Introduction
Immunotherapy in multiple myeloma
Immunotherapeutic agents are increasingly used for the treatment of both newly diagnosed and relapsed/refractory multiple myeloma (MM) patients [1–3]. This includes regimens containing CD38-targeting antibodies (e.g., daratumumab or isatuximab) for the treatment of newly diagnosed, transplant-eligible and transplant-ineligible, MM patients [4–6]. Furthermore, combination therapy with a CD38-targeting antibody or a SLAMF7-targeting antibody (elotuzumab) can also be used for patients with relapsed/refractory MM [7–11]. In addition, the FDA and EMA recently approved the B-cell maturation antigen (BCMA)-targeting antibody-drug conjugate, belantamab mafodotin, for the treatment of patients with advanced, heavily pretreated, MM [12]. Although these different immunotherapies have contributed to an improved outcome of MM patients, those with disease refractory to immunomodulatory drugs (IMiDs), proteasome inhibitors (PIs) and CD38-targeting antibodies have a very poor survival of less than one year [13]. The worst prognosis is observed in penta-refractory patients (refractory to 2 IMiDs, 2 PIs, and a CD38-targeting antibody) with a median overall survival of only 5.6 months. Importantly, T-cell redirecting therapies, such as chimeric antigen receptor (CAR) T-cells and bispecific antibodies (Figure 1), have significant clinical activity in these heavily pretreated patients.
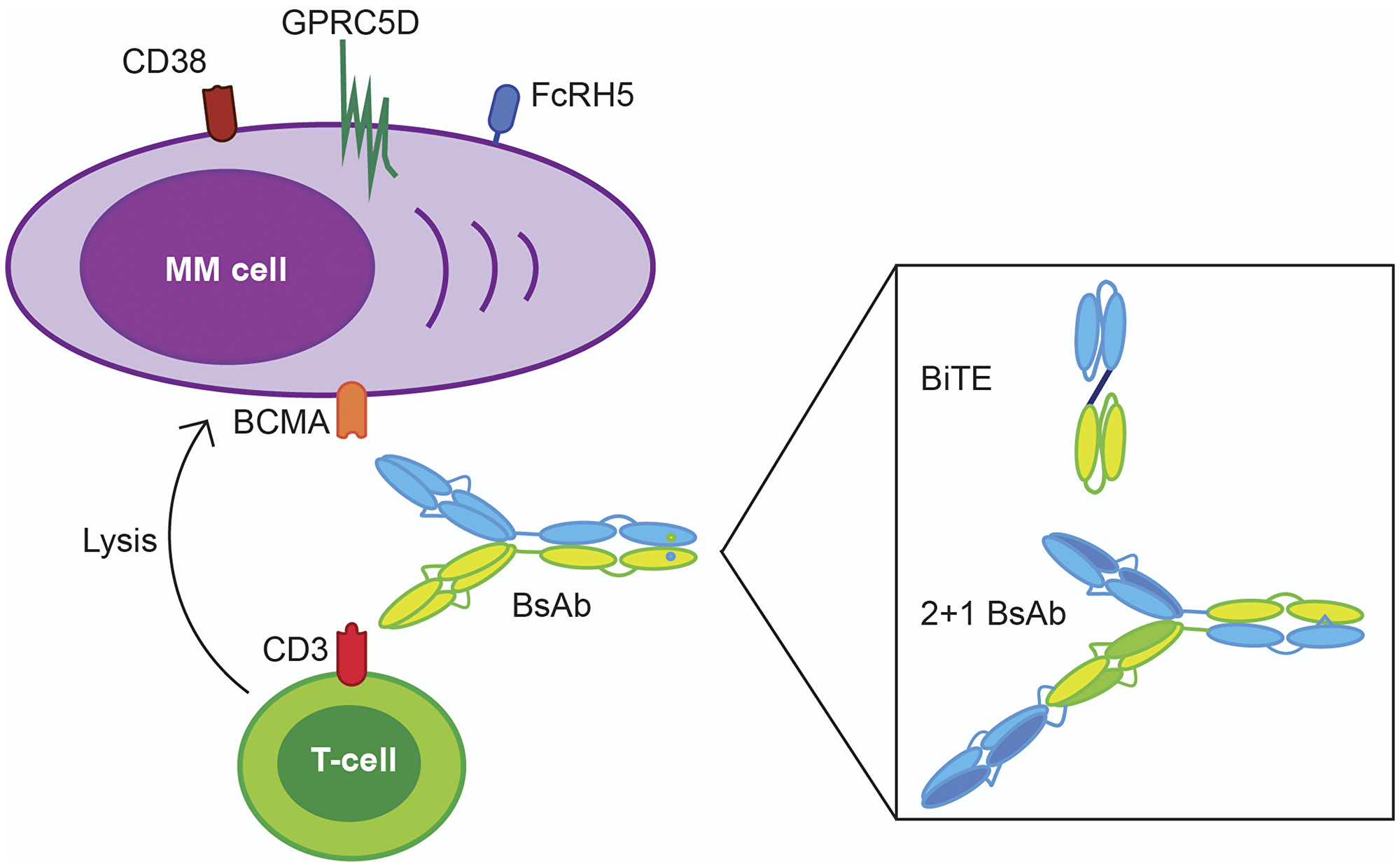
Figure 1: Examples of bispecific antibody designs used in clinical trials with multiple myeloma patients. Bispecific antibodies (BsAbs) bind with one arm to a CD3 antigen expressed on the T-cell surface and with another arm to a tumor-associated antigen (TAA) expressed on MM cells, such as B-cell maturation antigen (BCMA), CD38, G protein-coupled receptor of family C, group 5, member D (GPRC5D), and Fc receptor-homolog 5 (FcRH5). This coupling results in activation and degranualtion of T-cells and subsequent lysis of MM cells. Alternatives to the “classic” bivalent IgG-like BsAb design (e.g., teclistamab) are bispecific T-cell engagers (BiTEs, e.g., AMG420), which are comprised of two single-chain fragment variables and a peptide linker, and lack an Fc domain; and a 2+1 BsAb design (e.g., CC-93269), which is characterized by one bivalent arm that binds to both CD3 and a TAA and a monovalent arm that connects with a TAA.
Preclinical data on BCMA-targeting bispecific antibodies and BiTEs
We have recently provided the preclinical rationale for the use of the BCMAxCD3 bispecific antibody teclistamab (JNJ-7957) [14]. Teclistamab activates T-cells by connecting the CD3 antigen, present on T-cells, with BCMA, which is highly and selectively expressed on the surface of normal and clonal plasma cells. The redirected T-cells kill MM cells via various mechanisms including the release of perforins and granzymes. Teclistamab-mediated T-cell activation also resulted in a dose-dependent increase in several cytokines such as interferon-γ, tumor necrosis factor-α, interleukin-6, and interleukin-2. We also demonstrated that teclistamab effectively killed primary MM cells. Importantly, lysis was not only observed in samples obtained from newly diagnosed MM patients, but also in samples from heavily pretreated patients. This is a relevant finding since T-cell function in heavily pretreated patients is impaired due to the cumulative exposure to immunosuppressive anti-MM drugs (i.e., dexamethasone, proteasome inhibitors and alkylating drugs). In addition, patients with advanced disease have a more immunosuppressive tumor microenvironment with increased regulatory T-cell counts, as well as upregulation of inhibitory immune checkpoints, such as PD-L1. Because teclistamab-mediated MM cell lysis was heterogeneous, we assessed the impact of various tumor- and host-related factors on ex vivo response. The efficacy of teclistamab was not associated with cell surface expression of BCMA or cytogenetic risk status. However, samples with a high number of T-cells and thereby a higher T-cell to tumor cell ratio had superior teclistamab-mediated lysis, when compared to samples with low numbers of T-cells or low effector: target ratio. In agreement with our results, other bispecific antibodies or bispecific T-cell engagers (BiTE) targeting BCMA also showed high anti-MM activity in preclinical models [15–17].
Clinical studies with BCMA-targeting bispecific antibodies and BiTEs
Teclistamab is currently evaluated in a first-in-man study in heavily pretreated MM. The first results show high single agent activity and a favorable toxicity profile [18]. Teclistamab was administered via intravenous infusion to 78 patients with a median of 6 prior lines of therapy (80% of patients was triple-class refractory). Response was dose-dependent, with no responses observed in patients receiving teclistamab at a dose of 19.2 μg/kg or less, while the overall response rate was 30% in patients treated with 38.4–180 μg/kg. The best response was reported for the 12 patients treated at the dose of 270 μg/kg (highest dose reported up till now): at least partial response (PR) in 67%, at least very good partial response in 50%, and complete response (CR) in 25%. The most common adverse event related to teclistamab was cytokine release syndrome (CRS), which occurred in 56% of patients. Importantly, no grade ≥ 3 CRS was observed in these patients and most CRS events occurred during the step up dosing or first full dose [18]. Other toxicities included cytopenias and infections. The study is ongoing and currently exploring higher doses of teclistamab, as well as a subcutaneous administration, which may be the preferred route of administration because it is less invasive and faster, and will thereby reduce burden placed on the day care unit.
Preliminary data from other clinical trials also shows promising activity of BCMA-targeting bispecific antibodies and BiTEs [15]. One of these bispecific antibodies is CC-93269, which is characterized by bivalent binding to BCMA. The most recent update of the phase 1 study with this drug, reported results from the first 30 patients [19]. These patients had received a median of 5 prior lines of therapy (67% triple-class refractory). Similar to teclistamab, response to CC-93269 was dose-dependent with an overall response rate of 43% in all patients and 89% in the 9 patients treated with 10 mg CC-93269 including CR in 44%. CRS was observed in 77% of the patients, including one grade ≥ 3 event. Other adverse events included infections and cytopenias. This trial is ongoing to define the recommended phase 2 dose level.
AMG420 is the first-in-class BCMA-targeting BiTE. AMG420 has a short half-life because of its small size, and therefore needs to be administered via continuous intravenous infusion during 4 weeks of each 6-week cycle [20]. In the dose-escalation trial with 42 relapsed/refractory MM patients (median of 3.5 prior lines of therapy; 21% daratumumab-refractory), AMG420 induced a high response rate (≥ PR at the maximum-tolerated dose of 400 μg/day: 70%). CRS occurred in 38% of patients. Other adverse events included infections, bone marrow suppression, and polyneuropathy. Clinical development of AMG420 is stopped, because continuous intravenous infusion is logistically challenging. A half-life extended BCMA-targeting BiTE molecule, AMG701, is currently being evaluated in an ongoing phase 1 trial.
FUTURE DEVELOPMENTS
Combination treatment
Combination therapy is often used in cancer to prevent the outgrowth of resistant clones. Because of the high single agent activity and good tolerability profile of the BCMA-targeting bispecific antibodies, these agents are ideal components of a highly effective combination therapy. We showed that the direct combination of teclistamab with the CD38-targeting antibody daratumumab enhanced MM cell lysis in an additive fashion [14]. In addition, the activity of teclistamab was significantly enhanced in bone marrow samples obtained from patients with prior daratumumab treatment. Furthermore, teclistamab-mediated tumor cell lysis was superior when T-cells obtained from patients treated with daratumumab were used, compared to T-cells from daratumumab naïve MM patients [14]. This positive effect of daratumumab on the ability of teclistamab to kill MM cells, is probably related to the immune stimulating effects of daratumumab, which include the elimination of immune suppressor cells, induction of T-cell expansion, and increase in T-cell killing capacity [14, 21–23]. These data form the rationale for the ongoing phase 1 TRIMM study, which evaluates the efficacy of teclistamab plus daratumumab.
IMiDs, such as lenalidomide and pomalidomide, also have T-cell stimulating effects [24]. Indeed, pretreatment of effector cells with lenalidomide or pomalidomide enhanced AMG701-mediated lysis of MM cells [25, 26]. These data provide the rationale for clinical trials evaluating the combination of a bispecific antibody or BiTE with an IMiD. The cereblon E3 ligase modulator iberdomide (CC220) is a more potent immune stimulatory drug, compared to lenalidomide and pomalidomide [27], and is therefore also a promising potential combination partner for bispecific antibodies.
Earlier lines of therapy
Because of the high activity and favorable toxicity profile of bispecific antibodies in end-stage MM, these agents may also be beneficial for patients with early relapsed/refractory MM. Furthermore, newly diagnosed MM patients with high-risk cytogenetic abnormalities, who have a poor prognosis, may benefit from the incorporation of a BCMA-targeting bispecific antibody into their upfront treatment strategy. This is also supported by our finding that the ex vivo activity of teclistamab was not affected by presence of high-risk cytogenetic abnormalities. In addition, newly diagnosed patients, who do not achieve a deep response with a standard-of care treatment approach, may benefit from early access to a BCMA-targeting bispecific antibody with as ultimate goal the achievement of minimal residual disease-negativity.
Other bispecific and trispecific antibodies
Although the BCMA-targeting bispecific antibodies are most advanced in terms of clinical testing, several other T-cell redirecting antibodies are evaluated in early clinical trials. This includes bispecific or trispecific antibodies targeting GPRC5D, CD38, and FcRH5 [28–33].
Bispecific antibodies targeting other MM-associated antigens may be useful in patients with low baseline BCMA expression or heterogeneous BCMA expression. In these patients treatment with a BCMA-targeting bispecific antibody may lead to rapid outgrowth of BCMAlow clones. Furthermore, although a case report suggests that serial treatment with BCMA-targeting agents is feasible [34], we need more information on this important issue. If prior treatment with BCMA-targeted CAR T-cells or a BCMA-targeting antibody drug conjugate contributes to resistance to a BCMA-targeting bispecific antibody via reduced BCMA cell surface expression, then treatment with a bispecific antibody targeting another antigen may be more effective. In addition, a combination of bispecific antibodies, simultaneously targeting different antigens, may mitigate tumor antigen escape.
Conclusions
BCMA-targeting bispecific antibodies are highly active in extensively pretreated patients, including those who are refractory to all available drugs. These agents have a favorable safety profile, with CRS as most common adverse event, which can be easily managed with tocilizumab or steroids. A major advantage of bispecific antibodies or BiTEs over CAR T-cells is their direct “off-the-shelf” availability, while production of CAR T-cells takes several weeks. However, treatment with bispecific antibodies is typically continued until progression, while CAR T-cells are generally given as a single infusion and consequently patients will have a treatment-free interval, which is often preferred by patients and care givers. Finally, bispecific antibodies are promising combination partners with other immune stimulating drugs such as IMiDs and CD38-targeting antibodies. Clinical trials evaluating these combinations are ongoing.
CONFLICTS OF INTEREST
T.M. has received research support from Janssen Pharmaceuticals, Genmab, Takeda, Onkimmune, and Gadeta. S.Z. has received research funding from Celgene, Takeda and Janssen Pharmaceuticals; and serves in advisory boards for Janssen Pharmaceuticals, Sanofi, Celgene, Takeda and Oncopeptides. N.W.C.J.vdD. has received research support from Janssen Pharmaceuticals, AMGEN, Celgene, Novartis, and BMS; and serves in advisory boards for Janssen Pharmaceuticals, AMGEN, Celgene, BMS, Takeda, Roche, Novartis, Bayer, and Servier. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflicts of interest.
References
1. Verkleij CPM, Korst C, van de Donk N. Immunotherapy in multiple myeloma: when, where, and for who? Curr Opin Oncol. 2020; 32:664–671. https://doi.org/10.1097/CCO.0000000000000677. [PubMed].
2. van de Donk N, Richardson PG, Malavasi F. CD38 antibodies in multiple myeloma: back to the future. Blood. 2018; 131:13–29. https://doi.org/10.1182/blood-2017-06-740944. [PubMed].
3. van de Donk NW, Moreau P, Plesner T, Palumbo A, Gay F, Laubach JP, Malavasi F, Avet-Loiseau H, Mateos MV, Sonneveld P, Lokhorst HM, Richardson PG. Clinical efficacy and management of monoclonal antibodies targeting CD38 and SLAMF7 in multiple myeloma. Blood. 2016; 127:681–695. https://doi.org/10.1182/blood-2015-10-646810. [PubMed].
4. Moreau P, Attal M, Hulin C, Arnulf B, Belhadj K, Benboubker L, Bene MC, Broijl A, Caillon H, Caillot D, Corre J, Delforge M, Dejoie T, et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): a randomised, open-label, phase 3 study. Lancet. 2019; 394:29–38. https://doi.org/10.1016/S0140-6736(19)31240-1. [PubMed].
5. Facon T, Kumar S, Plesner T, Orlowski RZ, Moreau P, Bahlis N, Basu S, Nahi H, Hulin C, Quach H, Goldschmidt H, O’Dwyer M, Perrot A, et al, and MAIA Trial Investigators. Daratumumab plus Lenalidomide and Dexamethasone for Untreated Myeloma. N Engl J Med. 2019; 380:2104–2115. https://doi.org/10.1056/NEJMoa1817249. [PubMed].
6. Mateos MV, Dimopoulos MA, Cavo M, Suzuki K, Jakubowiak A, Knop S, Doyen C, Lucio P, Nagy Z, Kaplan P, Pour L, Cook M, Grosicki S, et al, and ALCYONE Trial Investigators. Daratumumab plus Bortezomib, Melphalan, and Prednisone for Untreated Myeloma. N Engl J Med. 2018; 378:518–528. https://doi.org/10.1056/NEJMoa1714678. [PubMed].
7. Dimopoulos MA, Lonial S, Betts KA, Chen C, Zichlin ML, Brun A, Signorovitch JE, Makenbaeva D, Mekan S, Sy O, Weisel K, Richardson PG. Elotuzumab plus lenalidomide and dexamethasone in relapsed/refractory multiple myeloma: Extended 4-year follow-up and analysis of relative progression-free survival from the randomized ELOQUENT-2 trial. Cancer. 2018; 124:4032–4043. https://doi.org/10.1002/cncr.31680. [PubMed].
8. Palumbo A, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, Spicka I, Hungria V, Munder M, Mateos MV, Mark TM, Qi M, Schecter J, et al, and CASTOR Investigators. Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2016; 375:754–766. https://doi.org/10.1056/NEJMoa1606038. [PubMed].
9. Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ, Rabin N, Orlowski RZ, Komarnicki M, Suzuki K, Plesner T, Yoon SS, Ben Yehuda D, et al, and POLLUX Investigators. Daratumumab, Lenalidomide, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2016; 375:1319–1331. https://doi.org/10.1056/NEJMoa1607751. [PubMed].
10. Attal M, Richardson PG, Rajkumar SV, San-Miguel J, Beksac M, Spicka I, Leleu X, Schjesvold F, Moreau P, Dimopoulos MA, Huang JS, Minarik J, Cavo M, et al, and ICARIA-MM study group. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, phase 3 study. Lancet. 2019; 394:2096–2107. https://doi.org/10.1016/S0140-6736(19)32556-5. [PubMed].
11. Dimopoulos MA, Dytfeld D, Grosicki S, Moreau P, Takezako N, Hori M, Leleu X, LeBlanc R, Suzuki K, Raab MS, Richardson PG, Popa McKiver M, Jou YM, et al. Elotuzumab plus Pomalidomide and Dexamethasone for Multiple Myeloma. N Engl J Med. 2018; 379:1811–1822. https://doi.org/10.1056/NEJMoa1805762. [PubMed].
12. Lonial S, Lee HC, Badros A, Trudel S, Nooka AK, Chari A, Abdallah AO, Callander N, Lendvai N, Sborov D, Suvannasankha A, Weisel K, Karlin L, et al. Belantamab mafodotin for relapsed or refractory multiple myeloma (DREAMM-2): a two-arm, randomised, open-label, phase 2 study. Lancet Oncol. 2020; 21:207–221. https://doi.org/10.1016/S1470-2045(19)30788-0. [PubMed].
13. Gandhi UH, Cornell RF, Lakshman A, Gahvari ZJ, McGehee E, Jagosky MH, Gupta R, Varnado W, Fiala MA, Chhabra S, Malek E, Mansour J, Paul B, et al. Outcomes of patients with multiple myeloma refractory to CD38-targeted monoclonal antibody therapy. Leukemia. 2019; 33:2266–2275. https://doi.org/10.1038/s41375-019-0435-7. [PubMed].
14. Frerichs KA, Broekmans MEC, Marin Soto JA, van Kessel B, Heymans MW, Holthof LC, Verkleij CPM, Boominathan R, Vaidya B, Sendecki J, Axel A, Gaudet F, Pillarisetti K, et al. Preclinical Activity of JNJ-7957, a Novel BCMA×CD3 Bispecific Antibody for the Treatment of Multiple Myeloma, Is Potentiated by Daratumumab. Clin Cancer Res. 2020; 26:2203–2215. https://doi.org/10.1158/1078-0432.CCR-19-2299. [PubMed].
15. Seckinger A, Delgado JA, Moser S, Moreno L, Neuber B, Grab A, Lipp S, Merino J, Prosper F, Emde M, Delon C, Latzko M, Gianotti R, et al. Target Expression, Generation, Preclinical Activity, and Pharmacokinetics of the BCMA-T Cell Bispecific Antibody EM801 for Multiple Myeloma Treatment. Cancer Cell. 2017; 31:396–410. https://doi.org/10.1016/j.ccell.2017.02.002. [PubMed].
16. Ramadoss NS, Schulman AD, Choi SH, Rodgers DT, Kazane SA, Kim CH, Lawson BR, Young TS. An anti-B cell maturation antigen bispecific antibody for multiple myeloma. J Am Chem Soc. 2015; 137:5288–5291. https://doi.org/10.1021/jacs.5b01876. [PubMed].
17. Hipp S, Tai YT, Blanset D, Deegen P, Wahl J, Thomas O, Rattel B, Adam PJ, Anderson KC, Friedrich M. A novel BCMA/CD3 bispecific T-cell engager for the treatment of multiple myeloma induces selective lysis in vitro and in vivo. Leukemia. 2017; 31:1743–1751. https://doi.org/10.1038/leu.2016.388. [PubMed].
18. Mateos MV, Usmani S, Nahi H, Krishnan A, San-Miguel J, Oriol A, Rosinol L, Chari A, Adams H 3rd, Girgis S, Wang Lin SX, Stephenson T, Kemmerer K, et al. A phase 1 study of teclistamab, a humanized B-cell maturation antigen (BCMA) X CD3 bispecific antibody, for the treatment of relapsed and/or refractory multipel myeloma (RRMM). EHA Library. 2020; 295026:S206.
19. Costa L, Wong S, Bermudez A, de la Rubia J, Mateos MV, Ocio E, Rodriguez Otero P, San Miguel J, Li S, Sarmiento R, Lardelli P, Gaudy A, Boss I, et al. Interim results from the first phase 1 clinical study of the B-cell maturation antigen (BCMA) 2+1 T-cell engager (TCE) CC-93269 in patients (pts) with relapsed/refractory multiple myeloma (RRMM). EHA Library. 2020; 295025:S205.
20. Topp MS, Duell J, Zugmaier G, Attal M, Moreau P, Langer C, Kronke J, Facon T, Salnikov AV, Lesley R, Beutner K, Kalabus J, Rasmussen E, et al. Anti-B-Cell Maturation Antigen BiTE Molecule AMG 420 Induces Responses in Multiple Myeloma. J Clin Oncol. 2020; 38:775–783. https://doi.org/10.1200/JCO.19.02657. [PubMed].
21. Krejcik J, Casneuf T, Nijhof IS, Verbist B, Bald J, Plesner T, Syed K, Liu K, van de Donk NW, Weiss BM, Ahmadi T, Lokhorst HM, Mutis T, Sasser AK. Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion, and skews T-cell repertoire in multiple myeloma. Blood. 2016; 128:384–394. https://doi.org/10.1182/blood-2015-12-687749. [PubMed].
22. Casneuf T, Adams HC 3rd, van de Donk N, Abraham Y, Bald J, Vanhoof G, Van der Borght K, Smets T, Foulk B, Nielsen KC, Rusbuldt J, Axel A, Lysaght A, et al. Deep immune profiling of patients treated with lenalidomide and dexamethasone with or without daratumumab. Leukemia. 2020. https://doi.org/10.1038/s41375-020-0855-4. [PubMed].
23. Adams HC 3rd, Stevenaert F, Krejcik J, Van der Borght K, Smets T, Bald J, Abraham Y, Ceulemans H, Chiu C, Vanhoof G, Usmani SZ, Plesner T, Lonial S, et al. High-Parameter Mass Cytometry Evaluation of Relapsed/Refractory Multiple Myeloma Patients Treated with Daratumumab Demonstrates Immune Modulation as a Novel Mechanism of Action. Cytometry A. 2019; 95:279–289. https://doi.org/10.1002/cyto.a.23693. [PubMed].
24. van de Donk NW, Görgün G, Groen RW, Jakubikova J, Mitsiades CS, Hideshima T, Laubach J, Nijhof IS, Raymakers RA, Lokhorst HM, Richardson PG, Anderson KC. Lenalidomide for the treatment of relapsed and refractory multiple myeloma. Cancer Manag Res. 2012; 4:253–268. https://doi.org/10.2147/CMAR.S27087. [PubMed].
25. Cho SF, Lin L, Xing L, Liu J, Yu T, Wen K, Hsieh P, Munshi N, Anderson K, Tai YT. Anti-BCMA BiTE® AMG 701 Potently Induces Specific T Cell Lysis of Human Multiple Myeloma (MM) Cells and Immunomodulation in the Bone Marrow Microenvironment. Blood. 2018; 132:592. https://doi.org/10.1182/blood-2018-99-118425.
26. Cho SF, Lin L, Xing L, Li Y, Wen K, Yu T, Hsieh PA, Munshi N, Wahl J, Matthes K, Friedrich M, Arvedson T, Anderson KC, Tai YT. The immunomodulatory drugs lenalidomide and pomalidomide enhance the potency of AMG 701 in multiple myeloma preclinical models. Blood Adv. 2020; 4:4195–4207. https://doi.org/10.1182/bloodadvances.2020002524. [PubMed].
27. Lonial S, Amatangelo M, Popat R, Minnema MC, Zonder JA, Larsen J, Oriol Rocafiguera A, Campagnaro EL, Rodriguez Otero P, Badros AZ, Siegel DS, Jagannath S, Bringhen S, et al. Translational and Clinical Evidence of a Differentiated Profile for the Novel CELMoD, Iberdomide (CC-220). Blood. 2019; 134:3119. https://doi.org/10.1182/blood-2019-124298.
28. Verkleij C, Broekmans M, van Duin M, Frerichs KA, Kuiper R, Kaiser M, Morgan G, Axel A, Chiu C, Sonneveld P, Zweegman S, Adams H 3rd, Mutis T, van de Donk N. Preclinical evaluation of the new gprc5dxcd3 (jnj-7564) bispecific antibody for the treatment of multiple myeloma. EHA Library. 2019; 266356:PF556.
29. Pillarisetti K, Edavettal S, Mendonça M, Li Y, Tornetta M, Babich A, Majewski N, Husovsky M, Reeves D, Walsh E, Chin D, Luistro L, Joseph J, et al. A T-cell-redirecting bispecific G-protein-coupled receptor class 5 member D x CD3 antibody to treat multiple myeloma. Blood. 2020; 135:1232–1243. https://doi.org/10.1182/blood.2019003342. [PubMed].
30. Li J, Stagg NJ, Johnston J, Harris MJ, Menzies SA, DiCara D, Clark V, Hristopoulos M, Cook R, Slaga D, Nakamura R, McCarty L, Sukumaran S, et al. Membrane-Proximal Epitope Facilitates Efficient T Cell Synapse Formation by Anti-FcRH5/CD3 and Is a Requirement for Myeloma Cell Killing. Cancer Cell. 2017; 31:383–395. https://doi.org/10.1016/j.ccell.2017.02.001. [PubMed].
31. Zuch de Zafra CL, Fajardo F, Zhong W, Bernett MJ, Muchhal US, Moore GL, Stevens J, Case R, Pearson JT, Liu S, McElroy PL, Canon J, Desjarlais JR, et al. Targeting Multiple Myeloma with AMG 424, a Novel Anti-CD38/CD3 Bispecific T-cell-recruiting Antibody Optimized for Cytotoxicity and Cytokine Release. Clin Cancer Res. 2019; 25:3921–3933. https://doi.org/10.1158/1078-0432.CCR-18-2752. [PubMed].
32. Fayon M, Martinez-Cingolani C, Abecassis A, Roders N, Nelson E, Choisy C, Talbot A, Bensussan A, Fermand JP, Arnulf B, Bories JC. Bi 38-3 is a novel CD38/CD3 bispecific T-cell engager with low toxicities for the treatment of multiple myeloma. Haematologica. 2020 Jul 16. https://doi.org/10.3324/haematol.2019.242453. [Epub ahead of print]. [PubMed]
33. Wu L, Seung E, Xu L, Rao E, Lord D, Wei R, Cortez-Retamozo V, Ospina B, Posternak V, Ulinski G, Piepenhagen P, Francesconi E, El-Murr N, et al. Trispecific antibodies enhance the therapeutic efficacy of tumor-directed T cells through T cell receptor co-stimulation. Nat Can. 2020; 1:86–98. https://doi.org/10.1038/s43018-019-0004-z.
34. Cohen AD, Garfall AL, Dogan A, Lacey SF, Martin C, Lendvai N, Vogl DT, Spear M, Lesokhin AM. Serial treatment of relapsed/refractory multiple myeloma with different BCMA-targeting therapies. Blood Adv. 2019; 3:2487–2490. https://doi.org/10.1182/bloodadvances.2019000466. [PubMed].

