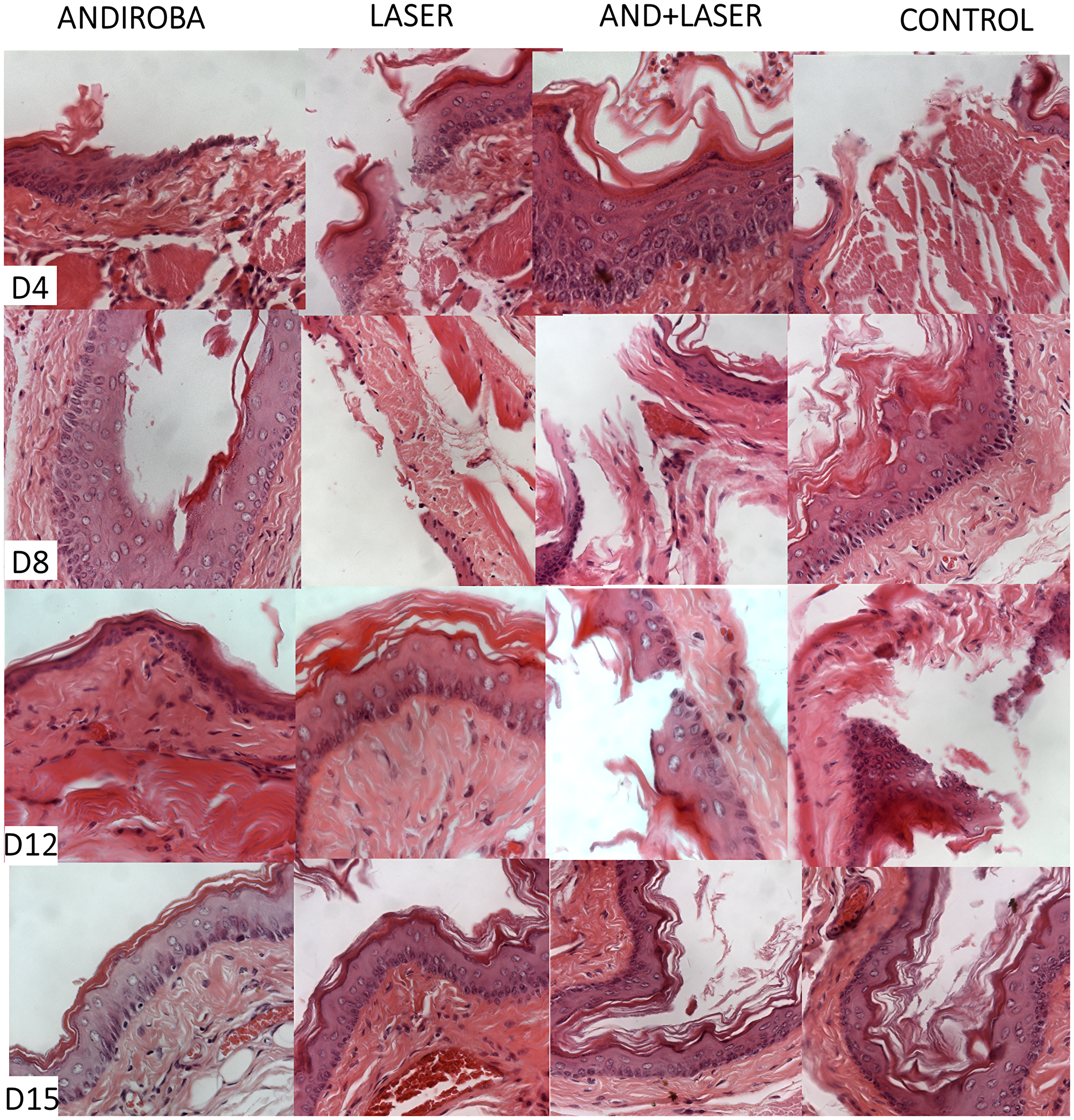
Figure 2: Histopathological evaluation of oral mucositis on days 4, 8, 12 and 15.
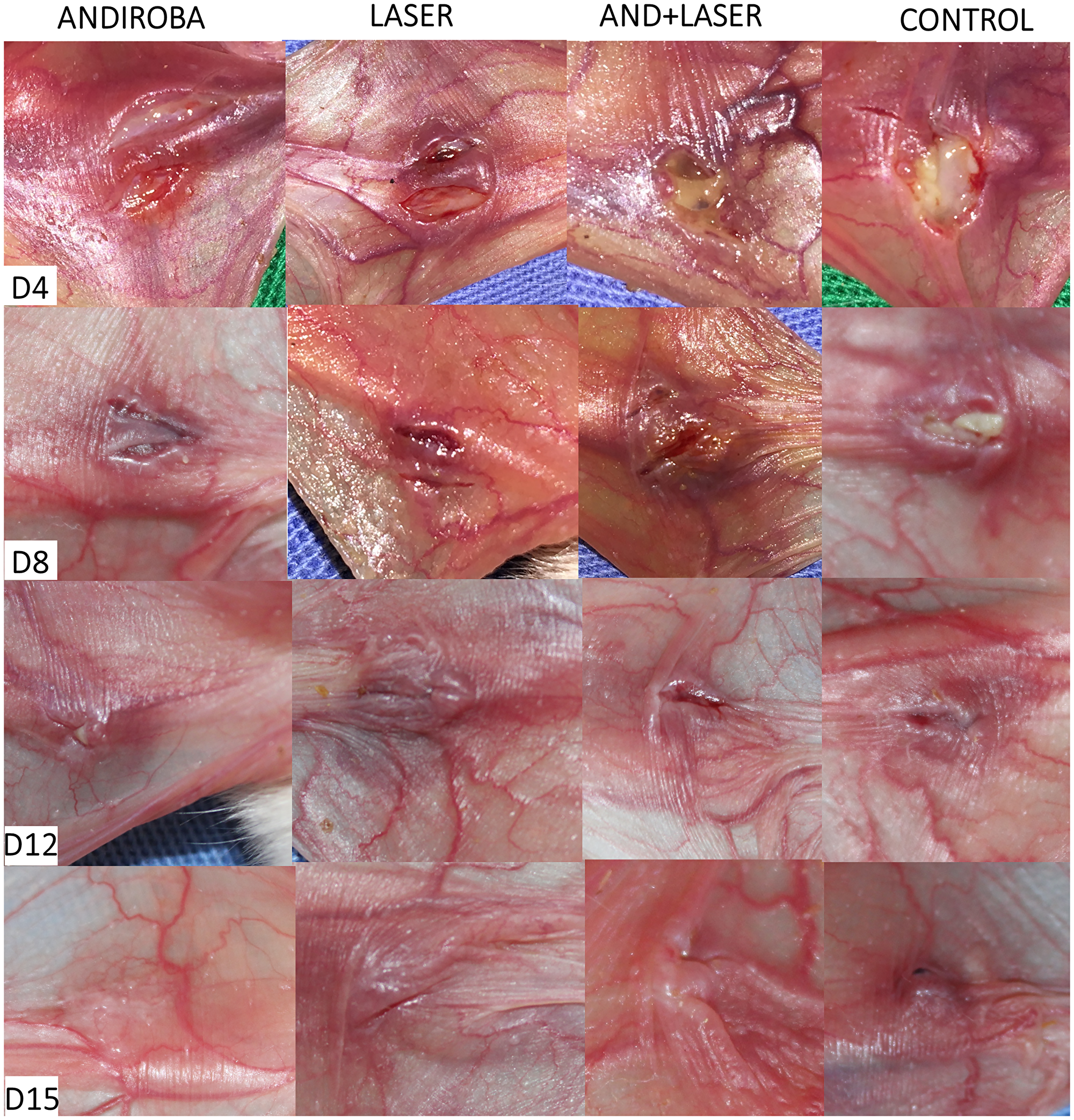
Figure 3: Clinical evaluation of oral mucositis in hamsters on days 4, 8, 12 and 15.
The images and glass slides analyzed corresponded to the mucositis and their respective treatments on days 4, 8, 12 and 15, as expressed in Table 2.
Table 2: Description of the analyzes performed for clinical evaluations of oral mucositis in hamsters treated on days 4, 8, 12 and 15
| Group | N | Mean | Median | SD | Variance | Minimum | Maximum | |
|---|---|---|---|---|---|---|---|---|
| D4 | ADB* | 14 | 2.29 | 2.00 | 0.469 | 0.220 | 2 | 3 |
| LASER* | 14 | 2.29 | 2.00 | 0.726 | 0.527 | 1 | 4 | |
| ADB + LASER | 14 | 2.57 | 3.00 | 0.514 | 0.264 | 2 | 3 | |
| CONTROL | 14 | 3.07 | 3.00 | 0.616 | 0.379 | 2 | 4 | |
| D8 | ADB* | 14 | 1.71 | 2.00 | 0.611 | 0.374 | 1 | 3 |
| LASER* | 14 | 1.86 | 2.00 | 0.864 | 0.747 | 1 | 4 | |
| ADB + LASER* | 14 | 1.93 | 2.00 | 0.616 | 0.379 | 1 | 3 | |
| CONTROL | 14 | 2.64 | 3.00 | 0.633 | 0.401 | 2 | 4 | |
| D12 | ADB | 14 | 1.50 | 1.00 | 0.650 | 0.423 | 1 | 3 |
| LASER | 14 | 1.50 | 1.50 | 0.519 | 0.269 | 1 | 2 | |
| ADB + LASER | 14 | 1.64 | 1.50 | 0.745 | 0.555 | 1 | 3 | |
| CONTROL | 14 | 1.71 | 2.00 | 0.726 | 0.527 | 1 | 3 | |
| D15 | ADB | 14 | 1.14 | 1.00 | 0.363 | 0.132 | 1 | 2 |
| LASER | 14 | 1.14 | 1.00 | 0.363 | 0.132 | 1 | 2 | |
| ADB + LASER | 14 | 1.21 | 1.00 | 0.426 | 0.181 | 1 | 2 | |
| CONTROL | 14 | 1.21 | 1.00 | 0.426 | 0.181 | 1 | 2 |
In the clinical analysis on days 4 and 8, the “laser” group and “andiroba” group presented significant statistical differences when compared to the control group (p < 0.05, respectively), evidencing a possible similarity in their clinical efficiencies. For the better enlightenment of this difference, we provide the Figure 2.
In the clinical analysis, significant difference was noticed (p < 0.05) between “laser” and “andiroba” groups compared to positive control group. By analyzing the means of positive control group (3.07) and “laser associated to andiroba” group (2.57), the groups “laser” and “andiroba” presented lower means (2.28), evidencing similar clinical results on day 4 for both treatments. On day 8, the positive control group (2.64) and “laser associated to andiroba” group (1.92) demonstrated higher means in comparison to “laser” (1.85) and “andiroba” (1.71) groups. On days 12 and 15, there was no significative difference to be found when comparing the groups, however the scores presented higher means in positive control group, followed by the treatments association – laser and andiroba.
On day 4 of histopathological analysis, it was observed that groups “andiroba” and “laser” presented significative statistical differences in comparison to control group (p < 0.05), which had the higher score means (4.07) in comparison to groups “laser associated to andiroba” (3.50), “laser” (3.21) and “andiroba” (3.07). On day 8, the groups “positive control” (3.28) and “laser associated to andiroba” (2.57) presented higher means in comparison to groups “laser” and “andiroba” (2.35 and 2.42, respectively). On days 12 and 15, there was no significative statistical difference between groups, however the groups “andiroba” and “laser” remained with the lower score means in comparison the other groups (Table 3) (Figure 3).
Table 3: Description of the histopathological analysis to oral mucositis in hamsters treated on days 4, 8, 12 and 15
| Groups | N | Mean | Median | SD | Variance | Minimum | Maximum | |
|---|---|---|---|---|---|---|---|---|
| D4 | ADB* | 14 | 3.07 | 3.00 | 0.829 | 0.687 | 2 | 4 |
| LASER* | 14 | 3.21 | 3.00 | 0.975 | 0.951 | 1 | 5 | |
| ADB + LASER | 14 | 3.50 | 4.00 | 0.650 | 0.423 | 2 | 4 | |
| CONTROL | 14 | 4.07 | 4.00 | 0.616 | 0.379 | 3 | 5 | |
| D8 | ADB* | 14 | 2.36 | 2.00 | 0.633 | 0.401 | 2 | 4 |
| LASER* | 14 | 2.43 | 2.00 | 1.016 | 1.033 | 1 | 5 | |
| ADB + LASER | 14 | 2.57 | 2.00 | 0.938 | 0.879 | 1 | 4 | |
| CONTROL | 14 | 3.29 | 3.00 | 0.726 | 0.527 | 2 | 4 | |
| D12 | ADB | 14 | 2.64 | 2.50 | 1.216 | 1.478 | 1 | 4 |
| LASER | 14 | 2.29 | 2.00 | 0.611 | 0.374 | 1 | 3 | |
| ADB + LASER | 14 | 2.93 | 3.00 | 0.917 | 0.841 | 1 | 4 | |
| CONTROL | 14 | 2.14 | 2.00 | 0.770 | 0.593 | 1 | 4 | |
| D15 | ADB | 14 | 2.00 | 2.00 | 0.784 | 0.615 | 1 | 3 |
| LASER | 14 | 1.86 | 1.50 | 0.949 | 0.901 | 1 | 3 | |
| ADB + LASER | 14 | 1.71 | 2.00 | 0.726 | 0.527 | 1 | 3 | |
| CONTROL | 14 | 1.86 | 2.00 | 0.663 | 0.440 | 1 | 3 |
The OM decrease on 4,8,12 and 15 days can be observed in Figures 4 and 5.
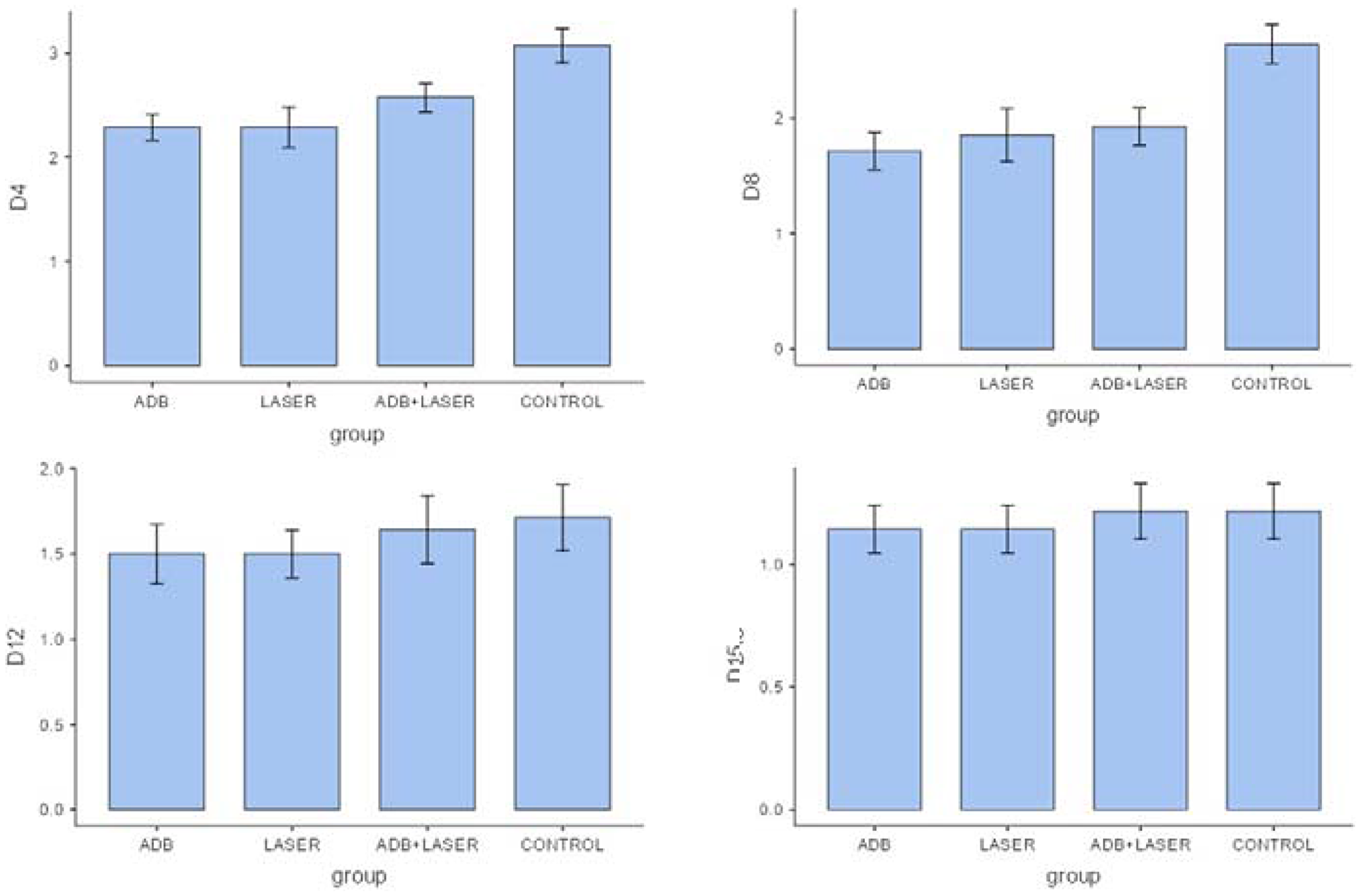
Figure 4: Comparison between clinical analyzes of different groups and days.
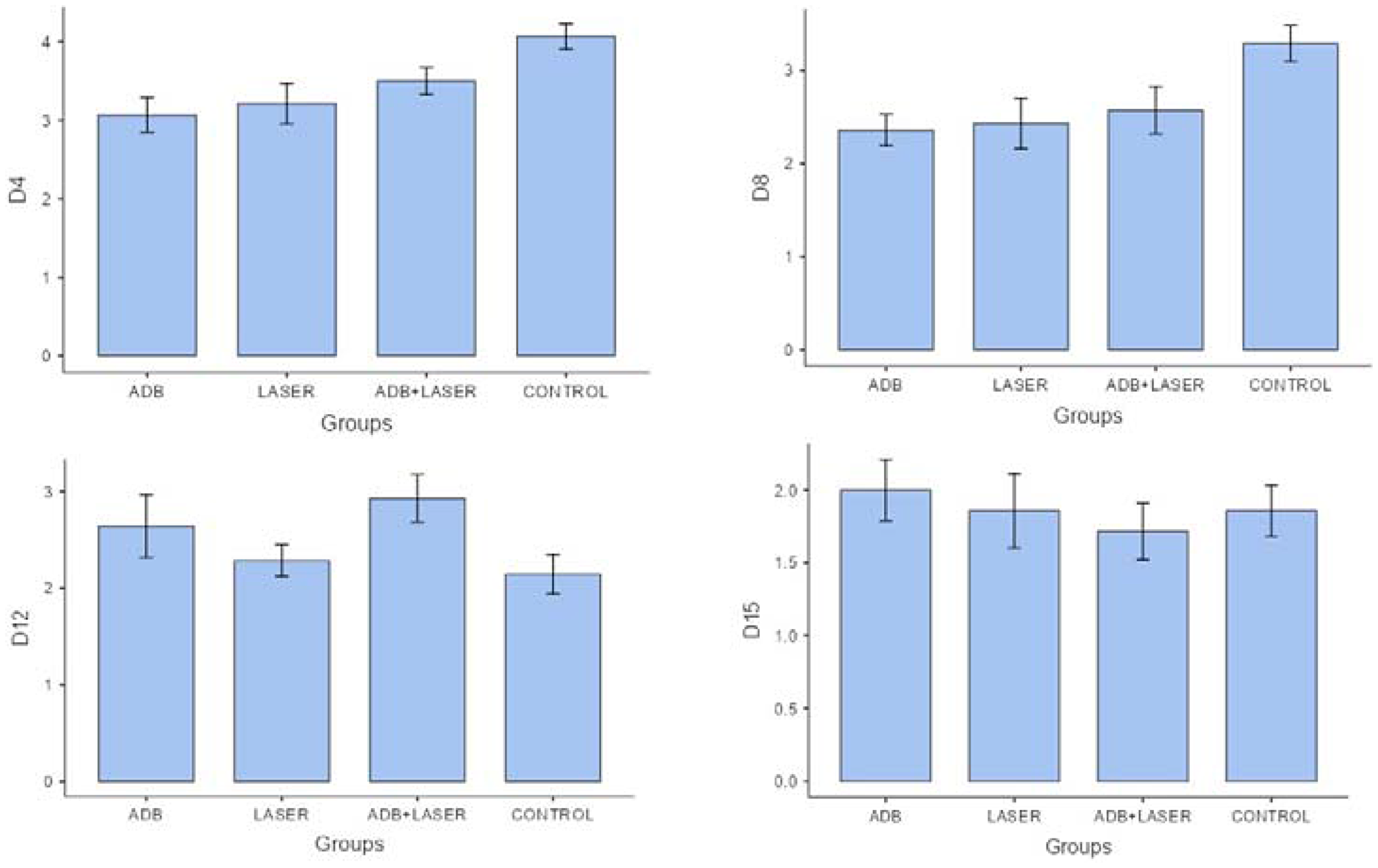
Figure 5: Comparison between histopathological analysis of different groups and days.
Comet test assay
To evaluate genotoxicity, an in vivo comet test was performed, which was followed by a third previously trained examiner analysis of the glass slides. The method applied for this study was based on the guidelines of Organization for Economic Co-operation and Development (OECD). After the counting of cells, a damage index was established (DI) and a final mean is obtained for each sample. The calculation of DI was made by the sum of the products from the score with the number of damages regarding to each level (Figure 6). For better comprehension of the test results, we provide the Table 4. To clearly observe these results, the Figure 7 is combined with a graphic of the DI means of the groups in matter.
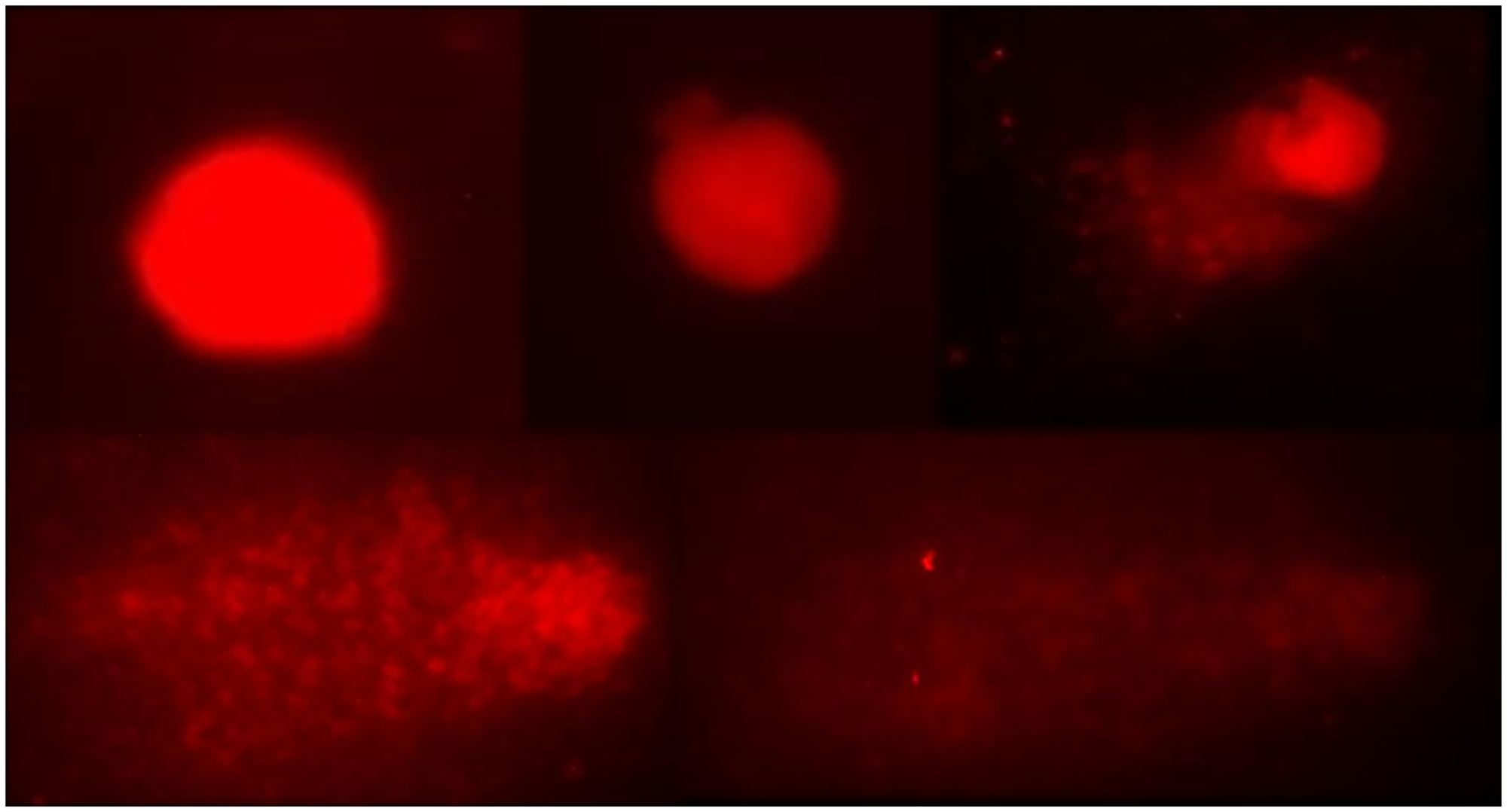
Figure 6: Representation of classes 0 to 4 in the visual classification of comets.
Table 4: Observation the DI (damage index) to the DNA molecule in different groups
| Groups | Average | Standard Deviation |
|---|---|---|
| NC | 24.62 | 1.88 |
| LASER | 30.37 | 7.72 |
| ADB | 19.16 | 2.08 |
| ADB + LASER | 47.83 | 8.09 |
| CICLOPHOS | 125.83 | 10.53 |
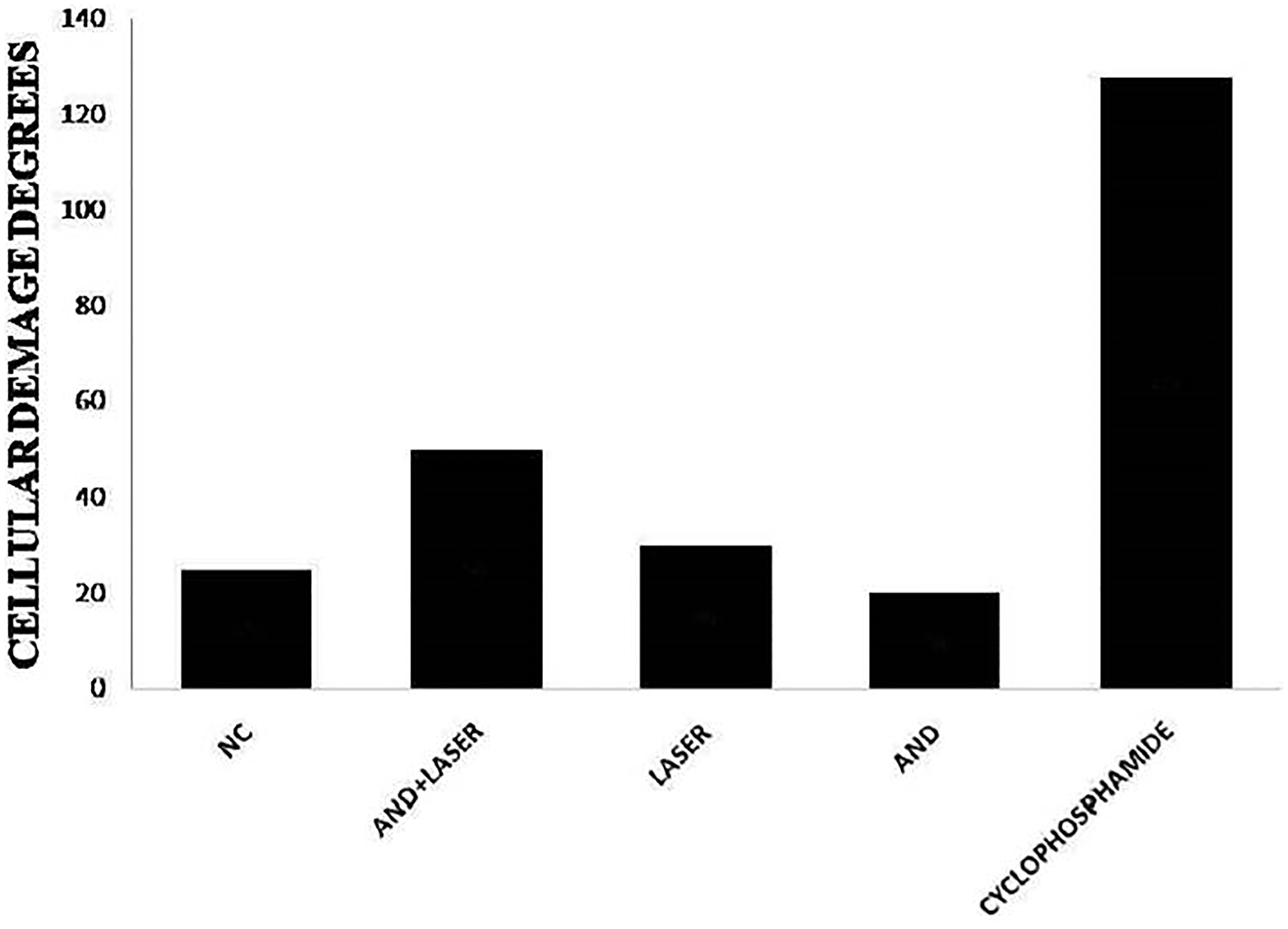
Figure 7: Analysis of the genotoxic effect on the DNA molecules of the cells analyzed in hamsters of the groups submitted to treatments and the cyclophosphamide group (control group).
In Table 4, it is possible to observe the DI to the DNA molecule in different groups. It is also possible to observe an expressive result on cyclophosphamide groups, however that is an expected result, due the fact that this group is a control for the test. The group cyclophosphamide, when compared to group negative control, presented significant statistical difference. The groups andiroba, laser and laser associated to andiroba did not present any significant statistical difference when compared to each other. Although the group andiroba presented lower means (19.16) when compared to the group negative control (24.62), they did not present any significant difference. However, it was observed that the association between laser andiroba treatments, when compared to the control, presented statistical difference (p < 0.003), and the association presented expressive means (47.83), when compared to the groups negative control means (24.62).
DISCUSSION
The evaluation of chemo inducted scarring activity of OM in hamster and the search for efficient treatment with decrease of genotoxicity were the guides for this research. This study was guided by clinical and histopathological investigations about the scarring activity on mucosae of hamsters subjected to grooving and OM induction using chemotherapy drugs (5-Fluorouracila), with posterior treatment by laser and andiroba oil. Besides, to evaluate the use viability of these treatments, their genotoxicity was analyzed, in addition to oil characteristics and its lipidic components for clearer knowledge of its particularities.
The low frequency laser therapy is considered a gold standard in OM, due to its efficiency in the process of healing acceleration, promoting inflammation and pain decrease. The laser luminous energy is converted into useful energy for the cell and is absorbed by chromophores inside mitochondria, essential in the cellular respiratory chain. This process results in the increase of adenosine triphosphate production (ATP), the source of cellular energy, aiding in the proliferation and production of proteins, intensifying cellular mitosis, therefore promoting tissue repair [62] and metabolism acceleration, promoting an anti-inflammatory effect and stimulating the production of collagen and angiogenesis [63–66]. However, due to its high cost, the OM treatment is restricted to a limited number of patients [67–69]. Thus, there is a constant search in the scientific community to enable and ease an accessible treatment for all.
Studies show that this oil presents saponifiable compounds, such as the fatty acids, emphasizing palmitic, oleic, stearic and linoleic acids and unsaponifiable compounds, like limonoids, which present higher visibility on therapeutic effects [36, 37]. This study analyzed the lipidic composition of andiroba oil and it was possible to observe that, in the saponifiable portion, the essential acids presenting more expressive percentages were the oleic (47.33%), palmitic (31.46%), linoleic (8.98%) and stearic (7.12%) oils. Among these, the linoleic oil presents an important role in healing acceleration process, since it is a fundamental component in the collagenase regulation, metalloprotein production and induction of granulation [70, 71].
In vitro and in vivo studies show benefits of fatty acids present in vegetable oils in wound healing, stimulation of cell proliferation and collagen production. In addition, its use is related to the fatty acids’ antimicrobial, anti-inflammatory and antioxidant action [39–48]. Oleic, linolenic and linoleic acids are known in literature to be initiators of anti-inflammatory mediators [32]. Studies have evaluated the topical application of linoleic acid in pressure ulcers in bedridden patients [72] and, experimentally, in mice with application of oleic or linoleic acid for 16 days [73]. In both studies, positive results were observed, with an acceleration of tissue repair within 48 hours of wound induction. It is believed that the positive results in the healing process associated to these fatty acids occurs through increased production of nitric oxide, which results in overexpression of free radicals, helping inflammatory response, in addition to activating macrophages and fibroblasts, stimulating collagen and keratinocyte production, as well as angiogenesis, providing the acceleration of local re-epithelialization process [74, 75]. Another fatty acid of great importance is arachidonic acid, which copes with the inflammatory cells migration, stimulates elastase, angiogenesis and consequent wound healing [76, 77].
Aiming the excellent effects of andiroba oil, this study evaluated its scarring potential in the OM treatment, in which can be observed that group “andiroba” and group “laser” presented statistical difference (p < 0.05) when compared to group “control” in histopathological and clinical analysis, on the 4th day of experiment. In this period, several injuries are clinically found with intense hyperemia and erythema, presenting hemorrhage, extensive ulcers and abscesses in some cases. At microscopic level, the injuries presented moderate to intense cellular engorgement, intense cellular infiltration, with polymorphonuclear leucocytes predominance. Thus, hemorrhagic areas, edemas with extensive ulcers predominance, and, in some cases, abscesses. The best result in the OM treatment was obtained by group “andiroba”, with laser treatment showing similar results, even though the associated “laser and andiroba” treatment presented higher means, showing more intense clinical injuries. This study agrees to Wanzeler et al. [27], whom, using clinical and histopathological evaluation of OM under treatment with several andiroba concentrations, observed an intense inflammatory process on 4º day of experiment, with extensive ulcers and presence of abscesses, and the group “andiroba” presented expressive results in comparison to group control.
On day 8, it is possible to notice, by the clinical images of the injuries, intense hyperemia and erythema, hemorrhage and small ulcers (up to 1 cm diameter), and yet the presence of scarring tissue and absence of abscesses. In microscopic analysis, it can be observed moderate presence of vascular engorgement with vacuolation, moderate to intense cellular infiltration with mononuclear leucocytes predominance, presence of hemorrhagic areas, edemas, and, in some cases, ulcers/abscesses. It is possible to observe that the means of andiroba and laser treatments, when isolated, remained low in comparison to the association of andiroba and the control groups. The “laser” and “andiroba” treatments presented the best results. In this scenario, studies regarding OM treatment, using similar therapeutic methods, also presented positive results and partial decrease in the use of topical andiroba [27, 30] and laser [78], both on 8th day of treatment, in agreement to this study, with decrease injury intensity, suggesting the acceleration of the healing process.
The data of this study regarding the laser therapy agree to data presented by literature, which can be explained by the increase in cellular division promoted by the laser, provoking the tissue regeneration, stimulation of fibroblast production and then, softening the OM peak gravity, reducing ulcers duration and promoting pain relief, which are essential in the healing process [78]. Thus, it is possible to comprehend the reason why on days 12 and 15 of experiment, the laser group presents expressive scarring results, with advanced process of tissue repair. The andiroba presented slightly better results than the laser, demonstrating its powerful anti-inflammatory and scarring actions.
The scientific literature demonstrates a relation between wound healing in injured tissues and the intracellular inhibition of nitric oxide (NO) [79]. These factors induce anti-inflammatory agents production and, consequently, inflammation modulation [80]. The limonoids contained in andiroba inhibit the production of nitric oxide induced by liposaccharides and inhibitors of macrophage activation [81, 82]. The effects of low power laser irradiation on cells are mediated by nitric oxide [83]. These factors can be corroborating to the similar healing results in andiroba group and laser group, isolated. The association of laser and andiroba did not show a promising effect, maybe because it could be inducing a dysregulated expression of nitric oxide. One study evaluated the acceleration of tissue repair in mice and related this efficacy to the increase in nitric oxide production, inflammatory pattern improvement, collagen production and subsequent re-epithelialization [84].
The toxicological and pharmacological research conduce the viability to use or not some phytotherapy products [85]. The evaluation of genotoxicity (comet assay) in this study demonstrated that andiroba oil treatment, without association, presents statistically significant difference when compared to the control group (cyclophosphamide) (p < 0.0001) and absence of DNA damage. The laser therapy presented no significant damage. These results corroborate with an experimental study, that evaluated andiroba oil use in Wistar rats, inducing acute and subacute toxicity, and reported no toxicity (no DNA damage), showing the therapeutic viability of andiroba oil’s use. [36, 86]. In this study it was also possible to observe that the association of treatments with laser and andiroba presented expressive genotoxicity when compared to control group, demonstrating a different result from treatments laser and andiroba, separately. However, no scientific evidence in agreement to such results has been found.
Thus, it is possible to conclude that both andiroba and laser are efficient treatments in the scarring potentiating effect in experimental models of chemo inducted OM, and andiroba group shows positive and similar results to the laser group for OM treatment, for being a low-cost and easy applied method. In contrast, the association of andiroba and laser, even though presenting better results than control group, presented inferior results, when compared to the groups using andiroba and laser separately. In conclusion, the use of andiroba oil and laser did not show genotoxic potential, when applied separately. However, genotoxic effects due to their association cannot be discarded.
Materials and Methods
Animals
We used 122 male golden Syrian hamsters (Mesocricetus auratus) for this study, with 90 days of age, weighting between 90 and 120 grams, from the Evandro Chagas Institute vivarium, Belém, Pará (PA), Brazil. The hamsters were kept in cages on the vivarium at the CESUPA, under controlled temperature (20°–24°C), relative air humidity (40–70%) and light (“12 h light /12 h dark” cycle). The animals had free access to food and water. The hamsters selection for the experiments was based on the easiness of observing and exposing their jugal mucosa, also, on their tolerance to chemotherapy drug doses for the induction of OM, without a high mortality rate.
Acquisition of andiroba and gaseous chromatography/mass spectometry (GC-MS) analysis
The andiroba’s oil used in this study belongs to the National Forest of Tapajós, located in west of Pará State (PA), comprehending the cities of Belterra, Aveiro, Rurópolis and Placas. It shares land borders with Tapajós river, with Santarém-Cuiabá road (BR-163) and with Cupari river, and coordinates 3° 31’ 1” S, 55° 4’ 23” W. Inside the forest, this oil is produced at the Comunidade Nossa Senhora do Rosário, Vila Santa Fé in km 200 north and km 67 of the BR-163 on Uruará-PA municipality. The community integrates the Sementes da Floresta Agroextrativist Association and their register is granted by Brazilian Company of Agroextrativist Researches of Brazil (BCAR). The BCAR company provided a technical report securing the quality of andiroba used.
The oil was analyzed by GC-MS using a gas chromatograph (Varian CP 3380 model) equipped with ions detector and capillary column CP-Sil 88 (60 m length, 0.25 mm internal diameter, 0.25 μm film thickness; Varian Inc., USA). This protocol promotes the conversion of fatty acids inside the oil into methyl ester fatty acids (MEFAs). The column temperature was adjusted to 80°C during 4’ and risen to 205°C in a 4°c/min rate. It was applied the Varian Star 3.4.1 software to quantify the fatty acids, with confection of chromatograms and mix of standard fatty acids (Nu-check-prep, Inc., USA). Fatty acid values were quantified in relative percentages of total acids.
Experimental groups
This study used a total of 122 animals, which were randomized and divided into 6 groups: Group “andiroba oil 100%”, Group “laser associated to andiroba oil 100%”, Group “laser”, Group “positive control”, Group “negative control” and Group “cyclophosphamide” (control group for genotoxicity analysis). The groups andiroba 100%, laser, laser associated to andiroba 100% and the positive control group presented a n = 28, each. The groups “negative control” and “cyclophosphamide control” had n = 5, each.
In the groups “andiroba oil”, “laser”, “laser associated to andiroba” and “positive control”, the OM was induced by the administration of chemotherapy drug 5-Fluorouracila (Fluoro-Uracil® 250 mg/10 ml, ICN Farmacêutica Ltda.) and a mechanical trauma on jugal mucosa of the animals, followed by their respective treatments from third to the fifteenth day. The “andiroba oil” group and “laser associated to andiroba oil” received the treatment three times a day (each oral mucosa) and approximately 0,5 ml per application (1,5 ml a day). The measurement and standardization required a plastic Pasteur pipette (0,5 ml) and the oil application was made using a plastic-wrapped cotton swab. Water and food were suspended for 1h for higher medication absorption. The laser group had applications of lasertherapy, once a day, without any kind of food restriction. In the group “laser associated to andiroba”, it was performed the application of laser once a day, and afterwards, it was applied andiroba oil three times a day, with food restriction. To perform the laser treatment, the animals were anaesthetized with ketamine in 80 mg/kg dose associated to xylazine in 20 mg/kg dose, by intraperitoneal via. For less risk of bias and in order to reduce the risk of interference from external factors, all groups were exposed to the same daily anesthetic factor.
In group “positive control” (PC), the animals were exposed to OM induction, however, without any kind of treatment. The group “negative control” (NC) did not received any OM induction protocol (absence of mechanical and chemotherapy induction), so their jugal mucosae served as normality standard and were used as negative control for the comet test. The group cyclophosphamide was the control for genotoxicity test (Comet Test) and the chemotherapy drug cyclophosphamide was used in these animals in a 1000 mg/kg dose by gavage 24 h before euthanasia, with the use of syringe and gavage tube.
Experimental protocol
The OM induction protocol performed on the animals was based on Soris et al. (1990) [87], which aims the most approximate reproducibility of the human body conditions. The administration of chemotherapy drug 5-FU (Fluoro-Uracil® 250 mg/10 ml, ICN Farmacêutica Ltda.) occurred by intraperitoneal injections on the animals, in the days 0, 5 and 10 of experiment, and 60 mg/kg doses of weight.
To a better representation of mucositis, it was performed a mechanical trauma on the oral mucosa of the animals, with previous anesthesia. The mucosa was exposed and attached in the surgical table and, with the aid of a sterile needle of caliber 18, it was performed two linear grooves, on right and left jugal mucosa of each animal, on days 1 and 2 of the experiment. This procedure was performed by only one previously trained operator. On days 4, 8, 12 and 15 seven random animals were selected from each group for photograph sessions and excisional biopsy on right and left jugal mucosae. The samples were submerged in 10% buffered formaldehyde. The euthanasia of the animals occurred by anesthetic overdosage. To perform the Comet Test, bone marrow, liver fragments and blood of the animals were removed on the 15th day, in all groups, including the negative control group.
Laser phototherapy protocol
The animals were previously anaesthetized and had their mucosae daily exposed for the performance of lasertherapy. The laser applied in this study was the continuous wave diode type (InGaA1P; MM Optics, São Carlos, São Paulo (SP), Brazil) with a wavelength of 660 nm (visible red), spot size of 0.04 cm², irradiance of 1 W/cm², output power of 40 mW, energy density of 6 J/cm², 6 seconds of exposition per spot, in a total of 0.24J, as applied by Weissheimer et al. (2017) [88].
Jugal mucosae clinical analysis
(The are several typing mistakes at the end of this section. The authors do not mention which clinical and histopathological parameters are going to be evaluated, nor how. Once again, there are problems concerning wording and text clarity).
The clinical evaluation was based on the analysis of severity of OM on the jugal mucosae of the animals, through the photographs taken on days 4, 8, 12 and 15. The images were evaluated by a previously trained examiner and blinded about the groups and treatment.
By performing the images analysis, there was not a complete score framing on the classified images using Lima et al. (2005) scale [61]. However, due to the necessity of more specific scores to the clinical reality of the experiment, it was opted for modifying the table, therefore denominated “Lima modified” – Score 0: absent or discreet hyperemia and erythema, absent hemorrhage, ulcer and abscess absence; Score 1: moderate hyperemia and erythema, absent hemorrhage, ulcer and abscess absence, presence of scarring tissue; Score 2: severe hyperemia and erythema, presence of hemorrhage and small ulcers (up to 1cm diameter), presence of larger area of scarring tissue and absence of abscesses; Score 3: severe hyperemia and erythema, presence of hemorrhage, extensive ulcers and absence of abscesses; and Score 4: severe hyperemia and erythema, presence of hemorrhage, extensive ulcers and abscesses.
Jugal mucosae histopathological analysis
The analysis was performed by the evaluation of histopathological glass slides which passed through laboratorial processing and were stained for hematoxylin-eosin. The glass slides were codified for the sample blinding, and the evaluation was performed by a third previously trained examiner.
During the histopathological analysis of inflammatory alterations, there were difficulties in the alteration framings in the criteria proposed by Lima et al. (2005) [61]. Due to it, we proposed a new table for the classification on inflammatory and scarring processes, which we denominated “Lima modified” – Score 0: epithelial and connective tissues with no vasodilation, absent or discreet inflammatory cellular infiltrate, absence of hemorrhage, edemas, ulcers or abscesses; Score 1: discreet vascular engorgement, reepithelization areas, discreet cellular infiltration, presence of mononuclear leukocytes, absence of hemorrhage, edemas, ulcers or abscesses; Score 2: moderate vascular engorgement, hydropic epithelial degeneration (vacuolation), discreet cellular infiltration, with mononuclear leucocytes predominance, presence of hemorrhagic areas, edemas, no ulcers and absence of abscesses; Score 3: moderate vascular engorgement, vacuolation, moderate or intense cellular infiltration, with mononuclear leucocytes predominance, presence of hemorrhagic areas, edemas, no ulcers and absence of abscesses; Score 4: moderate vascular engorgement, vacuolation, moderate or intense cellular infiltration, with polymorphonuclear leucocytes predominance, presence of hemorrhagic areas, edemas and eventual small ulcers and absence of abscesses; Score 5: intense vascular engorgement, intense vasodilation, intense cellular infiltration, with polymorphonuclear leucocytes predominance, presence of hemorrhagic areas, edemas, abscesses and extensive ulcers.
In vivo comet test
To evaluate the genotoxicity of treatments with laser and andiroba oil, as well as their association, during the 15 experimental days, a comet test was performed. The test was performed on the 15th day of experiment, in a sample of 30 animals, being 5 from each experimental group – (a) laser; (b) andiroba; (c) laser associated to andiroba; (d) negative control; (e) Cyclophosphamide, in which the animals were submitted to application of cyclophosphamide in a 1000 mg/kg dose by gavage 24 h before euthanasia, with the aid of syringe and gavage tube. For the test’s performance, the animals were “euthanized” and had their femur (bone marrow) removed, using a syringe (5 ml) and fetal bovine serum. Besides, blood was collected directly from the animal hearts and liver fragments.
The glass slides were previously covered in agarose solution in 1.5% normal melting point. The liver was macerated and mixed to the blood and bone marrow, until thorough mixing. After complete sample procedure, the glass slides remained in the lysis solution, in low luminosity environment, in which they were arranged in horizontal position in the electrophoresis tub, set in a 34V voltage in 300 mA electrical current for 20 minutes. The glass slides were, then, removed from the tub and submerged in icy distilled H2O (4°C) for electrophoresis solution removal. The washing procedure was repeated; however, the glass slides were immersed for 5 minutes for neutralization. The glass slides were fixed by being immersed in absolute ethanol for 3 minutes. After that, they were stained with 50 μL Ethidium bromide (20 μL) and covered in glass slide for analysis.
The glass slides were analyzed in duplicate of each sample, using a fluorescence microscope Olympus BX41 model (Olympus Co., Japan), and in each glass slide 50 cells were counted. The analysis was performed by the score standard, in which, according to size and intensity of the comet’s tail, the scores are classified in 5 categories, with variant damage of 0–4, according to the percentage of DNA in the comet’s tail, which indicates the injury degree suffered by the cell, as proposed by Singh et al. (1988) [89].
Statistical analysis
The sample size was determined by pilot study using the ANOVA test, in which it was adopted 0.05 of significance level and 80% power of evidence. Statistical tests of variance analysis (ANOVA) or Kruskal Wallis were performed, depending of sample normality distribution. In significant difference cases between groups, Tukey post-test (histopathological analysis and comet test) and Dunn post-test (Clinical Analysis) were applied. The significance level was established in 5% (p < 0.05) in all tests. Bioestat software, version 5.0 was used to perform such tests.
ACKNOWLEDGMENTS
To the Experimental Research Group and the Laboratory of Histopathology of the State University of Pará (CESUPA) for the technical assistance provided and to the Laboratory of Human Cytogenetics of the Federal University of Pará (LCH/UFPA). To the support of PROPESP/UFPA (PAPQ).
CONFLICTS OF INTEREST
Jessica Teixeira Gomes declares that she has no conflict of interests; Sergio Melo Alves Júnior declares that he has no conflicts of interests; Ana Márcia Viana Wanzeler declares that she has no conflicts of interest; Rosa Helena Figueiredo Chaves Soares declares that she has no conflict of interest; Carolina Pinheiro de Oliveira declares that she has no conflict of interest; Emanuelle de Matos Rodrigues declares that she has no conflicts of interest; Bruno Moreira Soares declares that he has no conflict of interest; Diego Di Felipe Ávila Alcantara declares that he has no conflicts of interest; Rommel Mario Rodríguez Burbano declares that he has no conflict of interest; Fabrício Mesquita Tuji declares that he has no conflicts of interest.
Ethical statement
The study was executed within the ethical standards care to the animals, as proposed by the law of animal use (Federal law nº 11,794, October 8, 2008); Decree nº 6,899; July 15, 2009 and under the standards followed by the National Council of Animal Experimentation Control (NCAEC), approved by the ANIMALS USING ETHICS COMISSION (AUEC) of University Center of Pará (CESUPA) under protocol nº 07/2016. The use of animals in the study, prioritized the minimum discomfort and suffering.
References
1. Duncan M, Grant G. Oral and intestinal mucositis - causes and possible treatments. Aliment Pharmacol Ther. 2003; 18:853–74. https://doi.org/10.1046/j.1365-2036.2003.01784.x. [PubMed].
2. Zhang N, Yin Y, Xu SJ, Chen WS. 5-Fluorouracil: mechanisms of resistance and reversal strategies. Molecules. 2008; 13:1551–69. https://doi.org/10.3390/molecules13081551. [PubMed].
3. Daugėlaitė G, Užkuraitytė K, Jagelavičienė E, Filipauskas A. Prevention and Treatment of Chemotherapy and Radiotherapy Induced Oral Mucositis. Medicina (Kaunas). 2019; 55:25. https://doi.org/10.3390/medicina55020025. [PubMed].
4. Riley P, Glenny AM, Worthington HV, Littlewood A, Clarkson JE, McCabe MG. Interventions for preventing oral mucositis in patients with cancer receiving treatment: oral cryotherapy. Cochrane Database Syst Rev. 2015; 2015:CD011552. https://doi.org/10.1002/14651858.CD011552. [PubMed].
5. Sant Ana G, Normando AGC, De Toledo I, Dos Reis PED, Guerra ENS. Topical Treatment of Oral Mucositis in Cancer Patients: A Systematic Review of Randomized Clinical Trials. Asian Pac J Cancer Prev. 2020; 21:1851–66. https://doi.org/10.31557/APJCP.2020.21.7.1851. [PubMed].
6. Anschau F, Webster J, Capra MEZ, de Azeredo da Silva ALF, Stein AT. Efficacy of low-level laser for treatment of cancer oral mucositis: a systematic review and meta-analysis. Lasers Med Sci. 2019; 34:1053–62. https://doi.org/10.1007/s10103-019-02722-7. [PubMed].
7. Redman MG, Harris K, Phillips BS. Low-level laser therapy for oral mucositis in children with cancer. Arch Dis Child. 2022; 107:128–33. https://doi.org/10.1136/archdischild-2020-321216. [PubMed].
8. Oberoi S, Zamperlini-Netto G, Beyene J, Treister NS, Sung L. Effect of prophylactic low level laser therapy on oral mucositis: a systematic review and meta-analysis. PLoS One. 2014; 9:e107418. https://doi.org/10.1371/journal.pone.0107418. [PubMed].
9. Legouté F, Bensadoun RJ, Seegers V, Pointreau Y, Caron D, Lang P, Prévost A, Martin L, Schick U, Morvant B, Capitain O, Calais G, Jadaud E. Low-level laser therapy in treatment of chemoradiotherapy-induced mucositis in head and neck cancer: results of a randomised, triple blind, multicentre phase III trial. Radiat Oncol. 2019; 14:83. https://doi.org/10.1186/s13014-019-1292-2. [PubMed].
10. Baharvand M, Jafari S, Mortazavi H. Herbs in Oral Mucositis. J Clin Diagn Res. 2017; 11:ZE05–11. https://doi.org/10.7860/JCDR/2017/21703.9467. [PubMed].
11. Wang G, Jia L. Herb medicine for relieving radiation induced oral mucositis: A systematic review and meta-analysis protocol. Medicine (Baltimore). 2019; 98:e18337. https://doi.org/10.1097/MD.0000000000018337. [PubMed].
12. Cheng YC, Wang YM, Lin YH, Cheng JY, Li SH, Huang YC, Tsai MY. Preventive effect and safety of Chinese herbal medicine for oral mucositis during radiotherapy in patients with head and neck cancer: Study protocol for a randomized trial. Contemp Clin Trials Commun. 2022; 27:100912. https://doi.org/10.1016/j.conctc.2022.100912. [PubMed].
13. Dos Reis PED, Ciol MA, de Melo NS, de Souza Figueiredo PT, Leite AF, de Melo Manzi N. Chamomile infusion cryotherapy to prevent oral mucositis induced by chemotherapy: a pilot study. Support Care Cancer. 2016; 24:4393–98. https://doi.org/10.1007/s00520-016-3279-y. [PubMed].
14. Elhadad MA, El-Negoumy E, Taalab MR, Ibrahim RS, Elsaka RO. The effect of topical chamomile in the prevention of chemotherapy-induced oral mucositis: A randomized clinical trial. Oral Dis. 2022; 28:164–72. https://doi.org/10.1111/odi.13749. [PubMed].
15. Tanideh N, Tavakoli P, Saghiri MA, Garcia-Godoy F, Amanat D, Tadbir AA, Samani SM, Tamadon A. Healing acceleration in hamsters of oral mucositis induced by 5-fluorouracil with topical Calendula officinalis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013; 115:332–38. https://doi.org/10.1016/j.oooo.2012.08.450. [PubMed].
16. Davarmanesh M, Miri R, Haghnegahdar S, Tadbir AA, Tanideh N, Saghiri MA, Garcia-Godoy F, Asatourian A. Protective effect of bilberry extract as a pretreatment on induced oral mucositis in hamsters. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013; 116:702–8. https://doi.org/10.1016/j.oooo.2013.07.004. [PubMed].
17. Mardani M, Afra SM, Tanideh N, Tadbir AA, Modarresi F, Koohi-Hosseinabadi O, Iraji A, Sepehrimanesh M. Hydroalcoholic extract of Carum carvi L. in oral mucositis: a clinical trial in male golden hamsters. Oral Dis. 2016; 22:39–45. https://doi.org/10.1111/odi.12375. [PubMed].
18. Piredda M, Facchinetti G, Biagioli V, Giannarelli D, Armento G, Tonini G, De Marinis MG. Propolis in the prevention of oral mucositis in breast cancer patients receiving adjuvant chemotherapy: A pilot randomised controlled trial. Eur J Cancer Care (Engl). 2017; 26. https://doi.org/10.1111/ecc.12757. [PubMed].
19. Tanideh N, Badie A, Habibagahi R, Koohi-Hosseinabadi O, Haghnegahdar S, Andisheh-Tadbir A. Effect of Topical 2% Eucalyptus Extract on 5-FU-Induced Oral Mucositis in Male Golden Hamsters. Braz Dent J. 2020; 31:310–18. https://doi.org/10.1590/0103-6440202003140. [PubMed].
20. Carvalho ACB, Perfeito JPS, Silva LVC, Ramalho LS, Marques RFO, Silveira D. Regulation of herbal medicines in Brazil: advances and perspectives. Braz J Pharm. 2011; 47. https://doi.org/10.1590/S1984-82502011000300004.
21. Li CL, Huang HL, Wang WC, Hua H. Efficacy and safety of topical herbal medicine treatment on recurrent aphthous stomatitis: a systemic review. Drug Des Devel Ther. 2016; 10:107–15. https://doi.org/10.2147/DDDT.S96589. [PubMed].
22. Silva CE, Santos OJ, Ribas-Filho JM, Tabushi FI, Kume MH, Jukonis LB, Cella IF. Effect of Carapa guianensis Aublet (Andiroba) and Orbignya phalerata (Babassu) in colonic healing in rats. Rev Col Bras Cir. 2015; 42:399–406. https://doi.org/10.1590/0100-69912015006009. [PubMed].
23. Pakravan F, Ghalayani P, Emami H, Isfahani MN, Noorshargh P. A novel formulation for radiotherapy-induced oral mucositis: Triamcinolone acetonide mucoadhesive film. J Res Med Sci. 2019; 24:63. https://doi.org/10.4103/jrms.JRMS_456_18. [PubMed].
24. Calixto JB. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz J Med Biol Res. 2000; 33:179–89. https://doi.org/10.1590/s0100-879x2000000200004. [PubMed].
25. Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014; 4:177. https://doi.org/10.3389/fphar.2013.00177. [PubMed].
26. Kikuchi T, Akita K, Koike H, In Y, Yamada T, Tanaka R. Carapanins A-C: new limonoids from andiroba (Carapa guianensis) fruit oil. Org Biomol Chem. 2020; 18:9268–74. https://doi.org/10.1039/d0ob01872d. [PubMed].
27. Wanzeler AMV, Júnior SMA, Gomes JT, Gouveia EHH, Henriques HYB, Chaves RH, Soares BM, Salgado HLC, Santos AS, Tuji FM. Therapeutic effect of andiroba oil (Carapa guianensis Aubl.) against oral mucositis: an experimental study in golden Syrian hamsters. Clin Oral Investig. 2018; 22:2069–79. https://doi.org/10.1007/s00784-017-2300-2. [PubMed].
28. das Graças Henriques M, Penido C. The therapeutic properties of Carapa guianensis. Curr Pharm Des. 2014; 20:850–56. https://doi.org/10.2174/13816128113199990048. [PubMed].
29. Tappin MR, Nakamura MJ, Siani AC, Lucchetti L. Development of an HPLC method for the determination of tetranortriterpenoids in Carapa guianensis seed oil by experimental design. J Pharm Biomed Anal. 2008; 48:1090–95. https://doi.org/10.1016/j.jpba.2008.08.027. [PubMed].
30. Soares ADS, Wanzeler AMV, Cavalcante GHS, da Silva Barros EM, Carneiro RCM, Tuji FM. Therapeutic effects of andiroba (Carapa guianensis Aubl) oil, compared to low power laser, on oral mucositis in children underwent chemotherapy: A clinical study. J Ethnopharmacol. 2021; 264:113365. https://doi.org/10.1016/j.jep.2020.113365. [PubMed].
31. Porfírio-Dias CL, Melo KM, Bastos CEM, Ferreira TAA, Azevedo LFC, Salgado HL, Santos AS, Rissino JD, Nagamachi CY, Pieczarka JC. Andiroba oil (Carapa guianensis Aubl) shows cytotoxicity but no mutagenicity in the ACPP02 gastric cancer cell line. J Appl Toxicol. 2020; 40:1060–66. https://doi.org/10.1002/jat.3966. [PubMed].
32. Klimas CA, de Oliveira Wadt LH, de Castilho CV, Lira-Guedes AC, da Costa P, da Fonseca FL. Variation in Seed Harvest Potential of Carapa guianensis Aublet in the Brazilian Amazon: A Multi-Year, Multi-Region Study of Determinants of Mast Seeding and Seed Quantity. Forests. 2021; 12:683. https://doi.org/10.3390/f12060683.
33. Pennington TD, Styles ΒX, Taylor DAH. Meliaceae. Flora Neotropica, monograph 28. New York, NY: The New York Botanical Garden. 1981. ISBN 10: 0893272353.
34. Sakamoto A, Tanaka Y, Inoue T, Kikuchi T, Kajimoto T, Muraoka O, Yamada T, Tanaka R. Andirolides Q-V from the flower of andiroba (Carapa guianensis, Meliaceae). Fitoterapia. 2013; 90:20–29. https://doi.org/10.1016/j.fitote.2013.07.001. [PubMed].
35. De Sousa RL, Silva SG, Costa JM, Da Costa WA, Maia AAB, De Oliveira MS, Andrade EHA. Chemical profile of manually extracted andiroba oil (Carapa guianensis Aubl., Meliaceae) from Mamangal community, located in Igarapé-Miri, Pará, Brazil. Scientia Plena. 2021; 17:127201. https://doi.org/10.14808/sci.plena.2021.127201.
36. Milhomem-Paixão SS, Fascineli ML, Roll MM, Longo JP, Azevedo RB, Pieczarka JC, Salgado HL, Santos AS, Grisolia CK. The lipidome, genotoxicity, hematotoxicity and antioxidant properties of andiroba oil from the Brazilian Amazon. Genet Mol Biol. 2016; 39:248–56. https://doi.org/10.1590/1678-4685-GMB-2015-0098. [PubMed].
37. Pereira JF. Estudo químico e biológico de Carapaguianensis Aubl. Dissertação de Mestrado, Universidade Federal do Rio de Janeiro. Rio de Janeiro. 1998.
38. Hatakana E, Curi R. Ácidos graxos e cicatrização: uma revisão. Rev Bras Farm. 2007; 88:53–58.
39. Cornily JC, Gilard M, Bezon E, Jan V, Pennec PY, Etienne Y, Vinsonneau U, Le Gal G, Mansourati J, Blanc JJ, Boschat J. Cardiac multislice spiral computed tomography as an alternative to coronary angiography in the preoperative assessment of coronary artery disease before aortic valve surgery: a management outcome study. Arch Cardiovasc Dis. 2010; 103:170–75. https://doi.org/10.1016/j.acvd.2010.01.004. [PubMed].
40. de Oliveira AP, de Souza Franco E, Barreto RR, Cordeiro DP, de Melo RG, de Aquino CMF, E Silva AAR, de Medeiros PL, da Silva TG, da Silva Góes AJ, de Sousa Maia MB. Effect of semisolid formulation of persea americana mill (avocado) oil on wound healing in rats. Evid Based Complement Alternat Med. 2013; 2013:472382. https://doi.org/10.1155/2013/472382. [PubMed].
41. Lamaud E, Huc A, Wepierre J. Effects of avocado and soya bean lipidic non-saponifiables on the components of skin connective tissue after topical application in the hairless rat: biophysical and biomechanical determination. Int J Cosmet Sci. 1982; 4:143–52. https://doi.org/10.1111/j.1467-2494.1982.tb00310.x. [PubMed].
42. Lewinska A, Zebrowski J, Duda M, Gorka A, Wnuk M. Fatty Acid Profile and Biological Activities of Linseed and Rapeseed Oils. Molecules. 2015; 20:22872–80. https://doi.org/10.3390/molecules201219887. [PubMed].
43. Lin TK, Zhong L, Santiago JL. Anti-Inflammatory and Skin Barrier Repair Effects of Topical Application of Some Plant Oils. Int J Mol Sci. 2017; 19:70. https://doi.org/10.3390/ijms19010070. [PubMed].
44. Mandawgade SD, Patravale VB. Formulation and evaluation of exotic fat based cosmeceuticals for skin repair. Indian J Pharm Sci. 2008; 70:539–42. https://doi.org/10.4103/0250-474X.44615. [PubMed].
45. Nasopoulou C, Karantonis HC, Detopoulou M, Demopoulos CA, Zabetakis I. Exploiting the anti-inflammatory properties of olive (Olea europaea) in the sustainable production of functional food and neutraceuticals. Phytochemistry Reviews. 2014; 13:445–58. https://doi.org/10.1007/s11101-014-9350-8.
46. Shinagawa FB, de Santana FC, Torres LRO, Mancini-Filho J. Grape seed oil: A potential functional food? Food Sci Technol (Campinas). 2015; 35:399–406. https://doi.org/10.1590/1678-457X.6826.
47. Vaughn AR, Clark AK, Sivamani RK, Shi VY. Natural Oils for Skin-Barrier Repair: Ancient Compounds Now Backed by Modern Science. Am J Clin Dermatol. 2018; 19:103–17. https://doi.org/10.1007/s40257-017-0301-1. [PubMed].
48. Poljšak N, Kreft S, Kočevar Glavač N. Vegetable butters and oils in skin wound healing: Scientific evidence for new opportunities in dermatology. Phytother Res. 2020; 34:254–69. https://doi.org/10.1002/ptr.6524. [PubMed].
49. Miot HA, Batistella RF, de Almeida Batista K, Volpato DEC, Augusto LST, Madeira NG, Haddad V Jr, Miot LDB. Comparative study of the topical effectiveness of the Andiroba oil (Carapa guianensis) and DEET 50% as repellent for Aedes sp. Rev Inst Med Trop Sao Paulo. 2004; 46:253–56. https://doi.org/10.1590/s0036-46652004000500004. [PubMed].
50. Cela EVS, de Britto da Rocha M, Gomes TM, Chia CY, Alves CF. Clinical evaluation of the effectiveness of andiroba oil in burns caused by hair removal with intense pulsed light: a prospective, comparative and double-blind study. Surg Cosmet Dermatol. 2012; 4:248–51.
51. Chia CY, Medeiros AD, Corraes AMS, Manso JEF, da Silva CSC, Takiya CM, Vanz RL. Healing effect of andiroba-based emulsion in cutaneous wound healing via modulation of inflammation and transforming growth factor beta 31. Acta Cir Bras. 2018; 33:1000–15. https://doi.org/10.1590/s0102-865020180110000007. [PubMed].
52. Nayak BS, Kanhai J, Milne DM, Pinto Pereira L, Swanston WH. Experimental evaluation of ethanolic extract of Carapa guianensis L. Leaf for its wound healing activity using three wound models. Evid Based Complement Alternat Med. 2011; 2011:419612. https://doi.org/10.1093/ecam/nep160. [PubMed].
53. Teixeira RK, Houat AP, Costa FL, Filho JC, Yasojima EY, Brito MV. Efeito do óleo de andiroba na sobrevida de camundongos submetidos à sepse abdominal. Rev Bras Clin Med. 2012; 10:407–9.
54. Silva OS, Romão PR, Blazius RD, Prohiro JS. The use of andiroba Carapa guianensis as larvicide against Aedes albopictus. J Am Mosq Control Assoc. 2004; 20:456–57. [PubMed].
55. de Mendonça FA, da Silva KF, dos Santos KK, Ribeiro Júnior KA, Sant’Ana AE. Activities of some Brazilian plants against larvae of the mosquito Aedes aegypti. Fitoterapia. 2005; 76:629–36. https://doi.org/10.1016/j.fitote.2005.06.013. [PubMed].
56. Penido C, Costa KA, Pennaforte RJ, Costa MF, Pereira JF, Siani AC, Henriques MG. Anti-allergic effects of natural tetranortriterpenoids isolated from Carapa guianensis Aublet on allergen-induced vascular permeability and hyperalgesia. Inflamm Res. 2005; 54:295–303. https://doi.org/10.1007/s00011-005-1357-6. [PubMed].
57. Penido C, Conte FP, Chagas MS, Rodrigues CA, Pereira JF, Henriques MG. Antiinflammatory effects of natural tetranortriterpenoids isolated from Carapa guianensis Aublet on zymosan-induced arthritis in mice. Inflamm Res. 2006; 55:457–64. https://doi.org/10.1007/s00011-006-5161-8. [PubMed].
58. da Poça JJG, Silva RC, de Souza EFM, de Almeida CP, do Reis Ferreira TC, Santos AS. Effects of andiroba (carapa guianenses) on joint articulate edema and ambulation of mice with experimental arthritis by zymozan induce. Revista Interdisciplinar em Saúde Cajazeiras. 2020; 7:1906–17. https://doi.org/10.35621/23587490.v7.n1.p1906-1917.
59. Zadik Y, Arany PR, Fregnani ER, Bossi P, Antunes HS, Bensadoun RJ, Gueiros LA, Majorana A, Nair RG, Ranna V, Tissing WJE, Vaddi A, Lubart R, et al. Systematic review of photobiomodulation for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 2019; 27:3969–83. https://doi.org/10.1007/s00520-019-04890-2. [PubMed].
60. Elad S, Cheng KKF, Lalla RV, Yarom N, Hong C, Logan RM, Bowen J, Gibson R, Saunders DP, Zadik Y, Ariyawardana A, Correa ME, Ranna V, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. 2020; 126:4423–31. https://doi.org/10.1002/cncr.33100. [PubMed].
61. Lima V, Brito GA, Cunha FQ, Rebouças CG, Falcão BA, Augusto RF, Souza ML, Leitão BT, Ribeiro RA. Effects of the tumour necrosis factor-alpha inhibitors pentoxifylline and thalidomide in short-term experimental oral mucositis in hamsters. Eur J Oral Sci. 2005; 113:210–17. https://doi.org/10.1111/j.1600-0722.2005.00216.x. [PubMed].
62. Pereira AN, de Paula Eduardo C, Matson E, Marques MM. Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg Med. 2002; 31:263–67. https://doi.org/10.1002/lsm.10107. [PubMed].
63. Khalighi HR, Mortazavi H, Mojahedi SM, Azari-Marhabi S, Moradi Abbasabadi F. Low Level Laser Therapy Versus Pharmacotherapy in Improving Myofascial Pain Disorder Syndrome. J Lasers Med Sci. 2016; 7:45–50. https://doi.org/10.15171/jlms.2016.10. [PubMed].
64. Wagner VP, Meurer L, Martins MA, Danilevicz CK, Magnusson AS, Marques MM, Filho MS, Squarize CH, Martins MD. Influence of different energy densities of laser phototherapy on oral wound healing. J Biomed Opt. 2013; 18:128002. https://doi.org/10.1117/1.JBO.18.12.128002. [PubMed].
65. Pellicioli AC, Martins MD, Dillenburg CS, Marques MM, Squarize CH, Castilho RM. Laser phototherapy accelerates oral keratinocyte migration through the modulation of the mammalian target of rapamycin signaling pathway. J Biomed Opt. 2014; 19:028002. https://doi.org/10.1117/1.JBO.19.2.028002. [PubMed].
66. Wagner VP, Curra M, Webber LP, Nör C, Matte U, Meurer L, Martins MD. Photobiomodulation regulates cytokine release and new blood vessel formation during oral wound healing in rats. Lasers Med Sci. 2016; 31:665–71. https://doi.org/10.1007/s10103-016-1904-0. [PubMed].
67. de Freitas Cuba L, Salum FG, Cherubini K, de Figueiredo MA. Antioxidant agents: a future alternative approach in the prevention and treatment of radiation-induced oral mucositis? Altern Ther Health Med. 2015; 21:36–41. [PubMed].
68. Rodríguez-Caballero A, Torres-Lagares D, Robles-García M, Pachón-Ibáñez J, González-Padilla D, Gutiérrez-Pérez JL. Cancer treatment-induced oral mucositis: a critical review. Int J Oral Maxillofac Surg. 2012; 41:225–38. https://doi.org/10.1016/j.ijom.2011.10.011. [PubMed].
69. Campos MI, Campos CN, Aarestrup FM, Aarestrup BJ. Oral mucositis in cancer treatment: Natural history, prevention and treatment. Mol Clin Oncol. 2014; 2:337–40. https://doi.org/10.3892/mco.2014.253. [PubMed].
70. Ferraris FK, Rodrigues R, da Silva VP, Figueiredo R, Penido C, Henriques Md. Modulation of T lymphocyte and eosinophil functions in vitro by natural tetranortriterpenoids isolated from Carapa guianensis Aublet. Int Immunopharmacol. 2011; 11:1–11. https://doi.org/10.1016/j.intimp.2010.09.010. [PubMed].
71. Barros FN, Farias MP, Tavares JP, Alves LC, Faustino MA. In vitro efficacy of oil from the seed of Carapa guianensis (andiroba) in the control of Felicola subrostratus . Rev Bras Farmacogn. 2012; 22:1130–33. https://doi.org/10.1590/S0102-695X2012005000047.
72. Moore ZE, Webster J. Dressings and topical agents for preventing pressure ulcers. Cochrane Database Syst Rev. 2018; 12:CD009362. https://doi.org/10.1002/14651858.CD009362.pub3. [PubMed].
73. Rodrigues HG, Vinolo MA, Magdalon J, Vitzel K, Nachbar RT, Pessoa AF, dos Santos MF, Hatanaka E, Calder PC, Curi R. Oral administration of oleic or linoleic acid accelerates the inflammatory phase of wound healing. J Invest Dermatol. 2012; 132:208–15. https://doi.org/10.1038/jid.2011.265. [PubMed].
74. Vitale S, Colanero S, Placidi M, Di Emidio G, Tatone C, Amicarelli F, D’Alessandro AM. Phytochemistry and Biological Activity of Medicinal Plants in Wound Healing: An Overview of Current Research. Molecules. 2022; 27:3566. https://doi.org/10.3390/molecules27113566. [PubMed].
75. Schilrreff P, Alexiev U. Chronic Inflammation in Non-Healing Skin Wounds and Promising Natural Bioactive Compounds Treatment. Int J Mol Sci. 2022; 23:4928. https://doi.org/10.3390/ijms23094928. [PubMed].
76. Sivamani RK. Eicosanoids and Keratinocytes in Wound Healing. Adv Wound Care (New Rochelle). 2014; 3:476–81. https://doi.org/10.1089/wound.2014.0523. [PubMed].
77. Silva JR, Burger B, Kühl CMC, Candreva T, Dos Anjos MBP, Rodrigues HG. Wound Healing and Omega-6 Fatty Acids: From Inflammation to Repair. Mediators Inflamm. 2018; 2018:2503950. https://doi.org/10.1155/2018/2503950. [PubMed].
78. Curra M, Pellicioli AC, Filho NA, Ochs G, Matte Ú, Filho MS, Martins MA, Martins MD. Photobiomodulation reduces oral mucositis by modulating NF-kB. J Biomed Opt. 2015; 20:125008. https://doi.org/10.1117/1.JBO.20.12.125008. [PubMed].
79. Cinelli MA, Do HT, Miley GP, Silverman RB. Inducible nitric oxide synthase: Regulation, structure, and inhibition. Med Res Rev. 2020; 40:158–89. https://doi.org/10.1002/med.21599. [PubMed].
80. Roeslan MO, Ayudhya TDN, Yingyongnarongkul BE, Koontongkaew S. Anti-biofilm, nitric oxide inhibition and wound healing potential of purpurin-18 phytyl ester isolated from Clinacanthus nutans leaves. Biomed Pharmacother. 2019; 113:108724. https://doi.org/10.1016/j.biopha.2019.108724. [PubMed].
81. Ninomiya K, Miyazawa S, Ozeki K, Matsuo N, Muraoka O, Kikuchi T, Yamada T, Tanaka R, Morikawa T. Hepatoprotective Limonoids from Andiroba (Carapa guianensis). Int J Mol Sci. 2016; 17:591. https://doi.org/10.3390/ijms17040591. [PubMed].
82. Inoue T, Ohmori S, Kikuchi T, Yamada T, Tanaka R. Carapanosins D-F from the Seeds of Andiroba (Carapa guianensis, Meliaceae) and Their Effects on LPS-Activated NO Production. Molecules. 2018; 23:1778. https://doi.org/10.3390/molecules23071778. [PubMed].
83. Karu TI, Pyatibrat LV, Afanasyeva NI. Cellular effects of low power laser therapy can be mediated by nitric oxide. Lasers Surg Med. 2005; 36:307–14. https://doi.org/10.1002/lsm.20148. [PubMed].
84. Most D, Efron DT, Shi HP, Tantry US, Barbul A. Characterization of incisional wound healing in inducible nitric oxide synthase knockout mice. Surgery. 2002; 132:866–76. https://doi.org/10.1067/msy.2002.127422. [PubMed].
85. Heinrich M, Appendino G, Efferth T, Fürst R, Izzo AA, Kayser O, Pezzuto JM, Viljoen A. Best practice in research - Overcoming common challenges in phytopharmacological research. J Ethnopharmacol. 2020; 246:112230. https://doi.org/10.1016/j.jep.2019.112230. [PubMed].
86. Costa-Silva JH, Lima CR, Silva EJ, Araújo AV, Fraga MC, Ribeiro E Ribeiro A, Arruda AC, Lafayette SS, Wanderley AG. Acute and subacute toxicity of the Carapa guianensis Aublet (Meliaceae) seed oil. J Ethnopharmacol. 2008; 116:495–500. https://doi.org/10.1016/j.jep.2007.12.016. [PubMed].
87. Sonis ST, Tracey C, Shklar G, Jenson J, Florine D. An animal model for mucositis induced by cancer chemotherapy. Oral Surg Oral Med Oral Pathol. 1990; 69:437–43. https://doi.org/10.1016/0030-4220(90)90376-4. [PubMed].
88. Weissheimer C, Curra M, Gregianin LJ, Daudt LE, Wagner VP, Martins MAT, Martins MD. New photobiomodulation protocol prevents oral mucositis in hematopoietic stem cell transplantation recipients-a retrospective study. Lasers Med Sci. 2017; 32:2013–21. https://doi.org/10.1007/s10103-017-2314-7. [PubMed].
89. Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988; 175:184–91. https://doi.org/10.1016/0014-4827(88)90265-0. [PubMed].

