Introduction
Neuroendocrine carcinomas (NECs) of the female genital tract are aggressive uncommon tumors that usually involve the uterine cervix and ovaries, though are very rarely seen in the endometrium [1]. A uterine cervix NEC is rare, accounting for only 1–6% of all cervical malignancies. According to the World Health Organization (WHO) classification, neuroendocrine tumors in the uterine cervix are categorized into 4 categories; typical carcinoid, atypical carcinoid, small cell neuroendocrine carcinoma (SCNEC), and large cell neuroendocrine carcinoma (LCNEC) [2]. The WHO defines SCNEC as an undifferentiated carcinoma with cellular and nuclear features that include small-sized cells, scant cytoplasm, hyperchromatic features, finely granular and molded nuclei, and inconspicuous nuclei. On the other hand, an LCNEC is defined as undifferentiated large cells that lack the cytologic and architectural features of small cell carcinoma, and show glandular or squamous differentiation. More simply, LCNEC is a malignant tumor composed of large cells that show neuroendocrine differentiation.
Cervical NEC has a higher frequency of lymphovascular invasion, lymphatic and distal metastasis, and recurrence as compared to other subtypes of uterine cervical malignancies, e. g., squamous cell carcinoma and adenocarcinoma [3, 4]. Clinical, Papanicolaou’s smear, and cervical biopsy findings are the primary screening and diagnostic tools for cervical malignancy cases. However, findings obtained in smear and cervical biopsy analyses are insensitive and inconclusive for diagnosis of cervical NEC in some patients, due to limited sample size or tumor heterogeneity [5]. Magnetic resonance imaging (MRI) is a well-established method for diagnosis and staging of uterine cervical tumors. Should MRI results be shown informative for correct diagnosis of NEC of the uterine cervix as well as accurate staging prior to performing treatment, the modality would be considered a useful clinical tool. To the best of our knowledge, 2 different reports have been presented that include discussion of MRI characteristics of uterine cervical SCNEC [6, 7], with a small number of patients in each (n = 7 and 26, respectively), while no known reports discussing MRI findings of cervical LCNEC are available. Therefore, features of uterine cervical NEC shown by MRI are not well recognized.
The aim of this study was to identify the distinct features and staging accuracy of uterine cervical NEC, including SCNEC and LCNEC, using MRI, as well as pathological correlations. Survival of affected patients was also evaluated.
Results
The clinicopathologic features of the present patients (n = 62) are summarized in Table 1. Their aged ranged from 26 to 82 years (median 39.5, mean 43.5 years), including 6 (9.7%) from 20–29 years old, 25 (40.3%) from 30–39, 15 (24.2%) from 40–49, 7 (11.3%) from 50–59, 5 (8.1%) from 60–69, 3 (4.8%) from 70–79, and 1 (1.6%) older than 79 years.
Table 1: Patient and tumor characteristics
| Character | N | % |
|---|---|---|
| Age at diagnosis (years) | ||
| Median (range) | 39.5 (26–82) | |
| NEC | ||
| pure type | 32 | 51.6 |
| SCNEC | 21 | 33.9 |
| LCNEC | 8 | 12.9 |
| Mixed SCNEC and LCNEC | 3 | 4.8 |
| mixed with other histotype | 30 | 48.4 |
| SCNEC with other histotype | 20 | 32.2 |
| LCNEC with other histotype | 10 | 16.1 |
| Positivity for synaptophysin | 57/62 | 91.9 |
| Positivity for chromogranin A | 48/60 | 80.0 |
| Positivity for CD56 | 42/53 | 79.2 |
| FIGO stage | ||
| IB1 | 32 | 51.6 |
| IB2 | 10 | 16.1 |
| IIA2 | 1 | 1.6 |
| IIB | 13 | 21.0 |
| IIIA | 1 | 1.6 |
| IVB | 5 | 8.1 |
| Treatment | ||
| Surgery | 8 | 12.9 |
| Surgery+chemotherapy | 31 | 50.0 |
| Surgery+chemotherapy+radiotherapy | 11 | 17.7 |
| NAC+Surgery | 3 | 4.8 |
| NAC+Surgery+chemotherapy | 7 | 11.3 |
| NAC+Surgery+chemotherapy+radiotherapy | 2 | 3.2 |
Presenting symptoms were known for 62 patients and included vaginal bleeding (n = 44), abnormal discharge (n = 8), abnormal pap smear results (n = 6), and inadvertent discovery in an examination for another disease (n = 4). Abnormal serum tumor marker levels for NSE were seen in 13 (27.1%) of 48, proGRP in 3 (17.6%) of 17, CA125 in 6 (13.3%) of 45, CA19-9 in 5 (11.9%) of 42, CEA in 4 (7.1%) of 56, SCC in 4 (7.1%) of 56, AFP in 0 (0%) of 5, and CYFRA in 0 (0%) of 2 patients. Cervical biopsy results revealed neuron-specific enolase (NEC) in 47 (75.8%) of the total 62 patients.
Pathological findings indicated pure NEC in 32 (51.6%), including pure SCNEC in 21, LCNEC in 8, and a mixture of SCNEC and LCNEC in 3 patients. Of the remaining 30 patients (48.4%), SCNEC combined with other pathologies was seen in 20, such as squamous cell carcinoma, adenocarcinoma, serous carcinoma, adenosquamous cell carcinoma, or carcinoma in situ, while LCNEC combined with other pathologies was noted in 10, including squamous cell carcinoma, adenocarcinoma, endometrioid adenocarcinoma, mucinous endocervical adenocarcinoma, or carcinoma in situ.
All tumors were positive for at least 1 neuroendocrine marker (synaptophysin, chromogranin A, CD56). Immunohistochemical findings of the specimens showed positive reactions for synaptophysin in 57 (91.9%) of 62, chromogranin A in 48 (80.0%) of 60, CD56 in 42 (79.2%) of 53, NSE in 5 (83.3%) of 6, p53 in 6 (75.0%) of 8, and SSRT2a in 2 (100%) of 2 patients examined for neuroendocrine markers. In addition, Ki-67 labelling index was identified in 16, ranging from 30% to 90% with a median of 80%. Also, the mitotic index was high for 10, in a range from 8 to 65 mitoses per 10 high power field (median, 17.5). Lymphovascular invasion was observed in 51 (82.3%) of 62 patients.
Early Federation of Gynecology and Obstetrics (FIGO) stage disease was noted in 56 patients (IB1, 32; IB2, 10; IIA2, 1; IIB, 13), while advanced FIGO stage disease was noted in 6 (IIIA, 1; IVB, 5). Of the 5 patients with stage IVB, 2 had liver metastasis, 1 had lung metastasis, 1 had liver and para-aortic nodal metastasis, and 1 had metastasis in the pancreas and breast.
MRI findings
MRI findings are summarized in Table 2. The largest tumor diameter was 36.3 ± 22.4 mm (range, 7–108 mm), with 19 (30.6%) patients showing a largest diameter greater than 4 cm and 43 (69.4%) that at 4 cm or less. Representative cases are presented in Figures 1, 2, and 3.
Table 2: MRI findings of cervical neuroendocrine carcinoma
| Character | N | % |
|---|---|---|
| Margin | ||
| Well-defined | 55 | 88.7 |
| Ill-defined | 7 | 11.3 |
| Shape | ||
| Round/Ovoid | 51 | 82.2 |
| Lobulated/Irregular | 11 | 17.7 |
| Internal appearance | ||
| Homogeneous | 50 | 80.6 |
| Heterogeneous | 12 | 19.4 |
| T1-weighted image | ||
| Slightly low | 10 | 16.1 |
| Iso | 48 | 77.4 |
| Slightly high | 4 | 6.5 |
| T2-weighted image | ||
| Slightly high | 50 | 80.6 |
| High | 12 | 19.4 |
| Diffusion weighted imaging | ||
| Intense high | 59 | 100 |
| Apparent diffusion coefficient map | ||
| Intense low | 59 | 100 |
| Dynamic contrast enhancement pattern | ||
| Gradual pattern | 30 | 88.2 |
| Washout/plateau pattern | 4 | 11.8 |
| The enhancement degree on the late phase | ||
| Mild | 37 | 66.1 |
| Moderate | 18 | 32.1 |
| Strong | 1 | 1.8 |
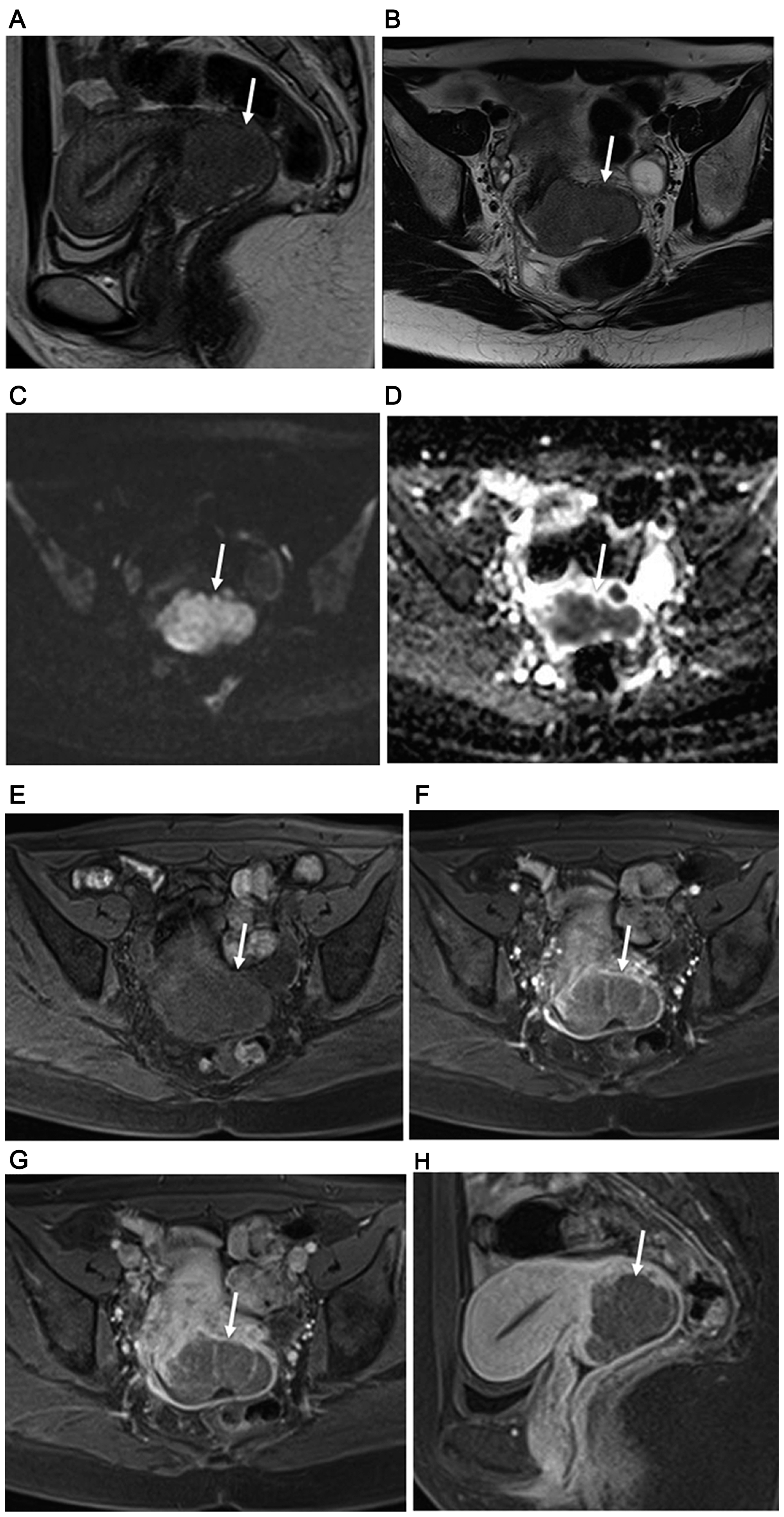
Figure 1: 34-year-old woman with FIGO stage IB2 uterine cervical large cell neuroendocrine carcinoma. (A) Sagittal and (B) axial T2-weighted imagings show homogeneous slightly hyperintense signal from large lobulated mass in uterine cervix (arrow). Subsequent surgery confirmed MRI staging of no invasion of vagina and parametrium. (C) Cervical mass with remarkably high signal intensity throughout tumor shown by axial DWI (arrow). (D) Obvious diffusion restriction throughout cervical tumor shown by axial ADC mapping (arrow). Results of dynamic contrast-enhanced axial fat-suppressed T1-weighted imaging found at (E) pre-contrast stage, and (F) 30 and (G) 120 seconds after administration of Gd-DTPA show a weak but gradual enhancement pattern (arrows). (H) Weak tumor enhancement (arrow) shown by sagittal late-phase enhanced fat-suppressed T1-weighted imaging (240 seconds after Gd-DTPA administration). Recurrence was noted 10.3 months after surgery and death at 20.5 months.
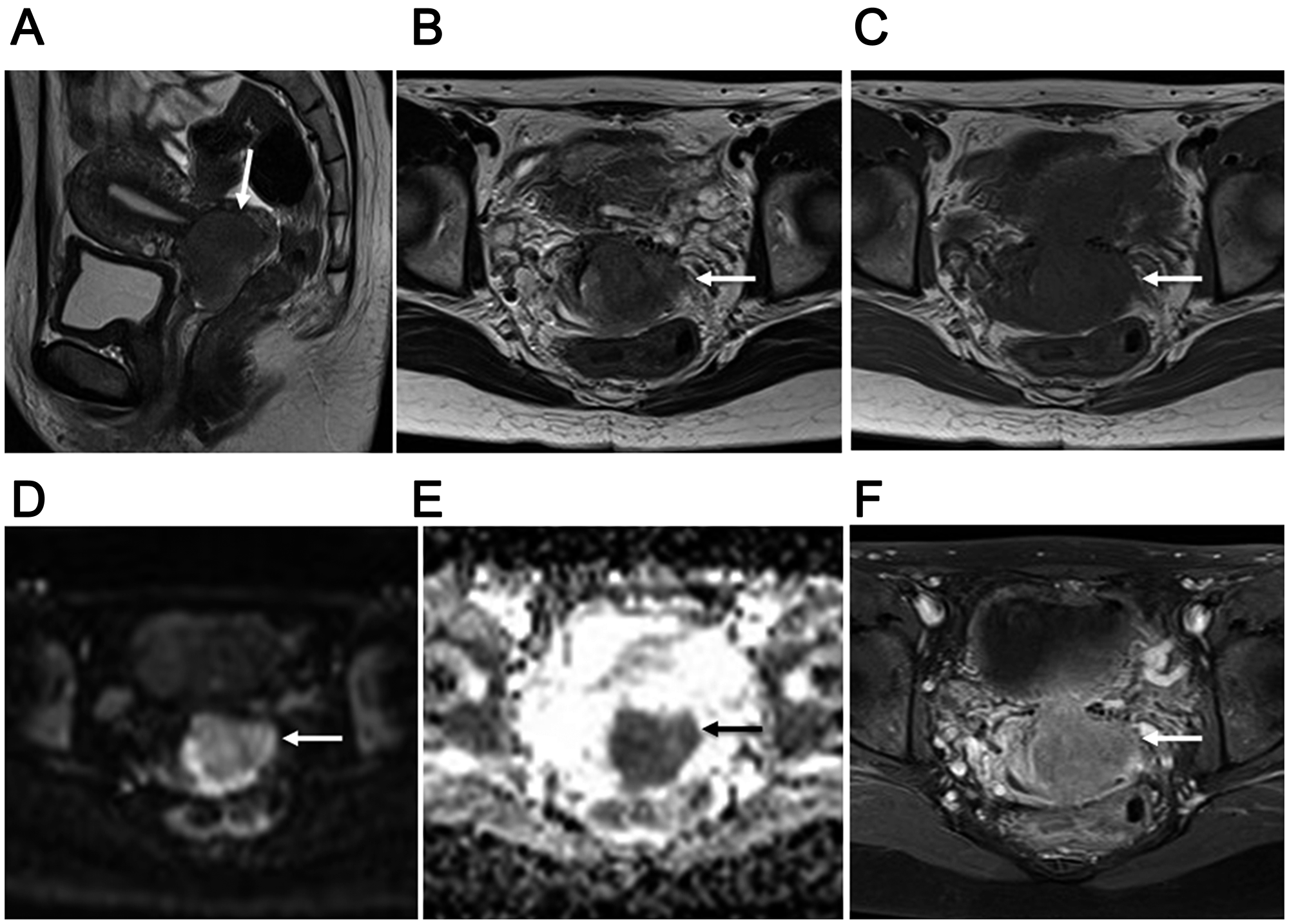
Figure 2: 34-year-old woman with FIGO stage IIB2 uterine cervical small cell neuroendocrine carcinoma. (A) Lobulated mass with homogeneous and slightly hyperintense signal in the uterine cervix (arrow) shown by sagittal T2-weighted imaging. (B) Ill-defined mass with invasion of left parametrium (arrow) in axial T2-weighted image. Subsequent surgery findings confirmed that MRI staging of parametrium invasion was correct. (C) Iso-intense mass invading left parametrium (arrow) shown by axial T1-weighted imaging. (D) Cervical mass with remarkably high signal intensity (arrow) shown by axial DWI. (E) Obvious diffusion restriction throughout tumor (arrow) shown by axial ADC map. (F) Weak tumor enhancement (arrow) shown by axial late-phase enhanced fat-suppressed T1-weighted imaging (240 seconds after Gd-DTPA administration). Recurrence was noted 10.8 months after surgery and death at 37.2 months.
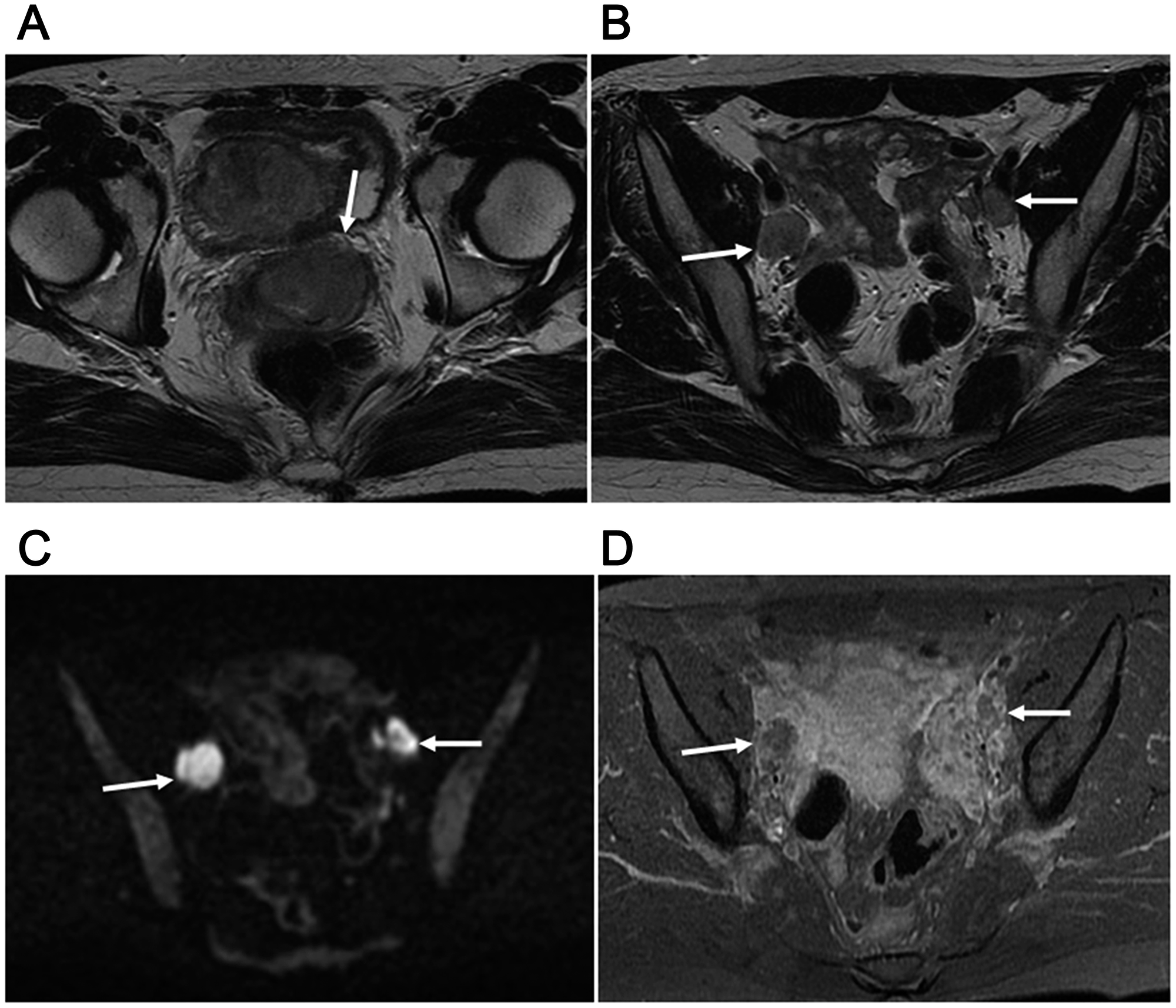
Figure 3: 30-year-old woman with T2bN1M0 (stage IIIB) and FIGO stage IIB2 uterine cervical small cell carcinoma mixed with squamous cell carcinoma. (A) Ill-defined mass in uterine cervix (arrow) with homogeneous slightly hyperintense signal shown by axial T2-weighted imaging. (B) Axial T2-weighted imaging showing 2 swollen lymph nodes in the bilateral obturator areas (arrows), both greater than 1 cm in the short diameter,, suggested to be nodal metastasis. (C) Two swollen lymph nodes with high signal intensity (arrows) shown by axial DWI. (D) Axial late-phase enhanced fat-suppressed T1-weighted imaging (240 seconds after Gd-DTPA) revealed weak enhancement of the 2 nodes (arrows). Surgery was performed after 2 courses of neoadjuvant chemotherapy, with pathological results showing nodal metastasis. No recurrence was seen at 113.8 months after initial neoadjuvant chemotherapy.
MRI and pathological staging
MRI diagnostic accuracy for tumor extension in a neighboring organ in the 50 patients without NAC is summarized in Table 3. Parametrial invasion was noted in 10 (20.0%) of 50 and correctly detected in 8 (80.0%) patients by MRI (Figure 2). Vaginal invasion was noted in 3 (6.0%) of 50 patients and correctly detected in 2 (66.7%) by MRINo invasion of the pelvic wall, urinary bladder, or rectum mucosa was observed in any of the present patients.
Table 3: MRI staging of cervical neuroendocrine carcinoma in 50 patients without NAC on a per patient basis
| Sensitivity | Specificity | PPV | NPV | Accuracy | |
|---|---|---|---|---|---|
| Invasion of parametrium | 80.0% (8/10) | 95.0% (38/40) | 80.0% (8/10) | 95.0% (38/40) | 92.0% (46/50) |
| 95% CI | 55.2–100 | 88.2–100 | 55.2–100 | 88.2–100 | 83.6–100 |
| Invasion of vagina | 66.7% (2/3) | 95.7% (45/47) | 50.0% (2/4) | 97.8% (45/46) | 94.0% (47/50) |
| 95% CI | 13.3–100 | 90.0–100 | 1.0–99.0 | 93.6–100 | 87.4–100 |
| *Metastatic pelvic lymph node | 38.5% (5/13) | 100% (35/35) | 100% (5/5) | 81.4% (35/43) | 83.3% (40/48) |
| 95% CI | 12.0–64.9 | 100 | 100 | 69.8–93.0 | 72.8–93.9 |
According to the most recently presented AJCC cancer staging [8], T stage in 50 patients without neoadjuvant chemotherapy (NAC) was classified as pT1b1 in 32, pT1b2 in 6, pT2a2 in 1, and pT2b in 11. Agreement was noted in 44 patients, with the overall accuracy of MRI local staging 88.0%. Details regarding agreement between MRI T staging and pathological staging are shown in Table 4.
Table 4: Correlation between MRI T staging and pathological staging in 50 patients without NAC
| Pathological staging | ||||||
|---|---|---|---|---|---|---|
| MRI staging | T1b1 | T1b2 | T2a1 | T2a2 | T2b | All |
| T1b1 | 30 | 0 | 0 | 0 | 1 | 31 |
| T1b2 | 0 | 5 | 0 | 1 | 0 | 6 |
| T2a1 | 1 | 0 | 0 | 0 | 1 | 2 |
| T2a2 | 0 | 0 | 0 | 0 | 0 | 0 |
| T2b | 1 | 1 | 0 | 0 | 9 | 11 |
| All | 32 | 6 | 0 | 1 | 11 | 50 |
Among the 48 patients who underwent a pelvic lymphadenectomy procedure without NAC, a total of 30 pelvic pathologically positive lymph node (LN) areas were found in 13 patients (27.1%), which consisted of the parametrial (n = 2), internal iliac (n = 8), external iliac (n = 7), common iliac (n = 3), and obturator fossa (n = 10) areas. In LN area-by-area analyses, the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy for detecting metastatic pelvic LN areas were 33.3% (10/30), 100% (450/450), 100%, (10/10) 95.7% (450/470), and 95.8% (460/480), respectively. As for patient-based sensitivity, specificity, PPV, NPV, and accuracy for detecting metastatic pelvic LN, those values were 38.5% (5/13), 100% (35/35), 100% (5/5) 81.4% (35/43), and 83.3% (40/48), respectively (Table 3) (Figure 3).
Survival analysis
Following surgery, 38 patients (31 without, 7 with NAC) received adjuvant chemotherapy and 13 (11 without, 2 with NAC) received chemoradiation therapy, while 11 patients (8 without, 3 with NAC) underwent no therapy. During a follow-up period of 45.6 months (4.3–151.0 months), 28 (45.2%) of 62 patients experienced recurrence or progression, including 15 (35.7%) of 42 classified as FIGO I, 7 (50.0%) of 14 as FIGO II, 1 (100%) of 1 as FIGO III, and 5 (100%) of 5 as FIGO IV. The sites of recurrence in those 28 patients were lung (n = 4), liver (n = 3), lung and liver (n = 3), lung, liver, and bone (n = 3), local (n = 2), LN (n = 2), local and LN (n = 2), liver and bone (n = 2), pancreas (n = 1), lung and bone (n = 1), LN and bone (n = 1), peritoneum and bone (n = 1), local, liver, and bone (n = 1), LN, lung, and bone (n = 1), and LN, lung, and liver (n = 1).
The median period from surgery to recurrence or progression for those 28 patients was 9.85 months (range 1.1 to 27.8 months). As for 3-year progression-free survival (PFS), those rates were 64.3% (27/42) for FIGO I, 50.0% (7/14) for FIGO II, 0% (0/1) for FIGO III, and 0% (0/5) for FIGO IV, while that was 54.8% (34/62) for all patients (Figure 4). Of those, 6 patients with advanced FIGO stage disease (III, IV) showed a shorter PFS than the 56 with early FIGO stage disease (I, II) and the difference was significant (p < 0.0001).
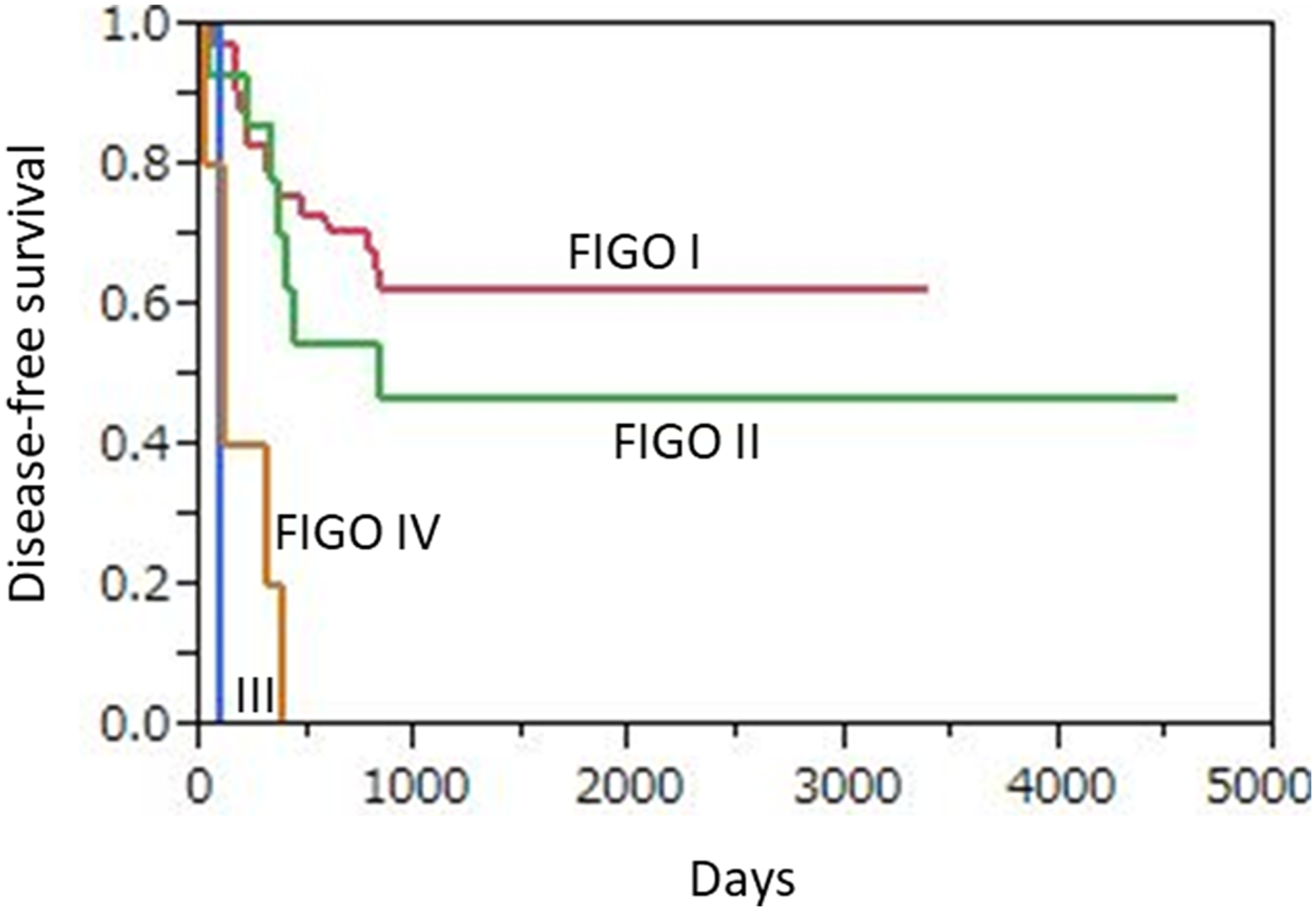
Figure 4: Kaplan-Meier disease-free survival curves for 62 uterine cervical neuroendocrine carcinoma patients according to FIGO stage. Advanced FIGO staging disease (III, IV) patients (n = 6) showed significantly shorter PFS as compared to those (n = 56) with early FIGO staging disease (I, II) (p < 0.0001).
During the follow-up period, 24 (38.7%) died of disease, including 12 (28.6%) of those classified as FIGO I, 6 (42.9%) as FIGO II, 1 (100%) as FIGO III, and 5 (100%) as FIGO IV. Median OS was 43.3 months for all patients, and 47.1, 46.6, 7.8, and 12.3 months when divided into FIGO I, II, III, and IV cases, respectively. The 3-year overall survival (OS) rates based on FIGO classification were 80.9% (34/42), 64.3% (9/14), 0% (0/1), and 0% (0/5), respectively, and 69.4% (43/62) for all patients (Figure 5). The 6 patients with advanced FIGO stage (III, IV) showed significantly worse OS than the 56 patients with early FIGO stage (I, II) disease (p < 0.0001).
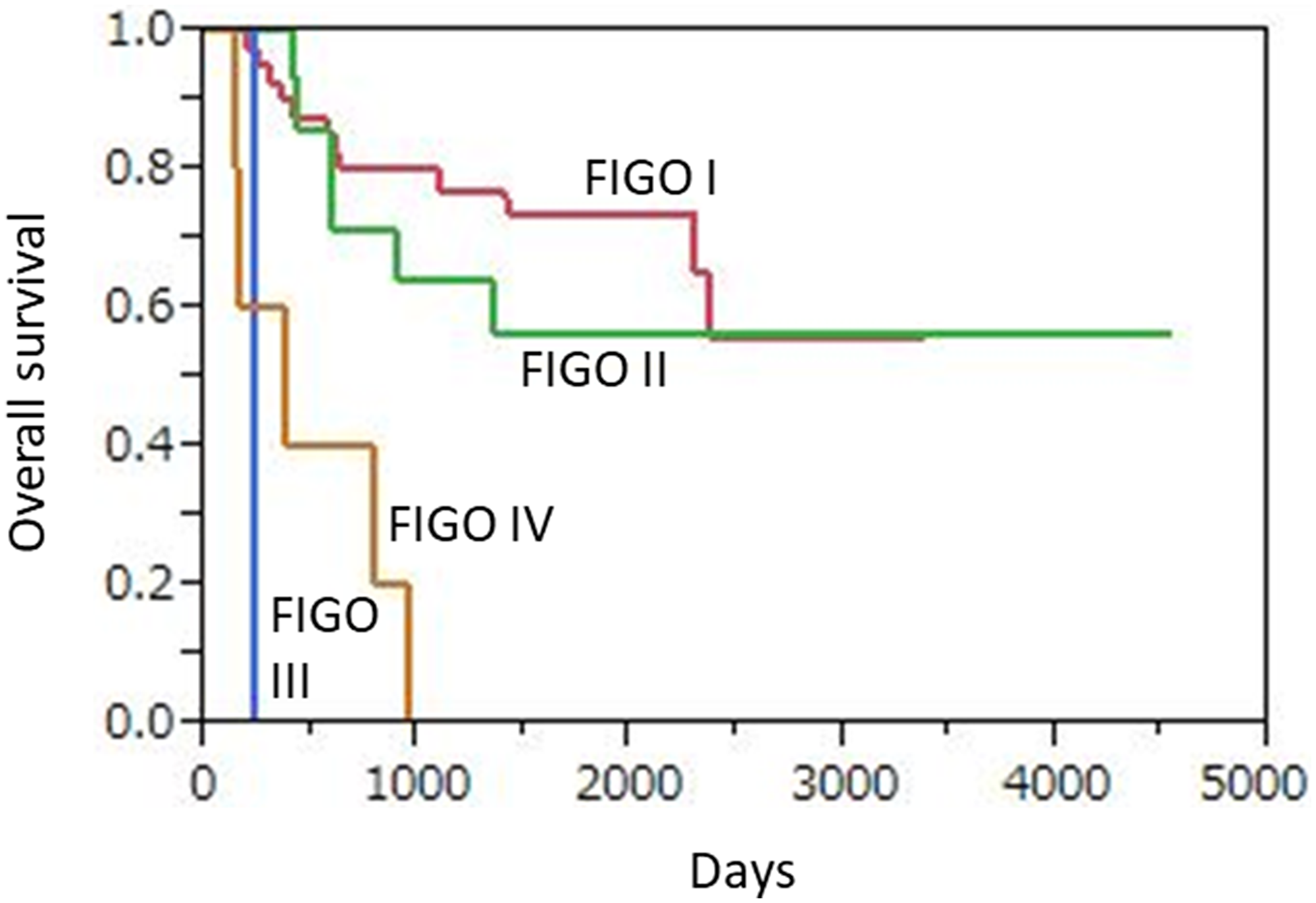
Figure 5: Kaplan-Meier overall survival curves for 62 uterine cervical neuroendocrine carcinoma patients according to FIGO stage. Advanced FIGO staging disease (III, IV) patients (n = 6) showed significantly worse OS as compare to those (n = 56) with early FIGO staging disease (I, II) (p < 0.0001).
Discussion and conclusions
This analysis of 62 patients with uterine cervical NEC proven by histological and surgery results revealed important findings related to MRI and patient prognosis. We found that likely suggestive features of uterine cervical NEC are a homogeneous lesion texture with an obvious restricted diffusion throughout the tumor, and consider that MRI is reliable for T staging of affected patients. Furthermore, it was confirmed that uterine cervical NEC is related to poor prognosis, especially in an advanced stage.
Two previous reports have presented MRI findings of patients with uterine cervical SCNEC [6, 7]. The present results were similar to those, as follows. First, advanced disease of the cervical NEC exhibited an ill-defined margin, irregular shape, infiltration into adjacent structures, and extensive lymphadenopathy in MRI results. These tumors also showed a significant tendency for homogeneous signal intensity with T2WI and homogeneous enhancement. Additionally, DWI showed abnormal high signal intensity with low ADC values throughout the tumor, reflecting a high level of cellularity of malignant cells. Duan et al. [7] reported that these MRI findings are nonspecific for indicating tumor malignancy, and that differentiation between squamous cell carcinoma and adenocarcinoma using MRI results is not easy in cases of cervical NEC. On the other hand, they also noted that lesion homogeneity and low ADC values shown by MRI are likely features suggestive of cervical NEC and the accuracy of MRI local T staging was very high (85.7%), very similar to the value (88.0%) noted in our study.
An NEC of the uterine cervix is aggressive and shows a highly malignant clinical behavior [3, 4]. Wang et al. [9] evaluated survival of 31 affected patients and reported a mean survival time of 32.2 months, with 2- and 5-year OS rates of 54.8% and 31.5%, respectively. Zivanovic et al. [10] evaluated survival of 17 patients with uterine cervical SCNEC and found a median OS of 14.6 months, along with 3-year relapse-free survival (RFS) and overall survival (OS) rates of 22% and 30%, respectively. In that study, the extent of disease was the only significant prognostic factor, and median OS was 31.2 months for patients with early stage (IA- IB2) and 6.4 months for those with advanced stage (IIB-IV) disease (p = 0.034). The same poor prognosis in advanced stage cases was identified in our series, as the 3-year OS rate for FIGO I and II was 76.8%, while that was 0% for FIGO III and IV cases. In the study by Wang et al. [9], the age of 31 patients with uterine cervical NEC was 42 ± 11.3 years, while that of the 25 reported by Duan et al. [7] was 46.3 ± 10.3 years (range 26–69 years). The mean age in the present cohort (43.5 years) was similar to those previous reports.
Although a clear treatment protocol for NEC of the uterine cervix has not been presented, it is recommended that patients be given multimodality therapy, including surgery, chemotherapy, and radiotherapy [4], with surgery generally thought to have the greatest impact. Zivanovic et al. [10] demonstrated that patients who received platinum-based chemotherapy had a 3-year RFS of 83% and OS as well of 83%, whereas those for patients who did not receive chemotherapy were 0% and 20%, respectively.
The present study results are limited by the retrospective design and variations in the MRI scan protocols used among the participating centers over the 13-year study period due to the rarity of this entity. The pathological diagnosis of 62 cervical NECs was made by each pathologist at each institution, not by one pathologist (central diagnosis).
In conclusion, NEC of the uterine cervix is rare and affected patients have a poor prognosis, especially advanced stage cases. A definitive diagnosis based on preoperative MRI results seems to be difficult. Nevertheless, the present as well as other previous findings indicate that a homogeneous lesion texture with an obvious restricted diffusion throughout the tumor are suggestive of uterine cervical NEC. Furthermore, we found pelvic MRI to provide reliable imaging findings for T staging in these patients.
Materials and Methods
Patient recruitment
This retrospective multicenter study of MRI findings of uterine cervical NEC included 24 institutions, and the appropriate review board at each gave approval to the protocol and waived the requirement for patient-informed consent. Sixty-two patients (average age at diagnosis 43.5 years, range 26–82 years) with uterine cervical NEC underwent pre-treatment pelvic MRI examinations between May 2006 and May 2018 at one of the participating institutions. For whole-body staging, 58 underwent chest/abdomen/pelvis computed tomography (CT) and 4 18F-fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT). Twelve (19.4%) of the 62 patients received neoadjuvant chemotherapy in 2 (n = 8), 3 (n = 3), or 4 (n = 1) courses. Of 50 patients without NAC, 2 underwent abdominal hysterectomy and bilateral salpingo-oophorectomy, 41 abdominal hysterectomy, bilateral salpingo-oophorectomy, and pelvic lymphadenectomy, and 7 abdominal hysterectomy, bilateral salpingo-oophorectomy, pelvic lymphadenectomy, and para-aortic lymphadenectomy procedures. As for the 12 patients with NAC, 1 underwent abdominal hysterectomy and bilateral salpingo-oophorectomy, 7 abdominal hysterectomy, bilateral salpingo-oophorectomy, and pelvic lymphadenectomy, and 4 abdominal hysterectomy, bilateral salpingo-oophorectomy, pelvic lymphadenectomy and para-aortic lymphadenectomy procedures. Conventional haematoxylin eosin and immunochemistry staining for various markers was applied to resected tumors and biopsy samples, including synaptophysin, chromogranin A, CD56, NSE, p53, SSRT2a, and Ki-67. Each tumor was staged according to the 2008 International FIGO staging system for carcinomas of the uterine cervix [11].
Pelvic MRI
Pelvic MRI examinations were performed using equipment available at the participating institutions during the course of this study, including a 1.5-T system (1.5T Gyroscan Intera NT, or Intera Achieva 1.5T nova dual; Philips Medical Systems, Best, The Netherlands, Optima MR450w, SIGNA HDxt 1.5T, or SIGNA EXCITE; GE Healthcare, Waukesha, WI, USA, MAGNETON Vision, MAGNETOM Avanto, or MAGNETOM ESSENZA; Siemens Medical Solutions, Erlangen, Germany, NT MRT200SP5; Canon Medical System, Ohtawara, Japan) in 36 patients or a 3-T system (Achieva 3.0T TX, Intera Achieva Quasar dual, or Ingenia CX; Philips Medical Systems, Best, The Netherlands, DISCOVERY MR750w or SIGNA EXCITE 3.0T-HD; GE Healthcare, Waukesha, WI, USA, MAGNETON Prisma, MAGNETON Skyra or MAGNETON Trio; Siemens Medical Solutions, Erlangen, Germany, Vantage Titan 3T; Cannon Medical System, Ohtawara, Japan) in 26 patients. For those examinations, a body coil was used for excitation and a pelvic phased-array coil for signal reception. Butyl scopolamine (Buscopan, Boehringer Ingelheim) was given intramuscularly to all patients immediately before the examination to reduce artifacts from bowel peristalsis, unless contraindicated. The MRI parameters varied depending on the institution. Unenhanced axial and sagittal fast-spin-echo T2WI was performed with a 4–5 mm slice thickness, and unenhanced T1WI results were acquired in the axial and sagittal planes with a spin-echo, and 4- to 5-mm slice thickness, In 59 patients, axial DWI was performed in 3 orthogonal directions using spin-echo-type single-shot echo planar imaging with a 4- to 5-mm slice thickness. Fifty-six patients underwent contrast-enhanced scanning. In 34 patients, following administration of 0.1 mmol/kg gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA) at a rate of 2.0–3.0 ml/second, followed by a saline flush (15 ml at 2.0–3.0 ml/second), multiphase dynamic images with fast-gradient-echo, fat-suppressed T1-weighted, axial, or sagittal sequences (2- to 3-mm slice thickness) were obtained. Delayed (4 to 6 minutes after Gd-DTPA administration) T1-weighted fat-suppressed axial and sagittal sequences were obtained sequentially, with parameters similar to those used before injection of Gd-DTPA. In the other 22 patients, following a single injection of Gd-DTPA (0.1 mmol/kg body weight), T1-weighted fat-suppressed axial and sagittal sequences in the late phase were obtained sequentially, using parameters similar to those used prior to injection of Gd-DTPA.
Imaging analysis
Pelvic MRI results were reviewed by 2 experienced radiologists with experience in gynecological MRI (K. K., 18 years; Y. K., 10 years) and blinded to patient information. Their decisions were made in a consensus manner. For expert review, MR images were displayed with a Digital Imaging and Communications in Medicine viewer (Osirix; Pixmeo, Geneva, Switzerland), and assessments of tumor location, size, margin (well-defined or ill-defined), shape (round/ovoid or lobular/irregular), signal intensity (compared with uterine myometrium), lesion texture (homogeneous or heterogeneous), contrast enhancement patterns, and degree of enhancement were made. Tumor size was measured as the greatest diameter in either the transverse or sagittal plane. Dynamic contrast enhancement patterns were categorized as washout/plateau or gradual. Time-intensity curves were categorized into progressive, plateau, washout, and indeterminate types. Progressive enhancement was defined as a gradual increase in signal throughout all phases of enhancement. Plateau enhancement was defined as an initial increase in signal followed by a plateau in which the MRI signal units remained unchanged within a range of a 5% difference. A washout curve was defined as a steep initial increase and peak in signal followed by a decrease of at least 5%–10% [12]. The degree of tumor enhancement in the late phase was compared with normal myometrium enhancement, then classified as mild, moderate, or strong.
The experienced readers also evaluated tumor extension into the parametrium, vagina, pelvic wall, and urinary bladder or rectum mucosa as well as pelvic lymph nodes (LNs), using methods detailed in previous reports [13, 14]. Lymphadenopathy location was defined as right or left parametrial, internal iliac, external iliac, common iliac, or obturator area. Lymphadenopathy was considered to be present based on size criteria (> 1 cm in short diameter) and/or signal intensity (with central necrosis).
Statistical analysis
Values are presented as the mean ± standard deviation (SD) and number (%). PFS was analyzed using Kaplan-Meier plots and a log-rank test, and defined as time from initial treatment to tumor recurrence or progression, determined by histologic or follow-up findings, including imaging results. OS was defined as time from initial treatment to date of death from the disease. Statistical analysis was performed using SAS, version 9.3 (SAS Institute Inc.). P values < 0.05 were considered to indicate significance.
Abbreviations
NEC: neuroendocrine carcinoma; NAC: neoadjuvant chemotherapy; WHO: World Health Organization; SCNEC: small cell neuroendocrine carcinoma; LCNEC: large cell neuroendocrine carcinoma; MRI: magnetic resonance imaging; NEC: neuron-specific enolase; FIGO: Federation of Gynecology and Obstetrics; T1WI: T1-weighted imaging; T2WI: T2-weighted imaging; ADC: apparent diffusion coefficient; DWI: diffusion weighted imaging; LN: lymph node; PPV: positive predictive value; NPV: negative predictive value; PFS: progression-free survival; OS: overall survival; RFS: relapse-free survival; SD: standard deviation.
ACKNOWLEDGMENTS
None.
Ethics statement
This retrospective study was approved by our institutional review board and the need for patient informed consent was waived.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
FUNDING
This work was supported by a Hyogo Science and Technology Association (1004) and a JSPS KAKENHI grant (number 19K08187).
References
1. Rouzbahman M, Clarke B. Neuroendocrine tumors of the gynecologic tract: select topics. Semin Diagn Pathol. 2013; 30:224–33. https://doi.org/10.1053/j.semdp.2013.06.007. [PubMed].
2. Kurman RJ, Carcanju ML, Herrington CS, Young R. World Health Organization classification of tumours of female reproductive organs. 4th ed. World Health Organization. Lyon: IARC Press; 2014.
3. Chan JK, Loizzi V, Burger RA, Rutgers J, Monk BJ. Prognostic factors in neuroendocrine small cell cervical carcinoma: a multivariate analysis. Cancer. 2003; 97:568–74. https://doi.org/10.1002/cncr.11086. [PubMed].
4. McCann GA, Boutsicaris CE, Preston MM, Backes FJ, Eisenhauer EL, Fowler JM, Cohn DE, Copeland LJ, Salani R, O’Malley DM. Neuroendocrine carcinoma of the uterine cervix: the role of multimodality therapy in early-stage disease. Gynecol Oncol. 2013; 129:135–9. https://doi.org/10.1016/j.ygyno.2013.01.014. [PubMed].
5. Wentzensen N, Walker JL, Gold MA, Smith KM, Zuna RE, Mathews C, Dunn ST, Zhang R, Moxley K, Bishop E, Tenney M, Nugent E, Graubard BI, et al. Multiple biopsies and detection of cervical cancer precursors at colposcopy. J Clin Oncol. 2015; 33:83–89. https://doi.org/10.1200/JCO.2014.55.9948. [PubMed].
6. Yang DH, Kim JK, Kim KW, Bae SJ, Kim KH, Cho KS. MRI of small cell carcinoma of the uterine cervix with pathologic correlation. AJR Am J Roentgenol. 2004; 182:1255–1258. https://doi.org/10.2214/ajr.182.5.1821255. [PubMed].
7. Duan X, Ban X, Zhang X, Hu H, Li G, Wang D, Wang CQ, Zhang F, Shen J. MR imaging features and staging of neuroendocrine carcinomas of the uterine cervix with pathological correlations. Eur Radiol. 2016; 26:4293–4302. https://doi.org/10.1007/s00330-016-4327-1. [PubMed].
8. Edge SB, Compton CC. The American joint committee on cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17:1471–1474. https://doi.org/10.1245/s10434-010-0985-4. [PubMed].
9. Wang KL, Yang YC, Wang TY, Chen JR, Chen TC, Chen HS, Su TH, Wang KG. Neuroendocrine carcinoma of the uterine cervix: A clinicopathologic retrospective study of 31 cases with prognostic implications. J Chemother. 2006; 18:209–216. https://doi.org/10.1179/joc.2006.18.2.209. [PubMed].
10. Zivanovic O, Leitao MM Jr, Park KJ, Zhao H, Diaz JP, Konner J, Alektiar K, Chi DS, Abu-Rustum NR, Aghajanian C. Small cell neuroendocrine carcinoma of the cervix: analysis of outcome, recurrence pattern and the impact of platinum-based combination chemotherapy. Gynecol Oncol. 2009; 112:590–593. https://doi.org/10.1016/j.ygyno.2008.11.010. [PubMed].
11. Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet. 2009; 105:103–104. https://doi.org/10.1016/j.ijgo.2009.02.012. [PubMed].
12. Garza A, Elsherif SB, Faria SC, Sagebiel T, Sun J, Ma J, Bhosale PR. Staging MRI of uterine malignant mixed Müllerian tumors versus endometrial carcinomas with emphasis on dynamic enhancement characteristics. Abdom Radiol (NY). 2020; 45:1141–1154. https://doi.org/10.1007/s00261-019-02096-y. [PubMed].
13. Freeman SJ, Aly AM, Kataoka MY, Addley HC, Reinhold C, Sala E. The revised FIGO staging system for uterine malignancies: implications for MR imaging. Radiographics. 2012; 32:1805–1827. https://doi.org/10.1148/rg.326125519. [PubMed].
14. Balleyguier C, Sala E, Da Cunha T, Bergman A, Brkljacic B, Danza F, Forstner R, Hamm B, Kubik-Huch R, Lopez C, Manfredi R, McHugo J, Oleaga L, et al. Staging of uterine cervical cancer with MRI: guidelines of the European Society of Urogenital Radiology. Eur Radiol. 2011; 21:1102–1110. https://doi.org/10.1007/s00330-010-1998-x. [PubMed].

