Introduction
Despite new treatment options, metastatic non-small cell lung cancer (NSCLC) continues to be associated with the highest cancer-related mortality rates worldwide [1]. In addition to the growing landscape of immuno-oncology, a multitude of driver mutations and alterations in genes such as EGFR (epidermal growth factor receptor), ALK (anaplastic lymphoma kinase), ROS1 (proto-oncogene tyrosine-protein kinase ROS1), BRAF (v-Raf murine sarcoma viral oncogene homolog B) and NTRK1-3 (neurotrophic tyrosine kinase receptors 1–3) represent targets for personalized treatment options. Therefore, molecular testing at diagnosis and at relapse is essential in advanced NSCLC [2] and is even gaining importance in early tumor stages [3].
Rearrangement of the ALK-gene can be detected in 3–8% of NSCLC patients [4], with the echinoderm microtubule-associated protein-like 4 (EML4) gene being the predominant genomic partner leading to the EML4-ALK fusion oncogene [5]. Aberrant ALK kinase activity can be specifically targeted with tyrosine kinase inhibitors (TKIs). The first-generation TKI crizotinib demonstrated superiority over standard platinum-based chemotherapy [6]. Subsequently, second- and third-generation ALK inhibitors such as alectinib [7], brigatinib [8] and lorlatinib [9] have shown increased benefit over crizotinib. Fourth-generation ALK inhibitors are currently being investigated [10]. However, the ALK-targeting approach loses effectiveness over time as on-target resistance mutations of ALK or ALK gene amplifications may occur which lead to disease progression [11–14].
Here, we report a novel ALK-exon 23 resistance mutation (p.A1200_G1201delinsW) that provided resistance to alectinib but was tractable by brigatinib. A subsequent off-target KRAS-exon 3 mutation (p.Q61K) was associated with secondary resistance to brigatinib but was basically sensitive to chemoimmunotherapy (CIT).
CASE REPORT
A 63-year-old man with a ten pack-year history of tobacco smoking presented in our clinic with immobilizing lower back pain, drenching night sweats, and a weight loss of three kilograms over a few weeks. Radiography and magnetic resonance imaging (MRI) of the spine showed a pathologic fracture of lumbar vertebra 1 (L1) with intrusion into the spinal canal. Follow-up computed tomography (CT) revealed tumor mass consistent with lung cancer in the left upper pulmonary lobe, as well as metastatic lesions in mediastinal lymph nodes, the liver and the left adrenal gland. 18F-fluorodeoxyglucose positron emission tomography (FDG PET), CT, and cerebral MRI scans revealed additionally multiple osseous and brain metastases. Both the pathological examination of tissues from vertebral bone resection and from a lung biopsy revealed a cytokeratine 7/TTF1-positive pulmonary adenocarcinoma with low PD-L1 expression (tumor proportion score: 20%) as determined by immunohistochemistry. Molecular genetic analyses using fluorescence-in-situ-hybridization (FISH) and RNA sequencing provided evidence of EML4-ALK fusion variant 1 (exon 13/ exon 20), which is the most frequent (33%) variant in lung cancer [15].
First-line treatment with the second-generation TKI alectinib was initiated with concurrent stereotactic radiotherapy of the bone and brain lesions. Treatment resulted in partial remission (PR) for 20 months (Figure 1). At this timepoint worsening of clinical status occurred and a CT scan showed isolated secondary progression of hepatic metastases. Liquid biopsy and tissue re-biopsy of a liver metastasis independently yielded evidence of a previously undescribed ALK-exon 23 indel mutation (p.A1200_G1201delinsW) via next-generation sequencing (NGS) (Figure 2). At this time, the patient rejected the recommended treatment with lorlatinib because of its potential adverse neurocognitive effects. Therefore, a second-line treatment with brigatinib was started leading to relief of clinical symptoms and a stable disease for four months. Subsequently, tertiary progression of the hepatic metastases was noted while a further liquid biopsy and liver sampling both failed to demonstrate the previous ALK resistance mutation. However, the EML4-ALK fusion was still detected in the liver specimen. Interestingly, a so far undescribed activating KRAS-exon 3 mutation (p.Q61K) was identified in both the liquid and liver biopsy via NGS analysis (Figure 2). An inactivating TP53 mutation (p.C238Y) was only found temporarily in a liquid biopsy but not in liver tissue at this time. Hence, therapy was switched to the third-generation TKI lorlatinib, which resulted in a stable disease for only two months. Due to further isolated hepatic disease progression the patient stopped TKI treatment and underwent combined chemoimmunotherapy and antiangiogenetic therapy according to the Impower150 protocol [16] based on discussion in a molecular tumor board at the West German Cancer Center. After four cycles of chemoimmunotherapy no further disease progression was observed, and the KRAS mutation disappeared from plasma as measured in a liquid biopsy. Hence, four cycles of maintenance therapy with atezolizumab and bevacizumab were conducted afterwards (Figure 2). Unfortunately, follow-up after four months revealed extensive hepatic and peritoneal disease progression, although cerebral MRI scans revealed no further evidence of previously detected brain metastases at all. Ultimately, rapid clinical deterioration resulted in patient’s death.
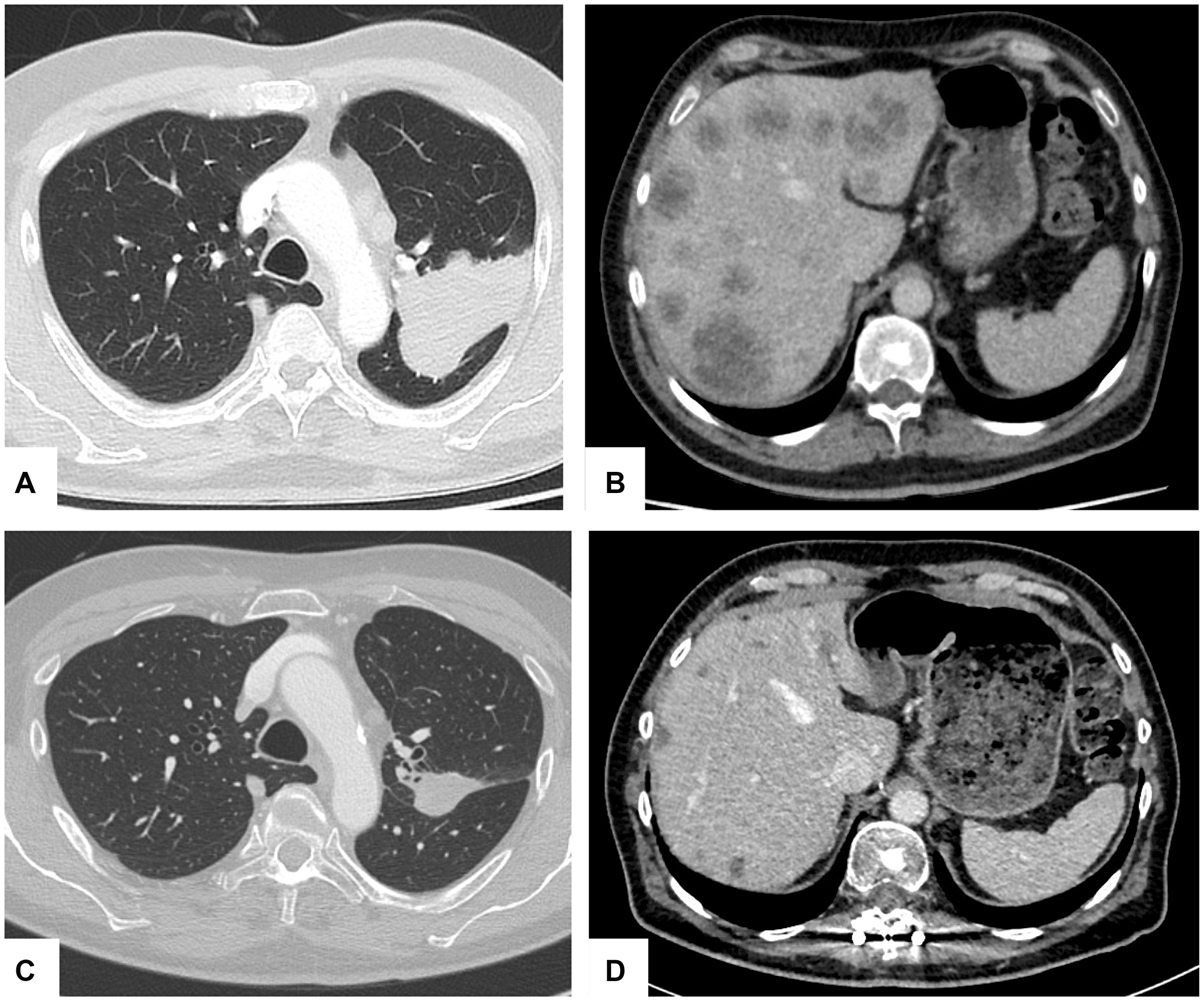
Figure 1: Hepatic and pulmonary tumor response during therapy. Axial computed tomography (CT) scans of left upper lobe pulmonary tumor and hepatic metastatic lesions at initial diagnosis (A, B) and after alectinib therapy ten months later (C, D) showing partial remission (PR).
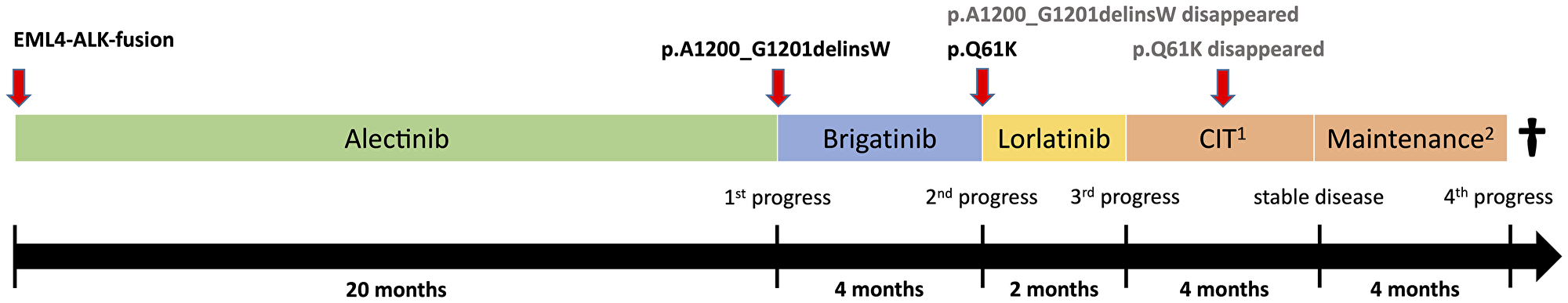
Figure 2: Therapy timeline and polyclonal tumor evolution. 1CIT: chemoimmuotherapy according to the Impower150 protocol with atezolizumab, carboplatin, paclitaxel and bevacizumab. 2Maintenance: Maintenance therapy with atezolizumab and bevacizumab.
DISCUSSION
On-target resistance mutations within the ALK gene have been shown to be common under treatment with second-generation ALK inhibitors such as alectinib [11]. There is a plethora of published data on preclinical characterization of these ALK mutations, and the activity of the common ALK-TKIs against these secondary mutations has been studied in vitro [11]. In contrast to this body of knowledge, molecular testing for ALK resistance mutations has not yet fully implemented in routine clinical practice. This is mainly due to the fact that second-line ALK-TKIs, namely brigatinib and lorlatinib, cover a nearly comparable spectrum of resistance mutations [11]. One exception is ALK-p.G1202R, which is the most common alteration after initial treatment with alectinib and which is thought to be more sensitive to lorlatinib than to brigatinib based on IC50 values [11, 13] and clinical data [12, 17]. However, there are no reports of any large-scale, head-to-head, randomized, controlled trials (RCT) in ALK-inhibitor-naïve NSCLC patients that compare currently the recommended dosages and administration frequency of the second-generation ALK inhibitor brigatinib with those of alectinib, which are currently both positioned as a first-line treatment option. Some of these questions are addressed by the ongoing prospective, randomized, open-label, phase II ABP study (NCT04318938). For the comparison of brigatinib and alectinib as second-line therapy after progression under crizotinib, the randomized, open label, phase III ALTA-3 study (NCT03596866) will add further information to this topic in future.
In this report, we describe a novel resistance mutation (ALK-p.A1200_G1201delinsW), which is located within the same genomic region encoding the kinase-domain. Based on our clinical findings from our patient we provide first evidence that i) this subtype of mutation confers resistance to alectinib as well and ii) brigatinib is obviously effective in this setting. We observed the disappearance of the ALK mutant clone and a clinical response of four months duration, which is basically in line with recently published progression free survival (PFS) data for brigatinib [18].
A tertiary progress of disease was induced in our patient by an unexpected off-target mutation, i.e., KRAS-p.Q61K. In contrast to other mutational subtypes [19, 20], the KRAS-p.Q61K mutation subtype has not yet been described as a resistance mutation in ALK-positive NSCLC but is known to be pathogenic from many other tumors [21, 22]. We found this specific KRAS mutation in both liquid biopsies and a liver tissue re-biopsy where the EML4-ALK fusion was still detectable. We decided to switch therapy to the third-generation TKI lorlatinib, as it has been shown efficacy in patients, who failed one or more second-generation TKIs [12]. However, this remained unsuccessful, which is in line with previous findings, that lorlatinib shows less efficacy in patients without ALK mutations [12]. Due to this short period (two months) under lorlatinib, we do not believe that lorlatinib treatment induced further resistance mutations in our patient. As the inactivating TP53 mutation temporarily presented in a liquid biopsy but not in liver tissue, we rather assumed the KRAS mutation, which was already detected before lorlatinib treatment both in liquid biopsy and liver tissue, to be the molecular driver of this hepatic tumor progression. Therefore, we stopped TKI treatment and subsequently started combined chemoimmunotherapy and antiangiogenetic treatment according to the Impower150 protocol [16]. For the subgroup of patients with baseline liver metastases, this trial protocol showed improved median overall survival (OS) [23]. Also in our patient, this regimen was clinically beneficial. After completion of four cycles stable disease was reached. Moreover, the KRAS-mutant clone disappeared from liquid biopsies after two cycles together with clinical improvement. However, this benefit lasted only a short period, as the tumor extensively progressed after four cycles of maintenance therapy. Follow-up liquid biopsies revealed no further on- and off-target resistance mutations (Table 1). In contrast to the remarkable five-year OS rate of 62,5% for patients with advanced ALK-positive NSCLC first-line treated with alectinib [24], the patient in this case report had a rather short survival. Although the Impower150 protocol has been shown to be beneficial for patients with liver metastases [23], the presence of liver metastases is still considered a negative prognostic marker in NSCLC [25] and in this patient, disease progression was related exclusively to the liver. As there was no detectable cerebral tumor progression, this did not impact on survival. Furthermore, the value of immunotherapy in the subgroup of ALK-positive NSCLC is yet unclear. The Impower150 trial was the first trial to show benefit of adding atezolizumab to chemotherapy in patients with EGFR or ALK mutation. However, the authors themselves assume that this might be explained by immunomodulatory effects of bevacizumab that augment the efficacy of atezolizumab [23]. For NSCLC patients with EGFR or ALK mutation previous trials did not show superiority of PD-L1 or PD-1 inhibitors compared to standard chemotherapy after failure of TKI therapy [26–28]. Further trials are needed as there are no RCTs yet that specifically focus on the value of immunotherapy in the subgroup of ALK-positive NSCLC, probably due to small patient numbers. In contrast, there are numerous RCTs that have shown the lacking benefit of immunotherapy in never smokers [29, 30]. The patient in this case report stopped smoking twenty years ago and had only a ten pack-year history of tobacco smoking. Thus, we assume that i) the baseline presence and progression of liver metastases, ii) the unknown value of immunotherapy in ALK-positive NSCLC as well as iii) it’s less efficacy in never smokers, explain why the patient in this case report had only a short-term benefit from CIT and progressed rapidly after maintenance therapy, resulting in patient’s death.
Table 1: Off-target resistance mechanisms and treatment options in ALK-positive NSCLC
| Off-target resistance mechanism | Treatment option | Literature |
|---|---|---|
| EGFR activation | Dual blockade of ALK and EGFR | Miyawaki, et al. 2017 [31] Tani, et al. 2016 [32] |
| HER activation | Dual blockade of ALK and EGFR | Tanizaki, et al. 2012 [33] Wilson, et al. 2015 [34] |
| KIT amplification | - | Katayama, et al. 2012 [35] |
| MET amplification | Dual blockade of ALK and MET | Dagogo-Jack, et al. 2020 [36] |
| MEK reactivation | Dual blockade of ALK and MEK | Hrustanovic, et al. 2015 [37] Shrestha, et al. 2020 [38] |
| PIK3CA mutations | - | Gainor, et al. 2016 [11] Crystal, et al. 2014 [39] |
| IGF-1R activation | Dual blockade of ALK and IGF-1R | Lovly, et al. 2014 [40] |
| SRC activation | Dual blockade of ALK and SRC | Crystal, et al. 2014 [39] Yoshida, et al. 2017 [41] |
| TP53 mutation | - | Gainor, et al. 2016 [11] |
| KRAS mutations (excluding G12C) | - | Doebele, et al. 2012 [20] Pailler, et al. 2019 [14] |
| KRAS G12C mutation | Sotorasib | McCoach, et al. 2018 [42] Hong, et al. 2020 [43] |
| RET fusion | Selpercatinib | Drilon, et al. 2020 [44] McCoach, et al. 2018 [42] |
Conclusions
In conclusion, this case report illustrates the development of two subsequent on- and off-target resistance mutations in a patient with ALK-fused NSCLC. We describe ALK-p.A1200_G1201delinsW as a so far undescribed mutation which confers resistance to alectinib but sensitivity to brigatinib. We provide further evidence that off-target KRAS-mutant clones i) give rise to resistance to ALK inhibitors in general and ii) might be addressed by chemoimmunotherapy. Therefore, our data highlights the importance of re-biopsies and molecular profiling to guide therapeutic decision-making according to clonal tumor evolution in ALK-positive NSCLC.
Consent for publication
The authors obtained written informed consent from the patient to publish information and images.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492. [PubMed].
2. Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, Mok TS, Reck M, Van Schil PE, Hellmann MD, Peters S. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018; 29:iv192–237. https://doi.org/10.1093/annonc/mdy275. [PubMed].
3. Wu YL, Tsuboi M, He J, John T, Grohe C, Majem M, Goldman JW, Laktionov K, Kim SW, Kato T, Vu HV, Lu S, Lee KY, et al, and ADAURA Investigators. Osimertinib in Resected EGFR-Mutated Non–Small-Cell Lung Cancer. N Engl J Med. 2020; 383:1711–23. https://doi.org/10.1056/NEJMoa2027071. [PubMed].
4. Devarakonda S, Morgensztern D, Govindan R. Genomic alterations in lung adenocarcinoma. Lancet Oncol. 2015; 16:e342–51. https://doi.org/10.1016/S1470-2045(15)00077-7. [PubMed].
5. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, Fujiwara S, Watanabe H, Kurashina K, Hatanaka H, Bando M, Ohno S, Ishikawa Y, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. 2007; 448:561–66. https://doi.org/10.1038/nature05945. [PubMed].
6. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, et al, and PROFILE 1014 Investigators. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014; 371:2167–77. https://doi.org/10.1056/NEJMoa1408440. [PubMed].
7. Peters S, Camidge DR, Shaw AT, Gadgeel S, Ahn JS, Kim DW, Ou SI, Pérol M, Dziadziuszko R, Rosell R, Zeaiter A, Mitry E, Golding S, et al, and ALEX Trial Investigators. Alectinib versus Crizotinib in Untreated ALK-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2017; 377:829–38. https://doi.org/10.1056/NEJMoa1704795. [PubMed].
8. Camidge DR, Kim HR, Ahn MJ, Yang JC, Han JY, Lee JS, Hochmair MJ, Li JY, Chang GC, Lee KH, Gridelli C, Delmonte A, Garcia Campelo R, et al. Brigatinib versus Crizotinib in ALK-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2018; 379:2027–39. https://doi.org/10.1056/NEJMoa1810171. [PubMed].
9. Shaw AT, Bauer TM, de Marinis F, Felip E, Goto Y, Liu G, Mazieres J, Kim DW, Mok T, Polli A, Thurm H, Calella AM, Peltz G, Solomon BJ, and CROWN Trial Investigators. First-Line Lorlatinib or Crizotinib in Advanced ALK-Positive Lung Cancer. N Engl J Med. 2020; 383:2018–29. https://doi.org/10.1056/NEJMoa2027187. [PubMed].
10. Cui JJ, Rogers E, Zhai D, Deng W, Ung J, Nguyen V, Zhang H, Zhang X, Parra A, Barrera M, Lee D, Murray B. Abstract 5226: TPX-0131: A next generation macrocyclic ALK inhibitor that overcomes ALK resistant mutations refractory to current approved ALK inhibitors. Cancer Res. 2020; 80:5226LP–5226. https://doi.org/10.1158/1538-7445.AM2020-5226.
11. Gainor JF, Dardaei L, Yoda S, Friboulet L, Leshchiner I, Katayama R, Dagogo-Jack I, Gadgeel S, Schultz K, Singh M, Chin E, Parks M, Lee D, et al. Molecular Mechanisms of Resistance to First- and Second-Generation ALK Inhibitors in ALK-Rearranged Lung Cancer. Cancer Discov. 2016; 6:1118–33. https://doi.org/10.1158/2159-8290.CD-16-0596. [PubMed].
12. Shaw AT, Solomon BJ, Besse B, Bauer TM, Lin CC, Soo RA, Riely GJ, Ou SI, Clancy JS, Li S, Abbattista A, Thurm H, Satouchi M, et al. ALK Resistance Mutations and Efficacy of Lorlatinib in Advanced Anaplastic Lymphoma Kinase-Positive Non-Small-Cell Lung Cancer. J Clin Oncol. 2019; 37:1370–79. https://doi.org/10.1200/JCO.18.02236. [PubMed].
13. Horn L, Whisenant JG, Wakelee H, Reckamp KL, Qiao H, Leal TA, Du L, Hernandez J, Huang V, Blumenschein GR, Waqar SN, Patel SP, Nieva J, et al. Monitoring Therapeutic Response and Resistance: Analysis of Circulating Tumor DNA in Patients With ALK+ Lung Cancer. J Thorac Oncol. 2019; 14:1901–11. https://doi.org/10.1016/j.jtho.2019.08.003. [PubMed].
14. Pailler E, Faugeroux V, Oulhen M, Mezquita L, Laporte M, Honoré A, Lecluse Y, Queffelec P, NgoCamus M, Nicotra C, Remon J, Lacroix L, Planchard D, et al. Acquired Resistance Mutations to ALK Inhibitors Identified by Single Circulating Tumor Cell Sequencing in ALK-Rearranged Non-Small-Cell Lung Cancer. Clin Cancer Res. 2019; 25:6671–82. https://doi.org/10.1158/1078-0432.CCR-19-1176. [PubMed].
15. Sabir SR, Yeoh S, Jackson G, Bayliss R. EML4-ALK variants: Biological and molecular properties, and the implications for patients. Cancers (Basel). 2017; 9:118. https://doi.org/10.3390/cancers9090118. [PubMed].
16. Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, Kelsch C, Lee A, et al, and IMpower150 Study Group. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N Engl J Med. 2018; 378:2288–301. https://doi.org/10.1056/nejmoa1716948. [PubMed].
17. Solomon BJ, Besse B, Bauer TM, Felip E, Soo RA, Camidge DR, Chiari R, Bearz A, Lin CC, Gadgeel SM, Riely GJ, Tan EH, Seto T, et al. Lorlatinib in patients with ALK-positive non-small-cell lung cancer: results from a global phase 2 study. Lancet Oncol. 2018; 19:1654–67. https://doi.org/10.1016/S1470-2045(18)30649-1. [PubMed].
18. Lin JJ, Zhu VW, Schoenfeld AJ, Yeap BY, Saxena A, Ferris LA, Dagogo-Jack I, Farago AF, Taber A, Traynor A, Menon S, Gainor JF, Lennerz JK, et al. Brigatinib in Patients With Alectinib-Refractory ALK-Positive NSCLC. J Thorac Oncol. 2018; 13:1530–38. https://doi.org/10.1016/j.jtho.2018.06.005. [PubMed].
19. Bordi P, Tiseo M, Rofi E, Petrini I, Restante G, Danesi R, Del Re M. Detection of ALK and KRAS Mutations in Circulating Tumor DNA of Patients With Advanced ALK-Positive NSCLC With Disease Progression During Crizotinib Treatment. Clin Lung Cancer. 2017; 18:692–97. https://doi.org/10.1016/j.cllc.2017.04.013. [PubMed].
20. Doebele RC, Pilling AB, Aisner DL, Kutateladze TG, Le AT, Weickhardt AJ, Kondo KL, Linderman DJ, Heasley LE, Franklin WA, Varella-Garcia M, Camidge DR. Mechanisms of resistance to crizotinib in patients with ALK gene rearranged non-small cell lung cancer. Clin Cancer Res. 2012; 18:1472–82. https://doi.org/10.1158/1078-0432.CCR-11-2906. [PubMed].
21. Wang Q, Zhong M, Lü YL, Yuan J, Wei LX. [Correlation of KRAS gene mutations and clinicopathologic parameters in colorectal carcinoma]. [Article in Chinese]. Zhonghua Bing Li Xue Za Zhi. 2012; 41:603–06. https://doi.org/10.3760/cma.j.issn.0529-5807.2012.09.007. [PubMed].
22. Sugimori M, Sugimori K, Tsuchiya H, Suzuki Y, Tsuyuki S, Kaneta Y, Hirotani A, Sanga K, Tozuka Y, Komiyama S, Sato T, Tezuka S, Goda Y, et al. Quantitative monitoring of circulating tumor DNA in patients with advanced pancreatic cancer undergoing chemotherapy. Cancer Sci. 2020; 111:266–78. https://doi.org/10.1111/cas.14245. [PubMed].
23. Reck M, Mok TSK, Nishio M, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, et al, and IMpower150 Study Group. Atezolizumab plus bevacizumab and chemotherapy in non-small-cell lung cancer (IMpower150): key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomised, open-label phase 3 trial. Lancet Respir Med. 2019; 7:387–401. https://doi.org/10.1016/S2213-2600(19)30084-0. [PubMed].
24. Mok T, Camidge DR, Gadgeel SM, Rosell R, Dziadziuszko R, Kim DW, Pérol M, Ou SI, Ahn JS, Shaw AT, Bordogna W, Smoljanović V, Hilton M, et al. Updated overall survival and final progression-free survival data for patients with treatment-naive advanced ALK-positive non-small-cell lung cancer in the ALEX study. Ann Oncol. 2020; 31:1056–64. https://doi.org/10.1016/j.annonc.2020.04.478. [PubMed].
25. Brueckl WM, Ficker JH, Zeitler G. Clinically relevant prognostic and predictive markers for immune-checkpoint-inhibitor (ICI) therapy in non-small cell lung cancer (NSCLC). BMC Cancer. 2020; 20:1185. https://doi.org/10.1186/s12885-020-07690-8. [PubMed].
26. Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE, Holgado E, Waterhouse D, Ready N, Gainor J, et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med. 2015; 373:123–35. https://doi.org/10.1056/NEJMoa1504627. [PubMed].
27. Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, Majem M, Fidler MJ, de Castro G Jr, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016; 387:1540–50. https://doi.org/10.1016/S0140-6736(15)01281-7. [PubMed].
28. Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols MC, Cortinovis DL, Leach J, Polikoff J, et al, and OAK Study Group. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017; 389:255–65. https://doi.org/10.1016/S0140-6736(16)32517-X. [PubMed].
29. Mo J, Hu X, Gu L, Chen B, Khadaroo PA, Shen Z, Dong L, Lv Y, Chitumba MN, Liu J. Smokers or non-smokers: who benefits more from immune checkpoint inhibitors in treatment of malignancies? An up-to-date meta-analysis. World J Surg Oncol. 2020; 18:15. https://doi.org/10.1186/s12957-020-1792-4. [PubMed].
30. Dai L, Jin B, Liu T, Chen J, Li G, Dang J. The effect of smoking status on efficacy of immune checkpoint inhibitors in metastatic non-small cell lung cancer: A systematic review and meta-analysis. EClinicalMedicine. 2021; 000:100990. https://doi.org/10.1016/j.eclinm.2021.100990.
31. Miyawaki M, Yasuda H, Tani T, Hamamoto J, Arai D, Ishioka K, Ohgino K, Nukaga S, Hirano T, Kawada I, Naoki K, Hayashi Y, Betsuyaku T, Soejima K. Overcoming EGFR Bypass Signal-Induced Acquired Resistance to ALK Tyrosine Kinase Inhibitors in ALK-Translocated Lung Cancer. Mol Cancer Res. 2017; 15:106–14. https://doi.org/10.1158/1541-7786.MCR-16-0211. [PubMed].
32. Tani T, Yasuda H, Hamamoto J, Kuroda A, Arai D, Ishioka K, Ohgino K, Miyawaki M, Kawada I, Naoki K, Hayashi Y, Betsuyaku T, Soejima K. Activation of EGFR Bypass Signaling by TGFα Overexpression Induces Acquired Resistance to Alectinib in ALK-Translocated Lung Cancer Cells. Mol Cancer Ther. 2016; 15:162–71. https://doi.org/10.1158/1535-7163.MCT-15-0084. [PubMed].
33. Tanizaki J, Okamoto I, Okabe T, Sakai K, Tanaka K, Hayashi H, Kaneda H, Takezawa K, Kuwata K, Yamaguchi H, Hatashita E, Nishio K, Nakagawa K. Activation of HER family signaling as a mechanism of acquired resistance to ALK inhibitors in EML4-ALK-positive non-small cell lung cancer. Clin Cancer Res. 2012; 18:6219–26. https://doi.org/10.1158/1078-0432.CCR-12-0392. [PubMed].
34. Wilson FH, Johannessen CM, Piccioni F, Tamayo P, Kim JW, Van Allen EM, Corsello SM, Capelletti M, Calles A, Butaney M, Sharifnia T, Gabriel SB, Mesirov JP, et al. A functional landscape of resistance to ALK inhibition in lung cancer. Cancer Cell. 2015; 27:397–408. https://doi.org/10.1016/j.ccell.2015.02.005. [PubMed].
35. Katayama R, Shaw AT, Khan TM, Mino-Kenudson M, Solomon BJ, Halmos B, Jessop NA, Wain JC, Yeo AT, Benes C, Drew L, Saeh JC, Crosby K, et al. Mechanisms of acquired crizotinib resistance in ALK-rearranged lung Cancers. Sci Transl Med. 2012; 4:120ra17. https://doi.org/10.1126/scitranslmed.3003316. [PubMed].
36. Dagogo-Jack I, Yoda S, Lennerz JK, Langenbucher A, Lin JJ, Rooney MM, Prutisto-Chang K, Oh A, Adams NA, Yeap BY, Chin E, Do A, Marble HD, et al. MET alterations Are a Recurring and Actionable Resistance Mechanism in ALK-Positive Lung Cancer. Clin Cancer Res. 2020; 26:2535–45. https://doi.org/10.1158/1078-0432.CCR-19-3906. [PubMed].
37. Hrustanovic G, Olivas V, Pazarentzos E, Tulpule A, Asthana S, Blakely CM, Okimoto RA, Lin L, Neel DS, Sabnis A, Flanagan J, Chan E, Varella-Garcia M, et al. RAS-MAPK dependence underlies a rational polytherapy strategy in EML4-ALK-positive lung cancer. Nat Med. 2015; 21:1038–47. https://doi.org/10.1038/nm.3930. [PubMed].
38. Shrestha N, Bland AR, Bower RL, Rosengren RJ, Ashton JC. Inhibition of Mitogen-Activated Protein Kinase Kinase Alone and in Combination with Anaplastic Lymphoma Kinase (ALK) Inhibition Suppresses Tumor Growth in a Mouse Model of ALK-Positive Lung Cancer. J Pharmacol Exp Ther. 2020; 374:134–40. https://doi.org/10.1124/jpet.120.266049. [PubMed].
39. Crystal AS, Shaw AT, Sequist LV, Friboulet L, Niederst MJ, Lockerman EL, Frias RL, Gainor JF, Amzallag A, Greninger P, Lee D, Kalsy A, Gomez-Caraballo M, et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science. 2014; 346:1480–86. https://doi.org/10.1126/science.1254721. [PubMed].
40. Lovly CM, McDonald NT, Chen H, Ortiz-Cuaran S, Heukamp LC, Yan Y, Florin A, Ozretić L, Lim D, Wang L, Chen Z, Chen X, Lu P, et al. Rationale for co-targeting IGF-1R and ALK in ALK fusion-positive lung cancer. Nat Med. 2014; 20:1027–34. https://doi.org/10.1038/nm.3667. [PubMed].
41. Yoshida R, Sasaki T, Minami Y, Hibino Y, Okumura S, Sado M, Miyokawa N, Hayashi S, Kitada M, Ohsaki Y. Activation of Src signaling mediates acquired resistance to ALK inhibition in lung cancer. Int J Oncol. 2017; 51:1533–40. https://doi.org/10.3892/ijo.2017.4140. [PubMed].
42. McCoach CE, Le AT, Gowan K, Jones K, Schubert L, Doak A, Estrada-Bernal A, Davies KD, Merrick DT, Bunn PA Jr, Purcell WT, Dziadziuszko R, Varella-Garcia M, et al. Resistance Mechanisms to Targeted Therapies in ROS1+ and ALK+ Non-small Cell Lung Cancer. Clin Cancer Res. 2018; 24:3334–47. https://doi.org/10.1158/1078-0432.CCR-17-2452. [PubMed].
43. Hong DS, Fakih MG, Strickler JH, Desai J, Durm GA, Shapiro GI, Falchook GS, Price TJ, Sacher A, Denlinger CS, Bang YJ, Dy GK, Krauss JC, et al. KRASG12C Inhibition with Sotorasib in Advanced Solid Tumors. N Engl J Med. 2020; 383:1207–17. https://doi.org/10.1056/NEJMoa1917239. [PubMed].
44. Drilon A, Oxnard GR, Tan DSW, Loong HHF, Johnson M, Gainor J, McCoach CE, Gautschi O, Besse B, Cho BC, Peled N, Weiss J, Kim YJ, et al. Efficacy of Selpercatinib in RET Fusion-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2020; 383:813–24. https://doi.org/10.1056/NEJMoa2005653. [PubMed].


