Introduction
The ability to detect actionable alterations in patients with advanced lung cancer has revolutionized the field of thoracic oncology and is in part responsible for a significant decline in mortality [1]. However, uptake of routine testing as per NCCN guidelines has been slow for a variety of reasons, including small biopsies, tissue exhaustion from sequential testing and slow diffusion of knowledge [2]. Timely identification of patients who might benefit from tyrosine kinase inhibitors (TKIs) is crucial, as it is known that TKIs selected on the basis of a driver alteration gives patients a much better chance at prolonged response rather than getting treatment following systemic chemotherapy. In addition, it is now being recognized that immunotherapy drugs given sequentially before TKIs but not after are associated with a much higher rate of pneumonitis and other adverse events [3, 4].
ALK translocations are very sensitive to a number of well tolerated oral medications [5, 6]. Responding patients often have prolonged non-trivial survival benefit—a recent study found a median OS of 48 months in a cohort of patients treated mostly with an older generation of ALK TKIs [7]. Clinical predictors, while useful, have been recognized for a long time as being insufficient for deciding which patients with advanced pulmonary adenocarcinoma should be tested [8]. We performed a retrospective study of a database to acquire real-world clinical data on the frequency of the translocation in a large pool of patients drawn primarily from community hospitals and practices.
Results
Patient characteristics
In total, 19,895 patients with aNSCLC diagnosed from 2015 to 2019 were included in this study. The mean age of patients was 68.5 ± 10.0 years. Men comprised 50.4% (10,029) of the patient cohort and 68.4% (13,599 of 19,895) of patients were Caucasian. A large proportion of patients had a non-squamous histology type (16,025 of 19,895 or 80.5%) and smoking history (17,003 of 19,895 or 85.5%), and the majority of patients were from community practices (18,350 of 19,895 or 92.2%) (Table 1).
Table 1: Prevalence of ALK rearrangement in aNSCLC according to patient characteristics
| Characteristics | N | % Total patients | N ALK+ patients | % ALK+ by characteristic |
|---|---|---|---|---|
| Total | 19,985 | 100.0 | 519 | 2.6 |
| Age at aNSCLC diagnosis1 | ||||
| 18–39 | 125 | 0.6 | 27 | 21.6 |
| 40–54 | 1,627 | 8.2 | 85 | 5.2 |
| 55+ | 18,143 | 91.2 | 407 | 2.2 |
| Gender | ||||
| Female | 9,866 | 49.6 | 275 | 2.8 |
| Male | 10,029 | 50.4 | 244 | 2.4 |
| Race1 | ||||
| White | 13,599 | 68.4 | 324 | 2.4 |
| Black or African American | 1,673 | 8.4 | 37 | 2.2 |
| Asian | 623 | 3.1 | 39 | 6.3 |
| Others | 2,043 | 10.3 | 43 | 2.1 |
| Unknown | 1,957 | 9.8 | 50 | 2.6 |
| Geographic Location1 | ||||
| Northeast | 3,892 | 19.6 | 98 | 2.5 |
| South | 7,988 | 40.2 | 154 | 1.9 |
| Midwest | 2,973 | 14.9 | 83 | 2.8 |
| West | 3,143 | 15.8 | 108 | 3.4 |
| Unknown | 1,899 | 9.5 | 76 | 4.0 |
| Practice Type1 | ||||
| Community | 18,350 | 92.2 | 452 | 2.5 |
| Academic | 1,545 | 7.8 | 67 | 4.3 |
| Smoking Status1 | ||||
| Nonsmoker | 2,862 | 14.5 | 257 | 8.9 |
| Smoker | 17,003 | 85.5 | 262 | 1.5 |
| Histology1 | ||||
| Non-squamous | 16,025 | 80.5 | 477 | 3.0 |
| NSCLC NOS | 903 | 4.5 | 17 | 1.9 |
| Squamous | 2,967 | 14.9 | 25 | 0.8 |
| PD-L1 Status | ||||
| PD-L1 positive | 1,839 | 9.2 | 56 | 3.0 |
| PD-L1 negative/unknown | 18,056 | 90.8 | 463 | 2.6 |
| Other Biomarker Status1,2 | ||||
| Any other biomarker positive | 6,240 | 31.4 | 33 | 0.5 |
| Other biomarker negative/unknown | 13,655 | 6806 | 486 | 3.6 |
Prevalence of ALK rearrangement
Overall, the prevalence of ALK rearrangement was 2.6% in aNSCLC patients (519 of 19,895 patients). Prevalence varied by patients’ demographic characteristics. The rate of ALK rearrangement was the highest (>20%) among patients younger than 40 years old. It decreased to over 10% in patients in their early 40s, and further decreased to a plateau of approximately 2% in patients older than 55 years old (Figure 1). Asian patients had a higher ALK rearrangement rate (39 of 623 or 6.3%) than other patients. There were no significant differences in the number of ALK rearrangements found in females (275 of 9866 or 2.8%) compared to male patients (244 of 10,029 or 2.4%). The prevalence of ALK translocation was higher in patients from academic centers (4.3%) than patients from community centers (2.5%).
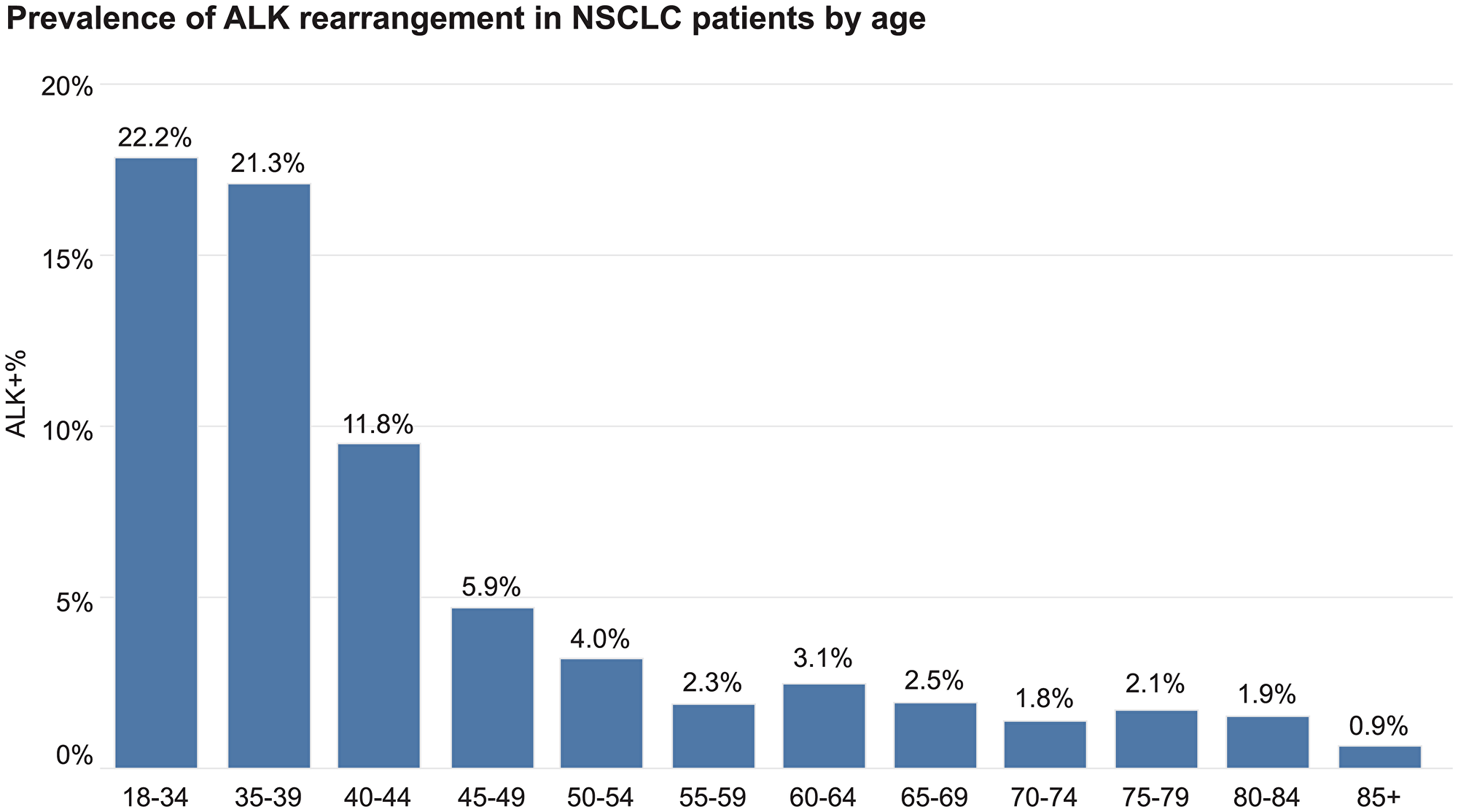
Figure 1: Prevalence of ALK rearrangement in aNSCLC patients by age.
ALK rearrangement rates also varied by some patients’ clinical characteristics. Patients with no smoking history had a higher ALK rearrangement rate compared to those with a history of smoking (8.9% vs. 1.5%). Overall, patients with squamous histology type had a lower ALK rearrangement rate (0.8%) than patients with non-squamous (3.0%) and nonspecific (1.9%) histology types. However, there was a subgroup of patients with squamous histology and no history of smoking that had a relatively high ALK rearrangement rate (3.3%) (Figure 2). Results from this study showed that the ALK rearrangement rate in NCCN guideline ALK testing ineligible patients (those with squamous histology and a history of smoking) was 0.7% and ranged from 1.4% to 9.3% in eligible patients (Figure 2).
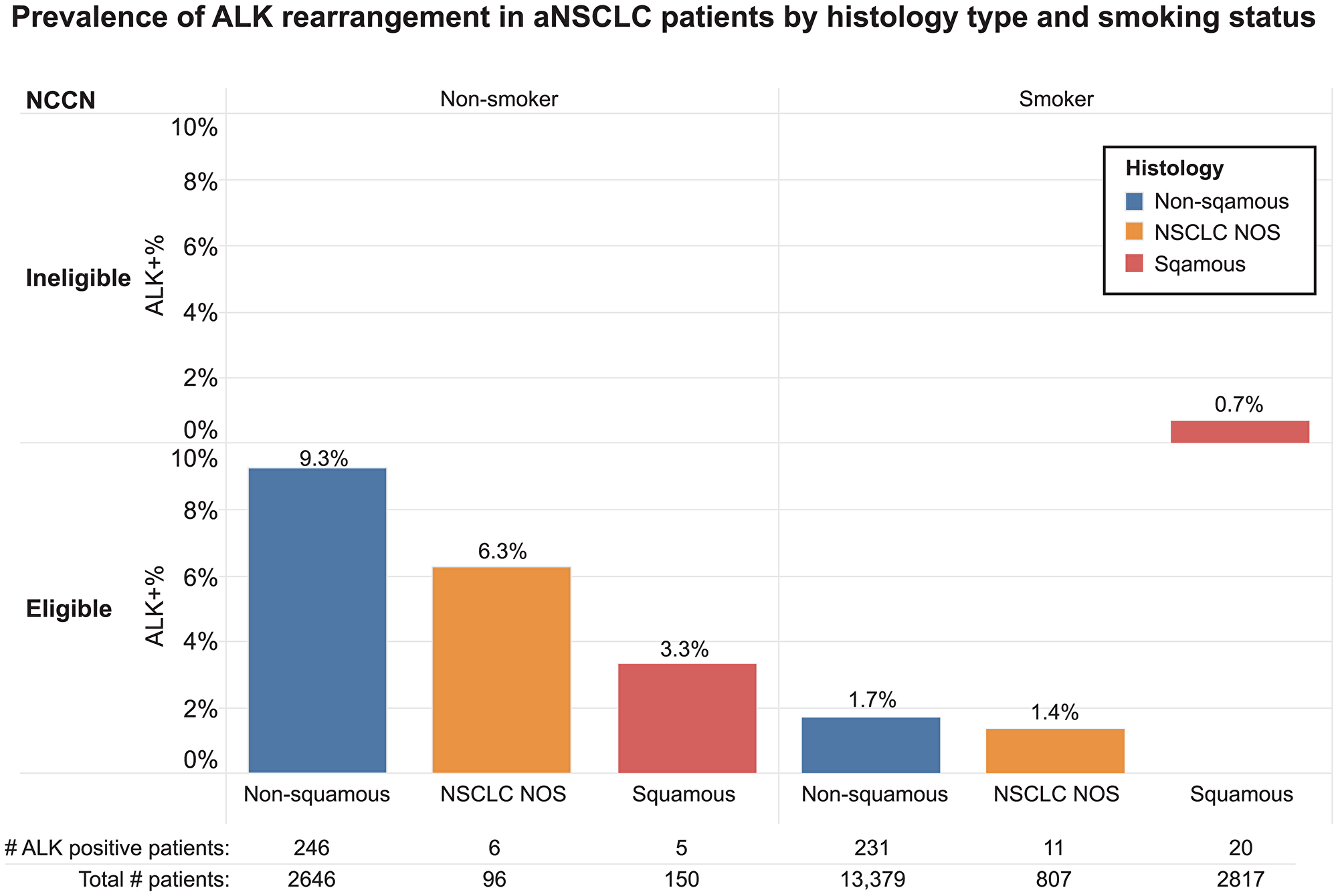
Figure 2: Prevalence of ALK rearrangement in aNSCLC patients by histology type and smoking status.
Patients who were positive for any other driver mutations or rearrangements (EGFR, KRAS, BRAF or ROS1) had a significantly lower ALK positivity rate (0.5%), while patients reported to be positive for PD-L1 had an ALK positivity rate of 3.0%.
DISCUSSION
Since the first descriptions of ALK rearrangements in lung cancer, prevalence estimates for this rearrangement have ranged relatively broadly from <2–13% [9–12]. The overall ALK rearrangement rate (2.6%) in this large retrospective data set was within the range observed previously in other non-selected populations [13–15]. Likewise, the younger age of the patients whose cancers harbored the ALK translocation has been reported by other groups as well [16, 17]. It is not surprising that, of those young patients with advanced lung cancer, there would be an enriched population of ALK positive patients and that was confirmed in the evaluation of this dataset. One in five patients in the 18–34 year old age group were found to be ALK fusion positive (Figure 1 for reference). This was cut in half by ages 40–44 (11.8%). But even in patients over 55, there was a 2% rate of positivity where treatment would be substantially different if an ALK rearrangement was detected. The fact that the prevalence of ALK rearrangement was higher in patients from academic centers than patients from community centers (4.3% vs. 2.5%) could be explained by patient self-referral patterns. Also, since the majority of the data is from patients in community settings, this finding could be attributed to the limited number of patients in this database that were seen in academic centers.
While our understanding of molecular pathophysiology has developed, clinical features such as histology, age and smoking history have been utilized when determining testing for driver alterations. As such, we examined histology and smoking status within this patient cohort. Regardless of documented histology, a higher ALK rearrangement rate (8.9%) was observed among patients who had no smoking history compared to patients with a smoking history (1.5% ALK positivity) which represent the largest number of patients in this cohort (17,003). Analyzing patients in this study by their histology, we observed that patients with non-squamous histology had an ALK rearrangement rate of 3%, patients in the NSCLC NOS category (most likely poorly differentiated adenocarcinomas and squamous cell carcinomas) 1.9%, and patients with squamous histology 0.8%. It is not discernable in our data if the patients with squamous histology had small biopsies, in which case, biomarker testing guidelines in place during the study period [18] recommended testing of these specimens as small biopsies with poorly differentiated areas of squamous histology may actually represent underdiagnosed poorly differentiated adenocarcinoma within an adenosquamous carcinoma. The percentage of ALK rearrangements in squamous histology as observed in our cohort is similar to a recent report in a smaller cohort [19].
We also examined the prevalence of ALK rearrangements as it related to patients deemed eligible for testing as per the NCCN guidelines in place during the study period [18]. The ALK positivity rate was greatest (9.3%) for non-smoking patients with non-squamous histology. Patients with squamous histology who were eligible as per guidelines (generally either nonsmokers or small biopsies) still had a 3.3% positivity rate, underscoring the importance, due to lung cancer heterogeneity, of not overlooking testing in patients with a diagnosis of squamous lung cancer on biopsy. Also, while a low rate of ALK positivity (1.7%) in patients with adenocarcinoma and a smoking history was observed, when coupled with the number of patients with both of these characteristics, this translates to a potentially large number of patients who would be a candidate for TKIs if appropriately identified through testing. In fact, these patients’ rate of ALK positivity (1.7%) is similar to the number of patients expected to have RET or ROS1 fusions [14, 16, 20–23]. The lowest number of ALK rearrangments (0.7%) were observed in patients with a history of smoking and squamous cell carcinoma. At the time of testing, these patients would have been ineligible for testing per NCCN guidelines in place. It is unknown as to why these patient specimens were sent for additional testing but it is likely that these were patients in which multigene panel testing was performed regardless of histology. Given that testing rates in patients with a history of smoking and squamous cell histology have increased in recent years in the US [24, 25], it is not surprising to find a low percentage of patients with ALK rearrangements. Because of lung cancer heterogeneity, in patients with squamous cell carcinoma diagnosed in small biopsies, NGS for actionable targets should be undertaken. Elderly patients and a substantial smoking history could potentially avoid testing but clinicians should use best judgment and if there are any questions about whether to test or not they should err on the side of testing. Younger patients should also be tested regardless of histology and smoking history. The presence of actionable co-mutations or rearrangements on diagnosis is relatively rare and was confirmed in this dataset [26–28].
These findings strongly reinforce the necessity of testing aNSCLC patients for ALK positivity regardless of the previously-depended upon clinical criteria of smoking history and tumor histology. Continuing to use these clinical criteria will deny some aNSCLC patients the opportunity to benefit from therapeutic improvements provided by ALK targeted therapy. NCCN guidelines were updated earlier this year with a recommendation to consider mutational testing in squamous histology without regard to smoking history [29]. This is a good step towards identifying all patients with a possible actionable mutation.
As tempting as it might be to start patients on immunotherapy in the presence of PD-L1 positivity, the number of patients we found who were both ALK positive and PD-L1 positive (3.0%) underscores the need to test for actionable alterations prior to initiation of immune checkpoint inhibitors. ALK positive patients benefit more from TKIs and one cannot simply assume that positive PD-L1 expression rules out the presence of a driver mutation. This underscores the vital need to continue to work nationally to shorten the molecular testing turnaround time for aNSCLC patients, both in academic and community practices.
Our study has the usual limitations of retrospective analyses of EHR databases, such as the potential for coding errors in oncology practices and missing data if procedures or treatments occurred outside the specific oncology practice. It is not possible to discern reasons for failure to test; certainly, some patients might not have been tested in the past due to having a very large burden of disease or impending hospice admission. Likewise, it was not possible to determine why patients with squamous histology and a history or smoking were tested. However, given the self-selection that often takes place with patients seeking care in major academic centers in big cities, having a broader population from primarily community centers from throughout the country provides a better snapshot of the actual incidence of ALK rearrangements in patients with aNSCLC. We are also unable to determine the sequence of testing for the various biomarkers.
In summary, this retrospective review of nearly 20,000 patients with aNSCLC and tested for ALK in the United States confirms that ALK rearrangements are found more commonly in younger nonsmokers and patients of Asian descent. We did not observe a significant difference in prevalence between males and females. However, a substantial minority of cases are found across all age groups, in patients with a history of smoking as well as patients with squamous histology and no history of smoking. Our findings strongly reinforce the need for earlier broad gene panel testing in all patients with advanced lung non-squamous aNSCLC regardless of clinical features. This approach has the potential to identify the maximum number of patients who could benefit from correct identification of actionable driver alterations, ultimately providing these patients with the maximum benefit of molecular therapeutic regimes.
Materials and Methods
Study design and data source
This cross-sectional observational study utilized Flatiron Health’s nationwide de-identified database derived from electronic health record (EHR) data originating from approximately 280 US cancer clinics (~800 sites of care). The Flatiron Health database is a longitudinal database, comprising de-identified patient-level structured and unstructured data, curated via technology-enabled abstraction [30, 31]. Institutional Review Board approval of the study protocol was obtained prior to study conduct, and included a waiver of informed consent.
Study population
Patients with advanced non-small cell lung cancer (stage IIIB, IIIC, or IV by American Joint Committee on Cancer 7th edition) [32] diagnosed from January 2015 to May 2019 were included. Eligible patients were also required to be ≥ 18 years old at aNSCLC diagnosis, have documented ALK testing (irrespective of test methodology) and known smoking status. ALK testing information was obtained from biomarker reports, pathology reports/addendums or physician notes.
Definitions
ALK translocation testing methodology was retrieved from EHR documentation. Various methods for testing were utilized including fluorescent in situ hybridization (FISH), next generation sequencing (NGS), immunohistochemistry (IHC) or other methods such as polymerase chain reaction (PCR) and sequencing other than NGS.
For patients with only one documented ALK result, results were categorized as positive (rearrangement/fusion present), negative (rearrangement/fusion not present) or other (unknown result/indeterminate test). If more than one ALK test result was documented, the preference was to accept a positive result over a negative or unknown test result. This sequence was utilized as it would mimic what is observed in practice, as there is not 100% concordance between different methods of testing [33–36].
Per NCCN guidelines that were in place during this study period, [18] a patient was eligible for ALK testing if they had an adenocarcinoma, large cell or NSCLC not otherwise specified (NOS); patients with squamous cell carcinoma and no history of smoking were also eligible. Patients with squamous histology and smoking history were ineligible for ALK testing under NCCN guidelines during the study time period. Although NCCN guidelines recommended ALK testing for patients with small biopsy specimens or mixed squamous histology, we did not include this in our definition of NCCN eligibility criteria as this data was not well-captured in the database.
The status of other driver mutations or rearrangements (EGFR, KRAS, BRAF or ROS1) as well as PD-L1 status was retrieved from the EHR as reported.
Statistical analysis
Descriptive statistics were used to summarize patient characteristics. Continuous variables were summarized with mean and standard deviation (SD). Frequency counts and the percentage of patients within each category were reported for categorical variables. To test the differences in ALK rearrangement rates by categorical variables, χ2 test was used. All analyses were undertaken using R statistical package version 3.5.3 (The R Foundation, Vienna, Austria). Prevalence of ALK rearrangement was assessed overall and then stratified by patient characteristics such as age, gender, race, smoking status and histology.
CONFLICTS OF INTEREST
Dr. Bernicker reports receiving personal fees from Novartis, Pfizer, AstraZeneca and Guardant Health outside of the submitted work. The remaining authors report no conflicts of interest.
FUNDING
This work was funded by Roche Diagnostics Corporation, Indianapolis, IN.
References
1. Howlader N, Forjaz G, Mooradian MJ, Meza R, Kong CY, Cronin KA, Mariotto AB, Lowy DR, Feuer EJ. The Effect of Advances in Lung-Cancer Treatment on Population Mortality. N Engl J Med. 2020; 383:640–49. https://doi.org/10.1056/NEJMoa1916623. [PubMed].
2. Smeltzer MP, Wynes MW, Lantuejoul S, Soo R, Ramalingam SS, Varella-Garcia M, Meadows Taylor M, Richeimer K, Wood K, Howell KE, Dalurzo ML, Felip E, Hollenbeck G, et al. The International Association for the Study of Lung Cancer Global Survey on Molecular Testing in Lung Cancer. J Thorac Oncol. 2020; 15:1434–48. https://doi.org/10.1016/j.jtho.2020.05.002. [PubMed].
3. Schoenfeld AJ, Arbour KC, Rizvi H, Iqbal AN, Gadgeel SM, Girshman J, Kris MG, Riely GJ, Yu HA, Hellmann MD. Severe immune-related adverse events are common with sequential PD-(L)1 blockade and osimertinib. Ann Oncol. 2019; 30:839–44. https://doi.org/10.1093/annonc/mdz077. [PubMed].
4. Lin JJ, Chin E, Yeap BY, Ferris LA, Kamesan V, Lennes IT, Sequist LV, Heist RS, Mino-Kenudson M, Gainor JF, Shaw AT. Increased Hepatotoxicity Associated with Sequential Immune Checkpoint Inhibitor and Crizotinib Therapy in Patients with Non-Small Cell Lung Cancer. J Thorac Oncol. 2019; 14:135–40. https://doi.org/10.1016/j.jtho.2018.09.001. [PubMed].
5. Shaw AT, Bauer TM, de Marinis F, Felip E, Goto Y, Liu G, Mazieres J, Kim DW, Mok T, Polli A, Thurm H, Calella AM, Peltz G, Solomon BJ, and CROWN Trial Investigators. First-Line Lorlatinib or Crizotinib in Advanced ALK-Positive Lung Cancer. N Engl J Med. 2020; 383:2018–29. https://doi.org/10.1056/NEJMoa2027187. [PubMed].
6. Peters S, Camidge DR, Shaw AT, Gadgeel S, Ahn JS, Kim DW, Ou SI, Pérol M, Dziadziuszko R, Rosell R, Zeaiter A, Mitry E, Golding S, et al, and ALEX Trial Investigators. Alectinib versus Crizotinib in Untreated ALK-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2017; 377:829–38. https://doi.org/10.1056/NEJMoa1704795. [PubMed].
7. Britschgi C, Addeo A, Rechsteiner M, Delaloye R, Früh M, Metro G, Banini M, Gautschi O, Rothschild SI, Wild PJ, Banna GL, Curioni-Fontecedro A. Real-World Treatment Patterns and Survival Outcome in Advanced Anaplastic Lymphoma Kinase (ALK) Rearranged Non-Small-Cell Lung Cancer Patients. Front Oncol. 2020; 10:1299. https://doi.org/10.3389/fonc.2020.01299. [PubMed].
8. Lindeman NI, Cagle PT, Aisner DL, Arcila ME, Beasley MB, Bernicker EH, Colasacco C, Dacic S, Hirsch FR, Kerr K, Kwiatkowski DJ, Ladanyi M, Nowak JA, et al. Updated Molecular Testing Guideline for the Selection of Lung Cancer Patients for Treatment With Targeted Tyrosine Kinase Inhibitors: Guideline From the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. J Mol Diagn. 2018; 20:129–59. https://doi.org/10.1016/j.jmoldx.2017.11.004. [PubMed].
9. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, Fujiwara S, Watanabe H, Kurashina K, Hatanaka H, Bando M, Ohno S, Ishikawa Y, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. 2007; 448:561–66. https://doi.org/10.1038/nature05945. [PubMed].
10. Shaw AT, Yeap BY, Mino-Kenudson M, Digumarthy SR, Costa DB, Heist RS, Solomon B, Stubbs H, Admane S, McDermott U, Settleman J, Kobayashi S, Mark EJ, et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J Clin Oncol. 2009; 27:4247–53. https://doi.org/10.1200/JCO.2009.22.6993. [PubMed].
11. Koivunen JP, Mermel C, Zejnullahu K, Murphy C, Lifshits E, Holmes AJ, Choi HG, Kim J, Chiang D, Thomas R, Lee J, Richards WG, Sugarbaker DJ, et al. EML4-ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin Cancer Res. 2008; 14:4275–83. https://doi.org/10.1158/1078-0432.CCR-08-0168. [PubMed].
12. Boland JM, Erdogan S, Vasmatzis G, Yang P, Tillmans LS, Johnson MR, Wang X, Peterson LM, Halling KC, Oliveira AM, Aubry MC, Yi ES. Anaplastic lymphoma kinase immunoreactivity correlates with ALK gene rearrangement and transcriptional up-regulation in non-small cell lung carcinomas. Hum Pathol. 2009; 40:1152–58. https://doi.org/10.1016/j.humpath.2009.01.012. [PubMed].
13. Dai Z, Kelly JC, Meloni-Ehrig A, Slovak ML, Boles D, Christacos NC, Bryke CR, Schonberg SA, Otani-Rosa J, Pan Q, Ho AK, Sanders HR, Zhang ZJ, et al. Incidence and patterns of ALK FISH abnormalities seen in a large unselected series of lung carcinomas. Mol Cytogenet. 2012; 5:44. https://doi.org/10.1186/1755-8166-5-44. [PubMed].
14. Takeuchi K, Soda M, Togashi Y, Suzuki R, Sakata S, Hatano S, Asaka R, Hamanaka W, Ninomiya H, Uehara H, Lim Choi Y, Satoh Y, Okumura S, et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med. 2012; 18:378–81. https://doi.org/10.1038/nm.2658. [PubMed].
15. Li T, Maus MK, Desai SJ, Beckett LA, Stephens C, Huang E, Hsiang J, Zeger G, Danenberg KD, Astrow SH, Gandara DR. Large-scale screening and molecular characterization of EML4-ALK fusion variants in archival non-small-cell lung cancer tumor specimens using quantitative reverse transcription polymerase chain reaction assays. J Thorac Oncol. 2014; 9:18–25. https://doi.org/10.1097/JTO.0000000000000030. [PubMed].
16. Sacher AG, Dahlberg SE, Heng J, Mach S, Jänne PA, Oxnard GR. Association Between Younger Age and Targetable Genomic Alterations and Prognosis in Non-Small-Cell Lung Cancer. JAMA Oncol. 2016; 2:313–20. https://doi.org/10.1001/jamaoncol.2015.4482. [PubMed].
17. Viñal D, Martínez D, Higuera O, de Castro J. Genomic profiling in non-small-cell lung cancer in young patients. A systematic review. ESMO Open. 2021; 6:100045. https://doi.org/10.1016/j.esmoop.2020.100045. [PubMed].
18. Ettinger DS, Wood DE, Akerley W, Bazhenova LA, Borghaei H, Camidge DR, Cheney RT, Chirieac LR, D’Amico TA, Demmy TL, Dilling TJ, Dobelbower MC, Govindan R, et al, and National comprehensive cancer network. Non-Small Cell Lung Cancer, Version 6.2015. J Natl Compr Canc Netw. 2015; 13:515–24. https://doi.org/10.6004/jnccn.2015.0071. [PubMed].
19. Lam VK, Tran HT, Banks KC, Lanman RB, Rinsurongkawong W, Peled N, Lewis J, Lee JJ, Roth J, Roarty EB, Swisher S, Talasaz A, Futreal PA, et al. Targeted Tissue and Cell-Free Tumor DNA Sequencing of Advanced Lung Squamous-Cell Carcinoma Reveals Clinically Significant Prevalence of Actionable Alterations. Clin Lung Cancer. 2019; 20:30–36.e3. https://doi.org/10.1016/j.cllc.2018.08.020. [PubMed].
20. Bergethon K, Shaw AT, Ou SH, Katayama R, Lovly CM, McDonald NT, Massion PP, Siwak-Tapp C, Gonzalez A, Fang R, Mark EJ, Batten JM, Chen H, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012; 30:863–70. https://doi.org/10.1200/JCO.2011.35.6345. [PubMed].
21. Rimkunas VM, Crosby KE, Li D, Hu Y, Kelly ME, Gu TL, Mack JS, Silver MR, Zhou X, Haack H. Analysis of receptor tyrosine kinase ROS1-positive tumors in non-small cell lung cancer: identification of a FIG-ROS1 fusion. Clin Cancer Res. 2012; 18:4449–57. https://doi.org/10.1158/1078-0432.CCR-11-3351. [PubMed].
22. Kohno T, Ichikawa H, Totoki Y, Yasuda K, Hiramoto M, Nammo T, Sakamoto H, Tsuta K, Furuta K, Shimada Y, Iwakawa R, Ogiwara H, Oike T, et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med. 2012; 18:375–77. https://doi.org/10.1038/nm.2644. [PubMed].
23. Wang R, Hu H, Pan Y, Li Y, Ye T, Li C, Luo X, Wang L, Li H, Zhang Y, Li F, Lu Y, Lu Q, et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol. 2012; 30:4352–59. https://doi.org/10.1200/JCO.2012.44.1477. [PubMed].
24. Illei PB, Wong W, Wu N, Chu L, Gupta R, Schulze K, Gubens MA. ALK testing trends and patterns among community practices in the United States. JCO Precision Oncology. 2018; 2:1–11. https://doi.org/10.1200/po.18.00159.
25. Bernicker EH, Xiao Y, Abraham A, Yang B, Croix DA, Redpath S, Engstrom-Melnyk J, Shah R, Madala J, Allen TC. Adherence to National Comprehensive Cancer Network ALK Testing Guidelines for Patients with Advanced Non-Small Cell Lung Cancer in U.S. Community Medical Centers. Oncologist. 2021; 26:e1050–57. https://doi.org/10.1002/onco.13779. [PubMed].
26. Ulivi P, Chiadini E, Dazzi C, Dubini A, Costantini M, Medri L, Puccetti M, Capelli L, Calistri D, Verlicchi A, Gamboni A, Papi M, Mariotti M, et al. Nonsquamous, Non-Small-Cell Lung Cancer Patients Who Carry a Double Mutation of EGFR, EML4-ALK or KRAS: Frequency, Clinical-Pathological Characteristics, and Response to Therapy. Clin Lung Cancer. 2016; 17:384–90. https://doi.org/10.1016/j.cllc.2015.11.004. [PubMed].
27. Deng H, Liu C, Zhang G, Wang X, Liu Y. Lung adenocarcinoma with concurrent ALK and ROS1 rearrangement: A case report and review of the literatures. Pathol Res Pract. 2018; 214:2103–05. https://doi.org/10.1016/j.prp.2018.09.028. [PubMed].
28. Guibert N, Barlesi F, Descourt R, Léna H, Besse B, Beau-Faller M, Mosser J, Pichon E, Merlio JP, Ouafik L, Guichard F, Mastroianni B, Moreau L, et al. Characteristics and Outcomes of Patients with Lung Cancer Harboring Multiple Molecular Alterations: Results from the IFCT Study Biomarkers France. J Thorac Oncol. 2017; 12:963–73. https://doi.org/10.1016/j.jtho.2017.02.001. [PubMed].
29. National Comprehensive Cancer Network. Non-small cell lung cancer (Version 6.2021). https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf (accessed September 30, 2021).
30. Ma X, Long L, Moon S, Adamson BJS, Baxi SS. Comparison of population characteristics in real-world clinical oncology databases in the US: Flatiron Health, SEER, and NPCR. medRxiv. 2020. https://doi.org/10.1101/2020.03.16.20037143.
31. Birnbaum B, Nussbaum N, Seidl-Rathkopf K, Agrawal M, Estévez M, Estola E, Haimson J, He L, Larson P, Richardson P. Model-assisted cohort selection with bias analysis for generating large-scale cohorts from the EHR for oncology research. 2020. https://arxiv.org/pdf/2001.09765.pdf.
32. Edge SB, Bryd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC Cancer Staging Manual (7th edition). New York, NY: Springer; 2010.
33. van der Wekken AJ, Pelgrim R, ‘t Hart N, Werner N, Mastik MF, Hendriks L, van der Heijden EHFM, Looijen-Salamon M, de Langen AJ, Staal-van den Brekel J, Riemersma S, van den Borne BE, Speel EJM, et al. Dichotomous ALK-IHC Is a Better Predictor for ALK Inhibition Outcome than Traditional ALK-FISH in Advanced Non-Small Cell Lung Cancer. Clin Cancer Res. 2017; 23:4251–58. https://doi.org/10.1158/1078-0432.CCR-16-1631. [PubMed].
34. Lin C, Shi X, Yang S, Zhao J, He Q, Jin Y, Yu X. Comparison of ALK detection by FISH, IHC and NGS to predict benefit from crizotinib in advanced non-small-cell lung cancer. Lung Cancer. 2019; 131:62–68. https://doi.org/10.1016/j.lungcan.2019.03.018. [PubMed].
35. Friedlaender A, Banna G, Patel S, Addeo A. Diagnosis and Treatment of ALK Aberrations in Metastatic NSCLC. Curr Treat Options Oncol. 2019; 20:79. https://doi.org/10.1007/s11864-019-0675-9. [PubMed].
36. An HJ, An E, Rabizadeh S, Liao WL, Burrows J, Hembrough T, Kang JH, Park CK, Kim TJ. Quantitative Multiplexed Proteomics Could Assist Therapeutic Decision Making in Non-Small Cell Lung Cancer Patients with Ambiguous ALK Test Results. Cancers (Basel). 2021; 13:2337. https://doi.org/10.3390/cancers13102337. [PubMed].


