INTRODUCTION
Rhabdomyosarcoma (RMS), a high-grade tumor of soft-tissue, is divided into four subtypes: embryonal RMS (ERMS), alveolar RMS (ARMS), pleomorphic RMS and spindle cell/sclerosing RMS [1]. Among these tumors, two types of chromosomal translocations, t(2;13)(q35;q14) and t(1:13)(p36;q14), have been detected in approximately 70–80% of ARMS resulting in the formation of either PAX3-FOXO1 or PAX7-FOXO1 fusion, respectively [2]. Fusion-positive ARMSs show a worse prognosis than fusion-negative ARMSs, and ARMSs with the PAX3-FOXO1 variant have a poorer outcome than those with PAX7–FOXO1 [2–5]. These PAX3-FOXO1/PAX7-FOXO1 fusion proteins are critical transcriptional factors and are thereby considered to have a central role in the pathogenesis of ARMS; however, the functions of these fusion proteins are unclear. Several gene expression studies have identified potential relevant genes in PAX3-FOXO1-positive-ARMS [6–9]. However, the protein expression signatures associated with these fusion proteins have not been clearly demonstrated.
The serine-threonine protein phosphatase 2A (PP2A) regulates multiple cell signaling cascades and its inactivation by viral oncoproteins, the mutation of specific structural subunits, or the upregulation of the cellular endogenous inhibitors may contribute to malignant transformation by regulating specific phosphorylation events [10]. PP2A dysfunction has been implicated in various malignancies [11]. We recently demonstrated that mutations of the PPP2R1A encoding part of PP2A frequently occurred in gastrointestinal stromal tumors (GISTs) and that these mutations were associated with a poorer prognosis in GISTs [12]. Furthermore, the inactivation of PP2A by these mutations was associated with the increased phosphorylation of its substrates in GISTs [12]. The pharmacological modulation of PP2A activity is becoming an attractive strategy for cancer treatment [10]. Some compounds targeting PP2A, such as FTY720, are able to induce PP2A reactivation and subsequent cell death in several types of cancer [10, 13–15].
In the present study, to better characterize the oncogenic role of the PAX3-FOXO1 fusion protein in the acquisition of aggressive behavior in ARMS, we conducted proteomic studies using a PAX3-FOXO1 knockdown system in ARMS cell lines and in vitro assays, and found that among the protein list, proteins encoded by PPP2R1A were downregulated in the native ARMS cell lines. The expression of PPP2R1A encoding alpha-subunit of PP2A was upregulated by the transfection of siRNAs against PAX3-FOXO1 at the mRNA and protein expression levels. Furthermore, the cell growth of ARMS cells was drastically increased by the transfection of siRNAs against PPP2R1A. In addition, the activation of PP2A by FTY720 treatment in ARMS cell lines inhibited cell growth. Taken together, PPP2R1A is negatively regulated by PAX3-FOXO1 fusion protein in ARMS and the activation of PP2A may represent a new therapeutic target in ARMS.
RESULTS
Candidate proteins associated with the PAX3-FOXO1 fusion product in ARMS
Four ARMS cell lines were transfected with 2 different siRNA targeted for PAX3-FOXO1 and incubated after transfection and collected after 72 h. The suppression of the fusion gene transcript was confirmed by a quantitative reverse transcription-PCR (Figure 1A). In this knockdown assay, the growth of all ARMS cell lines was decreased, following the knockdown of PAX3-FOXO1, to approximately 60% of that in the control groups (Figure 1A and Figure 1B). Proteins derived from each transfected ARMS cell line were analyzed using the i-TRAQ method and 8 distinct protein profiles were observed, in which 1,300–2,400 proteins showed altered expression levels (Supplementary Tables 1–8). In order to narrow down the candidate proteins, we selected proteins that were significantly altered in at least 2 of the 4 ARMS cell lines. This approach narrowed down the protein lists to 107 consistently upregulated and 114 consistently downregulated proteins (Supplementary Table 9). These upregulated or downregulated protein profiles were further analyzed using the Oncomine and IPA databases. Finally, a protein list consisting of 16 upregulated and 17 downregulated proteins that were expected to be regulated by PAX3-FOXO1 fusion protein was obtained (Table 1). Among these, we focused on PPP2R1A to test the biological significance in the setting of ARMS.
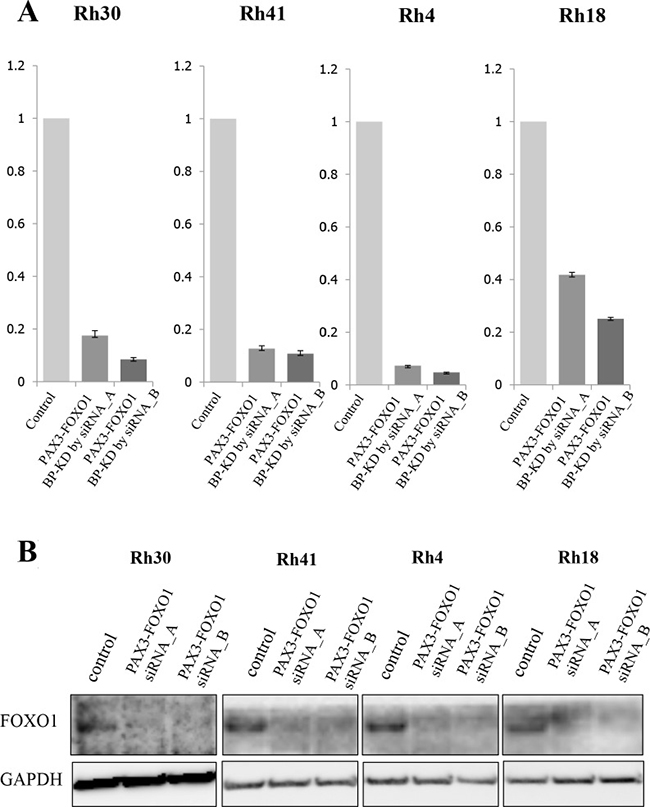
Figure 1: The expression of PAX3-FOXO1 and siRNA targeting PAX3-FOXO1 in ARMS cell lines. (A) Proteomic studies were performed using proteins extracted from four ARMS cell lines (Rh4, Rh18, Rh30 and Rh41) that were transfected with siRNAs targeting the PAX3-FOXO1 break point (BP). The quantitative PCRs (qPCRs) showed that PAX3-FOXO1 BP siRNA inhibited the mRNA expression of PAX3-FOXO1 in all four ARMS cell lines. (B) The protein expression of FOXO1 was verified by Western blotting. Western blotting of FOXO1 showed that the cell lines with PAX3-FOXO1 siRNA knockdown had lower expression levels of FOXO1 in comparison to control cells in the four ARMS cell lines.
Table 1: List of proteins related to the PAX3/FOXO3 fusion gene, determined based on data-mining
Accession |
Symbol |
Protein name |
Up/Down |
Fold difference |
P-value |
|---|---|---|---|---|---|
P80297 |
MT1X |
Metallothionein-1X |
Down |
0.47 |
3.29E-02 |
P42766 |
RPL35 |
60S ribosomal protein L35 |
Down |
0.57 |
1.98E-02 |
O75531 |
BANF1 |
Barrier-to-autointegration factor |
Down |
0.58 |
2.49E-02 |
P62753 |
RPS6 |
40S ribosomal protein S6 |
Down |
0.66 |
1.91E-02 |
P61254 |
RPL26 |
60S ribosomal protein L26 |
Down |
0.68 |
9.97E-03 |
P22087 |
FBL |
rRNA 2'-O-methyltransferase fibrillarin |
Down |
0.70 |
3.18E-02 |
P00367 |
GLUD1 |
Glutamate dehydrogenase 1, mitochondrial |
Down |
0.71 |
2.79E-02 |
Q02878 |
RPL6 |
60S ribosomal protein L6 |
Down |
0.72 |
1.74E-02 |
Q15233 |
NONO |
Non-POU domain-containing octamer-binding protein |
Down |
0.76 |
3.26E-02 |
P62633 |
CNBP |
Cellular nucleic acid-binding protein |
Down |
0.76 |
3.75E-02 |
P07900 |
HSP90AA1 |
Heat shock protein HSP 90-alpha |
Down |
0.78 |
1.95E-03 |
P08107 |
HSPA1A |
Heat shock 70 kDa protein 1A/1B |
Down |
0.81 |
1.50E-04 |
P06753 |
TPM3 |
Isoform 2 of Tropomyosin alpha-3 chain |
Down |
0.81 |
1.54E-02 |
Q99615 |
DNAJC7 |
DnaJ homolog subfamily C member 7 |
Down |
0.83 |
4.76E-02 |
P17987 |
TCP1 |
T-complex protein 1 subunit alpha |
Down |
0.84 |
2.25E-02 |
P48643 |
CCT5 |
T-complex protein 1 subunit epsilon |
Down |
0.86 |
1.80E-02 |
P13639 |
EEF2 |
Elongation factor 2 |
Down |
0.93 |
4.50E-02 |
Q14204 |
DYNC1H1 |
Cytoplasmic dynein 1 heavy chain 1 |
Up |
1.17 |
1.47E-02 |
P51858 |
HDGF |
Hepatoma-derived growth factor |
Up |
1.19 |
1.72E-02 |
P12956 |
XRCC6 |
X-ray repair cross-complementing protein 6 |
Up |
1.24 |
2.25E-02 |
Q13098 |
GPS1 |
COP9 signalosome complex subunit 1 |
Up |
1.27 |
2.49E-02 |
Q9BSJ8 |
ESYT1 |
Extended synaptotagmin-1 |
Up |
1.28 |
2.75E-03 |
P15121 |
AKR1B1 |
Aldose reductase |
Up |
1.29 |
1.58E-02 |
Q14315 |
FLNC |
Filamin-C |
Up |
1.31 |
7.14E-04 |
P07237 |
P4HB |
Protein disulfide-isomerase |
Up |
1.32 |
7.10E-03 |
P08758 |
ANXA5 |
Annexin A5 |
Up |
1.32 |
2.37E-02 |
P30153 |
PPP2R1A |
Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform |
Up |
1.37 |
1.60E-02 |
P37268 |
FDFT1 |
Squalene synthase |
Up |
1.43 |
4.10E-03 |
P40123 |
CAP2 |
Adenylyl cyclase-associated protein 2 |
Up |
1.43 |
6.33E-03 |
P52209 |
PGD |
6-phosphogluconate dehydrogenase, decarboxylating |
Up |
1.47 |
3.38E-03 |
O00299 |
CLIC1 |
Chloride intracellular channel protein 1 |
Up |
1.52 |
1.54E-02 |
P08962 |
CD63 |
CD63 antigen |
Up |
1.59 |
2.76E-02 |
P07108 |
DBI |
Acyl-CoA-binding protein |
Up |
1.64 |
4.10E-02 |
PPP2R1A inhibition by siRNA significantly promotes cell proliferation in ARMS cell lines
Before characterizing the role of PPP2R1A encoded by PPP2R1A in RMS cells, we confirmed the proteomics findings. PAX3-FOXO1 silencing upregulated the expression of PPP2R1A at both the mRNA and protein levels (Figure 2A). Next, we performed cell proliferation assays in ARMS cell lines using siRNA against PPP2R1A. This silencing of PPP2R1A significantly increased the cell growth of all ARMS cells (Figure 2B). These findings suggest that PPP2R1A has a tumor suppressive function in ARMS cells; however, the native expression of PPP2R1A was low in the presence of PAX3-FOXO1.
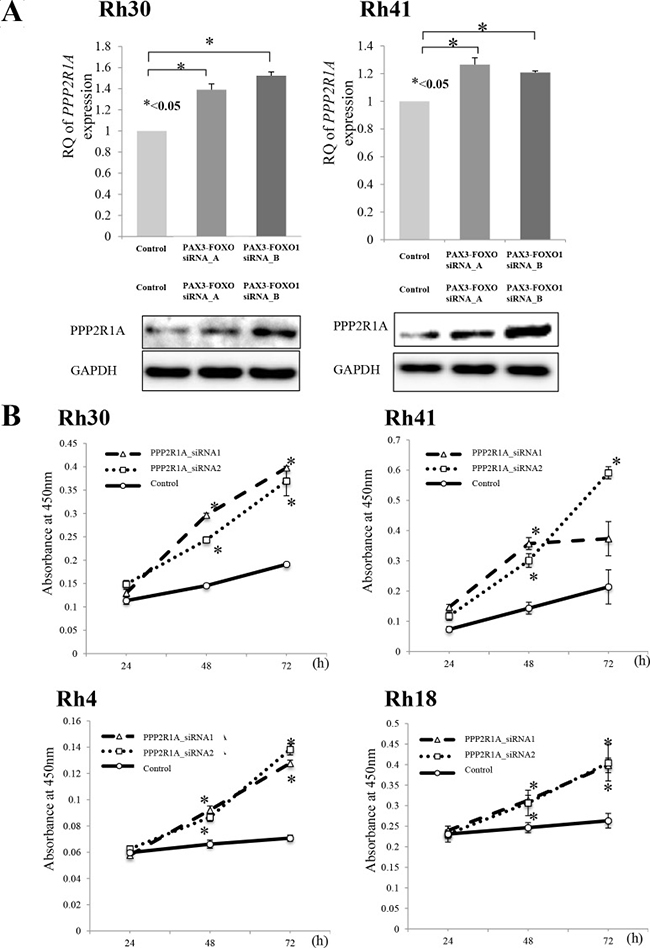
Figure 2: (A) Silencing PAX3-FOXO1 activated the expression of PPP2R1A and (B) cell viability following PPP2R1A knockdown in ARMS cell lines. (A) To investigate the association between PAX3-FOXO1 and PPP2R1A, siRNA PAX3-FOXO1 was performed and the mRNA and protein expression levels were measured by both a q-PCR and Western blotting. The silencing of PAX3-FOXO1 activated the expression of PPP2R1A in ARMS cell lines (Rh31 and Rh40); this was confirmed by both the mRNA and protein levels. (B) PPP2R1A siRNA knockdown in ARMS cell lines (Rh31 and Rh40) was performed to verify the associations between the PPP2R1A expression and cell viability. PPP2R1A siRNA suppressed the expression of PPP2R1A in ARMS cell lines and the silencing of PPP2R1A activated the cell viability in all four ARMS cell lines.
FTY720, a PP2A-activating drug, significantly inhibits the cellular proliferation of ARMS cell lines
Because it has been shown that FTY-720 reactivates PPP2R1A via SET that inhibits the function of PP2A [16], we first confirmed that SET was strongly expressed in ARMS cells (Figure 3A). We then checked whether the expression of SET could be affected by the PAX3-FOXO1 inhibition in ARMS cells. This experiment demonstrated that the SET expression significantly decreased according to the knockdown of PAX3-FOXO1 fusion (Figure 3A). This result also confirmed accuracy of our proteomic studies, as our proteomic profiles, obtained by silencing PAX3-FOXO1, also identified SET as one of the down regulated proteins (Supplementary Table 9). Next, we tried to see whether FTY720, a PP2A-activating drug, could affect the cellular proliferation in ARMS cells. This assay suppressed the cell growth in a dose-dependent manner in all ARMS cell lines and proved that FTY720 was effective under the presence of PAX3-FOXO1 fusion in ARMS (Figure 3B).
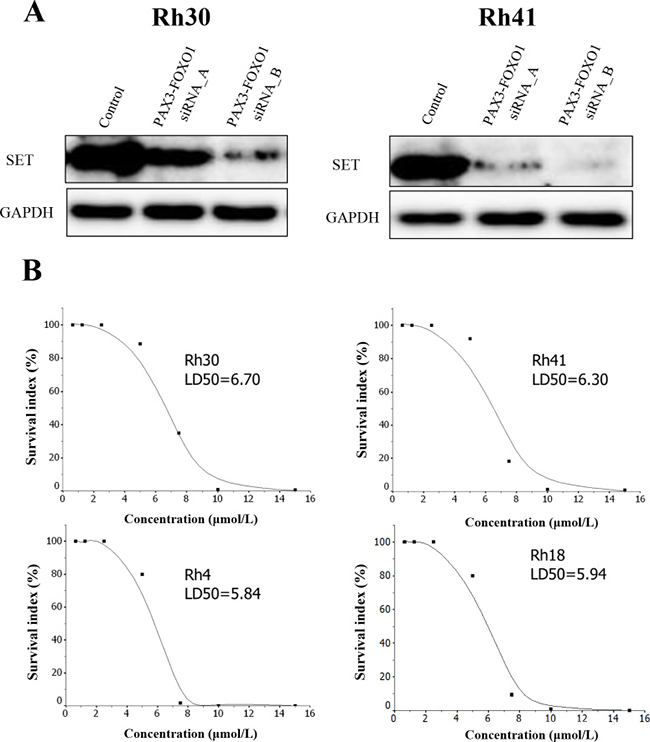
Figure 3: (A) The silencing of PAX3-FOXO1 suppressed the expression of SET and (B) the cell viability curve of FTY720 in 4 ARMS cell lines. (A) PAX3-FOXO1 siRNA knockdown in ARMS cell lines (Rh31 and Rh40) was performed to verify the association between the expression of PAX3-FOXO1 and SET. PAX3-FOXO1 siRNA suppressed the expression of SET in the two ARMS cell lines; this was confirmed by Western blotting. (B) The cell viability of FTY720 was investigated in four ARMS cell lines. FTY720 significantly inhibited the cell viability in all ARMS cell lines in a dose-dependent manner.
Human phospho-kinase array analysis
To identify the cellular signaling pathways that were activated in association with FTY-720 in ARMS cells, we performed a human phospho-kinase array analysis of 46 specific Ser/Thr and Tyr phosphorylation sites on 39 selected proteins. eNOS, AKT1/2/3, RSK1/2/3 and STAT3 phosphorylation were significant decreased following FTY-720 treatment (<55%) (Supplementary Figure 1).
DISCUSSION
Most ARMSs are characterized by chromosomal translocation—either t(2;13)(q35;q14) or t(1:13)(p36;q14)—leading to the formation of PAX3-FOXO1 or PAX7-FOXO1 fusion, respectively. It has been suggested that these PAX3-FOXO1/PAX7-FOXO1 fusion proteins have a central role in the pathogenesis of ARMS; however, the function of these fusion proteins remains unclear. In this regard, the previous studies demonstrated that PAX3-FOXO1 alone was not sufficient for tumorigenesis and that additional changes, such as increased MYCN and the expression of TERT were required [17, 18]. In addition, PAX3-FOXO1 alone did not transform immortalized human myoblasts in culture, and induced slow-growing tumors in immunocompromised mice [3, 19]. However, the combination of PAX3-FOXO1 and MYCN transformed the myoblasts and induced rapid tumorigenesis [19]. In this study, the knockdown of PAX3-FOXO1 in ARMS cell lines reduced the cell proliferation to approximately 60% in comparison to the control, which was partly in line with the previous reports suggesting that PAX3-FOXO1 alone is not responsible for the proliferative advantage. In sarcoma cell lines, the silencing of EWS-Fli1 [20] and PAX3-FOXO1 [21] induces the suppression of cell growth to almost the same level; however, the knockdown of KIT in GIST cells was associated with strong growth suppression (unpublished data). This growth inhibitory effect of gene silencing in ARMS seems to be insufficient for a single therapeutic target, and the drug delivery problem remains to be solved in this system. We therefore conducted a proteomic analyses of ARMS to characterize the protein profile associated with PAX3-FOXO1 fusion with cross-reference to the gene expression data, with the aim of identifying efficient therapeutic targets in this tumor that could be treated using easy methods of administration (i.e., oral administration). With this background, we focused on the PPP2R1A coding part of PP2A as a possible new therapeutic target in ARMSs. In this study, we found that phosphatase activity by PP2A was strongly repressed in this tumor; that is, the PPP2R1A expression was suppressed and that expression of SET, a PP2A suppressor, was upregulated by PAX3-FOXO1 in ARMS. Additionally, in our proteomic profiles regarding suppressing of PAX3-FOXO1, we successfully identified SET protein as a down regulated protein (Supplementary Table 9). Therefore, we believe that this concordance also indicated accuracy in our proteomic analyses.
PP2A consists of several isoforms, including structure (A) subunits, regulatory (B) subunits, and catalyst (C) subunits [22–24]. The regulatory (B) subunit has several subunit types [22–24]. Previous articles have revealed that genomic mutations in the Aα subunit of PP2A in human tumors interferes with the binding to other B and C subunits [22–24]. With respect to the associations among these A, B, and C subunits in expression, previous reports also revealed that the levels of the B and C subunits of PP2A were markedly reduced while the PP2A-Aα expression was suppressed [22]. This was because both the B and C subunits of PP2A were unstable unless bound to the A subunit PP2A [22]. Therefore, previous studies regarding the PP2A functions concluded that the expression of PPP2R1A were suppressed by PAX3/FOXO1 siRNA in ARMS cell lines might lead to lower expression of both the B and C subunits of PP2A.
FTY720 is an immunomodulatory agent used as an oral therapy for multiple sclerosis [22]; it is also a PP2A-activating drug [16]. FTY720 undergoes phosphorylation (FTY720-P) by sphingosine kinase 2 (SPHK2) to act as an immunosuppressant, and binds/internalizes the sphingosine-1-phosphate receptor (S1PR1) [25]. FTY720 also selectively induces apoptosis of neoplastic but not normal cells [11]. This anticancer activity does not require phosphorylation but mostly depends on its ability to activate PP2A [11]. The anticancer activity of FTY720 depends on the interaction/sequestration of the PP2A inhibitor SET; however, conversion into FTY720-P is not required [16]. In this study, the addition of FTY720 to ARMS cells drastically suppressed the cell growth in all 4 ARMS cell lines in a dose-dependent manner, confirming that SET surely disturbs the function of PP2A in ARMS. In addition to the expression of PPP2R1A. which encodes PP2A, we could confirm that SET is strongly expressed in ARMS cells and that the expression of SET in ARMS cell lines also decreased by the knockdown of PAX3-FOXO1. This finding suggests that the expression of SET is also positively regulated by PAX3-FOXO1 in ARMS. Because it has been shown that SET inhibits the function of PP2A, the reduced activity of PP2A is associated with not only the reduced expression of PP2A itself but also with the increased expression of SET in PAX3-FOXO1-positive-ARMS. These findings suggest that the function of PP2A in ARMS is strongly suppressed by PAX3-FOXO1 fusion.
The human phospho-kinase array analysis after FTY-720 treatment revealed eNOS, AKT1/2/3, RSK1/2/3 and STAT3 as a substrate of PP2A. A recent study revealed that AKT1/2/3 and PAX3-FOXO1 cooperation enforces myogenic differentiation blockade as a cause of differentiation failure in ARMS [26]. Furthermore, a study reported that STAT3 supported the growth and survival of RMS cells [27]. With regard to eNOS, Cunha et al. demonstrated that sarcomas frequently expressed eNOS [28]. A previous report found that mTOR inhibitor (rapamycin) inhibits insulin-like growth factor 1 (IGF-1)-stimulated cell motility through the PP2A pathway [29]. Furthermore, those functional studies showed that rapamycin inhibited the basal or IGF-1-induced motility of Ewing sarcoma (Rh1) and ARMS (Rh30) cell lines [29]. In addition, the treatment of cells with rapamycin activated the PP2A activity and concurrently inhibited the IGF-1 stimulated phosphorylation of several critical pathways [29]. Based on our findings and those of previous studies, combination therapy with FYT720 and other anticancer agents, including kinase inhibitors, would be beneficial to ARMS patients.
Finally, we believe that these protein signatures regulated by PAX3-FOXO1 will help to elucidate the malignant progression of ARMS with PAX3-FOXO1 fusion and lead to the development of novel therapeutic strategies. However, we are aware that this approach cannot identify proteins dysregulated by genetic alterations such as mutation and amplification, which are not expected to be regulated by fusion protein. For example, previous studies show that the expression of anaplastic lymphoma kinase (ALK) in RMS is frequently observed in alveolar RMS with PAX3-FOXO1 fusion and that the expression of ALK is associated with adverse clinical outcomes [30–33]. The high expression of ALK in ARMS with PAX3-FOXO1 fusion has been shown to be due to gene amplification [30, 31, 33]. However, the ALK protein level was not affected by the manipulation of the fusion gene in ARMS cells with PAX3-FOXO1 fusion.
In summary, the downregulation of PPP2R1A by PAX3-FOXO1 fusion protein plays an important role in the acquisition of malignant potential in ARMS cells with PAX3-FOXO1 fusion. In addition, strong expression of SET, PP2A inhibitor, positively regulated by PAX3-FOXO1 fusion protein also contributes to the dysfunction of phosphatase activity and would be involved in the acquisition of aggressive behavior of ARMS in a coordinated manner.
MATERIALS AND METHODS
Cell lines
We used 4 ARMS cell lines (Rh4, Rh18, Rh30 and Rh41) with PAX3-FOXO1 fusion cultured in DMEM with 10% FBS. Rh30 and Rh41were kindly provided by Dr. Peter Houghton (The Research Institute at Nationwide Children’s Hospital, Columbus, OH, USA). Rh4 and Th18 were obtained from Dr. Thomas Look (Dana-Farber Cancer Institute, Boston, MA, USA).
Knockdown of PAX3-FOXO1 in ARMS cell lines
To evaluate the function of the endogenous PAX3-FOXO1 fusion protein in ARMS, we performed RNA interference using siRNA duplexes against PAX3-FOXO1. Briefly, for preparation of the cell line, 24h before transfection, cells at 80% confluence were trypsinized and diluted with fresh medium, without antibiotics, to 3 × 105 cells/ml, and were then transferred into either 6-well plates (2.5 ml/well) or a 96-well plate (0.1 ml/well). Transfection of 2 different siRNAs for each target of PAX3-FOXO1 break point: PAX3-FOXO1_1 (s: GCCUCUCACCUCAGAAUUCdTdT, as: GAA UUCUGAGGUGAGAGGCdTdT, Sigma-Aldrich, MO, USA) and PAX3-FOXO1_2 (s: CCUCUCACCUCAGA AUUCAdTdT, as: UGAAUUCUGAGGUGAGAGGdTdT, Sigma-Aldrich, MO, USA) [21], PPP2R1A (SASI_Hs01_00206464, SASI_Hs01_00206465, Sigma-Aldrich MO, USA), and a scrambled siRNA as a negative control (Sigma-Aldrich MO, USA) was carried out using Lipofectamine™ RNAiMAX reagent (Thermo Fisher Scientific, CA, USA) and 30 pmol of each siRNA duplex. Cells were harvested at 24, 48, 72, and 96 h after transfection, then subjected to further analyses, including a proteomic analysis, Western blotting, a proliferation assay, and a real-time quantitative PCR.
Proteomic analysis by i-TRAQ
In order to identify the protein signatures regulated by the PAX3-FOXO1 fusion gene product in ARMS and to elucidate the function of PAX3-FOXO1, we performed proteomic analyses using isobaric tags for the relative and absolute quantitation (i-TRAQ) and determined the protein profiles regulated by PAX3-FOXO1 [34, 35].
RNA extraction and real-time PCR
Total RNA was extracted from cell pellets harvested at 72 h after transfection using TRIzol Reagent (Gibco/BRL, Tokyo, Japan) according to the manufacturer’s protocol. Five micrograms of RNA of each sample were used for the subsequent reverse transcription reaction (SuperScriptII) (Thermo Fisher Scientific, CA, USA). A semi-quantitative PCR was performed for PAX3-FOXO1 and PPP2R1A using a StepOne Real-Time PCR System (Applied Biosystems, CA, USA) and the predeveloped TaqMan assay reagents for PPP2R1A (Hs00204426_m1, Applied Biosystems CA, USA) and PAX3-FOXO1 (Hs03024825_ft, Applied Biosystems CA, USA). Human TBP was used as an endogenous control (Human TBP Endogenous Control, 4333769F, Applied Biosystems CA, USA). The comparative CT (ΔΔCT) method was used for the semi-quantification of the PCR samples.
Cell proliferation assay
Cells (1 × 106) of each cell line were plated in a 100-mm-diameter culture dish with 8 ml of RPMI supplemented with 10% calf serum and antibiotics (SM and PC). The number of cells was counted after 24 h, 72 h, and 120 h using a TC20 Automated Cell Counter (BIO-RAD); quantification was performed in triplicate.
Western blotting
The proteins were extracted from four ARMS cells and were separated via SDS-PAGE and transferred to nitrocellulose membranes. The membranes were incubated with either of the following antibodies: rabbit polyclonal antibodies against PPP2R1A (dilution 1:200, Santa Cruz, sc-168), SET (dilution 1: 200, Santa Cruz, sc-18076), mouse monoclonal antibody against GAPDH (dilution 1: 500, Santa Cruz, sc-32233) and FOXO1 (dilution 1: 100, Santa Cruz, sc-374427). After incubation, the membranes were washed three times with Tris-EDTA buffer and then reacted with horseradish peroxidase-conjugated secondary antibodies (1:1,000 dilution, GE Healthcare Biosciences).
Human phospho-kinase array analysis
We performed a human phospho-kinase array analysis using a RMS cell line (Rh31). The relative phosphorylation levels of 39 selected proteins on the array were acquired using a Proteome Profiler Human Phospho-Kinase Array Kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions. The expression levels of phosphorylation proteins were quantified using the Fuji Film Multi Gauge software program (Tokyo, Japan).
Statistical analysis
The Mann–Whitney U test were used to examine differences in cellular proliferation and gene expression levels according to the knockdown of each target gene.
ACKNOWLEDGMENTS AND FUNDING
This study was supported by Grant-in-Aid from the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant Number #15H04964, #16K15670, and #15KK0353 to Y.S.; #17K08730 to T.S.; #17K10987to K.K.; 17H07098 to K.A.; #16K20070 to T.O.; #16H07186 to K.M.) and Leading Advanced Projects for Medical Innovation (LEAP) (Grant Number # JP18am0001009) and the Practical Research for Innovative Cancer Control (Grant Number # JP18ck0106252) from the Japan Agency for Medical Research and Development. The authors thank Dr. Peter Houghton (The Research Institute at Nationwide Children's Hospital, Columbus, OH, USA) and Dr. Thomas Look (Dana-Farber Cancer Institute, Boston, MA, USA) for kindly providing ARMS cell lines.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest in association with this study.
REFERENCES
1. WHO Classification of Tumors of Soft Tissue and Bone. 4th edition. Edited by Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F. International Agency for Research on Cancer, Lyon, 2013.
2. Sorensen PH, Lynch JC, Qualman SJ, Tirabosco R, Lim JF, Maurer HM, Bridge JA, Crist WM, Triche TJ, Barr FG. PAX3-FKHR and PAX7-FKHR gene fusions are prognostic indicators in alveolar rhabdomyosarcoma: a report from the children’s oncology group. J Clin Oncol. 2002; 20:2672–79. https://doi.org/10.1200/JCO.2002.03.137.
3. Pandey PR, Chatterjee B, Olanich ME, Khan J, Miettinen MM, Hewitt SM, Barr FG. PAX3-FOXO1 is essential for tumour initiation and maintenance but not recurrence in a human myoblast model of rhabdomyosarcoma. J Pathol. 2017; 241:626–37. https://doi.org/10.1002/path.4867.
4. Fredericks WJ, Galili N, Mukhopadhyay S, Rovera G, Bennicelli J, Barr FG, Rauscher FJ 3rd. The PAX3-FKHR fusion protein created by the t(2;13) translocation in alveolar rhabdomyosarcomas is a more potent transcriptional activator than PAX3. Mol Cell Biol. 1995; 15:1522–35. https://doi.org/10.1128/MCB.15.3.1522.
5. Selfe J, Olmos D, Al-Saadi R, Thway K, Chisholm J, Kelsey A, Shipley J. Impact of fusion gene status versus histology on risk-stratification for rhabdomyosarcoma: retrospective analyses of patients on UK trials. Pediatr Blood Cancer. 2017; 64:e26386. https://doi.org/10.1002/pbc.26386.
6. De Pittà C, Tombolan L, Albiero G, Sartori F, Romualdi C, Jurman G, Carli M, Furlanello C, Lanfranchi G, Rosolen A. Gene expression profiling identifies potential relevant genes in alveolar rhabdomyosarcoma pathogenesis and discriminates PAX3-FKHR positive and negative tumors. Int J Cancer. 2006; 118:2772–81. https://doi.org/10.1002/ijc.21698.
7. Davicioni E, Anderson JR, Buckley JD, Meyer WH, Triche TJ. Gene expression profiling for survival prediction in pediatric rhabdomyosarcomas: a report from the children’s oncology group. J Clin Oncol. 2010; 28:1240–46. https://doi.org/10.1200/JCO.2008.21.1268.
8. Cao L, Yu Y, Bilke S, Walker RL, Mayeenuddin LH, Azorsa DO, Yang F, Pineda M, Helman LJ, Meltzer PS. Genome-wide identification of PAX3-FKHR binding sites in rhabdomyosarcoma reveals candidate target genes important for development and cancer. Cancer Res. 2010; 70:6497–508. https://doi.org/10.1158/0008-5472.CAN-10-0582.
9. Ebauer M, Wachtel M, Niggli FK, Schäfer BW. Comparative expression profiling identifies an in vivo target gene signature with TFAP2B as a mediator of the survival function of PAX3/FKHR. Oncogene. 2007; 26:7267–81. https://doi.org/10.1038/sj.onc.1210525.
10. Carratù MR, Signorile A, De Rasmo D, Reale A, Vacca A. Pharmacological Activation of Protein Phosphatase 2 A (PP2A): A Novel Strategy to Fight Against Human Malignancies? Curr Med Chem. 2016; 23:4286–96. https://doi.org/10.2174/0929867323666161014133423.
11. Perrotti D, Neviani P. Protein phosphatase 2A: a target for anticancer therapy. Lancet Oncol. 2013; 14:e229–38. https://doi.org/10.1016/S1470-2045(12)70558-2.
12. Toda-Ishii M, Akaike K, Suehara Y, Mukaihara K, Kubota D, Kohsaka S, Okubo T, Mitani K, Mogushi K, Takagi T, Kaneko K, Yao T, Saito T. Clinicopathological effects of protein phosphatase 2, regulatory subunit A, alpha mutations in gastrointestinal stromal tumors. Mod Pathol. 2016; 29:1424–32. https://doi.org/10.1038/modpathol.2016.138.
13. Beider K, Rosenberg E, Bitner H, Shimoni A, Leiba M, Koren-Michowitz M, Ribakovsky E, Klein S, Olam D, Weiss L, Wald H, Abraham M, Galun E, et al. The Sphingosine-1-Phosphate Modulator FTY720 Targets Multiple Myeloma via the CXCR4/CXCL12 Pathway. Clin Cancer Res. 2017; 23:1733–47. https://doi.org/10.1158/1078-0432.CCR-15-2618.
14. Gstalder C, Ader I, Cuvillier O. FTY720 (Fingolimod) Inhibits HIF1 and HIF2 Signaling, Promotes Vascular Remodeling, and Chemosensitizes in Renal Cell Carcinoma Animal Model. Mol Cancer Ther. 2016; 15:2465–74. https://doi.org/10.1158/1535-7163.MCT-16-0167.
15. Smith AM, Dun MD, Lee EM, Harrison C, Kahl R, Flanagan H, Panicker N, Mashkani B, Don AS, Morris J, Toop H, Lock RB, Powell JA, et al. Activation of protein phosphatase 2A in FLT3+ acute myeloid leukemia cells enhances the cytotoxicity of FLT3 tyrosine kinase inhibitors. Oncotarget. 2016; 7:47465–78. https://doi.org/10.18632/oncotarget.10167.
16. Oaks JJ, Santhanam R, Walker CJ, Roof S, Harb JG, Ferenchak G, Eisfeld AK, Van Brocklyn JR, Briesewitz R, Saddoughi SA, Nagata K, Bittman R, Caligiuri MA, et al. Antagonistic activities of the immunomodulator and PP2A-activating drug FTY720 (Fingolimod, Gilenya) in Jak2-driven hematologic malignancies. Blood. 2013; 122:1923–34. https://doi.org/10.1182/blood-2013-03-492181.
17. Linardic CM, Downie DL, Qualman S, Bentley RC, Counter CM. Genetic modeling of human rhabdomyosarcoma. Cancer Res. 2005; 65:4490–95. https://doi.org/10.1158/0008-5472.CAN-04-3194.
18. Naini S, Etheridge KT, Adam SJ, Qualman SJ, Bentley RC, Counter CM, Linardic CM. Defining the cooperative genetic changes that temporally drive alveolar rhabdomyosarcoma. Cancer Res. 2008; 68:9583–88. https://doi.org/10.1158/0008-5472.CAN-07-6178.
19. Xia SJ, Holder DD, Pawel BR, Zhang C, Barr FG. High expression of the PAX3-FKHR oncoprotein is required to promote tumorigenesis of human myoblasts. Am J Pathol. 2009; 175:2600–08. https://doi.org/10.2353/ajpath.2009.090192.
20. Tanaka K, Iwakuma T, Harimaya K, Sato H, Iwamoto Y. EWS-Fli1 antisense oligodeoxynucleotide inhibits proliferation of human Ewing’s sarcoma and primitive neuroectodermal tumor cells. J Clin Invest. 1997; 99:239–47. https://doi.org/10.1172/JCI119152.
21. Kikuchi K, Tsuchiya K, Otabe O, Gotoh T, Tamura S, Katsumi Y, Yagyu S, Tsubai-Shimizu S, Miyachi M, Iehara T, Hosoi H. Effects of PAX3-FKHR on malignant phenotypes in alveolar rhabdomyosarcoma. Biochem Biophys Res Commun. 2008; 365:568–74. https://doi.org/10.1016/j.bbrc.2007.11.017.
22. Chen W, Arroyo JD, Timmons JC, Possemato R, Hahn WC. Cancer-associated PP2A Aalpha subunits induce functional haploinsufficiency and tumorigenicity. Cancer Res. 2005; 65:8183–92. https://doi.org/10.1158/0008-5472.CAN-05-1103.
23. Cho US, Xu W. Crystal structure of a protein phosphatase 2A heterotrimeric holoenzyme. Nature. 2007; 445:53–57. https://doi.org/10.1038/nature05351.
24. Hahn K, Miranda M, Francis VA, Vendrell J, Zorzano A, Teleman AA. PP2A regulatory subunit PP2A-B’ counteracts S6K phosphorylation. Cell Metab. 2010; 11:438–44. https://doi.org/10.1016/j.cmet.2010.03.015.
25. Ingwersen J, Aktas O, Kuery P, Kieseier B, Boyko A, Hartung HP. Fingolimod in multiple sclerosis: mechanisms of action and clinical efficacy. Clin Immunol. 2012; 142:15–24. https://doi.org/10.1016/j.clim.2011.05.005.
26. Jothi M, Nishijo K, Keller C, Mal AK. AKT and PAX3-FKHR cooperation enforces myogenic differentiation blockade in alveolar rhabdomyosarcoma cell. Cell Cycle. 2012; 11:895–908. https://doi.org/10.4161/cc.11.5.19346.
27. Chen CL, Loy A, Cen L, Chan C, Hsieh FC, Cheng G, Wu B, Qualman SJ, Kunisada K, Yamauchi-Takihara K, Lin J. Signal transducer and activator of transcription 3 is involved in cell growth and survival of human rhabdomyosarcoma and osteosarcoma cells. BMC Cancer. 2007; 7:111. https://doi.org/10.1186/1471-2407-7-111.
28. Cunha IW, Lopes A, Falzoni R, Soares FA. Sarcomas often express constitutive nitric oxide synthases (NOS) but infrequently inducible NOS. Appl Immunohistochem Mol Morphol. 2006; 14:404–10. https://doi.org/10.1097/01.pai.0000190175.98576.a3.
29. Liu L, Chen L, Luo Y, Chen W, Zhou H, Xu B, Han X, Shen T, Huang S. Rapamycin inhibits IGF-1 stimulated cell motility through PP2A pathway. PLoS One. 2010; 5:e10578. https://doi.org/10.1371/journal.pone.0010578.
30. Yoshida A, Shibata T, Wakai S, Ushiku T, Tsuta K, Fukayama M, Makimoto A, Furuta K, Tsuda H. Anaplastic lymphoma kinase status in rhabdomyosarcomas. Mod Pathol. 2013; 26:772–81. https://doi.org/10.1038/modpathol.2012.222.
31. Gasparini P, Casanova M, Villa R, Collini P, Alaggio R, Zin A, Bonvini P, Antonescu CR, Boldrini R, Caserini R, Moro M, Centonze G, Meazza C, et al. Anaplastic lymphoma kinase aberrations correlate with metastatic features in pediatric rhabdomyosarcoma. Oncotarget. 2016; 7:58903–14. https://doi.org/10.18632/oncotarget.10368.
32. Bonvini P, Zin A, Alaggio R, Pawel B, Bisogno G, Rosolen A. High ALK mRNA expression has a negative prognostic significance in rhabdomyosarcoma. Br J Cancer. 2013; 109:3084–91. https://doi.org/10.1038/bjc.2013.653.
33. Lee JS, Lim SM, Rha SY, Roh JK, Cho YJ, Shin KH, Yang WI, Kim SH, Kim HS. Prognostic implications of anaplastic lymphoma kinase gene aberrations in rhabdomyosarcoma; an immunohistochemical and fluorescence in situ hybridisation study. J Clin Pathol. 2014; 67:33–39. https://doi.org/10.1136/jclinpath-2013-201655.
34. Mukaihara K, Suehara Y, Kohsaka S, Akaike K, Tanabe Y, Kubota D, Ishii M, Fujimura T, Kazuno S, Okubo T, Takagi T, Yao T, Kaneko K, Saito T. Protein Expression Profiling of Giant Cell Tumors of Bone Treated with Denosumab. PLoS One. 2016; 11:e0148401. https://doi.org/10.1371/journal.pone.0148401.
35. Tanabe Y, Suehara Y, Kohsaka S, Hayashi T, Akaike K, Mukaihara K, Kurihara T, Kim Y, Okubo T, Ishii M, Kazuno S, Kaneko K, Saito T. IRE1α-XBP1 inhibitors exerted anti-tumor activities in Ewing’s sarcoma. Oncotarget. 2018; 9:14428–43. https://doi.org/10.18632/oncotarget.24467.

