INTRODUCTION
Prostate cancer (PCa) is the most commonly diagnosed cancer in men in the United States, with an estimated 238,599 new cases and 29,720 deaths in the year 2013 [1]. Its global incidence is on the rise, especially in Asian countries [2, 3]. Once PCa invades local organs or spreads distantly, only palliative treatments can be offered. Patients with early stage PCa can survive more than 10 years after diagnosis. Thus, to reduce PCa-related mortality, efforts are being devoted to increase the detection rate of PCa at an early stage. The wide use of prostate-specific antigen (PSA) has greatly improved the early diagnosis of clinically localized PCa [4], resulting in a significant decrease in PCa-specific death [5]. However, as PSA is organ-specific rather than tumor-specific, elevated PSA is also associated with other conditions, such as prostatitis, benign prostatic hyperplasia and recent ejaculation [6]. Nevertheless, men with elevated serum PSA are recommended to undergo a biopsy for a definitive diagnosis. However, PSA has a low specificity of 25–40% in the so-called “diagnostic grey zone” (PSA 4–10 ng/ml), which has also resulted in many unnecessary biopsies and biopsy-related financial, social and psychological burdens. Therefore, there is an urgent need to develop more sensitive, specific biomarkers to diagnose PCa at an early stage while avoiding unnecessary biopsies. A wide range of promising PCa biomarkers has been reported, including CpG hypermethylation of GSTP1 [7], TMPRSS2:ERG gene fusion [8], AMACR [9], sarcosine [10], and the long noncoding RNA urine biomarker prostate cancer gene 3 (PCA3) [11]. Currently, PCA3 is the most extensively studied urine biomarker. PCA3 is a prostate-specific non-coding RNA that is highly overexpressed in PCa compared to the normal prostate [12, 13]. As prostate cells can be detected in the urine of men after a digital rectal examination (DRE), urine-based diagnostic tests have the advantage of being non-invasive or minimally invasive. The Progensa PCA3 test has been approved by the US Food and Drug Administration and is commercially available to guide repeat biopsy decision-making.
Metastasis-associated lung adenocarcinoma transcript 1 (MALAT-1) has been described as a regulator of metastasis and motility, and its expression is associated with metastasis in non-small cell lung cancer. It is a noncoding RNA of more than 8,000 nt derived from chromosome 11q13 [14]. MALAT-1 is overexpressed in multiple types of human malignancies, including hepatocellular cell carcinoma, lung adenocarcinoma, endometrial stromal sarcoma, and colorectal cancer [15-18]. Using RNA-Seq and quantitative RT-PCR, we found that MALAT-1 is upregulated in prostate cancer tissues compared with adjacent normal tissues and benign prostatic hyperplasia (BPH) tissues [19]. Higher MALAT-1 expression is correlated with aggressive characteristics of PCa in prostate cancer tissue. MALAT-1 silencing dramatically inhibited PCa cell growth, invasion and migration and induced cell cycle arrest in vitro and in vivo. Furthermore, we showed that a circulating MALAT-1 fragment (MD-miniRNA) outperformed PSA in predicting prostate biopsy outcomes, suggesting that MALAT-1 may be a promising biomarker for diagnosing PCa.
In this study, we investigated the potential diagnostic efficacy of urinary MALAT-1 transcript in a retrospective discovery phase (n=218) and a prospective multicenter cohort (n=216). We also evaluated the potential of the MALAT-1 score and the MALAT-1 model for PCa diagnosis.
RESULTS
Initially, 536 patients were included in this study, from which 29 samples were excluded for insufficient RNA extraction from the sediments. After quantitative RT-PCR analysis, another 73 patients were excluded, as their PSA Ct value was above 28 [20], indicating insufficient prostate cell collection. Therefore, 434 patients (218 patients in the discovery phase and 216 patients in the validation phase) were finally recruited in this study. The clinical and pathological data are listed in Table 1. PCa-associated risk factors (age, total PSA (tPSA), volume, percent free PSA (%fPSA) and DRE) were all significantly higher in patients with a positive biopsy compared with those with a negative biopsy in all the patients in the discovery phase. However, tPSA failed to discriminate positive biopsy results from negative biopsy results in the PSA 4-10 ng/ml cohort. These results were confirmed in the validation phase.

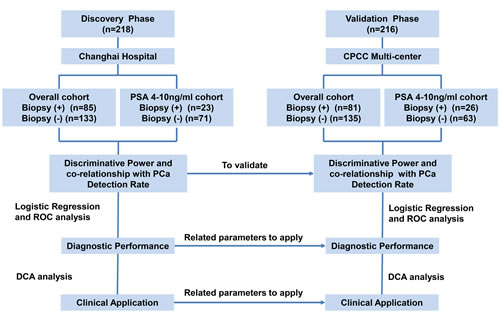
Figure 1: Study design. CPCC=Chinese Prostate Cancer Consortium; PSA=prostate-specific antigen; ROC=receiver operating characteristic curve; DCA=decision curve analysis.
MALAT-1 score sufficed to differentiate positive from negative prostate biopsy results and strongly correlated with the PCa detection rate
To explore whether the MALAT-1 score could be a useful biomarker to diagnose PCa in all patients and in the PSA 4-10 ng/ml cohort (which is of special clinical interest), the MALAT-1 score was tested in the discovery phase and evaluated in the validation phase. Our results revealed that the MALAT-1 score was significantly higher in positive biopsy patients in both the overall (Figure 2A) and the PSA 4-10 ng/ml cohorts (Figure 2B). The results were confirmed in the validation phase (Figure 2C for the overall cohort and Figure 2D for the PSA 4-10 ng/ml cohort).
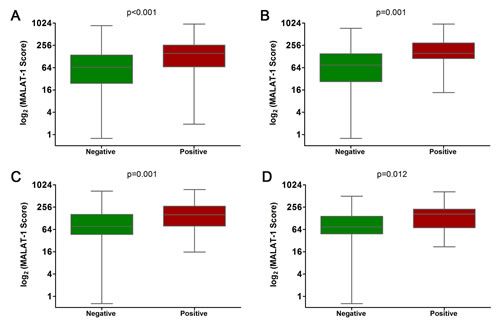
Figure 2: Comparison of MALAT-1 score between positive and negative biopsies in the discovery and validation phases. (A) Overall cohort in the discovery phase. (B) PSA 4-10 ng/ml cohort in the discovery phase. (C) Overall cohort in the validation phase. (D) PSA 4-10 ng/ml cohort in the validation phase.
Correlation analysis demonstrated that the MALAT-1 score did not correlate with other risk factors or the Gleason score (Supplemental Table 1). In addition, we evaluated the discriminative power of the MALAT-1 score in the overall cohort as well as in subsets of patients based on their PSA levels. In the overall cohort, the detection rate of PCa increased significantly with the MALAT-1 score: 24.1%, 27.3%, 42.6%, and 61.8% for men with low (0-25th percentile), intermediate (25th-50th percentile), high (50th-75th percentile), and very high (75th-100th percentile) MALAT-1 scores, respectively (Figure 3A, p<0.001). The association of increasing PCa detection rates with a higher MALAT-1 score was consistently observed in the PSA 4-10 ng/ml cohort (Figure 3A, p=0.001). In the validation phase, the same results were obtained and are shown in Figure 3B. The detection rate of PCa increased with the MALAT-1 score in the PSA >10 ng/ml cohort, but the increments did not meet significance in the discovery (p=0.085 for PSA 10-20 ng/ml and p=0.965 for PSA >20 ng/ml) or validation phases (p=0.117 for PSA 10-20 ng/ml and p=0.299 for PSA >20 ng/ml). Therefore, our results demonstrated that the MALAT-1 score was strongly correlated with prostate cancer risk in the overall group as well as in the subgroup with PSA=4-10 ng/ml and may serve as a noninvasive biomarker for the detection of PCa in urine.
Figure 3: Prostate cancer detection rate in subjects with low (green), intermediate (blue), high (purple) and very high (red) MALAT-1 scores in the overall cohort and in subgroups based on PSA levels. (A) Discovery phase. (B) Validation phase. §Pearson chi-square. ТFisher’s exact test. PSA=prostate-specific antigen.
Logistic regression evaluation of diagnosis performance of the MALAT-1 score in the discovery phase and applied in the validation phase
In the discovery phase, in univariable and multivariable logistic regression models, MALAT-1 score, age, tPSA, volume, %fPSA and DRE were independent risk factors in the overall cohort, while tPSA was excluded in the PSA grey zone cohort (Tables 2 and 3, Supplemental Figure 1). In univariable logistic regression models, the MALAT-1 score in the overall cohort represented a comparable informative parameter in the prediction of PCa (AUC: 0.688; Figure 4A) to serum PSA (AUC: 0.721) and was superior to serum PSA in the PSA grey zone cohort (MALAT-1 score AUC: 0.742; tPSA AUC: 0.545; Figure 4C) (Supplemental Table 2). There was a trend in the overall and PSA grey zone cohorts that the MALAT-1 score was superior to %fPSA (AUC: 0.639, 0.622), but it was not statistically significant. Each model’s predictive accuracy (PA) and the AUC are displayed in Table 3. The MALAT-1 score-based model demonstrated a higher AUC of 0.840 and PA of 77.20% in the prediction of PCa and resulted in an increased AUC of 0.0167 and increased PA of 2.59% in the overall cohort (Supplemental Figure 2). In the PSA grey zone cohort, the MALAT-1 score-based model demonstrated a higher AUC of 0.853 and PA of 79.79% in the prediction of PCa and resulted in an increased AUC of 0.0318 and increased PA of 5.32%.
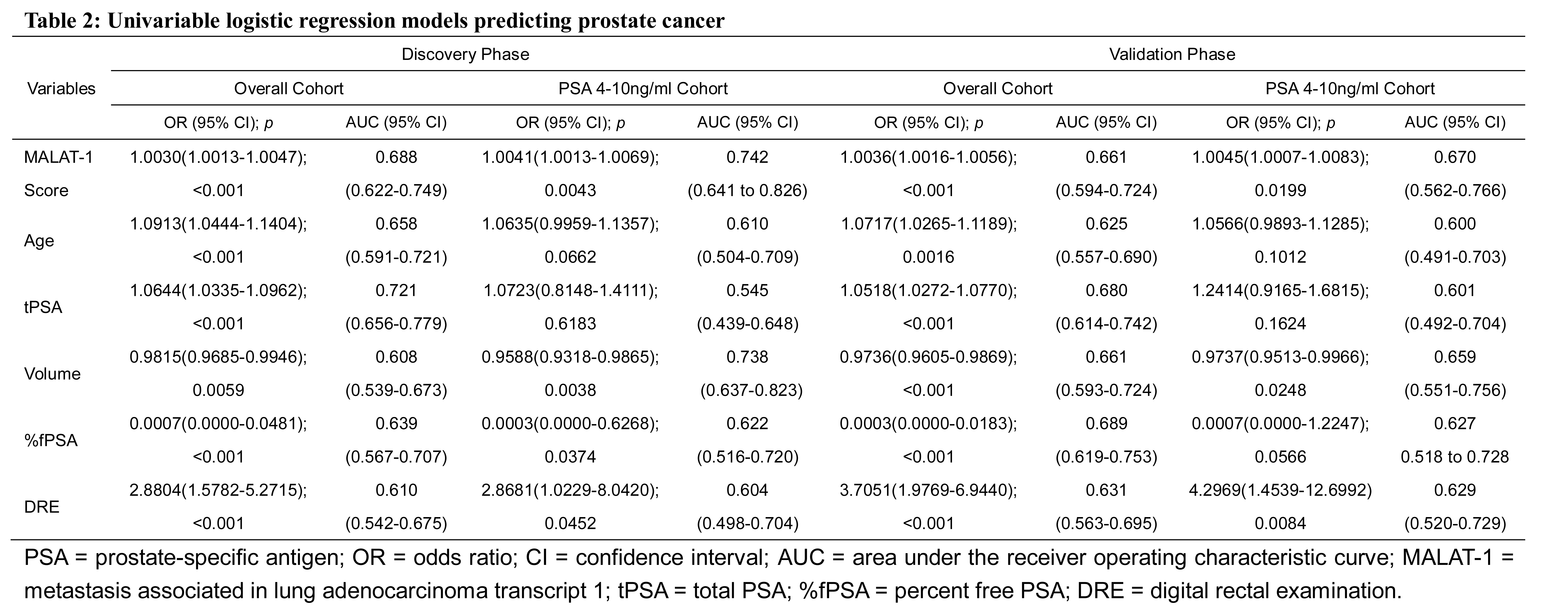

Figure 4: Receiver operating characteristic curve analysis for evaluating the diagnostic performance of the MALAT-1 score. Area under the curve (AUC) estimation for the MALAT-1 score in the overall cohort in the (A) discovery phase and (B) validation phase. Comparison of the diagnostic performance of the MALAT-1 score, tPSA and %fPSA in the PSA 4-10 ng/ml cohort in the (C) discovery phase and (D) validation phase.
The parameters estimated from the discovery phase were evaluated in the independent validation phase. In the univariable and multivariable logistic regression models, comparable independent risk factors were obtained in the overall and PSA grey zone cohorts. There was also a trend in the overall and PSA grey zone cohorts that the MALAT-1 score (AUC: 0.661; Figure 4B) was comparable to serum tPSA (AUC: 0.680) and %fPSA (AUC: 0.689), and the MALAT-1 score (AUC: 0.670) was superior to serum tPSA (AUC: 0.601) and %fPSA (AUC: 0.627) in the PSA grey zone (Figure 4D) (Table 2). Applying this model to the validation models, the MALAT-1 score-based model demonstrated a higher AUC of 0.833 and PA of 80.00% in the prediction of PCa and resulted in an increment AUC of 0.0156 and increase of PA of 2.56% in the overall cohort. In the PSA grey zone cohort, the MALAT-1 score-based model demonstrated a higher AUC of 0.799 and PA of 76.40% in the prediction of PCa and resulted in an increased AUC of 0.0269 and increased PA of 4.49% (Table 3).
Decision curve analysis evaluation of the accuracy of the diagnostic model with and without MALAT-1
As the diagnosis of prostate cancer in the PSA grey zone is of particular interest, the decision curve analysis focused on this group of patients, while the DCAs for the overall cohorts are reported in the supplemental data (Supplemental Figure 3 and Supplemental Tables 3, 4).
As the decision curve indicates, in the grey zone cohort discovery phase, the base model plus MALAT-1 was superior to the base model, with a higher net benefit for almost all threshold probabilities >10% (Figure 5A, Table 4). For the patients with PSA values of 4.0-10.0 ng/ml in the validation phase, the base model plus MALT-1 was better than the base model for all threshold probabilities >22% (Figure 5B).
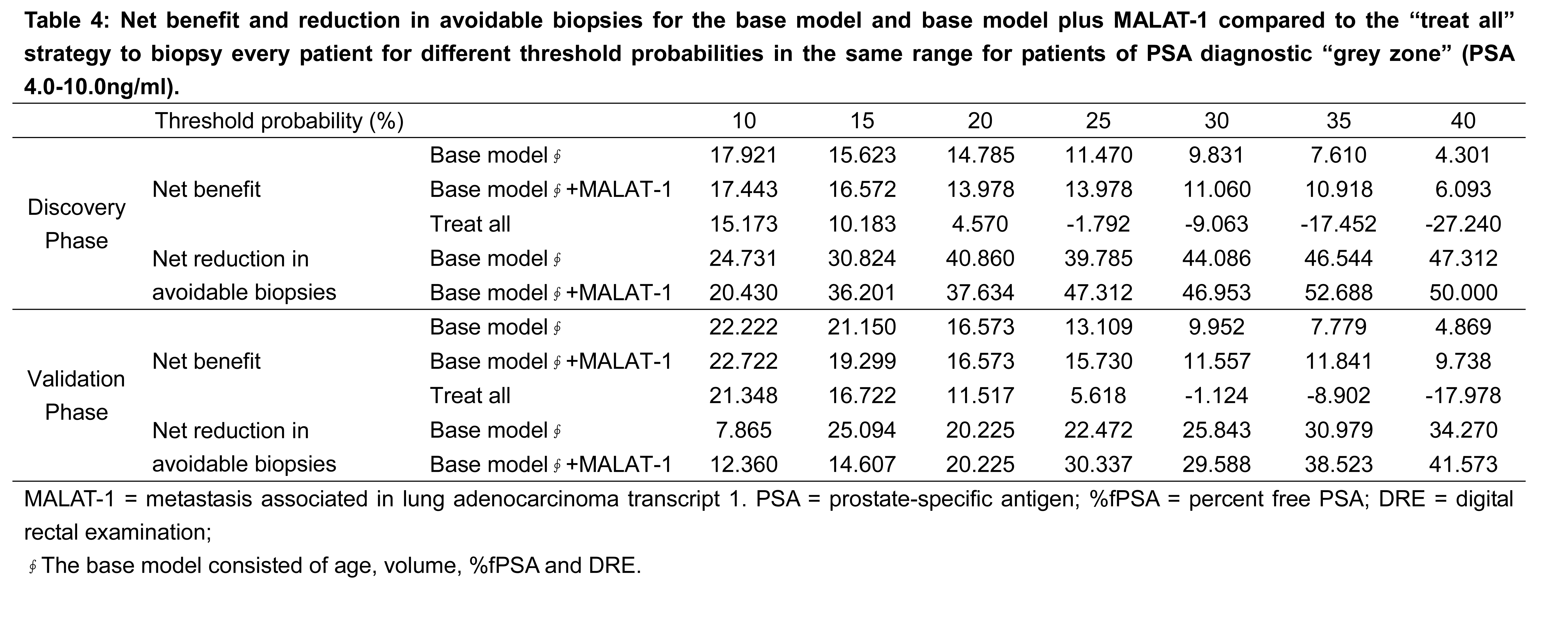
In the grey zone cohort of the discovery phase, the base model plus MALAT-1 detected more cancer than the base model at a threshold of 25% (13.97% vs. 11.47%) (Table 4). At this threshold, the base model plus MALAT-1 also prevented more unnecessary biopsies than the base model (47.32% vs. 39.78%) (Table 5). At the threshold of 25%, the base model with MALAT-1 could spare 8.5% more biopsies (46.5% vs. 38.0%), both at the risk of missing only one (4.3%) cancer patient in the discovery phase.
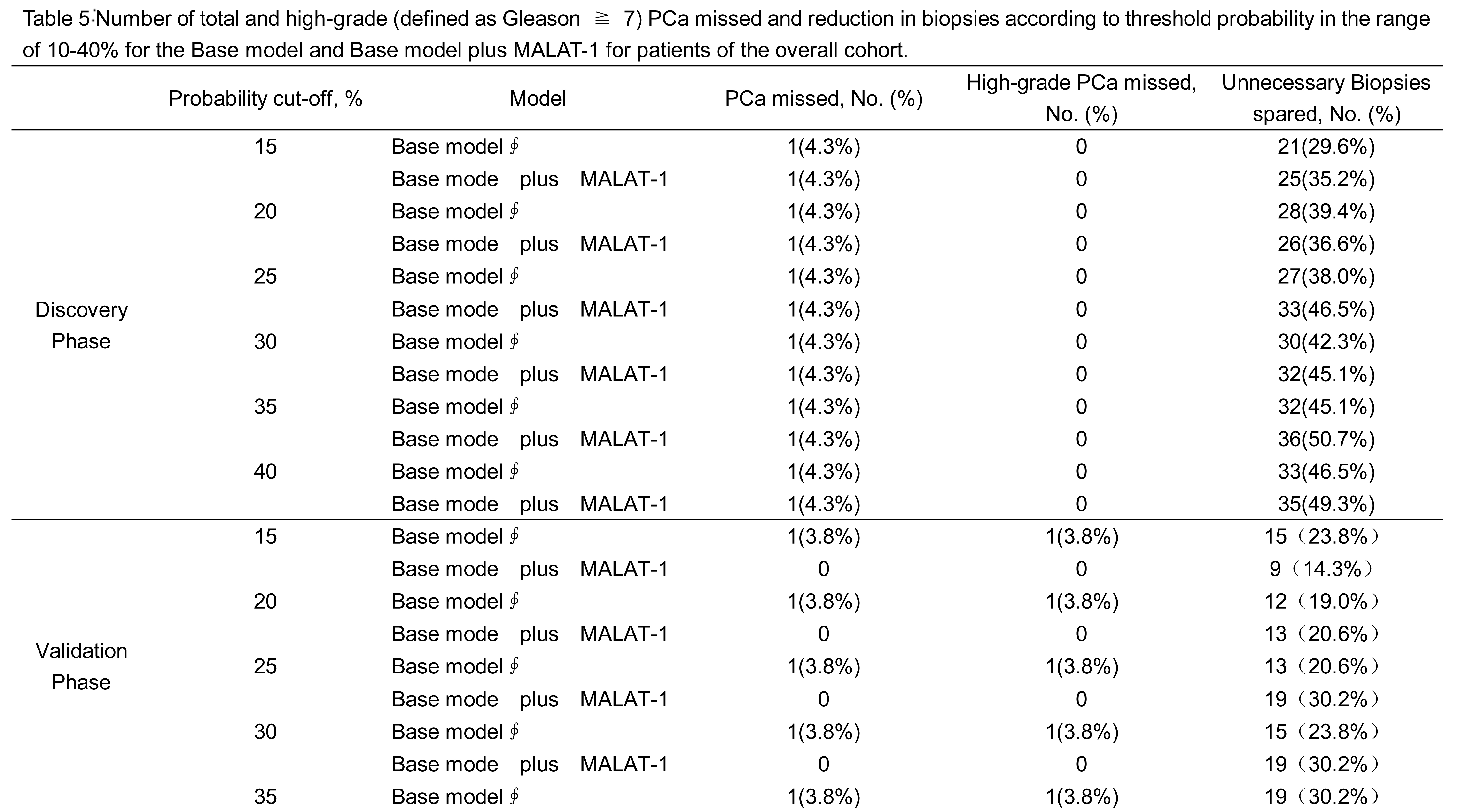

In the validation phase, when the same threshold of 25% was applied, the base model plus MALAT-1 could also detect more cancers (15.73% vs. 13.11%) and prevented more unnecessary biopsies than the base model (30.33% vs. 22.47%). At the same threshold, the MALAT-1 based model could spare more unnecessary biopsies (30.2% vs. 20.6%) while missing fewer cancer patients (0 vs. 3.8%).
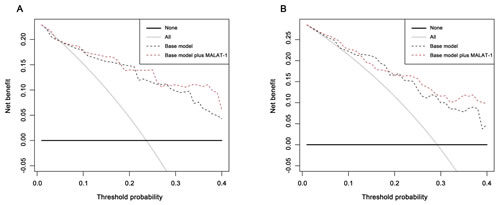
Figure 5: Decision curve analysis for positive biopsy prediction in the PSA 4-10 ng/ml cohorts by the base model (base model contains age, volume, %fPSA and DRE) in the (A) discovery phase and (B) validation phase. The dashed black line indicates the base model that contains age, volume, %fPSA and DRE; the solid black line shows the prediction model that includes only age, PSA level, DRE result, and prostate volume. The horizontal line along the x-axis assumes that no patient will have PCa (no patient should undergo a prostate biopsy), whereas the solid gray line assumes that all patients will have PCa (all patients will need to undergo a prostate biopsy).
Discussion
The current standard approach to diagnose PCa is prostate biopsy, which is mostly based on elevated PSA and/or abnormal DRE. Although the introduction of PSA has greatly improved early PCa detection and stage migration as well as reduced PCa mortality [4], PSA has low specificity in discriminating between benign and malignant prostatic diseases, leading to unnecessary and/or repeat biopsies [21]. As a result, the expansion of PSA screening has led to intense debate as to overdiagnosis and, ultimately, overtreatment of low-risk patients. A recent report from the European Randomized Study of Screening for Prostate Cancer (ERSPC) and Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial (PLCO) indicates a continued need for novel biomarkers to supplement serum PSA to accurately predict PCa risk and stratification of indolent and aggressive PCa [22, 23].
Although the incidence of PCa in China is reported to be 1.6/100,000 per year, its incidence has recently increased substantially, with an estimated incidence of 10/100,000 in 2010 compared with 1.71/100,000 in 1993 [24, 25]. With the implementation of more comprehensive screening programs in the developing cities and regions of China, the incidence rate may rapidly increase in the future, as evidenced by the fact that PCa ranks first among all types of urologic tumors and reaches 31.2/100,000 in Shanghai (2012) [26]. However, mass screening of PCa is not performed nationwide in China, especially in rural China, which results in a substantial portion of men who are diagnosed at an advanced stage with a high PSA level [27]. In our study, although the urine samples for the discovery and validation phases were collected from major hospitals in developed cities in China, the patients came from a variety of regions in China, including rural areas. As shown in Table 1, the PSA distribution of men who underwent prostate biopsy in both cohorts is dramatically different from those of western populations, with over 50% of men with a PSA>10 ng/ml. In contrast, with the implementation of PSA screening, over 90% of men referred for biopsy in western populations have PSA levels below 10 ng/ml. Apparently, the higher the PSA level is, the higher the risk of PCa. PSA is a good predictor when its level is >10 ng/ml. We compared the detection rate of prostate cancer in patients with different MALAT-1 scores in the PSA>10 ng/ml cohort. However, the differences between each group failed to meet the conventional levels of statistical significance. AUC-ROC analysis revealed that the diagnostic power of the MALAT-1 score (AUC in the discovery phase: 0.662; AUC in the validation phase: 0.640) was not superior to that of PSA (AUC in the discovery phase: 0.754; AUC in the validation phase: 0.785) in the PSA>10 ng/ml cohort in both the discovery (p=0.144, Z-test; Supplemental Figure 4A) and validation phases (p=0.022, Z-test; Supplemental Figure 4B). Thus, we particularly focused on evaluating the performance of the MALAT-1 score in patients with grey zone PSA, which is of great clinical interest.
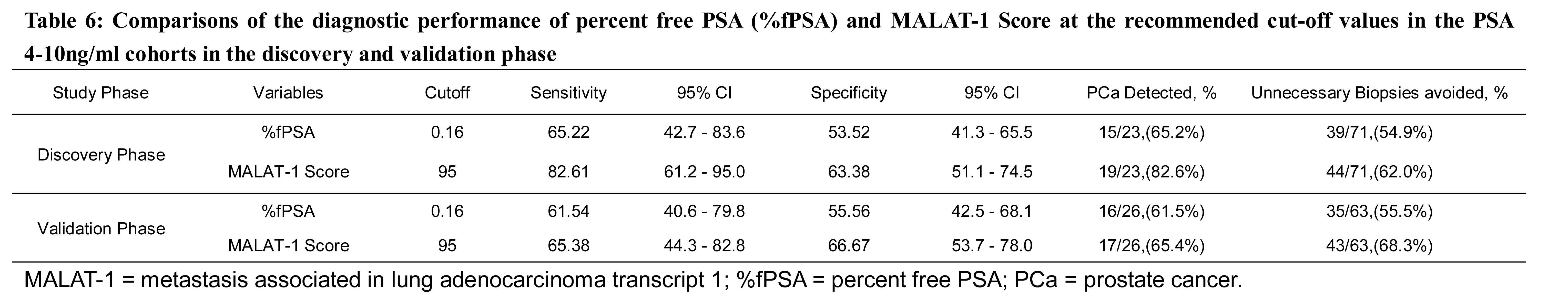
Multiple derivatives, including the ratio of the free-to-total (f/t) PSA, PSA density or PSA velocity, have been developed to improve the diagnostic accuracy of PSA. %fPSA is most widely used in clinical settings to stratify the risk of PCa for men who have total PSA levels between 4 and 10 ng/ml [28]. In our data set, the diagnostic accuracy of the MALAT-1 score outperformed %fPSA. Applying a MALAT-1 cut-off value of 95 achieved better sensitivity and specificity than the recommended %fPSA cut-off of 0.16 in grey zone cohorts in both the discovery and validation phases (Table 6). Using a MALAT-1 score cut-off of 95 rather than the 0.16 cut-off of %fPSA could detect more cancers (19/23 vs. 15/23 in the discovery phase; 17/26 vs. 16/26 in the validation phase) and, at the same time, reduce unnecessary biopsies (44/71 vs. 39/71 in the discovery phase; 43/63 vs. 35/63 in the validation phase). Collectively, the MALAT-1 score may be more accurate than %fPSA, the most widely used parameter for PSA grey zone patients.
Extensive efforts have been devoted to identify novel biomarkers to diagnose PCa more accurately and at early stages to predict tumor recurrence and progression. Numerous emerging biomarkers for PCa have been discovered and tested by clinicians recently, most notably, the urinary markers PCA3 and TMPRSS2:ERG [29, 30]. The Progensa PCA3 test was approved by the US Food and Drug Administration in 2012 to aid in the decision of taking repeat prostate biopsies when the patient had one or more negative biopsies previously. The consensus in most publications is that PCA3 can help distinguish between individuals with and without prostate cancer. A high PCA3 score has a high Gleason score and indicates clinically significant cancers [31]. The performance of PCA3 in Chinese populations has not been evaluated. On the basis of prior reports in Caucasian populations, the AUC of PCA3 in patients with grey zone PSA ranges from 0.64-0.69 [32], which is comparable with the AUC of the MALAT-1 score (0.67-0.74) in our study. However, the limitations of PCA3 include the lack of an appropriate cut-off level for clinical practice, and false negative results may miss some PCa, especially aggressive tumors.
However, several limitations to this study must be acknowledged. First, the sample size was limited in both cohorts, although the initial findings were validated in a prospective multicenter cohort. Second, we did not perform a head-to-head comparison of the MALAT-1 score and the PCA3 score in our study, due to the lack of a commercial PCA3 kit in China. Third, we did not compare the performance of the MALAT-1 score with clinical nomograms (e.g., Prostate Cancer Prevention Trial risk calculator-PCPT) to assess the ability of the MALAT-1 score to increase the AUC of the PCPT risk calculator for predicting PCa diagnosis on biopsy. Fourth, we did not look at the value of MALAT-1 in subgroups of patients classified by initial and repeat biopsy. Future large-scale studies are needed to confirm the efficacy of the MALAT-1 score in the diagnosis of prostate cancer.
In conclusion, we have clearly demonstrated that the MALAT-1 score could serve as a noninvasive biomarker for detecting PCa, especially in the PSA 4-10 ng/ml cohort. Applying a probability threshold of 25%, the MALAT-1-based model would prevent 30.2%-46.5% of unnecessary biopsies in the PSA 4–10 ng/ml cohort, while no high-grade cancers would be missed. This study is limited by the small sample size. Further large-scale studies are needed to confirm our findings.
METHODS
Patients and clinical specimens
This multicenter Chinese study was approved by the institutional review board of each participating hospital. Informed consent was obtained from all patients. Subjects were recruited between May 2012 and December 2013 at three urology centers in China (Changhai Hospital, Shanghai; Changzheng Hospital, Shanghai; and West China Hospital, Sichuan). All urine samples were collected from patients scheduled for prostate biopsy because of elevated serum PSA levels (≥4 ng/ml) and/or suspicious DRE. Patients with other known tumors and those receiving medical therapy known to affect serum PSA levels and/or previous PCa therapies were excluded from the study.
Study design
Our study was divided into two phases (Figure 1): (1) Discovery phase. The MALAT-1 score was tested in an independent cohort of urine sediment samples from 218 consecutive patients, including 85 PCa patients (positive prostate biopsies) and 133 patients with negative prostate biopsies in Shanghai Changhai Hospital who underwent a prostate biopsy. The discriminative power of the MALAT-1 score and its association with the PCa detection rate were evaluated. Then, the diagnostic performance and clinical value of the MALAT-1 score were assessed in the overall cohort and in the grey zone cohort (PSA 4-10 ng/ml) samples. (2) Validation phase. The association of the MALAT-1 score and other parameters with the PCa detection rate was validated in 216 samples (81 positive prostate biopsy and 135 negative prostate biopsies) from the Chinese Prostate Cancer Consortium (CPCC) multicenter samples. Then, the diagnostic accuracy of the MALAT-1 score and other parameters was validated.
Specimen collection and sample preparation
First-catch urine samples were collected following an attentive DRE (three strokes per lobe) before the biopsy was performed. The urine samples were immediately cooled on ice and were processed within two hours of collection. Biopsies were performed using an end-fire ultrasound transducer (Falcon 2101; B-K Medical, Inc.) and an automatic 18-gauge needle (Bard, Inc.). In all subjects, a 10-12 core systematic laterally directed transrectal ultrasound (TRUS)–guided biopsy was performed.
The urine samples were centrifuged at 2,500 x g for 15 min at 4°C and the pellets were washed twice with cold PBS (1×). The sediments were then homogenized in TRIzol reagent and were used for RNA extraction or stored at -80°C until further use.
Quantitative RT-PCR analysis
Total RNA was extracted from the urine sediments using TRIzol reagent (Invitrogen: No 15596-026, USA). Then, 50 ng of total RNA was treated with DNase I (TaKaRa: D2215, TaKaRa, Japan) prior to cDNA synthesis and was amplified using the TransPlex Complete Whole Transcriptome Amplification Kit (WTA2, Sigma-Aldrich, St. Louis, MO, USA) according to the manufacturer’s instructions. qRT-PCR was performed using SYBR® Premix Ex Taq™ (Perfect Real Time) (Takara: DRR081A TaKaRa, Japan) with an Applied Biosystems Step One Plus according to the manufacturer’s recommended cycling conditions. The gene-specific sequence information for the qRT-PCR primers is listed as follows: PSAKIT-forward primer GTCTGCGGCGGTGTTCTG, PSAKIT-reverse primer TGCCGACCCAGCAAGATC; MALAT-1 forward primer CTTCCCTAGGGGATTTCAGG, MALAT-1 reverse primer GCCCACAGGAACAAGTCCTA. Briefly, 2 µl of the cDNA solution was amplified using 10 µl of SYBR® Premix Ex Taq™ (Perfect Real Time) (2×) (Takara: DRR081A TaKaRa, Japan), 2 µl of primers, 0.4 µl of ROX Reference Dye (50×) and nuclease-free H2O at a final volume of 20 µl. The data were analyzed with StepOne Software version v2.1 (Applied BioSystems, USA). A melt-curve analysis was enabled at the end of the amplification. Samples with PSA Ct values of >28 [20] were excluded to ensure sufficient prostate cell collection. The MALAT-1 score was calculated as MALAT-1 mRNA/PSA mRNA×1000=2Ct(PSA)-Ct(MALAT-1)×1000. All experiments were performed in triplicate. No amplification of the signal was obtained when nuclease-free water was added instead of cDNA. The data were analyzed using StepOne Software version v2.1 (Applied BioSystems, USA).
Statistical analysis
The Mann-Whitney U-test, Student’s t-test, Pearson’s chi-square test and Fisher’s exact test were used for statistical comparisons of continuous and categorical variables as appropriate. Univariate and multivariate logistic regressions were used to identify independent predictors of PCa upon biopsy. Co-relationships between MALAT-1 and the clinical variables were assessed by the Spearman rank correlation test. Receiver operating characteristic (ROC) curves were constructed to discriminate among different groups of patients. The area under the ROC curve (AUC) was used to assess the predictive power. The sensitivity and specificity were calculated according to the standard formulas. Decision curve analysis was used to evaluate the clinical effects of the calculators.
All of the p values were two-sided, and p<0.05 was considered to be statistically significant. All of the statistical calculations were performed using SPSS (Statistical Package for the Social Sciences) v.17.0 (SPSS Inc., Chicago, IL, USA), MedCalc v.10.4.7.0 (MedCalc Software bvba, Mariakerke, Belgium) and R software v.3.1.1 (The R Foundation for Statistical Computing).
Author contributions
Y.S., S.R., and F.W. designed the experiments. R.C, F.W., J.L., X.S., Y.Z., W.Z., T.J, C.Z., J.S., H.W., H.W., Y.W, B.L., Y.L., Z.F., F.G., Q.W., D.X., D.S. and X.L. collected and prepared the clinical samples. F.W., R.C, J.L., X.S., M.Q, Y.Z., W.Z., T.J, C.Z., C.W. and J.S. performed the qRT-PCR. F.W., R.C., X.S, and Y.Z analyzed the data. Y.S., F.W., S.R., R.C, X.G. D.X, Q.W., C.X. and J.H. drafted and revised the manuscript. All authors read and approved the final version of the manuscript.
Funding
This work was supported by the Program for Changjiang Scholars and Innovative Research Team in University scheme of the Ministry of Education of China (NO.IRT1111, Yinghao Sun), the National Basic Research Program of China (2012CB518300, 2012CB518306, Yinghao Sun), the National Natural Science Foundation of China (81430058 for Yinghao Sun, 81101946, 81472397 for Shancheng Ren, 81172447 for Bing Liu), the Shanghai Pujiang Program (12PJD008, Shancheng Ren), Prostate Cancer Foundation Young Investigator Award (Shancheng Ren), Shanghai Municipal Health and Family Planning Commission Outstanding Young Investigator (XYQ2013077, Shancheng Ren), Shanghai Municipal Education Commission (Shancheng Ren), and the Shanghai Natural Science Foundation (11ZR1447800, Bing Liu).
References
1. Siegel R, Naishadham D and Jemal A. Cancer statistics, 2013. CA: a cancer journal for clinicians. 2013; 63(1):11-30.
2. Jemal A, Bray F, Center MM, Ferlay J, Ward E and Forman D. Global cancer statistics. CA: a cancer journal for clinicians. 2011; 61(2):69-90.
3. Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O and Bray F. International variation in prostate cancer incidence and mortality rates. European urology. 2012; 61(6):1079-1092.
4. Catalona WJ, Smith DS, Ratliff TL, Dodds KM, Coplen DE, Yuan JJ, Petros JA and Andriole GL. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. The New England journal of medicine. 1991; 324(17):1156-1161.
5. Stamey TA, Caldwell M, McNeal JE, Nolley R, Hemenez M and Downs J. The prostate specific antigen era in the United States is over for prostate cancer: what happened in the last 20 years? J Urol. 2004; 172(4 Pt 1):1297-1301.
6. Loeb S, Gashti SN and Catalona WJ. Exclusion of inflammation in the differential diagnosis of an elevated prostate-specific antigen (PSA). Urologic oncology. 2009; 27(1):64-66.
7. Chen R, Ren S, Meng T, Aguilar J and Sun Y. Impact of glutathione-S-transferases (GST) polymorphisms and hypermethylation of relevant genes on risk of prostate cancer biochemical recurrence: a meta-analysis. PloS one. 2013; 8(9):e74775.
8. Saramaki OR, Harjula AE, Martikainen PM, Vessella RL, Tammela TL and Visakorpi T. TMPRSS2:ERG fusion identifies a subgroup of prostate cancers with a favorable prognosis. Clinical cancer research : an official journal of the American Association for Cancer Research. 2008; 14(11):3395-3400.
9. Zielie PJ, Mobley JA, Ebb RG, Jiang Z, Blute RD and Ho SM. A novel diagnostic test for prostate cancer emerges from the determination of alpha-methylacyl-coenzyme a racemase in prostatic secretions. The Journal of urology. 2004; 172(3):1130-1133.
10. Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009; 457(7231):910-914.
11. Marks LS and Bostwick DG. Prostate Cancer Specificity of PCA3 Gene Testing: Examples from Clinical Practice. Reviews in urology. 2008; 10(3):175-181.
12. Bussemakers MJ, van Bokhoven A, Verhaegh GW, Smit FP, Karthaus HF, Schalken JA, Debruyne FM, Ru N and Isaacs WB. DD3: a new prostate-specific gene, highly overexpressed in prostate cancer. Cancer research. 1999; 59(23):5975-5979.
13. Hessels D, Verhaegh GW, Schalken JA and Witjes JA. Applicability of biomarkers in the early diagnosis of prostate cancer. Expert Rev Mol Diagn. 2004; 4(4):513-526.
14. Ji P, Diederichs S, Wang W, Boing S, Metzger R, Schneider PM, Tidow N, Brandt B, Buerger H, Bulk E, Thomas M, Berdel WE, Serve H and Muller-Tidow C. MALAT-1, a novel noncoding RNA, and thymosin beta4 predict metastasis and survival in early-stage non-small cell lung cancer. Oncogene. 2003; 22(39):8031-8041.
15. Lai MC, Yang Z, Zhou L, Zhu QQ, Xie HY, Zhang F, Wu LM, Chen LM and Zheng SS. Long non-coding RNA MALAT-1 overexpression predicts tumor recurrence of hepatocellular carcinoma after liver transplantation. Med Oncol. 2011.
16. Tano K, Mizuno R, Okada T, Rakwal R, Shibato J, Masuo Y, Ijiri K and Akimitsu N. MALAT-1 enhances cell motility of lung adenocarcinoma cells by influencing the expression of motility-related genes. FEBS Lett. 2010; 584(22):4575-4580.
17. Yamada K, Kano J, Tsunoda H, Yoshikawa H, Okubo C, Ishiyama T and Noguchi M. Phenotypic characterization of endometrial stromal sarcoma of the uterus. Cancer Sci. 2006; 97(2):106-112.
18. Xu C, Yang M, Tian J, Wang X and Li Z. MALAT-1: a long non-coding RNA and its important 3’ end functional motif in colorectal cancer metastasis. Int J Oncol. 2011; 39(1):169-175.
19. Ren S, Peng Z, Mao JH, Yu Y, Yin C, Gao X, Cui Z, Zhang J, Yi K, Xu W, Chen C, Wang F, Guo X, Lu J, Yang J, Wei M, et al. RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings. Cell Res. 2012; 22(5):806-821.
20. Laxman B, Morris DS, Yu J, Siddiqui J, Cao J, Mehra R, Lonigro RJ, Tsodikov A, Wei JT, Tomlins SA and Chinnaiyan AM. A first-generation multiplex biomarker analysis of urine for the early detection of prostate cancer. Cancer research. 2008; 68(3):645-649.
21. Djavan B, Zlotta A, Remzi M, Ghawidel K, Basharkhah A, Schulman CC and Marberger M. Optimal predictors of prostate cancer on repeat prostate biopsy: a prospective study of 1,051 men. The Journal of urology. 2000; 163(4):1144-1148; discussion 1148-1149.
22. Andriole GL, Crawford ED, Grubb RL, 3rd, Buys SS, Chia D, Church TR, Fouad MN, Gelmann EP, Kvale PA, Reding DJ, Weissfeld JL, Yokochi LA, O’Brien B, Clapp JD, Rathmell JM, Riley TL, et al. Mortality results from a randomized prostate-cancer screening trial. The New England journal of medicine. 2009; 360(13):1310-1319.
23. Schroder FH, Hugosson J, Roobol MJ, Tammela TL, Ciatto S, Nelen V, Kwiatkowski M, Lujan M, Lilja H, Zappa M, Denis LJ, Recker F, Berenguer A, Maattanen L, Bangma CH, Aus G, et al. Screening and prostate-cancer mortality in a randomized European study. The New England journal of medicine. 2009; 360(13):1320-1328.
24. Jian L, Xie LP, Lee AH and Binns CW. Protective effect of green tea against prostate cancer: a case-control study in southeast China. International journal of cancer Journal international du cancer. 2004; 108(1):130-135.
25. Shao Q, Ouyang J, Fan Y, Xie J, Zhou J, Wu J, Karim Kader A, Xu J, Liu G, Shan Y, Wen D and Zhang Y. Prostate cancer in the senior men from rural areas in east district of China: contemporary management and 5-year outcomes at multi-institutional collaboration. Cancer letters. 2012; 315(2):170-177.
26. Control SCoD. Cancer incidence in Shanghai Urban area, 2009. TumorSci (in Chinese). 2012; 32(10):854.
27. Na R, Wu Y, Xu J, Jiang H and Ding Q. Age-specific prostate specific antigen cutoffs for guiding biopsy decision in chinese population. PloS one. 2013; 8(6):e67585.
28. Catalona WJ, Smith DS, Wolfert RL, Wang TJ, Rittenhouse HG, Ratliff TL and Nadler RB. Evaluation of percentage of free serum prostate-specific antigen to improve specificity of prostate cancer screening. JAMA : the journal of the American Medical Association. 1995; 274(15):1214-1220.
29. Hansen J, Auprich M, Ahyai SA, de la Taille A, van Poppel H, Marberger M, Stenzl A, Mulders PF, Huland H, Fisch M, Abbou CC, Schalken JA, Fradet Y, Marks LS, Ellis W, Partin AW, et al. Initial prostate biopsy: development and internal validation of a biopsy-specific nomogram based on the prostate cancer antigen 3 assay. European urology. 2013; 63(2):201-209.
30. Tomlins SA, Aubin SM, Siddiqui J, Lonigro RJ, Sefton-Miller L, Miick S, Williamsen S, Hodge P, Meinke J, Blase A, Penabella Y, Day JR, Varambally R, Han B, Wood D, Wang L, et al. Urine TMPRSS2:ERG fusion transcript stratifies prostate cancer risk in men with elevated serum PSA. Science translational medicine. 2011; 3(94):94ra72.
31. Chevli KK, Duff M, Walter P, Yu C, Capuder B, Elshafei A, Malczewski S, Kattan MW and Jones JS. Urinary PCA3 as a Predictor of Prostate Cancer in a Cohort of 3,073 Men Undergoing Initial Prostate Biopsy. The Journal of urology. 2013.
32. Auprich M, Bjartell A, Chun FK, de la Taille A, Freedland SJ, Haese A, Schalken J, Stenzl A, Tombal B and van der Poel H. Contemporary role of prostate cancer antigen 3 in the management of prostate cancer. European urology. 2011; 60(5):1045-1054.

