INTRODUCTION
Lung cancer is among the most common cancers in both man and woman, and is the leading cause of cancer-related death worldwide [1]. Non-small cell lung cancer (NSCLC), with two main histological types of adenocarcinoma and squamous-cell (epidermoid) carcinoma, accounts for about 80% of primary lung cancer, and a majority present with advanced stage (III/IV) when they were first diagnosed. Only 10–15% newly diagnosed NSCLC patients were adopted with a potentially curative resection [2]. Despite the introduction of targeted therapy on specific mutant molecules such as EGFR, platinum agents combined with another cytotoxic compound are still the first-line chemotherapy for advanced NSCLC. The action mode of platinating agents is known to bind the DNA with intra- or interstrand crosslinks and the Pt-DNA adduct lead to DNA lesions, activation of multiple pathways and result in cell apoptosis ultimately [3]. Despite efforts to improve therapeutic efficacy, the platinum-based chemotherapy brings modest benefits but also adverse effects, with the five-year survival rates less than 15%. And an increasing evidence showed that the limited efficacy for advanced NSCLC is due to drug resistance and toxicological side effects such as thrombocytopenia, nausea/vomiting, ototoxicity, nephrotoxicity and peripheral neurotoxicity, etc [3].
Mechanistically, therapeutic efficacy and response to platinum-based chemotherapy could be linked to these biological pathways, altered cellular accumulation, cytosolic inactivation of platinum drugs, inactivation of DNA repair pathway, altered apoptosis or increased tolerance to DNA damage [3]. The aberrant regulation and dysfunction of candidate genes in these pathway, which are largely ascribed to their functional polymorphisms, may influence interindividual differences in clinical outcomes. Thus there is a growing need toward tailoring chemotherapy to identify these functional genetic polymorphisms as predictive pharmacogenetic markers for better efficacy and minimal toxicity. To this end, the influence of sequence variants in pharmacodynamics pathways, such as nucleotide excision repair pathway, on clinical outcome has been extensively investigated [4]. In contrast, the biological function and clinical relevance of genetic polymorphisms in pharmacokinetics pathway for platinum metabolism and disposition are largely elusive.
Human SLC31A1 (solute carrier family 31 member 1) gene, also known as CTR1 (copper transporter 1), encodes a high-affinity copper transporter in cell membrane that functions as a homotrimer to effect the uptake of dietary copper. Recently, an interesting possibility has clearly emerged that the copper transporter SLC31A1 also acts as the major plasma-membrane transporter for platinum drug intake, including cisplatin, oxaliplatin and carboplatin [5]. Deletion of Slc31a1 in yeast and murine cells results in reduced cisplatin accumulation and increased resistance [6]. On the contrary, transfection of small cell lung cancer cell lines with SLC31A1 gene correlates with enhanced uptake of carboplatin and oxaliplatin [7]. Consistently, Slc31a1 knockout in murine model has also been shown to completely eliminate cisplatin tumor response in vivo [8]. Furthermore, in clinical setting, NSCLC patients with undetectable SLC31A1 expression in their tumors had reduced platinum concentration and tumor response, and lower platinum concentration in clinical specimens correlates directly with reduced tumor response and shorter survival time [9, 10]. Recently, in two pilot studies with relatively small sample size (two to three hundreds) in Chinese patients with NSCLC who received Pt-based therapy, Xu et al. documented that SLC31A1 polymorphisms are associated with Pt-resistance or toxicity and poor clinical outcomes [11, 12]. But these results have not been validated in larger cohort of patients and functional implication of these associated variants are not clear.
In the present study, we assessed the association of tagging and potentially functional SNPs of SLC31A1 gene with toxicological phenotypes, objective response and survivals of 1004 Chinese NSCLC patients receiving platinum-based treatment. We also functionally characterized a common variant at 3ʹ untranslated region (3ʹUTR) that modulates the microRNA-3ʹUTR interaction and decreases gene expression, proposing a possible underlying mechanism for the genetic association of SLC31A1 with clinical outcomes.
RESULTS
Patient characteristics and clinical outcomes
A total of 1004 eligible NSCLC patients of Chinese population were recruited in the study to investigate the genetic association between SCL31A1 polymorphisms and clinical outcomes of platinum-based chemotherapy. The main characteristics and clinical outcomes of patients are summarized in Table 1. Ever smokers account for 57.5% of the patients. Adenocarcinoma was the most common histological type (62.9%). Severe gastrointestinal toxicity (nausea/vomiting) was observed in 8.3% of the evaluated patients (n = 964). Severe hematological toxicity was observed in 23.9% of the evaluated patients (n = 969), among which 29 (3.1%), 149 (15.2%), 115 (12.3%) and 34 (3.6%) patients suffered from grade 3 or 4 anemia, leukopenia, neutropenia, thrombocytopenia, respectively. Grade 3 or 4 overall toxicity was observed in 29.9% of the evaluated patients (n = 952). For survival analysis, by the time of final data collection (July 2012), the median follow-up time was 46.5 month, and death had occurred in 74.9% of enrolled patients. The median progression-free survival (PFS) was 9.1 months, and the median overall survival (OS) was 19.3 months. The rates of clinical phenotypes such as toxicities in our study were quite comparable to those previously reported in large randomized clinical trials [13].
Table 1: Patient characteristics and clinical outcomes
Characteristic |
Total Number |
Number |
Percent |
|---|---|---|---|
All patients |
1004 |
||
Sex |
1004 |
||
Male |
706 |
70.3 |
|
Female |
298 |
29.7 |
|
Age |
1004 |
||
≤58 |
518 |
51.6 |
|
>58 |
486 |
48.4 |
|
Smoking Status |
1000 |
||
Ever Smoker |
575 |
57.5 |
|
Nonsmoker a |
425 |
42.5 |
|
ECOG performance status b |
990 |
||
0–1 |
904 |
91.3 |
|
2 |
86 |
8.7 |
|
TNM stage |
999 |
||
IIIA |
81 |
8.1 |
|
IIIB |
293 |
29.3 |
|
IV |
625 |
62.6 |
|
Histological type |
1004 |
||
Adenocarcinoma (AC) |
632 |
62.9 |
|
Squamous cell carcinoma (SCC) |
221 |
22.1 |
|
Adenosquamocarcinoma |
20 |
2.0 |
|
Others c |
131 |
13.0 |
|
Chemotherapy regimens |
1004 |
||
Platinum (cisplatin)-navelbine |
316 |
31.5 |
|
Platinum (cisplatin)-gemcitabine |
239 |
23.8 |
|
Platinum (carboplatin)-paclitaxel |
313 |
31.2 |
|
Platinum-docetaxel |
87 |
8.7 |
|
Other platinum combinations |
49 |
4.9 |
|
Objective response |
976 |
||
Complete response (CR) |
1 |
0.1 |
|
Partial response (PR) |
176 |
18.0 |
|
Stable disease (SD) |
611 |
62.6 |
|
Progressive disease (PD) |
188 |
19.3 |
|
Toxicity outcome |
|||
Grade 3 or 4 gastrointestinal toxicity |
|||
Nausea/vomiting |
964 |
80 |
8.3 |
Grade 3 or 4 hematologic toxicity |
969 |
232 |
23.9 |
Anemia |
944 |
29 |
3.1 |
Leukopenia |
980 |
149 |
15.2 |
Neutropenia |
935 |
115 |
12.3 |
Thrombocytopenia |
950 |
34 |
3.6 |
Grade 3 or 4 overall toxicity |
952 |
285 |
29.9 |
Median time to outcomes (months) |
972 |
||
Progression-free survival (PFS) |
9.1 |
||
Overall survival (OS) |
19.3 |
a Nonsmokers were defined as those who had smoked <1 cigarette per day and for <1 year in their lifetime.
b ECOG PS, Eastern Cooperative Oncology Group performance status.
c Other carcinomas included mixed cell or undifferentiated carcinoma.
In the NSCLC patient cohort, we genotyped eight tagging and potentially functional SNPs of SLC31A1 gene, three at 5ʹflanking region (rs4979223, rs4978536 and rs2233914), three at the intron (rs10817464, rs10981699 and rs10817465), and two at 3ʹUTR (rs10513202 and rs10759637), all of which were in Hardy-Weinberg equilibrium (HWE) (Supplementary Table 1), and six of which are common variants (minor allele frequency, MAF >0.05) in this study population. The linkage disequilibrium analysis showed that the eight candidate SNPs could been partitioned into one haplotype block. For example, rs10759637 at 3ʹUTR are strictly associated with rs4979223 (r2 = 0.98) and rs2233914 (r2 = 0.56) at 5ʹflanking region (Figure 1A).
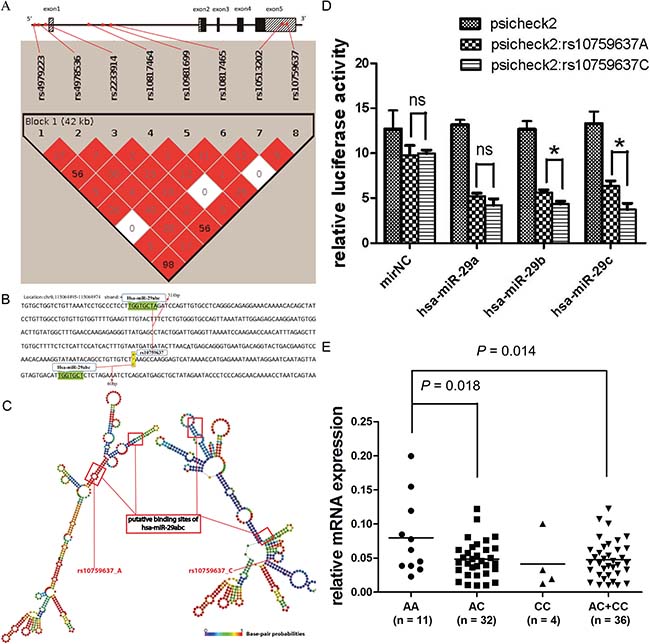
Figure 1: rs10759637 in SLC31A1 3ʹUTR decreases microRNA-medicated gene expression. (A) Schematic structure of SLC31A1 gene and linkage disequilibrium of its candidate SNPs (color intensity is proportional to D′ and percent numbers represent r2). (B) Local sequence of SLC31A1 3ʹUTR annotated with the close locations of rs10759637 and two putative binding sites for hsa-miR-29 family. (C) Comparison of secondary structures, built with RNAfold program, of the local SLC31A1 3ʹUTRs harboring hsa-miR-29 binding sites and the wild or the variant alleles of rs10759637. (D) Luciferase reporter assays in 16HBE cells to analyze the regulatory role hsa-miR-29 on SLC31A1 3ʹUTR that could be modulated by rs10759637. Co-transfection with hsa-miR-29 member (a, b, or c) and SLC31A1 3ʹUTR construct consistently resulted in reduced luciferase activity. In the cases of hsa-miR-29 b or c, rs10759637 allelic state significantly affected the cis-regulation toward reporter expression. (E) Real-time quantitative RT-PCR analysis of SLC31A1 mRNA expression in lung tumorous tissues from an independent cohort of patients, which was significantly correlated with rs10759637 genotypes.
Association of SLC31A1 gene polymorphisms with toxicity and objective response
In the patient cohort that had been evaluated for the seven toxicological phenotypes after platinum-based chemotherapy, we compared the genotypic distributions for the eight candidate SNPs of SLC31A1 between groups with respective mild or severe toxicological outcomes (Supplementary Table 2). Interestingly, we observed that rs4979223 (P = 0.007), rs4978536 (P = 0.063, with marginal significance), rs2233914 (P = 0.039), rs10817464 (P = 0.006) and rs10759637 (P = 0.006) showed significantly divergent genotypic distribution between groups with mild or severe thrombocytopenia, some of which remained statistically significant after multiple test correction. Based on these five candidate SNPs, we then assessed the association between SLC31A1 polymorphism and toxicological outcomes with multivariate unconditional logistic regression analysis (Table 2). As to the tightly linked rs4979223 and rs10759637, we found their heterozygous genotype (A/C), as compared to the grouped homozygous genotypes (A/A+C/C), were significantly associated with severe thrombocytopenia, with ORs (95% CI) being 2.60 (1.20−5.64) and 2.69 (1.24−5.83), respectively. The variant containing genotype group (A/G+G/G) of rs4978536, as compared to the wild A/A, was also associated with severe thrombocytopenia (OR 2.59; 95% CI 1.23−5.47). In alike manner, the variant genotypes of rs10817464 were associated with increased risks of severe thrombocytopenia (OR 3.09; 95% CI 1.20−7.93), leucopenia (OR 2.16; 95% CI 1.19−3.92), hematologic toxicity (OR 1.98; 95% CI 1.19−3.29) and overall toxicity (OR 1.98; 95% CI 1.21−3.22). On the contrary. The variant A/A genotype of rs2233914, as referred to the wild allele containing genotypes (G/G+G/A), was associated with mild overall toxicity (OR 0.59; 95% CI 0.36−0.95).
Table 2: Association between SLC31A1 SNPs and toxicity outcomes
Reference SNP |
Genotype |
Toxicity |
Toxicity grade (G3-4/G0-2) |
P valuea |
OR (95% CI)b |
P valueb |
|---|---|---|---|---|---|---|
rs4979223 |
A/A |
Thrombocytopenia |
3/282 |
0.007 c |
1.00 (ref) |
|
A/C |
24/417 |
4.90 (1.43−16.82) |
0.012 |
|||
C/C |
7/214 |
3.00 (0.75−12.05) |
0.121 |
|||
A/C vs A/A+C/C |
10/496 |
0.004 c |
2.60 (1.20−5.64) |
0.015 |
||
rs4978536 |
A/A |
Thrombocytopenia |
20/701 |
0.063 |
1.00 (ref) |
|
A/G |
13/195 |
2.71 (1.26−5.81) |
0.010 |
|||
G/G |
1/20 |
1.63 (0.19−13.89) |
0.654 |
|||
A/G+G/G vs A/A |
14/215 |
0.018 |
2.59 (1.23−5.47) |
0.012 |
||
rs2233914 |
G/G |
Overall toxicity |
135/299 |
0.150 |
1.00 (ref) |
|
G/A |
124/277 |
0.88 (0.64−1.20) |
0.409 |
|||
A/A |
26/91 |
0.55 (0.33−0.91) |
0.020 |
|||
A/A vs G/G+G/A |
259/576 |
0.052 |
0.59 (0.36−0.95) |
0.029 |
||
rs10817464 |
A/A |
Leucopenia |
130/767 |
0.069 |
1.00 (ref) |
|
A/G |
19/62 |
2.21 (1.22−4.01) |
0.009 |
|||
G/G |
0/2 |
NA |
NA |
|||
A/G+G/G vs A/A |
19/64 |
0.041 |
2.16 (1.19−3.92) |
0.011 |
||
A/A |
Thrombocytopenia |
27/842 |
0.006 c |
1.00 (ref) |
||
A/G |
6/73 |
2.75 (1.02−7.42) |
0.045 |
|||
G/G |
1/1 |
13.70 (0.69−220.29) |
0.085 |
|||
A/G+G/G vs A/A |
7/74 |
0.024 |
3.09 (1.20−7.93) |
0.019 |
||
A/A |
Hematologic toxicity |
204/683 |
0.088 |
1.00 (ref) |
||
A/G |
27/53 |
1.92 (1.15−3.21) |
0.012 |
|||
G/G |
1/1 |
6.34 (0.36−110.34) |
0.205 |
|||
A/G+G/G vs A/A |
28/54 |
0.024 |
1.98 (1.19−3.29) |
0.008 |
||
A/A |
Overall toxicity |
251/619 |
0.050 |
1.00 (ref) |
||
A/G |
33/47 |
1.95 (1.19−3.19) |
0.008 |
|||
G/G |
1/1 |
3.62 (0.20−66.75) |
0.387 |
|||
A/G+G/G vs A/A |
34/48 |
0.017 |
1.98 (1.21−3.22) |
0.006 |
||
rs10759637 |
A/A |
Thrombocytopenia |
3/286 |
0.006 c |
1.00 (ref) |
|
A/C |
24/414 |
5.10 (1.49−17.52) |
0.010 |
|||
C/C |
7/216 |
3.05 (0.76−12.24) |
0.116 |
|||
A/C vs A/A+C/C |
10/502 |
0.004 c |
2.69 (1.24−5.83) |
0.012 |
a P values of Pearson χ2 tests for overall distributions for SNPs genotypes between severe and mild toxicological phenotypes.
b Odds ratios (OR) and their 95% confidence intervals (CIs) and P values were calculated with unconditional logistic regression analysis, with adjustment of gender, age, smoking status, ECOG performance status, TNM status, histological types, and treatment regimen.
c Statistical significance remained after multiple tests adjustment taking into account linkage disequilibrium between polymorphisms.
Because these five candidate SNPs of SLC31A1 could be partitioned into one haplotype block (Figure 1), we also estimated their haplotype and diplotype frequencies in the cohort, and analyzed their association with toxicological outcomes (Table 3). We predicted only four common haplotypes for the five SNPs in the 1004 individuals, which is consistent with their strong linkage disequilibrium. Agreeing with the genotype-based association results as above, the Hap4_CGGGC, which is composed of rs4979223-variant-C, rs4978536-variant-G, rs2233914-wild-G, rs10817464-variant-G and rs10759637-variant-C alleles, was significantly associated with severe toxicological outcomes such as leucopenia (OR 1.88; 95% CI 1.04−3.40), thrombocytopenia (OR 2.81; 95% CI 1.19−6.60), hematologic toxicity (OR 1.88; 95% CI 1.14−3.08) and overall toxicity (OR 1.89; 95% CI 1.17−3.06). As compared to non-Hap4 carrier diplotype, the Hap4 carrier diplotype was also consistently associated with these severe toxicological phenotypes, with ORs (95% CI) being 2.16 (1.19−3.92), 3.09 (1.20−7.93), 1.98 (1.19−3.29) and 1.98 (1.21−3.22), respectively.
Table 3: Association between SLC31A1 haplotype and diplotype and toxicity outcomes
Haplotype or Diplotype a |
Leucopenia |
Thrombocytopenia |
Hematologic toxicity |
Overall toxicity |
||||
|---|---|---|---|---|---|---|---|---|
G3-4/0-2 |
OR (95% CI)b |
G3-4/0-2 |
OR (95% CI)b |
G3-4/0-2 |
OR (95% CI)b |
G3-4/0-2 |
OR (95% CI)b |
|
Haplotype frequency |
||||||||
Hap1_AAGAA |
164/880 |
1.00 |
30/983 |
1.00 |
255/780 |
1.00 |
311/702 |
1.00 |
Hap2_CAAAC |
92/560 |
0.75 (0.57−1.00) |
23/608 |
0.97 (0.56−1.68) |
142/500 |
0.79 (0.62−0.99) |
176/459 |
0.77 (0.62−0.97) |
Hap3_CGGAC |
23/149 |
0.75 (0.46−1.23) |
7/159 |
1.37 (0.60−3.12) |
37/133 |
0.83 (0.56−1.23) |
47/118 |
0.88 (0.61−1.28) |
Hap4_CGGGC |
19/66 |
1.88 (1.04−3.40) |
8/75 |
2.81 (1.19−6.60) |
29/55 |
1.88 (1.14−3.08) |
35/49 |
1.89 (1.17−3.06) |
Hap5_others |
0/7 |
NA |
0/7 |
NA |
1/6 |
0.55 (0.06−4.91) |
1/6 |
0.39 (0.04−3.51) |
Diplotype frequency |
||||||||
Non-Hap4 carriers |
130/767 |
1.00 |
27/842 |
1.00 |
204/683 |
1.00 |
251/619 |
1.00 |
Hap4 carriers |
19/64 |
2.16 (1.19−3.92) |
7/74 |
3.09 (1.20−7.93) |
28/54 |
1.98 (1.19−3.29) |
34/48 |
1.98 (1.21−3.22) |
aHaplotypes were predicted with PHASE basing on rs4979223 (A/C), rs4978536 (A/G), rs2233914 (G/A), rs10817464 (A/G) and rs10759637(A/C) that were associated with toxicity outcomes as shown in Table 2.
bOdds ratios (OR) and their 95% confidence intervals (CIs) were calculated with unconditional logistic regression analysis, with adjustment of gender, age, smoking status, ECOG performance status, TNM status, histological types, and treatment regimen.
In the further stratification analysis by demographic and clinical characteristics (Table 4), rs10759637 was associated in under-dominant model with thrombocytopenia in males (OR 5.08; 95% CI 1.62−15.94) and smoking patients (OR 4.06; 95% CI 1.40−15.11). Very similar association with thrombocytopenia was also observed for rs4979223. In alike manner, rs4978536 was associated in dominant model with thrombocytopenia in males (OR 3.16; 95% CI 1.18−8.47) and patients with adenocarcinoma (OR 3.05; 95% CI 1.26−7.37). Interestingly, in the subgroup patients with age ≤58 year or with TNM stage IV, rs10817464 consistently manifested significant association, in dominant model, with leucopenia, hematological toxicity, and overall toxicity, with ORs (95% CI) being 2.96 (1.26−6.96) and 2.88 (1.35−6.13), 2.27 (1.10−4.66) and 2.50 (1.31−4.76), 2.50 (1.25−4.96) and 2.56 (1.36−4.83), respectively. On the other hand, rs2233914 was associated with reduced overall toxicity in males (OR 0.63; 95% CI 0.44−0.90), patients treated with platinum-navelbine (OR 0.45; 95% CI 0.27−0.75) in dominant model, and in patients with adenocarcinoma (OR 0.41; 95% CI 0.21−0.81) in additive model. These data suggest that common variants of SLC31A1 gene are associated with hematological toxicological outcomes of platinum-based chemotherapy for NSCLC patients.
Table 4: Stratification analysis of association between SLC31A1 SNPs and toxicity outcomes
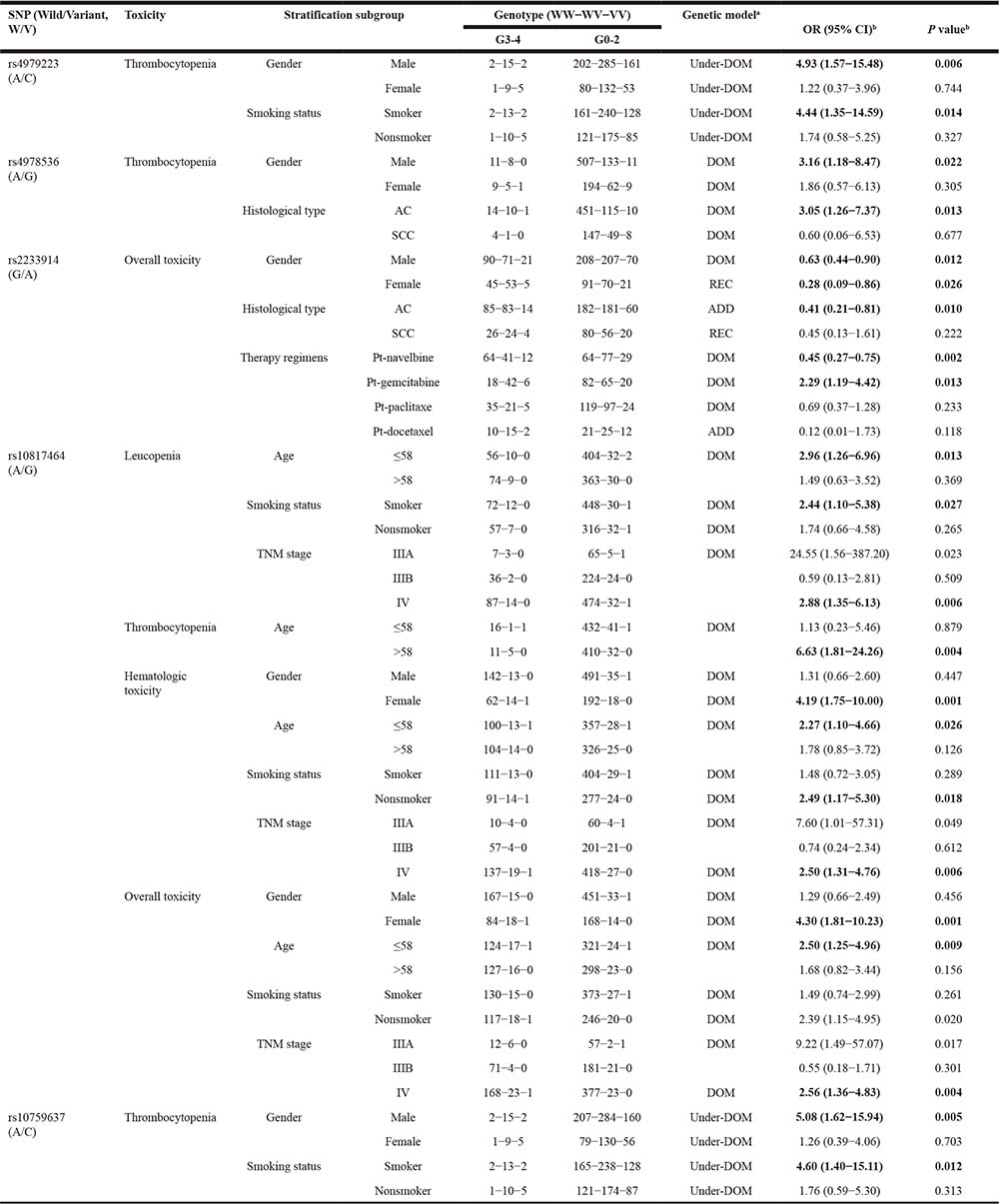
aFor each SNP, three different genetic models (dominant, recessive and additive) were analyzed, and the model with lowest P values was considered the best-fitting model. Under-dominant model was also analyzed for rs4979223 and rs10759637.
bOdds ratios (OR) and their 95% confidence intervals (CIs) and P values were calculated with unconditional logistic regression analysis, with adjustment of gender, age, smoking status, ECOG performance status, TNM status, histological types, and treatment regimen.
We also analyzed the association between SLC31A1 polymorphism and chemotherapy objective response. None of the eight candidate SNPs of SLC31A1 displayed statistically significant difference in genotypic distribution between the complete or partial response group and the stable or progressive disease group (Supplementary Table 3). These results do not support genetic correlation between SLC31A1 and objective response of platinum-based chemotherapy in the patient cohort.
Association of SLC31A1 gene polymorphisms with survival
We measured genetic association of SLC31A1 polymorphism with overall survival (OS) and progression-free survival (PFS) in the patient cohort with platinum-based chemotherapy by using log-rank test and Cox proportional hazards regression model (Table 5). As to the OS dataset, the log-rank test showed that rs4979223 (P = 0.006), rs2233914 (P = 0.016) and rs10759637 (P = 0.010) were significantly associated with survival. The median OS time of patients with heterozygous A/C of rs10759637 was significantly shorter than those patients with homozygous A/A or C/C (17.7 vs 20.2, P = 0.004). Cox proportional hazards regression analysis further showed that rs10759637 A/C was associated with increased risk of disease progression as compared with the homozygous genotype group (HR 1.24; 95% CI 1.07−1.44). In alike manner, rs4979223 heterozygous genotype was also a risk factor for OS with HR of 1.25 (1.08−1.45) and log-rank P value of 0.002 when assuming under-dominant model. On the contrary, rs2233914 variant A/A homozygote, in recessive model, was a beneficial factor for OS with HR of 0.73 (0.58−0.92), the median OS time of patients A/A genotype was significantly longer than those with G/G or G/A genotypes (22.3 vs 18.8, log-rank P = 0.011). As to the PFS dataset, we did not observe any association between progression-free survival and the eight candidate SNPs of SLC31A1.
Table 5: Association between SLC31A1 SNPs and survival
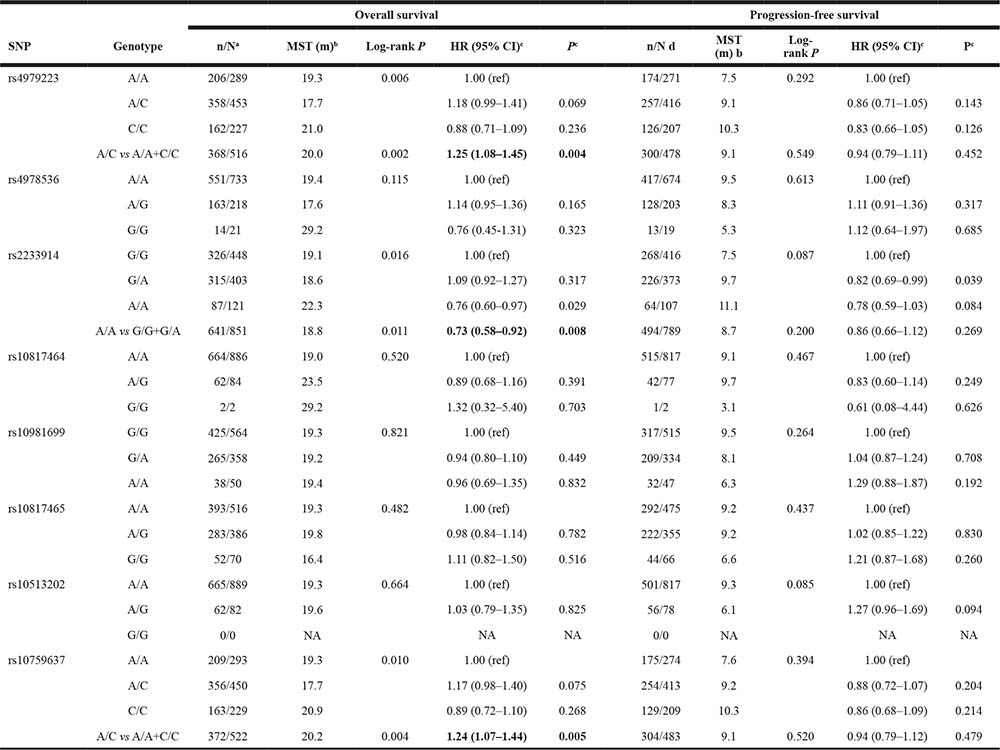
a Numbers indicate the death event for NSCLC patients during the following-up time among all individuals in the same genotype group.
b MST: median survival time.
c Hazard ratios (HR) and their 95% confidence intervals (CIs) and P values were calculated with by multivariate Cox proportional hazards regression with adjustment for covariates.
d Numbers indicate patients who suffered of disease progression (including death) during the following-up time among all individuals in the same genotype group.
Notably, in further stratification analysis of survival data, rs4979223, rs2233914 and rs10759637 consistently manifested significant association in concordant stratification spectrum (Table 6 and Figure 2A–2F). Influence of rs10759637 heterozygous A/C genotype on OS were pronounced in male patients (log-rank P = 0.019; HR 1.23; 95% CI 1.04−1.47), patients older than 58 (log-rank P = 0.004; HR 1.38; 95% CI 1.10−1.73), ever smoker (log-rank P = 0.013; HR 1.28; 95% CI 1.05−1.55), patients with ECOG PS 0–1 (log-rank P = 0.005; HR 1.24; 95% CI 1.06−1.45), patients with squamous cell cancer (log-rank P = 0.010; HR 1.57; 95% CI 1.12−2.20), and patients treated with platinum-docetaxel (log-rank P = 0.013; HR 1.95; 95% CI 1.03−3.67), respectively. A very similar stratification spectrum with association signal was also observed for rs4979223. And also in the six subgroups of stratification, rs2233914 variant A/A homozygote, in recessive model, was beneficial modifier of OS with HRs (95% CI) being 0.75 (0.58−0.97), 0.74 (0.53−1.05), 0.73 (0.55−0.98), 0.76 (0.60−0.96), 0.38 (0.22−0.66) and 0.38 (0.17−0.87), respectively. These results clearly demonstrate that non-coding variants of SLC31A1 are associated with overall survival of NSCLC patients with platinum-based chemotherapy.
Table 6: Stratification analysis of association between SLC31A1 SNPs and overall survival
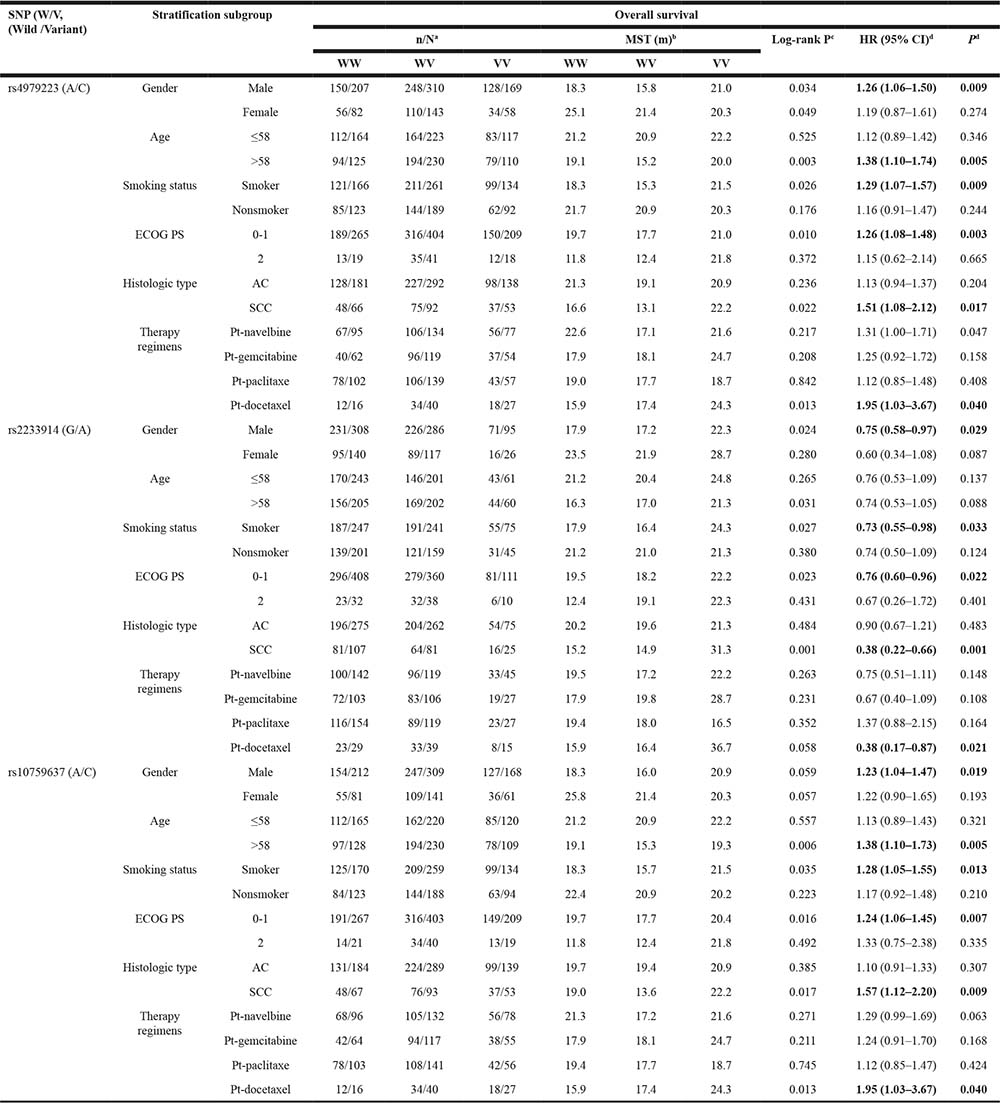
aNumbers indicate the death event for NSCLC patients during the following-up time among all individuals in the same genotype group.
bMST: median survival time.
cLog-rank tests for association between survival and overall genotypes.
dHazard ratios (HR) and their 95% confidence intervals (CIs) and P values were calculated with by multivariate Cox proportional hazards regression with adjustment for covariates. Under-dominant, recessive and under-dominant models were used to estimate the HRs for the three SNPs, respectively. For rs4979223, A/C heterozygote was compared to A/A+C/C homozygotes as reference; For rs2233914, A/A homozygote was compared to G/G+G/A group as reference. For rs10759637, A/C heterozygote was compared to A/A+C/C homozygotes as reference.
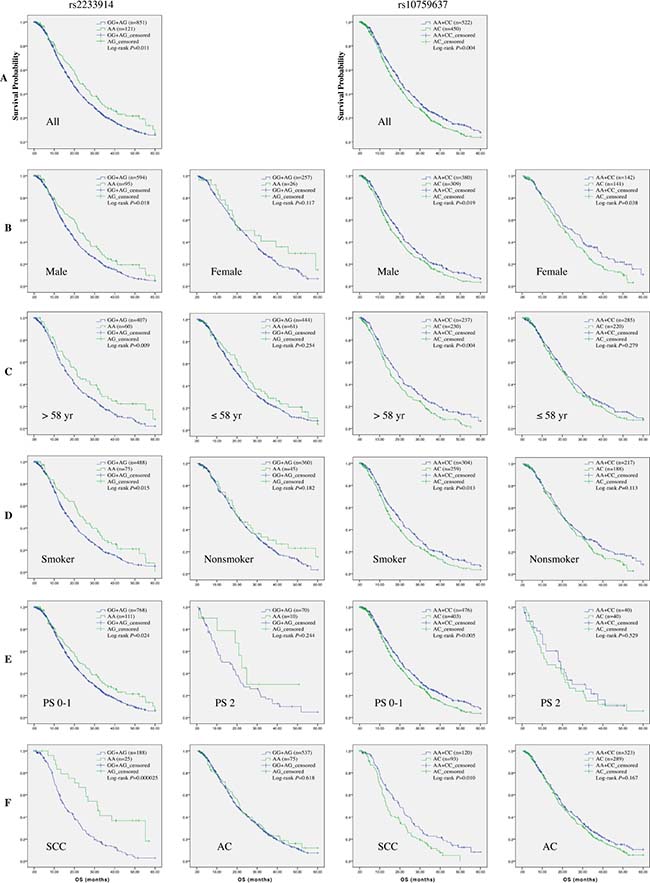
Figure 2: Kaplan-Meier curve of estimated overall survival for the NSCLC cohort according to SLC31A1 polymorphisms. In the entire patients (A) and subgroups stratified respectively with gender (B), age (C), smoking status (D), ECOG performance status (E), and histological type (F), the curves were plotted with SPSS software according to genotypes of two SNPs. For rs2233914, the A/A homozygote was compared to the G/G+G/A group as reference. For rs10759637, the A/C heterozygote was compared to the A/A+C/C homozygotes group as reference.
rs10759637 at SLC31A1 3ʹUTR affects hsa-miR-29 mediated expression regulation
In this pharmacogenetic study, clinical outcomes such as severe thrombocytopenia hematological toxicity and overall survival of NSCLC patients with platinum-based chemotherapy were associated with linked non-coding variants of SLC31A1 gene that codes the transporter for platinum agent intake. We hypothesized that the underlying mechanism for the genetic association might be linked to functional interplay between polymorphic cis-elements and putative trans-acting factors such as transcription factor and microRNA. In order to identify putative microRNA targeting sequences in SLC31A1 3ʹUTR and assess potential effect of rs10759637 on microRNA-mediated regulation, we used the miRdSNP database and TargetScanHuman 6.2 program to screen potential microRNA binding sites at SLC31A1 3ʹUTR. Interestingly, as shown in Figure 1B, we predicted two putative binding sites, 314 bp upstream and 60 bp downstream to rs10759637, respectively, for the hsa-miR-29 family that was reported to be expressed in lung tissue and implicated in lung development and carcinogenesis [14, 15]. Further prediction of local secondary structure of SLC31A1 mRNA 3ʹUTR with RNAfold program revealed that rs10759637 could alter the local mRNA secondary structure in the binding sites of hsa-miR-29abc by changing the minimum free energy from -133.40 kcal/mmol at the wild A allele state to -131.00 kcal/mmol at the variant C allele state (Figure 1C), suggesting that rs10759637 could modulate the regulatory function of hsa-miR-29 on SLC31A1 3ʹUTR. As to rs4979223 at 5ʹflanking region that was strictly associated with rs10759637, we did not observe putative binding sites containing it or other nearby linked SNPs in the 1000genome dataset for any transcription factor (data not shown). These results suggest that rs10759637 at SLC31A1 3ʹUTR might modulate the miRNA-mRNA interaction and affect miRNA-mediated expression regulation.
Using luciferase reporter gene assays in human bronchial epithelial cell line 16HBE, which, like lung epithelial cell, originates from primitive pluripotent pulmonary epithelial cells, we further investigated the functional modulation of rs10759637 on hsa-miR-29−SLC31A1 3ʹUTR interaction. In accordance with results of structural prediction as above, co-transfection with hsa-miR-29 member (a, b, or c) and SLC31A1 3ʹUTR construct (of rs10759637 A or C allele), as compared to the negative control microRNA mimic, consistently resulted in remarkably decreased luciferase activity, demonstrating that the hsa-miR-29 family is indeed a negative regulator for SLC31A1 expression by targeting its 3ʹUTR. Interestingly, in the setting of cotransfection with hsa-miR-29 b or c, rs10759637 A/C at 3ʹUTR significantly affected the cis-regulation toward reporter expression, the variant C allele correlated with much lowered luciferase activity as compared to the wild A allele (Figure 1D, P < 0.05). Finally, in lung tumorous tissues from an independent cohort of patients, we analyzed the correlation between rs10759637 genotypes and transcriptional expression level for SLC31A1 using real-time quantitative RT-PCR. Consistent with the results of structure prediction and reporter assays, we found that, as compared with the wild A/A genotype, the heterozygous A/C (P = 0.018) and the A/C+C/C group (P = 0.014) were significantly correlated with lowered expression of SLC31A1 transcript in lung cancer tissues, suggesting that the modulation of rs10759637 on hsa-miR-29−SLC31A1 3ʹUTR interaction observed in cell line has relevance in clinical samples.
Taken together, these functional studies revealed that rs10759637 in SLC31A1 3ʹUTR could modulate the microRNA-3ʹUTR interaction and thereby decrease gene expression, thus proposing a possible underlying mechanism for the genetic association between SLC31A1 polymorphism and clinical outcomes of platinum-based chemotherapy in NSCLC patients.
DISCUSSION
In this multi-institutions based pharmacogenetic study of NSCLC patients with platinum-based chemotherapy, we found linked non-coding variants of SLC31A1 gene, which encodes the major platinum uptake transporter, are associated with severe hematological toxicities such as thrombocytopenia and shorter overall survival. Further mechanistic analysis revealed that one SNP in 3ʹUTR correlated with downregulated gene expression through modulating the microRNA−3ʹUTR interaction. The clinically relevant and biologically functional SLC31A1 polymorphism provides potential biomarker for outcome prediction of platinum-based chemotherapy and lung cancer management.
As a well-defined transporter for platinum import, the expression and function of SLC31A1 are involved in platinum disposition and clinical response to platinum-based chemotherapy. Chen et al reported that in a cohort of stage Ⅲ NSCLC patients in Chinese population receiving first-line platinum-based doublet chemotherapy, SLC31A1 protein overexpression is associated with better therapeutic response and better survival [16]. In a cohort of American patients who underwent neoadjuvant platinum-based chemotherapy, Kim et al also observed correlation between SLC31A1 expression in tumor size and tissue platinum concentration, undetectable protein expression and reduced drug load are associated with reduced tumor response and shorter survival time [9, 10]. Consistently, low levels of SLC31A1 mRNA are also associated with poor clinical response to platinum-based therapy in ovarian cancer patients [17]. These clinical studies demonstrate that the expression status in situ of SLC31A1 is a predictor for platinum-based therapy response and a significant prognostic factor for advanced NSCLC patients. However, the resectable cases only account for 10-15% of diagnosed NSCLC patients, and routine measure of SLC31A1 mRNA or protein expression in lung cancer tissues is not convenient. Thus, it is of very limited clinical utility for SLC31A1 expression as a putative biomarker of response and outcome for platinum-based chemotherapy in NSCLC patients. We here present evidences that rs10759637 in SLC31A1 3ʹUTR functionally decreased microRNA-mediated gene expression, and this common non-coding variants as well as linked SNPs were associated with severe hematological toxicities such as thrombocytopenia and shorter overall survival in the Chinese cohort of NSCLC patients with platinum-based chemotherapy. Notably, the genetic associations with survival were consistently pronounced in male patients, patients ever-smoking, older than 58, or with squamous cell carcinoma. Therefore, relevant variants of SLC31A1 will be pertinent pharmacogenetic biomarkers, which is much more actionable than gene expression, for platinum-based therapy in NSCLC patients especially in these subgroups.
The association of rs10759637 in SLC31A1 3ʹUTR with clinical outcomes in NSCLC patients with platinum-based therapy is biologically plausible. In a screening in silico of potential interaction between microRNA and SNPs in 3ʹUTR of xenobiotic metabolism enzyme and transporter genes, Wei et al reported five putative miRNAs associated with rs10759637 of SLC31A1 in liver [18]. However, we could not experimentally validate these results in reporter gene assays in 16HBE cells (data not shown). Interestingly, we predicted two putative binding sites near rs10759637 for hsa-miR-29 members, and the structure of local SLC31A1 3ʹUTR harboring these sites could be affected by rs10759637. Furthermore, the C allele, which was associated with poor outcomes of platinum-based therapy in NSCLC patients, correlated with reductions in luciferase activity in reporter assay and in SLC31A1 transcript expression in clinical lung cancer tissues as well. Similarly, it was reported that one functional miRSNP, SNPs located at or near a microRNA binding site in 3ʹUTR of the target gene or in a microRNA, near the miR-24 binding site in the 3’UTR of dihydrofolate reductase gene (DHFR) interferes with miR-24 function, resulting in DHFR overexpression and methotrexate resistance [19]. A recent study also reported that the miRSNP rs1062980 may alter the expression of iron-responsive element binding protein 2 gene (IREB2) potentially through modulating the binding of miR-29a, and thereby modify risk of lung cancer [20]. The miR-29 family is expressed in lung tissues, and plays important roles in the pathogenesis of lung diseases by directly targeting genes such as those encoding extracellular matrix proteins and those associated with tissue invasion and metastasis in the contexts of fibrosis and adenocarcinoma, respectively [21, 22]. The regulation mechanism for SLC31A1 expression is largely unknown, two transcription factors, Sp1 and hypoxia-induced factor 2α (HIF2α), have been shown to mediate its basal and inducible expression [23, 24]. We noticed that rs4979223 and rs2233914 at the at 5ʹflanking region of SLC31A1 are not located at or near the cis-elements for Sp1 or HIF2α, their associations with toxicological phenotypes and survival could be largely due to their strong linkage disequilibrium with the functional rs10759637 at 3ʹUTR and genetic hitchhiking effect. Therefore, the modulation of rs10759637 as miRSNP on miR-29−SLC31A1 3ʹUTR interaction not also proposes underlying mechanism for the association of SLC31A1 polymorphism with clinical outcomes of platinum-based therapy, but also provides first evidence to link SLC31A1 gene to the miR-29 regulatory network.
Another notable aspect of this study is that genotypes of rs10759637 and linked variants, as well as their combined haplotype and diplotype, were consistently associated with severe hematological toxicities especially thrombocytopenia. Thrombocytopenia is one of major common hematologic complications of cancer chemotherapy and is associated with increased morbidity, mortality and health care costs [25]. In a large cohort of solid patients (total n = 47159; NSCLC n = 7001) undergoing chemotherapy including platinum-based regimens (accounted for about 1/3 and were most common for NSCLC), NSCLC patients had the highest prevalence of severe thrombocytopenia (10.7%) cross the four major chemotherapy regimens, suggesting that the burden of thrombocytopenia remains high [26]. Although the precise mechanism for chemotherapy-induced thrombocytopenia is not well established, many cytotoxic agents including platinum are now known to cause thrombocytopenia by predominantly inducing apoptosis of the progenitors of megakaryocytes, the platelet precursor, at early stages of differentiation [27, 28]. As genetic evidence supporting this toxicological mechanism, a recent whole-exome sequencing study reported that severe thrombocytopenia in NSCLC patients treated with gemcitabine/carboplatin are associated with variant of DDX53 gene, a modulator of p53 mediated apoptosis [29]. Considering that platinum agents are intravenously administered, their biotransformation and dynamic distribution in the bloodstream are likely to be directly involved inducing hematologic toxicity. Because SLC31A1 is ubiquitously expressed in all tissues of vertebrates [30], part of administered platinum agents could be imported into all tissue cells including lung cancer cells as target, the other fraction remains bound with plasma protein in circulation [31]. In this scenario, we could cautiously postulate that, through interfering miRNA mediated expression, the naturally occurring germline SNP rs10759637 in SLC31A1 3ʹUTR might render reduced expression of transporter to all tissue cells, thus result in lowered platinum uptake in overall mass tissues, and accumulated platinum agents in bone marrow and peripheral blood plasma, and consequently be associated with platinum resistance of target tissue cells and hematologic toxicity such as thrombocytopenia.
We should cautiously point out that, in the study population of Chinese NSCLC patient cohort, we observed slight deviation from HWE for the genotypic distribution of two clinically relevant SNPs of SLC31A1, rs4979223 (P = 0.026) in 5ʹflanking region and rs10759637 (P = 0.011) in 3ʹUTR, with about 43 kb distance but nearly complete linkage disequilibrium, which was due to significant underrepresentation of their putatively disadvantageous heterozygotes in patients that were pharmacogenetically associated with poor outcomes such as severe thrombocytopenia and shorter survival. These HWE tests could be statistically well powered by the large sample of the patient cohort and the common allelic frequencies of the two SNPs [32]. However, in the SNP datasets for natural populations, we found that genotypic distribution of rs10759637 complies with HWE both in the overall global population (the 1000 genome dataset) and in Chinese Han population (the HapMap CHB dataset) (data not shown). The patient ascertainment was well defined, and the two common SNPs were of independent genotyping calling but are in nearly complete linkage disequilibrium. It is therefore at least possible that the HWE departure was due to ascertainment bias or genotyping errors, but instead, possible explanations could be sampling bias, underlying genetic model at the susceptibility locus, and other unknown factors in view of population genetics and demography [33]. Consistent with this scenario, rs10759637 manifested unique association with clinical outcomes in the NSCLC cohort with heterozygote disadvantage, which is also referred to as under-dominance, a genetic model where heterozygote has a lower overall fitness than either homozygote [34]. Although being rare cases, heterozygote disadvantage was also reported to account for the deviation from HWE of a common variant (c.677C>T) of the 5,10-methylenetetrahydrofolate reductase gene (MTHFR) in subfertile patients and its association with embryo aneuploidy [35]. However, the biological mechanism by which the SLC31A1 rs10759637 A/C heterozygote could be disadvantage is unclear, especially considering that the variant C allele correlated with diminished gene expression. Furthermore, it has been reported of remarkable divergence in SLC31A1 expression in situ as well as related drug disposition and response among populations with different ethnic ancestry. In the cohort of NSCLC patients in American population who underwent neoadjuvant platinum-based chemotherapy, Kim et al. reported that the African American had significantly reduced SLC31A1 expression in tumor (P = 0.001), lowered tissue platinum concentration (P = 0.009) and decreased tumor shrinkage (P = 0.016) as compared to Caucasians [9]. We also noticed that, in public SNP database, the low-expression-related C allele of rs10759637 is the ancestral allele that is common in African population, the high-expression-related A allele is the derived allele that is dominant in Caucasian and Chinese Han population, suggesting of significant population differentiation at this locus. These observations not only support the function mode of rs10759637 on SLC31A1 expression, but also provide comparative and evolutionary pharmacogenetics implications for the association of SLC31A1 polymorphism with clinical outcomes of platinum-based chemotherapy in lung cancer.
In summary, this pharmacogenetic study on advanced NSCLC patients with platinum-based chemotherapy has identified linked variants of platinum import transporter SLC31A1 gene that were associated with severe hematological toxicities such as thrombocytopenia and shorter survival, one of which decreased gene expression through modulating microRNA-3ʹUTR interaction. Given that platinum-based chemotherapy is routinely used in clinical management of many types of cancers, and SLC31A1 is ubiquitously expressed in human tissues, validation of these findings in cohorts of lung cancer and other relevant types of cancer with larger sample size would be warranted in different ethnic populations.
MATERIALS AND METHODS
Patient recruitment and follow-up
This study encompassed in total 1004 eligible patients (Chinese Han) histologically diagnosed with stage III-IV NSCLC between March 2005 and January 2010 from five hospitals in the East of China: Shanghai Chest Hospital, Shanghai Zhongshan Hospital, Shanghai Changhai Hospital, Shanghai Changzheng Hospital, and Cancer Hospital of Jiangsu Province. The recruitment criteria for enrolling eligible patients and their demographic characteristics such as gender, age at diagnosis, smoking status, ECOG performance status, clinical TNM stage, and tumor histological type were described in detail in our previous reports [36–39]. No statistically significant difference was observed in the distribution of demographic features among the patients from the six hospitals (Pgender = 0.698, Page = 0.321). In addition, previously prepared lung cancer tissues from 47 surgically resectable patients recruited at Shanghai Changzheng Hospital were used for gene expression analyses [40].
The patients enrolled in the pharmacogenetic study were inoperable and received first-line, platinum-based chemotherapy (no prior surgery, radiotherapy, or concurrent chemoradiotherapy). The chemotherapeutic regimens were as follows: either cisplatin (75 mg/m2) or carboplatin (at an area under the curve 5), both administered on day 1 every 3 weeks, in combination with navelbine (25 mg/m2) on days 1 and 8 every 3 weeks, or gemcitabine (1250 mg/m2) on days 1 and 8 every 3 weeks, or paclitaxel (175 mg/m2) on day 1 every 3 weeks, or docetaxel (75 mg/m2) on day 1 every 3 weeks. A few patients received other platinum-based treatment (n = 49). All chemotherapeutic drugs were administered intravenously, and all treatments lasted for 2 to 6 cycles.
Clinical outcomes including toxicities, responses and survival were assessed in this study. The incidence of grade 3 or 4 chemotherapy toxicity was assessed for the patients from the end of the first two cycles of treatment according to the Common Terminology Criteria for Adverse Events version 3.0 (CTCAE v3.0), including overall toxicity, gastrointestinal toxicity (nausea and vomiting), and hematologic toxicity (leucopenia, neutropenia, anemia, and thrombocytopenia) [41]. No grade 5 toxicity (death) was observed. Responses to platinum-based chemotherapy were evaluated after the first two cycles of the course, which are classified into four categories namely complete response (CR), partial response (PR), stable disease (SD) and progressive disease (PD) according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines version 1.0 [42]. Object response rate (ORR) is defined as the percentage of patients with CR or PR. Survival data were collected from several sources including follow-up calls, the Social Security Death Index, and clinical medical records of inpatient and outpatient. Progression-free survival (PFS) was calculated from the date of chemotherapy beginning to the date of disease progression or death (whichever occurred first) or the last progression-free follow-up. Overall survival (OS) was calculated from the date of chemotherapy beginning to the date of death. The research assistants who performed the genotyping assays were blinded to the clinical state of patients and the clinical investigators were blinded to the genotypic state of patients. The study protocol was approved by the Ethical Review Committees of Fudan University School of Life Sciences and the participating hospitals, and written informed consent was obtained from each subjects.
SNPs selection and genotyping
Eight polymorphisms were selected by an approach combining both tagging and potentially functional SNPs of SLC31A1 gene. The tagging SNPs were screened from the Han Chinese in Beijing (CHB) population dataset of the HapMap database (http://www.hapmap.org) using a minor allele frequency (MAF) cutoff of 0.05 and a correlation coefficient (r2) threshold of 0.8. Genomic DNA was extracted from whole blood using the QIAamp DNA Maxi Kit (Qiagen GmbH, Hilden, Germany). Genotyping was performed using iSelect HD BeadChip (Illumina, San Diego, CA, USA) with the following quality-control criteria: genotyping call rate of SNP >0.95, GenCall score >0.2, and P value of Hardy-Weinberg equilibrium (HWE) >0.01. Genotyping of one SNP (rs10759637) at the 3ʹUTR for lung cancer tissues of the 47 surgically resectable patients was conducted using PCR-direct sequencing with forward primer 5ʹ-GGAGAGCAAGGAATGTGGACT-3ʹ and the reverse primer 5ʹ-GCAGCTCATGCTGAGATTTCTA-3ʹ.
In silico prediction of microRNA binding sites at the 3ʹUTR of SLC31A1
In order to identify putative microRNA targeting sequences in SLC31A1 3ʹUTR, and assess potential effect of rs10759637 on microRNA-mediated regulation, we used the miRdSNP database (http://mirdsnp.ccr.buffalo.edu/index.php) and TargetScanHuman 6.2 program (http://www.targetscan.org/) to screen microRNA binding sites, and used RNAfold (http://rna.tbi.univie.ac.at/) to predict the local secondary structure of the SLC31A1 mRNA.
Luciferase reporter assays
We performed luciferase reporter assays to characterize the effect of rs10759637 at SLC31A1 3ʹUTR on microRNA-mediated regulation. We constructed reporter gene vectors for rs10759637 by amplifying the target sequence about 1200bp around rs10759637 at SLC31A1 3ʹUTR from human genomic DNA, with the forward primer 5ʹ-CCGCTCGAGTCTACCAGTCATGGGCCAGAAGG-3ʹ and the reverse primer 5ʹ-ATTTGCGGCCGCGCTTCATCCTTTGGCAGCCAGTC-3ʹ, and after digestion with Xho I and Not I, cloning into the psiCHECK2 vector (Promega, Madison, WI, USA) near 3ʹUTR of Renilla luciferase. Reporter construct for rs10759637 variant was made through point mutation (C/A) with primeSTAR using primers 5ʹ-CAGCCTGTTGTCTCAAGCCAAGGAGTC-3ʹ (forward) and 5ʹ-GACTCCTTGGCTTGAGACAACAGGCTG-3ʹ (reverse). Empty vector psiCHECK2 was used as negative control. Three microRNA mimics (hsa-miR-29a, 5ʹ- UAGCACCAUCUGAAAUCGGUUA-3ʹ; hsa-miR-29b, 5ʹ-UAGCACCAUUUGAAAUCAGUGUU-3ʹ and hsa-miR-29c, 5ʹ-UAGCACCAUUUGAAAUCGGUUA-3ʹ) and one non-specific microRNA (miR-NC, 5ʹ-UUG UACUACACAAAAGUACUG-3ʹ) as negative control were synthetized from GenePharma, Shanghai China. Human bronchial epithelial cell line 16HBE (1.0 × 105 cells) was cotransfected with wild or variant rs10759637 report vector (400 ng) and microRNA (40pmol miRNA) using Lipofectamine™ 2000 (Invitrogen). Luciferase activity was measured with Dual-Luciferase Reporter Assay System (Promega) on Envision 2104 Multilabel Reader (PerkinElmer). Every treatment was set three replications and three independent experiments were performed.
Real-time quantitative RT-PCR
We quantified transcript level of SLC31A1 in lung cancer tissues from 47 patients using real-time quantitative RT-PCR assay. Total RNA was extracted from tumorous lung tissues with RNeasy Plus Mini Kit (QIAGEN, Hilden, Germany) and was then reverse transcribed into cDNA with PrimeScript RT Master Mix (TaKaRa, Dalian, China) using primers 5ʹ-GAGAGAGCCTGCTGCGTAAG-3ʹ (forward) and 5ʹ-AATGCAGAGGTACCCGTTGT-3ʹ (reverse). qRT-PCR was performed with SYBR® Premix Ex Taq Kit (Takara) on an Applied Biosystems 7900HT. We measured the expression of β-actin as internal control using primers 5ʹ-CAGAGCCTCGCCTTTGCC-3ʹ (forward) and 5ʹ-ATGCCGGAGCCGTTGTCG-3ʹ (reserve). Every sample was set in triplicate. Expression of SLC31A1 was normalized to β-actin with ΔΔCT method.
Statistical analysis
We used Pearson χ2 tests to examine deviations of genotype frequencies from those expected under HWE, and to test differences in the distributions of genotypes between patient groups with divergent clinical phenotypes or outcomes. We used the PHASE version 2.1 program to estimate haplotypes from the genotype data [43], and constructed the linkage disequilibrium (LD) plots using Haploview (http://www.broadinstitute.org/haploview), in which pairwise linkage disequilibrium relations among SNPs were examined using D′ and r2. We measured the association between genetic variants and dichotomous clinical phenotypes or outcomes by calculating odds ratios (OR) and their 95% confidence intervals (CIs) in unconditional logistic regression analysis, with adjustment of gender, age, smoking status, ECOG performance status, TNM status, histological types, and treatment regimen. Haplotype-based genetic association analysis was performed using Haplo.stats package in R-plus (available at http://cran.r-project.org/web/packages/haplo.stats.index.html; Version: 1.6.8). For each SNP, three different genetic models (dominant, recessive and additive) were analyzed, and the model with lowest P values was considered the best-fitting model. Under-dominant model was also analyzed for rs4979223 and rs10759637. The association between genetic variant and survival data (overall survival and progression-free-survival) was calculated by log-rank test with adjustment for covariates. The multivariate Cox proportional hazards regression was used for calculating the hazard ratios (HR) and 95% CI, Kaplan-Meier method was used to plot survival curve. All the statistical analysis was performed by SPSS (version 20). We use the two-side test for all P values. A P values < 0.05 was considered statistically significant. To account for the issue of multiple testing of SNPs, we used SNPSpD to correct the significance threshold taking into account LD between polymorphisms [44].
Abbreviations
3ʹUTR: 3ʹuntranslated region; AC: adenocarcinoma; CI: confidence interval; CR: complete response; ECOG PS: Eastern Cooperative Oncology Group performance status; HWE: Hardy-Weinberg equilibrium; HR: hazard ratio; LD: linkage disequilibrium; NSCLC: non-small-cell lung cancer; OR: odds ratio; OS: overall survival; PD: progressive disease; PFS: progression-free survival; PR: partial response; SCC: squamous cell carcinoma; SD: stable disease; SLC31A1: solute carrier family 31 member 1
Author contributions
HW, DL, JW, BH, CB, QL and LJ designed this study. JQ, ZG, JW and JX collected the samples and clinical information. CS, ZZ and YW analyzed the data. CS took the bioinformatics analysis on microRNA. JQ took experiments on functional analysis of gene expression. CS, ZZ and HW wrote the paper.
ACKNOWLEDGMENTS
We thank all the enrolled patients who make this study possible, and acknowledge greatly the collaboration from the participating hospitals and staff. This work was supported by the National Natural Science Foundation of China (No. 81172093, No. 81372526, No. 81572404, No. 81372236), the National Key Research Development Program (No. 2016YFC0905000), and the International S&T Cooperation Program of China (No. 2015DFE32790).
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
REFERENCES
1. Stewart BW. World Cancer Report 2014. International Agency for Research on Cancer (IARC). 2014.
2. Spiro SG, Silvestri GA. The treatment of advanced non-small cell lung cancer. Current Opinion in Pulmonary Medicine. 2005; 11:287–91. https://doi.org/10.1097/01.mcp.0000166590.03042.56.
3. Rabik CA, Dolan ME. Molecular mechanisms of resistance and toxicity associated with platinating agents. Cancer Treatment Reviews. 2007; 33:9–23. https://doi.org/10.1016/j.ctrv.2006.09.006.
4. Perez-Ramirez C, Canadas-Garre M, Molina MA, Robles AI, Faus-Dader MJ, Calleja-Hernandez MA. Contribution of genetic factors to platinum-based chemotherapy sensitivity and prognosis of non-small cell lung cancer. Mutat Res. 2017; 771:32–58. https://doi.org/10.1016/j.mrrev.2016.11.003.
5. Holzer AK, Manorek GH, Howell SB. Contribution of the major copper influx transporter CTR1 to the cellular accumulation of cisplatin, carboplatin, and oxaliplatin. Molecular Pharmacology. 2006; 70:1390–4. https://doi.org/10.1124/mol.106.022624.
6. Ishida S, Lee J, Thiele DJ, Herskowitz I. Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc Natl Acad Sci U S A. 2002; 99:14298–302. https://doi.org/10.1073/pnas.162491399.
7. Song IS, Savaraj N, Siddik ZH, Liu P, Wei Y, Wu CJ, Kuo MT. Role of human copper transporter Ctr1 in the transport of platinum-based antitumor agents in cisplatin-sensitive and cisplatin-resistant cells. Molecular Cancer Therapeutics. 2004; 3:1543–9.
8. Larson CA, Blair BG, Safaei R, Howell SB. The role of the mammalian copper transporter 1 in the cellular accumulation of platinum-based drugs. Mol Pharmacol. 2009; 75:324–30. https://doi.org/10.1124/mol.108.052381.
9. Kim ES, Tang X, Peterson DR, Kilari D, Chow CW, Fujimoto J, Kalhor N, Swisher SG, Stewart DJ, Wistuba II, Siddik ZH. Copper transporter CTR1 expression and tissue platinum concentration in non-small cell lung cancer. Lung Cancer. 2014; 85:88–93. https://doi.org/10.1016/j.lungcan.2014.04.005.
10. Kim ES, Lee JJ, He G, Chow CW, Fujimoto J, Kalhor N, Swisher SG, Wistuba II, Stewart DJ, Siddik ZH. Tissue platinum concentration and tumor response in non-small-cell lung cancer. J Clin Oncol. 2012; 30:3345–52. https://doi.org/10.1200/jco.2011.40.8120.
11. Xu X, Duan L, Zhou B, Ma R, Zhou H, Liu Z. Genetic polymorphism of copper transporter protein 1 is related to platinum resistance in Chinese non-small cell lung carcinoma patients. Clin Exp Pharmacol Physiol. 2012; 39:786–92. https://doi.org/10.1111/j.1440-1681.2012.05741.x.
12. Xu X, Ren H, Zhou B, Zhao Y, Yuan R, Ma R, Zhou H, Liu Z. Prediction of copper transport protein 1 (CTR1) genotype on severe cisplatin induced toxicity in non-small cell lung cancer (NSCLC) patients. Lung Cancer. 2012; 77:438–42. https://doi.org/10.1016/j.lungcan.2012.03.023.
13. Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. The New England Journal of Medicine. 2002; 346:92–8.
14. Guo W, Benlhabib H, Mendelson CR. The MicroRNA 29 Family Promotes Type II Cell Differentiation in Developing Lung. Molecular and Cellular Biology. 2016; 36:2141–50. https://doi.org/10.1128/mcb.00096-16.
15. Fabbri M, Garzon R, Cimmino A, Liu Z, Zanesi N, Callegari E, Liu S, Alder H, Costinean S, Fernandez-Cymering C, Volinia S, Guler G, Morrison CD, et al. MicroRNA-29 family reverts aberrant methylation in lung cancer by targeting DNA methyltransferases 3A and 3B. Proceedings of the National Academy of Sciences of the United States of America. 2007; 104:15805–10. https://doi.org/10.1073/pnas.0707628104.
16. Chen HH, Yan JJ, Chen WC, Kuo MT, Lai YH, Lai WW, Liu HS, Su WC. Predictive and prognostic value of human copper transporter 1 (hCtr1) in patients with stage III non-small-cell lung cancer receiving first-line platinum-based doublet chemotherapy. Lung Cancer. 2012; 75:228–34. https://doi.org/10.1016/j.lungcan.2011.06.011.
17. Ishida S, McCormick F, Smith-McCune K, Hanahan D. Enhancing tumor-specific uptake of the anticancer drug cisplatin with a copper chelator. Cancer Cell. 2010; 17:574–83. https://doi.org/10.1016/j.ccr.2010.04.011.
18. Wei R, Yang F, Urban TJ, Li L, Chalasani N, Flockhart DA, Liu W. Impact of the Interaction between 3’-UTR SNPs and microRNA on the Expression of Human Xenobiotic Metabolism Enzyme and Transporter Genes. Front Genet. 2012; 3:248. https://doi.org/10.3389/fgene.2012.00248.
19. Mishra PJ, Humeniuk R, Mishra PJ, Longo-Sorbello GS, Banerjee D, Bertino JR. A miR-24 microRNA binding-site polymorphism in dihydrofolate reductase gene leads to methotrexate resistance. Proc Natl Acad Sci U S A. 2007; 104:13513–8. https://doi.org/10.1073/pnas.0706217104.
20. Zhang L, Ye Y, Tu H, Hildebrandt MA, Zhao L, Heymach JV, Roth JA, Wu X. MicroRNA-related genetic variants in iron regulatory genes, dietary iron intake, microRNAs and lung cancer risk. Ann Oncol. 2017; 28:1124–9. https://doi.org/10.1093/annonc/mdx046.
21. Cushing L, Kuang PP, Qian J, Shao F, Wu J, Little F, Thannickal VJ, Cardoso WV, Lu J. miR-29 is a major regulator of genes associated with pulmonary fibrosis. Am J Respir Cell Mol Biol. 2011; 45:287–94. https://doi.org/10.1165/rcmb.2010-0323OC.
22. Plaisier CL, Pan M, Baliga NS. A miRNA-regulatory network explains how dysregulated miRNAs perturb oncogenic processes across diverse cancers. Genome Research. 2012; 22:2302–14. https://doi.org/10.1101/gr.133991.111.
23. Song IS, Chen HH, Aiba I, Hossain A, Liang ZD, Klomp LW, Kuo MT. Transcription factor Sp1 plays an important role in the regulation of copper homeostasis in mammalian cells. Mol Pharmacol. 2008; 74:705–13. https://doi.org/10.1124/mol.108.046771.
24. Pourvali K, Matak P, Latunde-Dada GO, Solomou S, Mastrogiannaki M, Peyssonnaux C, Sharp PA. Basal expression of copper transporter 1 in intestinal epithelial cells is regulated by hypoxia-inducible factor 2alpha. FEBS Lett. 2012; 586:2423–7. https://doi.org/10.1016/j.febslet.2012.05.058.
25. Elting LS, Rubenstein EB, Martin CG, Kurtin D, Rodriguez S, Laiho E, Kanesan K, Cantor SB, Benjamin RS. Incidence, cost, and outcomes of bleeding and chemotherapy dose modification among solid tumor patients with chemotherapy-induced thrombocytopenia. J Clin Oncol. 2001; 19:1137–46. https://doi.org/10.1200/jco.2001.19.4.1137.
26. Wu Y, Aravind S, Ranganathan G, Martin A, Nalysnyk L. Anemia and Thrombocytopenia in Patients Undergoing Chemotherapy for Solid Tumors: A Descriptive Study of a Large Outpatient Oncology Practice Database, 2000-2007. Clinical Therapeutics. 2009; 31:2416–32. https://doi.org/10.1016/j.clinthera.2009.11.020.
27. Zeuner A, Signore M, Martinetti D, Bartucci M, Peschle C, De Maria R. Chemotherapy-induced thrombocytopenia derives from the selective death of megakaryocyte progenitors and can be rescued by stem cell factor. Cancer Res. 2007; 67:4767–73. https://doi.org/10.1158/0008-5472.can-06-4303.
28. Jelic S, Radulovic S. Chemotherapy-Associated Thrombocytopenia_ Current and Emerging Management Strategies. Am J Cancer. 2006; 5:371–82.
29. Green H, Hasmats J, Kupershmidt I, Edsgard D, de Petris L, Lewensohn R, Blackhall F, Vikingsson S, Besse B, Lindgren A, Branden E, Koyi H, Peterson C, et al. Using Whole-Exome Sequencing to Identify Genetic Markers for Carboplatin and Gemcitabine-Induced Toxicities. Clin Cancer Res. 2016; 22:366–73. https://doi.org/10.1158/1078-0432.ccr-15-0964.
30. Kuo YM, Zhou B, Cosco D, Gitschier J. The copper transporter CTR1 provides an essential function in mammalian embryonic development. Proc Natl Acad Sci U S A. 2001; 98:6836–41. https://doi.org/10.1073/pnas.111057298.
31. Sooriyaarachchi M, Narendran A, Gailer J. Comparative hydrolysis and plasma protein binding of cis-platin and carboplatin in human plasma in vitro. Metallomics. 2011; 3:49–55. https://doi.org/10.1039/c0mt00058b.
32. Salanti G, Amountza G, Ntzani EE, Ioannidis JP. Hardy-Weinberg equilibrium in genetic association studies: an empirical evaluation of reporting, deviations, and power. Eur J Hum Genet. 2005; 13:840–8. https://doi.org/10.1038/sj.ejhg.5201410.
33. Wittke-Thompson JK, Pluzhnikov A, Cox NJ. Rational inferences about departures from Hardy-Weinberg equilibrium. Am J Hum Genet. 2005; 76:967–86. https://doi.org/10.1086/430507.
34. Laruson AJ, Reed FA. Stability of underdominant genetic polymorphisms in population networks. J Theor Biol. 2016; 390:156–63. https://doi.org/10.1016/j.jtbi.2015.11.023.
35. Enciso M, Sarasa J, Xanthopoulou L, Bristow S, Bowles M, Fragouli E, Delhanty J, Wells D. Polymorphisms in the MTHFR gene influence embryo viability and the incidence of aneuploidy. Hum Genet. 2016; 135:555–68. https://doi.org/10.1007/s00439-016-1652-z.
36. Song X, Wang S, Hong X, Li X, Zhao X, Huai C, Chen H, Gao Z, Qian J, Wang J, Han B, Bai C, Li Q, et al. Single nucleotide polymorphisms of nucleotide excision repair pathway are significantly associated with outcomes of platinum-based chemotherapy in lung cancer. Sci Rep. 2017; 7:11785. https://doi.org/10.1038/s41598-017-08257-7.
37. Li X, Shao M, Wang S, Zhao X, Chen H, Qian J, Song X, Wang J, Jin L, Wu J, Li Q, Bai C, Han B, et al. Heterozygote advantage of methylenetetrahydrofolate reductase polymorphisms on clinical outcomes in advanced non-small cell lung cancer (NSCLC) patients treated with platinum-based chemotherapy. Tumour Biol. 2014; 35:11159–70. https://doi.org/10.1007/s13277-014-2427-6.
38. Zhao X, Wang S, Wu J, Li X, Wang X, Gao Z, Wu W, Wang H, Wang J, Qian J, Ma K, Li H, Han B, et al. Association of TERT Polymorphisms with Clinical Outcome of Non-Small Cell Lung Cancer Patients. PLoS One. 2015; 10:e0129232. https://doi.org/10.1371/journal.pone.0129232.
39. Wang S, Song X, Li X, Zhao X, Chen H, Wang J, Wu J, Gao Z, Qian J, Han B, Bai C, Li Q, Lu D. RICTOR polymorphisms affect efficiency of platinum-based chemotherapy in Chinese non-small-cell lung cancer patients. Pharmacogenomics. 2016; 17:1637–47. https://doi.org/10.2217/pgs-2016-0070.
40. Xiang C, Wang J, Kou X, Chen X, Qin Z, Jiang Y, Sun C, Xu J, Tan W, Jin L, Lin D, He F, Wang H. Pulmonary expression of CYP2A13 and ABCB1 is regulated by FOXA2, and their genetic interaction is associated with lung cancer. FASEB J. 2015; 29:1986–98. https://doi.org/10.1096/fj.14-264580.
41. Carbone DP, Minna JD. Chemotherapy for non-small cell lung cancer. BMJ. 1995; 311:889–90.
42. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000; 92:205–16.
43. Stephens M, Donnelly P. A comparison of bayesian methods for haplotype reconstruction from population genotype data. Am J Hum Genet. 2003; 73:1162–9. https://doi.org/10.1086/379378.
44. Nyholt DR. A simple correction for multiple testing for single-nucleotide polymorphisms in linkage disequilibrium with each other. Am J Hum Genet. 2004; 74:765–9. https://doi.org/10.1086/383251.

