INTRODUCTION
Resveratrol (trans-3, 4′, 5-trihydroxystilbene) is a naturally occurring polyphenol whose complex cellular actions are cell cycle arrest, differentiation, and apoptosis [1]. These cellular actions may reflect a reduction in intracellular reactive oxygen species (ROS) and mitochondrial trans-membrane potential Δ ψm, as well as reduced phosphorylation of mTOR, RP-S6 and 4EBP1 [2]. In addition, the compound is also reported to have anti-inflammatory, anti-leukemic, neuroprotective and antiviral properties [3–5]. A cell surface receptor for the stilbene has been identified on integrin αvβ3 and has been shown to be linked to activation of the MAPK signal transduction pathway [6]. The Arg-Gly-Asp (RGD) recognition site on the integrin blocks actions of the stilbene, indicating proximity of the resveratrol receptor to the RGD site [6, 7]. The mechanisms of anti-tumor effects of resveratrol are incompletely understood, but an end result in a variety of tumor cell models of resveratrol treatment is apoptosis [8–10].
Other beneficial effects attributed to resveratrol relate to aging, e.g., cataract and bone mass loss, and to neurodegeneration, obesity and diabetes [11]. Resveratrol induces expression of multiple genes and confers metabolic changes that mimic caloric restriction, a state which promotes longevity across species [12]. These benefits appear to be dependent upon sirtuin 1 (SIRT1), a NAD+-dependent deacetylase. How resveratrol activates SIRT1 protein is not entirely clear, but the binding of lamin A by SIRT1 induces an allosteric change in SIRT1 that exposes its deacetylase site to native substrates [11]. Modulation of SIRT1 activity is also dependent on physiological substrate sequence and these substrates may contribute to actions of resveratrol [13].
The pharmacokinetics of resveratrol have been studied in pre-clinical models and human subjects [14, 15] and appear unfavorable for application to clinical disease states [14]. The systemic bioavailability of resveratrol is poor, as is the case with many polyphenols. Studies in mice, rats and dogs consistently suggest that resveratrol is well-absorbed, but is avidly glucuronidated and sulfated in both liver and intestinal epithelial cells [16, 17]. Studies in human subjects also indicate poor bioavailability of unmodified resveratrol [18, 19]. Resveratrol has a very short half-life in the systemic circulation.
In the current studies, we have examined the anti-cancer properties of resveratrol in chemotherapy-resistant human breast cancer MDA-MB-231 cells, using an in vitro perfused cell model in which concentrations of the agent and durations of exposure to the agent can be critically adjusted. Ambient pH and medium composition are also readily manipulated and are important to tumor cell function [20]. Resistance of cancers to therapy can involve both biochemical and microenvironmental factors. Studies here include effects on resveratrol’s activity of factors such as environmental pH of tumor cells, duration of cell exposure to resveratrol, and effectiveness of drug concentrations in terms of anti-proliferation. The microarray and real-time PCR studies are focused on genes whose expression is highly upregulated by resveratrol (such as NLRP1, CASP2, LSM4 and SLC12A4) and genes that support apoptosis, e.g., BAD, p53, TP53I3, p21, c-fos and COX-2.
RESULTS
Effect of time of exposure on resveratrol-induced signal transduction, gene expression and anti-proliferation in MDA-MB-231 cells
Resveratrol has limited bioavailability, which has caused concern about its biological activity. Cellular uptake and metabolism of resveratrol is rapid [21]. To determine the minimum incubation time for resveratrol-activated signal transduction and anti-proliferation, MDA-MB-231 cells were treated with 10 μM resveratrol for 0.5 − 4 h prior to harvest. Exposure of cells for 0.5 h to resveratrol importantly activated ERK1/2 (Fig. 1A). To determine the duration of resveratrol-induced ERK1/2 activation after removal of the stilbene, we treated cells with resveratrol for 0.5 − 4 h, then removed the agent at specific time points. Cells were then washed twice with fresh medium before they were re-fed with medium for the remainder of the time prior to harvest. The activation of ERK1/2 exposed to resveratrol for 0.5 h did not decay as compared to that in the 4 h exposed cells (Fig. 1B).
The effect of duration of incubation with resveratrol on drug-modulated gene expression was determined by incubating cells with resveratrol for either 1 h or 6 h. The resveratrol was then removed and cells were washed twice with fresh medium and re-fed with fresh medium for another 5 h. There was no significant difference in resveratrol-upregulared and -downregulated gene expression with exposure to 10 μM resveratrol for 6 h and 1 h (Fig. 1C). When MDA-MB-231 cells were treated with 10 μM resveratrol for short periods of time (0.5 to 4 h) daily for 6 d, we found that exposure to the drug for 4 h reduced cell counts by more than 60%, compared to untreated control cells (1.28 × 106 ± 3.46 × 105 [resveratrol] vs. 4.03 × 106 ± 3.29 × 105 [control]). On the other hand, exposure of cells to 10 μM resveratrol for 24 h daily for 6 d caused 80% reduction in cell counts (7.72 × 105 ± 5.44 × 104) compared to untreated control (Fig. 1D). The full-term exposure to resveratrol increased only 20% more in anti-proliferative effect than those treated with 4 h daily. Those results suggest that short-term exposure to resveratrol is sufficient to induce cellular activities such as gene expression and anti-proliferation.
Effect of acid-alkaline culture conditions on resveratrol-induced anti-proliferation in MDA-MB-231 cells
It has been reported that the acidic condition of tumor microenvironment affects the efficacy of chemotherapy [20]. MDA-MB-231 cells were cultured in media with pHs of 6.8, 7.4, 7.5 and 8.6. Cell proliferation in the absence of resveratrol (10 μM) was significantly affected by pH culture condition, but the anti-proliferation effect of the stilbene was only minimally affected by pH (Fig. 2A). The resveratrol-induced activation of ERK1/2 was not affected by extracellular pH change (Fig. 2B).
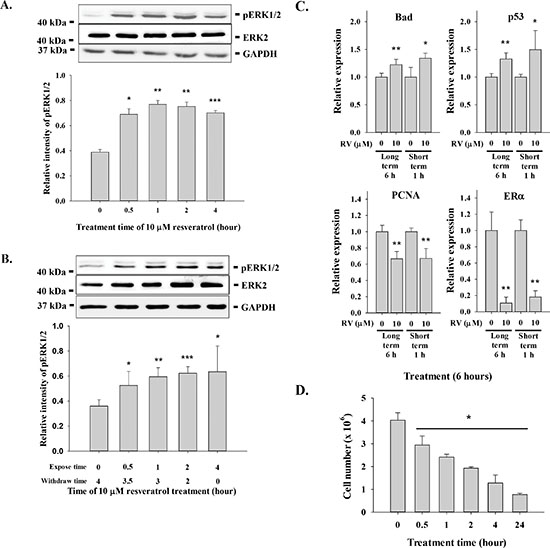
Figure 1: Incubation effect on resveratrol-induced signal transduction, gene expression and anti-proliferation. (A) MDA-MB-231 cells were treated with 10 μM resveratrol for different time periods (0.5 to 4 h) prior to harvest. Cell extracts were separated by SDS-PAGE and blotted with anti-pERK1/2. Number of independent experiments (N) = 3. (B) MDA-MB-231 cells were treated with 10 μM resveratrol for different time periods (0.5 to 4 h) prior to removal of resveratrol and re-fed with fresh medium before harvest. Cell extracts were separated by SDS-PAGE and blotted with anti-pERK1/2 and anti-ERK2. N = 4. (C) MDA-MB-231 cells were treated with 10 μM resveratrol for different time periods (1 or 4 h) prior to removal of resveratrol and re-fed with fresh medium before harvest. Total RNA was extracted and qPCR was conducted as described. Number of independent experiments (N) = 3. (D) MDA-MB-231 cells were seeded in Petri dish and re-fed with DMEM containing 10% serum. 10 μM (228.24 × 10-6 mg/ml) of resveratrol was added to cell culture daily for 24, 4, 2, 1 and 0.5 h. After incubation, cells were re-fed with fresh medium. Cells were harvested at the 6th day and cell numbers were counted. N = 4 (*p < 0.05, compared to control with vehicle solvent)
Microarray gene profiles in resveratrol-treated MDA-MB-231 cells
MDA-MB-231 cells were exposed to 10 μM resveratrol for 6 h. Microarray experiments were conducted as described in the Materials and Methods and identified 25 highly up-regulated genes (≥6-fold increase in mRNA abundance) as major gene targets for resveratrol (Fig. 3A). The second most up-regulated mRNA with resveratrol treatment was encoded by the NALP1 gene, also known as NLRP1 and DEFCAP, an essential component of the inflammasome and known to play an important role in innate immunity. Recent hallmark reports clarify the role of this gene in caspase-1 activation and interleukin-1β production and in inflammasome assembly and function demonstrating that NALP1 is a direct sensor of bacterial components in host defense against pathogens [22]. The third most highly up-regulated mRNA was encoded by the CASP2 (caspase 2) gene, a well-known apoptosis regulatory protein. It has been demonstrated by using in vitro coimmunoprecipitation experiments that NALP1 (DEFCAP) protein is capable of strongly interacting with caspase-2, and transient overexpression of full-length DEFCAP-L, but not DEFCAP-S, in breast adenocarcinoma cells MCF-7 resulted in significant levels of apoptosis [23]. Other up-regulated genes, such as LSM4, which is involved in RNA processing, may function in a chaperone-like manner [24]. SLC12A4 (human potassium chloride cotransporter 1) is activated by cell swelling and may be associated with apoptosis [25, 26]. These results convey the anti-inflammation and anti-cancer properties of the stilbene. In order to further investigate whether environmental conditions affect the expression of those up-regulated genes induced by resveratrol, expression of NALP1, CASP2, SLC12A4 and LSM4 was studied.
MDA-MB-231 cells grown in different pH conditions were treated with 10 μM resveratrol for 6 h and total RNA was harvested for real-time PCR. Expression of NALP1, CASP2, SLC12A4 and LSM4 mRNAs were induced significantly under various conditions (Fig. 3B). We also studied pro-apoptotic protein abundance in resveratrol-treated cells harvested from the perfusion bellows system and demonstrated activation of several pro-apoptotic genes (Fig. 3C). Thus, resveratrol-induced gene expression and chemical/cellular activity are minimally affected by changes in the cancer microenvironment.
Pharmacodynamic studies of resveratrol in the perfusion bellows cell culture system
MDA-MB-231 cells were seeded in the non-perfused bellows cell culture system, re-fed with medium containing 10% FBS after 24 h and refreshed daily thereafter. Cells were counted to confirm the numbers of cells initially attached to flakes. Resveratrol concentration in the medium was constant throughout each experiment and the resveratrol concentrations tested were 0.01 μM to 10 μM. An anti-proliferative effect of resveratrol was not detectable on cell counts at 2 d (Fig. 4A), regardless of stilbene concentration (up to 10 μM). Anti-proliferation was apparent by 4 d of treatment, at which time resveratrol caused a 1.3 to 20-fold (1.59 × 108 ± 2.03 × 107 to 1.01 × 107 ± 2.75 × 105) decrease in treated cells compared to unexposed control cells (2.02 × 108 ± 2.36 × 107). The resveratrol effect was concentration-dependent (Fig. 4A). In a second protocol, cells were exposed to 10% FBS-containing DMEM and treated with different concentrations of resveratrol (0.1 to 10 μM) daily for 8 d. Cells were then harvested from the perfusion bottles and counted. There were significantly different growth patterns of control cell cultures between perfusion and non-perfusion cell culture systems in extending the culture condition (Fig. 4B).
Because short-term exposure to resveratrol induced anti-proliferation in MDA-MB-231 cells (Fig. 1D), we determined whether short-term exposure to resveratrol induced anti-proliferation of cancer cells in the perfusion bellows cell culture system. MDA-MB-231 cells were cultured in cell culture system as described above. Different concentrations of resveratrol were injected into perfusion bellows bottles over a 1 h period and incubation continued for another 23 h without resveratrol in influx medium. We determined the concentrations of resveratrol harvested from perfusion bellows bottle by LCMS/MS. Media were harvested at various time points. The concentration of resveratrol in efflux tubes from bellows cell culture was far below the influx tube in the range between 0.5 μM to 100 μM (Fig. 4C). However, the concentrations of resveratrol in efflux tubes of the 50 μM and 100 μM input resveratrol concentrations followed the expected patterns (Fig. 4C). These results confirm previous reports that the turnover rate of resveratrol inside the cells is rapid. Interestingly, the results of 50 μM and 100 μM resveratrol experiments also suggest that resveratrol may not need to enter cells to induce apoptosis. For the anti-proliferation studies, the short-term 1 h-injection of resveratrol procedures were repeated for 6 d. Results presented in Fig. 4D indicate that although cells were exposed to resveratrol for a short period of time, resveratrol induced anti-proliferation.
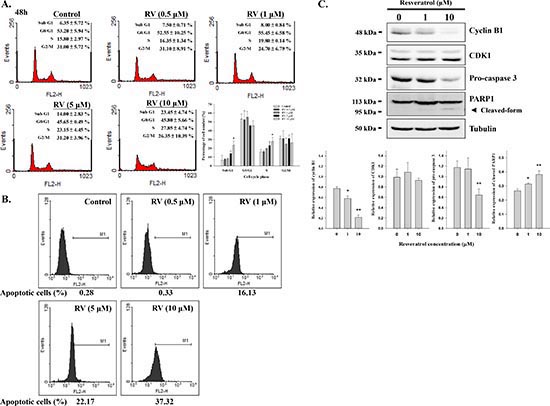
In order to determine the effect of resveratrol-induced apoptosis and cell cycle arrest, cells were treated with different concentration of resveratrol for 24 h. Cells were harvested and flow cytometry was conducted. Resveratrol increased the fraction of cells in S phase from 15.80 ± 2.97% in control to 27.85 ± 4.74% at the 48 h time point in a concentration-dependent manner (Fig. 5A). Cells in G1 and G2/M phases were slightly decreased after resveratrol treatment indicating that resveratrol may induce S phase growth arrest in MDA-MB-231 cells. Resveratrol also induced apoptosis in a concentration-dependent manner (Fig. 5A and B), which may result from modulation of cell cycle progression. Resveratrol inhibited MDA-MB-231 cell proliferation and increased the fraction of cells in sub-G1 by 23.45 ± 4.74%, compared to untreated control. Furthermore, the effects of resveratrol on cyclin B1, CDK1, caspase 3, and PARP1 were also addressed. Results indicated that 10 μM resveratrol significantly inhibited cyclin B1 expression, decreased the content of caspase 3, and activated PARP1 (Fig. 5C).
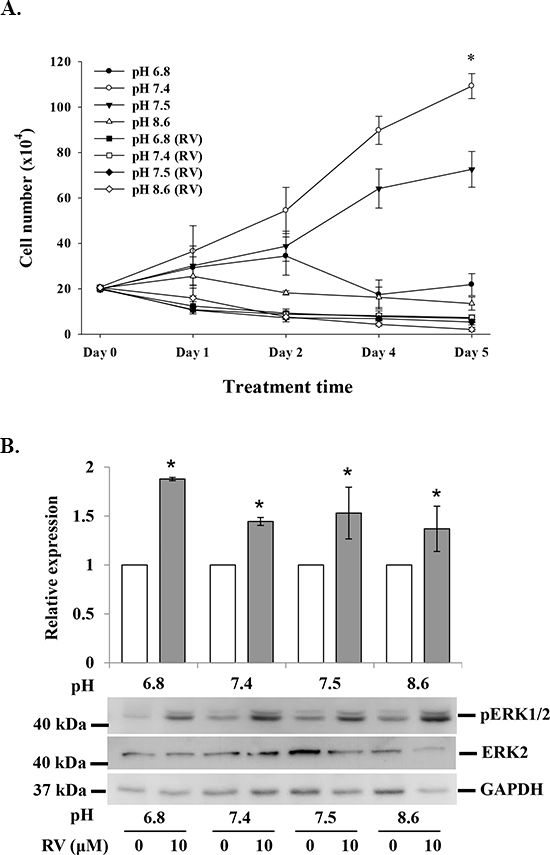
Figure 2: Effect of acid-alkaline culture conditions on resveratrol-induced anti-proliferation in MDA-MB-231 cells. (A) MDA-MB-231 cells cultured in different pH conditions were treated with or without 10 μM resveratrol daily for 5 d. At the end of the culture period, cells were harvested and counted (N = 4). (B) MDA-MB-231 cells cultured in different pH conditions were treated with 10 μM resveratrol for 4 h. Cells were harvested and total protein was extracted and separated by SDS PAGE. pERK1/2 and ERK2 were evaluated. GAPDH was used as an internal control. N = 4 (*p < 0.05, compared to control with vehicle solvent)
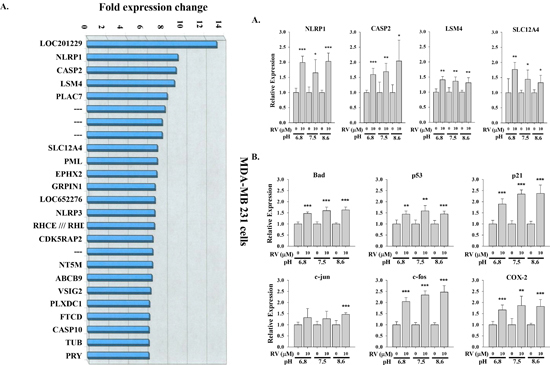
Figure 3: Resveratrol-induced gene expression is not affected by culture media pH. (A) The most up-regulated genes induced by resveratrol in human breast cancer MDA-MB-231 cells. MDA-MB-231 cells were treated with or without 10 μM resveratrol for 6 h. Total RNA was harvested and hybrided with Affymetrix HG-U133 plus 2.0 microarray platform. 25 top up-regulated genes from 54675 probe sets. N = 2. (B) MDA-MB-231 cells cultured in different pH conditions were treated with 10 μM resveratrol for 6 h. Cells were harvested and total RNA was extracted for qPCR. Expression of 4 most up-regulated genes induced by resveratrol was examined. N = 4. (C) resveratrol-induced 6 p53-dependent genes were also examined. N = 3 (*p < 0.05; **p < 0.005; ***p < 0.001, compared to control with vehicle solvent)
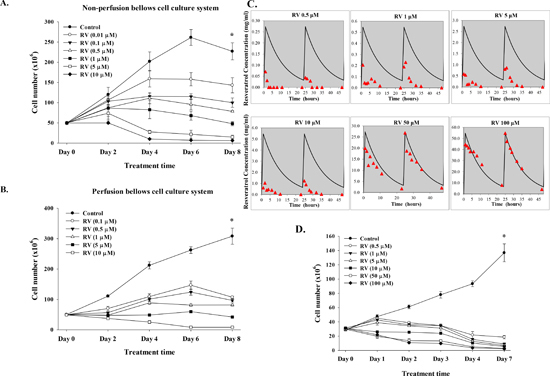
Figure 4: Resveratrol induces anti-proliferation in breast cancer MDA-MB-231 cells. Cells were cultured in (A) non-perfusion (B) perfusion bellows cell culture system with 10% fetal bovine serum in culture medium for 8 d. Different concentrations (0.01 to 10 μM) of resveratrol were added in culture media daily, together with refreshed medium. Cells were harvested and cell numbers were counted every other day. Points, mean; bars, ± SD; N = 4 (C) Short-term exposure to resveratrol induces anti-proliferation of cancer cells in the perfusion bellows cell culture system. Determination of resveratrol concentration was performed by LC/MS/MS. Cells were cultured in the system with 10% fetal bovine serum in culture medium. Different concentrations of resveratrol were added in culture media daily by syringe pump in 1 h and no fresh resveratrol in influx medium for the another 23 h. One ml of medium was drawn from the efflux tube of the central reservoirs at different time points. Samples were stored at −20°C until examination. Concentrations of resveratrol were measured by LC/MS/MS as described in the Materials and Methods. N = 2. (D) Pharmacodynamic studies of resveratrol-induced anti-proliferation in perfusion bellows cell culture system. Cells were cultured in the perfusion bellows culture system with 10% fetal bovine serum in culture medium for 6 d. Different concentrations of resveratrol were added in culture media daily by syringe pump in 1 h and there was no fresh resveratrol in the influx medium for another 23 h; the final concentrations in the media were 0.5 to 100 μM. Cells were harvested and cell numbers were counted every day. N = 3 (*p < 0.05, compared to control with vehicle solvent)
DISCUSSION
This study investigated effects of resveratrol-induced anti-proliferation in triple-negative MDA-MB-231 breast cancer cells, but the results shown may be applicable to other cancer cells. Included were studies of changes in the cell microenvironment to determine whether such changes altered drug activity.
When low concentrations of resveratrol were used in the current studies, the efflux concentrations of resveratrol were far below the projected concentrations (Fig. 4C); this is consistent with the short biological half-life of the agent described by others [27]. Studies have indicated that resveratrol in double-distilled water is stable for up to 8 h; on the other hand, resveratrol in incubation medium containing 10% FBS decays after 2-3 h and only 10% of the activity persists at 8 h at 37°αβ
μ<<
®™'°
μμ®°®
–
μμ×–––°–
μ'
μ×−×°−°μμ°™
μ'–
μμ'μ®°°°’′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′′α′′′′′′′′ΔΔ
’<
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
–
'–
–
–
–
'′–
–
–
–
–
–

