INTRODUCTION
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma (NHL), accounting for 30% to 40% of all newly diagnosed cases [1]. DLBCL is an aggressive malignancy, very heterogeneous in genetic abnormalities, clinical features and response to treatment. This heterogeneity results in highly variable outcomes among patients [2].
Combined chemotherapy of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) is considered as the standard first line therapy for DLBCL. The application of R-CHOP has led to complete remission for 75-80% of patients [3]. However, 30-40% of patients that achieve complete remission relapse at a later stage and some patients have refractory disease. Alarmingly, those patients tend to respond poorly to additional chemotherapy lines [4].
In order to predict the outcome of patients with DLBCL, different tools have been developed. For instance, the International Prognostic Index (IPI) identifies four risk groups with predicted five-year survival ranging from 26% to 73% using different clinical factors (age, stage, serum LDH, ECOG performance status and number of extranodal sites) [5]. Nevertheless, some patients present an unfavorable course of disease despite a good prognostic index. Another molecular prognostic tool uses gene expression profiling (GEP) and immunohistochemical analysis to discriminate two molecular subtypes with different clinical outcomes independent of IPI stratification: the germinal center B-cell-like (GCB) DLBCL, and the activated B-cell-like (ABC) DLBCL, with 5-year survival rates of 60% and 35% respectively [6, 7]. However, these tools are not able to identify all the patients that will not respond to therapy and, hence, novel biomarkers to enable a better prognostic stratification are needed.
During the past decades, several novel molecular and biological candidates with diagnostic, predictive and prognostic potential in DLBCL have been suggested, including microRNAs (miRNAs). MiRNAs are small, regulatory, non-coding RNAs that can negatively regulate gene expression at the post-transcriptional level by binding to the 3´ untranslated region (UTR) of a target mRNA and leading to increased degradation or inhibition of translation [8]. Nowadays, hundreds of miRNAs have been identified as biomarkers in cancer [9–11]. Interestingly, recent evidence has emerged showing that tumor-associated miRNAs can also be detected in body fluids such as serum or plasma [12]. This is possible thanks to the fact that miRNAs are protected from degradation in serum or plasma through association with RNA-binding proteins and/or packaging inside extracellular vesicles [13], providing them with high stability. This characteristic makes it possible to use non-invasive techniques for their evaluation, which is a great advantage regarding disease monitoring. Therefore, circulating miRNAs have emerged as candidate non-invasive biomarkers for diagnosis, and prognosis in cancer [14–17].
To date, some groups have investigated the potential role of circulating microRNAs in DLBCL [18–28]. Nevertheless, these studies present controversial results. We considered that it would be of interest to clarify these discrepancies. Therefore, herein, we review the potential role of circulating miRNAs as biomarkers for diagnosis, subtype characterization, prediction of treatment response and prognosis in patients with DLBCL.
RESULTS
A total of 487 records were initially identified (Figure 1). Among them, 328 records remained after duplicates were removed. Among these records, 211 were discarded after abstract review because they clearly did not meet the required inclusion criteria. The full texts of the remaining 117 studies, which studied miRNAs in human DLBCL, were examined in detail. Further 104 articles were excluded because they were focused in tumor tissue and did not include circulating miRNAs, were focused in specific groups of patients with additional pathologies, or did not analyze microRNAs as diagnosis, subtype, treatment response, or prognosis biomarkers. After reviewing the references of the identified articles, one additional study was included. Finally, 12 articles investigating the role of circulating miRNAs as biomarkers in DLBCL were included. Eleven of them considered miRNAs as putative DLBCL diagnosis biomarkers, two articles searched for markers for subtype classification, and five articles considered treatment response or prognosis as their endpoint.
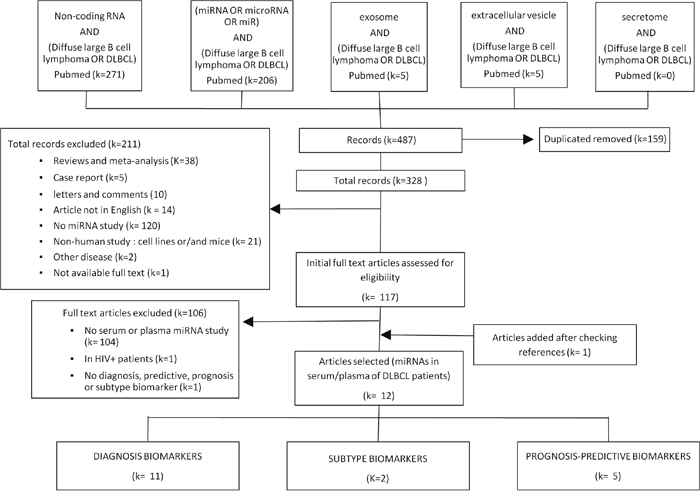
Figure 1: Flowchart of study selection.
Circulating miRNAs as non-invasive biomarkers for diagnosis in DLBCL
Eleven articles searched for circulating miRNAs deregulated in DLBCL patients vs. healthy controls (Table 1) [18-20, 22-29]. Those deregulated miRNAs could be used as biomarkers for diagnosis in this malignancy.
Table 1: Circulating miRNAs as non-invasive biomarkers for diagnosis in DLBCL
miRNAs |
Result |
Sample |
miRNA source |
Method |
Reference |
|---|---|---|---|---|---|
159 miRNAs |
upregulation |
14 DLBCL vs. 20 controls |
Plasma/plasma exosomes (RNeasy MinElute) |
Illumina HISeq sequencing |
Khare D, et al (2017) [28] |
2588 miRNAs |
51 DEMs |
3 DLBCL vs. |
Serum |
Illumina HISeq sequencing and qRT-PCR |
Meng Y, et al (2017) [27] |
miR-21 |
upregulation |
203 DLBCL vs. 100 controls |
Serum |
qRT-PCR |
Zheng Z, et al. (2017) [26] |
miR-155 |
NS |
5 DLBCL vs. |
Serum extracellular vesicles |
qRT-PCR |
Caivano A, et al. (2017) [25] |
miR-21 |
upregulation |
56 DLBCL vs. 20 controls |
Serum |
qRT-PCR |
Yuan WX, et al. (2016) [24] |
miR-21 |
upregulation |
112 DLBCL vs. 45 controls |
Serum |
qRT-PCR |
Li J, et al (2015) [23] |
miR-17 |
NS |
21 DLBCL vs. 6 controls |
Serum |
qRT-PCR |
Borges NM, et al. (2016) [22] |
miR-15a-3p |
upregulation |
33 DLBCL vs. 22 controls |
Serum |
qRT-PCR |
Inada, et al (2015) [29] |
miR-21 |
upregulation |
62 DLBCL vs. 50 controls |
Serum |
qRT-PCR |
Chen W, et al (2014) [20] |
miR-15a |
upregulation |
75 DLBCL vs. 77 controls |
Serum |
qRT-PCR |
Fang C, et al (2012) [19] |
miR-21 |
upregulation |
60 DLBCL vs. 43 controls |
Serum |
qRT-PCR |
Lawrie CH, et al. (2008) [18] |
upregulation: Significantly increased expression in DLBCL patients.
downregulation: Significantly decreased expression in DLBCL patients.
NS: no significant difference in expression between patients and controls.
DEM: differentially expressed miRNAs.
*: analyzed in a subset of 20 patients vs. 20 controls.
Considering common miRNAs analyzed across different studies, we only identified seven miRNAs found to be significantly deregulated in DLBCL patients in at least two studies (miR-15a, miR-21, miR-29c, miR-34a, miR-145, miR-155, and miR-210) (Table 2) [18-20, 22-29]. Among them, five miRNAs showed conflicting results. On the one hand, miR-29c and miR-34a were found contradictorily upregulated or downregulated in DLBCL patients in different studies. On the other hand, miR-15a, miR-155 and miR-210 were found upregulated or unchanged in patients in the different studies in which they were analyzed.
Table 2: Circulating miRNAs explored by 2 or more studies as non-invasive biomarkers for diagnosis in DLBCL
miRNAs |
upregulation |
downregulation |
NS |
|---|---|---|---|
miR-15a |
[28] |
||
miR-21 |
|||
miR-29c |
[19] |
||
miR-34a |
[27] |
[19] |
[28] |
miR-145 |
|||
miR-155 |
|||
miR-210 |
upregulation: Significantly increased expression in DLBCL patients.
downregulation: Significantly decreased expression in DLBCL patients.
NS: no significant difference in expression between patients and controls.
Promisingly, other two miRNAs showed more homogeneous results. For instance, miR-145 was consistently downregulated in DLBCL patients. However, it must be noted that this result was only supported by the two studies in which it was studied [24, 28]. Interestingly, miR-21 was found accordingly upregulated in six of the eight studies in which it was analyzed [18, 20, 23, 24, 26, 29], pointing to this miRNA as a potential diagnosis biomarker in DLBCL.
Circulating miRNAs as non-invasive biomarkers for subtype classification in DLBCL
The potential role of circulating microRNAs in DLBCL classification has been analyzed in a limited set of only two studies (Table 3) [20, 21]. A total of six microRNAs were studied comparing patients with ABC and GCB subtypes to search for differences among them. Each miRNA was analyzed in only one study. Among them, only miR-21 was found to be differentially expressed between subtypes, being upregulated in the subgroup of patients with ABC subtype [20].
Table 3: Circulating miRNAs as non-invasive biomarkers for ABC/GCB subtype classification in DLBCL
miRNAs |
Result |
Sample |
miRNA source |
Method |
Reference |
|---|---|---|---|---|---|
miR-33a |
NS |
173 DLBCL |
Serum |
qRT-PCR |
Song G, et al. (2014) [21] |
miR-21 |
upregulation |
62 DLBCL |
Serum |
qRT-PCR |
Chen W, et al (2014) [20] |
upregulation: Significantly increased expression in ABC subtype.
NS: no significant difference in expression between subtypes.
Circulating miRNAs as non-invasive biomarkers for prediction of response to R-CHOP therapy in DLBCL
Only two studies have focused in circulating miRNAs as predictive biomarkers of response to R-CHOP treatment in DLBCL patients [21, 24] (Table 4).
Table 4: Circulating miRNAs as non-invasive biomarkers for prediction of of response to R-CHOP therapy in DLBCL
miRNAs |
Result |
Sample |
miRNA source |
Method |
Reference |
|---|---|---|---|---|---|
miR-125b |
upregulation |
56 DLBCL |
Serum |
qRT-PCR |
Yuan WX, et al. (2016) [24] |
736 miRNAs |
upregulation |
133 DLBCL |
Serum |
qRT-PCR |
Song G, et al. (2014) [21] |
upregulation: Significantly increased expression in drug resistant DLBCL patients.
downregulation: Significantly decreased expression in drug resistant DLBCL patients.
NS: no significant difference in expression between resistant and sensitive patients.
*Studied in a subgroup of 20 DLBCL (10 chemo-resistant vs 10 chemo-sensitive).
**Studied in a subgroup of 40 DLBCL (20 chemo-resistant vs 20 chemo-sensitive).
The first study analyzed 736 miRNAs in serum samples of 20 complete remission and 20 primarily refractory DLBCL patients. Five miRNAs were differentially expressed between both groups (miR-224, miR-1236, miR-520d-3p, miR-33a, and miR-455-3p) and were validated in an independent group of 133 patients. Upregulation of miR-455-3p and miR-33a was found to be associated with chemosensitivity while upregulation of miR-224, miR-1236, and miR-520d-3p was associated with chemoresistance [21].
The second study analyzed a group of eight miRNAs (miR-21, miR-29, miR-125b, miR-130a, miR-145, miR-155, miR-200c and miR-451) finding the upregulation of miR-125b and miR-130a to be associated with resistance to R-CHOP treatment [24].
MiR-21 was the only miRNA analyzed in both studies. Nevertheless, its expression was not significantly associated with treatment response in any of the two studies.
Circulating miRNAs as non-invasive biomarkers for prognosis prediction in DLBCL
Finally, the relevance of circulating microRNAs for prognosis prediction in DLBCL patients was considered in five studies [18, 20, 21, 23, 24] (Table 5). A total of ten microRNAs were analyzed. Among them, only miR-21 was studied in more than one article, providing discordant results. Upregulated miR-21 has been described as an independent poor prognostic factor in one of the studies [23]. However, the other two studies [18, 20] have shown its upregulation associated with good prognosis. Regarding the other miRNAs, which were analyzed each of them by an only study, upregulation of miR-125b was found to be associated with poor prognosis in patients with DLBCL [24]. On the other hand, high expression of miR-224, miR-1236, and miR-520d-3p and low expression of miR-455-3p and miR-33a were found to be individually associated with unfavorable outcome and a score based on this five-miRNAs was proposed to predict the clinical outcome of DLBCL patients treated with R-CHOP regimen, independent from the IPI score [21].
Table 5: Circulating miRNAs as non-invasive biomarkers for prognosis prediction in DLBCL
miRNAs |
Result |
Outcome |
Sample |
miRNA source |
Method |
Reference |
|---|---|---|---|---|---|---|
miR-125b |
- |
OS |
56 DLBCL |
Serum |
qRT-PCR |
Yuan WX, et al. (2016) [24] |
miR-21 |
- |
OS |
112 DLBCL |
Serum |
qRT-PCR |
Li J, et al (2015) [23] |
miR-33a |
+ |
MRT/PR |
133 DLBCL |
Serum |
qRT-PCR |
Song G, et al. (2014) [21] |
miR-21 |
+ |
RFS |
62 DLBCL |
Serum |
qRT-PCR |
Chen W, et al (2014) [20] |
miR-21 |
+* |
RFS / OS |
52 DLBCL |
Serum |
qRT-PCR |
Lawrie CH, et al. (2008) [18] |
OS: overall survival; MRT: median remission time; PR: probability of remission; PFS: progression free survival ; RFS: relapse free survival; -: high expression of the miRNA significantly associated with worse outcome; +: high expression of the miRNA significantly associated with better outcome; * statistically significant association with better RFS but no significant association with OS.
DISCUSSION
In this systematic review, we have performed a deep analysis of the current literature in relation to the potential role of circulating miRNAs as non-invasive biomarkers for diagnosis, subtype characterization, treatment response and prognosis in patients with DLBCL.
Regarding the suitability of circulating miRNAs as diagnostic biomarkers in DLBCL, eleven articles were identified, in which a total of seven miRNAs (miR-15a, miR-29c, miR-34a, miR-155 and miR-210) were found at least twice to be significantly deregulated in DLBCL patients [18-20, 22-29]. Among them, only miR-145 and miR-21 presented concordant results. Contradictory results could be due to different factors, including differences in methodology, or miRNA source, though in these studies those factors are quite homogeneous. On the other hand, it must be noted that most studies were performed with limited sample sizes and statistical power. Consequently, further larger studies are needed to clarify the results.
MiR-145, which was downregulated in DLBCL patients in the two studies in which it was analyzed [24, 28], is a tumor-suppressor miRNA that has been found to be downregulated in several cancer types, i.e. colorectal cancer, bladder cancer, or ovarian cancer [30–32]. In fact, some of the validated targets of this miRNA include known hallmarks of DLBCL, such as c-MYC or ETS1 [33–39]. However, such a limited number of studies with modest sample sizes does not allow to reach final conclusions. Further studies would be required in order to validate the putative role of mir-145 as a non-invasive biomarker for diagnosis in DLBCL.
On the other hand, it is noteworthy that miR-21, which was analyzed in eight independent studies, was significantly upregulated in DLBCL patients in six of them [18, 20, 23, 24, 26, 29]. In agreement with this observation, miR-21 is one of the most frequently up-regulated miRNAs in solid tumors and high levels are also observed in B-NHLs, being considered an oncomiR. In fact, overexpression of miR21 experimentally leads to a pre-B malignant lymphoid-like phenotype [40]. In addition, miR-21 has been described as a key regulator of disease progression in B-cell lymphoma [41]. Its role in tumorigenesis and disease progression has been associated to the inhibition of the expression of phosphatases, limiting the activity of signaling pathways such as PI3K/AKT, recently suggested to play a crucial role in mediating growth, proliferation and cell survival in a substantial number of DLBCL patients [42, 43]. In summary, the biological role of miR-21 and its replicated upregulation in circulating fluids suggest that miR-21 could be a good biomarker for DLBCL diagnosis.
The utility of circulating microRNAs for DLBCL classification has been analyzed by only two studies with no coincidence in the miRNAs considered [20, 21]. Only miR-21 was found to be upregulated in the ABC subgroup, which is associated with lower survival, in a subset of 62 DLBCL patients [20]. Since this association was observed in only one study with a limited sample size, it would need to be further explored by additional research.
Focusing on circulating miRNAs as predictive biomarkers of response to R-CHOP treatment, two studies were identified [21, 24]. The only miRNA analyzed in both investigations, mir-21, was not associated with treatment response in any of the studies. Interestingly, among the 736 miRNAs analyzed in the first study, upregulation of miR-455-3p and miR-33a was associated with chemosensitivity while upregulation of miR-224, miR-1236, and miR-520d-3p was associated with chemoresistance [21]. In the second study, eight miRNAs were analyzed, finding the upregulation of miR-125b and miR-130a to be associated with R-CHOP chemoresistance [24]. These results would need to be confirmed in additional studies.
Finally, the implication of circulating microRNAs in prognosis in DLBCL has been analyzed in five studies including ten miRNAs [18, 20, 21, 23, 24]. Among them, only miR-21 has been studied in more than one study. However, the results for this miRNA are contradictory. On the one hand, a multivariate analysis, showed upregulated miR-21 as an independent poor prognostic factor [23]. On the contrary, the other two studies that considered this miRNA have found its upregulation to be associated with good prognosis [18, 20]. Surprisingly, in the first of these studies, upregulation of miR-21 was also associated with ABC-DLBCL subgroup, which usually behaves more aggressively and is associated with worse outcome [20]. A possible explanation for this apparently contradictory situation is that serum miR-21 expression was higher in patients with DLBCL stage I and II in comparison to those with III and IV, which could be a confounding factor. On the other hand, Lawrie et al also showed miR-21 upregulation associated with good prognosis [18]. In this case, even though no multivariate analysis was performed, no association between microRNA levels and clinicopathological features was observed (i.e. sex, IPI, stage and presence of extranodal disease). As a result, the significance of miR-21 in prognosis of DLBCL remains uncertain.
Focusing in miRNAs analyzed by a single study, upregulation of miR-125b has been associated with poor prognosis [24]. In addition, a predictor score based on a signature of five miRNAs, among which high expression of miR-224, miR-1236, and miR-520d-3p and low expression of miR-455-3p and miR-33a were individually associated with unfavorable outcome, has been proposed to predict the clinical outcome of DLBCL patients, independent from the IPI score [21]. However, all these results need to be confirmed, given the limited evidence provided.
In brief, the limited number of studies performed, which usually consider a limited set of miRNAs, and the lack of consistency in the results obtained make it difficult to draw final conclusions about the role of circulating miRNAs as non-invasive biomarkers in DLBCL. Although the preliminary associations observed could be of interest, the only result that seems sounder so far is the upregulation of mir-21 in DLBCL patients, which could be used for diagnosis. Consequently, it would be of particular interest to perform large-scale studies including larger sample sizes and a wider array of miRNAs. We can conclude that even if this is a very promising field of study, published evidence is very limited and further studies are needed.
MATERIALS AND METHODS
A systematic search in PubMed database was performed to identify articles published between November 1975 and November 2017 using the following strategy: [((‘Non-coding RNA’) OR (‘microRNA’ OR ‘miRNAs’ OR ‘miR’) OR (‘exosome’) OR (‘extracellular vesicle’) OR (‘secretome’)) AND (‘Diffuse large B cell lymphoma’ OR ‘DLBCL’)].
Articles were included if they presented independent original studies and evaluated the impact of circulating miRNAs as diagnosis, subtype, treatment response or prognosis biomarkers in DLBCL in human population. Reviews and meta-analyses, case reports, letters, comments, abstracts, and articles not published in English were not included. Studies were also excluded if they did not analyze miRNAs, did not include data from human populations, or were focused on other diseases. After full text assessment, articles that did not analyze circulating miRNAs, included other diseases, or did not assess the role of miRNAs in diagnosis, subtype, treatment response, or prognosis, were excluded. All references within the identified studies were then reviewed in order to identify additional matches.
Each eligible manuscript was assessed independently by two researchers (ML and AL). Disagreements were resolved by consensus. Data extracted from each study included: publication year, type of blood-based fluid analyzed (serum or plasma), characteristics of the study population (Table 6), technical methodology, miRNAs assessed, and the list of specific miRNAs that could be used as biomarkers in DLBCL. In order to define associations between the expression of the miRNAs and the phenotypes, a p value < 0.05 was considered statistically significant.
Table 6: Clinical characteristics of the study population in each included study
Study |
Sample |
Age |
Sex (%) |
Hans’ algorithm (%) |
Performance status (%) |
Ann Arbor staging (%) |
IPI (%) |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Median (range) or mean ± SD |
Female |
Male |
GCB |
non-GCB |
nap |
PS 0-2 |
PS 3-4 |
I |
II |
III |
IV |
0-2 |
3-5 |
||
Khare D, et al (2017) [28] |
14 DLBCL |
64 (34-85) |
36 |
64 |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
50 |
50 |
20 controls |
37 (26-63) |
45 |
55 |
||||||||||||
Meng Y, et al (2017) [27] |
3 DLBCL |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
3 controls |
NA |
NA |
NA |
||||||||||||
Zheng Z, et al. (2017) [26] |
203 DLBCL |
NA |
39.4 |
60.6 |
NA |
NA |
NA |
NA |
NA |
56.7 |
43.3 |
66 |
34 |
||
100 controls |
NA |
NA |
NA |
||||||||||||
Caivano A, et al. (2017) [25] |
5 DLBCL |
66 (53–80) |
60 |
40 |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
18 controls |
60 (43–77) |
55.6 |
44.4 |
||||||||||||
Yuan WX, et al. (2016) [24] |
56 DLBCL |
54.7 (23-74) |
43 |
57 |
NA |
NA |
NA |
NA |
NA |
10 |
24 |
38 |
28 |
28 |
72 |
20 controls |
NA |
NA |
NA |
||||||||||||
Li J, et al (2015) [23] |
112 DLBCL |
NA |
48.2 |
51.8 |
NA |
NA |
NA |
NA |
NA |
42 |
58 |
37.5 |
62.5 |
||
45 controls |
NA |
NA |
NA |
||||||||||||
Borges NM, et al. (2016) [22] |
21 DLBCL |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
6 controls |
NA |
NA |
NA |
||||||||||||
Inada, et al (2015) [29] |
33 DLBCL |
67 (36-84) |
39 |
61 |
42.4 |
48.5 |
9.1 |
91 |
9 |
9.1 |
21.2 |
18.2 |
51.5 |
55.6 |
45.4 |
22 controls |
62 (20-76) |
41 |
59 |
||||||||||||
Chen W, et al (2014) [20] |
62 DLBCL |
66.1 ±13.6 |
33.8 |
66.1 |
51.6 |
48.4 |
0 |
NA |
NA |
30.7 |
17.7 |
19.3 |
32.3 |
71 |
29 |
50 controls |
NA |
NA |
NA |
||||||||||||
Fang C, et al (2012) [19] |
75 DLBCL |
54 (19–85) |
45.3 |
54.7 |
NA |
NA |
NA |
NA |
NA |
13.3 |
16 |
22.7 |
48 |
60 |
40 |
77 controls |
50 (36–68) |
41.5 |
58.4 |
||||||||||||
Lawrie CH, et al. (2008) [18] |
60 DLBCL |
63 ±11 |
35 |
65 |
NA |
NA |
NA |
NA |
NA |
33 |
23 |
30 |
13 |
70 |
30 |
43 controls |
NA |
NA |
NA |
||||||||||||
Song G, et al. (2014) (21) |
173 DLBCL |
56.4 (19-78) |
47.4 |
52.6 |
53.2 |
46.8 |
0 |
85.5 |
14.5 |
14.5 |
25.4 |
35.3 |
24.8 |
22.3 |
67.7 |
NA: Data not available. Nap: unclassified.
Abbreviations
DLBCL: Diffuse large B-cell lymphoma; miRNAs: microRNAs; R-CHOP: Combined chemotherapy of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone; IPI: International Prognostic Index; GEP: gene expression profiling; GCB: center B-cell-like DLBCL; ABC: activated B-cell-like DLBCL; UTR: untranslated region.
Author contributions
AGO contributed to the conception and design of the study. MLS, ALE, and JAM acquired and analyzed the data. MLS, ELL and AGO interpreted the results and drafted the article. AGO acquired and administrated the funding. All authors have read, reviewed critically and approved the final manuscript.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
FUNDING
This study was funded by the Basque Government (IT989-16), and UPV/EHU (UFI11/35). The funding source did not have any role in study design, collection, analysis and interpretation of data, writing of the report, or decision to submit the article for publication.
REFERENCES
1. Swerdlow SH, Campmo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th Edition, Volume 2. Lyon, France: IARC Press; 2008.
2. Lossos IS, Morgensztern D. Prognostic biomarkers in diffuse large B-cell lymphoma. J Clin Oncol. 2006; 24:995-1007. https://doi.org/10.1200/JCO.2005.02.4786.
3. Coiffier B, Thieblemont C, Van Den Neste E, Lepeu G, Plantier I, Castaigne S, Lefort S, Marit G, Macro M, Sebban C, Belhadj K, Bordessoule D, Fermé C, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d'Etudes des Lymphomes de l'Adulte. Blood. 2010; 116:2040-5. https://doi.org/10.1182/blood-2010-03-276246.
4. Raut LS, Chakrabarti PP. Management of relapsed-refractory diffuse large B cell lymphoma. South Asian J Cancer. 2014; 3:66-70. https://doi.org/10.4103/2278-330X.126531.
5. Project IN-HsLPF. A predictive model for aggressive non-Hodgkin's lymphoma. N Engl J Med. 1993; 329:987-94. https://doi.org/10.1056/NEJM199309303291402.
6. Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A, Boldrick JC, Sabet H, Tran T, Yu X, Powell JI, Yang L, Marti GE, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000; 403:503-11. https://doi.org/10.1038/35000501.
7. Rosenwald A, Wright G, Chan WC, Connors JM, Campo E, Fisher RI, Gascoyne RD, Muller-Hermelink HK, Smeland EB, Giltnane JM, Hurt EM, Zhao H, Averett L, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med. 2002; 346:1937-47. https://doi.org/10.1056/NEJMoa012914.
8. Martin-Guerrero I, Gutierrez-Camino A, Lopez-Lopez E, Bilbao-Aldaiturriaga N, Pombar-Gomez M, Ardanaz M, Garcia-Orad A. Genetic variants in miRNA processing genes and pre-miRNAs are associated with the risk of chronic lymphocytic leukemia. PLoS One. 2015; 10:e0118905. https://doi.org/10.1371/journal.pone.0118905.
9. Gutierrez-Camino A, Oosterom N, den Hoed MAH, Lopez-Lopez E, Martin-Guerrero I, Pluijm SMF, Pieters R, de Jonge R, Tissing WJE, Heil SG, García-Orad A, van den Heuvel-Eibrink MM. The miR-1206 microRNA variant is associated with methotrexate-induced oral mucositis in pediatric acute lymphoblastic leukemia. Pharmacogenet Genomics. 2017; 27:303-6. https://doi.org/10.1097/FPC.0000000000000291.
10. Shirafkan N, Mansoori B, Mohammadi A, Shomali N, Ghasbi M, Baradaran B. MicroRNAs as novel biomarkers for colorectal cancer: New outlooks. Biomed Pharmacother. 2018; 97:1319-30. https://doi.org/10.1016/j.biopha.2017.11.046.
11. Hao NB, He YF, Li XQ, Wang K, Wang RL. The role of miRNA and lncRNA in gastric cancer. Oncotarget. 2017; 8:81572-82. https://doi.org/10.18632/oncotarget.19197.
12. Weber JA, Baxter DH, Zhang S, Huang DY, Huang KH, Lee MJ, Galas DJ, Wang K. The microRNA spectrum in 12 body fluids. Clin Chem. 2010; 56:1733-41. https://doi.org/10.1373/clinchem.2010.147405.
13. Yáñez-Mó M, Siljander PR, Andreu Z, Zavec AB, Borràs FE, Buzas EI, Buzas K, Casal E, Cappello F, Carvalho J, Colás E, Cordeiro-da Silva A, Fais S, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015; 4:27066.
14. Wang H, Wang T, Shi W, Liu Y, Chen L, Li Z. Comprehensive analysis on diagnostic value of circulating miRNAs for patients with ovarian cancer. Oncotarget. 2017; 8:66620-8. https://doi.org/10.18632/oncotarget.18129.
15. Santangelo A, Tamanini A, Cabrini G, Dechecchi MC. Circulating microRNAs as emerging non-invasive biomarkers for gliomas. Ann Transl Med. 2017; 5:277. https://doi.org/10.21037/atm.2017.06.15.
16. Montagnana M, Benati M, Danese E. Circulating biomarkers in epithelial ovarian cancer diagnosis: from present to future perspective. Ann Transl Med. 2017; 5:276. https://doi.org/10.21037/atm.2017.05.13.
17. Peng Q, Zhang X, Min M, Zou L, Shen P, Zhu Y. The clinical role of microRNA-21 as a promising biomarker in the diagnosis and prognosis of colorectal cancer: a systematic review and meta-analysis. Oncotarget. 2017; 8:44893-909. https://doi.org/10.18632/oncotarget.16488.
18. Lawrie CH, Gal S, Dunlop HM, Pushkaran B, Liggins AP, Pulford K, Banham AH, Pezzella F, Boultwood J, Wainscoat JS, Hatton CS, Harris AL. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br J Haematol. 2008; 141:672-5. https://doi.org/10.1111/j.1365-2141.2008.07077.x.
19. Fang C, Zhu DX, Dong HJ, Zhou ZJ, Wang YH, Liu L, Fan L, Miao KR, Liu P, Xu W, Li JY. Serum microRNAs are promising novel biomarkers for diffuse large B cell lymphoma. Ann Hematol. 2012; 91:553-9. https://doi.org/10.1007/s00277-011-1350-9.
20. Chen W, Wang H, Chen H, Liu S, Lu H, Kong D, Huang X, Kong Q, Lu Z. Clinical significance and detection of microRNA-21 in serum of patients with diffuse large B-cell lymphoma in Chinese population. Eur J Haematol. 2014; 92:407-12. https://doi.org/10.1111/ejh.12263.
21. Song G, Gu L, Li J, Tang Z, Liu H, Chen B, Sun X, He B, Pan Y, Wang S, Cho WC. Serum microRNA expression profiling predict response to R-CHOP treatment in diffuse large B cell lymphoma patients. Ann Hematol. 2014; 93:1735-43. https://doi.org/10.1007/s00277-014-2111-3.
22. Borges NM, do Vale Elias M, Fook-Alves VL, Andrade TA, de Conti ML, Macedo MP, Begnami MD, Campos AH, Etto LY, Bortoluzzo AB, Alves AC, Young KH, Colleoni GW. Angiomirs expression profiling in diffuse large B-Cell lymphoma. Oncotarget. 2016; 7:4806-16. https://doi.org/10.18632/oncotarget.6624.
23. Li J, Fu R, Yang L, Tu W. miR-21 expression predicts prognosis in diffuse large B-cell lymphoma. Int J Clin Exp Pathol. 2015; 8:15019-24.
24. Yuan WX, Gui YX, Na WN, Chao J, Yang X. Circulating microRNA-125b and microRNA-130a expression profiles predict chemoresistance to R-CHOP in diffuse large B-cell lymphoma patients. Oncol Lett. 2016; 11:423-32. https://doi.org/10.3892/ol.2015.3866.
25. Caivano A, La Rocca F, Simeon V, Girasole M, Dinarelli S, Laurenzana I, De Stradis A, De Luca L, Trino S, Traficante A, D'Arena G, Mansueto G, Villani O, et al. MicroRNA-155 in serum-derived extracellular vesicles as a potential biomarker for hematologic malignancies - a short report. Cell Oncol (Dordr). 2017; 40:97-103. https://doi.org/10.1007/s13402-016-0300-x.
26. Zheng Z, Xu PP, Wang L, Zhao HJ, Weng XQ, Zhong HJ, Qu B, Xiong J, Zhao Y, Wang XF, Janin A, Zhao WL. MiR21 sensitized B-lymphoma cells to ABT-199 via ICOS/ICOSL-mediated interaction of Treg cells with endothelial cells. J Exp Clin Cancer Res. 2017; 36:82. https://doi.org/10.1186/s13046-017-0551-z.
27. Meng Y, Quan L, Liu A. Identification of key microRNAs associated with diffuse large B-cell lymphoma by analyzing serum microRNA expressions. Gene. 2018; 642:205-11. https://doi.org/10.1016/j.gene.2017.11.022.
28. Khare D, Goldschmidt N, Bardugo A, Gur-Wahnon D, Ben-Dov IZ, Avni B. Plasma microRNA profiling: Exploring better biomarkers for lymphoma surveillance. PLoS One. 2017; 12:e0187722. https://doi.org/10.1371/journal.pone.0187722.
29. Inada K, Okoshi Y, Cho Y, Saito H, Iijima T, Hori M, Kojima H. Availability of Circulating MicroRNAs as a Biomarker for Early Diagnosis of Diffuse Large B-Cell Lymphoma. Open Journal of Blood Diseases. 2015; 5:48-58.
30. Akao Y, Nakagawa Y, Naoe T. MicroRNA-143 and -145 in colon cancer. DNA Cell Biol. 2007; 26:311-20. https://doi.org/10.1089/dna.2006.0550.
31. Ichimi T, Enokida H, Okuno Y, Kunimoto R, Chiyomaru T, Kawamoto K, Kawahara K, Toki K, Kawakami K, Nishiyama K, Tsujimoto G, Nakagawa M, Seki N. Identification of novel microRNA targets based on microRNA signatures in bladder cancer. Int J Cancer. 2009; 125:345-52. https://doi.org/10.1002/ijc.24390.
32. Liang H, Jiang Z, Xie G, Lu Y. Serum microRNA-145 as a novel biomarker in human ovarian cancer. Tumour Biol. 2015; 36:5305-13. https://doi.org/10.1007/s13277-015-3191-y.
33. Rosenquist R, Beà S, Du MQ, Nadel B, Pan-Hammarström Q. Genetic landscape and deregulated pathways in B-cell lymphoid malignancies. J Intern Med. 2017; 282:371-94. https://doi.org/10.1111/joim.12633.
34. Amin AD, Peters TL, Li L, Rajan SS, Choudhari R, Puvvada SD, Schatz JH. Diffuse large B-cell lymphoma: can genomics improve treatment options for a curable cancer? Cold Spring Harb Mol Case Stud. 2017; 3:a001719. https://doi.org/10.1101/mcs.a001719.
35. Chou CH, Shrestha S, Yang CD, Chang NW, Lin YL, Liao KW, Huang WC, Sun TH, Tu SJ, Lee WH, Chiew MY, Tai CS, Wei TY, et al. miRTarBase update 2018: a resource for experimentally validated microRNA-target interactions. Nucleic Acids Res. 2018; 46:D296-D302. https://doi.org/10.1093/nar/gkx1067.
36. Zheng L, Pu J, Qi T, Qi M, Li D, Xiang X, Huang K, Tong Q. miRNA-145 targets v-ets erythroblastosis virus E26 oncogene homolog 1 to suppress the invasion, metastasis, and angiogenesis of gastric cancer cells. Mol Cancer Res. 2013; 11:182-93. https://doi.org/10.1158/1541-7786.MCR-12-0534.
37. Sachdeva M, Zhu S, Wu F, Wu H, Walia V, Kumar S, Elble R, Watabe K, Mo YY. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci U S A. 2009; 106:3207-12. https://doi.org/10.1073/pnas.0808042106.
38. Chen Z, Zeng H, Guo Y, Liu P, Pan H, Deng A, Hu J. miRNA-145 inhibits non-small cell lung cancer cell proliferation by targeting c-Myc. J Exp Clin Cancer Res. 2010; 29:151. https://doi.org/10.1186/1756-9966-29-151.
39. Shao Y, Qu Y, Dang S, Yao B, Ji M. MiR-145 inhibits oral squamous cell carcinoma (OSCC) cell growth by targeting c-Myc and Cdk6. Cancer Cell Int. 2013; 13:51. https://doi.org/10.1186/1475-2867-13-51.
40. Medina PP, Nolde M, Slack FJ. OncomiR addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature. 2010; 467:86-90. https://doi.org/10.1038/nature09284.
41. Musilova K, Mraz M. MicroRNAs in B-cell lymphomas: how a complex biology gets more complex. Leukemia. 2015; 29:1004-17. https://doi.org/10.1038/leu.2014.351.
42. Uddin S, Hussain AR, Siraj AK, Manogaran PS, Al-Jomah NA, Moorji A, Atizado V, Al-Dayel F, Belgaumi A, El-Solh H, Ezzat A, Bavi P, Al-Kuraya KS. Role of phosphatidylinositol 3'-kinase/AKT pathway in diffuse large B-cell lymphoma survival. Blood. 2006; 108:4178-86. https://doi.org/10.1182/blood-2006-04-016907.
43. Hasselblom S, Hansson U, Olsson M, Torén L, Bergström A, Nilsson-Ehle H, Andersson PO. High immunohistochemical expression of p-AKT predicts inferior survival in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Br J Haematol. 2010; 149:560-8. https://doi.org/10.1111/j.1365-2141.2010.08123.x.

