Introduction
Extramammary Paget’s disease (EMPD) is a rare intraepithelial adenocarcinoma, localized in apocrine gland-rich sites, most commonly perianal, vulvar and penoscrotal. The clinical presentation is discreet and eczematous in nature, often causing a delay in diagnosis. Most cases of EMPD are in-situ and have an excellent prognosis. In 5-25%, dermal invasion is visible, correlating with a poor prognosis. Due to the rarity of the disease, evidence-based management in both in-situ and metastatic disease is scarce. In the past, various combinatorial chemotherapeutic regimens were applied, with limited success. Recently, growing evidence suggests a central role of human epidermal growth factor receptor (HER) 2 in the disease. Yet incomplete responsiveness and the occurrence of treatment resistance to anti-HER2 treatment frequently occurs.
Results
We report on an 80-year-old patient in excellent general condition (ECOG 0), who was referred to our clinic for further treatment of a chronic scrotal eczema. Upon physical examination, erythematous plaque with multiple nodules affecting the skin of the scrotum was seen, along with prominent inguinal lymphadenopathy (Figure 1A). Histologic analysis showed an ulcerated and invasive adenocarcinoma with Paget cells, consistent with the diagnosis of extramammary Paget’s disease. Furthermore, enlarged and metabolic active inguinal, iliacal and interaortocaval lymph-nodes were detectable on PET/CT-imaging (Figure 2A). Indeed, fine needle aspiration (FNA) confirmed the presence of lymph node metastasis. Fluorescence in situ (FISH) for HER2 amplification was negative and immune-histochemical staining for HER2 was 2+, classifying the patient as HER2 negative, according to current ASCO/CAP Guidelines [1]. Urological and gastroenterological examination did not reveal underlying malignancy of the prostate or gastrointestinal tract, respectively. After interdisciplinary discussion, the patient was opted for radiotherapy of the penoscrotal region and metabolic active lymph nodes (cumulative 60Gy), which was well tolerated.
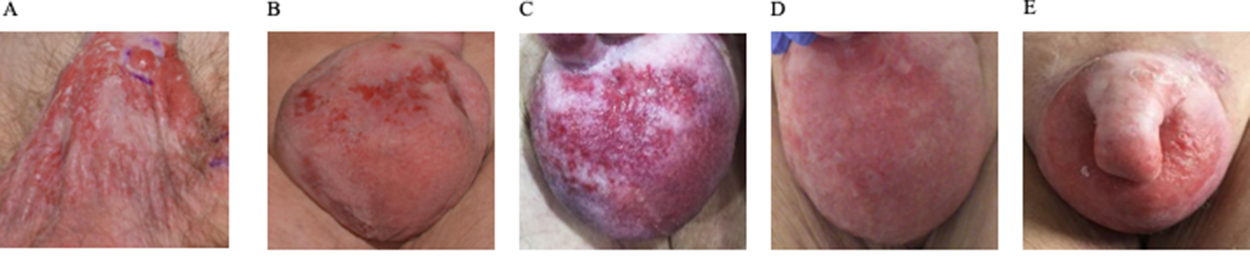
Figure 1: Therapeutic efficacy assessed by clinical penoscrotal examination. Imaging of the penoscrotal region (A) at the initial presentation, (B) following recurrent disease after radiation therapy (C) after lapatinib treatment (D) best response during trastuzumab and carboplatin treatment, and (E) disease progression after 10 cycles of trastuzumab and carboplatin.
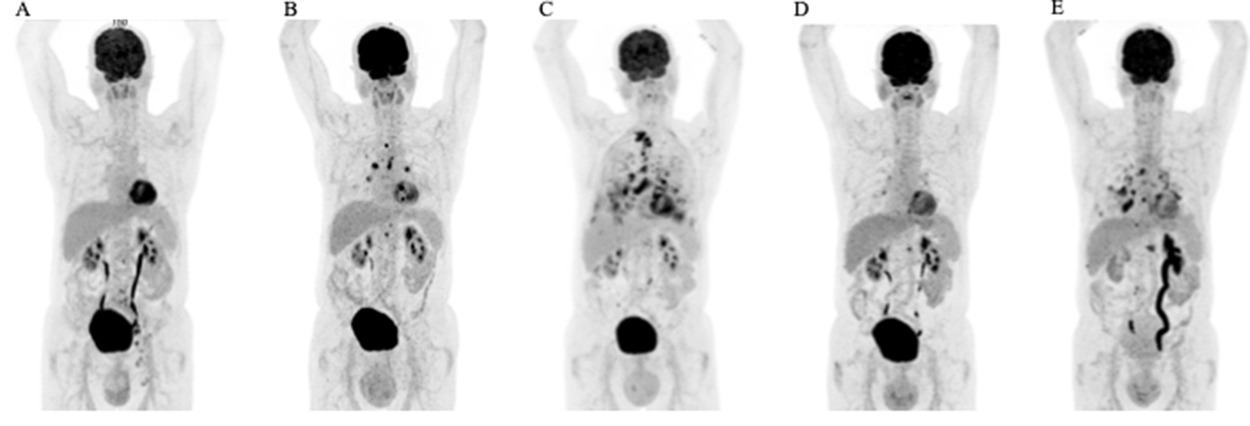
Figure 2: Therapeutic efficacy assessed by FDG-PET/CT scans. Coronal maximum intensity projection images (A) at the initial presentation, (B) following recurrent disease after radiation therapy (C) after lapatinib treatment (D) best response during trastuzumab and carboplatin treatment, and (E) disease progression after 10 cycles of trastuzumab and carboplatin.
6 months into follow-up, scrotal inflammation re-occurred (Figure 1B). Multiple mapping skin biopsies revealed apocrine adenocarcinoma in situ. PET/CT imaging showed new metabolic activity of the thoracal and retrocrural lymph-nodes, and a metabolically active nodulous in the superior right lobe of the lung (Figure 2B). FNA of the pulmonal nodule confirmed the presence of pulmonary metastasis. In order to identify targetable molecular alterations, next-generation sequencing (Oncomine Focus Assay) of the scrotal tumor was performed, revealing an activating ERBB2S310F point mutation. Accordingly lapatinib treatment (1250mg daily) was initiated [2]. However, the patient’s general condition deteriorated continuously along with respiratory insufficiency, leading to the necessity of continuous oxygen supplementation. While scrotal inflammation worsened, PET/CT imaging showed disease progression with pleural effusions and bone metastasis (Figure 1C and 2C, respectively). Again, histological analysis of FDG-avid bronchial mucosa was performed, confirming the presence of Paget-cells. Interestingly, HER2 immune-histochemical staining was strongly positive (3+) classifying the patient as HER2 positive. Therefore, lapatinib was discontinued and the patient was treated with trastuzumab (6mg/kg body weight) and carboplatin (300mg) every 3 weeks. Within one month, the patient’s general condition improved rapidly and oxygen supplementation was not further needed. In addition, scrotal inflammation resolved (Figure 1D). PET/CT imaging showed partial response of all lymph node metastasis, as well as subtotal response of pulmonary and bone metastasis (Figure 2D). A single bone metastasis at the cervical vertebra was irradiated due to local disease progression.
After 10 cycles of trastuzumab and carboplatin, follow-up PET/CT imaging displayed disease progression, with re-occurrence of multiple lymph-node and pulmonary metastasis (Figure 2E). Scrotal inflammation was mildly increased (Figure 1E). Molecular analysis (Oncomine Focus Assay) from a newly developing and metabolic active hilar lymph node metastasis was performed. Apart from the known ERBB2S310F mutation, additional genomic alterations were identified, including ERBB3A232V and PIK3CAG106V point mutations, and amplification of CDK6. In order to identify further potential treatable targets, Foundation One analysis was performed, revealing additional, equivocal MET amplification. Given the fact, that ERRB3 mutations have been associated with resistance to ERBB2 targeted treatment strategies, trastuzumab / carboplatin was discontinued and afatinib, an ERRB family Inhibitor, was initiated [3]. Unfortunately, 8 days after initiation of afatinib, the patient died of community acquired pneumonia.
DISCUSSION
Advances have been made in the treatment of metastatic extramammary Paget’s disease. The identification of ERBB2 amplifications and somatic mutations in EMPD has enabled disease specific, targeted treatment [4]. Indeed, 15-80% of all EMPD patients show immune-histochemical HER2 positivity, associated with a biologically aggressive phenotype [5]. Accordingly, anecdotal anti-HER2 treatment has significantly improved the outcome of metastasized EMPD, emphasizing its pathogenic role the disease. However, incomplete responsiveness and the occurrence of treatment resistance to anti-HER2 therapies demand the need for further knowledge.
The tyrosine receptor kinase HER2 causes increased MAPK/ERK and PI3K/mTOR pathway signaling, accelerating cell growth and survival. Historically, HER2 status is assessed by immuno-histochemistry (IHC) and fluorescence in-situ hybridization (FISH) for the detection of overexpression and amplification, respectively. However, nonamplified, activating ERBB2-mutations are not detected by IHC / FISH. Furthermore, only 30% of all ERBB2 alterations are amplifications, and nearly 2% of all tumors carry ERBB2 mutations [4, 6]. The activating S310F point mutation is the most common somatic mutation in ERBB2 and has been successfully targeted in EMPD [6, 7]. Given the similar beneficial response to anti-HER2 treatment in amplified and non-amplified ERBB2 alterations, patients with a potential benefit to anti-HER2 treatment may not be identified using IHC / FISH.
Here we report on the clinical efficacy of trastuzmab / carboplatin in an 80-year-old male patient with metastatic penoscrotal EMPD, harboring a somatic ERBB2S310F mutation, with primary resistance to lapatinib. Upon disease progression, sequential genetic profiling revealed additional somatic point mutations in ERBB3 (p.A232V) and PIK3CA (p.G106V) as well as MET and CDK6 amplification. ERBB3A232V is a missense, hot-spot mutation within the extracellular domain, causing anchorage-independent growth and signaling when HER2 kinase activity is present [3]. Accordingly, in the presence of ERBB2S310F, additional somatic ERBB3A232V may have caused a compensatory mechanism of resistance to ERBB2 targeted therapy. Indeed, ERBB3 mutations have been shown to cause resistance to ERBB2 targeted therapy [3]. PIK3CAG106V is a missense, activating hot-spot mutation within the adaptor-binding domain of the catalytic subunit of the phosphoinositide 3-kinase, stimulating its lipid kinase activity [8]. Activation of downstream signaling pathways are a known mechanism of secondary resistance and mutations in PIK3CA are associated with resistance to trastuzumab [9]. In addition, both MET and CDK6 amplification have been described to contribute to trastuzumab resistance in HER2-overexpressing breast cancer [10, 11].
This is the first description of acquired somatic alterations occurring after secondary treatment resistance in a patient with non-amplified, ERBB2 mutated, metastatic EMPD. Interestingly, these acquired genetic alterations may have caused treatment resistance and contribute to the understanding of commonly occurring secondary treatment failure of anti-HER2 treatments in metastatic EMPD. Furthermore, this case provides rationale for repetitive genomic analysis in treatment resistant EMPD lesions and contributes to the growing evidence of ERBB2 in the pathogenesis of metastatic extramammary Paget’s disease.
Materials and Methods
DNA from FFPE tumor tissue was isolated using the automated Maxwell isolation system (Promega). Next generation sequencing was performed on formalin-fixed paraffin embedded (FFPE) samples, using the Oncomine Focus Assay (Life Technologies / Thermo Fisher) and the FoundationOne panel. Library preparation and sequencing was performed according to the manufacturers’ manuals.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
References
1. Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, et al; American Society of Clinical Oncology; College of American Pathologists. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol. 2013; 31: 3997–4013. https://doi.org/10.1200/JCO.2013.50.9984. [PubMed].
2. Jenni D, Karpova MB, Muhleisen B, Mangana J, Dreier J, Hafner J, Dummer R. A prospective clinical trial to assess lapatinib effects on cutaneous squamous cell carcinoma and actinic keratosis. ESMO Open. 2016; 1: e000003. https://doi.org/10.1136/esmoopen-2015-000003. [PubMed].
3. Jaiswal BS, Kljavin NM, Stawiski EW, Chan E, Parikh C, Durinck S, Chaudhuri S, Pujara K, Guillory J, Edgar KA, Janakiraman V, Scholz RP, Bowman KK, et al. Oncogenic ERBB3 mutations in human cancers. Cancer Cell. 2013; 23: 603–17. https://doi.org/10.1016/j.ccr.2013.04.012. [PubMed].
4. Mishra R, Hanker AB, Garrett JT. Genomic alterations of ERBB receptors in cancer: clinical implications. Oncotarget. 2017; 8: 114371–92. https://doi.org/10.18632/oncotarget.22825. [PubMed].
5. Tanaka R, Sasajima Y, Tsuda H, Namikawa K, Tsutsumida A, Otsuka F, Yamazaki N. Human epidermal growth factor receptor 2 protein overexpression and gene amplification in extramammary Paget disease. Br J Dermatol. 2013; 168: 1259–66. https://doi.org/10.1111/bjd.12249. [PubMed].
6. Chmielecki J, Ross JS, Wang K, Frampton GM, Palmer GA, Ali SM, Palma N, Morosini D, Miller VA, Yelensky R, Lipson D, Stephens PJ. Oncogenic alterations in ERBB2/HER2 represent potential therapeutic targets across tumors from diverse anatomic sites of origin. Oncologist. 2015; 20: 7–12. https://doi.org/10.1634/theoncologist.2014-0234. [PubMed].
7. Vornicova O, Hershkovitz D, Yablonski-Peretz T, Ben-Itzhak O, Keidar Z, Bar-Sela G. Treatment of metastatic extramammary Paget's disease associated with adnexal adenocarcinoma, with anti-HER2 drugs based on genomic alteration ERBB2 S310F. Oncologist. 2014; 19: 1006–7. https://doi.org/10.1634/theoncologist.2014-0054. [PubMed].
8. Burke JE, Perisic O, Masson GR, Vadas O, Williams RL. Oncogenic mutations mimic and enhance dynamic events in the natural activation of phosphoinositide 3-kinase p110alpha (PIK3CA). Proc Natl Acad Sci U S A. 2012; 109: 15259–64. https://doi.org/10.1073/pnas.1205508109. [PubMed].
9. Shi W, Jiang T, Nuciforo P, Hatzis C, Holmes E, Harbeck N, Sotiriou C, Peña L, Loi S, Rosa DD, Chia S, Wardley A, Ueno T, et al. Pathway level alterations rather than mutations in single genes predict response to HER2-targeted therapies in the neo-ALTTO trial. Ann Oncol. 2017; 28: 128–35. https://doi.org/10.1093/annonc/mdw434. [PubMed].
10. Minuti G, Cappuzzo F, Duchnowska R, Jassem J, Fabi A, O'Brien T, Mendoza AD, Landi L, Biernat W, Czartoryska-Arlukowicz B, Jankowski T, Zuziak D, Zok J, et al. Increased MET and HGF gene copy numbers are associated with trastuzumab failure in HER2-positive metastatic breast cancer. Br J Cancer. 2012; 107: 793–9. https://doi.org/10.1038/bjc.2012.335. [PubMed].
11. Goel S, Wang Q, Watt AC, Tolaney SM, Dillon DA, Li W, Ramm S, Palmer AC, Yuzugullu H, Varadan V, Tuck D, Harris LN, Wong KK, et al. Overcoming Therapeutic Resistance in HER2-Positive Breast Cancers with CDK4/6 Inhibitors. Cancer Cell. 2016; 29: 255–69. https://doi.org/10.1016/j.ccell.2016.02.006. [PubMed].

