Introduction
Epithelial ovarian cancer (EOC) is a lethal disease with 5-year survival rates of 40% [1]. Only 15% of EOC patients are diagnosed with Stage I disease and have favorable 5 year survival rate of 92% [2]. However, lack of early detection methods leads to late stage disease at diagnosis and poor outcomes in majority of the patients. Women at a high risk for EOC are offered regular pelvic exams in combination with transvaginal ultrasound and a blood test for CA-125 tumor marker, but these strategies have failed to significantly improve patient survival [3]. Developing strategies to identify women with a high risk of developing EOC and to prevent tumor development is critical for reducing EOC-related mortality.
About 24% of EOCs are associated with hereditary conditions [4] and inherited heterozygous mutations in breast cancer susceptibility gene (BRCA) 1 and 2 account for 65–85% of hereditary ovarian tumors [5]. BRCA1 maintains genomic stability and exhibits tumor suppressive activities by regulating DNA repair, chromatin reorganization, transcription and ubiquitination [6]. BRCA1 mutation (BRCA1mut)-associated EOC is typically characterized by serous histology [7], and is more likely to be high grade and advanced stage [8]. Women with inherited BRCA1mut have a 44% probability of developing EOC by age 80 compared to a 2% probability in the general population [9], and also have an increased probability of early onset of EOC [10]. To date, the only reliable risk-reducing method is surgery, i. e., bilateral salpingo-oophorectomy [11], which involves high financial and emotional costs. Based on a cumulative EOC risk of 0.55% to age 35 for BRCA1mut carriers, an international registry study recommended bilateral salpingo-oophorectomy before age 40 to maximize prevention and to minimize adverse effects [12]. For pre-menopausal women, removing ovaries is associated with early menopause and infertility, and an increase in cardiovascular and neurological disease and osteoporosis [13]. Thus, there is an urgent need to develop other non-invasive and cost-effective methods to prevent EOC, particularly in young women carrying BRCA1mut.
Vitamin D (VD), a steroid hormone initially identified for its role in maintaining calcium homeostasis, exhibits pleotropic functions [14]. VD (cholecalciferol) is primarily produced in the skin upon UV light exposure, which is converted to calcifediol (major circulating form of VD) in the liver and then to calcitriol (biologically active form of VD) in the kidney [14]. VD binds to VD receptor (VDR), a member of the nuclear receptor superfamily, to activate downstream signaling and regulates expression of genes critical for cell proliferation, differentiation and apoptosis [14]. VDR expression has been reported in the ovary [15] and the fallopian tubes [16], from which EOC originates [17].
VD regulates expression of genes involved in the major hallmarks of cancer [18]. Epidemiological studies have demonstrated an inverse correlation between circulating calcifediol concentrations and cancer incidence in ovarian and other cancers [19, 20]. A Mendelian randomization study showed that genetically abated VD production was associated with increased risk of ovarian cancer in European women carrying specific single nucleotide polymorphisms (SNPs) of genes critical for VD biosynthesis [21]. Absence of VDR expression in BRCA1mut breast tumors was associated with impaired overall patient survival [22] and a shorter disease-free interval [23], suggesting a possible relationship between VD signaling and cancer development in BRCA1mut carriers. However, VD levels and actions in EOC development have not been reported in women with BRCA1mut.
Considering the regulatory effects of VD on cell proliferation, we investigated the potential for VD in the prevention of EOC in BRCA1mut carriers. We sought to determine circulating VD levels and VDR expression in ovarian surface epithelial (OSE)/fallopian tube epithelial (FTE) cells in healthy women with BRCA1mut. We also examined whether VD supplementation can regulate VDR expression and the proliferation of OSE cells from healthy BRCA1mut carriers.
Results
Reduced serum VD levels in healthy women with BRCA1mut
We analyzed serum calcifediol levels in healthy pre-menopausal women with BRCA1mut and BRCAwt patients. The BRCA1mut types and related functional consequences are summarized in Table 1. Calcifediol, the major circulating form of VD with a half-life of 2–3 weeks, is a common measure of systemic VD levels [24]. Calcifediol was detectable in all serum samples. Although ages were comparable between BRCA1mut carriers and BRCAwt patients (36.1 ± 1.7 versus 37.1 ± 1.4 years; p = 0.33), BRCA1mut carriers had ~33% lower (p = 0.003) serum calcifediol concentrations than those of BRCAwt patients (29.4 ± 2.3 versus 44.1 ± 4.0 ng/ml). Serum calcifediol level of 20–29.9 ng/ml is defined as VD insufficiency [25]. While ~92% (12 of 13) of BRCAwt patients had healthy levels of VD (> 30 ng/ml serum calcifediol), 54% (6 of 11) of BRCA1mut carriers exhibited VD insufficiency (21.7–27.7 ng/ml serum calcifediol) (Table 2).
Table 1: Summary of BRCA1 mutations in cancer-free women
| Experiment | Patient | BRCA1 mutation | Mutation Type | Clinical Significance | Functional consequence |
|---|---|---|---|---|---|
| Serum assay and immunohisto-chemistry | 1 | c.3916_3917del | Frameshift | Pathogenic | Truncated Protein |
| 2* | c.2722 G>T | Nonsense | Pathogenic | Truncated Protein | |
| 3 | c.5266dupC | Frameshift | Pathogenic | Truncated Protein | |
| 4 | c.1556del | Frameshift | Pathogenic | Truncated protein | |
| 5 | c.4065_4068del | Frameshift | Pathogenic | Truncated Protein | |
| 6* | c.4065_4068del | Frameshift | Pathogenic | Truncated Protein | |
| 7 | c.66_67AG [1] | Frameshift | Pathogenic | Truncated Protein | |
| 8 | c.2035A>T | Nonsense | Pathogenic | Truncated Protein | |
| 9* | c.66_67AG [1] | Frameshift | Pathogenic | Truncated Protein | |
| 10 | c.213-11T>G | Aberrant Splicing | Pathogenic | Nonsense mediated mRNA decay | |
| 11* | c.2722G>T | Nonsense | Pathogenic | Truncated Protein | |
| Ovarian surface epithelial cell culture | 1 | c.3112G>T | Nonsense | Pathogenic | Truncated protein |
| 2 | c.66dupA | Frameshift | Pathogenic | Truncated Protein | |
| 3 | c.851_852del | Frameshift | Pathogenic | Truncated protein |
Table 2: Serum 25-hydroxyvitamin D3 (calcifediol) levels in pre-menopausal cancer-free women with BRCA1 (BRCA1mut) or without (control) germline mutations in BRCA1 (BRCAwt)
| Patient | Age (years) | Calcifediol (ng/ml) | |
|---|---|---|---|
| BRCAwt | 1 | 41 | 56.96 |
| 2 | 44 | 45.94 | |
| 3 | 30 | 46.06 | |
| 4 | 37 | 53.35 | |
| 5 | 43 | 45.82 | |
| 6 | 35 | 69.80 | |
| 7 | 34 | 33.00 | |
| 8 | 28 | 34.25 | |
| 9 | 35 | 40.75 | |
| 10 | 42 | 34.65 | |
| 11 | 36 | 61.35 | |
| 12 | 43 | 14.69 | |
| 13 | 34 | 36.39 | |
| BRCA1mut | 1 | 41 | 23.44 |
| 2 | 45 | 30.32 | |
| 3 | 30 | 22.12 | |
| 4 | 36 | 33.47 | |
| 5 | 43 | 23.46 | |
| 6 | 35 | 21.71 | |
| 7 | 32 | 45.99 | |
| 8 | 28 | 38.50 | |
| 9 | 33 | 27.66 | |
| 10 | 41 | 30.09 | |
| 11 | 33 | 26.53 |
Reduced VDR expression in OSE and FTE in healthy women with BRCA1mut
VD is known to regulate cellular VDR expression [26] and is essential for the biological actions of VD [14]. Therefore, VDR protein levels in OSE and FTE were examined in healthy BRCA1mut and BRCAwt patients. VDR immunostaining (brown) was detected in the nucleus (DNA-bound) and cytoplasm (free form) of OSE (Figure 1A and 1B) and FTE (Figure 1D and 1E) cells, in addition to granulosa cells of ovarian follicles known for VDR expression (Figure 1A and 1B) [27]. VDR staining was intense in OSE and FTE cells, as well as in granulosa cells of ovarian follicles, in BRCAwt patients (Figure 1A and 1D). In contrast, VDR staining intensity was reduced in BRCA1mut carriers (Figure 1B and 1E). VDR immunostaining was absent in the negative control ovarian and fallopian tube sections with and without counterstaining, respectively (Figure 1C and 1F).
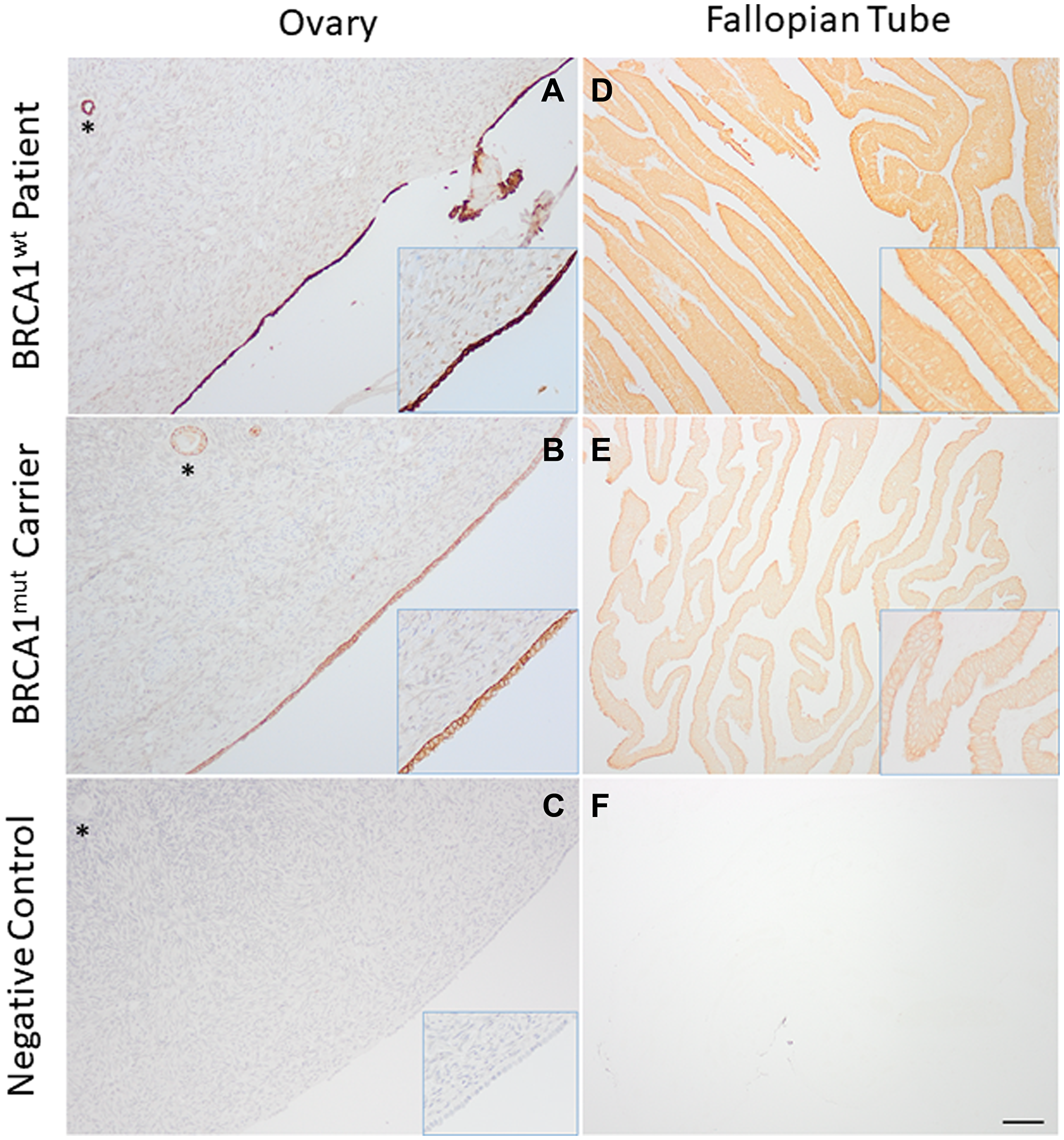
Figure 1: Vitamin D receptor (VDR) expression in the ovary and fallopian tube. Ovarian surface (A and B) and fallopian tube (D and E) epithelial cells in healthy women with BRCA1 mutations (BRCA1mut; A and D) and women without BRCA mutations (BRCAwt; B and E) exhibited positive immunostaining of VDR (brown in representative sections). Immunohistochemistry was performed with (A and B) or without (D and E) counterstain hematoxylin to allow for visualization of VDR nuclear and cytoplasmic staining. Phosphate-buffered saline was used instead of anti-VDR antibody for the negative control ovarian (C) and fallopian tube (F) sections. Immunohistochemistry was performed with (C) or without (F) counterstain hematoxylin. *ovarian follicle. Scale bar = 150 μm for A–F, and 75 μm for the insert (higher magnification).
Increased VDR protein levels in BRCA1mut OSE in response to VD treatment
To further examine VD regulated VDR expression, OSE cells from 3 healthy women with different germline BRCA1mut (Table 1) were cultured without (control) or with calcitriol, the biologically active form of VD that binds to VDR and activates downstream events [14]. VDR protein was detectable in OSE cells from all 3 BRCA1mut patients after 72 hrs of culture without calcitriol supplementation (Figure 2A). VDR protein levels in BRCA1mut OSE cells treated with calcitriol were elevated by ~95% relative to those of cells cultured under control conditions (VDR/αTubulin = 0.53 ± 0.05 versus 0.27 ± 0.03; p = 0.005) (Figure 2B).
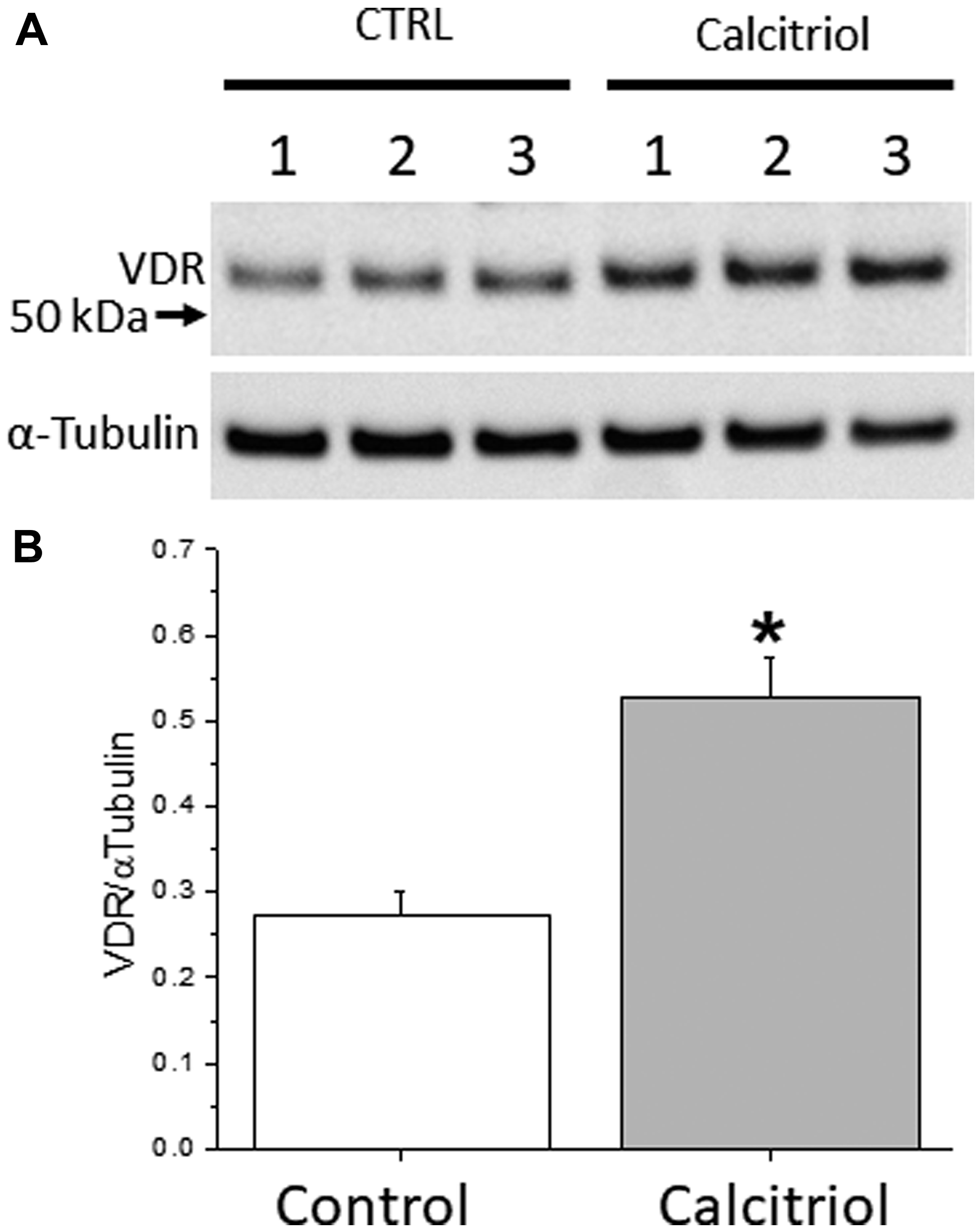
Figure 2: Vitamin D receptor (VDR) expression in cultured ovarian surface epithelial (OSE) cells. OSE cells derived from 3 healthy women with different germline BRCA1 mutations (patient 1–3) were cultured without (control) or with 5 nM calcitriol supplementation in the medium for 72 hrs. VDR protein levels were analyzed by Western blot (A) followed by densitometry analysis (B). The α-Tubulin served as the loading control. *significant difference between culture groups, p < 0.05. Data are presented as the mean ± SEM.
Reduced BRCA1mut OSE cell proliferation in response to VD treatment
VD is known to attenuate cell proliferation in cultured ovarian cancer cell lines by inducing cell cycle arrest [28]. Therefore, a dose-response experiment was performed in OSE cells from 3 healthy women with different germline BRCA1mut to examine cell proliferation in response to 72 hrs of calcitriol supplementation. The number of cultured BRCA1mut OSE cells decreased (p < 0.05) with the calcitriol level as low as 1 nM, and further declined with the calcitriol dose increased to 10 nM (Figure 3). Similar dose-dependent cell proliferation patterns were observed in cultured BRCA1mut OSE cells from all 3 patients regardless of their BRCA1mut types (Figure 3).
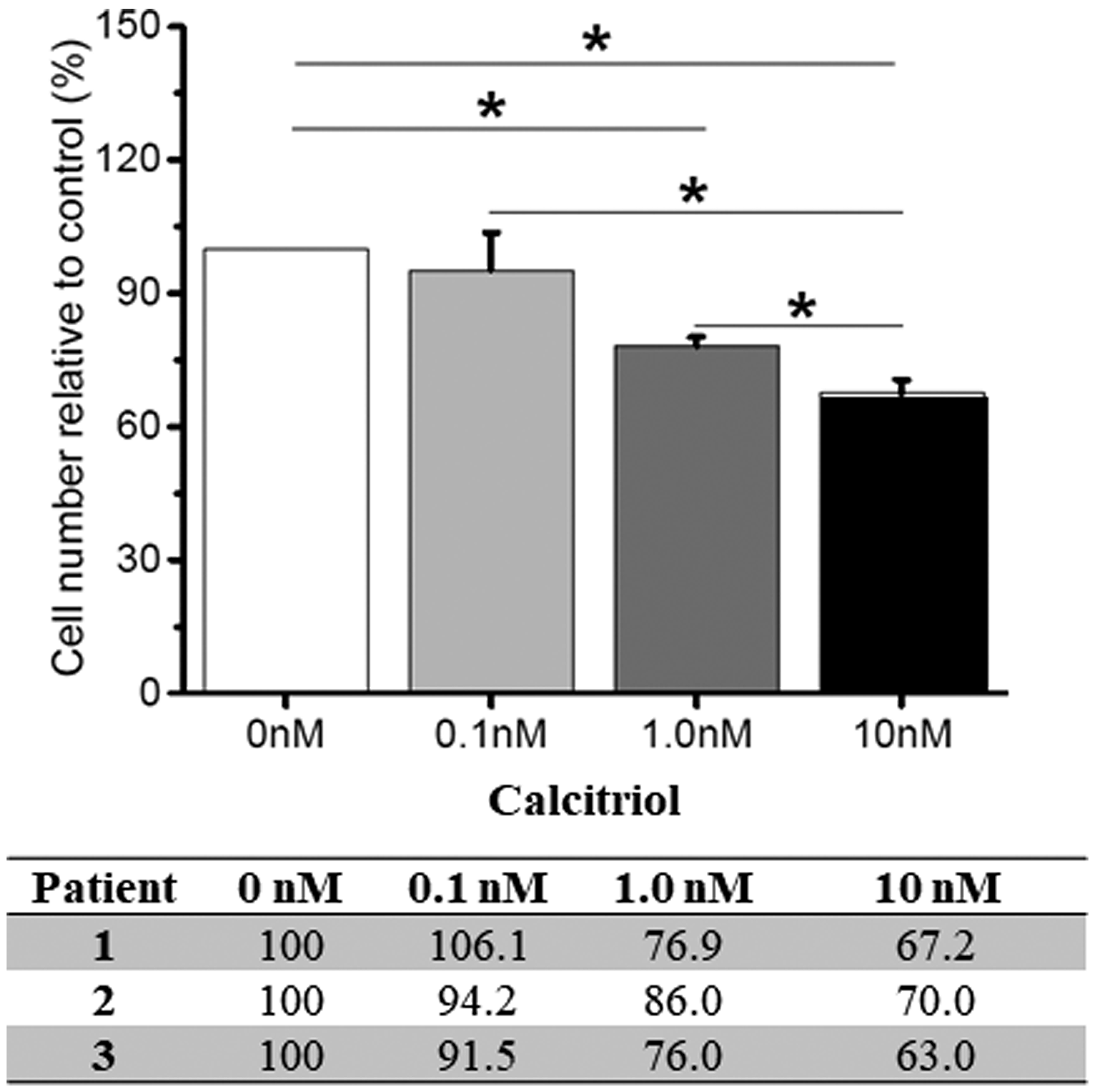
Figure 3: Vitamin D-regulated proliferation of cultured ovarian surface epithelial (OSE) cells. OSE cells derived from 3 healthy women with different germline BRCA1 mutations (patient 1–3) were cultured with 0 (control), 0.1, 1.0 or 10 nM calcitriol supplementation in the medium for 72 hrs. Cells were counted and cell proliferation was presented as the ratio (%) of number of cells in the calcitriol culture to those of the control (0 nM) culture. Data from individual patients were presented in the table. *significant difference between culture groups, p < 0.05. Data are presented as the mean ± SEM.
Unchanged BRCA1mut OSE cell viability upon VD treatment
As high-dose VD can induce apoptosis in cultured ovarian cancer cell lines [29], cytotoxic effects of VD were evaluated in OSE cells from 3 healthy women with different germline BRCA1mut cultured without (control) or with calcitriol. After 72 hrs of culture, the majority of BRCA1mut OSE cells were alive (green) regardless of BRCA1mut types of patients or cell culture conditions (Figure 4). The numbers of dead (red) BRCA1mut OSE cells were comparable between control and calcitriol treatment group (11.2 ± 0.8 versus 11.1 ± 0.5 cells; p = 0.45) (Figure 4).
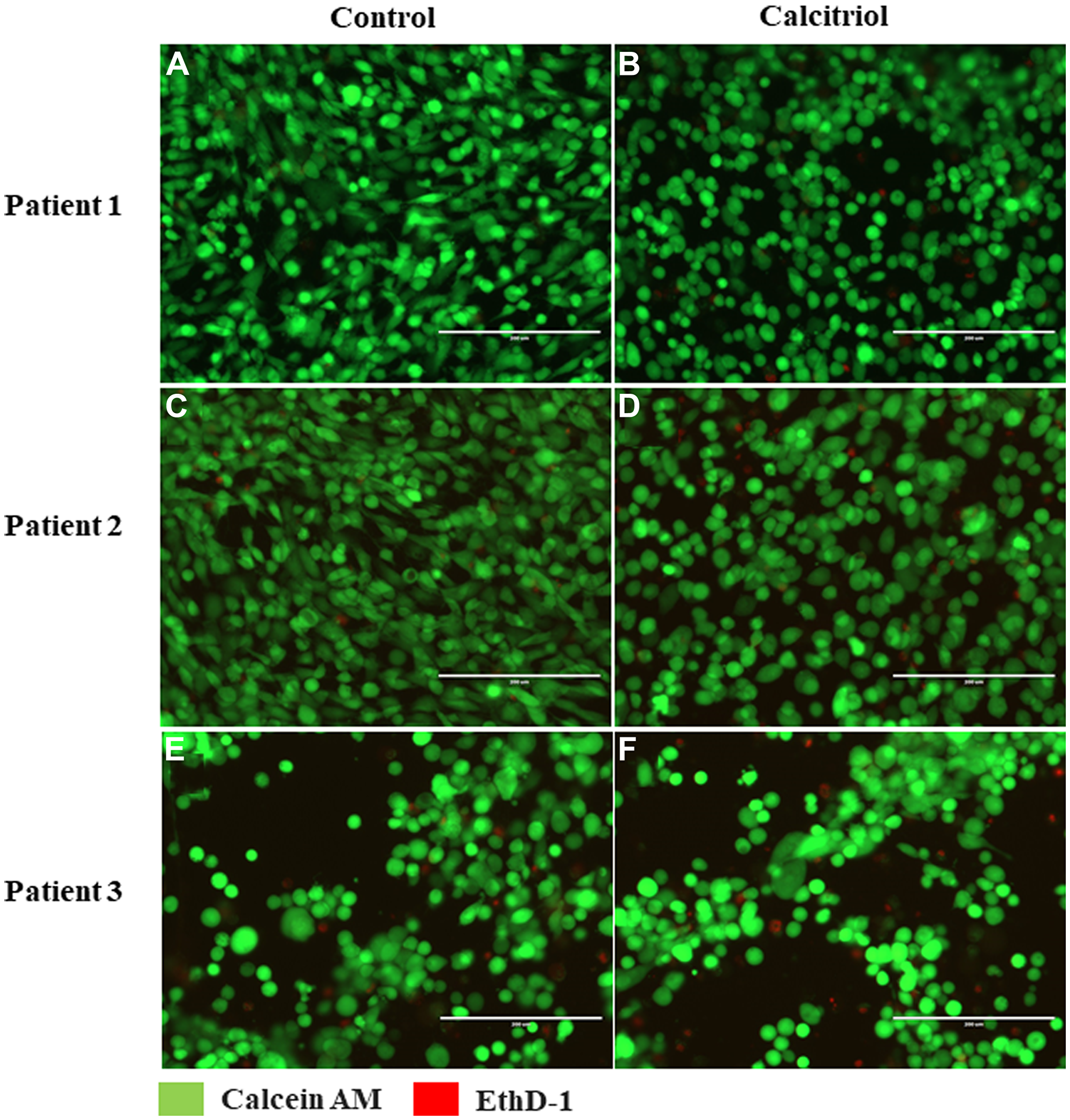
Figure 4: Vitamin D-regulated viability of cultured ovarian surface epithelial (OSE) cells. OSE cells derived from 3 healthy women with different germline BRCA1 mutations (patient 1–3) were cultured without (control (A, C, E)) or with 5 nM calcitriol supplementation (B, D, F) in the medium for 72 hrs. Cell viability was assessed by live/dead cell analysis via incubation with calcein AM (green; live cells) and EthD-1 (red; dead cells). Scale bar = 200 μm.
DISCUSSION
Women with BRCA1mut face a difficult decision regarding risk-reducing surgery. Although bilateral salpingo-oophorectomy is associated with a substantial reduction in the risk of ovarian, fallopian tube, or peritoneal cancer in BRCA1mut carriers [30], the incidence of primary peritoneal carcinoma has been reported after surgery [31]. As FTE has been identified as a site of origin for many serous EOC, so-called “staged” surgery with removal of the fallopian tubes at an earlier age, followed by removal of ovaries later before the age of 45, is being evaluated in clinical trials [32]. However, this protocol has potential risks of failing to eliminate EOC completely and to reduce the risk of breast cancer due to preserving ovaries till a later stage [33]. Therefore, the development and application of non-surgical preventive approaches are needed [34]. The role of oral contraceptives, non-steroidal anti-inflammatory drugs and vitamin A analogues and VD have been investigated in reducing the risk of EOC development in the general population [35]. Nevertheless, data in BRCA1mut carriers is limited.
We demonstrate for the first time that VD biosynthesis and/or metabolism is compromised in BRCA1mut carriers. While BRCAwt women had a normal VD status (44.1 ng/ml on average), BRCA1mut carriers exhibited VD-insufficiency (29.4 ng/ml on average) according to the clinical definition (20–29.9 ng/ml serum calcifediol) [25], despite a lack of data on UV light exposure and diet supplementation. Ineffective conversion of skin-derived and dietary cholecalciferol into calcifediol in the liver may result from previously reported BRCA1mut-related liver dysfunction [36]. In addition, a prior study showed that plasma levels of VD-binding protein isotypes 1 and 2 were significantly reduced in BRCA1mut carriers compared to BRCAwt patients [37]. The impaired VD metabolite transportation between the skin, liver and kidney are likely to limit VD metabolism [14]. These observations have important implications for calcifediol or calcitriol, instead of cholecalciferol, as an effective VD supplementation in BRCA1mut carriers.
VDR protein expression was reduced in OSE and FTE of healthy women with BRCA1mut relative to BRCAwt patients, as suggested by VDR immunostaining intensity. The difference in VDR protein expression was also observed in ovarian follicles between patient groups. Our data are consistent with in vitro studies showing that the number of DNA binding sites occupied by VDR are dynamically controlled by VD availability to cells [38]. Low systemic VD levels may lead to reduced VD signaling activity and diminished VDR expression in OSE, FTE and ovarian follicle granulosa cells in BRCA1mut carriers.
The dynamic VDR expression in response to VD exposure was further examined in cultured OSE cells from healthy women with BRCA1mut. In vitro treatment with a relatively low dose calcitriol significantly increased VDR protein expression in OSE cells compared to that of cells cultured without calcitriol. The data are consistent with previous observation of VD-dependent VDR expression in the nonhuman primate ovary [27]. Granulosa cell VDR gene expression in cultured rhesus macaque ovarian follicles was elevated following calcitriol treatment, which improved VD actions [39]. Studies in mouse pre-osteoblastic cells [40] and human lymphoblastoid cell lines [41] have reported VDR expression and binding to its specific response DNA elements under VD-depleted conditions, which are enhanced by calcitriol treatment resulting in increased VDR occupancy and elevated downstream gene expression. Similar dynamic VDR expression could occur in vivo wherein the number of accessible DNA binding sites for VDR positively corelates to cellular VD stimulation. Thus, VD activity via VDR in OSE and FTE cells could be improved by VD supplementation in BRCA1mut carriers.
The role of VD in prevention of cancers, including EOC, has remained inconclusive despite significant research effort [42]. Clinically, a tentative inverse association between circulating VD levels and ovarian cancer incidence was found by meta-analysis of existing data [19]. VD is known to regulate the expression of multiple proteins involved in the regulation of cell cycle and apoptosis [18]. Our data suggests that VD treatment may limit cell growth and/or division via maintaining cell cycle arrest. In addition, studies have previously demonstrated VD-enhanced DNA damage repair in uterine fibromas [43] and Ras oncogene-induced senescent fibroblasts [44]. Lending further support to linking VD to BRCA1, Deng et al. reported that BRCA1 mediated nuclear transport of VDR and VD-induced VDR expression in rat osteosarcoma cells [45]. Campbell et al. proposed the model in which the anti-proliferative effect of VD was associated with induction of BRCA1 gene expression via VDR in breast and prostate cancer cells [46]. Although the underlying mechanism requires further investigation, VD supplementation appears to have a potential to prevent or limit EOC development in BRCA1mut carriers.
Supplementation of VD and calcium was found to reduce all cancer risk in postmenopausal women [47]. A randomized clinical trial showed that cancer incidence was significantly lower in patients receiving VD plus calcium supplementation as compared to the control and calcium-only supplementation groups [48]. Another study reported a 14–20% decrease in breast cancer and colorectal cancer incidence in women taking 400 IU/day calcifediol and 1000 mg/day calcium for 8 years [49]. Thus, investigating the effect of VD supplementation on EOC incidence in BRCA1mut carriers will further the knowledge on precise role of VD in EOC prevention.
Studies on VD-associated cancer prevention have generated conflicting results due to the limitations of combining different types of cancers which are heterogeneous and arise from the dysregulation of multiple cellular processes. Our study focuses on VD action in BRCA1mut carriers using biological materials from healthy women with distinct BRCA1mut. Studies of VD action are limited to in vitro experiments using cell culture, which show promising results. Experiments in animal models with a BRCA1mut background are needed to further explore VD effects on cancer development in vivo.
Collectively, our results suggest a potential chemo-preventive role of VD in EOC development in women with BRCA1mut. Future studies will investigate the relationship between impaired VD action and EOC incidence in BRCA1mut carriers, as well as the mechanism and role of VD in EOC prevention in women with BRCA1mut. The availability of a chemo preventive strategy for BRCA1mut carriers, such as VD supplementation, will be a significant improvement on current risk reducing strategies. As mutations in BRCA1 gene also result in an increased risk of breast cancer [9], our findings may provide important implications for breast cancer prevention in BRCA1mut carriers.
Materials and Methods
Patient sample collection
Ovarian and fallopian tube tissue samples and corresponding blood samples, as well as clinical information, were obtained from the Oregon Ovarian Cancer Registry and Tissue Repository (OOCRTR) and the Oregon Clinical and Translational Research Institute (OCTRI) at the Oregon Health & Science University (OHSU). All samples were collected with informed consent, and the experimental protocol was approved by the Institutional Review Board (IRB; #921 and #3485).
Systemic VD levels in patients
Systemic VD levels of patients were determined using electrochemiluminescence immunoassay. Blood samples were collected from heathy pre-menopausal (≤ 45 years old) women with BRCA1mut (n = 11) (Table 1) and women without BRCA mutations (BRCAwt; n = 13). Serum calcifediol (major circulating form of VD) concentrations were measured using a Cobas Elecsys kit (catalog number: 06506780160; Roche Diagnostics, Indianapolis, IN) by the Endocrine Technologies Core at the Oregon National Primate Research Center, OHSU, as previously described [39].
VDR expression in OSE and FTE
VDR expression in OSE and FTE were evaluated using immunohistochemistry, as previously reported [27]. Ovarian and fallopian tube tissues were collected from BRCAwt patients (n = 5 for the ovary; n = 3 for the fallopian tube) and BRCA1mut carriers (n = 4) (Table 1), and fixed for paraffin embedding. Deparaffinized 5 μm sections were rehydrated in ethanol, followed by incubation at 4°C overnight with mouse anti-human VDR antibody (1:200; sc-13133; Santa Cruz Biotechnology, Inc., Santa Cruz, CA). Phosphate-buffered saline was used instead of the first antibody as negative control. Sections were then incubated with the secondary antibody and processed using the ImmPress HRP Reagent Kit (MP-7402, Vector Laboratories, Burlingame, CA). The antigen-antibody complex was visualized by incubation with 3,3′-diaminobenzidine. Ovarian tissue sections were counterstained using hematoxylin to demonstrate the nuclear versus cytoplasmic VDR staining. Images were captured via an Olympus BX40 inverted microscope and an Olympus DP72 digital camera (Olympus Imaging America Inc., Center Valley, PA, USA).
OSE cell culture
OSE cells from healthy women with BRCA1mut (n = 3) (Table 1) who underwent risk-reducing bilateral salpingo-oophorectomy were kindly provided by Dr. Elizabeth Swisher, University of Washington, Seattle, WA. Cells were cultured in defined medium: 50/50 (v/v) DMEM/RPMI 1640 supplemented with 20% (v/v) fetal bovine serum, epidermal growth factor (0.01 μg/ml), gentamycin (50 μg/ml), ciprofloxacin (10 μg/ml), insulin (10 μg/ml) and penicillin/streptomycin (100 μg/ml) (Sigma-Aldrich, St. Louis, MO) in 5% CO2 at 37°C. Cells grew to approximately 80% confluence prior to harvesting.
VDR expression in cultured OSE cells
OSE cells (n = 3 patients) were plated at 100,000 cells/well in a 6-well plate. Twenty-four hours post plating, cells were treated with 0 (control) or 5 nM calcitriol (biologically active form of VD; Sigma-Aldrich) supplementation in the culture medium for 72 hrs. Cells were harvested for Western blot, as previously described [50]. Briefly, total protein was extracted using a RIPA buffer containing 150 mM NaCl, 1% (v/v) NP-40, 1% (w/v) sodium deoxycholate, 0.1% (w/v) SDS, and 25 mM Tris-HCl (pH 7.6), electrophoresed on 4–12% Bis-Tris gel, and transferred to a nitrocellulose membrane. The membrane was blocked with 5% (w/v) nonfat milk in 10 mM Tris-buffered saline (pH 8.0), and then incubated with mouse anti-human VDR (1:250; sc-13133; Santa Cruz Biotechnology, Inc.) or α-Tubulin (loading control; 1:24,000; T6074; Sigma-Aldrich) antibody at 4°C overnight. The membrane was subsequently incubated with horseradish peroxidase-conjugated secondary antibody (Abcam, Burlingame, CA, USA). Images developed on the film were scanned using the Konica Minolta Bizhub C368 (Konica Minolta, Wayne, NJ, USA). Densitometry analysis was performed using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Viability of cultured OSE cells
OSE cells (n = 3 patients) were plated at 5,000 cells/well in a 96-well plate. Twenty-four hours post plating, cells were treated with 0 (control) or 5 nM calcitriol supplementation in the culture medium for 72 hrs. Cell viability was assessed using the LIVE/DEAD Viability/Cytotoxicity Kit (Catalogue # 10237012, Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s directions. Briefly, cells were incubated with 1 μM calcein AM and 2 μM EthD-1 at room temperature for 30 min. Images were captured using an EVOS FL imaging system (Themo Fisher Scientific, Waltham, MA, USA). All experiments were carried out in triplicate.
Proliferation of cultured OSE cells
OSE cells (n = 3 patients) were plated at 100,000 cells/well in a 6-well plate. Twenty-four hours post plating, cells were treated with 0, 0.1, 1 or 10 nM calcitriol supplementation in the culture medium for 72 hrs. Cells were then harvested and counted using a hemocytometer under a Leica DM IL microscope (Leica, Wetzler, Germany). All experiments were carried out in triplicate.
Statistical analysis
Data involving two and multiple experimental groups were analyzed by a Student’s t-test and one-way analysis of variance, respectively, using SigmaPlot 11 software (SPSS, Chicago, IL).
ACKNOWLEDGMENTS
We would like to thank Dr. Elizabeth M. Swisher at the University of Washington, Seattle, WA for providing us with OSE cells. We would also like to thank the Endocrine Technologies Core at the Oregon National Primate Research Center, OHSU for measuring serum calcifediol levels.
CONFLICTS OF INTEREST
Authors have no conflicts of interest to declare.
FUNDING
Support for this research was generously provided by the Sherie Hildreth Ovarian Cancer Foundation and the OHSU Foundation (award number 14020) (T.P.). This project was also supported by the National Institutes of Health Office of the Director P51OD011092 (J.X.).
References
1. Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD, Gaudet MM, Jemal A, Siegel RL. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018; 68:284–296. https://doi.org/10.3322/caac.21456. [PubMed].
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020; 70:7–30. https://doi.org/10.3322/caac.21590. [PubMed].
3. Elias KM, Guo J, Bast RC Jr. Early Detection of Ovarian Cancer. Hematol Oncol Clin North Am. 2018; 32:903–914. https://doi.org/10.1016/j.hoc.2018.07.003. [PubMed].
4. Walsh T, Casadei S, Lee MK, Pennil CC, Nord AS, Thornton AM, Roeb W, Agnew KJ, Stray SM, Wickramanayake A, Norquist B, Pennington KP, Garcia RL, et al. Mutations in 12 genes for inherited ovarian, fallopian tube, and peritoneal carcinoma identified by massively parallel sequencing. Proc Natl Acad Sci U S A. 2011; 108:18032–18037. https://doi.org/10.1073/pnas.1115052108. [PubMed].
5. Toss A, Tomasello C, Razzaboni E, Contu G, Grandi G, Cagnacci A, Schilder RJ, Cortesi L. Hereditary ovarian cancer: not only BRCA 1 and 2 genes. Biomed Res Int. 2015; 2015:341723. https://doi.org/10.1155/2015/341723. [PubMed].
6. Huen MS, Sy SM, Chen J. BRCA1 and its toolbox for the maintenance of genome integrity. Nat Rev Mol Cell Biol. 2010; 11:138–148. https://doi.org/10.1038/nrm2831. [PubMed].
7. Mavaddat N, Barrowdale D, Andrulis IL, Domchek SM, Eccles D, Nevanlinna H, Ramus SJ, Spurdle A, Robson M, Sherman M, Mulligan AM, Couch FJ, Engel C, et al. Pathology of breast and ovarian cancers among BRCA1 and BRCA2 mutation carriers: results from the Consortium of Investigators of Modifiers of BRCA1/2 (CIMBA). Cancer Epidemiol Biomarkers Prev. 2012; 21:134–147. https://doi.org/10.1158/1055-9965.EPI-11-0775. [PubMed].
8. Soegaard M, Kjaer SK, Cox M, Wozniak E, Hogdall E, Hogdall C, Blaakaer J, Jacobs IJ, Gayther SA, Ramus SJ. BRCA1 and BRCA2 mutation prevalence and clinical characteristics of a population-based series of ovarian cancer cases from Denmark. Clin Cancer Res. 2008; 14:3761–3767. https://doi.org/10.1158/1078-0432.CCR-07-4806. [PubMed].
9. Kuchenbaecker KB, Hopper JL, Barnes DR, Phillips KA, Mooij TM, Roos-Blom MJ, Jervis S, van Leeuwen FE, Milne RL, Andrieu N, Goldgar DE, Terry MB, Rookus MA, et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA. 2017; 317:2402–2416. https://doi.org/10.1001/jama.2017.7112. [PubMed].
10. Petrucelli N, Daly MB, Pal T. BRCA1- and BRCA2-Associated Hereditary Breast and Ovarian Cancer. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, and Amemiya A, eds. GeneReviews((R)). Seattle, WA. 1993.
11. Rebbeck TR, Kauff ND, Domchek SM. Meta-analysis of risk reduction estimates associated with risk-reducing salpingo-oophorectomy in BRCA1 or BRCA2 mutation carriers. J Natl Cancer Inst. 2009; 101:80–87. https://doi.org/10.1093/jnci/djn442. [PubMed].
12. Kotsopoulos J, Gronwald J, Karlan B, Rosen B, Huzarski T, Moller P, Lynch HT, Singer CF, Senter L, Neuhausen SL, Tung N, Eisen A, Foulkes WD, et al. Age-specific ovarian cancer risks among women with a BRCA1 or BRCA2 mutation. Gynecol Oncol. 2018; 150:85–91. https://doi.org/10.1016/j.ygyno.2018.05.011. [PubMed].
13. Faubion SS, Kuhle CL, Shuster LT, Rocca WA. Long-term health consequences of premature or early menopause and considerations for management. Climacteric. 2015; 18:483–491. https://doi.org/10.3109/13697137.2015.1020484. [PubMed].
14. Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol Rev. 2016; 96:365–408. https://doi.org/10.1152/physrev.00014.2015. [PubMed].
15. Silvagno F, Poma CB, Realmuto C, Ravarino N, Ramella A, Santoro N, D’Amelio P, Fuso L, Pescarmona G, Zola P. Analysis of vitamin D receptor expression and clinical correlations in patients with ovarian cancer. Gynecol Oncol. 2010; 119:121–124. https://doi.org/10.1016/j.ygyno.2010.06.008. [PubMed].
16. Uberti F, Morsanuto V, Lattuada D, Colciaghi B, Cochis A, Bulfoni A, Colombo P, Bolis G, Molinari C. Protective effects of vitamin D3 on fimbrial cells exposed to catalytic iron damage. J Ovarian Res. 2016; 9:34. https://doi.org/10.1186/s13048-016-0243-x. [PubMed].
17. Zhang S, Dolgalev I, Zhang T, Ran H, Levine DA, Neel BG. Both fallopian tube and ovarian surface epithelium are cells-of-origin for high-grade serous ovarian carcinoma. Nat Commun. 2019; 10:5367. https://doi.org/10.1038/s41467-019-13116-2. [PubMed].
18. Krishnan AV, Feldman D. Mechanisms of the anti-cancer and anti-inflammatory actions of vitamin D. Annu Rev Pharmacol Toxicol. 2011; 51:311–336. https://doi.org/10.1146/annurev-pharmtox-010510-100611. [PubMed].
19. Yin L, Grandi N, Raum E, Haug U, Arndt V, Brenner H. Meta-analysis: Circulating vitamin D and ovarian cancer risk. Gynecol Oncol. 2011; 121:369–375. https://doi.org/10.1016/j.ygyno.2011.01.023. [PubMed].
20. Yin L, Ordonez-Mena JM, Chen T, Schottker B, Arndt V, Brenner H. Circulating 25-hydroxyvitamin D serum concentration and total cancer incidence and mortality: a systematic review and meta-analysis. Prev Med. 2013; 57:753–764. https://doi.org/10.1016/j.ypmed.2013.08.026. [PubMed].
21. Ong JS, Cuellar-Partida G, Lu Y. Association of vitamin D levels and risk of ovarian cancer: a Mendelian randomization study. Int J Epidemiol. 2016; 45:1619–1630. https://doi.org/10.1093/ije/dyw207. [PubMed].
22. Heublein S, Mayr D, Meindl A, Kircher A, Jeschke U, Ditsch N. Vitamin D receptor, Retinoid X receptor and peroxisome proliferator-activated receptor gamma are overexpressed in BRCA1 mutated breast cancer and predict prognosis. J Exp Clin Cancer Res. 2017; 36:57. https://doi.org/10.1186/s13046-017-0517-1. [PubMed].
23. Berger U, McClelland RA, Wilson P, Greene GL, Haussler MR, Pike JW, Colston K, Easton D, Coombes RC. Immunocytochemical determination of estrogen receptor, progesterone receptor, and 1,25-dihydroxyvitamin D3 receptor in breast cancer and relationship to prognosis. Cancer Res. 1991; 51:239–244. [PubMed].
24. Dawson-Hughes B, Heaney RP, Holick MF, Lips P, Meunier PJ, Vieth R. Estimates of optimal vitamin D status. Osteoporos Int. 2005; 16:713–716. https://doi.org/10.1007/s00198-005-1867-7. [PubMed].
25. Kuriacose R, Olive KE. Vitamin D insufficiency/deficiency management. South Med J. 2014; 107:66–70. https://doi.org/10.1097/SMJ.0000000000000051. [PubMed].
26. Zella LA, Kim S, Shevde NK, Pike JW. Enhancers located in the vitamin D receptor gene mediate transcriptional autoregulation by 1,25-dihydroxyvitamin D3. J Steroid Biochem Mol Biol. 2007; 103:435–439. https://doi.org/10.1016/j.jsbmb.2006.12.019. [PubMed].
27. Xu J, Lawson MS, Xu F, Du Y, Tkachenko OY, Bishop CV, Pejovic-Nezhat L, Seifer DB, Hennebold JD. Vitamin D3 Regulates Follicular Development and Intrafollicular Vitamin D Biosynthesis and Signaling in the Primate Ovary. Front Physiol. 2018; 9:1600. https://doi.org/10.3389/fphys.2018.01600. [PubMed].
28. Li P, Li C, Zhao X, Zhang X, Nicosia SV, Bai W. p27(Kip1) stabilization and G(1) arrest by 1,25-dihydroxyvitamin D(3) in ovarian cancer cells mediated through down-regulation of cyclin E/cyclin-dependent kinase 2 and Skp1-Cullin-F-box protein/Skp2 ubiquitin ligase. J Biol Chem. 2004; 279:25260–25267. https://doi.org/10.1074/jbc.M311052200. [PubMed].
29. Jung YS, Kim HJ, Seo SK, Choi YS, Nam EJ, Kim S, Kim SW, Han HD, Kim JW, Kim YT. Anti-Proliferative and Apoptotic Activities of Mullerian Inhibiting Substance Combined with Calcitriol in Ovarian Cancer Cell Lines. Yonsei Med J. 2016; 57:33–40. https://doi.org/10.3349/ymj.2016.57.1.33. [PubMed].
30. Finch A, Beiner M, Lubinski J, Lynch HT, Moller P, Rosen B, Murphy J, Ghadirian P, Friedman E, Foulkes WD, Kim-Sing C, Wagner T, Tung N, et al. Salpingo-oophorectomy and the risk of ovarian, fallopian tube, and peritoneal cancers in women with a BRCA1 or BRCA2 Mutation. JAMA. 2006; 296:185–192. https://doi.org/10.1001/jama.296.2.185. [PubMed].
31. Perez-Lopez FR, Ceausu I, Depypere H, Kehoe S, Lambrinoudaki I, Mueck A, Senturk LM, Simoncini T, Stevenson JC, Stute P, Rees M. Interventions to reduce the risk of ovarian and fallopian tube cancer: A European Menopause and Andropause Society Postition Statement. Maturitas. 2017; 100:86–91. https://doi.org/10.1016/j.maturitas.2017.03.003. [PubMed].
32. Harmsen MG, Arts-de Jong M, Hoogerbrugge N, Maas AH, Prins JB, Bulten J, Teerenstra S, Adang EM, Piek JM, van Doorn HC, van Beurden M, Mourits MJ, Zweemer RP, et al. Early salpingectomy (TUbectomy) with delayed oophorectomy to improve quality of life as alternative for risk-reducing salpingo-oophorectomy in BRCA1/2 mutation carriers (TUBA study): a prospective non-randomised multicentre study. BMC Cancer. 2015; 15:593. https://doi.org/10.1186/s12885-015-1597-y. [PubMed].
33. Temkin SM, Bergstrom J, Samimi G, Minasian L. Ovarian Cancer Prevention in High-risk Women. Clin Obstet Gynecol. 2017; 60:738–757. https://doi.org/10.1097/GRF.0000000000000318. [PubMed].
34. Tschernichovsky R, Goodman A. Risk-Reducing Strategies for Ovarian Cancer in BRCA Mutation Carriers: A Balancing Act. Oncologist. 2017; 22:450–459. https://doi.org/10.1634/theoncologist.2016-0444. [PubMed].
35. Beral V, Doll R, Hermon C, Peto R, Reeves G, and Collaborative Group on Epidemiological Studies of Ovarian Cancer. Ovarian cancer and oral contraceptives: collaborative reanalysis of data from 45 epidemiological studies including 23,257 women with ovarian cancer and 87,303 controls. Lancet. 2008; 371:303–314. https://doi.org/10.1016/S0140-6736(08)60167-1. [PubMed].
36. Friedenson B. BRCA1 and BRCA2 pathways and the risk of cancers other than breast or ovarian. MedGenMed. 2005; 7:60. [PubMed].
37. Custodio A, Lopez-Farre AJ, Zamorano-Leon JJ, Mateos-Caceres PJ, Macaya C, Caldes T, de la Hoya M, Olivera E, Puente J, Diaz-Rubio E, Perez-Segura P. Changes in the expression of plasma proteins associated with thrombosis in BRCA1 mutation carriers. J Cancer Res Clin Oncol. 2012; 138:867–875. https://doi.org/10.1007/s00432-012-1161-y. [PubMed].
38. Pike JW, Meyer MB. Fundamentals of vitamin D hormone-regulated gene expression. J Steroid Biochem Mol Biol. 2014; 144:5–11. https://doi.org/10.1016/j.jsbmb.2013.11.004. [PubMed].
39. Xu J, Hennebold JD, Seifer DB. Direct vitamin D3 actions on rhesus macaque follicles in three-dimensional culture: assessment of follicle survival, growth, steroid, and antimullerian hormone production. Fertil Steril. 2016; 106:1815–20.e1. https://doi.org/10.1016/j.fertnstert.2016.08.037. [PubMed].
40. Meyer MB, Goetsch PD, Pike JW. Genome-wide analysis of the VDR/RXR cistrome in osteoblast cells provides new mechanistic insight into the actions of the vitamin D hormone. J Steroid Biochem Mol Biol. 2010; 121:136–141. https://doi.org/10.1016/j.jsbmb.2010.02.011. [PubMed].
41. Ramagopalan SV, Heger A, Berlanga AJ, Maugeri NJ, Lincoln MR, Burrell A, Handunnetthi L, Handel AE, Disanto G, Orton SM, Watson CT, Morahan JM, Giovannoni G, et al. A ChIP-seq defined genome-wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res. 2010; 20:1352–1360. https://doi.org/10.1101/gr.107920.110. [PubMed].
42. Minisola S, Ferrone F, Danese V, Cecchetti V, Pepe J, Cipriani C, Colangelo L. Controversies Surrounding Vitamin D: Focus on Supplementation and Cancer. Int J Environ Res Public Health. 2019; 16:189. https://doi.org/10.3390/ijerph16020189. [PubMed].
43. Ali M, Shahin SM, Sabri NA, Al-Hendy A, Yang Q. Hypovitaminosis D exacerbates the DNA damage load in human uterine fibroids, which is ameliorated by vitamin D3 treatment. Acta Pharmacol Sin. 2019; 40:957–970. https://doi.org/10.1038/s41401-018-0184-6. [PubMed].
44. Graziano S, Johnston R, Deng O, Zhang J, Gonzalo S. Vitamin D/vitamin D receptor axis regulates DNA repair during oncogene-induced senescence. Oncogene. 2016; 35:5362–5376. https://doi.org/10.1038/onc.2016.77. [PubMed].
45. Deng C, Ueda E, Chen KE, Bula C, Norman AW, Luben RA, Walker AM. Prolactin blocks nuclear translocation of VDR by regulating its interaction with BRCA1 in osteosarcoma cells. Mol Endocrinol. 2009; 23:226–236. https://doi.org/10.1210/me.2008-0075. [PubMed].
46. Campbell MJ, Gombart AF, Kwok SH, Park S, Koeffler HP. The anti-proliferative effects of 1alpha,25(OH)2D3 on breast and prostate cancer cells are associated with induction of BRCA1 gene expression. Oncogene. 2000; 19:5091–5097. https://doi.org/10.1038/sj.onc.1203888. [PubMed].
47. Lappe JM, Travers-Gustafson D, Davies KM, Recker RR, Heaney RP. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007; 85:1586–1591. https://doi.org/10.1093/ajcn/85.6.1586. [PubMed].
48. Lappe J, Watson P, Travers-Gustafson D, Recker R, Garland C, Gorham E, Baggerly K, McDonnell SL. Effect of Vitamin D and Calcium Supplementation on Cancer Incidence in Older Women: A Randomized Clinical Trial. JAMA. 2017; 317:1234–1243. https://doi.org/10.1001/jama.2017.2115. [PubMed].
49. Bolland MJ, Grey A, Gamble GD, Reid IR. Calcium and vitamin D supplements and health outcomes: a reanalysis of the Women’s Health Initiative (WHI) limited-access data set. Am J Clin Nutr. 2011; 94:1144–1149. https://doi.org/10.3945/ajcn.111.015032. [PubMed].
50. Wang K, Xu F, Campbell SP, Hart KD, Durham T, Maylie J, Xu J. Rapid actions of anti-Mullerian hormone in regulating synaptic transmission and long-term synaptic plasticity in the hippocampus. FASEB J. 2020; 34:706–719. https://doi.org/10.1096/fj.201902217R. [PubMed].

