INTRODUCTION
Cancer is a major risk factor for venous thromboembolism (VTE) [1], and cancer patients who develop VTE tend to have a poorer prognosis and diminished life expectancy [1]. These patients are also at increased risk of experiencing VTE recurrence and major bleeding complications [1]. Overall, VTE is the second leading cause of death in cancer patients in both medical and surgical settings, after metastasis [1]. Current national and international clinical practice guidelines for the management of VTE in patients with cancer recommend administering LMWH for at least 3 months [2–4]. After 3–6 months, the decision to continue or terminate anticoagulant treatment should be based on the benefit-to-risk ratio, tolerability, drug availability, patient preference, and cancer activity. These recommendations are based on evidence that LMWH is the safest and most efficacious anticoagulant for the treatment and prophylaxis of VTE in this patient population, and that it is associated with a lower incidence of bleeds and recurrent VTE compared to unfractionated heparin (UFH) or vitamin K antagonists (VKAs) [5–7].
There is discrepancy, however, between evidence-based recommendations and clinical practice [8]. The French CARMEN study, which assessed 500 cancer patients treated for VTE two years after the release of French national guidelines, indicated that while physicians administered appropriate anticoagulation for established VTE during the initial phase of treatment (first 10 days) in 98% of cases, guideline adherence dropped to 62% during the treatment maintenance phase (10 days-3 months) [9]. Similarly, an analysis of data from the RIETE registry reported that only 66% of cancer patients were maintained on LMWH for the appropriate recommended treatment duration [10]. Results from a recent study of insurance claims records in the United states indicated that LMWH continues to be under-prescribed in cancer patients; VKA was the most commonly used anticoagulant (50%), with LMWH prescribed in 40% of cases [11]. Similar recent studies carried out on a global scale suggest that LMWH may be prescribed in as few as 25% of cases worldwide[12]. These continued failures in guideline adherence appear to stem in part from 1) concerns about whether cancer patients can tolerate long-term LMWH treatment and their potential side effects, despite their grade A evidence-based established benefit, [2] 2) beliefs about patients’ lack of willingness to accept treatment regimens involving daily subcutaneous injections [13], and 3) preconceptions about the overall impact LMWH treatment will have on QoL. Potential complications of LMWH treatment include heparin-induced thrombocytopenia (HIT), allergic reactions, pain and ecchymosis at injection sites. A number of prospective studies examining long-term use of LMWH in cancer patients have not reported any cases of severe thrombocytopenia and, overall, the incidences of thrombocytopenia in patients on long-term LMWH or receiving VKAs are similar [5–7]. Risk of allergic reaction was not found to differ between LMWH and placebo arms in the MALT study [14]. Pain and development of indurations at injection sites can develop in 30–90% of patients [15], which could prove to be a barrier to treatment compliance. Health-related Quality of life (QoL) has never been assessed in this patient population, although it is an important variable in evaluating treatment effectiveness and compliance in oncology [16, 17]. We therefore designed this prospective, longitudinal, multicenter, observational study to assess QoL in cancer patients initiated on long-term anticoagulant therapy for VTE in a real-world setting with treatment management determined by the local attending physicians.
RESULTS
400 consecutive cancer patients at participating GFTC centers, diagnosed with a VTE and put on an anticoagulant, were included in the study (Figure 1, patient flow chart). At inclusion, 88.8% (355/400) of patients were treated with LMWH, 5.5% (22/400) received a vitamin K antagonist (VKA), 1.5% (6/400) an unfractionated heparin (UFH), 3.75% (15/400) an anti-Xa direct oral anticoagulant, and 0.5% (2/400) another anticoagulant therapy. The overall number of deaths at 6 months was 84, with a mortality rate of 23.7% (95% CI 19.1%–28.1%) (Figure 2A). Seventy-nine deaths were attributed to cancer progression, 3 to VTE, and 2 to others causes (Figure 2A). Kaplan–Meier survival analysis indicated that patients on LMWH (n =337) had numerically better survival outcomes relative to other treatments (n = 41), but these were not significant (p = 0.08 by log-rank test) (Figure 2B). Only 378 patients were considered in the survival analysis because vital status was not available for twenty-one patients and date of death was not available for one patient (Figure 1).
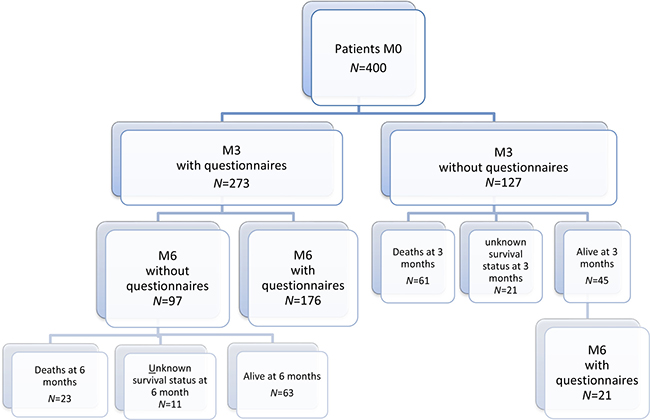
Figure 1: Patient flow chart. Flow chart depicts the distribution of completed questionnaires at inclusion, and 3- and 6-month follow-ups.
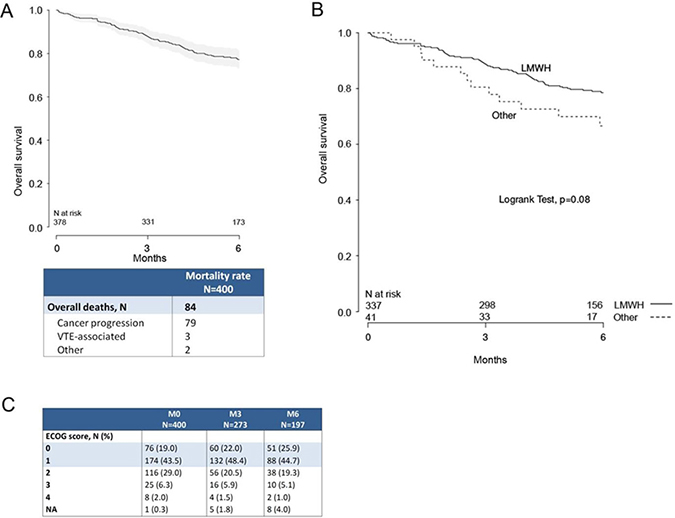
Figure 2: Survival at 3 and 6 months after initiation of anticoagulant therapy. (A) Kaplan–Meier survival plot of the entire patient population. Only 378 patients were considered in the survival analysis because vital status was not available for twenty-one patients and date of death was not available for one patient. (B) Kaplan–Meier survival plot of LMWH group (n = 337) versus a pooled group of patients receiving other anticoagulant therapies (n = 41). Patients receiving LMWH had numerically better survival outcomes (log-rank test, p = 0.08). (C) Distribution of ECOG scores at inclusion, 3-month, and 6-month follow-ups.
Among the 355 patients given LMWH, 59.4% had metastatic disease (181/355) and 67.1% were on chemotherapy (237/355). Baseline patient characteristics are provided in Table 1 and Supplementary Table 1. During treatment follow-up, 18.9% (67/355) of patients on LMWH reported at least one side effect of injections (number of reports: pain at injection site, 26 (7.3%); ecchymosis, 57 (16.1%); pruritis, 2(0.6%); nodules, 28 (7.9%)).
Table 1: Baseline patient characteristics at time of VTE diagnosis for participants receiving LMWH
LMWH |
|
|---|---|
Age (years, SD) |
63 (14) |
Female (N, %) |
183 (51.6) |
Weight (kg, SD) |
70 (15) |
Height (cm, SD) |
170 (9) |
VTE (N, %) |
|
Symptomatic |
266 (75.4) |
Asymptomatic |
87 (24.7) |
NA |
2 |
Type of VTE (N, %) |
|
Superficial vein thrombosis |
19 (5.4) |
Deep vein thrombosis (DVT) |
253 (71.3) |
Bilateral thrombosis |
20 (5.6) |
Pulmonary embolism |
117 (33.0) |
Central venous catheter-related thrombosis |
29 (8.2) |
Cancer dissemination (N, %) |
|
Local |
52 (17.1) |
Regional |
72 (23.6) |
Metastatic |
181 (59.3) |
NA |
50 |
Histology (N, %) |
|
Adenocarcinoma |
223 (62.8) |
Squamous |
18 (5.1) |
Undifferentiated |
4 (1.1) |
Neuroendocrine |
5 (1.4) |
Sarcoma |
4 (1.1) |
Other |
101 (28.5) |
Cancer treatment-related risk factors (N, %) |
|
Surgery < 1 month |
36 (10.2) |
Radiotherapy < 1 month |
26 (7.4) |
Hormone therapy |
16 (4.5) |
Tyrosine kinase inhibitors |
8 (2.3) |
Chemotherapy |
237 (67.1) |
Anti-angiogenic |
19 (5.4) |
Risk factors (N, %) |
|
Prior DVT risk |
45 (12.7) |
Prior PE risk |
10 (2.8) |
Obesity (BMI > 35 kg/m2) |
11 (3.1) |
Acute infection |
27 (7.7) |
Surgery < 3 months |
75 (21.3) |
Bedridden |
51 (14.5) |
Varicose veins inferior (arms) |
36 (10.3) |
Central venous catheter |
202 (56.9) |
Heart failure |
4 (1.1) |
Acute respiratory failure |
9 (2.6) |
Thrombophilia |
1 (0.3) |
Descriptive baseline statistics. Categorical variables are reported as frequencies with percentages. Continuous variables are reported as means with standard deviations (SD).
Improvement in QoL over the 6-month observation period
Three standardized questionnaires, all previously translated and comprehensively validated into different languages, including French [18–23], were used to measure generic and disease-specific QoL (Supplementary Materials).
In the Medical Outcome Study 36-item Short-Form Health Survey (MOS SF-36), [18] global health-related quality of life (HRQoL) scores significantly increased 1.3-fold [95% CI 0.81–1.79] per month over the 6-month observation period (P < 0.0001) (Table 2, Figure 3). Improvements in both the physical (1.32-fold [95% CI 0.80–1.84]; P < 0.0001) and mental (1.33-fold [95% CI 0.79–1.87]; P < 0.0001) health summary scores were observed, as well as across all specific dimensions examined (Table 2). In the patients who answered the questionnaires at 3-month follow-up, there was a mean 2.9 (19) point increase in global HRQoL scores compared to their respective scores at baseline, with a 2.7 (20) point increase in the physical health status summary score, and a 3.1 (22) point increase in the mental health status summary score (Supplementary Table 2). At 6 month follow-up, patients on LMWH showed a mean 7.6 (22) point increase relative to their respective scores at M0, with increases in both the physical (7.7 (23)) and mental health status (7.6 (24)) summary scores (Supplementary Table 2).
Table 2: QoL assessments at 0, 3 and 6-month follow-ups in patients treated with LMWH, mean (SD)
M0 |
M3 |
M6 |
||||||
|---|---|---|---|---|---|---|---|---|
N |
Mean (SD) |
N |
Mean (SD) |
N |
Mean (SD) |
Slope of change per month (95%CI) |
P-value |
|
355 |
252 |
181 |
||||||
MOS SF-36 |
||||||||
Global HRQoL |
355 |
42 (20) |
252 |
48 (20) |
181 |
51 (22) |
1.30 (0.81; 1.79) |
<0.0001 |
Physical Health summary score |
355 |
38 (19) |
252 |
44 (20) |
181 |
48 (22) |
1.32 (0.80; 1.84) |
<0.0001 |
Mental Health summary score |
355 |
45 (23) |
252 |
52 (22) |
181 |
55 (24) |
1.33 (0.79; 1.87) |
<0.0001 |
Physical functioning |
355 |
47 (31) |
252 |
54 (28) |
181 |
56 (29) |
0.98 (0.31; 1.66) |
0.005 |
Physical roles limitation |
355 |
20 (33) |
252 |
28 (39) |
181 |
35 (42) |
2.35 (1.37; 3.34) |
<0.0001 |
Bodily pain |
355 |
54 (30) |
252 |
62 (28) |
181 |
66 (29) |
1.64 (0.87; 2.41) |
<0.0001 |
General health |
355 |
32 (8.3) |
252 |
34 (9) |
181 |
35 (10) |
0.54 (0.29; 0.78) |
<0.0001 |
Vitality |
355 |
37 (22) |
252 |
44 (22) |
181 |
49 (22) |
1.62 (1.10; 2.14) |
<0.0001 |
Social functioning |
355 |
55 (29) |
252 |
63 (26) |
181 |
63 (28) |
1.33 (0.63; 2.03) |
0.0002 |
Emotional functioning |
355 |
32 (42) |
252 |
38 (45) |
181 |
41 (45) |
1.52 (0.43; 2.62) |
0.007 |
Mental health |
355 |
58 (21) |
252 |
63 (19) |
181 |
65 (21) |
1.07 (0.60; 1.53) |
<0.0001 |
EORTC QLQ-C30 |
||||||||
Global health status /QOL |
351 |
47 (24) |
250 |
56 (23) |
178 |
61 (24) |
2.25 (1.63; 2.88) |
<0.0001 |
Physical functioning |
348 |
66 (27) |
249 |
72 (22) |
178 |
72 (24) |
0.74 (0.12; 1.37) |
0.02 |
Role functioning |
343 |
53 (37) |
248 |
64 (33) |
178 |
66 (33) |
1.93 (1.03; 2.84) |
<0.0001 |
Emotional functioning |
353 |
67 (26) |
250 |
73 (24) |
179 |
77 (23) |
1.51 (0.93; 2.10) |
<0.0001 |
Cognitive functioning |
352 |
76 (26) |
250 |
82 (21) |
179 |
82 (24) |
0.87 (0.31; 1.42) |
0.002 |
Social functioning |
351 |
60 (32) |
250 |
69 (29) |
179 |
74 (30) |
1.99 (1.18; 2.79) |
<0.0001 |
Fatigue |
344 |
55 (31) |
249 |
43 (28) |
176 |
36 (29) |
–2.78 (–3.53; –2.03) |
<0.0001 |
Nausea and vomiting |
346 |
17 (26) |
249 |
13 (24) |
177 |
9.3 (19) |
–1.11 (–1.75; –0.47) |
0.0008 |
Pain |
354 |
35 (33) |
249 |
26 (27) |
180 |
24 (29) |
–1.68 (–2.49; –0.87) |
<0.0001 |
Dyspnea |
343 |
36 (37) |
246 |
28 (33) |
177 |
23 (28) |
–1.92 (–2.68; –1.15) |
<0.0001 |
Insomnia |
342 |
39 (36) |
246 |
28 (31) |
176 |
25 (30) |
–2.26 (–3.15; –1.38) |
<0.0001 |
Appetite loss |
345 |
38 (39) |
248 |
24 (33) |
177 |
19 (29) |
–3.12 (–4.04; –2.21) |
<0.0001 |
Constipation |
350 |
26 (33) |
248 |
18 (25) |
178 |
18 (28) |
–1.39 (–2.15; –0.62) |
0.00041 |
Diarrhea |
351 |
21 (31) |
249 |
18 (28) |
176 |
15 (26) |
–0.91 (–1.70; –0.12) |
0.02 |
Financial difficulties |
349 |
12 (25) |
248 |
10 (23) |
178 |
7.9 (21) |
–0.55 (–1.02; –0.08) |
0.02 |
Veines-QOL |
||||||||
VEINES-QOL |
340 |
50 (10) |
240 |
50 (10) |
168 |
50 (10) |
–0.20 (–0.43; 0.04) |
0.10 |
VEINES-Sym |
344 |
50 (10) |
241 |
50 (10) |
169 |
50 (10) |
–0.08 (–0.33; 0.16) |
0.52 |
Random-effects linear regression model with time as a covariate was performed on the QoL outcomes in the three questionnaires (MOS SF-36, EORTC QLQ-C30, VEINES-QOL). Estimated change in QoL scores per month (95% confidence Intervals) and corresponding p-values are provided.
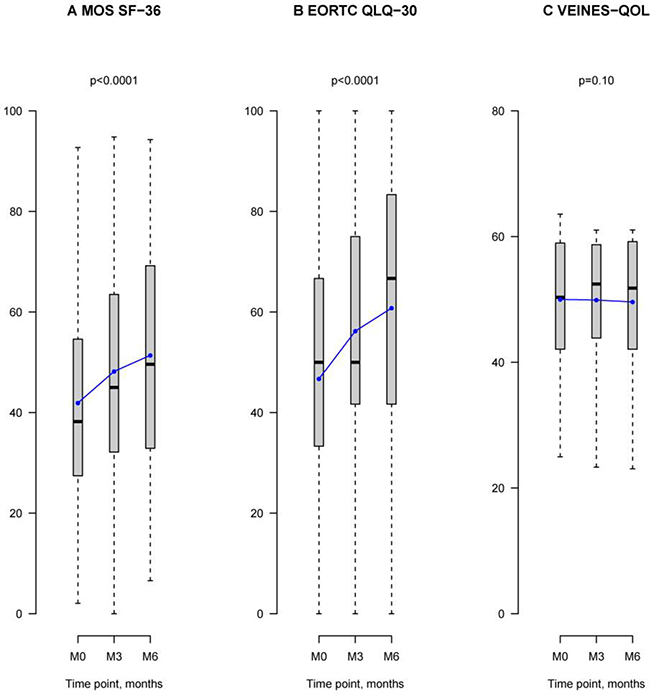
Figure 3: QoL assessments at 0, 3, and 6-month follow-ups in patients treated with LMWH. Grey box-and-whisker plots show the median, interquartile, and overall spread of all the QoL data at M0, M3, and M6. Blue linear plots illustrate the mean of the distribution of QoL scores at each observation period M0, M3, and M6. (A) Graph illustrates health-related quality of life (HRQoL) scores in the MOS SF-36 survey. At M0, HRQoL mean score was 42 with a median score of 38 [27; 55]. At M3, the mean score was 48 with a median of 45 [32; 63]. At M6, the mean score was 51 with a median of 50 [33; 69]. HRQoL scores increased significantly over the 6-month observation period (P < 0.0001). (B) Graph shows global health status/QoL scores in the EORTC QLQ-C30 questionnaire which assessed cancer-related QoL. At M0, the mean QoL score was 47, with a median of 50 [33; 67]. At M3, the mean QoL score was 56, with a median of 50 [42; 75]. At M6, the mean score was 61, with a median of 67 [44; 83]. Global health status/QoL scores significantly changed over the 6-month observation period (P < 0.0001). (C) Graph shows VEINES-QOL scores. At M0, the mean VEINES-QOL score was 50, with median of 50 [42; 59]. At M3, the mean VEINES-QOL score was 50, with a median of 52 [44; 59]. At M6, the mean of VEINES-QOL scores was 50, with a median of 52 [42; 59]. VEINES-QOL scores did not change during the 6-month LMWH treatment period.
In the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30cancer-related QoL survey, global Health status/QoL significantly increased 2.25-fold [95% CI 1.63–2.88] per month over the 6-month study period (P < 0.0001) (Table 2, Figure 3). Underlying improvements across all functioning and symptom scales of the EORTC QLQ-C30 survey were observed (Table 2). Compared to scores at M0, global Health status/QoL scores increased by a mean 6.6 (26) points at 3-month follow-up, and by 11 (29) points at 6-month follow-up (Supplementary Table 2).
In the Venous Insufficiency Epidemiological and Economic Study (VEINES)-QOL questionnaire, [21–23] scores did not significantly change during the study (-0.20-fold [95% CI–0.43–0.04]; p = 0.10) (Table 2). This was also the case in the symptom-specific scale of the questionnaire, VEINES-Sym (-0.08-fold [95% CI–0.33–0.16]; p = 0.52).
Predictors of QoL outcomes in cancer patients with established VTE receiving Long-term LMWH
Independent predictors of QoL outcomes in the MOS SF-36 and the EORTC QLQ-C30 questionnaires during the study period were identified (Supplementary Tables 3–5). In the MOS SF-36 survey, decreased functional activity at inclusion (ie.: bedridden; = 0.016) and symptomatic as opposed to asymptomatic VTE (P < 0.001) were independent predictors of improvement in HRQoL scores (Supplementary Tables 3, 4). Cancer dissemination (local, regional, metastatic; p = 0.022) and cancer histological type (adenocarcinoma, squamous, neuroendocrine, sarcoma, other; p = 0.001) were also predictors of QoL outcomes (Supplementary Tables 3, 4).
In the EORTC QLQ-C30, presence of the following factors at inclusion were independent predictors of improvement in global Health status/QoL scores over the 6-month study period: decreased functional activity measured by ECOG scores (ie.: 2, 3, 4 versus 0, 1 score, p = 0.011), symptomatic as opposed to asymptomatic VTE (p = 0.045), presence of acute infection (p = 0.033), and not undergoing anti-angiogenic therapy (p = 0.039) (Supplementary Tables 3 and 5). Cancer dissemination (p = 0.044) and cancer histology (p = 0.002) were also associated with QoL outcomes in the cancer-specific EORTC QLQ-C30 (Supplementary Tables 3, 5).
DISCUSSION
QUAVITEC is the first study to assess HRQoL in cancer patients with acute VTE undergoing anticoagulant treatment over six months. In this independent prospective observational study, QoL was assessed in a real-world setting with anticoagulant therapy determined by expert treating clinicians who were members of the Groupe Francophone Thrombose et Cancer (GFTC), which aims to improve good clinical practices for the treatment of VTE in cancer patients. The majority of patients were treated with LMWH (88.8%), consistent with evidence-based clinical practice guidelines. In the MOS SF-36 questionnaire, significant improvements in HRQoL were observed over the 6-month study period in patients receiving LMWH, relative to their respective scores at inclusion. The EORTC QLQ-C30 survey similarly indicated a robust significant increase in cancer-related global health status/QoL over time in patients on LMWH. Studies estimating the minimal important differences (MID) in QoL scores suggest that the significant increases in QoL observed here in the MOS SF-36 and EORTC QLQ-C30 questionnaires are clinically meaningful. Studies in cancer patients have estimated that the MID in the EORTC QLQ-C30 questionnaire to range between 6 and 15 points [17, 24, 25] and that statistically significant changes in scores that are less than 6 are unlikely to be of clinical significance for patients [26]. In the MOS SF-36, the physical and mental health summary scores we observed also exceeded reported estimated MID (3–5 points) [27]. Scores in the disease-specific VEINES-QOL survey, which assesses QoL and symptom burden in patients with DVT, did not change during the 6-month study observation period. The Kaplan–Meier survival analysis indicated that patients treated with LMWH had non-significant better survival outcomes than patients receiving other anticoagulant therapies. However, the number of patients receiving treatments other than LMWH were few (n = 41). Furthermore, these data are difficult to interpret because the patients on other treatments, were patients in whom LMWH was contra-indicated and therefore likely to be more medically ill, such as hospitalized patients on iv unfractionated heparin, or patients with severe renal insufficiency on warfarin.
ECOG scores indicate that at follow-up the surviving study population had shifted toward improved health, with the percentage of patients with ECOG scores of 0 or 1 increasing from 62.5% at M0 to 70.6% at M6 (Figure 2C). The observation that QoL improved with increasing ECOG scores over the six-month study period suggests that the LMWH treatment regimen did not have a negative impact on QoL. In our study, 18.9% of patients on LMWH reported at least one side effect of injection, and 7.3% reported experiencing pain associated with LMWH injection. There is the possibility that painful side effects associated with daily LMWH injections may have an impact on treatment compliance in certain patients. However, an international qualitative survey of anticoagulant healthcare providers and their patients reported that physicians underestimate patient willingness to follow long-term treatment regimens that require injection, when it is the optimal treatment choice [13]. A later study assessed which features of anticoagulant treatment are most important to patients in making anticoagulant treatment decisions. Patients reported that avoiding interference with their cancer treatment was the most important factor (39%). This was followed by anticoagulant safety and efficacy (ie.: low thrombosis rate (24%) and minimizing bleed risk (19%)), which were rated to be of greater importance over convenience of route of administration (oral over injection, 13%) [28]. In the TROPIQUE study, patients reported that long-term LMWH was very convenient (mean score, 79.7 (SD:17.1)), and treatment satisfaction was relatively high (62.9 (SD:16.7)), particularly on measures of reassurance about treatment efficacy and experience with side effects. However, 23.4% of patients did report being unsatisfied or very unsatisfied with daily LMWH injections, indicating that a subpopulation of patients may significantly benefit from oral anticoagulant treatment as appropriate options become available. Studies in non-cancer patients are confirming that DOACs are associated with improved QoL compared to LMWH and VKA [29]. Although DOACS may be effective and safe in cancer patients, dedicated studies are needed before DOACS can be used with certainty in the cancer patient population. At present, LMWH is the appropriate first-line choice for the majority of cancer patients with VTE [2]. Our results indicate that HRQoL, as assessed by the MOS SF-36, EORTC QLQ-C30, and VEINES-QOL surveys, is not negatively affected by the use of LMWH; HRQoL increased in surviving patients despite LMWH treatment.
Our study had a number of limitations. This independent prospective observational real-life study could not be randomized due to the fact that this patient population required anticoagulant treatment with LMWH, unless contra-indicated, according to evidence-based clinical practice guidelines. While the drop-out rate was relatively low (32 of 400), a significant proportion of patients died over the 6-month study period, mainly due to cancer progression (23.7%). Notably, the mortality rate observed in our study was similar to the rates reported in the CLOT and CATCH trials, which also had 6-month follow-ups. ECOG scores improved in the group of surviving patients over the study period. Thus, QoL improved with ECOG scores, suggesting that LMWH did not hinder an increased QoL associated with a better global health.
MATERIALS AND METHODS
Patients
In this prospective, longitudinal, observational multicenter study, all consecutive cancer patients (>18 years), objectively diagnosed with symptomatic or asymptomatic deep vein thrombosis (DVT), pulmonary embolism (PE), or superficial vein thrombosis, were recruited at 22 participating centers from the Groupe Francophone Thrombose and Cancer (www.thrombose-cancer.com) between February 2011 and 2012. Patients with less than 3 months life-expectancy or with unfeasible follow-up, those incapable of answering the questionnaires or who did not provide written informed consent were excluded from the study. The protocol was approved by the ethics committees of each participating center, according to local laws.
Procedures
The study primary objective was to evaluate QoL in cancer patients at the time of VTE diagnosis and start of anticoagulant therapy (M0), after 3 (M3) and 6 (M6) months of anticoagulant treatment for objectively confirmed VTE. Secondary objectives were survival after 3 and 6 months of anticoagulant therapy. Anticoagulant treatment was determined by the attending physicians, based on patient characteristics and comorbidities, and consistent with evidence-based clinical practice guidelines, most patients were under long-term LMWH, unless contraindicated.
Three standardized questionnaires, all previously translated and comprehensively validated into different languages, including French [18–23], were used to measure generic and disease-specific QoL (Supplementary Materials).
In the Medical Outcome Study 36-item Short-Form Health Survey (MOS SF-36), [18], for generic Health-Related Quality of Life (HRQoL), generates 8 subscales (from scores of 0 to 100) and two summary scores that measure physical health (physical component score (PCS)) and mental health (mental component score (MCS)) status. The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) [20] is used to assess QoL in cancer patients, through 9 multi-item scales that span three dimensions: functional, symptoms and a global health status and quality-of-life scale (HRQoL). The Venous Insufficiency Epidemiological and Economic Study (VEINES)-QOL questionnaire, for venous disease-specific QOL, is a 26-item questionnaire measuring the global impact of chronic venous disease [21] or recent DVT [22] on QoL with questions on the intensity and severity (VEINES-Sym subscale) and the impacts of venous symptoms [21, 23].
Statistical analysis
Categorical variables were reported as frequencies with percentages and continuous variables as means with standard deviations (SD). To assess the impact of time on QoL scores, random-effects linear regression models with time as covariate were used. Patients’ random effects were considered on the intercept and on the time coefficients to take in account the intra-patient correlations. Impact of patient characteristics on change in QoL were assessed using a random-effects linear regression model including a cross-level interaction term for each characteristic and time. Interaction terms for binary characteristics were tested using Wald tests, while interaction terms for non-binary qualitative characteristics were globally tested using likelihood ratio tests. All analyses were performed in the group of patients treated by LWMH only. The Kaplan Meier estimator was used to estimate the probability of survival and the log-rank test to compare survival between LMWH and other treatments. All statistical tests were two-sided and a significance level of α = 0.05 was applied. Analyses were run using R 2.15.2 [30].
CONCLUSIONS
VTE in patients with cancer has a negative impact on morbidity and survival, and appropriate anticoagulant therapy is crucial for improving health outcomes in these patients. Despite this, studies continue to consistently show that LMWH is under-utilized in cancer patients. One barrier to conforming to evidence-based clinical practice guidelines has been physician concern about the burden of LMWH injections on QoL. Our study findings show that LMWH did not hinder QoL improvements in cancer patients who survived to 6-month follow-up and exhibited increased health overall. Given how crucial anticoagulant treatment is to decreasing morbidity and mortality, these data contribute to dispelling concerns about the negative impact of LMWH treatment regimens on overall patient well-being and QoL.
Author contributions
DF designed the study and wrote the manuscript; NF, TB, FC, OB, LV, HC, IB, DPP, MC, VL, and FC did clinical follow-up of patients and provided data for the study; MRR performed statistical analysis; IB collected data and helped with data management; HR and JC reviewed and edited the manuscript, and helped with submission. All authors read and approved the manuscript.
ACKNOWLEDGMENTS
The authors wish to thank Bouthayna T’Batou and Kristelle Dessaux for their technical assistance in data monitoring and statistical analysis.
CONFLICTS OF INTEREST
All authors declare that they have no conflicts of interest.
FUNDING
This work was supported by the “Groupe Francophone Thrombose et Cancer”.
REFERENCES
1. Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013; 122:1712–23.
2. Farge D, Bounameaux H, Brenner B, Cajfinger F, Debourdeau P, Khorana AA, Pabinger I, Solymoss S, Douketis J, Kakkar A. International clinical practice guidelines including guidance for direct oral anticoagulants in the treatment and prophylaxis of venous thromboembolism in patients with cancer. Lancet Oncol. 2016; 17:e452–e66.
3. Lyman GH, Bohlke K, Khorana AA, Kuderer NM, Lee AY, Arcelus JI, Balaban EP, Clarke JM, Flowers CR, Francis CW, Gates LE, Kakkar AK, Key NS, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update 2014. J Clin Oncol. 2015; 33:654–6.
4. Kearon C, Akl EA, Ornelas J, Blaivas A, Jimenez D, Bounameaux H, Huisman M, King CS, Morris TA, Sood N, Stevens SM, Vintch JRE, Wells P, et al. Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. Chest. 2016; 149:315–52.
5. Meyer G, Marjanovic Z, Valcke J, Lorcerie B, Gruel Y, Solal-Celigny P, Le Maignan C, Extra JM, Cottu P, Farge D. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002; 162:1729–35.
6. Lee AY, Levine MN, Baker RI, Bowden C, Kakkar AK, Prins M, Rickles FR, Julian JA, Haley S, Kovacs MJ, Gent M. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003; 349:146–53.
7. Hull RD, Pineo GF, Brant RF, Mah AF, Burke N, Dear R, Wong T, Cook R, Solymoss S, Poon MC, Raskob G. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006; 119:1062–72.
8. Rabinovich E, Bartholomew JR, Wilks ML, Tripp BL, McCrae KR, Khorana AA. Centralizing care of cancer-associated thromboembolism: The Cleveland Clinic experience. Thromb Res. 2016; 147:102–3.
9. Sevestre MA, Belizna C, Durant C, Bosson JL, Vedrine L, Cajfinger F, Debourdeau P, Farge D. Compliance with recommendations of clinical practice in the management of venous thromboembolism in cancer: the CARMEN study. J Mal Vasc. 2014; 39:161–8.
10. Mahe I, Sterpu R, Bertoletti L, Lopez-Jimenez L, Mellado Joan M, Trujillo-Santos J, Ballaz A, Hernández Blasco LM, Marchena PJ, Monreal M; RIETE Investigators. Long-Term Anticoagulant Therapy of Patients with Venous Thromboembolism. What Are the Practices? PLoS One. 2015; 10:e0128741.
11. Khorana AA, Yannicelli D, McCrae KR, Milentijevic D, Crivera C, Nelson WW, Schein JR. Evaluation of US prescription patterns: Are treatment guidelines for cancer-associated venous thromboembolism being followed? Thromb Res. 2016; 145:51–3.
12. Khorana A, McCrae K, Milentijevic D, Fortier J, Nelson W, Laliberté F, Crivera C, Lefebvre P, Yannicelli D, Schein J. Current practice patterns and patient persistence on anticoagulant treatments for cancer-associated thrombosis. 2015 ASH Annual Meeting. 2015; Abstract 626.
13. Noble SI, Finlay IG. Is long-term low-molecular-weight heparin acceptable to palliative care patients in the treatment of cancer related venous thromboembolism? A qualitative study. Palliat Med. 2005; 19:197–201.
14. Klerk CP, Smorenburg SM, Otten HM, Lensing AW, Prins MH, Piovella F, Prandoni P, Bos MM, Richel DJ, van Tienhoven G, Büller HR. The effect of low molecular weight heparin on survival in patients with advanced malignancy. J Clin Oncol. 2005; 23:2130–5.
15. Hadley SA, Chang M, Rogers K. Effect of syringe size on bruising following subcutaneous heparin injection. Am J Crit Care. 1996; 5:271–6.
16. Food and Drug Administration. Guidance for Industry. Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims 2009. Available from: https://www.fda.gov/downloads/drugs/guidances/ucm193282.pdf.
17. Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998; 16:139–44.
18. Ware J, Kosinski M, Keller S. SF-36 Physical and Mental Health Summary Scales: A User’s Manual. Boston, MA: The Health Institute; 1994.
19. Leplege A, Ecosse E, Verdier A, Perneger TV. The French SF-36 Health Survey: translation, cultural adaptation and preliminary psychometric evaluation. J Clin Epidemiol. 1998; 51:1013–23.
20. Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman SB, de Haes JC, Kaasa S, Klee M, Osoba D. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993; 85:365–76.
21. Lamping DL, Schroter S, Kurz X, Kahn SR, Abenhaim L. Evaluation of outcomes in chronic venous disorders of the leg: development of a scientifically rigorous, patient-reported measure of symptoms and quality of life. J Vasc Surg. 2003; 37:410–9.
22. Kahn SR, Ducruet T, Lamping DL, Arsenault L, Miron MJ, Roussin A, Desmarais S, Joyal F, Kassis J, Solymoss S, Desjardins L, Johri M, Shrier I. Prospective evaluation of health-related quality of life in patients with deep venous thrombosis. Arch Intern Med. 2005; 165:1173–8.
23. van Korlaar I, Vossen C, Rosendaal F, Cameron L, Bovill E, Kaptein A. Quality of life in venous disease. Thromb Haemost. 2003; 90:27–35.
24. Jayadevappa R, Malkowicz SB, Wittink M, Wein AJ, Chhatre S. Comparison of distribution- and anchor-based approaches to infer changes in health-related quality of life of prostate cancer survivors. Health Serv Res. 2012; 47:1902–25.
25. Maringwa JT, Quinten C, King M, Ringash J, Osoba D, Coens C, Martinelli F, Vercauteren J, Cleeland CS, Flechtner H, Gotay C, Greimel E, Taphoorn MJ, et al. Minimal important differences for interpreting health-related quality of life scores from the EORTC QLQ-C30 in lung cancer patients participating in randomized controlled trials. Support Care Cancer. 2011; 19:1753–60.
26. Kvam AK, Fayers P, Wisloff F. What changes in health-related quality of life matter to multiple myeloma patients? A prospective study. Eur J Haematol. 2010; 84:345–53.
27. Hays RD, Morales LS. The RAND-36 measure of health-related quality of life. Ann Med. 2001; 33:350–7.
28. Noble S, Matzdorff A, Maraveyas A, Holm MV, Pisa G. Assessing patients’ anticoagulation preferences for the treatment of cancer-associated thrombosis using conjoint methodology. Haematologica. 2015; 100:1486–92.
29. Monreal M, Folkerts K, Diamantopoulos A, Imberti D, Brosa M. Cost-effectiveness impact of rivaroxaban versus new and existing prophylaxis for the prevention of venous thromboembolism after total hip or knee replacement surgery in France, Italy and Spain. Thromb Haemost. 2013; 110:987–94.
30. R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing 2016. Available from: https://www.R-project.org/.

