INTRODUCTION
Pancreatic cancer ranks as the 7th most common human cancer, with a 5-year survival rate of 5% encompassing all stages of disease [1]. In 2012, 338,000 new cases were reported, while 330,000 deaths occurred despite chemotherapy, with or without surgery [1]. Europe and North America account for more than 40% of the worldwide burden of pancreatic cancer disease and mortality [1]. More than 90 % of newly diagnosed cases with pancreatic cancer represent ductal adenocarcinoma [1]. Only 1/5 of patients (20% of cases) qualify for surgery; the majority of patients present with metastatic disease at the time of diagnosis [2]. Improved treatment modalities for patients with pancreatic cancer are therefore urgently needed.
Current T cell-based immunotherapies have shown clinically durable responses not only in patients with melanoma, yet more recently also in patients with tumors of different histologies, associated with the activation and mobilization of T cells recognizing tumor-associated antigens (TAAs) [3]. Since not all TAAs elicit an equally efficacious immune response, novel targets that can mediate biologically and clinically relevant cellular immune responses are needed. Importantly, T cells targeting the patient’s ‘private antigens’ i.e. tumor-associated mutations, including tumor ‘driver’ mutations such as V-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog (KRAS) genetic aberrations, are associated with tumor regression and long-term survival in patients with gastrointestinal tumors [4].
Mature mesothelin represents a non-mutated, cell surface-bound TAA that is expressed by mesothelial cells lining various internal organs, including the pleura, pancreas, peritoneum and the pericard [5]. Overexpression of mesothelin has been identified in a number of human cancers e.g mesothelioma, lung cancer, colorectal cancer, ovarian cancer as well as pancreatic cancer [5-9]. Virtually all pancreatic adenocarcinomas/ductal adenocarcinomas have been found to be positive for mesothelin expression [10-12]. Mesothelin comprises several components: a 36-amino acid (aa)-long signal peptide, (i) the megakaryocyte-potentiating factor (MPF), which is shed into the bloodstream upon enzymatic cleavage (aa 37-286) and (ii) the GPI-anchored mature mesothelin, which is overexpressed in tumor tissues (aa 296-606; with a pro-peptide, aa 607-630, that is cleaved during maturation/activation) [5]. Mesothelin-specific T cell responses have been observed in peripheral blood lymphocytes from patients with pancreatic cancer, including individuals who were administered a cancer vaccine candidate overexpressing mesothelin peptides and granulocyte-macrophage colony-stimulating factor [6, 13-15]. Here we show that mesothelin-specific interferon gamma (IFN-γ) production by peripheral blood lymphocytes is a reliable predictor of survival among patients with pancreatic cancer. We also provide evidence that mesothelin-specific cellular immune responses can be amplified in vitro with interleukin (IL)-21 conditioning, possibly with preferential expansion of T-cells directed against TAAs. This is the first report to establish a clinically relevant link between mesothelin-directed cellular immune responses in peripheral blood lymphocytes and increased survival of patients with pancreatic cancer. Mesothelin-specific immune responses may also aid to identify patients i) at increased risk for tumor progression ii) who may benefit from a more frequent followup and alternate treatment strategies, including immunotherapeutic strategies with the aim to expand anti-cancer T-cells.
RESULTS
A description of the patient cohort is provided in Table 1. Whole blood assays were used for gauging immune responses, since peripheral blood mononuclear cells (PBMCs) constitute a reliable readout of cellular immune responses. IL-21 has been shown to promote preferential expansion of high-affinity, antigen-specific T cells and to enrich antigen-experienced (memory) CD8+ and CD4+ T cells in the host [16-18]. These effects are also observed in association with IL-15 [19] as well as IL-7 that promote antigen-specific T-cell proliferation and anti-tumor T-cell functions [20]. In addition, we have previously reported a cytokine cocktail, consisting of IL-2, IL-15 and IL-21, that leads to the accumulation of central memory tumor infiltrating T-cells (TILs), obtained from patients with glioblastoma as well as pancreatic tumors [21, 22]. Therefore, we decided to use IL-21 in the experiments in order to investigate the ‘conditioning effect’ of IL-21 on mesothelin-specific T cells from patients with pancreatic cancer. Pertaining to evaluation of the patients’ cellular immune responses, strong IFN-γ production in response to anti-human CD3 antibody (OKT3) stimulation in peripheral blood from patients with pancreatic cancer appeared to be associated with a survival benefit in the presence, but not in the absence of IL-21 conditioning (Figure 1A & 1B). While robust IFN-γ responses to OKT3 without IL-21 conditioning did not correlate with patient survival, the patients whose peripheral blood lymphocytes produced more IFN-γ after OKT3 stimulation (with IL-21 conditioning) displayed a negative survival benefit (p=0.016). In contrast, increased IFN-γ responses to the precursor mesothelin peptide pool stimulation, with IL-21 conditioning, was associated with increased survival (p=0.007) (Figure 1C). A survival benefit in relation to precursor mesothelin-directed IFN-γ production was also observed when the patients’ peripheral blood was not conditioned with IL-21 (p=0.002) (Figure 1D). The positive correlation between survival and the precursor mesothelin-induced cellular immune response proved to be linked to the 23 peptides belonging to the cell-bound, mature mesothelin component (p=0.018) only in the presence of IL-21 conditioning (Figure 1E and 1F). We did not find an association between IFN-γ responses to the mesothelin MPF component and improved survival of patients with pancreatic cancer (data not shown).
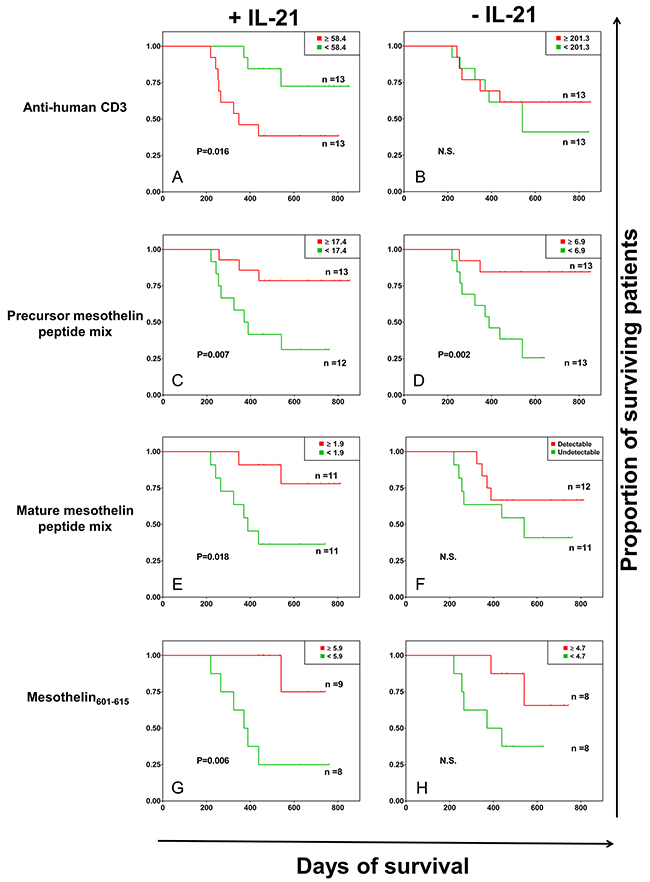
Figure 1: Kaplan-Meier survival analysis of patients with pancreatic cancer based on antigen-specific IFN-γ responses. A whole blood assay was performed (7-day incubation) in 96-well microtiter plates, with or without IL-21 conditioning. The following stimulation conditions were applied: (A and B) anti-human CD3 antibody/OKT3 as positive control; (C and D) full-length mesothelin, the 42 peptides corresponding to the MPF and the mature mesothelin components; (E and F) mature mesothelin peptide mix, the 23 peptides spanning the GPI-anchored, cell surface-bound mature component of the mesothelin molecule –expressed on cancer cells; (G and H) mesothelin601-615, which was found to be highly immunogenic in an initial screen (presented in Supplementary Table 2 and Supplementary Figure 1). Culture medium was used as negative control. Supernatants were harvested for IFN-γ measurement by a standard sandwich ELISA, and the negative control values were subtracted from the final values reported. Shown are the survival of patients with pancreatic cancer in relation to IFN-γ production by peripheral blood T cells, based on the median cut-off values of IFN-γ levels (in pg/ml of cytokine concentration). P<0.05 was considered significant, while ‘N.S.’ denotes a non-significant p value.
Table 1: Clinical characteristics of participating patients
Patient ID (#) |
Age (years) |
Gender (M/F) |
Diagnosis |
|---|---|---|---|
2 |
68 |
M |
Adenocarcinoma |
5 |
74 |
M |
Adenocarcinoma |
7 |
71 |
M |
Ductal adenocarcinoma |
19 |
62 |
F |
Ductal adenocarcinoma |
24 |
81 |
M |
Ductal adenocarcinoma |
31 |
77 |
M |
Ductal adenocarcinoma |
34 |
50 |
M |
Ductal adenocarcinoma |
41 |
75 |
F |
Ductal adenocarcinoma |
50 |
75 |
F |
Ductal adenocarcinoma |
51 |
58 |
F |
Ductal adenocarcinoma |
59 |
60 |
M |
Ductal adenocarcinoma |
62 |
50 |
M |
Ductal adenocarcinoma |
63 |
61 |
M |
Ductal adenocarcinoma |
68 |
56 |
F |
Ductal adenocarcinoma |
72 |
48 |
F |
Ductal adenocarcinoma |
74 |
55 |
F |
Ductal adenocarcinoma |
76 |
54 |
M |
Ductal adenocarcinoma |
81 |
63 |
M |
Ductal adenocarcinoma + BD-IPMN |
87 |
66 |
F |
Pancreatobiliary ductal adenocarcinoma |
99 |
50 |
F |
Pancreatobiliary ductal adenocarcinoma |
104 |
76 |
F |
Pancreatobiliary ductal adenocarcinoma |
107 |
81 |
M |
Pancreatobiliary ductal adenocarcinoma |
114 |
62 |
F |
Pancreatobiliary ductal adenocarcinoma |
116 |
67 |
M |
Pancreatobiliary ductal adenocarcinoma |
117 |
70 |
M |
Pancreatobiliary ductal adenocarcinoma |
120 |
72 |
M |
Pancreatobiliary ductal adenocarcinoma |
Age median |
64.5 years |
||
Male:female ratio |
1.36:1 |
(M = 15, F = 11) |
|
In an initial in vitro screen to identify immune recognition hotspots within the mesothelin molecule (pertaining to T-cell reactivity), the mesothelin601-615 peptide (MQEALSGTPCLLGPG) which is part of the mature mesothelin molecule expressed on the surface of pancreatic cancer cells, was identified as an immunodominant mesothelin peptide (Supplementary Figure 1 and Supplementary Table 2). The mesothelin586-600 peptide (LQGGIPNGYLVLDLS), which was also tested, resulted in strong cytokine production from T cells in the mesothelin screening experiments, and was thus used as an internal control for the activity of mesothelin601-615 in the present study with regard to survival of patients with pancreatic cancer. We observed that patient survival was associated with T-cell responses directed against the mesothelin601-615 peptide (p=0.006) (Figure 1G). A weaker but similar trend of patient survival associated with mesothelin601-615-specific IFN-γ production was also found without IL-21 conditioning albeit absence of statistical significance (Figure 1H). We did not observe a survival benefit for patients with pancreatic cancer in relation to T-cell responses directed to the mesothelin586-600 peptide, in the presence or absence of IL-21 conditioning (Figure 2A and 2B).
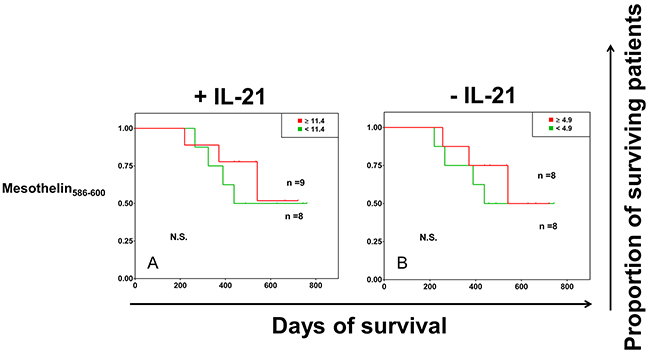
Figure 2: (A and B). Identical experimental layout as in Figure 1. The peptide derived from mesothelin586-600 was used as a control peptide which was found to be recognized in the ‘hotspot identification’ (Supplementary Table 2 and Supplementary Figure 1). Culture medium was used as negative control. Supernatants were harvested for IFN-γ measurement by a standard sandwich ELISA, and the negative control values were subtracted from the final values reported. Shown are the survival of patients with pancreatic cancer in relation to IFN-γ production by peripheral blood T cells, based on the median cut-off values of IFN-γ levels (in pg/ml of cytokine concentration). ‘N.S.’ denotes a non-significant p value.
In the absence of IL-21 conditioning, the mesothelin601-615 peptide induced the strongest IFN-γ responses in peripheral blood of patients with pancreatic cancer, as compared to the precursor mesothelin peptide mix, the mature mesothelin peptide mix or the control mesothelin586-600 peptide (Supplementary Table 1). With IL-21 conditioning, the general pattern of the IFN-γ response to mesothelin586-600 was increased by more than 3-fold, while the response to mesothelin601-615 remained unchanged. With regard to differences between individual patients, we observed a general increase of IFN-γ production using the precursor mesothelin- or mature mesothelin stimulation, i.e. between a 2- and 6-fold increase of IFN-γ production in some samples. While there were some blood samples that exhibited a robust increase in IFN-γ production in the OKT3 group with IL-21 conditioning, other samples displayed total abrogation of cytokine responses after IL-21 conditioning. Interestingly, such a stark difference in IFN-γ responses, i.e. with or without IL-21 conditioning, was not seen among the individual peripheral blood samples when treated with medium alone (i.e. no specific stimulus was applied and therefore the spontaneous IFN-γ production was measured).
DISCUSSION
Immunoreactivity studies gauging T-cell responses to mesothelin have been previously reported. Cytotoxic CD8+ T-cell responses to mesothelin-derived epitopes restricted by human leukocyte antigens (HLA)-A2, A3 and A24 have been described in surgically treated patients with pancreatic adenocarcinoma who received granulocyte-macrophage colony-stimulating factor-immunotherapy [23]. In addition, CD4+ T-cell mesothelin epitopes were also shown to induce IFN-γ production in peripheral blood from patients with pancreatic cancer [15]. In the present study, we show for the first time that IL-21 conditioning enhances mesothelin-specific T-cell responses, reflected by IFN-γ production in peripheral blood mononuclear cells, which correlates with increased survival of patients with pancreatic cancer who undergo surgery. The immunological recognition pattern, i.e. that only mature mesothelin (but not the MPF component of mesothelin), and particularly the immunodominant MQEALSGTPCLLGPG peptide (mesothelin aa 601-615) within the mature mesothelin is independently associated with survival, lends support that mesothelin-directed IFN-γ production provides a clinically relevant immune signature associated with improved clinical outcome.
IL-21 has been studied in preclinical models of cancer pertaining to the maintenance of antigen-specific CD8 T-cell responses [24], as well as in relation to the proliferation, survival and functionality of cancer-specific T cells isolated from patients with melanoma or pancreatic cancer [22, 25]. Additionally, activation of the IL-21/IL-21 receptor (IL-21R) pathway in T cells facilitates better control of human immunodeficiency virus (HIV) replication [26], while children with IL-21R deficiency are predisposed to opportunistic infections due to impaired cellular immunity [27].
More recently, exogenous IL-21 was shown to inhibit the expression of FoxP3, the cardinal transcription factor associated with regulatory T cells, in human CD4+ T cells following encounter with cancer cells – in a manner similar to TGF-β blockade [28]. We reported previously previously that a cytokine cocktail comprising IL-2, IL-15 and IL-21 selectively increases the population of central/effector memory tumor-reactive tumor-infiltrating lymphocytes (TIL) from pancreatic cancer as well as TIL from WHO grade 4 glioma after ex vivo expansion [21, 22]. Thus, IL-21 potentially selects for the long-term maintenance of a restricted, but highly efficient pool of antigen-specific T cells in the host – with a strong influence on disease control (cancer, chronic infections) and survival, a key observation with regard to the cellular therapy of cancer.
Importantly, the enhanced survival benefit seen among patients whose IFN-γ response to mesothelin601-615 was increased in the presence of IL-21 conditioning shows that peripheral blood immune cells recognizing this peptide are likely to express IL-21R on their surface. Interestingly, a study published in 2014 showed that mesothelin601-615 was exclusively recognized by circulating CD4+ T cells from healthy individuals, but not by T cells from patients with pancreatic cancer [15]. However, this was performed in the absence of IL-21 – conditioning, but with IL-2 supplementation for 9-10 days – which expands T-cells with a broad spectrum of TCR specificities, while IL-21 is more selective, particularly for memory CD8+ T cells.
Specific mesothelin epitopes, that are recognized by human CD8+ T cells, have been described before, although the amino acid residues 601-615 (as well as 586-600) are not among those previously reported [13, 29, 30]. It is thus possible that T cells with TCRs that are directed against mesothelin601-615 respond better to IL-21, and can be expanded to produce a measurable immunological readout in the presence of IL-21. Targeted immune responses to mesothelin601-615 may also bear clinical relevance in the diagnosis of patients with pancreatic cancer, since the use of a single peptide antigen may provide highly accurate immune-based readouts - in addition to being financially feasible for routine applications. T-cell reactivity to mesothelin601-615 could therefore be exploited in the context of immune monitoring and treatment outcomes of neoadjuvant therapy, chemotherapy as well as targeted immunotherapies in patients with cancer. Although we observed a significant increase in IFN-γ production in response to mesothelin586-600 after IL-21 conditioning, which was used as an internal control for comparison, we did not observe a correlation of cytokine response(s) to mesothelin586-600 with improved survival of patients with pancreatic cancer. It is possible that mesothelin586-600 does not encode a T-cell epitope for T cells that can recognize, kill or contain tumor cells and may therefore fail to evoke a clinically relevant and measurable immune reactivity in vitro.
Increased IFN-γ production, following OKT3 stimulation, was shown to be associated with decreased survival, a situation that may reflect non-productive inflammation and antigen-specific T-cell exhaustion [31]. The cell response to OKT3 is polyclonal due to activation of the TCR, and is therefore non-specific i.e. not related to a particular antigenic stimulus. Conversely, the antigen peptide mix represents a much more targeted cellular immune response i.e. by TCRs that recognize and respond to their cognate epitope(s). Since patients with pancreatic cancer are immunologically disadvantaged compared to healthy individuals, the OKT3 response allows for a more general overview of the functional T-cell status, and whether the T cells are able to respond to TCR stimulation at all.
We also observed that IL-21 conditioning of peripheral blood resulted in decreased IFN-γ production among circulating immune cells activated with the anti-human CD3 antibody (OKT3) – which may reflect the general immunological status of the cell-mediated immune response. This is plausible, since IL-21 can selectively inhibit IFN-γ production by memory Th17 cells and CD4+ Th1 effector cells and promote cytokine production by memory T-cell populations [32]. Furthermore, IL-21 can disrupt the activation and maturation of dendritic cells during an immune response, in addition to downregulation of MHC class II molecules [33]. This supports the case that memory CD8+ T cells are more likely to benefit from ‘IL-21 conditioning’, since they do not require APC-dependent activation, nor are they dependent on MHC class II-restricted antigen presentation.
Peripheral blood T cells from some patients produced more IFN-γ than others. This difference can be attributed to several factors: the extent of disease in each patient (at diagnosis, when blood was drawn), the general immunological status (in part reflected by the OKT3-directed cellular immune response), as well as the age of the patient. Nevertheless, we also observed that the background of IFN-γ production in peripheral blood i.e. in the absence of IL-21 conditioning, was rather low in the majority of patients. Thus, we hypothesize that IL-21 exhibits a biologically relevant and generally positive effect on circulating immune cells in patients with pancreatic cancer, associated with expansion of certain TCR specificities, such as specific mesothelin epitopes, described in this report.
MATERIALS AND METHODS
Patients
The Regional Ethics Review Board (Regionala etikprövningsnämnden) at Karolinska Institutet, Stockholm, Sweden, approved the study (diary number: 2013/977-31/1). Peripheral blood was obtained by venepuncture from 26 patients with pancreatic ductal adenocarcinoma (PDA) prior to starting on chemotherapy and undergoing surgery. A description of the patient cohort is provided in Table 1.
Whole-blood assay
Whole blood assays (WBA) were performed by first diluting peripheral blood 5x with RPMI 1640 L-glutamine medium (ThermoFisher Scientific, Carlsbad, CA), followed by co-culture with 42 peptides spanning the full-length precursor mesothelin molecule before furin cleavage (19 peptides - MPF component; 23 peptides – mature mesothelin component), (Peptide and Elephants, Berlin, Germany) with or without conditioning with recombinant human IL-21 (10ng/ml) (Prospec, Rehovot, Israel) in 96-well plates (200μl diluted blood/well). The mature mesothelin epitope MQEALSGTPCLLGPG (mesothelin601-615) was also tested separately since we found this peptide to induce strong T-cell reactivity, defined by IFN-γ production, in peripheral blood obtained from eight patients with pancreatic cancer in an initial screen (Supplementary Information). In the same screen, another mesothelin peptide, LQGGIPNGYLVLDLS (mesothelin586-600), was also found to induce IFN-γ production, and was thus selected as an internal control for comparison in the present study. Other WBA antigens included in the study are listed in Table 2. Anti-human CD3 antibody (OKT3) was used as positive control while medium alone served as negative control. WBA plates were incubated for 7 days at 37°C with 5% CO2. Supernatants were harvested for IFN-γ detection by sandwich ELISA (Mabtech, Stockholm, Sweden). Final concentrations of antigen-specific IFN-γ production (pg/ml) were recorded after subtracting medium control values.
Table 2: List of antigens and controls used in the whole blood assay
Antigen |
Type |
Final concentration |
Access NO. |
Company |
|---|---|---|---|---|
Anti-human CD3 antibody |
antibody |
30ng/ml |
Clone:OKT3 |
Biolegend |
Precursor mesothelin |
peptide mix |
1μg/peptide/ml |
Q13421 (UniprotKB) |
Peptides&Elephants |
Mature mesothelin |
peptide mix |
1μg/peptide/ml |
A12-114LB |
Peptides&Elephants |
MPF |
peptide mix |
1μg/peptide/ml |
A12-114LB |
Peptides&Elephants |
Mesothelin 586-600 |
single peptide |
1μg/ml |
Seq: LQGGIPNGYLVLDLS |
Peptides&Elephants |
Mesothelin 601-615 |
single peptide |
1μg/ml |
Seq: MQEALSGTPCLLGPG |
Peptides&Elephants |
Statistical analysis
Survival probability was evaluated using Kaplan-Meier curves and long-rank test. The cut-off median values of antigen-specific IFN-γ production were determined based on the intensity of response to each antigenic stimulus tested.
CONCLUSIONS
IFN-γ responses in T-cells from peripheral blood, along with IL-21 conditioning and OKT3 stimulation, reflect pro-inflammatory cellular immune reactivity and decreased survival. Patients with subdued mesothelin-specific IFN-γ production appear to be at higher risk for cancer progression and would most likely benefit adapted monitoring and more targeted treatment strategies. Importantly, we are able to show that a single epitope (mesothelin601-615) can predict the probability of survival in patients with pancreatic cancer. This finding has major implications for novel diagnostic tests, whereby the use of a single epitope to measure clinically relevant immune responses in patients with pancreatic cancer increases feasibility, technical applicability and robustness. For patients undergoing immune checkpoint therapy i.e. with anti-programmed cell death 1 (PD-1), an increase in mesothelin601-615-specific T cells could be indicative of improved clinical outcomes, although this needs to be formally evaluated. Preclinical (murine model of human pancreatic ductal adenocarcinoma) evaluation of a TCR-engineered T-cell product specific for mesothelin406-414 showed increased immune cell infiltration into the tumor site, coupled with improved survival of mice [12]. Furthermore, mesothelin-directed chimeric antigen receptors (CAR) are also in clinical trials for various cancers [34]. A clinical trial in its recruitment phase for investigating the safety, efficacy and tolerability of anti-mesothelin antibody (BMS-986148) in conjunction with anti-PD-1 (nivolumab) in patients with advanced solid tumors including patients with pancreatic cancer (NCT02341625) is underway. From an immunotherapy viewpoint, the absence of naturally-occurring mesothelin-directed cellular immune responses in the patient may be augmented with supportive strategies, if an appropriate T-cell repertoire exists that can be measured in vitro by expansion of mesothelin-specific T cells, e.g. using adjunctive IL-21 conditioning. Our findings call for a more tailored follow-up of patients with pancreatic cancer and encourage host-directed therapies for patients with advanced gastrointestinal malignancies.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
FUNDING
This study was supported by grants from the Swedish Research Council (VR), the Swedish Heart and lung Foundation (Hjärtlungfonden) and the Söderberg Foundation to MM; Swedish Childhood Cancer Foundation (Barncancerfonden) to IE; China Scholarship Council to QM, ZL and SX.
REFERENCES
1. WHO. World cancer report 2014. Lyon: International Agency for Research on Cancer, World Health Organisation; 2014.
2. American Cancer Society. Pancreatic cancer stages: american cancer society; 2016 [cited 2016 16.11.2016]. Available from: http://www.cancer.org/cancer/pancreaticcancer/detailedguide/pancreatic-cancer-staging.
3. Rosenberg SA, Restifo NP. Adoptive cell transfer as personalized immunotherapy for human cancer. Science. 2015; 348:62–68. https://doi.org/10.1126/science.aaa4967.
4. Tran E, Robbins PF, Lu YC, Prickett TD, Gartner JJ, Jia L, Pasetto A, Zheng Z, Ray S, Groh EM, Kriley IR, Rosenberg SA. T-cell transfer therapy targeting mutant KRAS in cancer. N Engl J Med. 2016; 375:2255–62. https://doi.org/10.1056/NEJMoa1609279.
5. Pastan I, Hassan R. Discovery of mesothelin and exploiting it as a target for immunotherapy. Cancer Res. 2014; 74:2907–12. https://doi.org/10.1158/0008-5472.CAN-14-0337.
6. Johnston FM, Tan MC, Tan BR Jr, Porembka MR, Brunt EM, Linehan DC, Simon PO Jr, Plambeck-Suess S, Eberlein TJ, Hellstrom KE, Hellstrom I, Hawkins WG, Goedegebuure P. Circulating mesothelin protein and cellular antimesothelin immunity in patients with pancreatic cancer. Clin Cancer Res. 2009; 15:6511–8. https://doi.org/10.1158/1078-0432.CCR-09-0565.
7. Kachala SS, Bograd AJ, Villena-Vargas J, Suzuki K, Servais EL, Kadota K, Chou J, Sima CS, Vertes E, Rusch VW, Travis WD, Sadelain M, Adusumilli PS. Mesothelin overexpression is a marker of tumor aggressiveness and is associated with reduced recurrence-free and overall survival in early-stage lung adenocarcinoma. Clin Cancer Res. 2014; 20:1020–8. https://doi.org/10.1158/1078-0432.CCR-13-1862.
8. Morello A, Sadelain M, Adusumilli PS. Mesothelin-targeted CARs: driving T cells to solid tumors. Cancer Discov. 2016; 6:133–46. https://doi.org/10.1158/2159-8290.CD-15-0583.
9. Servais EL, Colovos C, Rodriguez L, Bograd AJ, Nitadori J, Sima C, Rusch VW, Sadelain M, Adusumilli PS. Mesothelin overexpression promotes mesothelioma cell invasion and MMP-9 secretion in an orthotopic mouse model and in epithelioid pleural mesothelioma patients. Clin Cancer Res. 2012; 18:2478–89. https://doi.org/10.1158/1078-0432.CCR-11-2614.
10. Argani P, Iacobuzio-Donahue C, Ryu B, Rosty C, Goggins M, Wilentz RE, Murugesan SR, Leach SD, Jaffee E, Yeo CJ, Cameron JL, Kern SE, Hruban RH. Mesothelin is overexpressed in the vast majority of ductal adenocarcinomas of the pancreas: identification of a new pancreatic cancer marker by serial analysis of gene expression (SAGE). Clin Cancer Res. 2001; 7:3862–8.
11. Hassan R, Laszik ZG, Lerner M, Raffeld M, Postier R, Brackett D. Mesothelin is overexpressed in pancreaticobiliary adenocarcinomas but not in normal pancreas and chronic pancreatitis. Am J Clin Pathol. 2005; 124:838–45. https://doi.org/10.1309/F1B64CL7H8VJKEAF.
12. Stromnes IM, Schmitt TM, Hulbert A, Brockenbrough JS, Nguyen H, Cuevas C, Dotson AM, Tan X, Hotes JL, Greenberg PD, Hingorani SR. T cells engineered against a native antigen can surmount immunologic and physical barriers to treat pancreatic ductal adenocarcinoma. Cancer Cell. 2015; 28:638–52. https://doi.org/10.1016/j.ccell.2015.09.022.
13. Thomas AM, Santarsiero LM, Lutz ER, Armstrong TD, Chen YC, Huang LQ, Laheru DA, Goggins M, Hruban RH, Jaffee EM. Mesothelin-specific CD8(+) T cell responses provide evidence of in vivo cross-priming by antigen-presenting cells in vaccinated pancreatic cancer patients. J Exp Med. 2004; 200:297–306. https://doi.org/10.1084/jem.20031435.
14. Jaffee EM, Hruban RH, Biedrzycki B, Laheru D, Schepers K, Sauter PR, Goemann M, Coleman J, Grochow L, Donehower RC, Lillemoe KD, O’Reilly S, Abrams RA, et al. Novel allogeneic granulocyte-macrophage colony-stimulating factor-secreting tumor vaccine for pancreatic cancer: a phase I trial of safety and immune activation. J Clin Oncol. 2001; 19:145–56. https://doi.org/10.1200/JCO.2001.19.1.145.
15. Chen Y, Ayaru L, Mathew S, Morris E, Pereira SP, Behboudi S. Expansion of anti-mesothelin specific CD4+ and CD8+ T cell responses in patients with pancreatic carcinoma. PLoS One. 2014; 9:e88133. https://doi.org/10.1371/journal.pone.0088133.
16. Allard EL, Hardy MP, Leignadier J, Marquis M, Rooney J, Lehoux D, Labrecque N. Overexpression of IL-21 promotes massive CD8+ memory T cell accumulation. Eur J Immunol. 2007; 37:3069–77. https://doi.org/10.1002/eji.200637017.
17. Yuan Y, Yang Y, Huang X. IL-21 is required for CD4 memory formation in response to viral infection. JCI Insight. 2017; 2:e90652. https://doi.org/10.1172/jci.insight.90652.
18. Novy P, Huang X, Leonard WJ, Yang Y. Intrinsic IL-21 signaling is critical for CD8 T cell survival and memory formation in response to vaccinia viral infection. J Immunol. 2011; 186:2729–38. https://doi.org/10.4049/jimmunol.1003009.
19. Moroz A, Eppolito C, Li Q, Tao J, Clegg CH, Shrikant PA. IL-21 enhances and sustains CD8+ T cell responses to achieve durable tumor immunity: comparative evaluation of IL-2, IL-15, and IL-21. J Immunol. 2004; 173:900–09. https://doi.org/10.4049/jimmunol.173.2.900.
20. Liu S, Lizée G, Lou Y, Liu C, Overwijk WW, Wang G, Hwu P. IL-21 synergizes with IL-7 to augment expansion and anti-tumor function of cytotoxic T cells. Int Immunol. 2007; 19:1213–21. https://doi.org/10.1093/intimm/dxm093.
21. Liu Z, Meng Q, Bartek J Jr, Poiret T, Persson O, Rane L, Rangelova E, Illies C, Peredo IH, Luo X, Rao MV, Robertson RA, Dodoo E, Maeurer M. Tumor-infiltrating lymphocytes (TILs) from patients with glioma. OncoImmunology. 2016; 6:e1252894. https://doi.org/10.1080/2162402X.2016.1252894.
22. Meng Q, Liu Z, Rangelova E, Poiret T, Ambati A, Rane L, Xie S, Verbeke C, Dodoo E, Del Chiaro M, Löhr M, Segersvärd R, Maeurer MJ. Expansion of tumor-reactive T cells from patients with pancreatic cancer. J Immunother. 2016; 39:81–89. https://doi.org/10.1097/CJI.0000000000000111.
23. Lutz E, Yeo CJ, Lillemoe KD, Biedrzycki B, Kobrin B, Herman J, Sugar E, Piantadosi S, Cameron JL, Solt S, Onners B, Tartakovsky I, Choi M, et al. A lethally irradiated allogeneic granulocyte-macrophage colony stimulating factor-secreting tumor vaccine for pancreatic adenocarcinoma. A Phase II trial of safety, efficacy, and immune activation. Ann Surg. 2011; 253:328–35. https://doi.org/10.1097/SLA.0b013e3181fd271c.
24. Croce M, Rigo V, Ferrini S. IL-21: a pleiotropic cytokine with potential applications in oncology. J Immunol Res. 2015; 2015:696578. https://doi.org/10.1155/2015/696578.
25. Li Y, Bleakley M, Yee C. IL-21 influences the frequency, phenotype, and affinity of the antigen-specific CD8 T cell response. J Immunol. 2005; 175:2261–69. https://doi.org/10.4049/jimmunol.175.4.2261.
26. Yue FY, Lo C, Sakhdari A, Lee EY, Kovacs CM, Benko E, Liu J, Song H, Jones RB, Sheth P, Chege D, Kaul R, Ostrowski MA. HIV-specific IL-21 producing CD4+ T cells are induced in acute and chronic progressive HIV infection and are associated with relative viral control. J Immunol. 2010; 185:498–506. https://doi.org/10.4049/jimmunol.0903915.
27. Kotlarz D, Ziętara N, Milner JD, Klein C. Human IL-21 and IL-21R deficiencies: two novel entities of primary immunodeficiency. Curr Opin Pediatr. 2014; 26:704–12. https://doi.org/10.1097/MOP.0000000000000160.
28. Kannappan V, Butcher K, Trela M, Nicholl I, Wang W, Attridge K. Interleukin 21 inhibits cancer-mediated FOXP3 induction in naïve human CD4 T cells. Cancer Immunol Immunother. 2017; 66:637–45. https://doi.org/10.1007/s00262-017-1970-6.
29. Wang B, Kuroiwa JM, He LZ, Charalambous A, Keler T, Steinman RM. The human cancer antigen mesothelin is more efficiently presented to the mouse immune system when targeted to the DEC-205/CD205 receptor on dendritic cells. Ann N Y Acad Sci. 2009; 1174:6–17. https://doi.org/10.1111/j.1749-6632.2009.04933.x.
30. Yokokawa J, Palena C, Arlen P, Hassan R, Ho M, Pastan I, Schlom J, Tsang KY. Identification of novel human CTL epitopes and their agonist epitopes of mesothelin. Clin Cancer Res. 2005; 11:6342–51. https://doi.org/10.1158/1078-0432.CCR-05-0596.
31. Jiang Y, Li Y, Zhu B. T-cell exhaustion in the tumor microenvironment. Cell Death Dis. 2015; 6:e1792. https://doi.org/10.1038/cddis.2015.162.
32. Kastirr I, Maglie S, Paroni M, Alfen JS, Nizzoli G, Sugliano E, Crosti MC, Moro M, Steckel B, Steinfelder S, Stölzel K, Romagnani C, Botti F, et al. IL-21 is a central memory T cell-associated cytokine that inhibits the generation of pathogenic Th1/17 effector cells. J Immunol. 2014; 193:3322–31. https://doi.org/10.4049/jimmunol.1400775.
33. Brandt K, Bulfone-Paus S, Foster DC, Rückert R. Interleukin-21 inhibits dendritic cell activation and maturation. Blood. 2003; 102:4090–98. https://doi.org/10.1182/blood-2003-03-0669.
34. O’Hara M, Stashwick C, Haas AR, Tanyi JL. Mesothelin as a target for chimeric antigen receptor-modified T cells as anticancer therapy. Immunotherapy. 2016; 8:449–60. https://doi.org/10.2217/imt.16.4.

