INTRODUCTION
Hepatic angiosarcoma (HAS) is a rare disease, with an annual incidence of approximately 200 cases worldwide [1]. It accounts for 2% of all primary malignancies of the liver and is the third most common type of liver cancer [2]. While exposure to vinyl chloride, arsenic, thorotrast, and radium have been identified as risk factors for the development of HAS, most cases arise in patients without exposure [1, 2]. Advanced HAS carries a poor prognosis, with 2 year overall survival (OS) of approximately 33%. This is related to presentation at advanced stages of disease, high rate of spontaneous tumor rupture resulting in intra-abdominal hemorrhage, and limited response to chemotherapy or radiation [2]. The standard treatment for patients with localized disease is complete surgical resection, often followed by adjuvant chemotherapy. While there is no universally accepted standard treatment in the metastatic setting, commonly used chemotherapies include doxorubicin, paclitaxel, or gemcitabine [3].
Although targeted therapies have revolutionized the treatment of other malignancies, such as non-small cell lung cancer (NSCLC) or breast cancer, there has been little progress towards applying these treatments to angiosarcoma. This is partially due to the limited knowledge regarding the landscape of genomic abnormalities from small cohorts of specimens that examined a select panel of genes. Frequent alterations of the RAS-RAF-MAPK pathway, TP53 and CDKN2A/p16 genes have been observed [4]. Increased expression of vascular endothelial growth factor (VEGF) and its receptor (VEGFR) is also common and supported clinical investigation of drugs that inhibit angiogenesis such as bevacizumab (a monoclonal antibody against VEGF-A) [5], sorafenib (tyrosine kinase inhibitor (TKI) that targets VEGFR and BRAF) [6, 7] and pazopanib (TKI that targets VEGFR, fibroblast growth factor receptor, and platelet derived growth factor) [8]. Tumor responses to these agents are seen in 10–25% of patients and progression-free survival (PFS) limited to 1–4 months.
Abnormalities in the RAS-RAF-MAPK pathway have been targeted with the mTOR (mammalian target of rapamycin) inhibitor everolimus. Two patients with angiosarcoma were treated with everolimus and both had a partial response (PR) lasting 6–12 months [9]. In a subgroup analysis of 3 patients with angiosarcoma enrolled in a multicenter phase II trial of everolimus for metastatic soft tissue sarcomas, one had PR and another stable disease (SD) [10]. Both of these patients had continued disease control for at least 16 weeks.
We describe the first case of HAS harboring a fusion of ROS1 with GOPC/FIG and review of the role of ROS1 rearrangements in cancer and evidence supporting the use of therapeutics that target ROS1.
CLINICAL CASE
A 50 year-old woman presented to her primary care physician for evaluation of a 2 week history of right upper quadrant abdominal pain and weight loss. CT of the abdomen and pelvis revealed multifocal hepatic disease and a dominant 7 cm lesion in the right lobe of the liver (Figure 1). CT scans of chest and brain showed no evidence of extrahepatic disease.
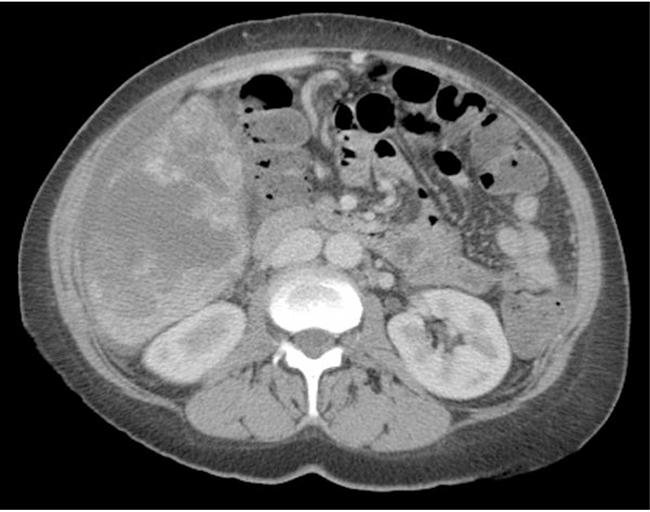
Figure 1: CT scan of the abdomen and pelvis with intravenous contrast showing multiple masses throughout the liver, later biopsy-proven hepatic angiosarcoma.
Percutaneous liver biopsy showed an extensively hemorrhagic and necrotic tumor composed of irregular, anastomosing vascular channels lined with atypical cuboidal to flattened endothelial cells with irregular hyperchromatic nuclei (Figure 2). Occasional mitoses were identified in vascular lining. The neoplastic cells were positive for vascular endothelial markers CD31 and CD34, but negative for cytokeratin E1/AE3, cytokeratins 7 and 20, hep-par1, AFP, and CA19.9. Proliferation index as detected by Ki-67 immunostaining was variable, ranging from <10% to focal areas of 40%. These morphologic features and immunophenotype were consistent with diagnosis of HAS.
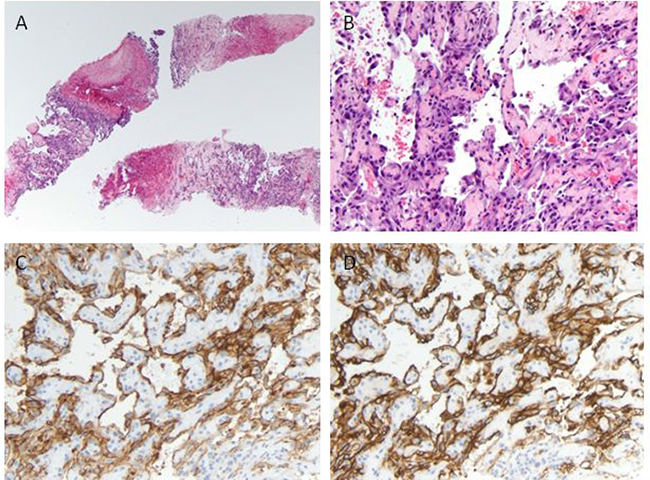
Figure 2: Histologic and immunohistochemical analysis of the liver tumor. (A) Low-power view of the biopsy cores containing hemorrhagic and necrotic tumor, hematoxylin and eosin (H&E), original magnification ×40. (B) On higher magnification tumor cells form irregular anastomosing vascular channels lined by cells with hyperchromatic atypical nuclei (H&E, original magnification ×200). Tumor cells express the endothelial markers CD31 (C), and CD34 (D).
Before starting therapy, patient had an acute decrease in hemoglobin (Hgb) from 8 to 6.5 and repeated CT scan that showed progression of dominant lesion to 12 cm, ascites and a small area of subcapsular hemorrhage compared to scan one month prior. The patient underwent hepatic artery embolization with post-procedural stabilization of Hgb and received chemotherapy with paclitaxel. Shortly thereafter, the patient experienced gradual deterioration of performance status with progressive abdominal pain, ascites and lower extremity edema. She chose to not receive additional cancer-directed therapy and pursued hospice care. The patient expired 2 months after initial diagnosis.
Comprehensive genomic profiling (CGP) performed on liver biopsy specimen revealed a ROS1 rearrangement involving GOPC that had not been previously described in HAS. [11]. CGP of the liver biopsy specimen was performed in a Clinical Laboratory Improvement Amendments (CLIA)-certified pathology laboratory (Foundation Medicine, Cambridge, MA), as previously described [11, 12]. In brief, ≥50 ng DNA was extracted from 40 microns of tumor sample in formalin-fixed, paraffin-embedded tissue blocks. Next generation sequencing was performed and targeted all coding exons in 405 cancer-related genes plus select introns within 31 genes that often display rearrangements in malignancy (FoundationOne) using Illumina HiSeq (Illumina, San Diego, CA) technology [13].
Additional abnormalities included mutations in MLL2 and PRDM1 genes, CDKN2A loss, and a low overall tumor mutational burden (4 mutations per megabase). Variants of unknown significance were noted in the following genes BRSK1, JARID2, KIT, MLH1, PPP2R1A, and ZNF217. Since these results became available only after the patient’s decline and subsequent transition to hospice care, she was unable to receive targeted therapy against ROS1.
LANDSCAPE OF ROS1 AND GOPC GENOMIC ALTERATIONS IN CANCERS
To investigate the frequency of ROS1 gene fusions among multiple cancer types, we queried MSK-IMPACT Clinical Sequencing Cohort [14], a pan cancer cohort of tumor samples with sequencing and copy number alterations (CNA) data (10336 patients/10945 samples) available at cBioportal cancer genomics database (www.cbioportal.org). ROS1 gene fusions with any gene partner occurred among 40 patients (0.4%) with the highest frequency of occurrences in non-small cell lung cancer (NSCLC; 31 of 1668 cases:1.8%) and salivary gland cancer (2 of 114 cases: 1.7%). Among the two cases of soft tissue sarcoma identified, one patient with perivascular epithelioid cell tumor had both ROS1-NETO1 and SLC4A1-ROS1 fusions while the other patient with synovial sarcoma carried COL4A3BP-ROS1 fusion. In this analysis we found only two cases carrying ROS1-GOPC fusions: high grade glioma and cancer of unknown primary (Figure 3A). The list of gene fusion partners with ROS1 from this dataset is summarized in Table 1. Specifically, a small cohort of angiosarcoma project dataset in cBioportal showed homodeletion of ROS1 in two out of 12 sequenced patients. One of the cases was a breast angiosarcoma patient with co-occurring GOPC deletion at 6q22.1 (Figure 3B).
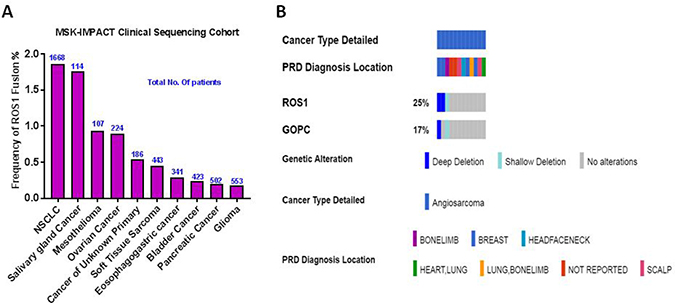
Figure 3: Frequency of ROS1 gene fusions in cancer. (A) Combined ROS1 fusion frequency with all known gene partners in a large pan-cancer dataset. Number of patients for each cancer subtype is shown in blue. NSCLC: Non-Small Cell Lung Cancer. (B) Oncoprint of ROS1 and GOPC copy number alterations in a small angiosarcoma (n = 12) project dataset taken from cBioportal is shown. PRD Diagnosis refers to patient reported response to initial diagnosis location of angiosarcoma.
Table 1: ROS1 gene fusion partners in various cancer subtypes
Gene fusion pair |
Cancer type |
Total No. of patients |
|---|---|---|
CD74-ROS1 |
Lung Adenocarcinoma |
16 |
EZR-ROS1 |
Lung Adenocarcinoma |
6 |
ROS1-SDC4 |
Lung Adenocarcinoma |
4 |
GOPC-ROS1 |
High-Grade Glioma, NOS |
2 |
Cancer of Unknown Primary |
||
SLC34A2-ROS1 |
Lung Adenocarcinoma |
2 |
SLC4A10-ROS1 |
Perivascular Epithelioid Cell Tumor |
1 |
SLC4A4-ROS1 |
Pancreatic Adenocarcinoma |
1 |
SLC6A17-ROS1 |
Lung Adenocarcinoma |
1 |
ROS1-NETO1 |
Perivascular Epithelioid Cell Tumor |
1 |
ROS1-SLC16A10 |
Bladder Urothelial Carcinoma |
1 |
TMEM181-ROS1 |
Pleural Mesothelioma, Epithelioid Type |
1 |
TPM3-ROS1 |
Lung Adenocarcinoma |
1 |
C6orf204-ROS1 |
Salivary Carcinoma |
1 |
COL4A3BP-ROS1 |
Synovial Sarcoma |
1 |
GOLGB1-ROS1 |
Serous Borderline Ovarian Tumor |
1 |
ROS1-FAM135B |
Lung Adenocarcinoma |
1 |
ROS1-HS3ST5 |
Stomach Adenocarcinoma |
1 |
List of ROS1 fusion partners and total number of patients identified with each gene fusion pair is shown. Data is extracted from MSK-IMPACT Clinical Sequencing Cohort at cBioportal. Gene abbreviations in order of appearance in table – CD74, Cluster of Differentiation 74; EZR, Ezrin; ROS1, ROS Proto-Oncogene 1; SDC4, Syndecan-4; GOPC, Golgi Associated PDZ And Coiled-Coil Motif Containing; SLC34A2, Solute Carrier Family 34 Member 2; SLC4A10, Solute Carrier Family 4 Member 10; SLC4A4, Solute Carrier Family 4 Member 4; SLC6A17, Solute carrier family 6 member 17; NETO1, Neuropilin And Tolloid Like 1; SLC16A10, Solute Carrier Family 16 Member 10; TMEM181, Transmembrane Protein 181; TPM3, Tropomyosin 3; C6orf204, chromosome 6 open reading frame 204; COL4A3BP, Collagen type IV alpha-3-binding protein; GOLGB1, Golgin B1; FAM135B, Family With Sequence Similarity 135 Member B; HS3ST5, Heparan Sulfate-Glucosamine 3-Sulfotransferase 5.
To further explore the incidence of ROS1 and GOPC concurrent alterations in primary hepatobiliary tumors and soft tissue sarcomas, we queried available datasets in cBioportal. In TCGA sarcoma dataset (265 patients with soft tissue sarcomas and 9 patients with nerve sheath tumors), we identified concurrent gene amplification or gain of ROS1 and GOPC involving 6q22.1 in 53 (21%) of the patients. In the TCGA hepatocellular carcinoma dataset, we identified 51 (14%) of 366 sequenced patients, of which 50 had low level gain and 1 patient had high level amplification of both genes. Among the 51 patients with concurrent gain of ROS1 and GOPC, we identified a patient with hepatocellular carcinoma plus intrahepatic cholangiosarcoma. Notably, Kaplan–Meier survival estimate showed a poor overall survival in both soft tissue sarcoma and hepatocellular carcinoma patients with ROS1 and GOPC gain/amplification (Figure 4). This database analysis highlights the previously unknown involvement of ROS1 and GOPC copy number alterations in soft tissue sarcomas and hepatic cancers.
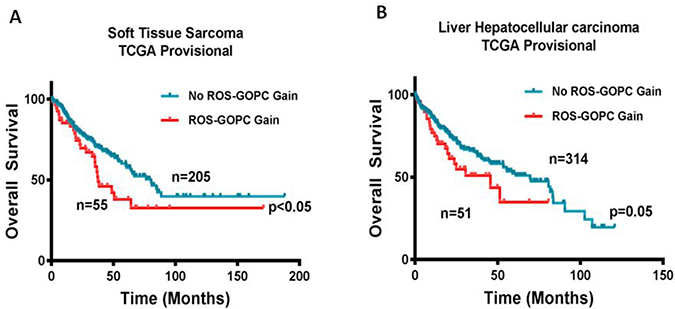
Figure 4: Gain in ROS1 and GOPC copy number is associated with poor overall survival. Kaplan–Meier survival curves for overall survival of patients with or without co-occurring ROS1 and GOPC copy number gain from (A) TCGA Soft Tissue Sarcoma dataset and (B) TCGA Hepatocellular carcinoma provisional dataset from cBioportal database were generated. Total number of patients in the two categories is shown. *P ≤ 0.05 for Log-rank (Mantel Cox) test.
DISCUSSION
ROS1 is a proto-oncogene located on the long arm of chromosome 6 and encodes a receptor tyrosine kinase involved in the regulation of cancer cell growth and differentiation. ROS1 is often involved in genomic rearrangements resulting in constitutionally active kinases that stimulate multiple pathways such as JAK-STAT, PI3K-AKT-mTOR, and RAS-RAF-MEK-ERK [15]. Fusion products of ROS1 have been observed in a variety of types of cancer, including tumors of the lung, gastrointestinal tract, hepatobiliary tree, and central nervous system [15]. Fusion partners that participate in ROS1 rearrangements include CD74, SLC34A2, SDC4 in addition to GOPC/FIG, which was observed in this patient’s tumor [15]. ROS1 rearrangement with another gene (CEP85L) has been previously described in angiosarcoma (primary site unknown) [16].
The GOPC/FIG (Fused In Glioblastoma) gene also resides on the short arm of chromosome 6 and encodes a protein displaying coiled-coil and PDZ domains. Through interaction with the PDZ domain (structural domain that recognize amino acid motifs), this protein localizes to the Golgi apparatus where it facilitates the intracellular trafficking of proteins to the cell surface and lysosomes [17, 18]. Among the proteins under control of GOPC/FIG are the cystic fibrosis transmembrane conductance regulator (CFTR), frizzled 5 and 8, beta-1-adrenergic receptor, cadherin 23, and somatostatin receptor subtype 5 [18].
GOPC/FIG was initially named following its discovery as a ROS1 fusion partner in a patient-derived astrocytoma cell line [17]. As a result of a 240 kb deletion on 6q21, exon 36 of ROS1 was fused to exon 3 of GOPC [17, 19]. Since this initial “long” fusion was described, a second “short” isoform that attaches exon 7 of FIG to exon 35 of ROS1 has also been identified [19]. ROS1-GOPC fusions have been observed in anaplastic astrocytoma [20], NSCLC [21], ovarian serous tumor [22], cholangiocarcinoma [19], and acral lentiginous melanoma [23]. Similar to other ROS1 fusions, ROS1- GOPC fusion proteins are believed to represent major drivers of carcinogenesis [19, 24]. Preclinical research has demonstrated that these gene fusions induce tumorigenesis both in vitro and in vivo [19, 24].
The identification of a ROS1- GOPC fusion has significant clinical implications due to the established anti-cancer activity of TKIs in tumors that carry ROS1 rearrangements. Crizotinib is a small molecule inhibitor of multiple tyrosine kinases, most notably ALK and ROS1. In a study of 50 patients with NSCLC and ROS1 rearrangement, treatment with crizotinib resulted in a 72% response rate and median PFS of 19.2 months [25]. Crizotinib has since gained FDA approval in NSCLC with ROS1 rearrangement. Although the prevalence of ROS1-GOPC fusion is too low for a dedicated clinical investigation, research in NSCLC cell lines has demonstrated in-vitro inhibition of cell growth following exposure to crizotinib [21].
While most of the research supporting ROS1 inhibition was conducted in NSCLC, there is anecdotal evidence of efficacy in other diseases. One patient with heavily pretreated acral lentiginous melanoma carrying a ROS1-GOPC fusion was treated with entrectinib (TKI with activity against tropomyosin receptor kinase (Trk), ROS1 and ALK), leading to a PR with 38% reduction in tumor burden at 3 months and 55% at 11 months [23]. The response was ongoing at the time of publication.
Unfortunately, the presence of a ROS1 fusion in this patient’s tumor was identified only after she had experienced significant decline in her performance status and quality of life. As a result, she did not receive targeted therapy against ROS1. Such treatment has a rather modest side effect profile and has produced prolonged responses in patients with other malignancies. This case illustrates the importance of obtaining comprehensive genetic and molecular profiling early in the course of disease. This is particularly crucial for malignancies that are often resistant to standard treatments such as angiosarcoma, or frequently harbor actionable mutations such as ROS1 fusions. Although this is the first report of ROS1-GOPC fusion in HAS, database analysis highlights the previously unknown frequency of ROS1 and GOPC molecular alterations in soft tissue sarcomas and hepatic cancers. This includes both fusions, which typically confer sensitivity to treatment with TKI’s, and copy number alterations of ROS1. Further study is needed to better characterize the relationship between amplification of ROS1 and response to targeted inhibitors. In addition, our analysis found amplification of these genes to correlate with reduced overall survival. Research in NSCLC has been inconclusive on this subject, with one study finding ROS1 copy number gain (CNG) to correlate with impaired disease-free and overall survival [26] but another finding no significant relationship [27]. However, when considered in tandem with the findings that ROS1 CNG does not always correlate to over-expression of the protein [26] and the most common mechanism of ROS1 CNG is polysomy of chromosome 6 [27], it seems most likely that ROS1 CNG is a surrogate marker of an aggressive tumor with complex karyotype.
CONFLICTS OF INTEREST
None.
REFERENCES
1. Chaudhary P, Bhadana U, Singh RA, Ahuja A. Primary hepatic angiosarcoma. Eur J Surg Oncol. 2015; 41:1137–43. https://doi.org/10.1016/j.ejso.2015.04.022.
2. Chien CY, Hwang CC, Yeh CN, Chen HY, Wu JT, Cheung CS, Lin CL, Yen CL, Wang WY, Chiang KC. Liver angiosarcoma, a rare liver malignancy, presented with intraabdominal bleeding due to rupture- a case report. World J Surg Oncol. 2012; 10:23. https://doi.org/10.1186/1477-7819-10-23.
3. Zheng YW, Zhang XW, Zhang JL, Hui ZZ, Du WJ, Li RM, Ren XB. Primary hepatic angiosarcoma and potential treatment options. J Gastroenterol Hepatol. 2014; 29:906–11. https://doi.org/10.1111/jgh.12506.
4. Murali R, Chandramohan R, Möller I, Scholz SL, Berger M, Huberman K, Viale A, Pirun M, Socci ND, Bouvier N, Bauer S, Artl M, Schilling B, et al. Targeted massively parallel sequencing of angiosarcomas reveals frequent activation of the mitogen activated protein kinase pathway. Oncotarget. 2015; 6:36041–52. https://doi.org/10.18632/oncotarget.5936.
5. Agulnik M, Yarber JL, Okuno SH, von Mehren M, Jovanovic BD, Brockstein BE, Evens AM, Benjamin RS. An open-label, multicenter, phase II study of bevacizumab for the treatment of angiosarcoma and epithelioid hemangioendotheliomas. Ann Oncol. 2013; 24:257–63. https://doi.org/10.1093/annonc/mds237.
6. Maki RG, D’Adamo DR, Keohan ML, Saulle M, Schuetze SM, Undevia SD, Livingston MB, Cooney MM, Hensley ML, Mita MM, Takimoto CH, Kraft AS, Elias AD, et al. Phase II study of sorafenib in patients with metastatic or recurrent sarcomas. J Clin Oncol. 2009; 27:3133–40. https://doi.org/10.1200/jco.2008.20.4495.
7. Ray-Coquard I, Italiano A, Bompas E, Le Cesne A, Robin YM, Chevreau C, Bay JO, Bousquet G, Piperno-Neumann S, Isambert N, Lemaitre L, Fournier C, Gauthier E, et al, and French Sarcoma Group (GSF/GETO). Sorafenib for patients with advanced angiosarcoma: a phase II Trial from the French Sarcoma Group (GSF/GETO). Oncologist. 2012; 17:260–6. https://doi.org/10.1634/theoncologist.2011-0237.
8. Kollar A, Jones RL, Stacchiotti S, Gelderblom H, Guida M, Grignani G, Steeghs N, Safwat A, Katz D, Duffaud F, Sleijfer S, van der Graaf WT, Touati N, et al. Pazopanib in advanced vascular sarcomas: an EORTC Soft Tissue and Bone Sarcoma Group (STBSG) retrospective analysis. Acta Oncol. 2017; 56:88–92. https://doi.org/10.1080/0284186x.2016.1234068.
9. Zhang SL, Liang L, Ji Y, Wang ZM, Zhou YH. The benefit of everolimus in recurrent/epithelioid angiosarcoma patients: Case reports and literature review. Oncotarget. 2017; 8:95023–9. https://doi.org/10.18632/oncotarget.21832.
10. Yoo C, Lee J, Rha SY, Park KH, Kim TM, Kim YJ, Lee HJ, Lee KH, Ahn JH. Multicenter phase II study of everolimus in patients with metastatic or recurrent bone and soft-tissue sarcomas after failure of anthracycline and ifosfamide. Invest New Drugs. 2013; 31:1602–8. https://doi.org/10.1007/s10637-013-0028-7.
11. He J, Abdel-Wahab O, Nahas MK, Wang K, Rampal RK, Intlekofer AM, Patel J, Krivstov A, Frampton GM, Young LE, Zhong S, Bailey M, White JR, et al. Integrated genomic DNA/RNA profiling of hematologic malignancies in the clinical setting. Blood. 2016; 127:3004–14. https://doi.org/10.1182/blood-2015-08-664649.
12. Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, Schnall-Levin M, White J, Sanford EM, An P, Sun J, Juhn F, Brennan K, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013; 31:1023–31. https://doi.org/10.1038/nbt.2696.
13. Ross JS, Ali SM, Fasan O, Block J, Pal S, Elvin JA, Schrock AB, Suh J, Nozad S, Kim S, Jeong Lee H, Sheehan CE, Jones DM, et al. ALK Fusions in a Wide Variety of Tumor Types Respond to Anti-ALK Targeted Therapy. Oncologist. 2017; 22:1444–50. https://doi.org/10.1634/theoncologist.2016-0488.
14. Zehir A, Benayed R, Shah RH, Syed A, Middha S, Kim HR, Srinivasan P, Gao J, Chakravarty D, Devlin SM, Hellmann MD, Barron DA, Schram AM, et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat Med. 2017; 23:703–13. https://doi.org/10.1038/nm.4333.
15. Davies KD, Doebele RC. Molecular Pathways - ROS1 Fusion Proteins in Cancer. Clin Cancer Res. 2013; 19:4040–5. https://doi.org/10.1158/1078-0432.CCR-12-2851.
16. Giacomini CP, Sun S, Varma S, Shain AH, Giacomini MM, Balagtas J, Sweeney RT, Lai E, Del Vecchio CA, Forster AD, Clarke N, Montgomery KD, Zhu S, et al. Breakpoint analysis of transcriptional and genomic profiles uncovers novel gene fusions spanning multiple human cancer types. PLoS Genet. 2013; 9:e1003464. https://doi.org/10.1371/journal.pgen.1003464.
17. Charest A, Lane K, McMahon K, Park J, Preisinger E, Conroy H, Housman D. Fusion of FIG to the receptor tyrosine kinase ROS in a glioblastoma with an interstitial del(6)(q21q21). Genes Chromosomes Cancer. 2003; 37:58–71. https://doi.org/10.1002/gcc.10207.
18. Herrmann S, Ninkovic M, Kohl T, Pardo LA. PIST (GOPC) modulates the oncogenic voltage-gated potassium channel KV10.1. Front Physiol. 2013; 4:201. https://doi.org/10.3389/fphys.2013.00201.
19. Gu TL, Deng X, Huang F, Tucker M, Crosby K, Rimkunas V, Wang Y, Deng G, Zhu L, Tan Z, Hu Y, Wu C, Nardone J, et al. Survey of tyrosine kinase signaling reveals ROS kinase fusions in human cholangiocarcinoma. PLoS One. 2011; 6:e15640.
20. Shao L, Miller S, Koschmann C, Camelo-Piragua S. Clinical Application of Whole Genome Array Improves the Diagnosis of Pediatric Brain Tumors. Int J Surg Pathol. 2017; 25:688–95. https://doi.org/10.1177/1066896917727349.
21. Rimkunas VM, Crosby KE, Li D, Hu Y, Kelly ME, Gu TL, Mack JS, Silver MR, Zhou X, Haack H. Analysis of receptor tyrosine kinase ROS1-positive tumors in non-small cell lung cancer: identification of a FIG-ROS1 fusion. Clin Cancer Res. 2012; 18:4449–57. https://doi.org/10.1158/1078-0432.ccr-11-3351.
22. Birch AH, Arcand SL, Oros KK, Rahimi K, Watters AK, Provencher D, Greenwood CM, Mes-Masson AM, Tonin PN. Chromosome 3 anomalies investigated by genome wide SNP analysis of benign, low malignant potential and low grade ovarian serous tumours. PLoS One. 2011; 6:e28250. https://doi.org/10.1371/journal.pone.0028250.
23. Couts KL, McCoach CE, Murphy D, Christiansen J, Turner J, Lewis KD, Robinson WA, Doebele RC. Acral Lentiginous Melanoma Harboring a ROS1 Gene Fusion With Clinical Response to Entrectinib. JCO Precision Oncology. 2017; 1–7. https://doi.org/10.1200/PO.16.00013.
24. Charest A, Wilker EW, McLaughlin ME, Lane K, Gowda R, Coven S, McMahon K, Kovach S, Feng Y, Yaffe MB, Jacks T, Housman D. ROS fusion tyrosine kinase activates a SH2 domain-containing phosphatase-2/phosphatidylinositol 3-kinase/mammalian target of rapamycin signaling axis to form glioblastoma in mice. Cancer Res. 2006; 66:7473–81. https://doi.org/10.1158/0008-5472.can-06-1193.
25. Shaw AT, Ou SH, Bang YJ, Camidge DR, Solomon BJ, Salgia R, Riely GJ, Varella-Garcia M, Shapiro GI, Costa DB, Doebele RC, Le LP, Zheng Z, et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. 2014; 371:1963–71. https://doi.org/10.1056/NEJMoa1406766.
26. Jin Y, Sun PL, Kim H, Park E, Shim HS, Jheon S, Kim K, Lee CT, Chung JH. ROS1 gene rearrangement and copy number gain in non-small cell lung cancer. Virchows Arch. 2015; 466:45–52. https://doi.org/10.1007/s00428-014-1679-2.
27. Clavé S, Gimeno J, Muñoz-Mármol AM, Vidal J, Reguart N, Carcereny E, Pijuan L, Menéndez S, Taus Á, Mate JL, Serrano S, Albanell J, Espinet B, et al. ROS1 copy number alterations are frequent in non-small cell lung cancer. Oncotarget. 2016; 7:8019–28. https://doi.org/10.18632/oncotarget.6921.

