INTRODUCTION
Neuroendocrine neoplasms (NENs) arise most frequently in the gastroentero-pancreatic system and lungs. These neoplasms originate in scattered endocrine cells of the diffuse neuroendocrine system and are biologically very heterogeneous [1]. Two groups of NEN behave biologically and clinically distinctively different; the well differentiated slowly growing neuroendocrine tumors with an excellent to good prognosis and poorly differentiated fast growing neuroendocrine carcinomas with a poor prognosis. Grading of NENs is performed by Ki67 index. G1 and G2 neuroendocrine tumors have a Ki67 index of 20% or below whereas neuroendocrine carcinomas have a Ki 67 index of more than 20%. Neuroendocrine carcinomas are defined by small or large cell morphology. In addition, the new WHO classification of pancreatic NENs includes the newly created well differentiated neuroendocrine tumors with a proliferation of up to 50% as neuroendocrine tumor G3 [2, 3]. NENs may be functionally active due to the autonomous secretion of biogenic amines or peptide hormones such as serotonin, gastrin or insulin causing characteristic functional syndromes [4].
The majority of well-differentiated NENs overexpress receptors of the 5 subclasses of somatostatin receptors (SSTRs), most frequently receptors 2 and 5. Activated SSTRs have anti-secretory and anti-proliferative activity, and are targeted by stable somatostatin analogues for treatment of functional syndromes and to reduce tumor growth of NENs [5–7]. In addition, SSTR are utilized to visualize NENs by molecular imaging. SSTR scintigraphy uses stable somatostatin analogues coupled to short-lived radionuclides such as 111Indium or 99mTechnetium. The more recently developed SSTR PET/CT involves 68Gallium (68Ga) as a positron emitter, coupled to stable somatostatin analogues. This technique is more sensitive, faster with much lesser radiation exposure, compared to the standard SSTR scintigraphy [8]. Netspot, the first kit for the preparation of 68Ga-DOTATATE for PET/CT imaging of NENs was recently approved by the US FDA (http://www.fda.gov/Drugs/InformationOnDrugs/ucm508921.htm).
Coupling beta-and/or gamma-emitting radionuclides to stable somatostatin analogues permits internal radiation of SSTR-expressing NENs by peptide receptor radionuclide therapy (PRRT). 90Yttrium (90Y) and 177Lutetium (177Lu) are used as radionuclides and chelated to stable somatostatin analogues such as DOTATATE and DOTATOC [9, 10]. Numerous phase I and phase II studies have shown favorable progression-free survival (PFS) and overall survival (OS) in NEN patients compared to historical controls [9–11]. Recently, a phase III study (NETTER-1 trial) has compared 4 cycles of PRRT, each using 7.4 GBq of 177Lu-DOTATATE (Lutathera®) together with 30 mg Octreotide-LAR every 4 weeks, to high dose somatostatin analogue therapy with 2 x 30 mg Octreotide-LAR every 4 weeks. Patients with progressive NENs of small bowel under treatment with somatostatin analogues and with SSTR expression were included, and 230 were randomized in a 1:1 ratio. In the control group, median PFS was 8.4 months, whereas it was not reached yet in the Lutathera® group (estimation 40 months). The hazard ratio was 0.209 (95% CI, 0.129-0.33) in favor of Lutathera®. Adverse events were rare and the treatment was well tolerated [12].
Standardized uptake values (SUVs) of SSTR PET/CT predict absorbed dose and response to PRRT [13–16]. Here, we present OS and PFS in 1048 patients treated with at least one cycle of PRRT using 90Y or 177Lu, which were selected and re-staged by 68Ga-SSTR PET/CT (Figure 1). Response to PRRT evaluated by radiological criteria (RECIST 1.1) has been reported previously in two subgroups of 56 and 200 patients, respectively [11, 17].
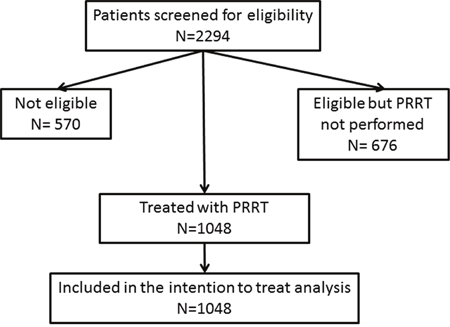
Figure 1: Flow chart of trial characteristics.
RESULTS
Patient characteristics
The majority of patients (593/1048) were males (56.6%) and 455 (43.4%) were females. Most patients (559/1048) were between 41 and 60 years at diagnosis (53.3%) whereas 393 (37.5%) were older than 60 years. At diagnosis, 96 patients (9.2%) were 40 years or younger. This group included 1 patient less than 10 years of age, 5 patients between 11 and 20 years, 24 patients between 21 and 30 and 76 patients aged between 31 and 40 years. Most patients received PRRT with 177Lu; 378 as monotherapy (36.1%) and 513 (48.9%) in combination with 90Yttrium either as a combination of both radionuclides in one cycle (TANDEM) or sequential (DUO). Only 157 patients (15%) received 90Y-DOTATATE or -DOTATOC as monotherapy. Mean administered radioactivity was 18.84 GBq with a minimum of 1.4 GBq and a maximum of 63.9 GBq. Most patients (247 and 399, respectively) had well differentiated NENs of grade 1 (23.6%) or grade 2 (38.1%). A minority of 67 patients (6.4%) had well differentiated NENs grade 3. Grading was not available in 335 patients (31.9%), which were diagnosed before WHO criteria were published and no tissue was available anymore for analysis. These patients were included as a separate group. The majority of patients had been pretreated by one or more other therapies (surgery, somatostatin analogs, chemotherapy, molecular therapy). PRRT was performed as first-line treatment in 119 patients (11.4%) with mostly ileum NENs. More than 3 other therapies before PRRT had been performed in 209 patients (19.9%), whereas 417 patients (39.8%) had received 2-3 therapies and 303 (28.9%) had had one therapy. Primary tumors were localized in pancreas (384 or 36.6%), small intestine (315 or 30.1%), lung (75 or 7.2%), colon and rectum (52 or 5.0%), duodenum (22 or 2.1%), thymus and mediastinum (16 or 1.5%) stomach (15 or 1.4%), caecum and appendix (5 or 0.5%), others (13 or 1.2%) and 151 patients (14.4%) had tumors with unknown primary site. Other primary tumors included paragangliomas/pheochromocytomas (15 patients) and 25 patients with medullary thyroid cancer. For statistical analysis, the group of others included other primary tumor as described above and primaries of duodenum, stomach, thymus, mediastinum, stomach, caecum and appendix.
Functional syndromes were present in 244 patients (23.3%), whereas 804 (76.7%) had no symptoms of hormone overproduction. Carcinoid syndrome was predominant (158 or 15.0%), followed by Zollinger-Ellison syndrome/gastrinoma (46 or 4.4%), the syndromes caused by glucagonoma (16 or 1.5%), insulinoma (15 or 1.4%) and the Werner-Morrison syndrome caused by VIPoma (8 or 0.7%).
Overall survival
Data and statistical analysis on overall survival (OS) is presented in Table 1. Of 1048 patients, 573 (54.7%) were alive with a median OS of 51 months (Figure 2A). Univariate analysis suggested a significant difference in OS for female gender, which was not corroborated by multivariate analysis (Table 1, Supplementary Figure 1A). Compared to the main age group at diagnosis between 40 and 60 years, patients older than 60 years at diagnosis had a significantly shorter survival. Longer survival of patients younger than 40 years at diagnosis was not statistically significant in multivariate analysis (Table 1, Supplementary Figure 1B). Best OS was achieved by a combination of 90Y- and 177Lu- for PRRT. Shortest survival was observed in patients treated exclusively with 90Y, while treatment with solely 177Lu resulted in an intermediate survival between the two extreme curves. These differences were highly statistically significant in univariate and multivariate analysis (Table 1, Figure 3A). Grading of NENs was a strong predictor of overall survival. Best overall survival was achieved in the G1 group, followed by G2. G3 NENs had the shortest overall survival with 23 months compared to 88 months in the G1 group. Compared to G2, overall survival of G1 patients was significantly longer and significantly shorter in the G3 group. The group of patients with G3 NENs included 18 patients with a proliferation rate of more than 50%. These patients had a median overall survival of 8.7 months (95% confidence interval 0-21.1).
Table 1: Patient characteristics and results of overall survival after start of PRRT
Analysis |
Number |
% |
Death patients |
Median |
95% CI |
Univariate analysis |
Multivariate analysis |
||
|---|---|---|---|---|---|---|---|---|---|
p |
HR |
95% CI |
p |
||||||
All Patients |
1048 |
100 |
475 |
51 |
47.0-54.9 |
||||
Gender |
|||||||||
Male |
593 |
56.6 |
276 |
50 |
45.4-54.5 |
0.029 |
1 |
||
Female |
455 |
43.4 |
199 |
53 |
47.3-58.6 |
0.84 |
0.7-1.02 |
0.07 |
|
Age |
|||||||||
≤40 years |
96 |
9.1 |
40 |
70 |
33.3-106.6 |
0.018 |
0.91 |
0.65-1.28 |
0.6 |
>40 and ≤60 years |
559 |
53.4 |
248 |
54 |
46.6-61.3 |
1 |
|||
>60 years |
393 |
37.5 |
187 |
46 |
41.8-50.2 |
1.32 |
1.08-1.61 |
0.01 |
|
Radionuclide |
|||||||||
Lutetium-177 |
378 |
36 |
143 |
44 |
36-1-52 |
<0.001 |
1.67 |
1.33-2.08 |
<0.001 |
Yttrium-90 |
157 |
15 |
100 |
24 |
17.4-30.6 |
2.89 |
2.27-3.69 |
<0.001 |
|
combined |
513 |
49 |
232 |
64 |
51.7-64.3 |
1 |
|||
Grading |
|||||||||
G1 (Ki67<2%) |
247 |
23.5 |
76 |
88 |
69.3-106.6 |
<0.001 |
0.65 |
0.49-0.86 |
0.0025 |
G2 (Ki67 3-20%) |
399 |
38.1 |
162 |
51 |
44-57.9 |
1 |
|||
G3 (Ki67>20%) |
67 |
6.4 |
22 |
23 |
10.8-35.2 |
1.71 |
1.21-2.4 |
0.0023 |
|
unknown |
335 |
32 |
143 |
46 |
41.5-50.4 |
1.04 |
0.84-1.29 |
0.74 |
|
Previous therapies |
|||||||||
0 |
119 |
11.4 |
23 |
55 |
37.4-72.5 |
<0.001 |
0.72 |
0.46-1.12 |
0.15 |
1 |
303 |
29 |
119 |
62 |
52.1-71.8 |
0.7 |
0.55-0.88 |
0.0028 |
|
2-3 |
417 |
39.7 |
218 |
51 |
45-56.9 |
1 |
|||
>3 |
209 |
19.9 |
115 |
41 |
35.3-46.6 |
1.39 |
1.1-1.76 |
0.01 |
|
Primary tumor |
|||||||||
Bronchial |
75 |
7.2 |
41 |
40 |
30.5-49.4 |
0.01 |
1.08 |
0.76-1.54 |
0.67 |
Pancreas |
384 |
36.7 |
200 |
44 |
37.7-50.2 |
1 |
|||
Small Intestine |
315 |
30 |
120 |
69 |
53.7-84.2 |
0.57 |
0.44-0.75 |
<0.001 |
|
CUP |
151 |
14.4 |
60 |
53 |
37.5-68.4 |
0.78 |
0.58-1.06 |
0.11 |
|
Other |
123 |
11.7 |
54 |
47 |
38.8-55.1 |
0.7 |
0.51-0.95 |
0.02 |
|
Functional syndromes |
|||||||||
Carcinoid syndrome |
158 |
15 |
82 |
49 |
38.7-59.2 |
0.012 |
1.17 |
0.89-1.52 |
0.26 |
Gastrinoma |
46 |
4.3 |
21 |
66 |
48.9-83 |
0.8 |
0.51-1.26 |
0.34 |
|
Insulinoma |
15 |
1.4 |
10 |
32 |
9.7-54.2 |
1.06 |
0.56-2.03 |
0.86 |
|
Glucagonoma |
16 |
1.5 |
7 |
127 |
nr |
0.65 |
0.3-1.4 |
0.27 |
|
VIPoma |
8 |
0.7 |
3 |
46 |
14.6-77.3 |
0.78 |
0.25-2.46 |
0.68 |
|
Other |
1 |
0.1 |
1 |
52 |
nr |
5.64 |
0.87-36.6 |
0.07 |
|
None |
804 |
76.7 |
351 |
51 |
47-54.9 |
1 |
|||
Abbreviations: nr: not reached; 95 CI: 95% confidence interval bold figures indicate significant results.
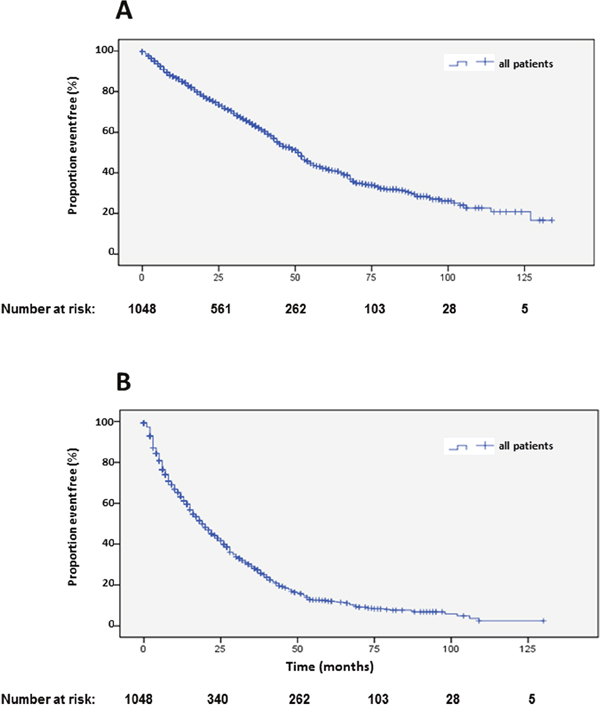
Figure 2: Kaplan-Meier plots of overall survival (A) and progression-free survival 1 (B) after start of PRRT.
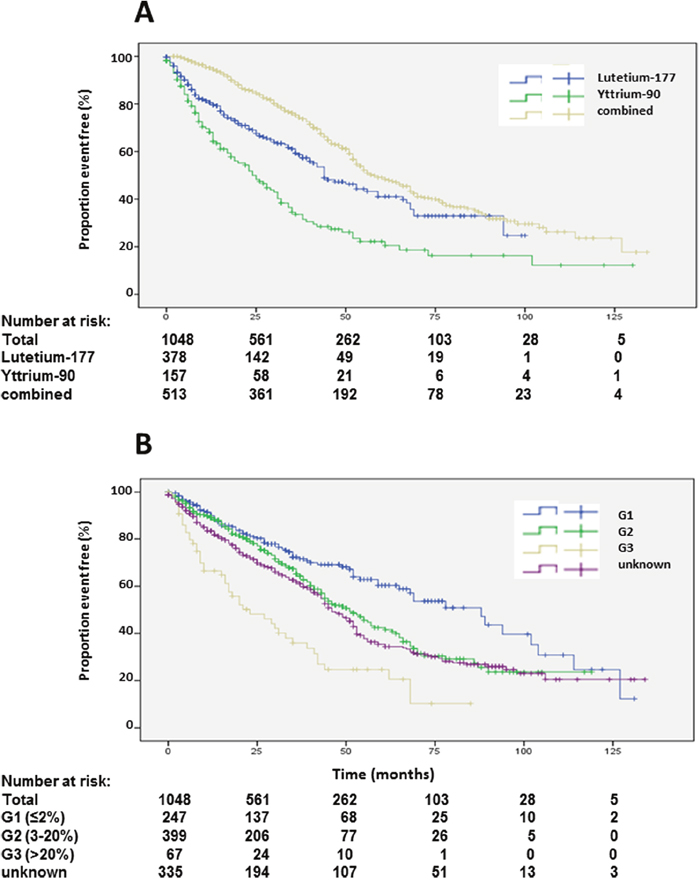
Figure 3: Kaplan-Meier plots of overall survival according to radioisotope (A) and grading (B).
Patients with unknown grading had similar overall survival as G2 patients (Table 1, Figure 3B). Patients with more than 3 prior therapies had significantly lower survival whereas patients with only one previous therapy had a significantly longer survival (Table 1, Figure 4A). Site of origin of NENs was a predictor of median overall survival in one group only. Best survival was observed in patients with NENs of small bowel (69 months), which was statistically significant followed by CUP, others, pancreas and lung. In between the later groups, differences did not achieve statistical significance except for the heterogeneous group of other neuroendocrine neoplasias (Table 1, Figure 4B). This group included 25 patients with medullary thyroid cancer. These patients had a median overall survival of 32 months (23.8-43.5).
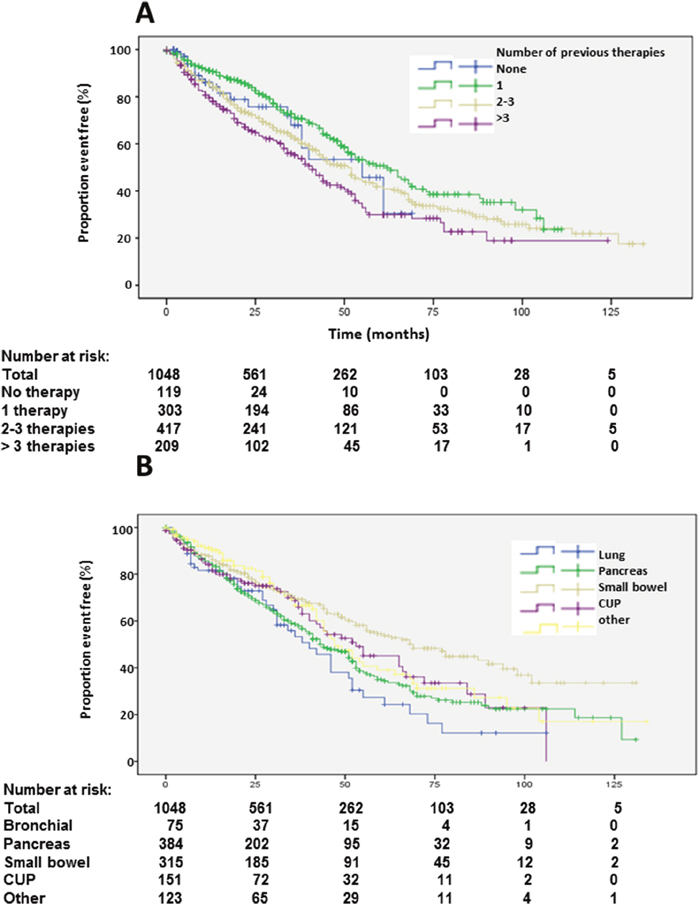
Figure 4: Kaplan-Meier plots of overall survival according to number of previous therapies (A) and primary tumors (B).
The majority of patients had no functional syndrome and a median overall survival of 51 months. Patients with carcinoid syndrome had a slightly lower median overall survival of 49 months whereas patients with gastrinomas and glucagonomas had longer median overall survival of 66 and 127 months, respectively. Patients with insulinomas had a shorter median overall survival of 32 months whereas patients with VIPomas were similar to non-functional patients or patients with carcinoid syndrome with a median overall survival of 46 months (Table 1, Supplementary Figure 3A).
Progression-free survival
Patients were restaged by 68Ga-SSTR PET/CT and tumor status was determined by RECIST and EORTC criteria. In addition, progression was defined as resumption of PRRT after an interval of more than 6 months or of any other therapy such as surgery, chemotherapy, local ablative therapies or molecular therapies according to the decision of the tumor board. As such, we determined progression-free survival after at least one cycle of PRRT (PFS1) including all 1048 patients (Table 2). Resumption of PRRT (2nd phase) was performed in 470 patients (PFS2; Supplementary Table 1) and again (3rd phase) in 184 patients (PFS3; Supplementary Table 2).
Table 2: Patient characteristics and results of progression-free survival after start of PRRT
Number |
% |
Progressive patients |
Median |
95% CI |
Univariate analysis |
Multivariate analysis |
|||
|---|---|---|---|---|---|---|---|---|---|
p |
HR |
95% CI |
p |
||||||
All Patients |
1048 |
100 |
774 |
19 |
16.9-21 |
||||
Gender |
|||||||||
Male |
593 |
56.6 |
447 |
18 |
15.9-20 |
0.108 |
1 |
||
Female |
455 |
43.4 |
327 |
22 |
18.8-25.1 |
0.87 |
0.75-1 |
0.05 |
|
Age |
|||||||||
≤40 years |
96 |
9.1 |
68 |
18 |
13.9-22 |
0.281 |
0.92 |
0.71-1.19 |
0.51 |
>40 and ≤60 years |
559 |
53.4 |
420 |
21 |
17.7-24.2 |
1 |
|||
>60 years |
393 |
37.5 |
286 |
19 |
16.4-21.5 |
1.04 |
0.89-1.22 |
0.64 |
|
Radionuclide |
|||||||||
Lutetium-177 |
378 |
36 |
236 |
17 |
14-19.9 |
<0.001 |
1.12 |
0.94-1.33 |
0.20 |
Yttrium-90 |
157 |
15 |
122 |
13 |
9.8-16.1 |
1.41 |
1.15-1.74 |
0.001 |
|
combined |
513 |
49 |
416 |
24 |
21.2-26.7 |
1 |
|||
Grading |
|||||||||
G1 (Ki67<2%) |
247 |
23.5 |
162 |
22 |
16.6-27.3 |
<0.001 |
0.86 |
0.71-1.05 |
0.14 |
G2 (Ki67 3-20%) |
399 |
38.1 |
292 |
21 |
18.1-23.9 |
1 |
|||
G3 (Ki67>20%) |
67 |
6.4 |
58 |
7 |
5-8.9 |
1.72 |
1.29-2.3 |
<0.001 |
|
unknown |
335 |
32 |
262 |
19 |
15.1-22.8 |
0.91 |
0.77-1.08 |
0.3 |
|
Previous therapies |
|||||||||
0 |
119 |
11.4 |
45 |
24 |
8.3-39.6 |
<0.001 |
0.78 |
0.57-1.08 |
0.13 |
1 |
303 |
29 |
231 |
22 |
18.8-25.1 |
0.91 |
0.77-1.09 |
0.32 |
|
2-3 |
417 |
39.7 |
334 |
18 |
14.8-21.1 |
1 |
|||
>3 |
209 |
19.9 |
164 |
17 |
12.4-21.5 |
1.18 |
0.97-1.43 |
0.10 |
|
Primary tumor |
|||||||||
Bronchial |
75 |
7.2 |
62 |
11 |
6.3-15.6 |
<0.001 |
1.43 |
1.08-1.91 |
0.01 |
Pancreas |
384 |
36.7 |
303 |
20 |
17-22.9 |
1 |
|||
Small Intestine |
315 |
30 |
211 |
22 |
17.6-26.3 |
0.75 |
0.61-0.92 |
0.01 |
|
CUP |
151 |
14.4 |
115 |
13 |
9.5-16.4 |
1.27 |
1.01-1.58 |
0.04 |
|
Other |
123 |
11.7 |
83 |
20 |
13.9-26 |
0.83 |
0.64-1.06 |
0.14 |
|
Functional syndromes |
|||||||||
Carcinoid syndrome |
158 |
15 |
128 |
16 |
12.6-19.3 |
0.612 |
1.18 |
0.95-1.46 |
0.13 |
Gastrinoma |
46 |
4.3 |
39 |
26 |
19.1-32.8 |
1.01 |
0.72-1.42 |
0.95 |
|
Insulinoma |
15 |
1.4 |
13 |
28 |
12.5-43.4 |
0.95 |
0.54-1.66 |
0.86 |
|
31Glucagonoma |
16 |
1.5 |
13 |
20 |
nr-40.1 |
0.94 |
0.54-1.66 |
0.85 |
|
VIPoma |
8 |
0.7 |
6 |
22 |
14.6-29.3 |
0.79 |
0.35-1.79 |
0.59 |
|
Other |
1 |
0.1 |
1 |
16 |
nr |
1.96 |
0.27-14.3 |
0.52 |
|
None |
804 |
76.7 |
574 |
19 |
16.9-21 |
1 |
|||
Abbreviations: nr: not reached; 95 CI: 95% confidence interval bold figures indicate significance results.
Median PFS1 based on the EORTC PET criteria was 19 months (Figure 2B). Detailed subgroup and statistical analysis of PFS1 is depicted in Table 2. Uni-variate analysis depicted statistically significant differences according to radionuclides (Figure 5A), grading (Figure 5B), number of previous therapies (Figure 6B) and origin of primary tumors (Figure 6A) but not gender (Supplementary Figure 2A), age (Supplementary Figure 2B) and functionality (Supplementary Figure 3B) (Table 2). Multi-variate analysis revealed that patients treated with 90Y exclusively had a significantly shorter PFS than patients treated with 177Lu or a combination of both radionuclides (Table 2, Figure 5A). Grading determined a significantly lower PFS only in patients with G3 tumors, but there was no significant difference in PFS between G1 and G2 tumors, or between G2 tumors and tumors with unknown proliferation rate (Figure 5B, Table 2). Patients with G3 NENs with more than 50% proliferation rate had a median PFS1 of 5.7 months (nr-12.1) compared to all G3 patients with a median PFS1 of 7 months (Table 2).
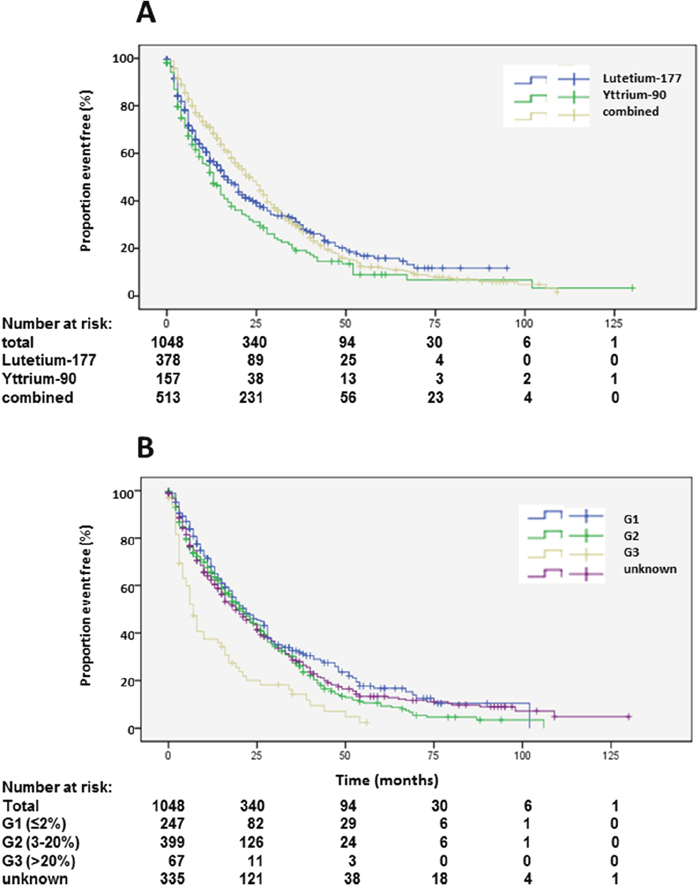
Figure 5: Kaplan-Meier plots of progression-free survival 1 according to radioisotope (A) and grading (B).
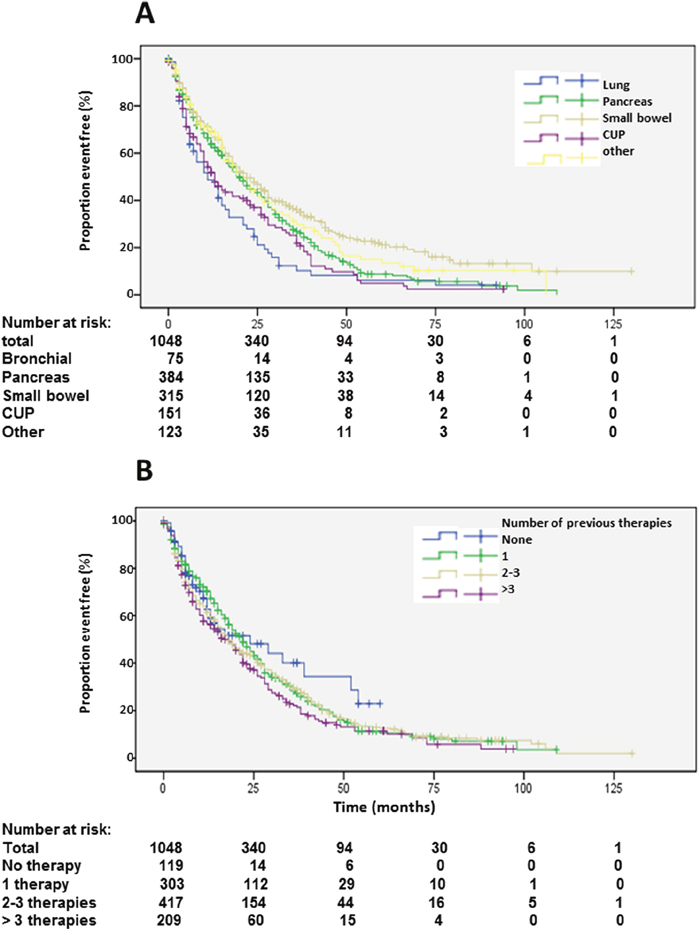
Figure 6: Kaplan-Meier plots of progression-free survival 1 according to primary tumors (A) and number of previous therapies (B).
Origin of primary tumors influenced PFS. Patients with neuroendocrine neoplasias of small bowel had a longer PFS compared to patients with pancreatic NENs whereas bronchial origin und unknown origin was associated with a significantly shorter PFS in multi-variate analysis (Figure 6A, Table 2). The 25 patients with medullary thyroid cancer had a median PFS1 of 10.1 months (2-18.2). After an interval of more than 6 months, 470 patients underwent re-PRRT. Median PFS2 was 11 months (Figure 7A; Supplementary Table 1). Univariate analysis indicated statistically significant differences according to radionuclides, grading and origin of neuroendocrine neoplasia but no influence of gender, age, number of previous therapies and functionality (Supplementary Table 1). In multi-variate analysis, these differences were attributable to significantly lower PFS in patients treated with 90Y solely (Supplemantary Table 1). Patients with unknown grading had a significantly longer progression free survival with a median of 11 months, as compared to 9 months in patients with G2 tumors (Supplementary Figure 4A, Supplementary Table 1).
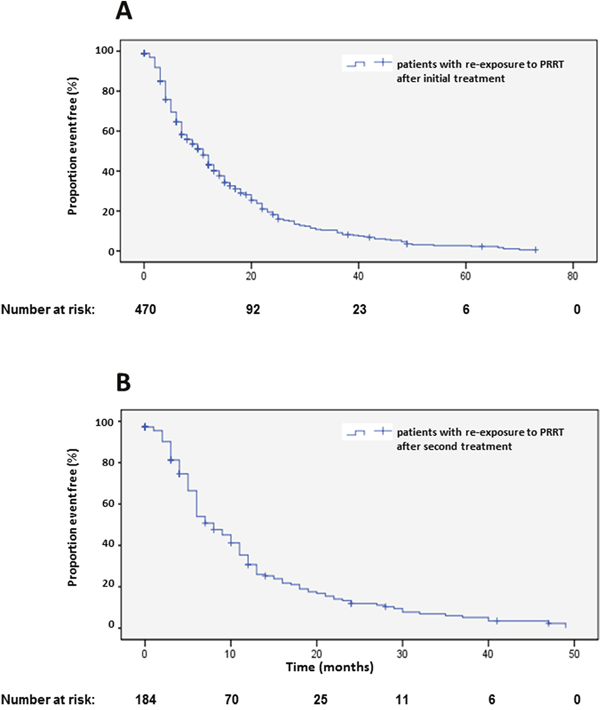
Figure 7: Kaplan-Meier plots of progression-free survival 2 (A) and 3 (B) after re-start of PRRT.
In 184 patients, a re-re-PRRT was performed after an interval of more than 6 months after re-PRRT (Supplementary Table 2). Median PFS was 8 months (Figure 7B). Here, the small size of the different groups limited the analysis of statistical significance. Uni-variate and multi-variate analysis suggested a significantly longer PFS in female patients (Supplementary Table 2). Multi-variate analysis indicated a shorter PFS in patients younger than 40 years and longer PFS in patients with unknown grading (Supplementary Figure 4B, Supplemenary Table 2), however, these results were limited by the small numbers of sub-groups.
Adverse events
Adverse events were recorded according to patient and treatment cycle according to CTCAE criteria (Table 3). Hemoglobin levels, number of leucocytes, platelets and creatinine levels were determined before and after PRRT. After the initial PRRT (1048 cycles), grade 3 and 4 adverse events were rare. Dialysis was performed before PRRT in 3 patients and continued thereafter. After 2633 follow-up cycles, grade 3 and 4 adverse events were still prevalent in less than 1% of patients. Laboratory values were then studied across all 3692 cycles. Here, grade 3 and 4 adverse events did not increase and were still below 1%. Missing values were less than 1% (Table 3). Dialysis was necessary at any time after PRRT in additional 5 patients of 1048 (0.4%) patients. During follow-up, MDS or leukemia developed in 22 (2%) patients (Table 4), which developed after a mean of 8 years after diagnosis of the neuroendocrine neoplasia and was most prevalent in patients with NEN of pancreatic or small bowel. Possible risk factors included previous chemotherapy and external beam radiation. Most patients developing MDS or leukemia had a high tumor load with hepatic metastases. Prognosis of patients with MDS/leukemia was dismal with a mean overall survival of 14.4 months after diagnosis (Table 4).
Table 3: Number of patients with adverse events according to CTCAE criteria
Before first PRRT (1048) |
||||
|---|---|---|---|---|
Normal, G1, G2 |
G3 |
G4 |
No information |
|
Leucocytes |
1047 (99.9%) |
0 |
0 |
1 |
Thrombocytes |
1047 (99.9%) |
0 |
3 |
1 |
Hemoglobin |
1040 (99.2%) |
3 |
3 |
1 |
Chronic kidney disease |
1040 (99.2%) |
1 |
2 |
4 (0.3%) |
According to treatment cycles (3692) |
||||
|---|---|---|---|---|
Normal, G1, G2 |
G3 |
G4 |
No information |
|
Leucocytes |
3680 (99.6%) |
8 (0.2%) |
0 |
4 (0.1%) |
Thrombocytes |
3679 (99.6%) |
1 |
8 (0.2%) |
4 (0.1%) |
Hemoglobin |
3666 (99.2%) |
10 (0.2%) |
7 |
9 (0.2%) |
Chronic kidney disease |
3664 (99.2%) |
7 (0.1%) |
7 (0.2%) |
14 (0.3) |
Table 4: Characteristics of patients with MDS or leukemia after PRRT
Age at diagnosis of neuroendocrine neoplasia |
Age at start of PRRT |
Age at diagnosis of MDS/leukemia |
Radionuclides and dose |
Type of MDS/leukemia |
Type and extend of neuroendocrine neoplasia |
Other risk factors |
Sex |
Survival after diagnosis of MDS/leukemia (death or date last seen) |
|
|---|---|---|---|---|---|---|---|---|---|
1 |
40 years |
42 years |
49 years |
131Iodine MIBG 22.7 GBq90Yttrium 7.4 GBq |
MDS |
paraganglioma hepatic, lymphatic and osseous metastases |
chemotherapy external beam radiation (40 Gy) |
female |
15 months |
2 |
67 years |
68 years |
69 years |
90Yttrium 8 GBq177Lutetium 6 GBq |
RAEB/T |
neuroendocrine carcinoma pancreas hepatic, lymphatic and osseous metastases |
right sided heart disease hypertension |
female |
6 months |
3 |
66 years |
79 years |
80 years |
90Yttrium 9.5 GBq 177Lutetium 6.3 GBq |
RAEB-1 |
neuroendocrine tumor of unknown primary hepatic metastases |
right sided heart disease hypertension |
female |
16 months |
4 |
63 years |
64 years |
66 years |
90Yttrium 14 GBq |
RAEB/AML |
neuroendocrine tumor pancreas extensive hepatic metastases |
chemotherapy interferon alpha |
male |
8 months |
5 |
66 years |
67 years |
69 years |
90Yttrium 10.5 GBq 177Lutetium 4 GBq |
acute leukemia |
neuroendocrine carcinoma pancreas hepatic and lymphatic metastases |
male |
1 month |
|
6 |
64 years |
64 years |
75 years |
177Lutetium 8 GBq |
RAEB-1 |
neuroendocrine tumor of the ileum lymphatic and hepatic metastases |
external beam radiation (50 Gy) |
female |
11months |
7 |
50 years |
68 years |
71 years |
90Yttrium 2.5 GBq 177Lutetium 17 GBq |
RAEB-1 |
neuroendocrine tumor of the ileum lymphatic and osseus metastases |
arterial hypertension obesity |
female |
43 months |
8 |
69 years |
69 years |
70 years |
90Yttrium 11.4 GBq 177Lutetium 6.5 GBq |
P-CLL |
neuroendocrine tumor of unknown primary hepatic metastases |
diabetes coronary heart disease arterial hypertension |
male |
96 months |
9 |
63 years |
63 years |
67 years |
90Yttrium 6.9 GBq 177Lutetium 27.3 GBq |
MDS (CMML-1) |
neuroendocrine tumor of the pancreas lymphatic and bone metastases |
chemotherapy external beam radiation |
male |
8 months |
10 |
69 years |
70 years |
70 years |
90Yttrium 6 GBq |
MDS RAEB-1 |
neuroendocrine tumor of the ileum extensive hepatic metastases |
female |
24 months |
|
11 |
45 years |
57 years |
66 years |
90Yttrium 16.6 GBq 177Lutetium 11 GBq |
MDS RAEB-1 |
neuroendocrine carcinoma of the duodenum hepatic metastases |
multiple chemotherapies Interferon alpha |
female |
16 months |
12 |
51 years |
52 years |
58 years |
90Yttrium 8 GBq 177Lutetium 22.5 GBq |
MDS RAEB-1 |
pancreatic neuroendocrine neoplasma Lymphatic, osseus and extensive hepatic metastases |
diabetes |
male |
8 months |
13 |
69 years |
70 years |
71 years |
90Yttrium 9.2 GBq 177Lutetium 12.7 GBq |
t-MDS RAEB-2 |
pancreatic neuroendocrine neoplasma osseus and extensive hepatic metastases |
chemotherapy |
male |
16 months |
14 |
58 years |
73 years |
79 years |
177Lutetium 22.5 GBq |
AML MO |
neuroendocrine neoplasma of ileum peritoneal and hepatic metastases |
chemotherapy arterial hypertension |
female |
10 months |
15 |
63 years |
63 years |
66 years |
177Lutetium 26.3 GBq |
MDS RAEB-1 |
neuroendocrine neoplasma of ileum Peritoneal, pleural, osseus and lymphatic metastases |
external beam radiation carcinoid heart disease |
female |
6 months |
16 |
49 years |
52 years |
57 years |
90Yttrium 9.5 GBq 177Lutetium 11.7 GBq |
MDS RAEB-1 |
pancreatic neuroendocrine neoplasma extensive lymphatic and hepatic metastases |
female |
16 months |
|
17 |
71 years |
73 years |
75 years |
90Yttrium 6.7 GBq 177Lutetium 6.9 GBq |
AML EGIL |
pancreatic neuroendocrine neoplasma extensive lymphatic, osseus and hepatic metastases |
arterial hypertension diabetes mellitus |
male |
24 months |
18 |
41 years |
46 years |
50 years |
90Yttrium 8 GBq 177Lutetium 17.5 GBq |
MDS RAEB-1 |
functionally active neuroendocrine neoplasm of ileum (carcinoid) extensive hepatic, bone, lymphatic and pleural metastases |
external beam radiation diabetes mellitus |
male |
4 months |
19 |
68 years |
69 years |
75 years |
90Yttrium 5.2 GBq 177Lutetium 27.6 GBq |
MDS RAEB-1 |
functionally active pancreatic neuroendocrine neoplasma (gastrinoma) lymphatic and extensive hepatic metastases |
chemotherapy transarterial chemoembolisation molecular therapy |
female |
9 months |
20 |
52 years |
59 years |
61 years |
177Lutetium 21.7 GBq |
MDS RAEB-2 |
typical carcinoid of the lung extensive hepatic and osseus metastases |
selective internal radiotherapy external beam radiation hypertension |
male |
6 months |
21 |
61 years |
64 years |
65 years |
177Lutetium 8.5 GBq |
MDS RAEB-1 AML |
functionally active neuroendocrine neoplasm of ileum (carcinoid) extensive hepatic metastases |
multiple TACE carcinoid heart disease arterial hypertension interferon therapy |
male |
16 months |
22 |
53 years |
53 years |
63 years |
90Yttrium 2 GBq 177Lutetium 48 GBq |
MDS RAEB-1 |
pancreatic neuroendocrine neoplasma extensive lymphatic, osseous and hepatic metastases |
chemotherapy |
female |
2 months |
Ø |
58.9 years |
62.9 years |
66.9 years |
21.7 GBq |
MDS unspecified 1 RAEB-1 12 RAEB-2 3 RAEB-T 1 AML 3 pCLL 1 |
Paraganglioma 1 SiNET 7 pNET (& duodenum) 9 CUP 2 Lung 1 Functionally active 2/21 |
ext. beam radiation 5 chemotherapy 8 carcinoid heart disease 4 interferon 3 art. hypertension 6 diabetes 3 |
12 female 10 male |
14.4 months |
Abbreviations: AML: acute myeloid leukemia; Ci: Curie; MDS: myelodysplastic syndrome; Gy: Gray; RAEB: refractory anemia with excess blasts; extensive hepatic metastases were defined as more than 50% infiltration of the liver data cut off for survival after diagnosis of MDS was 06-23-2016.
DISCUSSION
In our study, we report OS, PFS and adverse events of 1048 patients treated by PRRT over ten years. Limitations of our study is it retrospective nature and lack of control arms and random selectin for therapies. On the other hand, this is a large selection of patients treated in day-today practice yielding results useful in clinical settings. Just more than half of the cohort has survived during follow-up yielding exact overall survival data of patients and sub-groups. To ensure that all deceased patients were included in the analysis, several tracking mechanisms were employed at our institution as described above. Compared to historical controls [11], OS observed in our study is favorable implying that PRRT is effective. On the other hand, OS observed in our study may be mainly related to better differentiation of NEN since we included only patients with somatostatin receptor-expressing NEN. However, there appears to be no survival advantage of patients with somatostatin receptor-expressing mid-gut neuroendocrine tumors compared to non-expressing tumors [18].
Treatment with both radioisotopes resulted in better OS than with 177Lu or 90Y solely. These results were also reported in another study. Combined application of 90Y- and 177Lu-based PRRT in patients with different NENs resulted in a median OS of 66.1 months [19], which is almost identical to the median OS of 64 months in our study. These results indicate that personalized treatment adapting radioisotopes and dose to tumor load and location is superior than more restricted protocols. However, it should be noted that PRRT as monotherapy may be not as effective as combinatory protocols using radiosensitizers such as Everolimus (Afinitor®) or a combination of capecitabine and temozolomide achieving higher response rates compared to PRRT alone [20, 21].
Another molecular target, the serine-threonine kinase mTOR plays a pivotal role in pathogenesis of NENs and is effectively inhibited by Everolimus (Afinitor®). A prospective placebo-controlled multi-center trial in patients with pancreatic NENs has shown prolongation of PFS in patients treated with Everolimus (Afinitor®) versus patients treated with placebo (11.0 versus 4.6 months). Although median PFS in patients treated with Everolimus (Afinitor®) is lower in patients with pancreatic NENs than 20 months observed in our study, a recent analysis has shown that patients treated initially with Everolimus (Afinitor®) have a median OS of 44 months [22], which corresponds exactly to the median OS in our study. Albeit these studies are difficult to compare due to different design, these results corroborated clinical significance of hitting key molecular pathways in treatment of NENs.
OS was significantly determined by gender, age, radionuclides, number of previous therapies and origin of tumors in uni- and multi-variate analysis whereas functionality of NENs was significantly different only in uni-variate but not in multi-variate analysis. The patients with G1 tumors had the best OS, and so also did those with an age of a less than 40 years at diagnosis, patients treated with a combination of 90Y and 177Lu, and in NENs of small bowel. The OS of patients with G3 NENs was favorable. However, it is likely that this subgroup reflects mainly well differentiated neuroendocrine carcinomas due to the expression of somatostatin receptors. This group most likely reflects the newly created classification of neuroendocrine tumor G3, which is limited to NENs with a proliferation rate of up to 50% [3]. Indeed, OS of patients with a proliferation rate of more than 50% had a shorter OS than the whole group. These differences reflect biological characteristics of NENS, which should be included in clinical practice when planning PRRT and follow-up.
Follow-up in our patients was performed with 68Ga-SSTR PET/CT and EORTC criteria as well as RECIST [23]. Median PFS was 19 months in all patients, which is considerably shorter than the median of 41 months determined by RECIST 1.1 in a multi-institutional study in Germany involving 450 patients [11]. Shorter PFS by SSTR PET/CT follow-up reflects the increased sensitivity of this novel method. Although, 68Ga-SSTR PET/CT is very accurate for estimating the therapy response and especially for patient selection for PRRT, we postulate that PFS calculated solely on the basis of PET/CT be judged with caution. It should be kept in mind that EORTC criteria were primarily meant for the assessment of tumor response in lymphomas on 18F-FDG PET/CT. The different principle of assessment (SSTR status vs glucose metabolism) of NENs by this molecular imaging technique has to be born in mind. Probably, there is a need for newer response assessment criteria (for e.g., based on SUV tumor-to-spleen ratio, molecular tumor volume), designed on the basis of future prospective studies. In fact, median PFS determined by SSTR PET/CT using EORTC criteria does not mandatorily mirror OS in sub-groups with divergent prognosis. In fact, median PFS was not significantly different according to gender and age. Overall survival advantage of patient treated with a combination of 90Y and 177Lu (64 months) compared to sole usage of 177Lutetium (44 months) was not exactly reflected in median PFS times of 24 and 17 months, respectively, although showing a similar trend. Only exclusive usage of 90Y as radionuclide for PRRT resulted in significant lower PFS of 7 months compared to combined use (24 months). Likewise, OS advantage of patients with G1 NEN compared to G2 NEN (88 months versus 55 months) was not translated into PFS, which was 22 months for G1 tumors and 21 months for G2 NENs. However, G3 NENs had a lower OS and PFS. Number of previous therapies and functionality had no significant effect upon median PFS in multi-variate analysis. Median PFS in NENs of bronchial or unknown primary was significantly lower compared to pancreatic NENs in multi-variate analysis. Interestingly, the significantly lower median PFS of NENs of unknown primary (13 months versus 20 months in pancreatic NENs) was not reflected in median OS, which was not significantly different in both sub-groups (53 versus 44 months). We were able to include a subgroup of patients with somatostatin receptor positive medullary thyroid cancer in our analysis. Median OS of these patients was promising with 32 months median OS and 10.1 months PFS1, which should be examined in larger studies comparing standard treatments and peptide receptor radionuclide therapy.
These results indicate that progression documented by a sensitive method does not necessarily affect OS and should be considered in clinical practice. A criteria/scoring for somatostatin receptor PET/CT in NETs including for molecular response assessment, similar to for e.g., Krenning’s score for somatostatin receptor scintigraphy, needs to be developed and standardized.
Patients with NENs often require multi-disciplinary treatment with several lines of systemic therapy, often resulting in side effects depending on the therapy involved. Hemo- and nephrotoxicity are well known adverse events of PRRT [11, 24–27]. We monitored adverse events in our study by screening laboratory measurements during the 3692 treatment cycles. Here, we found grade 3 and 4 adverse events in less than 1% of PRRT cycles. There could be a possible underestimation of the real extent, since long term effects may be potentially missed. Therefore, we performed an additional search in all patient files and also obtained extensive information from the family physician to look into any hematological and nephrological toxicities during follow-up. We identified 22 subjects with leukemia/MDS and 5 additional patients (0.4%) with chronic kidney disease requiring hemodialysis after PRRT, whereas 3 patients had already been on hemodialysis before PRRT due to other reasons. Recently, incidence of leukemia and MDS after PRRT was reviewed by Bodei et al. [26] and Kesavan & Turner [25] reporting rates of 2% and 1.4%, respectively which corresponds to the 2.1% in our study. Risk factors identified in our study were chemotherapy and external beam radiation, as well as high tumor loads defined as involvement of more than 50% of the liver, multiple/multifocal (<10) bone and lymph node metatases. Development of leukemia/MDS after PRRT is associated with a limited mean overall survival of 14.4 months in our study. Rates of permanent severe nephrotoxicity of grade ≥4 have been described in as high as 9.2% in a study applying high doses of 90Y exclusively [24]. In contrast, severe nephrotoxicity is rare in patients treated by PRRT with 177Lu [28]. Incidence of severe renal impairments in our study was much lower, due to the mandatory renal protection followed as well as proper hydration of the patient after therapy.
MATERIALS AND METHODS
Patients
Between 2004 and 2014, 2294 patients were referred to Zentralklinik Bad Berka, Germany for diagnosis and treatment of NENs. Of these, 570 were found to be not eligible for PRRT and 676 patients, although eligible, did not undergo the treatment for various reasons (e.g. decision was reached in the interdisciplinary tumor board to perform first a different kind of therapy like transcatheter arterial chemoembolisation due to high functionality of the tumor). The criteria used to define eligibility for PRRT were in conformation with the published guidelines for PRRT [29] and included a Karnofsky of more than 60%, life expectancy of more than 6 months, somatostatin receptor positive NENs and adequate renal and bone marrow function. Excluded were patients not matching these criteria and somatostatin receptor negative NENs. Thus, PRRT was performed in 1048 patients and all of them were included in the intention to treat analysis (Figure 1). Before PRRT, patients were extensively informed about the procedure and the possible side effects. Written informed consent was obtained from all patients, which included permission for data storage and analysis. In 2007, the responsible ethical committee permitted retrospective and prospective data collection and analysis by the German neuroendocrine tumor registry, which was renewed in 2014. Data collection was performed in accordance with the registry. Decision to treat the patients by PRRT was taken by internal or external tumor boards. All patients were studied by SSTR PET/CT using 68Ga-DOTANOC, 68Ga-DOTATOC, or 68Ga-DOTATATE prior to PRRT, which was also used for patient follow-up. All patients were either progressive before PRRT, as determined by morphological imaging (CT or MRI) or by SSTR PET/CT, or were severely symptomatic due to extensive tumor mass or functional syndromes.
Radiopharmaceuticals
The DOTA-conjugated somatostatin analogues DOTA-TOC,-NOC and -TATE were labeled with 68Ga, 177Lu and 90Y in our radiopharmacy department in accordance with good medical practice. The radionuclide 68Ga was obtained in house from the 68Geranium-68Ga generator (Eckert and Ziegler GmbH, Berlin, Germany). A highly efficient NaCl-based labeling procedure for the radiopharmaceutical production of 68Ga labeled ligand-conjugate has been developed in our hospital [30, 31]. 177Lu and 90Y were obtained from different manufacturers. The labeling of DOTA-conjugated peptides with 177Lu (also applicable to 90Y) was performed according to a previously published method [32].
68Ga-SSTR PET/CT
PET/CT (until January 2014 with Siemens Biograph and since then with Biograph mCT Flow 64; Siemens Medical Solutions AG, Erlangen, Germany) was performed in all cases 45-90 minutes after the intravenous injection of 46-260 MBq of 68Ga-DOTANOC, -DOTATOC or DOTATATE. PET/CT images were acquired from the skull to the middle part of the thigh. Contrast-enhanced CT (ceCT, spiral CT using Biograph mCT Flow 64) was acquired after intravenous administration of 60-100 mL nonionic iodinated contrast. Images were evaluated by two experienced nuclear medicine specialists. Any area with intensity greater than background and that could not be explained by physiologic activity was considered to be indicative of tumor tissue. Maximum standardized uptake values (SUVmax) were obtained by drawing circular regions of interest (ROIs), which were automatically adapted (40% isocontour) to a 3D volume of interest using commercial software provided by the vendor.
Infusion and renal protection
Kidney protection was performed using an infusion of 1600 mL of a renoprotective amino-acid mixture of 5% lysine HCl and 10% L-arginine HCl. The infusion was started at least 30 min prior to administration of the therapeutic dose and continued for 4 h thereafter. The radiopharmaceutical was co-administered over 10–15 min by using a second infusion pump system. The activity to administer was individually chosen based on the uptake in the tumor lesions as shown by 68Ga-SSTR PET/CT (performed before each treatment cycle), kidney function (assessed using the following: tubular extraction rate determined by 99mTc-MAG3 scintigraphy, glomerular filtration determined by 99mTc-DTPA clearance, and serum creatinine), hematological reserve, previous treatments and general status of the patient (Karnofsky Performance Scale) [32–34]. Decision to use 90Y and/or 177Lu depended upon tumor mass, SUV and radionuclide availability [24]. Both radionuclides were used in subsets of patients sequentially (DUO) or in combination (TANDEM)[19, 35].
PRRT
The intention to treat analysis included 1048 patients, who received 3692 cycles of PRRT. Most patients received a combination of 90Y and 177Lu (49%), whereas 36% patients were only treated with 177Lu and 15% with 90Y, exclusively. PRRT with 90Y was performed in 371 cycles, applying a mean activity of 3.18 GBq, whereas with 177Lu was administered solely in 1043 cycles with a mean applied activity of 6.54 GBq. Dosimetry was performed after each cycle. Administered activity depended on patient and tumor-related factors like weight, renal function, tumor burden, clinical course and previous dosimetry. DUO-PRRT, i.e., application of 177Lu and 90Y in subsequent cycles in a particular patient, comprised 1235 cycles with 177Lu (mean administered activity 6.55 GBq) and 1008 cycles with 90Y (mean activity 3.36 GBq). TANDEM-PRRT, i.e., treatment with a combination of 177Lu and 90Y-labeled somatostatin analogues administered on the same day, was performed in 35 cycles with a mean implemented activity of 3.06 and 4.65 GBq, respectively.
Efficacy and safety
Restaging was performed with SSTR-PET/CT 3-6 months after PRRT. In case of stable disease or remission (complete or partial), the patient was restaged with SSTR PET/CT every 6-12 months until disease progression was evident on imaging. In addition, MRI in selected cases (allergy to iodinated contrast or poor detectability of liver metastases on CT scan) and routine sonography were performed for additional diagnostic evaluation. Resumption of PRRT after detection of progression after a therapy interruption of more than 6 months (2nd phase of PRRT) was performed in 470 patients. The next (second) resumption of PRRT (3rd phase of PRRT) after progression post 2nd phase of PRRT after an additional interval of more than 6 months was implemented in 184 patients. Laboratory parameters (erythrocytes, haemoglobin, platelets, leucocytes, creatinine, BUN, SGOT, SGPT, bilirubin, SAP, TSH, SGamma-GT and respective tumor markers) were evaluated prior to each cycle and at restaging. Renal function was monitored by tubular extraction rate (TER) using 99mTc-MAG3 renography and in addition the glomerular filtration rate (GFR) was determined using 99mTc-DTPA renography.
Method of patient documentation
A database was established in 2004 including more than 250 items for all patients with at least one cycle of PRRT. Out of this database, items were extracted to allow analysis of overall and progression-free survival according to age, sex, number and kind of pretreatments, grading, differentiation, methods of PRRT and administered radioactivity as well as origin of primary tumors and presence and kind of functional syndromes. Adverse events were determined according to CTCAE criteria version 4.03 by assessing all available laboratory measurements. In addition, adverse events were documented in an additional dataset by incoming patient reports during follow-up. Clinical data from the patient file as well as during patient registration by trained physician assistants. Data analysis was performed on anonymized data sets. All patients irrespective of tumor stage or prognosis, received an appointment for follow-up. When patients did not appear, their family and/or physicians were contacted. In addition, all patients were invited once yearly to a patient conference organized by our hospital. Date of death was reported by family physicians. No patient was lost to follow-up.
Statistical analysis
We assessed overall survival and progression-free survival with Kaplan-Meier curves and compared subgroups with one-sided log rank test. Univariate and multivariate analyses were done in R version 3.0.0 and rms package [36]. Uni-variate analysis was done by fitting an analysis of variance model to each variable under investigation [37]. Multi-variate analysis was conducted using Cox models [38]. Responses were evaluated with EORTC criteria (PET component of PET/CT) as well as by RECIST (CT component of PET/CT or MRI) [39]. Adverse events were assessed from lab data and graded according to CTCAE criteria.
CONCLUSIONS
Treatment of patients with NENs by PRRT has demonstrated favorable and clinical significant median OS and PFS with minimal acceptable side effects, observed during a treatment period of a decade. Follow-up by somatostatin receptor PET/CT is feasible and effective, however, progression documented by this sensitive method should be always evaluated in clinical context.
Abbreviations
177Lu: 177Lutetium; 68Ga: 68Gallium; 68Ga-DOTANOC: 68Gallium [DOTA, 1-Nal3]-octreotide (DOTANOC); 68Ga-DOTATATE: 68Gallium [DOTA, Tyr3]-octreotate; 90Y: 90Yttrium; 99mTc-DTPA: Technetium-99mTc-diethylenetriaminepentaacetic acid; 99mTc-MAG3: Technetium-99m (99mTc) mercaptoacetyltriglycine; Bq: Bequerel; BUN: blood urea nitrogen; CTCAE: Common Terminology Criteria for Adverse Events; DOTATOC: (DOTA(0)-Phe(1)-Tyr(3))octreotid; EORTC: European Organisation for Research and Treatment of Cancer; MRI: magnetic resonance imaging; NEN: neuroendocrine neoplasms; OS: overall survival; PET/CT: positron-emission tomography/computed tomography; PFS: progression-free survival; PRRT: peptide receptor radionuclide therapy; SAP: serum alkaline phosphatase; SGamma-GT: serum γ-Glutamyltransferase; SGOT: serum glutamic-oxaloacetic transaminase; SGPT: glutamic-pyruvic transaminase; SSTR: somatostatin receptor; SUV: standardized uptake values; TER: tubular extraction rate; TSH: tyroidea stimulating hormone: WHO: world health organization.
Author contributions
R.P.B., H.R.K. and D.H. wrote the manuscript, K. N., H. F., A.J., and P.L. analysed data, D.K., D.M., V.P., M.H., F.R., and A.S. collected the data and patient characteristics.
ACKNOWLEDGMENTS
We are indebted to all patients and their families during the conduct of the study for their continuous trust and support.
CONFLICTS OF INTEREST
R.P. Baum received honoraria from Ipsen Pharma, ROTOP Pharmaka, OctreoPharm Sciences GmbH and Advanced Accelerator Applications outside the submitted work; H.R. Kulkarni has nothing to disclose; K. Niepsch has nothing to disclose; D. Kaemmerer received travel support by the companies IPSEN Pharma GmbH and Pfizer; D. Mueller is inventor of the herein mentioned NaCl based 68Ga labeling method. In some countries, patent applications have been registered; V. Prasad has nothing to disclose; M. Hommann has nothing to disclose; F. Robiller has nothing to disclose; A. Singh has nothing to disclose; H. Franz reports personal fees from Zentralklinik Bad Berka, during the conduct of the study; A. Jochems has nothing to disclose; P. Lambin is member is the Scientific advisory board of the companies DNAmito and Oncoradiomics and is inventor of several radiomics patents; D. Hörsch reports personal fees from Lexicon Pharma Inc, grants and personal fees from Ipsen Pharma Inc, during the conduct of the study; grants and personal fees from Novartis Pharma Inc, personal fees from Pfizer Pharma Inc, grants and personal fees from Ipsen Pharma Inc, personal fees from Lexicon Pharma Inc, outside the submitted work.
FUNDING
This study was designed by academic investigators. Data was collected by trained medical documentaries. Collection and analysis was made possible by a generous grant of the Wilhelm and Ingeburg Dinse memory foundation in Hamburg.
REFERENCES
1. Merola E, Rinzivillo M, Cicchese N, Capurso G, Panzuto F, Delle Fave G. Digestive neuroendocrine neoplasms: A 2016 overview. Dig Liver Dis. 2016; 48:829-835.
2. Rindi G, Petrone G, Inzani F. The 2010 WHO classification of digestive neuroendocrine neoplasms: a critical appraisal four years after its introduction. Endocr Pathol. 2014; 25:186-192.
3. Klöppel G. Neuroendocrine Neoplasms: Dichotomy, Origin and Classifications. Visc Med. 2017; 33:324-330.
4. de Herder WW, Rehfeld JF, Kidd M, Modlin IM. A short history of neuroendocrine tumours and their peptide hormones. Best Pract Res Clin Endocrinol Metab. 2016; 30:3-17.
5. Rinke A, Krug S. Neuroendocrine tumours - Medical therapy: Biological. Best Pract Res Clin Endocrinol Metab. 2016; 30:79-91.
6. Rinke A, Wittenberg M, Schade-Brittinger C, Aminossadati B, Ronicke E, Gress TM, Müller HH, Arnold R; PROMID Study Group. Placebo-Controlled, Double-Blind, Prospective, Randomized Study on the Effect of Octreotide LAR in the Control of Tumor Growth in Patients with Metastatic Neuroendocrine Midgut Tumors (PROMID): Results of Long-Term Survival. Neuroendocrinology. 2017; 104:26-32.
7. Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, Cadiot G, Wolin EM, Capdevila J, Wall L, Rindi G, Langley A, Martinez S, et al; CLARINET Investigators. Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study. Endocr Relat Cancer. 2016; 23:191-199.
8. Deppen SA, Liu E, Blume JD, Clanton J, Shi C, Jones-Jackson LB, Lakhani V, Baum RP, Berlin J, Smith GT, Graham M, Sandler MP, Delbeke D, et al. Safety and Efficacy of 68Ga-DOTATATE PET/CT for Diagnosis, Staging, and Treatment Management of Neuroendocrine Tumors. J Nucl Med. 2016; 57:708-714.
9. Bodei L, Kwekkeboom DJ, Kidd M, Modlin IM, Krenning EP. Radiolabeled Somatostatin Analogue Therapy Of Gastroenteropancreatic Cancer. Semin Nucl Med. 2016; 46:225-238.
10. Brabander T, Teunissen JJ, Van Eijck CH, Franssen GJ, Feelders RA, de Herder WW, Kwekkeboom DJ. PRRT of neuroendocrine tumours. Best Pract Res Clin Endocrinol Metab. 2016; 30:103-114.
11. Hörsch D, Ezziddin S, Haug A, Gratz KF, Dunkelmann S, Miederer M, Schreckenberger M, Krause BJ, Bengel FM, Bartenstein P, Biersack HJ, Pöpperl G, Baum RP. Effectiveness and side-effects of PRRT for neuroendocrine neoplasms in Germany: A multi-institutional registry study with prospective follow-up. Eur J Cancer. 2016; 58:41-51.
12. Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, Mittra E, Kunz PL, Kulke MH, Jacene H, Bushnell D, O’Dorisio TM, Baum RP, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017; 376:125-135.
13. Haug AR, Auernhammer CJ, Wängler B, Schmidt GP, Uebleis C, Göke B, Cumming P, Bartenstein P, Tiling R, Hacker M. 68Ga-DOTATATE PET/CT for the early prediction of response to somatostatin receptor-mediated radionuclide therapy in patients with well-differentiated neuroendocrine tumors. J Nucl Med. 2010; 51:1349-1356.
14. Ezziddin S, Lohmar J, Yong-Hing CJ, Sabet A, Ahmadzadehfar H, Kukuk G, Biersack HJ, Guhlke S, Reichmann K. Does the pretherapeutic tumor SUV in 68Ga DOTATOC PET predict the absorbed dose of 177Lu octreotate? Clin Nucl Med. 2012; 37:e141-147.
15. Sainz-Esteban A, Prasad V, Schuchardt C, Zachert C, Carril JM, Baum RP. Comparison of sequential planar 177Lu-DOTA-TATE dosimetry scans with 68Ga-DOTA-TATE PET/CT images in patients with metastasized neuroendocrine tumours undergoing PRRT. Eur J Nucl Med Mol Imaging. 2012; 39:501-511.
16. Kratochwil C, Stefanova M, Mavriopoulou E, Holland-Letz T, Dimitrakopoulou-Strauss A, Afshar-Oromieh A, Mier W, Haberkorn U, Giesel FL. SUV of [68Ga]DOTATOC-PET/CT Predicts Response Probability of PRRT in Neuroendocrine Tumors. Mol Imaging Biol. 2015; 17:313-318.
17. Baum RP, Kluge AW, Kulkarni H, Schorr-Neufing U, Niepsch K, Bitterlich N, van Echteld CJ.[(177)Lu-DOTA](0)-D-Phe(1)-Tyr(3)-Octreotide ((177)Lu-DOTATOC) For Peptide Receptor Radiotherapy in Patients with Advanced Neuroendocrine Tumours: A Phase-II Study. Theranostics 2017; 13:501-510.
18. Wang YZ, Beyer DT. Does receptor status impact survival of patients with mid-gut neuroendocrine tumors. J Clin Oncol. 2016; 34:227.
19. Villard L, Romer A, Marincek N, Brunner P, Koller MT, Schindler C, Ng QK, Mäcke HR, Müller-Brand J, Rochlitz C, Briel M, Walter MA. Cohort study of somatostatin-based radiopeptide therapy with [(90)Y-DOTA]-TOC versus [(90)Y-DOTA]-TOC plus [(177)Lu-DOTA]-TOC in neuroendocrine cancers. J Clin Oncol. 2012; 30:1100-1106.
20. Claringbold PG, Turner JH. NeuroEndocrine Tumor Therapy with Lutetium-177-octreotate and Everolimus (NETTLE): A Phase I Study. Cancer Biother Radiopharm. 2015; 30:261-269.
21. Claringbold PG, Turner JH. Pancreatic Neuroendocrine Tumor Control: Durable Objective Response to Combination 177Lu-Octreotate-Capecitabine-Temozolomide Radiopeptide Chemotherapy. Neuroendocrinology 2016; 103:32-439.
22. Yao JC, Pavel M, Lombard-Bohas C, Van Cutsem E, Lam D, Kunz T, Brandt U, Capdevila J, De Vries EGE, Tomassetti P, Hobday T, Pommier R, Öberg K. Everolimus for the Treatment of Advanced Pancreatic Neuroendocrine Tumors: Overall Survival and Circulating Biomarkers From the Randomized, Phase III RADIANT-3 Study. J Clin Oncol. 2016; 34:3906-3913.
23. Aras M, Erdil TY, Dane F, Gungor S, Ones T, Dede F, Inanir S, Turoglu HT. Comparison of WHO, RECIST 1.1, EORTC, and PERCIST criteria in the evaluation of treatment response in malignant solid tumors. Nucl Med Commun. 2016; 37:9-15.
24. Imhof A, Brunner P, Marincek N, Briel M, Schindler C, Rasch H, Mäcke HR, Rochlitz C, Müller-Brand J, Walter MA. Response, survival, and long-term toxicity after therapy with the radiolabeled somatostatin analogue [90Y-DOTA]-TOC in metastasized neuroendocrine cancers. J Clin Oncol 2011; 29:2416-2423.
25. Kesavan M, Turner JH. Myelotoxicity of PRRT of Neuroendocrine Tumors: A Decade of Experience. Cancer Biother Radiopharm. 2016; 31:189-198.
26. Bodei L, Modlin IM, Luster M, Forrer F, Cremonesi M, Hicks RJ, Ezziddin S, Kidd M, Chiti A. Myeloid neoplasms after chemotherapy and PRRT: myth and reality. Endocr Relat Cancer. 2016; 23:C1-7.
27. Sabet A, Ezziddin K, Pape UF, Ahmadzadehfar H, Mayer K, Pöppel T, Guhlke S, Biersack HJ, Ezziddin S. Long-term hematotoxicity after PRRT with 177Lu-octreotate. J Nucl Med. 2013; 54:1857-1861.
28. Bergsma H, Konijnenberg MW, van der Zwan WA, Kam BL, Teunissen JJ, Kooij PP, Mauff KA, Krenning EP, Kwekkeboom DJ. Nephrotoxicity after PRRT with (177)Lu-DOTA-octreotate. Eur J Nucl Med Mol Imaging. 2016; 43:1802-1811.
29. Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Hörsch D, O’Dorisio MS, O’Dorisio TM, Howe JR, Cremonesi M, Kwekkeboom DJ. The joint IAEA, EANM, and SNMMI practical guidance on PRRT (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2013; 40:800-816.
30. Mueller D, Klette I, Baum RP, Gottschaldt M, Schultz MK, Breeman WA. Simplified NaCl based (68)Ga concentration and labeling procedure for rapid synthesis of (68)Ga radiopharmaceuticals in high radiochemical purity. Bioconjug Chem. 2012; 23:1712–1717.
31. Mueller D, Breeman WAP, Klette I, Gottschaldt M, Odparlik A, Baehre M, Schultz MK. Radiolabeling of DOTA-like conjugated peptides with generator-produced Ga-68 and using NaCl-based cationic elution method. Nat Protoc. 2016; 11:1057–1066.
32. Wehrmann C, Senftleben S, Zachert C, Müller D, Baum RP. Results of individual patient dosimetry in PRRT with 177Lu DOTA-TATE and 177Lu DOTA-NOC. Cancer Biother Radiopharm 2007; 22:406-416.
33. Kulkarni HR, Schuchardt C, Baum RP. PRRT with (177)Lu labeled somatostatin analogs DOTATATE and DOTATOC: contrasting renal dosimetry in the same patient. Recent Results Cancer Res. 2013; 194:551-559.
34. Schuchardt C, Kulkarni HR, Prasad V, Zachert C, Müller D, Baum RP. The Bad Berka dose protocol: comparative results of dosimetry in PRRT using (177)Lu-DOTATATE, (177)Lu-DOTANOC, and (177)Lu-DOTATOC. Recent Results Cancer Res. 2013;194:519-36.
35. Baum RP, Kulkarni HR, Carreras C. Peptides and receptors in image-guided therapy: theranostics for neuroendocrine neoplasms. Semin Nucl Med. 2012; 42:190-207.
36. R Core Team. R: A language and environment for statistical computing. R foundation for Statistical Computing, Vienna, Austria. 2012. Available online at http://www.R-project.org/.
37. Chambers JM, Freeny AE, Heiberger RM. Analysis of variance; designed experiments. In Statistical Models Eds JM Chambers & TJ Hastie. Pacific Grove, California: Wadsworth & Brooks/Cole. 1992.
38. Cox DR. Regression models and life tables (with discussion). Journal of the Royal Statistical Society, Series B. 1972; 34:187–220.
39. Young H, Baum R, Cremerius U, Herholz K, Hoekstra O, Lammertsma AA, Pruim J, Price P. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur J Cancer 1999; 35:1773-1782.

