Introduction
The cancer stem cell (CSC) model was proposed over 30 years ago [1] and is a very important field of study in cancer research. The frequency of CSCs varies from 27% to 100% in highly tumorigenic cancers, such as hematopoietic and melanoma primary tumors, as well as in some cancer cell lines [2]. Although CSCs account for less than 1% of the total cells in solid tumors [3], they have several roles in tumor generation and progression, such as in the capacity for self-renewal, asymmetric cell division, slow division kinetics, increased capacity of invasion, metastasis, tumor formation, proliferation, resistance to conventional chemotherapy, and radiotherapy and immunotherapy [4–7]. For the small amount of these cells inside the tumor, however, several studies have been conducted on the identification of CSC markers [8, 9].
The most considered CSC markers have been aldehyde dehydrogenase (ALDH), and CD44+ [10]. In particular, ALDH was described in 2010 by Sullivan et al. as a cancer stem cell marker in a panel of 11 non-small-cell lung cancer (NSCLC) tumor samples, 45 NSCLC lines, and 7 small-cell lung cancer (SCLC) lines [11]. Additionally, this small population of CSCs was better identified using multiple markers [12]. In particular, Wang et al. in 2013 established a panel of lung cancer cell lines from primary tumors and characterized a small subpopulation as strongly positive for CD44 (CD44high), with the main population being weakly positive or negative for CD44. Co-expression of CD90 (CD90+) further narrowed down the putative stem cell population. This CD44 and CD90 positive subpopulation showed several CSC characteristics. In fact, the CD44high CD90+ subpopulation was considered a good candidate for a CSC marker [4].
In 2015, N. Zakaria et al. showed the putative lung CSC phenotypes of CD166+/CD44+ and CD166+/EPCAM+ with multipotent characteristics of stem cells in lung adenocarcinoma cells (A549 and H2170) [8].
The identification of multiple markers is due to the complexity of highlighting the entire cancer stem cell population. Hence, a triple-positive marker, EPCAM+/CD166+/CD44+, has recently been described in the human non-small cell lung cancer cell line [12].
Nevertheless, although ALDH is considered an intracellular enzyme and is the most used marker to identify CSCs in lung cancer [7, 11, 13], the scientific community has never correlated this intracellular marker with an epithelial marker, which may be very useful for targeting lung cancer stem cells. The aim of the present research was to compare the ALDHhigh cells with the double-positive CD44/EPCAM cells, in order to better define the superficial antigens of ALDHhigh cells. The use of superficial antigens may trigger new therapeutic approaches against CSCs in NSCLC.
Results
Patients’ and specimens’ characteristics
The major clinical and demographic characteristics of the 24 patients enrolled from December 2017 to January 2019 are reported in Table 1. The average age was 70.3 years (SD 9.3, range 52 to 84); 62.5% were males, and all were smokers. Patients harboring stage I (37.5%) and stage III (37.5%) NSCLC were more prevalent than those harboring stage II (25.0%), whereas adenocarcinoma (75.0%) was more frequent than squamous cell carcinoma (25.0%). The surgical tumor specimens’ average weight was 1.3 grams (SD 1.9, range 0.1 to 9.6), and the average cellular yield was 33.8 million cells per gram (SD 35.9, range 7 to 150).
Table 1: Descriptive characteristics of the patients and specimens included in the study
| Characteristics of patients | All patients (n = 24) | |
|---|---|---|
| Age (years) | mean ± SD | 70.3 ± 9.3 |
| median (range) | 70 (52; 84) | |
| Sex – M | n (%) | 15 (62.5%) |
| Smoker – Yes | n (%) | 24 (100.0%) |
| Adenocarcinoma | n (%) | 18 (75.0%) |
| Squamous cell carcinoma | n (%) | 6 (25.0%) |
| Stage - I | n (%) | 9 (37.5%) |
| Stage - II | n (%) | 6 (25.0%) |
| Stage - III | n (%) | 9 (37.5%) |
| Characteristics of specimens | All samples (n = 24) | |
| Weight (grams) | mean ± SD | 1.3 ± 1.9 |
| median (range) | 0.8 (0.1; 9.6) | |
| Cellular yield (million cells per gram) | mean ± SD | 33.8 ± 35.9 |
| median (range) | 19.7 (7.0; 150.0) | |
| Cytofluorimetric analysis | All samples (n = 23) | |
| ALDH+ (% on 7AAD- cells) | mean ± SD | 3.2 ± 3.4% |
| median (range) | 1.9% (0.4; 12.5%) | |
| CD44+/EPCAM+ (% on 7AAD- cells) | mean ± SD | 3.1 ± 2.5% |
| median (range) | 2.6% (0.1; 10.1%) | |
| CD44+/EPCAM– (% on 7AAD- cells) | mean ± SD | 11.7 ± 22.9% |
| median (range) | 2.5% (0.2; 86.0%) | |
| CD44–/EPCAM+ (% on 7AAD- cells) | mean ± SD | 18.5 ± 19.4% |
| median (range) | 12.5% (0.0; 64.0%) |
Cytofluorimetric analysis of ALDHhigh and CD44+/EPCAM+ in primary lung cancer cells
The putative CSCs were physically separated from the bulk parental tumor cells and recovered by fluorescence-activated cell sorting (FACS) according to the following gating strategy. Tumor cells were first identified based on their morphological parameters (forward scatter versus side scatter (FSC/SSC)), and ALDH activity was measured in the 7-AAD-negative cell subpopulation only. ALDHlow and ALDHhigh cells were selected and sorted. Results obtained in the main cytofluorimetric analysis are reported in Table 1. An ALDHhigh subpopulation was identified for all patients, and the average expression was 3.2% (SD 3.4%, range 0.4% to 12.5%, with 69.6% of samples above 1%). A CD44+/EPCAM+ subpopulation was also identified in all samples, exhibiting an average expression equal to 3.1% (SD 2.5%, range 0.1% to 10.1%, with 82.6% of samples above 1%), similar to that of ALDHhigh cells (Figure 1). We also identified CD44+/EPCAM– and CD44–/EPCAM+ subpopulations with average expressions equal to 11.7% and 18.5% (SD 22.9% and 19.4%), respectively (Table 1) (Figure 1). The present results did not relevantly change if considering only the subgroup of 18 patients harboring adenocarcinoma (Supplementary Table 1, Supplementary Figure 1).
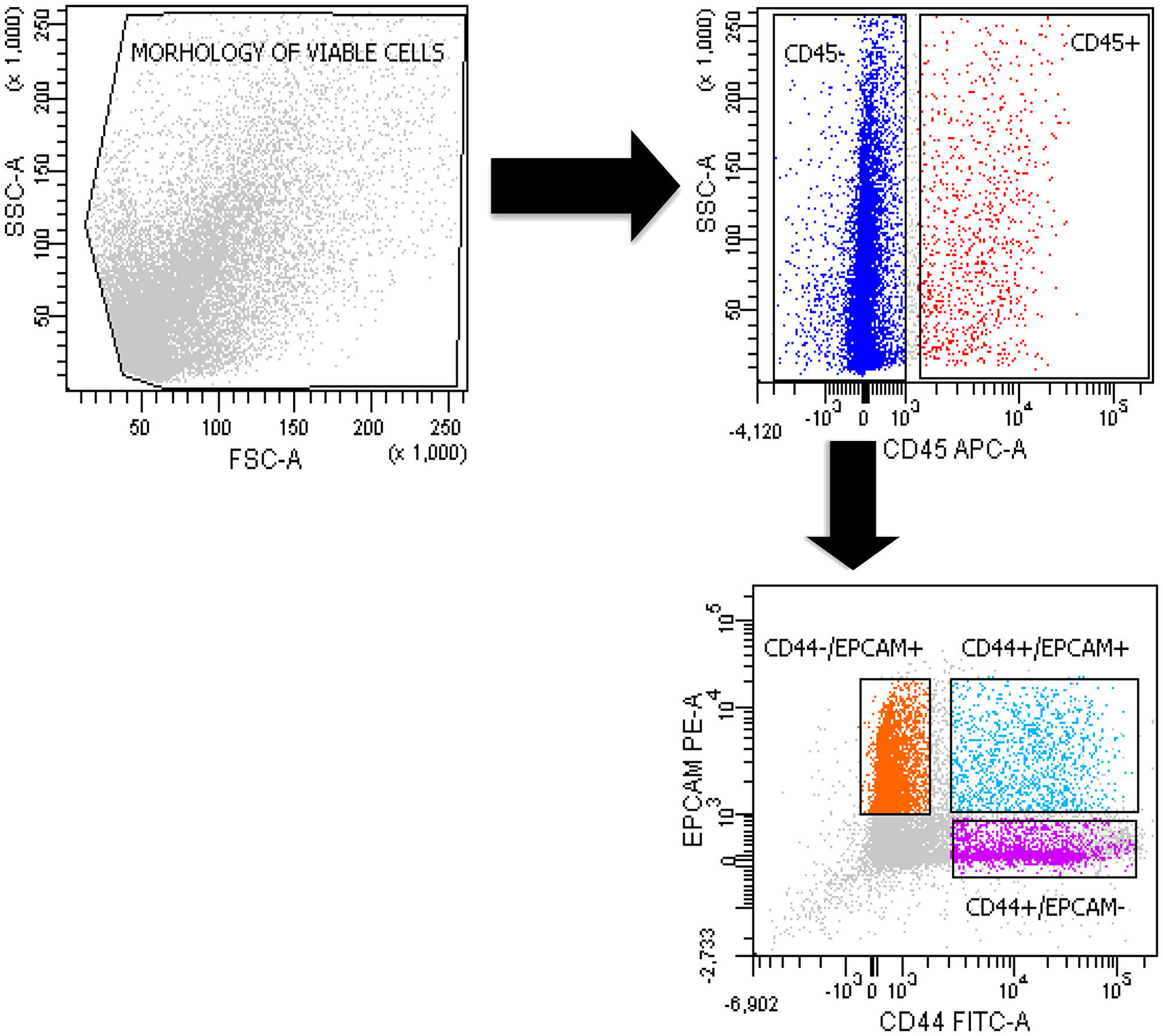
Figure 1: Sorting of double-positive CD44+/EPCAM+ and negative CD44+/EPCAM– and CD44–/EPCAM+ cells. The gating strategy of a representative FACS analysis of a primary tumor cell suspension in 1 patient. We used 7-AAD to detect live cells and CD45 to exclude the hematopoietic cell populations.
Relationship between ALDHhigh cancer stem-like cells and CD44+/EPCAM+ cells in primary lung cancer
As per the data reported in Table 2, there was a highly positive correlation between the expression of ALDHhigh cells and the expression of CD44+/EPCAM+ cells, with a Pearson’s linear correlation coefficient equal to 0.69 (95% CI 0.39–0.86; P = 0.0002), and Spearman’s rank correlation coefficient was equal to 0.52 (P = 0.0124). Conversely, no correlation was observed between ALDHhigh and CD44+/EPCAM– cell subpopulations or between ALDHhigh and CD44–/EPCAM+ cell subpopulations (Table 2).
Table 2: Correlation of ALDHhigh cells expression with expression of CD44+ and EPCAM+ cells
| Antigens | Pearson’s correlation | Spearman’s correlation | ||
|---|---|---|---|---|
| r (95% CI) | p | r | p | |
| CD44+/EPCAM+ | 0.69 (0.39; 0.86) | 0.0002 | 0.52 | 0.0124 |
| CD44+/EPCAM– | 0.07 (–0.35; 0.48) | 0.7429 | 0.19 | 0.3918 |
| CD44–/EPCAM+ | –0.29 (–0.63; 0.14) | 0.1810 | -0.30 | 0.1663 |
The results of the assessment of the differences in the expression of ALDHhigh and CD44+/EPCAM+ cells are reported in Table 3. The average paired difference between the expression of ALDHhigh and CD44+/EPCAM+ cells was very close to 0, being 0.1% (SD 2.5%, range –5.2% to 5.0%). Moreover, 43.5% of samples had a difference of less than 1% between the expression of ALDHhigh cells and the expression of CD44+/EPCAM+ cells, and 69.6% of samples had a difference of less than 2% between the expression of ALDHhigh cells and the expression of CD44+/EPCAM+ cells. Based on the paired t-test, there was no difference between these subpopulations in terms of means (P = 0.8464), and the 95% confidence interval for the paired difference was very narrow, ranging from –1.0% to 1.2%. Considering a target difference equal to 2% and based on the observed standard deviation for the difference between ALDHhigh and CD44+/EPCAM+ cells, the power of the paired t-test would be equal to 95.9%; if considering a target difference of 1.5% or 1%, the corresponding powers would be equal to 79.2% and 45.6%, respectively.
Table 3: Analysis of equality of expressions of ALDHhigh and CD44+/EPCAM+ cells
| All samples (n = 23) | p-value | ||
|---|---|---|---|
| Δ (ALDHhigh minus CD44+/EPCAM+) | mean ± SD (95% CI) | 0.1 ± 2.5% (–1.0 ; 1.2%) | 0.8464 |
| median (range) | 0.0% (–5.2 ; 5.0%) | ||
| |Δ| ≤ 1% | n (%) (cum %) | 10 (43.5%) (43.5%) | |
| |Δ| ≤ 2.5% | n (%) (cum %) | 6 (26.1%) (69.6%) | |
| |Δ| ≤ 5% | n (%) (cum %) | 5 (21.7%) (91.3%) | |
| |Δ| ≤ 7.5% | n (%) (cum %) | 2 (8.7%) (100.0%) |
Finally, based on the linear regression model, we estimated that a 1% increase in CD44+/EPCAM+ expression yields on average a 0.9% increase in ALDH+ expression (95% CI 0.5%–1.3%). The estimated regression equation was very similar to the line representing equality in the expression of the 2 types of cells (Figure 2).
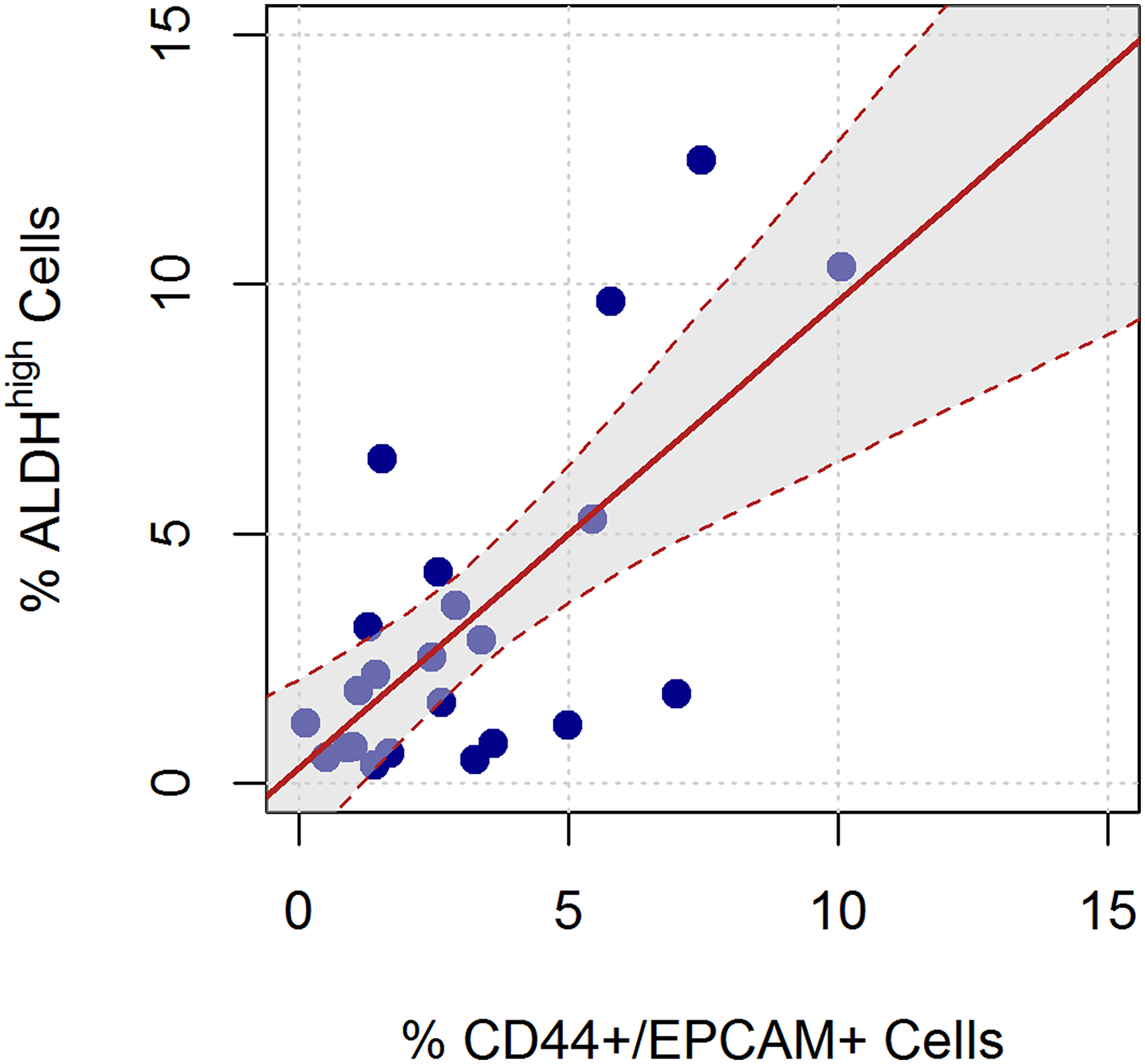
Figure 2: Linear relation between the ALDHhigh and CD44+/EPCAM+ sorted cells. The red line represents the linear regression equation, considering ALDHhigh as the dependent variable and CD44+/EPCAM+ as the independent variable. The shaded area represents the confidence interval for the regression equation.
Cytofluorimetric analysis of ALDH high/low sorted cell subpopulations for the identification of CD44+/EPCAM+ cells
Primary lung cancer cells derived from a 65-year-old male patient who had undergone surgery for stage IIB NSCLC were sorted by FACS to isolate ALDHhigh and ALDHlow cells. Subsequently, both ALDHhigh and ALDHlow cells were further analyzed for CD44+/EPCAM+ expression by FACS, setting a gate on 7AAD- and CD45. The results showed that a total of 49.4% of ALDHhigh cells were CD44+/EPCAM+, whereas only 2.6% of ALDHlow cells were CD44+/EPCAM+ (ratio 19:1) (Figure 3A).
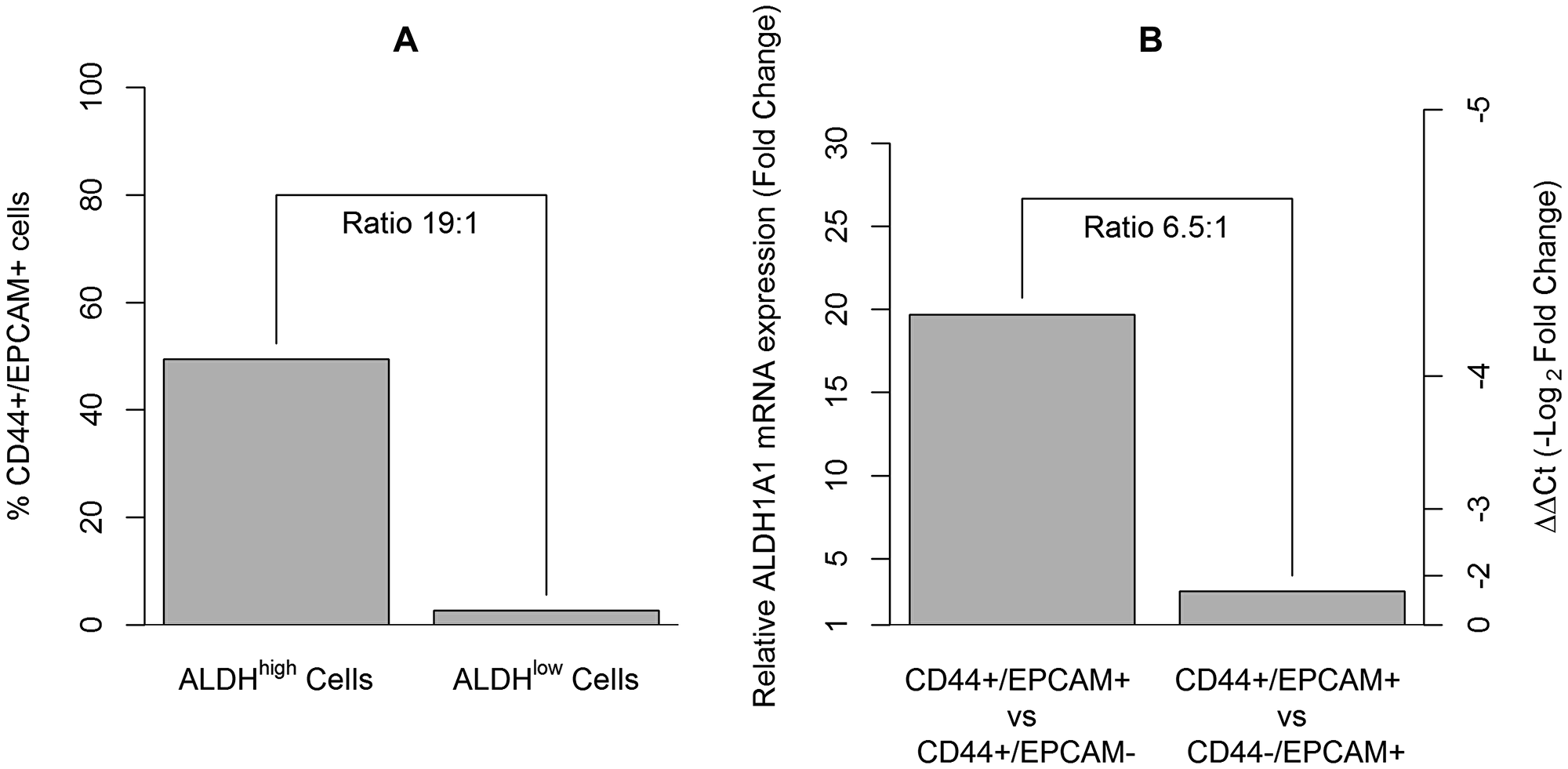
Figure 3: The assessment of CD44+/EPCAM+ cells in ALDHhigh sorted cells and their enrichment for the ALDH1A1 cancer stem cell gene. (A) Approximately 50% of the ALDHhigh sorted cells were CD44+/EPCAM+ cells, whereas only 2.6% of the the ALDHlow cells were CD44+/EPCAM+ cells. (B) Gene expression analysis of ALDH1A1 assessed in CD44+/EPCAM+, CD44+/EPCAM–, and CD44–/EPCAM+ sorted cells. Histogram on the left axis showed a fold change equal to 19.3 times (ΔΔCt = –4.3) when comparing CD44+/EPCAM+ to CD44+/EPCAM– cells and a fold change equal to 3.0 times (ΔΔCt = –1.6) when comparing CD44+/EPCAM+ to CD44–/EPCAM+ cells.
CD44+/EPCAM+ sorted cells have the ability to form tumor spheres in vitro
Primary sorted CD44+/EPCAM+ cells derived from a 74-year-old male patient, who underwent surgery for stage IIIA NSCLC, were cultured to assess the growth ability of these cells to form tumor spheres in vitro (Figure 4, Panel 1A–1C). At the time point of 2 days after the seeding, the CD44+/EPCAM+ cells had formed non-adherent spheres in culture, and more than 50% of these tumor spheres were either small or medium in size, with a total area below 16.000 μm2. At the time point of 7 days after the seeding, there were no more spheres of 20.000 μm2 or below in area, and the spheres belonging to the other classes had not increased in number since the time point of 2 days after the seeding. At the end point, 21 days after the seeding of the cells, only the tumor spheres of medium/large area remained, with spheres belonging to the classes of 16.000–32.000 μm2 in area and 32.000-64.000 μm2 in area representing 50% of the total spheres in culture (Figure 4, Panel 2A–2C).
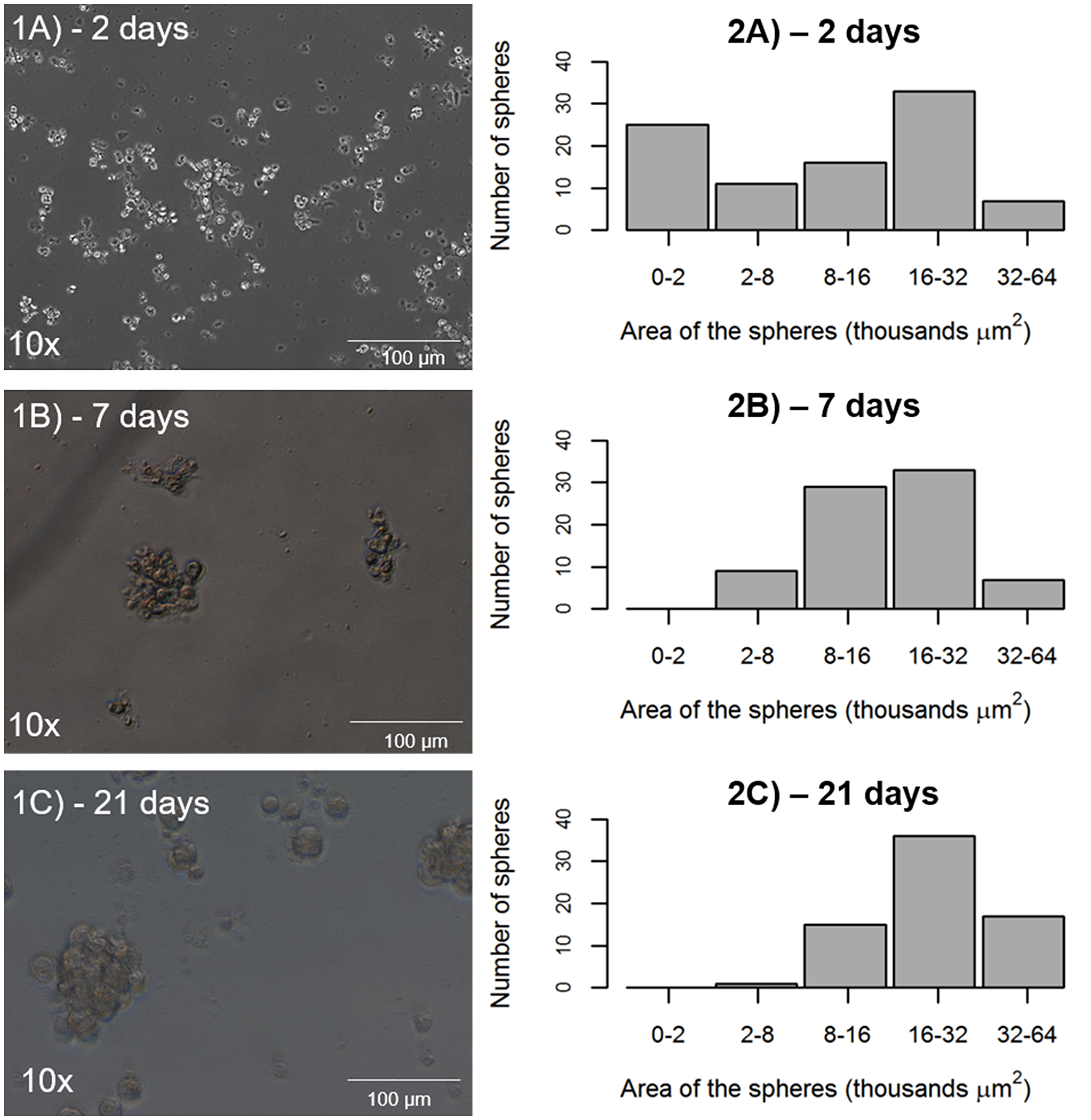
Figure 4: Potential of tumor sphere formation by CD44+/EPCAM+ sorted cells. (1A–1C) representative tumor spheres from CD44+/EPCAM+ sorted cells at 2, 7, and 21 days. (2A–2C), tumor spheres exhibit the same number over time and increased area, as measurement at 2, 7, and 21 days indicates.
Gene expression of ALDH1A1 cancer stem cell marker according to positivity for CD44/EPCAM
As an additional experiment, primary cells derived from a 76-year-old male patient, who underwent surgery for stage IA2 NSCLC, were co-stained for anti-CD44 and anti-EPCAM antibodies for selection by cell sorting. The gene expression of ALDH1A1 was assessed in the CD44+/EPCAM+ cells in comparison with the other cell subpopulations sorted, the CD44+/EPCAM– cells and the CD44–/EPCAM+ cells. A fold change equal to 19.3 times (ΔΔCt = –4.3) was found when comparing CD44+/EPCAM+ cells to CD44+/EPCAM– cells, whereas a fold change equal to 3.0 times (ΔΔCt = –1.6) was observed when comparing CD44+/EPCAM+ cells to CD44–/EPCAM+ cells. The ratio of these two fold changes was approximately 6.5:1 (Figure 3B).
DISCUSSION
Lung cancer has remained the most commonly occurring cancer globally. Traditional methods such as surgery, radiotherapy, and chemotherapy are treatment methods for lung cancer. Resistance to radiotherapy and chemotherapy and cancer relapse, however, remain challenging issues in lung cancer treatment. Chemoresistance is a major problem in the treatment of cancer patients, as cancer cells become resistant to the chemical substances used in treatment, thereby limiting the efficiency of chemo agents [14]. This resistance is attributable to a class of cells known as cancer stem cells. In fact, accumulating evidence suggests that the cancer stem cell population is responsible for chemoresistance and cancer relapse, as CSCs have the ability to self-renew and to differentiate into heterogeneous lineages of cancer cells in response to chemotherapeutic agents [15, 16]. CSCs are also able to induce cell cycle arrest (quiescent state), which supports their ability to become resistant to chemotherapy and radiotherapy [17, 18].
Although successful cancer therapy abolishes the bulk of proliferating tumor cells, a subset of remaining CSCs can survive and promote cancer relapse due to their ability to establish higher invasiveness and chemoresistance [18, 19]. Understanding the features of CSCs will be important in establishing the foundation for a new era in the treatment of cancer. To achieve this goal, we strongly believe that an accurate identification of CSC antigens may be helpful in improving knowledge of this small but harmful population. This is why we conducted our experiments comparing ALDHhigh and CD44+/EPCAM+ cell subpopulations.
The choice to use these markers derived from recent studies demonstrating that ALDH and CD44 are the most common CSC markers in many solid tumors [18, 20]. Nevertheless, in our previous study [15], we found comparable expression of ALDHhigh and CD44+ cells without a significant positive correlation, hence the necessity to add another antigen for identifying the cells that are the most similar to ALDHhigh cells, which are actually considered cancer stem cells [21, 22]. The choice to use EPCAM derived from a specific characteristic of this marker: it is an epithelial cell adhesion molecule, expressed by solid tumors of epithelial origin, such as non-small-cell lung cancer, breast cancer, or ovarian cancer [23]. Moreover, it is well recognized that EPCAM positive cells possess tumor-initiating potential, and EPCAM has already been used as a key marker of ovarian cancer stem cells [24]. These findings strongly support the notion that EPCAM is an ideal therapeutic target for ovarian cancer. In fact, various EPCAM antagonists have been developed as EPCAM-targeted antibodies serving as an effective treatment in both experimental models and clinical trials. From these findings, we decided to use CD44 together with EPCAM, comparing these selected cell subpopulations with the ALDHhigh cell subpopulation.
Interestingly, we found that there is a highly positive linear correlation between ALDHhigh cells and CD44+/EPCAM+ cells. Furthermore, a very strong similarity between these cell subpopulations in terms of means was found, and their linear relationship was assessed as very close to equality. Conversely, we observed no correlation between ALDHhigh and CD44+/EPCAM– cells or between ALDHhigh and CD44–/EPCAM+ cells. These results suggested to us that the closest cells to ALDHhigh cells are CD44+/EPCAM+ cells. We regard this as a relevant result, since to the authors’ knowledge this is the first study that investigated and identified a robust correlation between these populations of cancer stem cells. Based on our observed results, this is the first study, which demonstrates that these two cancer stem cells populations seem to match closely with each other.
As performed previously [12], we decided to assess the tumor sphere formation ability of CD44+/EPCAM+ cells. We observed a growth of tumor spheres for up to 3 weeks, with results that were similar to those previously recorded for ALDHhigh cells in terms of the number and size of spheres [7].
As proof of concept of the ALDH enrichment in the population identified by CD44+/EPCAM+ cells, we performed an additional experiment in which primary lung cancer cells were sorted for CD44/EPCAM antigens. The ALDH mRNA expression was evaluated by real-time PCR in CD44+/EPCAM+, CD44+/EPCAM–, and CD44–/EPCAM+ cell subpopulations. Our results showed an abundant overexpression of the ALDH1A1 gene (fold change of 19.3) for CD44+/EPCAM+ cells versus CD44+/EPCAM– cells; meanwhile, a slighter overexpression of the ALDH1A1 gene (fold change of 3) was observed for CD44+/EPCAM+ cells versus CD44–/EPCAM+ cells. Strong evidence also derived from the cytofluorimetric analysis of the ALDHhigh sorted cells stained for CD44/EPCAM. Approximately 50% of the ALDHhigh sorted cells were CD44+/EPCAM+, whereas only 2.6% of the ALDHlow sorted cells were CD44+/EPCAM+.
Taken together, the results of the present research highlight a strong similarity between ALDHhigh and CD44+/EPCAM+ cells, as suggested by cytofluorimetric analyses, tumor sphere-forming assays, and RT-PCR experiments.
The identification of surface markers in lung cancer can contribute to personalized medicine. Although there is still uncertainty in the identification of cancer stem cells especially in lung, the scientific community have pointed out their important role in prognosis and recurrence [25]. Furthermore, the identification of these markers may be helpful for a better epidemiological stratification of oncological patients. Hence, the identification of cancer stem cell markers should be considered as a crucial step in the development of novel cancer-specific molecular targeted therapies [25].
Our experiments, performed as proof of concept, showed that the superficial marker CD44+/EPCAM+ detected about half of the ALDHhigh cell population. These two populations seem to be very similar in terms of stemness gene expression as well as their capacity to make spheroids. In a recent study, MacDonagh et al. reported that ALDH1-positive cells can survive under cisplatin treatment [26]. This resistance to cancer treatments is daily confirmed in many oncologic patients. Although ALDH is nowadays considered an important marker for cancer stem cells as well as also for epithelial cancer cells [27] and that some clinical trials are running at the present time for analyzing ALDH-targeting treatments effects against cancer [28, 29], we strongly agree with the scientific community about the importance to identify markers, in order to develop target treatments against cancer stem cells [30–32]. In particular, with regards of our study, even if we hypothesized that the percentage of CD44+/EPCAM+ in ALDHhigh cell population would be less than 50% in other patients, the similarity of an intracellular marker highlighting ALDHhigh cell population with a superficial marker CD44+/EPCAM+ is a very important concept in terms of target treatment. In summary, our research is an important starting point for further studies that are needed to better define the CD44+/EPCAM+ superficial marker highlighting lung cancer stem cells.
Limitations
A limitation of our study was that we did not perform formal sample size calculations for our main cytofluorimetric analysis. Nevertheless, the statistical power was calculated ex post and was satisfactory.
Materials and Methods
The study was conducted by analyzing the expression of ALDHhigh/low cells and the positivity for CD44 and EPCAM antigens in a sample of surgical lung cancer tissue. The main approach was a cytofluorimetric analysis of ALDH expression and positivity for CD44/EPCAM on primary cell population obtained from 23 patients harboring NSCLC. Moreover, the following additional experiments were performed, on 1 patient each: 1) a cytofluorimetric analysis of CD44+/EPCAM+ on ALDHhigh/low sorted cell subpopulations; 2) a tumor sphere-forming assay on CD44+/EPCAM+ sorted cells; 3) a real-time polymerase chain reaction (RT-PCR) for the ALDH1A1 mRNA expression on CD44+/EPCAM+ sorted cells.
Study design and sample size
The present study was a cross-sectional study and was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines [33].
The outcomes evaluated in the study were the expressions of ALDHhigh, CD44+/EPCAM+, CD44+/EPCAM–, and CD44–/EPCAM+ cells. The study primarily aimed at assessing the correlation and the pairwise difference between the expression of ALDHhigh and CD44+/EPCAM+ cells. Secondarily, correlations between ALDHhigh and CD44+/EPCAM– cells and between ALDH+ and CD44–/EPCAM+ cells were considered.
A formal calculation for sample size was not performed. A sample of patients who met the inclusion criteria during a 14-month timeframe was used. Statistical power calculations were performed ex post and were reported.
Study population
Overall, 24 patients met our inclusion criteria within those harboring NSCLC undergone major lung resection by lateral thoracotomy at the Division of Thoracic Surgery of Modena University Hospital (Italy) for stage I, II, or IIIA non-small-cell lung cancer (8th tumor, node, metastasis (TNM) staging system) in the period from December 2017 to January 2019 were included in the study. A main cytofluorimetric analysis was performed in 23 patients; for one patient an additional cytofluorimetric analysis was added and in another one, the tumor sphere-forming assay was carried out. The last patient enrolled was only included for the additional RT-PCR experiment. Inclusion criteria were aged between 18 and 85, R0 resection, availability of formalin fixed, paraffin embedded surgery specimen from the primary tumor, and availability of fresh surgical specimen for cytofluorimetric analysis. Exclusion criteria were incomplete resection, unknown tumor, node, metastasis (TNM) status, synchronous tumors, and previous lung cancer.
Primary cells isolated from human lung cancer
Tumor tissues were obtained within 1 to 2 h after surgical removal, washed in sterile Dulbecco’s phosphate-buffered saline (PBS) (L1825-BC – Merck Millipore, Italy), and mechanically minced into small pieces (2 to 4 mm). Minced samples were digested using a tumor dissociation kit in a disposable gentle MACS™ C-Tube (Miltenyi Biotec, Italy) according to the manufacturer’s instructions. Samples were digested for 60 min at 37°C in a gentle MACS Octo dissociator, filtered through 70-μm sterile cell strainers, centrifuged at 300 × g for 5 min, and resuspended in a DMEM and HAM’S F12 media mixture (2:1) (Gibco) containing 50 IU/mL penicillin-streptomycin and 4 mM glutamine. Viable cells were counted using an optic phase-contrast microscope [7].
ALDEFLUOR assay
Single-cell suspensions of primary tumor cells were diluted in ALDEFLUOR assay buffer containing BODIPY-aminoacetaldehyde (STEMCELL Technologies, Vancouver, BC). The assay was performed according to the manufacturer’s protocol. Briefly, at least 5,000,000 tumor cells were resuspended in ALDEFLUOR BUFFER (5 μl/106) and stained with ALDEFLUOR substrate. Immediately, 5 × 105 cells were transferred to a control tube containing 5 μl of diethylaminobenzaldehyde (DEAB), which is a specific inhibitor of ALDH. Control and test samples were incubated for 45 min at 37°C and protected from light. Cells were centrifuged at 300 × g for 5 min. The cell pellet was resuspended in 1 ml of ALDEFLUOR assay buffer [7, 13].
Cytofluorimetric analysis and cell sorting
Primary tumor cell suspensions were stained with ALDEFLUOR (Stem Cell) and allophycocyanin (APC)-conjugated anti-CD45 (Becton Dickinson, Franklin Lakes, NJ), phycoerythrin (PE)-conjugated anti-EPCAM (Becton Dickinson), and Brilliant Blue 515 (BB515) anti-CD44 (Becton Dickinson). An isotype control sample for each condition was used to exclude the autofluorescence background. Cell morphology was evaluated using side scatter (SSC) and forward scatter (FSC). Dead cells were excluded using 7-amino-actinomycin D (7-AAD) staining. Both types of cells, ALDHhigh and double-positive CD44/EPCAM, were analyzed and sorted. The gate was set based on ALDHhigh cells in 1 case and on CD45-negative-CD44+/EPCAM+, CD45-negative cells-CD44+/EPCAM– and CD45-negative cells-CD44–/EPCAM+ in the other cases.
Cell sorting was performed using a FACSAria III (Becton Dickinson). The results were analyzed using fluorescence-activated cell sorting (FACS) Diva software (Becton Dickinson, Franklin Lakes, NJ). Sorted cells were directly lysed for gene expression analyses and seeded in culture for tumor sphere assay.
Tumor sphere-forming assay
A tumor sphere-forming assay on CD44+/EPCAM+ cells was performed, as previously assessed by our research group for ALDHhigh cells [7]. The CD44+/EPCAM+ single-cell suspension was seeded in 24 ultralow attachment well plates. Cells were cultured in a mixture of serum-free Dulbecco’s modified Eagle’s medium (DMEM) and HAM’S F12 media (2:1) (Gibco) containing 50 IU/mL penicillin-streptomycin and 4 mM glutamine supplemented with 5 μg/ml insulin, 10 ng/ml epidermal growth factor (EGF), 20 ng/ml basic fibroblast growth factor (bFGF), 0.18 nM adenine, and 2 nM triiodotironin. The cells were cultured in 5% CO2 at 37°C for 3 weeks, and the media were replaced or supplemented with fresh growth factors twice per week. The entire well was digitally photographed using inverted phase-contrast microscopy (Zeiss Axioskop and Axiocam ICc3 color camera). All images were analyzed using the AxioVision software (Zeiss). The total number of spheres was counted, and the spheres’ areas were manually measured at 2, 7, and 21 days from seeding.
Real-time PCR (RT-PCR)
Total cellular RNA was extracted from double-positive CD44/EPCAM sorted cells using the RNeasy Mini Kit (Qiagen) according to the manufacturer’s instructions. Total RNA (2 μg) was reverse transcribed using the RevertAid™ First Strand cDNA Synthesis Kit (Thermo Scientific). Following cDNA synthesis, real-time PCR (RT-PCR) was performed in technical replicates for each sample using FAST SYBR™ Green detection chemistry (Applied Biosystems) on Step One instrument. Human ALDH1A1 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were amplified using gene-specific primers (ALDH1A1: forward primer 5′- TGTTAGCTGATGCCGACTTG-3′, reverse primer 5′-TTCTTAGCCCGCTCAACACT-3′; GAPDH: forward primer 5′-ACATCGCTCAGACACCATG-3′, reverse primer 5′TGTAGTTGAGGTCAATGAAGGG-3′). The forward and reverse primers were designed using IDT PrimerQuest1 (http://eu.idtdna.com/PrimerQuest/Home/Index). The cycling parameters consisted of denaturation at 95°C for 10 min and 40 cycles of 15 sec at 94°C, 30 sec at 60°C, and 1 min at 72°C, followed by a continuous melting curve.
Statistical analysis
The continuous variables were described in terms of mean, standard deviation (SD), median, and range. The categorical variables were described as absolute and percentage frequencies.
Expression of all cells was measured as the percentages of 7-AAD negative (7-AAD-) cells. We calculated the correlations of the expression of ALDHhigh cells with the expression of CD44+/EPCAM+, CD44+/EPCAM–, and CD44–/EPCAM+ cells. Both Pearson’s linear correlation and Spearman’s rank correlation coefficients were considered. The difference between the expression of ALDHhigh cells and the expression of CD44+/EPCAM+ cells was assessed using a paired t-test. The statistical power of the test was calculated ex post based on the observed standard deviation of the mean differences and considering 3 target differences equal to 2%, 1.5%, and 1%. A linear regression model that considers ALDHhigh cells expression as the dependent variable and CD44+/EPCAM+ cells expression as the independent variable was used to assess the linear relationship of the two cells subpopulations. The regression line was reported graphically.
RT-PCR data were analyzed according to the methods described in [34]. The relative mRNA expression of ALDH+ cells was reported as Delta Delta Ct (ΔΔCt) and as fold changes (FC, equal to 2-ΔΔCt). Two relative mRNA expressions of ALDHhigh cells were calculated: 1) CD44+/EPCAM+ compared to CD44+/EPCAM– cells; 2) CD44–/EPCAM+ compared to CD44+/EPCAM+ cells.
The analyses were conducted using R 3.6.1 statistical software (The R Foundation for Statistical Computing, Wien) at the 95% confidence level.
Conclusions
Our study is the first attempt toward the utility of a double superficial marker such as CD44+/EPCAM+ for the identification and further the targeting of lung cancer stem cells. This will be helpful for the setting of new treatments against lung cancer stem cells as well as also for a better control of the tumor growth.
Consent for publication
This Study, involving human subjects, human material, and human data, has been performed in accordance with the Declaration of Helsinki and has been approved by the Ethics committee at University Hospital of Modena, MODENA, Italy, on 17 March 2017, Prot. N. 914/C.E. Further information and documentation to support this is available to the Editor on request. All the patients included in this Study have signed an informed consent before being enrolled. Further information and documentation to support this is available to the Editor on request. Consent for publication of data has been obtained from study participants. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
NSCLC: non-small cell lung cancer; ALDH: aldehyde dehydrogenase; CD44: cluster of differentiation 44; EPCAM: epithelial cell adhesion molecule; FACS: fluorescence-activated cell sorting; CSCs: cancer stem cells; SSC: side scatter; FSC: forward scattered.
ACKNOWLEDGMENTS
The Project has been supported in parts by funds from the Division of Thoracic Surgery of the University Hospital of Modena and from the Laboratory of Cellular Therapy of the University of Modena and Reggio Emilia, from unrestricted grant from Myriad Inc. (US) and from the Italian Ministry of Education, University and Research: Departments of Excellence 2017.
CONFLICTS OF INTEREST
None of the Authors have any competing interests.
FUNDING
The Project has been supported by funds from the Division of Thoracic Surgery of the University Hospital of Modena and from the Laboratory of Cellular Therapy of the University of Modena and Reggio Emilia, Italy. These funds were used to buy laboratory equipment and to support the project and people involved.
References
1. Codony-Servat J, Verlicchi A, Rosell R. Cancer stem cells in small cell lung cancer. Transl Lung Cancer Res. 2016; 5:16–25. https://doi.org/10.3978/j.issn.2218-6751.2016.01.01. [PubMed].
2. Kelly PN, Dakic A, Adams JM, Nutt SL, Strasser A. Tumor growth need not be driven by rare cancer stem cells. Science. 2007; 317:337. https://doi.org/10.1126/science.1142596. [PubMed].
3. Salcido CD, Larochelle A, Taylor BJ, Dunbar CE, Varticovski L. Molecular characterisation of side population cells with cancer stem cell-like characteristics in small-cell lung cancer. Br J Cancer. 2010; 102:1636–1644. https://doi.org/10.1038/sj.bjc.6605668. [PubMed].
4. Wang P, Gao Q, Suo Z, Munthe E, Solberg S, Ma L, Wang M, Westerdaal NA, Kvalheim G, Gaudernack G. Identification and characterization of cells with cancer stem cell properties in human primary lung cancer cell lines. PLoS One. 2013; 8:e57020. https://doi.org/10.1371/journal.pone.0057020. [PubMed].
5. Sales KM, Winslet MC, Seifalian AM. Stem cells and cancer: an overview. Stem Cell Rev. 2007; 3:249–255. https://doi.org/10.1007/s12015-007-9002-0. [PubMed].
6. Masciale V, Grisendi G, Banchelli F, D’Amico R, Maiorana A, Sighinolfi P, Pinelli M, Lovati E, Stefani A, Morandi U, Dominici M, Aramini B. Correlating tumor-infiltrating lymphocytes and lung cancer stem cells: a cross-sectional study. Ann Transl Med. 2019; 7:619. https://doi.org/10.21037/atm.2019.11.27. [PubMed].
7. Masciale V, Grisendi G, Banchelli F, D'Amico R, Maiorana A, Sighinolfi P, Stefani A, Morandi U, Dominici M, Aramini B. Isolation and Identification of Cancer Stem-Like Cells in Adenocarcinoma and Squamous Cell Carcinoma of the Lung: A Pilot Study. Front Oncol. 2019; 9:1394. https://doi.org/10.3389/fonc.2019.01394. [PubMed].
8. Zakaria N, Yusoff NM, Zakaria Z, Lim MN, Baharuddin PJ, Fakiruddin KS, Yahaya B. Human non-small cell lung cancer expresses putative cancer stem cell markers and exhibits the transcriptomic profile of multipotent cells. BMC Cancer. 2015; 15:84. https://doi.org/10.1186/s12885-015-1086-3. [PubMed].
9. Wang Y, Jiang M, Du C, Yu Y, Liu Y, Li M, Luo F. Utilization of lung cancer cell lines for the study of lung cancer stem cells. Oncol Lett. 2018; 15:6791–6798. https://doi.org/10.3892/ol.2018.8265. [PubMed].
10. Leung EL, Fiscus RR, Tung JW, Tin VP, Cheng LC, Sihoe AD, Fink LM, Ma Y, Wong MP. Non-Small Cell Lung Cancer Cells Expressing CD44 Are Enriched for Stem Cell-Like Properties. PLoS One. 2010; 5:e14062. https://doi.org/10.1371/journal.pone.0014062. [PubMed].
11. Sullivan JP, Spinola M, Dodge M, Raso MG, Behrens C, Gao B, Schuster K, Shao C, Larsen JE, Sullivan LA, Honorio S, Xie Y, Scaglioni PP, et al. Aldehyde dehydrogenase activity selects for lung adenocarcinoma stem cells dependent on Notch signalling. Cancer Res. 2010; 70:9937–9948. https://doi.org/10.1158/0008-5472.CAN-10-0881. [PubMed].
12. Satar NA, Fakiruddin KS, Lim MN, Mok PL, Zakaria N, Fakharuzi NA, Abd Rahman AZ, Zakaria Z, Yahaya BH, Baharuddin P. Novel Triple positive Markers Identified in Human Non small Cell Lung Cancer Cell Line With Chemotherapy-Resistant and Putative Cancer Stem Cell Characteristics. Oncol Rep. 2018; 40:669–681. https://doi.org/10.3892/or.2018.6461. [PubMed].
13. Masciale V, Grisendi G, Banchelli F, D'Amico R, Maiorana A, Morandi U, Dominici M, Aramini B. Cancer stem-neuroendocrine cells in an atypical carcinoid case report. Transl Lung Cancer Res. 2019; 8:1157–1162. https://doi.org/10.21037/tlcr.2019.12.07. [PubMed].
14. Tredan O, Galmarini CM, Patel K, Tannock IF. Drug resistance and the solid tumor microenvironment. J Natl Cancer Inst. 2007; 99:1441–1454. https://doi.org/10.1093/jnci/djm135. [PubMed].
15. Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008; 8:755–768. https://doi.org/10.1038/nrc2499. [PubMed].
16. Dean M, Fojo T, Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer. 2005; 5:275–284. https://doi.org/10.1038/nrc1590. [PubMed].
17. Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene. 2010; 29:4741–4751. https://doi.org/10.1038/onc.2010.215. [PubMed].
18. Shlush LI, Mitchell A, Heisler L, Abelson S, Ng SWK, Trotman-Grant A, Medeiros JJF, Rao-Bhatia A, Jaciw-Zurakowsky I, Marke R, McLeod JL, Doedens M, Bader G, et al. Tracing the origins of relapse in acute myeloid leukaemia to stem cells. Nature. 2017; 547:104–108. https://doi.org/10.1038/nature22993. [PubMed].
19. Pattabiraman DR, Weinberg RA. Tackling the cancer stem cells — what challenges do they pose? Nat Rev Drug Discov. 2014; 13:497–512. https://doi.org/10.1038/nrd4253. [PubMed].
20. Templeton AK, Miyamoto S, Babu A, Munshi A, Ramesh R. Cancer stem cells: progress and challenges in lung cancer. Stem Cell Investig. 2014; 1:9. https://doi.org/10.3978/j.issn.2306-9759.2014.03.06. [PubMed].
21. Chen D, Wang CY. Targeting cancer stem cells in squamous cell carcinoma. Precis Clin Med. 2019; 2:152–165. https://doi.org/10.1093/pcmedi/pbz016. [PubMed].
22. Vassalli G. Aldehyde Dehydrogenases: Not Just Markers, but Functional Regulators of Stem Cells. Stem Cells Int. 2019; 2019:3904645. https://doi.org/10.1155/2019/3904645. [PubMed].
23. Marcato P, Dean CA, Giacomantonio CA, Lee PW. Aldehyde dehydrogenase: Its role as a cancer stem cell marker comes down to the specific isoform. Cell Cycle. 2011; 10:1378–1384. https://doi.org/10.4161/cc.10.9.15486. [PubMed].
24. Zheng J, Zhao S, Yu X, Huang S, Liu HY. Simultaneous targeting of CD44 and EpCAM with a bispecific aptamer effectively inhibits intraperitoneal ovarian cancer growth. Theranostics. 2017; 7:1373–1388. https://doi.org/10.7150/thno.17826. [PubMed].
25. Imrich S, Hachmeister M, Gires O. EpCAM and its potential role in tumor-initiating cells. Cell Adhes Migr. 2012; 6:30–38. https://doi.org/10.4161/cam.18953. [PubMed].
26. MacDonagh L, Gallagher MF, Ffrench B, Gasch C, Breen E, Gray SG, Nicholson S, Leonard N, Ryan R, Young V, O'Leary JJ, Cuffe S, Finn SP, et al. Targeting the cancer stem cell marker, aldehyde dehydrogenase 1, to circumvent cisplatin resistance in NSCLC. Oncotarget. 2017; 8:72544–72563. https://doi.org/10.18632/oncotarget.19881. [PubMed].
27. Toledo-Guzmán ME, Hernández MI, Gómez-Gallegos ÁA, Ortiz-Sánchez E. ALDH as a Stem Cell Marker in Solid Tumors. Curr Stem Cell Res Ther. 2019; 14:375–388. https://doi.org/10.2174/1574888X13666180810120012. [PubMed].
28. Evaluation of the Effectiveness of an Aldehyde Inhibitor Dehydrogenases (DIMATE) on the Cell Population Leukemic or Normal Stem (DIMATE). 2016. https://clinicaltrials.gov/ct2/show/NCT02748850.
29. Marcucci F, Rumio C, Lefoulon F. Anti-Cancer Stem-like Cell Compounds in Clinical Development – An Overview and Critical Appraisal. Front Oncol. 2016; 6:115. https://doi.org/10.3389/fonc.2016.00115. [PubMed].
30. Saygin C, Matei D, Majeti R, Reizes O, Lathia JD. Targeting Cancer Stemness in the Clinic: From Hype to Hope. Cell Stem Cell. 2019; 24:25–40. https://doi.org/10.1016/j.stem.2018.11.017. [PubMed].
31. Turdo A, Veschi V, Gaggianesi M, Chinnici A, Bianca P, Todaro M, Stassi G. Meeting the Challenge of Targeting Cancer Stem Cells. Front Cell Dev Biol. 2019; 7:16. https://doi.org/10.3389/fcell.2019.00016. [PubMed].
32. Talukdar S, Emdad L, Das SK, Sarkar D, Fisher PB. Evolving Strategies for Therapeutically Targeting Cancer Stem Cells. Adv Cancer Res. 2016; 131:159–191. https://doi.org/10.1016/bs.acr.2016.04.003. [PubMed].
33. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008; 61:344–349. https://doi.org/10.1016/j.jclinepi.2007.11.008. [PubMed].
34. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25:402–08. https://doi.org/10.1006/meth.2001.1262. [PubMed].

