INTRODUCTION
Head and neck cancer accounts for 500,000 new cases and nearly 300,000 deaths annually worldwide [1] .
Squamous cell carcinoma is the most frequent histological subtype of head and neck tumors [2].
About two thirds of head and neck squamous cell carcinoma patients are diagnosed with locally advanced disease [3] and are treated with a combination of surgery, radiation therapy and chemotherapy. Despite this primary treatment, more than a third of these patients have locoregional recurrences or distant metastases [4–7] and in this situation treatment is mostly palliative and disappointing, with a median overall survival (OS) of less than 1 year [8].
About 10% of the patients present with distant metastases at diagnosis [9]. The most frequent site of distant metastasis is the lung [3].
Platinum based chemotherapy is the usual first-line palliative treatment. Currently, the standard of care is the combination of Cetuximab, Cisplatin and 5-Fluorouracil, followed by maintenance with Cetuximab (the “EXTREME” regimen) [10] .
A large proportion of patients are ineligible to the EXTREME regimen because of their age>70yo, Performance Status (PS)>1 or their severe comorbidities (cardiac or renal insufficiency etc…). In this population, despite replacement of Cisplatin with Carboplatin in the EXTREME regimen, tolerability remains poor.
In advanced non-small cell lung cancer, the standard of care is platinum based chemotherapy too. In elderly population, Carboplatin with weekly Paclitaxel is a safe and recommended option [11, 12].
Similarly to this indication in lung cancer, we propose in our institution Carboplatin with weekly Paclitaxel for patients unfit to Cisplatin and in some patients eligible to Cisplatin who refuse the EXTREME regimen for various reasons (mostly due to the 4 days of continuous perfusion of Fluorouracil).
This association isn’t validated in head and neck cancer but several studies have shown the efficacy of Paclitaxel as monotherapy or in combination with Cisplatin [13] with promising results.
In this retrospective study we evaluated the efficacy and the tolerability of Carboplatin and Paclitaxel as first-line treatment in patients with recurrent or metastatic head and neck squamous cell carcinoma (RMHNSCC), mostly for patients who are unfit to the EXTREME regimen.
RESULTS
Patients’ characteristics
Between August 2009 and December 2016, 117 patients with RMHNSCC were treated at the “Centre Léon Bérard “(Lyon, France) with Carboplatin and Paclitaxel combination as first-line therapy.
Patients’ characteristics are summarized in Table 1.
Table 1: Baseline demographics of the patient population
N (%) |
|
|---|---|
Median age, years [range] |
66,2 [27.1–94.3] |
Sex |
|
Female |
25 (21) |
Male |
92 (79) |
Localization at initial diagnosis |
|
Oral cavity |
31 (26) |
Oropharynx |
33 (28) |
Hypopharynx |
25 (21) |
Larynx |
21 (18) |
Other localization |
6 (5) |
Unknown |
1 (1) |
Tumor stage at initial diagnosis |
|
I |
7 (6) |
II |
22 (19) |
III |
36 (31) |
IVa |
23 (20) |
IVb |
7 (6) |
IVc |
14 (12) |
Unknown |
8 (7) |
Initial treatment |
|
Neoadjuvant chemotherapy |
31 (26) |
Platinium based |
28 (24) |
Unknown |
3 (3) |
Surgery |
83 (71) |
Radiotherapy |
85 (73) |
Alone |
44 (38) |
With Cisplatin |
26 (23) |
With Cetuximab |
11 (9) |
Other or unknown |
4 (3) |
Relapse before the introduction of Carboplatin+Paclitaxel |
|
Loco Regional only |
57 (49) |
Loco Regional and Metastatic |
35 (30) |
Metastatic only |
13 (11) |
Performance status at onset Carboplatin and Paclitaxel |
|
0 |
9 (8) |
1 |
57 (49) |
2 |
26 (22) |
3 |
12 (10) |
Unknown |
13 (11) |
Since 89% of patients experienced a recurrence, 41% received a platinum-agent previously for a localized disease (18% of those less than 6 months before relapse). Before palliative chemotherapy onset, 34 (29%) patients were treated locally for previous relapses (mainly surgery, radiotherapy potentiated or not by chemotherapy).The most common site of distant metastases was pulmonary .
All patients received Carboplatin and Paclitaxel strictly in first intent except for 8 patients. 2 of these patients should be retrospectively considered as initially metastatic: they had non-specific lung micronodules when they started induction chemotherapy with Cisplatin Docetaxel and Fluorouracil. After that they were treated with radiotherapy and one of them potentialized with Cisplatin. After 7.8 and 10 months of the last platin administration, the lung nodules progressed and they started carboplatin and paclitaxel. Despite a certain frailty, 5 patients received a first cycle of cisplatin-based chemotherapy and switched to Carboplatin and Paclitaxel due to severe toxicity. Finally 1 patient received a cycle of Cisplatin/Vinorelbine since the solitary metastasis was considered as a primary lung cancer and switched at the second cycle for Carboplatin with Paclitaxel.
Of the 117 patients treated with Carboplatin and Paclitaxel, 23 (20%) were eligible to a chemotherapy by Cisplatin Fluorouracil and Cetuximab (EXTREME regimen). 3 of those received firstly a Cisplatin-based chemotherapy, then, due to toxicity, Carboplatin and Paclitaxel. 94 (80%) were ineligible to EXTREME due at least to one adverse criteria among age >70 years, renal failure (creatinine clearance <60 ml/min), PS≥2 or severe comorbiditie(s) (cardiac insufficiency, cirrhosis, cisplatin allergy etc…). Details of ineligibility are summarized in Table 2.
Table 2: Frailty criteria of the 94 patients ineligible for the EXTREME schedule
N (%) |
|
|---|---|
All criteria listed : |
|
Age>70 yo |
47 (40%) |
Creatinine clearance <60ml/min |
24 (21%) |
Severe Comorbidities |
29 (25%) |
PS ≥2 |
38 (32%) |
Patients unfit for cisplatin |
94 |
Patients with only one criteria : |
70 (74%) |
Age |
30 (26%) |
Renal failure |
2 (1%) |
Severe comorbidities |
14 (12%) |
PS ≥2 |
24 (21%) |
Patients with ≥2 criteria |
21 (18%) |
Patients with 1 criteria previous listed and another criteria : |
3 (3%) |
Lung cancer associated |
2 (2%) |
High dose of corticosteroids contraindicating the inclusion in a protocol |
1 (1%) |
Carboplatin and paclitaxel delivery
Data on chemotherapy delivery are summarized in Figure 1. Initially, Carboplatin was administered on a monthly basis for 88 (75%) patients and weekly for 29 (25%) patients. Due to severe frailty, 35 patients received a 25% reduction of the dose of a least one agent. Of note, 3 patients received Cetuximab in combination with carboplatin and paclitaxel. Treatment toxicities are presented in Table 3.
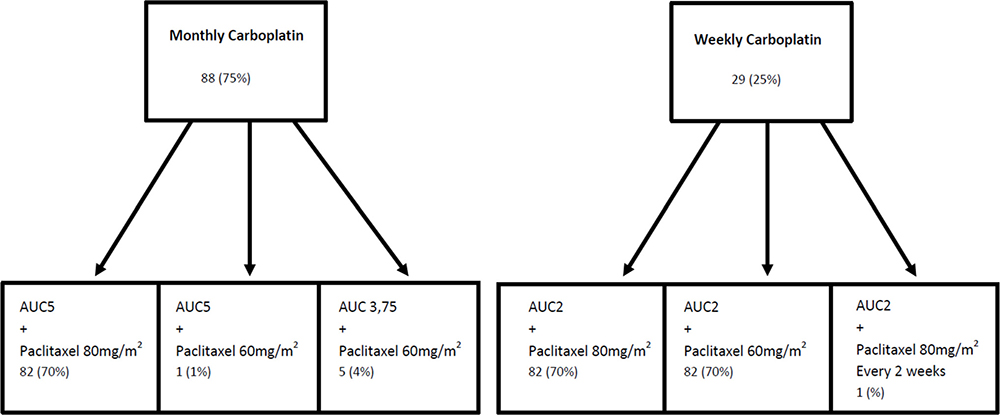
Figure 1: Delivery of carboplatin and paclitaxel.
Table 3: Treatment toxicities
Monthly Carboplatin N (%) |
Weekly Carboplatin N (%) |
|
|---|---|---|
Total of toxicities |
88 |
40 |
Any grade toxicity leading to modification of treatment |
||
Dose reduction |
53 (60%) |
7 (18%) |
With modification of monthly Carboplatin in weekly Carboplatin |
11 (13%) |
|
Cessation of treatment |
16 (18%) |
9 (23%) |
Anemia |
45 (51%) |
10 (25%) |
≥grade 3) |
23 (26%) |
1 (3% |
Thrombopenia |
26 (30%) |
8 (20%) |
≥grade 3 |
7 (8%) |
0 (0%) |
Neutropenia |
46 (52%) |
20 (50%) |
≥grade 3 |
35 (40%) |
11 (28%) |
Febrile neutropenia |
12 (14%) |
5 (13%) |
Leading death |
1 (1%) |
1 (3%) |
Infection without aplasia |
39 (44%) |
14 (35%) |
Digestive toxicity |
17 (19%) |
5 (13%) |
≥grade 3 |
1 (1%) |
0 (0%) |
Neuropathy |
10 (11%) |
3 (8%) |
≥grade 3 |
2 (2%) |
1 (3%) |
Dose reduction was needed for 60 (51%) patients, 11 of those resulted with a modification of monthly Carboplatin to weekly Carboplatin and 25 (21%) stopped treatment due to toxicity.
The most common side effects were hematologic, they are detailed in Table 4. There were 94 grade ≥ 3 toxicities with 17 (15%) febrile neutropenia, 2 of those were fatal (2%). 18 patients needed granulocyte colony stimulating factors.
Table 4: Hemato toxicities
Monthly Carboplatin |
Weekly Carboplatin |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
1 |
2 |
3 |
4 |
5 |
1 |
2 |
3 |
4 |
5 |
|
Anemia |
1 |
21 |
23 |
0 |
0 |
0 |
9 |
1 |
0 |
0 |
Thrombopenia |
11 |
8 |
4 |
3 |
0 |
3 |
5 |
0 |
0 |
0 |
Neutropenia |
1 |
10 |
26 |
9 |
0 |
1 |
8 |
9 |
2 |
0 |
Febrile neutropenia |
5 |
6 |
1 |
4 |
0 |
1 |
||||
Overall, weekly Carboplatin was better tolerated, inducing fewer severe toxicities than monthly Carboplatin.
Carboplatin and paclitaxel efficacy
In intent to treat analysis the overall response rate was 40%, 5(4%) complete responses and 42 (36%) partial responses, whereas 33 (28%) patients were stabilized and 16 (14%) progressed. There were 21 (18%) unevaluable patients because they died before the first assessment. With a median of follow up of 28.1 months [95% CI; 24.3–52.8 months], the median OS for all population was 9.1months [95% CI; 6.9–11.5] (Figure 2A) and it was 13.7 months [95% CI; 7.3–27.9] and 8 months [95% CI; 5.4–10.7] for patients eligible to Cisplatin and not respectively (Figure 2B). Among patients ineligible for cisplatin, median OS was 11.5 months [95% CI; 8.5–19.1] if they were of PS0–1, but was 3.6 months [95% CI; 2.2–6.9] and 1.7 months [95% CI; 0.3–5.1] if they were of PS2 and 3 respectively (Figure 2C).
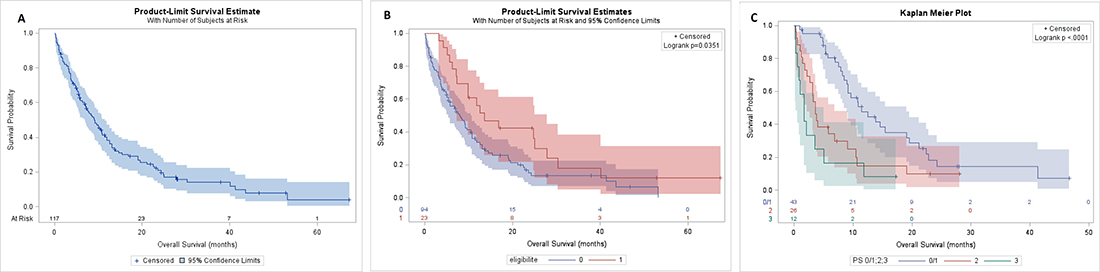
Figure 2: OS of population. OS of all population (A), in eligible and no eligible group (B), in PS 0/1, 2 and 3 of no eligible group (C).
The median progression-free survival (PFS) for all population was 4.7months [95% CI; 3.7–5.2] (Figure 3A) and it was 5.5 months [CI 95%; 1.5–6.6] and 4.4 months [CI 95% ; 3.4–5.0] for patients eligible to Cisplatin and not respectively (Figure 3B). Among patients ineligible for Cisplatin, median PFS was 5.5 months [CI 95%; 4.8–7.1] if they were of PS0/1, but 2.0 months [CI 95%; 1.-4.0] and 1.7 months [CI 95%; 0.3–4.9] if they were of PS2 and 3 respectively (Figure 3C).
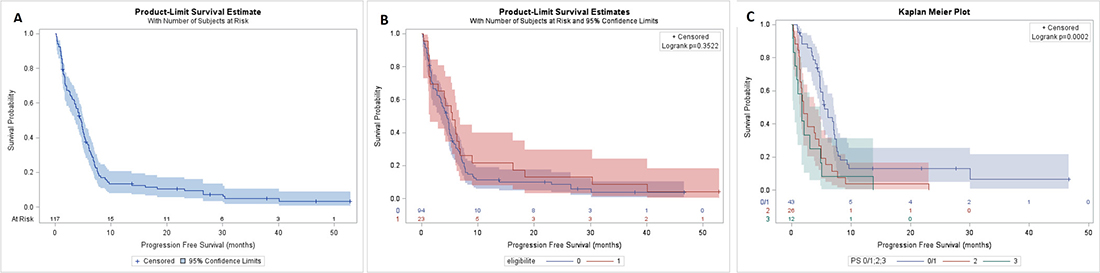
Figure 3: PFS of population. PFS of all population (A), in eligible and no eligible group (B), in PS 0/1, 2 and 3 of no eligible group (C).
DISCUSSION
The EXTREME (Platin-5FU-Cetuximab) regimen has become the standard of care in first-line therapy in patients with recurrent/metastatic head and neck squamous cell carcinoma with 36% responses and a median overall survival of 10.1 months [95% CI; 8.6–11.2] (versus 20% and 7.4 months [95% CI; 6,4–8,3] respectively for Platin-5FU) [10]. In subgroup analysis, OS was significantly better only with cisplatin at 10.6 months (vs 7.3 with chemotherapy alone) whereas there was not any significant difference with carboplatin (9.7 vs 8.3 months) suggesting that for patients ineligible to Cisplatin, the Carboplatin-EXTREME regimen is possibly not the best regimen.
Many patients aren’t eligible for Cisplatin and our study suggests Carboplatin with weekly Paclitaxel can be a good alternative for these unfit patients and even possibly for fit patients. Indeed the response rate of the whole population reaches 40% with a median OS of 13.7 months [95% CI; 7.3–27.9] and 8 months [95% CI; 5.4–10.7] for patients eligible and for patients ineligible to Cisplatin respectively. Evidently, the weak number of eligible patients (23) explains the large confidence interval forbidding definitive conclusion and does not authorize direct comparison with a prospective clinical trial. Further analysis are needed.
Again, even if a comparison isn’t possible between the results of a clinical trial and our retrospective study, Carboplatin and weekly Paclitaxel seems better for patients ineligible to Cisplatin with PS 0/1 compared to Carboplatin+5 Fluorouracil+Cetuximab in EXTREME study with a median OS of 11.5 months [95% CI; 8.5–19.1] vs 9.7 months and a median PFS of 5.5 months [CI 95%; 4.8; 7.1] vs 5.3 months. Survival was poor for patients with PS2 or 3 with OS of 3.6 and 1.7 months and PFS of 2.0 and 1.7 months respectively. Since this population is usually excluded from clinical trials we cannot draw definitive conclusion. Specific studies would be necessary to evaluate this population. Furthermore our definition of PFS was different than EXTEREME study, we considered the deaths only from cancer and not from any cause, it could be overestimated our results.
In our study, the tolerance of Carboplatin and weekly Paclitaxel appeared acceptable in this frail population with weekly Carboplatin regimen. In fact, the toxicities with monthly Carboplatin were more frequent and more severe. And a large part of the population of weekly Carboplatin group switched from monthly group after toxicities. Response rate of weekly Carboplatin group delivered from the beginning was slightly lower than for the overall population at 34% but we cannot conclude with only 29 patients. In further studies we propose to adopt a weekly regimen for both Paclitaxel and Carboplatin.
The main toxicities were hematologic with 26% and 3% anemia grade ≥3, 8% and 0% thrombopenia grade ≥3 and 40% and 28% neutropenia grade ≥3 in monthly and weekly Carboplatin group respectively.
Hepatotoxicity with Carboplatin and Paclitaxel is described in several studies [11, 14]. In our population, we didn’t observed any hepatic perturbation related to chemotherapy.
Toxicities of the Carboplatin group of the Extreme study are not available but in our practice it seems to be less toxic than Cisplatin, so we cannot formally compare our data.
Another study demonstrated safety and efficacy of weekly Carboplatin and Paclitaxel in 31 patients with locally advanced, distant metastases or recurrent head and neck squamous cell carcinoma. Median OS was 12.8 months [95% CI 8.6–15.5] and the major toxicity was hematologic too, with 22% neutropenia grade ≥3, 12% anemia grade ≥3 and 0% thrombopenia grade ≥3 [15].
With our results and since monotherapies with Paclitaxel, Cetuximab or Capecitabine are efficient after failure of platinum [16–18] there is sense to prefer bi-chemotherapy (by Carboplatin and Paclitaxel) to tri-chemotherapy (by Platin-5FU-Cetuximab) in order to allow sequential treatments that could increase survival [19]. Moreover, previous studies showed that Cetuximab could be combined with Paclitaxel with possible synergy and promising efficacy after failure of Platinum [6, 20] and thus could be better than EXTREME schedule for selected patients [21]. So, combination of the three agents Platin, Paclitaxel and Cetuximab could be of great interest. Indeed, a randomized phase II study in first-line reported 51.7% responses with Cisplatin-Paclitaxel-Cetuximab and an OS of 11 months [22]. Similarly, another phase II study combining Cisplatin Docetaxel and Cetuximab in first-line showed 44.4% responses and OS of 14 months [23].
After failure of Platinum, immunotherapy by checkpoint inhibitors has become the standard treatment. Indeed, the Checkmate 141 phase III randomized trial, showed that Nivolumab, a PD1 inhibitor, improved overall survival compared with single-agent therapy including Methotrexate, Docetaxel and Cetuximab [24] with a median overall survival of 7·5 months (95% CI, 5·5–9·1) and of 5·1 months (95% CI 4·0–6·0) (HR 0·70 [97·73% CI, 0·51–0·96]; p = 0·01) respectively. It is associated with fewer severe toxic effects and a better quality of life [25].
And currently, a phase III study compares Ipilimumab (CTLA 4 inhibitor) and Nivolumab with EXTREME schedule [26].
In conclusion, our study shows that weekly Carboplatin and Paclitaxel is a good option as first-line therapy for recurrent/metastatic head and neck squamous cell carcinoma, mostly for patients ineligible to Cisplatin. A future question would be the place of Cetuximab: in combination with Carboplatin and Paclitaxel or after failure of Nivolumab (or another anti-PD1 or PDL1). Similarly, combination of Carboplatin and Paclitaxel with immunotherapies could be relevant as in advanced non-small cell lung cancer [27, 28].
MATERIALS AND METHODS
Patient selection
We retrospectively reviewed the data in our institution between August 2009 and December 2016.
The inclusion criteria were as follows: (1) 18 or older patients with patients with histologically confirmed head and neck squamous cell carcinoma, (2) treated with Carboplatin and Paclitaxel in first-line for a recurrent or metastatic disease.
A first-line is defined by a first treatment in the recurrent/metastatic setting or by a switch from another treatment (due to toxicity of the first course) before any progression.
Prior treatment for a localized disease by surgery, radiotherapy or chemotherapy were permitted .
Treatment
Different 28-days schedules were used. Paclitaxel was administered at 80 mg/m2 at day 1 (D1), day 8 (D8) and day 15 (D15). The standard schedule was Carboplatin AUC5 (area under the curve = 5) at D1 and in the alternative schedule Carboplatin was administered at AUC2 at D1, D8 and D15.
The doses could be reduced at the beginning or during treatment according to the patient’s frailty or to toxicities.
Assessments
Response was evaluated every 6–8 weeks by repeated clinical and computed tomographic scan assessments on the basis of the extent of disease at presentation. Antitumor activity was evaluated according to the Response Evaluation Criteria In Solid Tumors criteria 1.1 [29].
Statistical design
Overall survival (OS) was defined as the time from the date of first Carboplatin and Paclitaxel administration to the date of death.
Progression-free survival (PFS) was calculated from the date of first Carboplatin and Paclitaxel administration to the date of progression or death secondary to the cancer, whichever occurred first. If progression or death did not occur before the cut-off date, data were censored at the time of the last valid assessment.
Survival distributions were estimated by the Kaplan–Meier method.
Abbreviations
RMHNSCC: recurrent/metastatic head and neck squamous cell carcinoma; D1: day 1; D8: day 8; D15: day 15; AUC5: area under the curve = 5; PS: performance status; PFS: progression free survival; OS: overall survival.
Author contributions
AP performed data analysis, wrote the manuscript. AK edited the manuscript. EVN, PER, MP, SD and PZ contributed to data acquisition. JF supervised data acquisition and analysis, wrote and edited the manuscript.
CONFLICTS OF INTEREST
Jérome Fayette has been paid honoraria by BMS.
FUNDING
This study was not supported by any pharmaceutical corporation or company.
REFERENCES
1. Fact Sheets by Population. Available from: http://globocan.iarc.fr/Pages/fact_sheets_population.aspx.
2. Shetty AV, Wong DJ. Systemic Treatment for Squamous Cell Carcinoma of the Head and Neck. Otolaryngol Clin North Am. 2017; 50:775–82. https://doi.org/10.1016/j.otc.2017.03.013.
3. Argiris A, Karamouzis MV, Raben D, Ferris RL. Head and neck cancer. Lancet. 2008; 371:1695–709. https://doi.org/10.1016/S0140-6736(08)60728-X.
4. Fayette J, Montella A, Chabaud S, Bachelot T, Pommier P, Girodet D, Racadot S, Montbarbon X, Favier B, Zrounba P. Paclitaxel is effective in relapsed head and neck squamous cell carcinoma: a retrospective study of 66 patients at a single institution. Anticancer Drugs. 2010; 21:553–58. https://doi.org/10.1097/CAD.0b013e3283388e60.
5. Magnes T, Melchardt T, Weiss L, Mittermair C, Neureiter D, Klieser E, Gampenrieder S, Moser G, Gaggl A, Greil R, Egle A. Prognostic score in patients with recurrent or metastatic carcinoma of the head and neck treated with cetuximab and chemotherapy. PLoS One. 2017; 12:e0180995. https://doi.org/10.1371/journal.pone.0180995.
6. Péron J, Ceruse P, Lavergne E, Buiret G, Pham BN, Chabaud S, Favier B, Girodet D, Zrounba P, Ramade A, Fayette J. Paclitaxel and cetuximab combination efficiency after the failure of a platinum-based chemotherapy in recurrent/metastatic head and neck squamous cell carcinoma. Anticancer Drugs. 2012; 23:996–1001. https://doi.org/10.1097/CAD.0b013e32835507e5.
7. Posner MR, Hershock DM, Blajman CR, Mickiewicz E, Winquist E, Gorbounova V, Tjulandin S, Shin DM, Cullen K, Ervin TJ, Murphy BA, Raez LE, Cohen RB, et al, and TAX 324 Study Group. Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl J Med. 2007; 357:1705–15. https://doi.org/10.1056/NEJMoa070956.
8. Price KA, Cohen EE. Current treatment options for metastatic head and neck cancer. Curr Treat Options Oncol. 2012; 13:35–46. https://doi.org/10.1007/s11864-011-0176-y.
9. Head and Neck Tumors | Cancer Network | The Oncology Journal. Available from: http://www.cancernetwork.com/cancer-management/head-and-neck-tumors.
10. Vermorken JB, Mesia R, Rivera F, Remenar E, Kawecki A, Rottey S, Erfan J, Zabolotnyy D, Kienzer HR, Cupissol D, Peyrade F, Benasso M, Vynnychenko I, et al. Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med. 2008; 359:1116–27. https://doi.org/10.1056/NEJMoa0802656.
11. Quoix E, Zalcman G, Oster JP, Westeel V, Pichon E, Lavolé A, Dauba J, Debieuvre D, Souquet PJ, Bigay-Game L, Dansin E, Poudenx M, Molinier O, et al, and Intergroupe Francophone de Cancérologie Thoracique. Carboplatin and weekly paclitaxel doublet chemotherapy compared with monotherapy in elderly patients with advanced non-small-cell lung cancer: IFCT-0501 randomised, phase 3 trial. Lancet. 2011; 378:1079–88. https://doi.org/10.1016/S0140-6736(11)60780-0.
12. Reck M, Popat S, Reinmuth N, De Ruysscher D, Kerr KM, Peters S, and ESMO Guidelines Working Group. Metastatic non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014 (Suppl 3); 25:iii27–39. https://doi.org/10.1093/annonc/mdu199.
13. Gibson MK, Li Y, Murphy B, Hussain MH, DeConti RC, Ensley J, Forastiere AA, and Eastern Cooperative Oncology Group. Randomized phase III evaluation of cisplatin plus fluorouracil versus cisplatin plus paclitaxel in advanced head and neck cancer (E1395): an intergroup trial of the Eastern Cooperative Oncology Group. J Clin Oncol. 2005; 23:3562–67. https://doi.org/10.1200/JCO.2005.01.057.
14. Govindan R, Szczesna A, Ahn MJ, Schneider CP, Gonzalez Mella PF, Barlesi F, Han B, Ganea DE, Von Pawel J, Vladimirov V, Fadeeva N, Lee KH, Kurata T, et al. Phase III Trial of Ipilimumab Combined With Paclitaxel and Carboplatin in Advanced Squamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2017; 35:3449–57. https://doi.org/10.1200/JCO.2016.71.7629.
15. Moosmann P, Egli F, Stahel RA, Jost L. Weekly paclitaxel and carboplatin combination chemotherapy in patients with advanced squamous cell carcinoma of the head and neck. Onkologie. 2003; 26: 568–72. https://doi.org/10.1159/000074153.
16. Sacco AG, Cohen EE. Current Treatment Options for Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma. J Clin Oncol. 2015; 33:3305–13. https://doi.org/10.1200/JCO.2015.62.0963.
17. Vermorken JB, Trigo J, Hitt R, Koralewski P, Diaz-Rubio E, Rolland F, Knecht R, Amellal N, Schueler A, Baselga J. Open-label, uncontrolled, multicenter phase II study to evaluate the efficacy and toxicity of cetuximab as a single agent in patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck who failed to respond to platinum-based therapy. J Clin Oncol. 2007; 25:2171–77. https://doi.org/10.1200/JCO.2006.06.7447.
18. Martinez-Trufero J, Isla D, Adansa JC, Irigoyen A, Hitt R, Gil-Arnaiz I, Lambea J, Lecumberri MJ, Cruz JJ. Phase II study of capecitabine as palliative treatment for patients with recurrent and metastatic squamous head and neck cancer after previous platinum-based treatment. Br J Cancer. 2010; 102:1687–91. https://doi.org/10.1038/sj.bjc.6605697.
19. Péron J, Polivka V, Chabaud S, Poupart M, Ceruse P, Ramade A, Girodet D, Zrounba P, Fayette J. An effective and well-tolerated strategy in recurrent and/or metastatic head and neck cancer: successive lines of active chemotherapeutic agents. BMC Cancer. 2014; 14:504. https://doi.org/10.1186/1471-2407-14-504.
20. Jiménez B, Trigo JM, Pajares BI, Sáez MI, Quero C, Navarro V, Llácer C, Medina L, Rueda A, Alba E. Efficacy and safety of weekly paclitaxel combined with cetuximab in the treatment of pretreated recurrent/metastatic head and neck cancer patients. Oral Oncol. 2013; 49:182–85. https://doi.org/10.1016/j.oraloncology.2012.09.003.
21. Nakano K, Marshall S, Taira S, Sato Y, Tomomatsu J, Sasaki T, Shimbashi W, Fukushima H, Yonekawa H, Mitani H, Kawabata K, Takahashi S. A comparison of weekly paclitaxel and cetuximab with the EXTREME regimen in the treatment of recurrent/metastatic squamous cell head and neck carcinoma. Oral Oncol. 2017; 73:21–26. https://doi.org/10.1016/j.oraloncology.2017.07.022.
22. Bossi P, Miceli R, Locati LD, Ferrari D, Vecchio S, Moretti G, Denaro N, Caponigro F, Airoldi M, Moro C, Vaccher E, Sponghini A, Caldara A, et al. A randomized, phase 2 study of cetuximab plus cisplatin with or without paclitaxel for the first-line treatment of patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck. Ann Oncol. 2017; 28:2820–26. https://doi.org/10.1093/annonc/mdx439.
23. Guigay J, Fayette J, Dillies AF, Sire C, Kerger JN, Tennevet I, Machiels JP, Zanetta S, Pointreau Y, Bozec Le Moal L, Henry S, Schilf A, Bourhis J. Cetuximab, docetaxel, and cisplatin as first-line treatment in patients with recurrent or metastatic head and neck squamous cell carcinoma: a multicenter, phase II GORTEC study. Ann Oncol. 2015; 26:1941–47. https://doi.org/10.1093/annonc/mdv268.
24. Ferris RL, Blumenschein G Jr, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE, Even C, Worden F, Saba NF, Iglesias Docampo LC, et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med. 2016; 375:1856–67. https://doi.org/10.1056/NEJMoa1602252.
25. Harrington KJ, Ferris RL, Blumenschein G Jr, Colevas AD, Fayette J, Licitra L, Kasper S, Even C, Vokes EE, Worden F, Saba NF, Kiyota N, Haddad R, et al. Nivolumab versus standard, single-agent therapy of investigator’s choice in recurrent or metastatic squamous cell carcinoma of the head and neck (CheckMate 141): health-related quality-of-life results from a randomised, phase 3 trial. Lancet Oncol. 2017; 18:1104–15. https://doi.org/10.1016/S1470-2045(17)30421-7.
26. Study of Nivolumab in Combination With Ipilimumab Compared to the Standard of Care (Extreme Study Regimen) as First Line Treatment in Patients With Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck - Full Text View - ClinicalTrials. gov. [cited 2018 Mar 3]. Available from: https://clinicaltrials.gov/ct2/show/NCT02741570.
27. Rizvi NA, Hellmann MD, Brahmer JR, Juergens RA, Borghaei H, Gettinger S, Chow LQ, Gerber DE, Laurie SA, Goldman JW, Shepherd FA, Chen AC, Shen Y, et al. Nivolumab in Combination With Platinum-Based Doublet Chemotherapy for First-Line Treatment of Advanced Non-Small-Cell Lung Cancer. J Clin Oncol. 2016; 34:2969–79. https://doi.org/10.1200/JCO.2016.66.9861.
28. Kanda S, Goto K, Shiraishi H, Kubo E, Tanaka A, Utsumi H, Sunami K, Kitazono S, Mizugaki H, Horinouchi H, Fujiwara Y, Nokihara H, Yamamoto N, et al. Safety and efficacy of nivolumab and standard chemotherapy drug combination in patients with advanced non-small-cell lung cancer: a four arms phase Ib study. Ann Oncol. 2016; 27:2242–50. https://doi.org/10.1093/annonc/mdw416.
29. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009; 45:228–47. https://doi.org/10.1016/j.ejca.2008.10.026.

