INTRODUCTION
The androgen receptor (AR) is a member of the nuclear receptor family of proteins that acts as ligand-inducible transcription factors, which comprises three main functional domains: the N-terminal transcriptional domain, the DNA binding domain (DBD) and the ligand binding domain (LBD) [1]. Testosterone and its more potent metabolite, dihydrotestosterone (DHT), can bind ar. Upon androgen binding, the ar forms a dimer, translocates to nucleus and recruits a wide variety of co-regulators to modulate androgen target genes [2]. Clinical disorders related to ar dysfunction include testicular feminization mutation syndrome (Tfm), prostate cancer and Kennedy’s disease [3–6]. In addition, the Tfm syndrome has been observed in other mammals [7–9].
To elucidate the molecular basis of ar-related disorders, in the last decade, generation and characterization of ar knockout mouse models (ARKO) revealed key roles of the ar in male and female reproduction [10–12]. Because the mammalian ar gene is located on the X chromosome, which is critical for male fertility, it is impractical to generate an ARKO mouse line using conventional gene targeting. To date, most data about ARKO mice are obtained from a Cre-loxP strategy for conditional KO, including global and cell-specific androgen receptor KO mice [11, 13–28]. The ar mediates androgen actions on different parts of the reproductive system at different stages of development. In males, ar-mediated androgen activity is involved in differentiation of efferent duct system for germ cells [29], spermatogenesis [25], sex and reproductive behavior, and secondary sex characteristics [12, 30, 31]. In females, the ar-mediated androgen actions are important for reproductive development and function, including folliculogenesis, and uterine and mammary gland development [16, 32–35].
The failure of product amplification from lampreys using many combinations of degenerate ar primers suggests that ar gene may be specific to jawed vertebrates [36]. A jawed vertebrate such as teleost fish have the ar gene, and androgens are involved in secondary sex characteristics and behavior [37, 38], spermatogenesis [39, 40], and Leydig cell androgen production [41]. Moreover, fish appear to be more sensitive to androgen with respect to sex differentiation because fully functional female-to-male sex reversal can be induced by exposure of juvenile or adult fish to androgens [42, 43].
Zebrafish (Danio rerio), as one of the model organisms, their ar gene has been isolated and its expression patterns and biochemical characteristics have been described [44–46]. However, whether zebrafish ar has effects on sex determination or gonad development is still largely unknown. Molecular control of sex determination and gonad differentiation in zebrafish appears to be complex and variable across domesticated strains versus wild populations [16, 47–49]. Zebrafish belong to undifferentiated gonochoristic fishes: during the juvenile period, all individuals develop undifferentiated ovary-like gonads containing immature oocytes. Between 20 and 30 days post fertilization (d.p.f.), some immature oocytes develop into ovaries, while the other immature oocytes degenerate and acquire testis morphology [50–52].
In this study, we used CRISPR/Cas9 technology to investigate whether ar plays a role in reproductive development in zebrafish. Loss of ar caused progressive loss of spermatogenesis and ovarian function, so we suggest that the ar is key to spermatogenesis and maintenance of ovarian function.
RESULTS
Targeted disruption of ar produces more female zebrafish and increases weight in male and female zebrafish
ar is evolutionarily conserved among zebrafish, mouse, rat and humans, particularly in its DNA-binding domain (DBD) and ligand-binding domain (LBD) (Supplementary Figure 1). After micro-injecting synthesized sgRNA and Cas9 mRNA into one-cell stage embryos, we initially used HMA (Heteroduplex mobility assay) to measure the efficiency of sgRNA/Cas9-mediated ar disruption in the F0 generation and then conducted sequencing confirmation of the F1 generation. After screening, we obtained two mutants in the ar gene (ar ihb1225/ihb1225 and ar ihb1226/ihb1226, respectively; Figure 1A and Supplementary Figure 2). In these two mutants, two truncated peptides were predicted which lacked most of the LBD (Figure 1A). Reduced ar mRNA in testes and ovaries was confirmed in ar ihb1225/ihb1225 adult ar-null zebrafish (Figure 1B), which might be due to non-sense mediated decay.
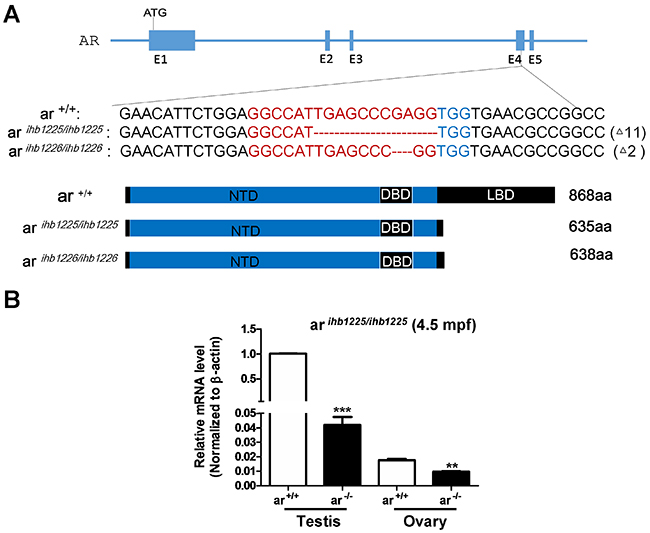
Figure 1: Generation of ar-null zebrafish via CRISPR/Cas9 technology. (A) Scheme of the genomic structure of zebrafish ar, the sequence information of targeting sites and the predicted protein products of ar in the mutants ar ihb1225/ihb1225 and ar ihb1226/ihb1226. gRNA binding site sequences are highlighted in red font. ar +/+, wild-type; ar ihb1225/ihb1225, homozygous mutant line 1 (ar -/-) with 11-bp nucleotides (TGAGCCCGAGG) deletion in the Exon 4 of ar; ar ihb1226/ihb1226, homozygous mutant line 2 (ar -/-) with 2-bp nucleotides (GA) deletion in the Exon 4 of ar. (B) Expression of ar in testes or ovaries from the wildtype (ar +/+) and the ar-null mutant 1 (ar-/-) zebrafish (4.5 mpf; n=3, respectively). Mpf, month post fertilization.
From the embryonic stage to adulthood, ar heterozygotes (ar+/-) were indistinguishable from wildtype siblings (ar+/+) with ar ihb1225/ihb1225 or ar ihb1226/ihb1226 background. To obtain ar homozygotes (ar-/-), we mated ar+/- × ar+/-. For offspring of ar+/- × ar+/-, numbers of ar+/+, ar+/- and ar-/- were determined by Mendel’s ratio (1:2:1) (Figure 2A), which suggested that knockout of ar in zebrafish had no effect on the survival. However, at the adult stage (from 3.5 mpf to 4.5 mpf), female ratio of ar -/- was higher than that of ar +/+ or ar+/- (Figure 2B). Notably, male and female ar-/- weights were greater than wildtype siblings (ar+/+) (Figure 2C). Dissection revealed more fatty abdomens in ar-/- males (Supplementary Figure 3). Therefore, targeted disruption of ar produces more female zebrafish and increases their weight.
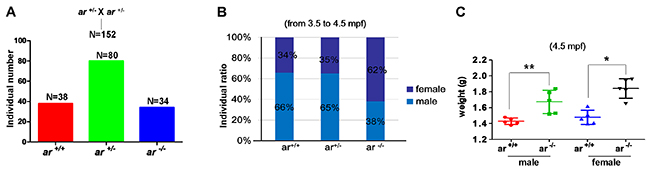
Figure 2: Targeted disruption of ar produces more female zebrafish and increases weight increased in male and female zebrafish. (A) The individual number of three genotypes from the offspring of intercrossing between ar heterozygous (ar +/-). (B) Comparison of sex ratio among wildtype (ar +/+), heterozygous (ar +/-) and homozygous (ar -/-) (M1)zebrafish similarly raised. Counts were from 3.5 to 4.5 mpf zebrafish. (C) Weights compared between wildtype (ar +/+) and homozygous (ar -/-; M1) male and female zebrafish at 4.5 mpf. Mpf, month post fertilization.
Ar-null male zebrafish display female secondary sex characteristics
Adult wildtype and heterozygous males (ar +/+ and ar +/-) had deep yellow pigmentation of the anal fin and breeding tubercles (BT) (or epidermal tubercles, ET) on pectoral fins (red arrows) (Figure 3A and 3B). However, ar-null males (ar -/-)(ar ihb1225/ihb1225) had light yellow pigmentation of the anal fin and no BTs of the pectoral fin (Figure 3C). In addition, ar-null male zebrafish (ar -/-)(ar ihb1225/ihb1225) had a female-like appearance with larger abdomens (Figure 3C). Mating activity of ar -/- male and wildtype females were not observed so we speculate this to mean that ar-null male zebrafish had female secondary sex characteristics and a female-like appearance.

Figure 3: ar-null male zebrafish have female secondary sex characteristics. (A-C) Wildtype (ar+/+) and heterozygous (ar+/-) male adult zebrafish have deep yellow pigmentation of the anal fin and breeding tubercles (BT) in the pectoral fin (red arrows); ar-null male zebrafish (ar -/-; ar ihb1225/ihb1225) had light yellow pigmentation of the anal fin and no pectoral BTs; ar-null male zebrafish (ar-/-; ar ihb1225/ihb1225) also had a female-like appearance and a larger abdomen. Mpf, month post fertilization.
Targeted disruption of ar in zebrafish causes male infertility with defective spermatogenesis
To understand the effect of ar on male reproductive development, we immunohistochemically and histologically studied testes. Grossly, testes in ar-/- adult males were smaller and more transparent compared to ar +/+ or ar +/- male siblings (Figure 4A-4C). Notably, a significant decrease of the gonadosomatic index (GSI) was found in ar-/- testes (Figure 4D). Sperm motility assessment showed that the motility of sperms isolated from ar -/- testes was much lower than that of sperms isolated from ar +/+ testes (Figure 4E). At 1.5 mpf (months post fertilization), no obvious difference were noted between ar -/- and their wildtype sibling’s (ar +/+) testes (Supplementary Figure 5A and 5B). However, at 70 dpf (days post fertilization), compared with the wildtype sibling testes, spermatogenesis in ar -/- testes was delayed and had more spermatogonia (SG) and fewer spermatocytes (SC), but no spermatozoa (SZ)(Supplementary Figure 5C and 5D). Apparently, spermatogenesis in ar-/- testes was arrested at the first meiosis. At 3 mpf, spermatogenetic cysts were smaller in ar -/- testes, the proportion of SC was increased, but fewer SZ. Spermatogenesis was arrested at the second meiosis (Figure 4F and 4G). In addition, the size of spermatogonia (SG) was bigger and the number of Sertoli cells was increased in ar -/- testes (Figure 4H and 4I). At 6 mpf, ar-/- testes degenerated and were loose (Supplementary Figure 5E and 5F).
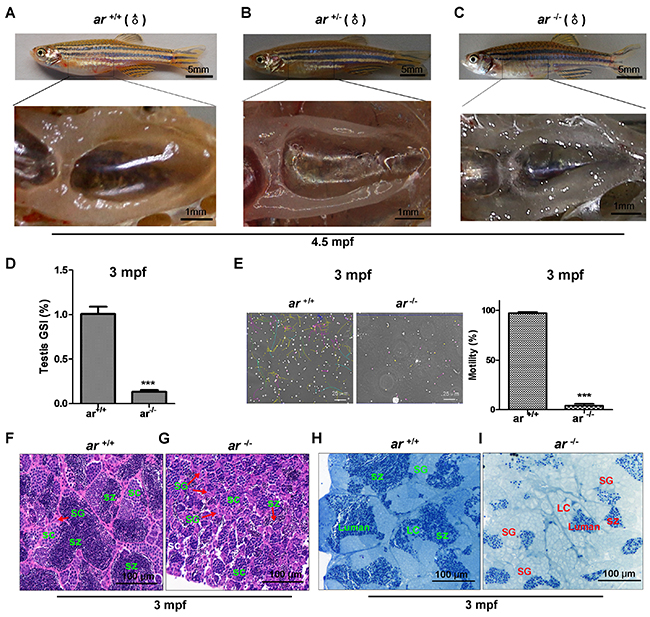
Figure 4: Targeted disruption of ar in zebrafish causes male infertility due to defective spermatogenesis. (A-C) Gross appearance of testes from wildtype (ar +/+), heterozygous (ar +/-) and homozygous (ar -/-) (ar ihb1225/ihb1225) zebrafish. Testes of ar -/- male zebrafish was smaller and more transparent compared with that in their ar +/+ or ar +/- male siblings. (D) Gonadosomatic index (GSI) in ar+/+ and ar-/- male zebrafish. (E) Sperm motility evaluation under a dark-phase microscope. (F, G) Histology of testes from wildtype (ar +/+) and homozygous (ar -/-) (ar ihb1225/ihb1225) zebrafish at 3 mpf. Compared with wildtype sibling testes, spermatogenesis in ar -/- testes was delayed as indicated by more spermatogonia (SG), fewer spermatocytes (SC) and fewer spermatozoa (SZ); the spermatogenetic cysts were smaller; and spermatogenesis was arrested at the second meiosis. (H, I) Toluidine blue staining testes from wildtype (ar +/+) and homozygous (ar -/-) (ar ihb1225/ihb1225) zebrafish at 3 mpf. Compared with wildtype sibling testes, the size of spermatogonia (SG) was bigger and the number of Sertoli cells was increased in ar -/- testes. Leydig cells (LC) and lumen are indicated. Dpf, days post fertilization; Mpf, months post fertilization.
Subsequently, we determined expression of germ-cell marker, vasa using immunofluorescent staining, which clearly marks germ cells at different stages as indicated by decreased intensity of fluorescence from primordial germ cell (PGC) to mature gametocyte [53]. At 70 dpf, ar +/+ testes developed normally as indicated by spermatid (SPD) filling, but, spermatogenesis in ar -/- testes was delayed as confirmed by greater spermatogonia (SG), primary spermatocytes (PSP) and secondary spermatocytes (SSP) (Figure 5A). At 4.5 mpf, many spermatoza (SZ) were filled in ar +/+ testes, but ar -/- testes remained filled with spermatogonia (SG), primary spermatocyte (PSP) and secondary spermatocyte (SSP) (Figure 5B). Although it appeared that some spermatozoa (SZ) were present in ar -/- testes, fewer sperm could be isolated from ar -/- males. When homogenous adult ar -/- testes were mixed with eggs from wildtype females, few fertilized eggs were observed (data not shown). These phenotypes were also observed in another ar mutant line (M2) (Supplementary Figure 6A). Thus, ar -/- testes are almost dysfunctional.
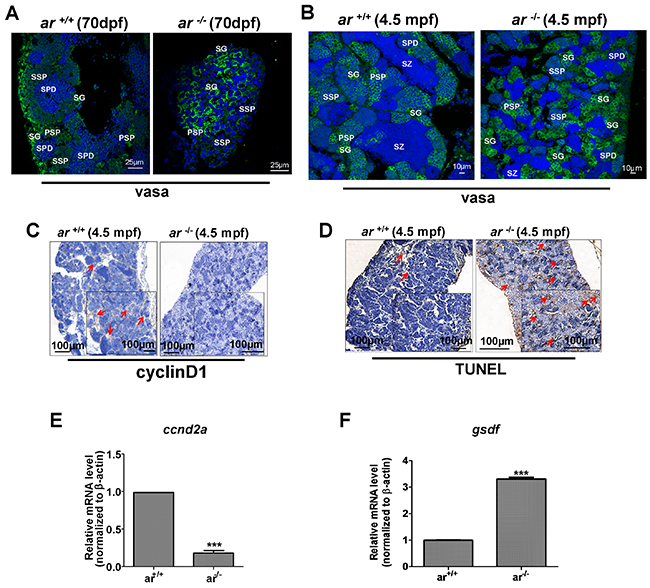
Figure 5: Proliferating germ cells were reduced, but apoptotic germ cells increased in ar-null zebrafish testes. (A, B) Immunofluorescent staining with anti-vasa antibody identified different types of spermatogenic cells in ar -/- testes and wildtype sibling (ar +/+) testes at 70 dpf or 4.5 mpf, respectively. SG, spermatogonia; SZ, spermatozoa; PSP, primary spermatocyte; SSP, secondary spermatocyte; SPD, spermatid. (C) Immunohistochemistry with anti-cyclin D1 antibody indicated that proliferating germ cells were reduced in ar -/- testes compared with wildtype (ar+/+) siblings at 4.5 mpf. (D) TUNEL assay indicated increased apoptosis of germ cells in ar -/- testes compared with wildtype (ar +/+) siblings at 4.5 mpf. Mpf, months post fertilization. (E, F) Expression of ccnd2a (cyclin d2a) was down-regulated, but expression of gsdf was up-regulated in ar-/- (ar ihb1225/ihb1225) testes compared with wildtype (ar +/+) sibling testes (4.5 mpf, n=3, respectively).
To determine the causes underlying the effects of ar on spermatogenesis, we quantified expression of cell proliferation marker, cyclin D1 by immunohistochemistry. Figure 5C shows that cell proliferation was greater in ar +/+ testes compared with ar -/- testes. In addition, TUNEL assay confirmed more germ cells in ar -/- testes were apoptotic (Figure 5D). Therefore, targeted disruption of ar causes progressive loss of spermatogenesis in zebrafish.
To understand how ar affects zebrafish spermatogenesis, we determined expression of two genes associated with meiosis. Figure 5E and 5F shows that expression of ccnd2a (cyclin d2a) was down-regulated, but expression of gsdf (gonadal somatic cell derived factor), a growth factor expressed in Sertoli cells of testes, was up-regulated in ar -/- (ar ihb1225/ihb1225) testes compared with that of wildtype (ar +/+) sibling testes (4.5 mpf, n=3, respectively) [54, 55].
Targeted disruption of ar in zebrafish causes premature ovarian failure during growth
To understand the effect of ar on female reproductive development, histology and immunohistochemistry were used to study ovaries. At 4.5 mpf, gross appearance of ar -/- female was indistinguishable from wildtype (ar+/+) and heterozygous (ar +/-) sibling females (Figure 6A-6C). However, ovaries of ar-/- females were smaller with fewer eggs at 4.5 mpf (Figure 6C). Consistently, a significant decrease of the gonadosomatic index (GSI) was found in ar -/- ovaries (Figure 6D). From 3 mpf to 4.5 mpf, the average egg number per female in ar -/- was decreased compared with that of wildtype siblings (Supplementary Figure 4A and 4B). After mating with wildtype males (AB line), less fertilization occurred in ar -/- eggs (Figure 6E). At 1.5 mpf, no obvious difference was noted between wildtype (ar +/+) and homozygous (ar -/-) (ar ihb1225/ihb1225) ovaries (Figure 6F and 6G). At 70 dpf, in wildtype ovaries (ar+/+), most oocytes developed to stage III and IV; but in ovaries of ar -/-, most oocytes developed to stage I or II, and some to III and IV (Figure 6H and 6I). At 3 mpf, in wildtype ovaries (ar +/+), most oocytes developed to stage IV; but in ovaries of the homozygous (ar -/-), most oocytes developed to stage II and follicles were partially atretic (Figure 6J and 6K). At 4 mpf, in the homozygous (ar -/-) ovaries, atretic follicles were observed and degeneration occurred (Figure 6L and 6M). At 3 to 4 mpf, ar -/- ovaries prematurely failed, and most follicles were arrested at stage I or II as indicated by female sub-fertility. In 5 mpf, in ar -/- homozygous ovaries (ar -/-), no mature oocytes were observed and severe degeneration occurred (30%, n=30). The ar -/- females at this age were completely infertile. These phenotypes were also observed in another ar mutant line (ar ihb1226/ihb1226) (Supplementary Figure 6B). Thus, targeted disruption of ar causes premature ovarian failure.
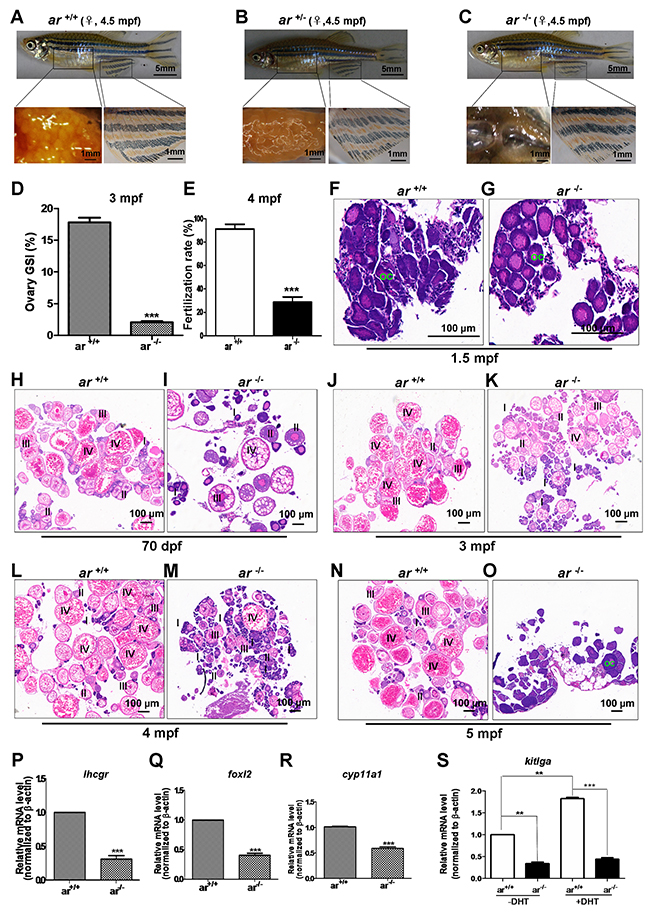
Figure 6: Targeted disruption of ar in zebrafish causes premature ovarian failure during growth. (A-C) Gross appearance and ovaries from wildtype (ar +/+), heterozygous (ar +/-) and homozygous (ar -/-) (ar ihb1225/ihb1225) female zebrafish. At 4.5 mpf, the gross appearance of ar -/- (ar ihb1225/ihb1225) female zebrafish was indistinguishable from wildtype siblings (ar +/+) or their heterozygous sibling (ar +/-), but the ovaries of ar -/- female were smaller with fewer eggs and became transparent compared with wildtype sibling (ar +/+) or heterozygous siblings (ar +/-). (D) Gonadosomatic index (GSI) in ar +/+ and ar -/- female zebrafish. (E) Fertilization rate of eggs from wildtype (ar +/+) and homozygous (ar -/-) female zebrafish at 4mpf. (F, G) Histology of ovaries from wildtype (ar +/+) and homozygous (ar -/-) (ar ihb1225/ihb1225) zebrafish at 1.5 mpf. No obvious difference between ar -/- and ar +/+ ovaries. OC, Oocyte. (H, I) Histology of ovary from the wildtype (ar +/+) and homozygous (ar -/-) (ar ihb1225/ihb1225) zebrafish at 70 dpf. Ovaries of wildtype (ar +/+), most oocytes developed to stage III and IV; but more ovaries of homozygous (ar -/-) (M1) developed to stage II or I, some developed to stage III and IV. (J, K) Histology of ovaries from wildtype (ar +/+) and homozygous (ar -/-) zebrafish at 4 mpf. In ovaries of wildtype (ar +/+), most oocytes developed to stage IV, but for ovaries of homozygous (ar -/-), most oocytes developed to stage II and partial degeneration occurred. (L, M) Histology of ovaries from wildtype (ar+/+) and homozygous (ar -/-) zebrafish at 4 mpf. In ovaries of homozygous (ar -/-), follicles atresia and degeneration occurred. (N, O) Histology of ovaries from wildtype (ar +/+) and homozygous (ar -/-) zebrafish at 5 mpf. In ovaries of homozygous (ar -/-), no follicles were observed and severe degeneration occurred (30%, n = 30). (P) lhcgr (luteinizing homone/choriogonadotropin receptor) expression in ovaries of wildtype (ar+/+) and homozygous (ar-/-) zebrafish at 4.5 mpf. (Q) foxl2 (forkhead box L2) expression in ovaries of wildtype (ar +/+) and homozygous (ar -/-) zebrafish at 4.5 mpf. (R) cyp11a1 (p450 side chain cleavage enzyme) expression in ovaries of wildtype (ar+/+) and homozygous (ar -/-) zebrafish at 4.5 mpf. (S) kitlga expression in ovaries of wildtype (ar+/+) and homozygous (ar -/-) zebrafish at 4.5 mpf injected with or without DHT (100 nM, 10μl for each). Dpf, days post fertilization; Mpf, months post fertilization.
To understand why disruption of ar in zebrafish caused atretic follicles and premature ovarian failure, we determined expression level of folliculogenesis markers, lhcgr, foxl2 and cyp11a1 in ovaries by qPCR. For 4.5-month old zebrafish ovaries, lhcgr, foxl2 and cyp11a1 expression was lower in homozygous ovaries (ar -/-) (ar ihb1225/ihb1225) compared with wildtype siblings (Figure 6P-6R). In addition, kitlga, a potential androgen responsive gene and a regulator of folliculogenesis [56–59] was assessed and expression was also lower in homozygous ovaries (ar -/-) (ar ihb1225/ihb1225) compared with wildtype siblings (Figure 6S). When DHT (100 nM, 10μl each ip) was given, kitlga expression was induced significantly in wildtype ovaries (ar+/+) (Figure 6S). Immunofluorescent assays for ovaries at 4.5 mpf with anti-vasa antibody confirmed histological observations (Supplementary Figure 4C and 4D). Thus, ar is essential for the maintenance of zebrafish ovarian function.
Targeted disruption of ar in zebrafish causes reproductive-endocrine disorder
Given the significant effect of ar on zebrafish spermatogenesis and ovarian function, we compared the levels of serum testosterone (T) and estradiol (E2) in mutant and control males and females, respectively. In males (5 mpf), serum testosterone was lower in ar -/- compared with ar +/+, but in females (5 mpf), serum testosterone was the opposite for ar -/- and ar +/+ (Figure 7A). In addition, in males (5 mpf), serum 11-ketotestosterone (11-KT) was higher in ar -/- compared with ar +/+(Figure 7B). Serum estradiol was not different between ar -/- and ar+/+ males, but was lower in ar -/- females compared with ar +/+ females (Figure 7C). Thus, targeted disruption of ar in zebrafish causes reproductive-endocrine disorder.
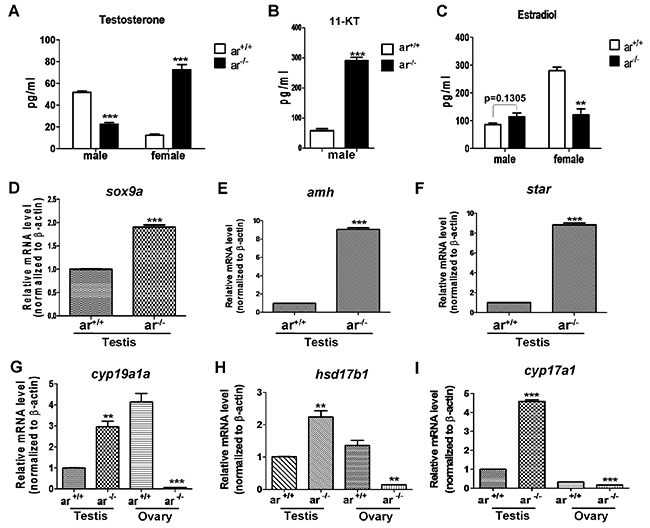
Figure 7: Serum sex hormone and gene expression of key steroidogenic enzymes in wildtype (ar+/+) and homozygous (ar-/-) (ar ihb1225/ihb1225) zebrafish at 5 months (serum hormone) or 4.5 mpf (gene expression). (A) In ar -/- male, serum testosterone (T) level was lower, but in ar -/- female, serum testosterone concentration increased. (B) In ar -/- male, serum 11-ketotestosterone (11-KT) was higher. (C) Serum estradiol (E2) was not altered obviously in ar +/+ and ar -/- males, but E2 in ar +/+ females is higher than that of ar -/- females. Serum testosterone (T) and estradiol (E2) were measured by ELASA. (D) sox9a expression in testes from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. (E) amh expression in testes from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. (F) star expression in testes from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. (G) cyp19a1a expression in testes and ovaries from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. (H) hsd17b1 expression in testes and ovaries from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. (I) cyp17a1 expression in testes and ovaries from wildtype (ar +/+) and homozygous (ar -/-) zebrafish. Mpf, months post fertilization.
Subsequently, we determined reproductive marker expression (sox9a and amh) [27, 60, 61], inducers of estradiol (cyp19a1a and hsd17b1) and hormone synthesis enzymes (star and cyp17a1a). sox9a, amh, cyp19a1a, hsd17b1, star and cyp17a1a expression was higher in ar -/- testes compared with ar +/+ testes (Figure 7D-7I). However, in ovaries, cyp19a1a, hsd17b1 and cyp17a1a expression was lower in ar -/- compared with ar+/+ (Figure 7G-7I). Thus, loss of ar causes dysregulation of genes related to reproductive development and hormone synthesis.
Therefore, the influence of ar KO on zebrafish might arise from disruption of ar roles in spermatogenesis, oogenesis and steroid biosynthesis.
DISCUSSION
The function of ar in vivo was first demonstrated in a mouse model [2, 14, 26]. Using tissue-specific male ARKO mice or male global ARKO mice, ar was confirmed to be essential for the development of male reproductive organs and spermatogenesis, although not required for testes formation [10, 12]. Female global ARKO mice appear normal, but develop premature ovarian failure (POF) phenotypes, have longer estrous cycles and reduced fertility, fewer follicles, impaired mammary development, delayed production of a first litter and fewer pups per litter [12, 15, 28, 42, 56, 62, 63]. In addition to reproductive defects, ARKO mice have late-onset obesity [14, 44], impaired cardiac growth [64], reduced neutrophils [65], accelerated wound healing [66], reduced bone size, thickness and volume of bone [67], and reduced muscle mass [68].
Even though the zebrafish ar gene has been isolated and its expression characterized [45, 46], its function in vivo is unclear. Thus, we used CRISPR/Cas9 technology to knock out ar in zebrafish and reveal important function in vivo. Of note, heterozygous ar+/- males were fertile and indistinguishable from wildtype males. In this study, even though we could not verified whether the truncated ar proteins were presented in heterozygous ar+/- zebrafish or due to lack of suitable zebrafish ar antibodies. The same phenotypes between heterozygous and wildtype indicated that knock out of ar in zebrafish might cause loss of function of ar completely instead of gain of function of the truncated ar proteins. In addition, disruption of ar resulted in more females; even though this alone does not suggest how zebrafish sex determination occurs, it does confirm the importance of ar in zebrafish sex differentiation. Actually, zebrafish sex determination is a complicated affair [47] [49, 69] [70].
It appears that the function of ar is evolutionarily conserved because targeted disruption of ar in zebrafish caused defects in spermatogenesis and maintenance of ovarian function, which were similar to ar-null mice. In addition, late onset of obesity phenotypes as indicated by fatty abdominal cavities were also evident in ar-null zebrafish, similar to that of ar-null mice [14]. Thus, in zebrafish, androgen receptor might also negatively regulate lipid metabolism. In addition, less energy is spent on gamete production in ar-null zebrafish, it might also cause zebrafish to gain weight.
Androgen action on zebrafish organogenesis has been described [71–73] and androgens are required for generation of breeding tubercles (breeding ornaments) on zebrafish pectoral fins [23, 73]. Here, we observed that targeted disruption of ar caused defects in the formation of breeding tubercles (BTs), which confirmed the role of androgen action on development of zebrafish secondary sex characteristics. However, we observed no defects of pectoral fin regeneration in ar-null male zebrafish, which is reported to be controlled by androgen/GSK3 signaling [71]. Such inconsistency may be due to difference among strains used [33].
Even though the effects of ar disruption on reproductive development have been described in a mouse model, the underlying mechanisms are unknown. Once androgens bind to their ar, a signaling cascade is initiated that directly or indirectly stimulates reproductive development, but how these genes work and how deregulation of the genes in ar-null mice (zebrafish) cause a series of defects is unclear. We noted expression of meiosis-related genes (ccnd2a and gsdf) were dysregulated in ar-null testes and fewer proliferated cells and more apoptotic cells were present. This may partially explain the effect of ar on zebrafish spermatogenesis. Also, we noticed that expression of genes involved in reproductive development (sox9a and amh) [27, 60, 61], estradiol induction (cyp19a1a and hsd17b1) and hormone synthesis (star and cyp17a1a) were deregulated in testes and ovaries after ar disruption. In addition, in ar-null male zebrafish, the level of testosterone was decreased, the level of 11-ketotestosterone was increased. By contrast, in ar-null female zebrafish, the level of androgenic sex hormone was increased dramatically, but the level of estrogenic sex hormone was decreased significantly. These phenomena not only suggested that the feminization of ar-null zebrafish was mediated by estrogen signaling, but also indicated that the importance of androgen signaling in maintenance of ovary function. The disorder of sexual hormone in ar-null zebrafish also confirmed that in zebrafish the synthesis of sexual hormone was inter-conversed during they were synthesized from cholesterol. In ar-null zebrafish, when testosterone was reduced, estradiol was increased or vice versa [74]. Therefore, dysfunction of the androgen signaling cascade due to ar disruption could explain phenotypes seen in ar-null zebrafish.
Intriguingly, we noticed some differences between ar-null male zebrafish and zebrafish with testis androgen deprivation using trilostane. In zebrafish with testis androgen deprevtion, Leydig cell marked gene (star) was increased, but the Sertoli cell gene marked genes (amh, gsdf) were not changed significantly. It seemed that androgen deprivationmainly affected androgen synthesis in Leydig cells [75]. However, in the testis of ar-null zebrafish, 11-KT was increased and the expression of genes related to steroidogenesis was increased in Leydig cells. In addition, the expressions of Sertoli cell marked gene (amh), and the spermatogonia-stimulating gene (gsdf) were also increased. Given that 11-ketotestosterone (11-KT) instead of testosterone serves as the prominent circulating androgen in zebrafish, the germ cell activities modulated by androgen action might mediate by paracrine factors.
Taken together, ar-null zebrafish may be a potential model for revealing pathological mechanisms of diseases related to ar defects. Using genetic approaches to understand mechanisms involved in ar-modulated reproductive development in a zebrafish model may help to elucidate the role of ar in hormone-dependent physiological development and pathogenesis, such as testicular feminization mutation syndrome (Tfm), oligozoospermia and stein-leventhal symdrome.
MATERIALS AND METHODS
Zebrafish and their maintenance
The wild type zebrafish (Danio rerio; AB strain) were used for generating ar mutant lines. The guidelines for zebrafish nomenclature (http://wiki.zfin.org/display/general/ZFIN+Zebrafish+Nomenclature+Guidelines) were followed for naming the two mutants (ar inb1225/ihb1225 and ar inb1226/ihb1226) (https://zfin.org/action/feature/view/ZDB-ALT-161228-1 and https://zfin.org/actin/feature/view/ZDB-ALT-170119-1).
Zebrafish were maintained in a re-circulating water system according to standard protocol. All experiments with zebrafish were approved by the animal care and use committee of Institute of Hydrobiology, Chinese Academy of Sciences.
Generation of ar -/- zebrafish
The zebrafish-Codon-Optimized Cas9 plasmid [63] was digested with XbaI, purified and transcribed using T7 mMessage Machine Kit (Ambion, Austin, USA). gRNA was designed using http://crispr.mit.edu, and the Exon 4 of ar was chosen as a targeting region. pUC19-gRNA scaffold was used for amplifying sgRNA template [76]. The gene name and the primers for PCR amplification were listed in Supplementary Tables 1 and 2. sgRNA was synthesized using Transcript Aid T7 High Yield Transcription Kit (Fermentas, Maryland, USA). Cas9 mRNA and sgRNA were mixed and injected into embryos at one or two-cell stage (500-800ng/μl and 50-80ng/μl, respectively). The mutations were initially detected by HMA (heteroduplex mobility assay) as previously described [77]. If the results were positive, the remainder embryos were raised up to adulthood as the F0, which were back-crossed with wild-type zebrafish (AB line) to generate the F1, which were genotyped by HMA initially and confirmed by sequencing of targeting sites. The heterozygous F1 were back-crossed to wild-type zebrafish (AB line; none of their own parents) to obtain F2. The F2 adult zebrafish carrying the same mutation were inter-crossed to generate the F3 offspring, which should contain wildtype (ar +/+), heterozygous (ar +/-) and homozygous (ar-/-).
Fertility assessment
Egg production assessment was conducted as described previously [78]. To evaluate female fertility between homozygous ar -/- and wildtype (ar +/+) siblings, we set 5 groups for ar -/- or ar+/+ female respectively. One ar -/- or ar+/+sibling female was mated with two wildtype males (AB line) every day for 2 weeks. The egg numbers were counted and the survival rate was calculated.
For artificial insemination, at 3 mpf, the sperm was obtained by crushing the dissected testes. Briefly, the testes were dissected from mutant male zebrafish under a stereo microscope and transferred to a culture dish. The testes were cut into small pieces in 100 μl of Hanks’ balanced salt solution (0.137 M NaCl, 5.4 mM KCl, 1.3 mM CaCl2, 1.0 mM MgSO4, 0.25 mM Na2HPO4, 0.44 mM KH2PO4, 4.2 mM NaHCO3, and 5.55 mM glucose, pH 7.2) and then were crushed using a forceps. Simultaneously, the eggs were obtained from wildtype female zebrafish (AB line). Artificial insemination was carried out according to the method reported previously [79].
For sperm motility evaluation, 1 μl of sperm suspension was placed on a glass slide, then, 1 μl of distilled water was added to the sperm suspension for activating sperms. The sperm motility was observed and evaluated under a dark-phase microscope (Optiphot 2, Nikon Inc., NY, USA) at 40×magnification. The motility value was presented as the percentage of sperms with active movement, the sperms only with vibration were not counted as motile sperms. For each sample, the sperm motility was measured in three different fields at least 2 times.
Sex determination
For sex determination of the wildtype (ar +/+) and homozygous (ar -/-) zebrafish, we firstly distinguished them by morphology. Female zebrafish had a big abdomen and prominent genital pore. To ensure the accuracy, we eventually verified their sex by dissection.
Morphological and histological analysis
Zebrafish (4.5 mpf) were weighted, photographed and dissected after anesthetization. Intact testes and ovaries from zebrafish (1.5 to 5 mpf) were dissected and fixed in 4 % PFA (Paraformaldehyde) over night at 4°C. Samples were dehydrated and embedded in paraffin, and cut into 4 μm. Hematoxylin-Eosin (H.E) staining was performed as described previously [78, 80]. Toluidine blue staining was performed as described previously [81].
Hormone measurement
Blood samples were collected from 5 month-old zebrafish as described [82]. For each zebrafish, 5 to 10 μl of blood could be collected. Blood collected from 3 individuals was used as one sample for measurement. The blood samples were centrifuged at 5000 g for 20 minutes at 4°C, and the supernatants were separated and purified according to the manufacturer’s extraction protocol (Cayman Chemical, Ann Arbor, USA). Testosterone, 11-ketotestosterone and estradiol were measured by competitive enzyme-linked immunosorbent assay (ELISA) kits (Cayman Chemical) following the manufacturer’s instructions. All standards and samples were measured by three independent experiments performed in triplicate.
Immunofluorescent, immunohistochemistry and apoptosis assay
Immunofluorescent staining for germ cells was performed using anti-Vasa antibody (DEAD-box helicase 4 (Cag_Vasa) antibody; Cat# Cag_Vasa, RRID:AB_2631966) as described previously [83, 84]. Immunohistochemistry staining for cell cycle was performed using anti-cyclinD1 antibody (GenWay Biotech Inc. Cat# 18-783-77526, RRID:AB_1009478).
Apoptotic cells were detected by terminal deoxynucleotidyltransferase-mediated dUTP nick end labeling (TUNEL) assay (Roche Applied Science, IN, USA).
DHT injection assay
Dihydrotestosterone (DHT) (Sigma-Aldrich) were prepared in the vehicle dimethoxyethane (DME) (Sigma-Aldrich, USA) and diluted in Sodium Chloride solution at 100 nM concentration [64]. Three female ar +/+ and ar -/- zebrafish (5 months) with similar weight were injected with 10 μl each (100nm/L). After 12 hours of intraperitoneal injection, intact ovaries from adult zebrafish were dissected after anesthetization, and total RNA was extracted from the ovaries for semi-quantitative RT-PCR assays.
Quantitative real-time PCR analysis
Total RNA was extracted using RNAiso Plus (TaKaRa, Tokyo, Japan) following the protocol provided by the manufacturer. cDNAs were synthesized using the Revert Aid First Strand cDNA Synthesis Kit (Fermentas). SYBR Green mix (Roche) was used for quantitative RT-PCR assays. The gene names and the primers are listed in Supplementary Tables 1 and 2. Actb1 (β-actin) was used as an internal control [85, 86].
Statistical analysis
Statistical analysis for sex ratio was performed using Microsoft Excel 2007, other statistical analysis was performed using GraphPad Prism, v5 (unpaired t-test) (GraphPad Software Inc). Data are reported as means ± S.E.M. of three independent experiments performed in triplicate. The difference was considered to be significant if the p values were less than 0.05. The p values are summarized with the following symbols: * p<0.05; ** p<0.01; ***p<0.001.
ACKNOWLEDGMENTS
We are grateful to Drs. Bo Zhang and Jingwei Xiong for their generous gifts of reagents.
CONFLICTS OF INTEREST
No competing interest declared.
GRANT SUPPORT
W. X is supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA08010208) and NSFC grant 31461163003, 91631102, 31671315 and 31721005.
REFERENCES
1. Davey RA, Grossmann M. Androgen Receptor Structure, Function and Biology: From Bench to Bedside. Clin Biochem Rev. 2016; 37:3-15.
2. Yeh S, Tsai MY, Xu Q, Mu XM, Lardy H, Huang KE, Lin H, Yeh SD, Altuwaijri S, Zhou X, Xing L, Boyce BF, Hung MC, et al. Generation and characterization of androgen receptor knockout (ARKO) mice: an in vivo model for the study of androgen functions in selective tissues. Proc Natl Acad Sci USA. 2002; 99:13498-503. https://doi.org/10.1073/pnas.212474399.
3. La Spada AR, Wilson EM, Lubahn DB, Harding AE, Fischbeck KH. Androgen receptor gene mutations in X-linked spinal and bulbar muscular atrophy. Nature. 1991; 352:77-79. https://doi.org/10.1038/352077a0.
4. Griffin JE. Androgen resistance-the clinical and molecular spectrum. N Engl J Med. 1992; 326:611-18. https://doi.org/10.1056/NEJM199202273260906.
5. Quigley CA, De Bellis A, Marschke KB, el-Awady MK, Wilson EM, French FS. Androgen receptor defects: historical, clinical, and molecular perspectives. Endocr Rev. 1995; 16:271-321. https://doi.org/10.1210/edrv-16-3-271.
6. McPhaul MJ. Molecular defects of the androgen receptor. J Steroid Biochem Mol Biol. 1999; 69:315-22. https://doi.org/10.1016/S0960-0760(99)00050-3.
7. Bardin CW, Bullock L, Schneider G, Allison JE, Stanley AJ. Pseudohermaphrodite rat: end organ insensitivity to testosterone. Science. 1970; 167:1136-37. https://doi.org/10.1126/science.167.3921.1136.
8. Lyon MF, Hawkes SG. X-linked gene for testicular feminization in the mouse. Nature. 1970; 227:1217-19. https://doi.org/10.1038/2271217a0.
9. Meyers-Wallen VN, Wilson JD, Griffin JE, Fisher S, Moorhead PH, Goldschmidt MH, Haskins ME, Patterson DF. Testicular feminization in a cat. J Am Vet Med Assoc. 1989; 195:631-34.
10. Zhou X. Roles of androgen receptor in male and female reproduction: lessons from global and cell-specific androgen receptor knockout (ARKO) mice. J Androl. 2010; 31:235-43. https://doi.org/10.2164/jandrol.109.009266.
11. Walters KA, Simanainen U, Handelsman DJ. Molecular insights into androgen actions in male and female reproductive function from androgen receptor knockout models. Hum Reprod Update. 2010; 16:543-58. https://doi.org/10.1093/humupd/dmq003.
12. Chang C, Lee SO, Wang RS, Yeh S, Chang TM. Androgen receptor (AR) physiological roles in male and female reproductive systems: lessons learned from ar-knockout mice lacking ar in selective cells. Biol Reprod. 2013; 89:21. https://doi.org/10.1095/biolreprod.113.109132.
13. Matsumoto T, Takeyama K, Sato T, Kato S. Androgen receptor functions from reverse genetic models. J Steroid Biochem Mol Biol. 2003; 85:95-99. https://doi.org/10.1016/S0960-0760(03)00231-0.
14. Sato T, Matsumoto T, Yamada T, Watanabe T, Kawano H, Kato S. Late onset of obesity in male androgen receptor-deficient (AR KO) mice. Biochem Biophys Res Commun. 2003; 300:167-71. https://doi.org/10.1016/S0006-291X(02)02774-2.
15. Hu YC, Wang PH, Yeh S, Wang RS, Xie C, Xu Q, Zhou X, Chao HT, Tsai MY, Chang C. Subfertility and defective folliculogenesis in female mice lacking androgen receptor. Proc Natl Acad Sci USA. 2004; 101:11209-14. https://doi.org/10.1073/pnas.0404372101.
16. Shiina H, Matsumoto T, Sato T, Igarashi K, Miyamoto J, Takemasa S, Sakari M, Takada I, Nakamura T, Metzger D, Chambon P, Kanno J, Yoshikawa H, Kato S. Premature ovarian failure in androgen receptor-deficient mice. Proc Natl Acad Sci USA. 2006; 103:224-29. https://doi.org/10.1073/pnas.0506736102.
17. Walters KA, Allan CM, Jimenez M, Lim PR, Davey RA, Zajac JD, Illingworth P, Handelsman DJ. Female mice haploinsufficient for an inactivated androgen receptor (AR) exhibit age-dependent defects that resemble the ar null phenotype of dysfunctional late follicle development, ovulation, and fertility. Endocrinology. 2007; 148:3674-84. https://doi.org/10.1210/en.2007-0248.
18. Walters KA, McTavish KJ, Seneviratne MG, Jimenez M, McMahon AC, Allan CM, Salamonsen LA, Handelsman DJ. Subfertile female androgen receptor knockout mice exhibit defects in neuroendocrine signaling, intraovarian function, and uterine development but not uterine function. Endocrinology. 2009; 150:3274-82. https://doi.org/10.1210/en.2008-1750.
19. Cheng XB, Jimenez M, Desai R, Middleton LJ, Joseph SR, Ning G, Allan CM, Smith JT, Handelsman DJ, Walters KA. Characterizing the neuroendocrine and ovarian defects of androgen receptor-knockout female mice. Am J Physiol Endocrinol Metab. 2013; 305:E717-26. https://doi.org/10.1152/ajpendo.00263.2013.
20. Xu Q, Lin HY, Yeh SD, Yu IC, Wang RS, Chen YT, Zhang C, Altuwaijri S, Chen LM, Chuang KH, Chiang HS, Yeh S, Chang C. Infertility with defective spermatogenesis and steroidogenesis in male mice lacking androgen receptor in Leydig cells. Endocrine. 2007; 32:96-106. https://doi.org/10.1007/s12020-007-9015-0.
21. Tsai MY, Yeh SD, Wang RS, Yeh S, Zhang C, Lin HY, Tzeng CR, Chang C. Differential effects of spermatogenesis and fertility in mice lacking androgen receptor in individual testis cells. Proc Natl Acad Sci USA. 2006; 103:18975-80. https://doi.org/10.1073/pnas.0608565103.
22. Fan W, Yanase T, Nomura M, Okabe T, Goto K, Sato T, Kawano H, Kato S, Nawata H. Androgen receptor null male mice develop late-onset obesity caused by decreased energy expenditure and lipolytic activity but show normal insulin sensitivity with high adiponectin secretion. Diabetes. 2005; 54:1000-08. https://doi.org/10.2337/diabetes.54.4.1000.
23. Tan KA, De Gendt K, Atanassova N, Walker M, Sharpe RM, Saunders PT, Denolet E, Verhoeven G. The role of androgens in sertoli cell proliferation and functional maturation: studies in mice with total or Sertoli cell-selective ablation of the androgen receptor. Endocrinology. 2005; 146:2674-83. https://doi.org/10.1210/en.2004-1630.
24. De Gendt K, Atanassova N, Tan KA, de França LR, Parreira GG, McKinnell C, Sharpe RM, Saunders PT, Mason JI, Hartung S, Ivell R, Denolet E, Verhoeven G. Development and function of the adult generation of Leydig cells in mice with Sertoli cell-selective or total ablation of the androgen receptor. Endocrinology. 2005; 146:4117-26. https://doi.org/10.1210/en.2005-0300.
25. De Gendt K, Swinnen JV, Saunders PT, Schoonjans L, Dewerchin M, Devos A, Tan K, Atanassova N, Claessens F, Lécureuil C, Heyns W, Carmeliet P, Guillou F, et al. A Sertoli cell-selective knockout of the androgen receptor causes spermatogenic arrest in meiosis. Proc Natl Acad Sci USA. 2004; 101:1327-32. https://doi.org/10.1073/pnas.0308114100.
26. Wang RS, Yeh S, Chen LM, Lin HY, Zhang C, Ni J, Wu CC, di Sant'Agnese PA, deMesy-Bentley KL, Tzeng CR, Chang C. Androgen receptor in sertoli cell is essential for germ cell nursery and junctional complex formation in mouse testes. Endocrinology. 2006; 147:5624-33. https://doi.org/10.1210/en.2006-0138.
27. Chang C, Chen YT, Yeh SD, Xu Q, Wang RS, Guillou F, Lardy H, Yeh S. Infertility with defective spermatogenesis and hypotestosteronemia in male mice lacking the androgen receptor in Sertoli cells. Proc Natl Acad Sci USA. 2004; 101:6876-81. https://doi.org/10.1073/pnas.0307306101.
28. Yeh S, Hu YC, Wang PH, Xie C, Xu Q, Tsai MY, Dong Z, Wang RS, Lee TH, Chang C. Abnormal mammary gland development and growth retardation in female mice and MCF7 breast cancer cells lacking androgen receptor. J Exp Med. 2003; 198:1899-908. https://doi.org/10.1084/jem.20031233.
29. Hannema SE, Hughes IA. Regulation of Wolffian duct development. Horm Res. 2007; 67:142-51. https://doi.org/10.1159/000096644.
30. Sato T, Matsumoto T, Kawano H, Watanabe T, Uematsu Y, Sekine K, Fukuda T, Aihara K, Krust A, Yamada T, Nakamichi Y, Yamamoto Y, Nakamura T, et al. Brain masculinization requires androgen receptor function. Proc Natl Acad Sci USA. 2004; 101:1673-78. https://doi.org/10.1073/pnas.0305303101.
31. O'Hara L, Smith LB. Androgen receptor roles in spermatogenesis and infertility. Best Pract Res Clin Endocrinol Metab. 2015; 29:595-605. https://doi.org/10.1016/j.beem.2015.04.006.
32. Walters KA, Simanainen U, Gibson DA. Androgen action in female reproductive physiology. Curr Opin Endocrinol Diabetes Obes. 2016; 23:291-96. https://doi.org/10.1097/MED.0000000000000246.
33. Prizant H, Gleicher N, Sen A. Androgen actions in the ovary: balance is key. J Endocrinol. 2014; 222:R141-51. https://doi.org/10.1530/JOE-14-0296.
34. Gervásio CG, Bernuci MP, Silva-de-Sá MF, Rosa-E-Silva AC. The role of androgen hormones in early follicular development. ISRN Obstet Gynecol. 2014; 2014:818010. https://doi.org/10.1155/2014/818010.
35. Sen A, Prizant H, Light A, Biswas A, Hayes E, Lee HJ, Barad D, Gleicher N, Hammes SR. Androgens regulate ovarian follicular development by increasing follicle stimulating hormone receptor and microRNA-125b expression. Proc Natl Acad Sci USA. 2014; 111:3008-13. https://doi.org/10.1073/pnas.1318978111.
36. Thornton JW. Evolution of vertebrate steroid receptors from an ancestral estrogen receptor by ligand exploitation and serial genome expansions. Proc Natl Acad Sci USA. 2001; 98:5671-76. https://doi.org/10.1073/pnas.091553298.
37. Páll MK, Mayer I, Borg B. Androgen and behavior in the male three-spined stickleback, Gasterosteus aculeatus. II. Castration and 11-ketoandrostenedione effects on courtship and parental care during the nesting cycle. Horm Behav. 2002; 42:337-44. https://doi.org/10.1006/hbeh.2002.1820.
38. Páll MK, Mayer I, Borg B. Androgen and behavior in the male three-spined stickleback, Gasterosteus aculeatus I.-changes in 11-ketotestosterone levels during the nesting cycle. Horm Behav. 2002; 41:377-83. https://doi.org/10.1006/hbeh.2002.1777.
39. Cavaco JE, Bogerd J, Goos H, Schulz RW. Testosterone inhibits 11-ketotestosterone-induced spermatogenesis in African catfish (Clarias gariepinus). Biol Reprod. 2001; 65:1807-12. https://doi.org/10.1095/biolreprod65.6.1807.
40. Cavaco JE, van Baal J, van Dijk W, Hassing GA, Goos HJ, Schulz RW. Steroid hormones stimulate gonadotrophs in juvenile male African catfish (Clarias gariepinus). Biol Reprod. 2001; 64:1358-65. https://doi.org/10.1095/biolreprod64.5.1358.
41. Cavaco JE, van Blijswijk B, Leatherland JF, Goos HJ, Schulz RW. Androgen-induced changes in Leydig cell ultrastructure and steroidogenesis in juvenile African catfish, Clarias gariepinus. Cell Tissue Res. 1999; 297:291-99. https://doi.org/10.1007/s004410051357.
42. Baroiller JF, Guiguen Y. Endocrine and environmental aspects of sex differentiation in gonochoristic fish. EXS. 2001; 91:177-201. https://doi.org/10.1007/978-3-0348-7781-7_9.
43. Kobayashi M, Aida K, Hanyu I. Induction of Gonadotropin Surge by Steroid-Hormone Implantation in Ovariectomized Goldfish. Zool Sci. 1987; 4:1084.
44. Gorelick DA, Watson W, Halpern ME. Androgen receptor gene expression in the developing and adult zebrafish brain. Dev Dyn. 2008; 237:2987-95. https://doi.org/10.1002/dvdy.21700.
45. Hossain MS, Larsson A, Scherbak N, Olsson PE, Orban L. Zebrafish androgen receptor: isolation, molecular, and biochemical characterization. Biol Reprod. 2008; 78:361-69. https://doi.org/10.1095/biolreprod.107.062018.
46. de Waal PP, Wang DS, Nijenhuis WA, Schulz RW, Bogerd J. Functional characterization and expression analysis of the androgen receptor in zebrafish (Danio rerio) testis. Reproduction. 2008; 136:225-34. https://doi.org/10.1530/REP-08-0055.
47. Liew WC, Orbán L. Zebrafish sex: a complicated affair. Brief Funct Genomics. 2014; 13:172-87. https://doi.org/10.1093/bfgp/elt041.
48. Orban L, Sreenivasan R, Olsson PE. Long and winding roads: testis differentiation in zebrafish. Mol Cell Endocrinol. 2009; 312:35-41. https://doi.org/10.1016/j.mce.2009.04.014.
49. Wilson CA, High SK, McCluskey BM, Amores A, Yan YL, Titus TA, Anderson JL, Batzel P, Carvan MJ 3rd, Schartl M, Postlethwait JH. Wild sex in zebrafish: loss of the natural sex determinant in domesticated strains. Genetics. 2014; 198:1291-308. https://doi.org/10.1534/genetics.114.169284.
50. Uchida D, Yamashita M, Kitano T, Iguchi T. Oocyte apoptosis during the transition from ovary-like tissue to testes during sex differentiation of juvenile zebrafish. J Exp Biol. 2002; 205:711-18.
51. Maack G, Segner H. Morphological development of the gonads in zebrafish. J Fish Biol. 2003; 62:895-906. https://doi.org/10.1046/j.1095-8649.2003.00074.x.
52. Wang XG, Bartfai R, Sleptsova-Freidrich I, Orban L. The timing and extent of ‘juvenile ovary’ phase are highly variable during zebrafish testis differentiation. J Fish Biol. 2007; 70:33-44. https://doi.org/10.1111/j.1095-8649.2007.01363.x.
53. Hartung O, Forbes MM, Marlow FL. Zebrafish vasa is required for germ-cell differentiation and maintenance. Mol Reprod Dev. 2014; 81:946-61. https://doi.org/10.1002/mrd.22414.
54. Gautier A, Sohm F, Joly JS, Le Gac F, Lareyre JJ. The proximal promoter region of the zebrafish gsdf gene is sufficient to mimic the spatio-temporal expression pattern of the endogenous gene in Sertoli and granulosa cells. Biol Reprod. 2011; 85:1240-51. https://doi.org/10.1095/biolreprod.111.091892.
55. Duffy KT, McAleer MF, Davidson WR, Kari L, Kari C, Liu CG, Farber SA, Cheng KC, Mest JR, Wickstrom E, Dicker AP, Rodeck U. Coordinate control of cell cycle regulatory genes in zebrafish development tested by cyclin D1 knockdown with morpholino phosphorodiamidates and hydroxyprolyl-phosphono peptide nucleic acids. Nucleic Acids Res. 2005; 33:4914-21. https://doi.org/10.1093/nar/gki799.
56. Matsumoto AM. Reproductive endocrinology: Estrogens-not just female hormones. Nat Rev Endocrinol. 2013; 9:693-94. https://doi.org/10.1038/nrendo.2013.211.
57. Matzuk MM, Burns KH, Viveiros MM, Eppig JJ. Intercellular communication in the mammalian ovary: oocytes carry the conversation. Science. 2002; 296:2178-80. https://doi.org/10.1126/science.1071965.
58. Sen A, Hammes SR. Granulosa cell-specific androgen receptors are critical regulators of ovarian development and function. Mol Endocrinol. 2010; 24:1393-403. https://doi.org/10.1210/me.2010-0006.
59. Matsumoto T, Sakari M, Okada M, Yokoyama A, Takahashi S, Kouzmenko A, Kato S. The Androgen Receptor in Health and Disease. Annu Rev Physiol. 2013;75:201-24. https://doi.org/10.1146/annurev-physiol-030212-183656.
60. Sun D, Zhang Y, Wang C, Hua X, Zhang XA, Yan J. Sox9-related signaling controls zebrafish juvenile ovary-testis transformation. Cell Death Dis. 2013; 4:e930. https://doi.org/10.1038/cddis.2013.456.
61. Skaar KS, Nóbrega RH, Magaraki A, Olsen LC, Schulz RW, Male R. Proteolytically activated, recombinant anti-mullerian hormone inhibits androgen secretion, proliferation, and differentiation of spermatogonia in adult zebrafish testis organ cultures. Endocrinology. 2011; 152:3527-40. https://doi.org/10.1210/en.2010-1469.
62. Xu N, Chua AK, Jiang H, Liu NA, Goodarzi MO. Early embryonic androgen exposure induces transgenerational epigenetic and metabolic changes. Mol Endocrinol. 2014; 28:1329-36. https://doi.org/10.1210/me.2014-1042.
63. Liu D, Wang Z, Xiao A, Zhang Y, Li W, Zu Y, Yao S, Lin S, Zhang B. Efficient gene targeting in zebrafish mediated by a zebrafish-codon-optimized cas9 and evaluation of off-targeting effect. J Genet Genomics. 2014; 41:43-46. https://doi.org/10.1016/j.jgg.2013.11.004.
64. Ikeda Y, Aihara K, Sato T, Akaike M, Yoshizumi M, Suzaki Y, Izawa Y, Fujimura M, Hashizume S, Kato M, Yagi S, Tamaki T, Kawano H, et al. Androgen receptor gene knockout male mice exhibit impaired cardiac growth and exacerbation of angiotensin II-induced cardiac fibrosis. J Biol Chem. 2005; 280:29661-66. https://doi.org/10.1074/jbc.M411694200.
65. Lai JJ, Lai KP, Zeng W, Chuang KH, Altuwaijri S, Chang C. Androgen receptor influences on body defense system via modulation of innate and adaptive immune systems: lessons from conditional ar knockout mice. Am J Pathol. 2012; 181:1504-12. https://doi.org/10.1016/j.ajpath.2012.07.008.
66. Lai JJ, Lai KP, Chuang KH, Chang P, Yu IC, Lin WJ, Chang C. Monocyte/macrophage androgen receptor suppresses cutaneous wound healing in mice by enhancing local TNF-alpha expression. J Clin Invest. 2009; 119:3739-51. https://doi.org/10.1172/JCI39335.
67. Venken K, De Gendt K, Boonen S, Ophoff J, Bouillon R, Swinnen JV, Verhoeven G, Vanderschueren D. Relative impact of androgen and estrogen receptor activation in the effects of androgens on trabecular and cortical bone in growing male mice: a study in the androgen receptor knockout mouse model. J Bone Miner Res. 2006; 21:576-85. https://doi.org/10.1359/jbmr.060103.
68. MacLean HE, Chiu WS, Notini AJ, Axell AM, Davey RA, McManus JF, Ma C, Plant DR, Lynch GS, Zajac JD. Impaired skeletal muscle development and function in male, but not female, genomic androgen receptor knockout mice. FASEB J. 2008; 22:2676-89. https://doi.org/10.1096/fj.08-105726.
69. Nagabhushana A, Mishra RK. Finding clues to the riddle of sex determination in zebrafish. J Biosci. 2016; 41:145-55. https://doi.org/10.1007/s12038-016-9593-1.
70. Luzio A, Coimbra AM, Benito C, Fontaínhas-Fernandes AA, Matos M. Screening and identification of potential sex-associated sequences in Danio rerio. Mol Reprod Dev. 2015; 82:756-64. https://doi.org/10.1002/mrd.22508.
71. Nachtrab G, Czerwinski M, Poss KD. Sexually dimorphic fin regeneration in zebrafish controlled by androgen/GSK3 signaling. Curr Biol. 2011; 21:1912-17. https://doi.org/10.1016/j.cub.2011.09.050.
72. Kang J, Nachtrab G, Poss KD. Local Dkk1 crosstalk from breeding ornaments impedes regeneration of injured male zebrafish fins. Dev Cell. 2013; 27:19-31. https://doi.org/10.1016/j.devcel.2013.08.015.
73. McMillan SC, Xu ZT, Zhang J, Teh C, Korzh V, Trudeau VL, Akimenko MA. Regeneration of breeding tubercles on zebrafish pectoral fins requires androgens and two waves of revascularization. Development. 2013; 140:4323-34. https://doi.org/10.1242/dev.095992.
74. Tokarz J, Möller G, de Angelis MH, Adamski J. Zebrafish and steroids: what do we know and what do we need to know? J Steroid Biochem Mol Biol. 2013; 137:165-73. https://doi.org/10.1016/j.jsbmb.2013.01.003.
75. García-López A, de Jonge H, Nóbrega RH, de Waal PP, van Dijk W, Hemrika W, Taranger GL, Bogerd J, Schulz RW. Studies in zebrafish reveal unusual cellular expression patterns of gonadotropin receptor messenger ribonucleic acids in the testis and unexpected functional differentiation of the gonadotropins. Endocrinology. 2010; 151:2349-60. https://doi.org/10.1210/en.2009-1227.
76. Chang N, Sun C, Gao L, Zhu D, Xu X, Zhu X, Xiong JW, Xi JJ. Genome editing with RNA-guided Cas9 nuclease in zebrafish embryos. Cell Res. 2013; 23:465-72. https://doi.org/10.1038/cr.2013.45.
77. Ota S, Hisano Y, Muraki M, Hoshijima K, Dahlem TJ, Grunwald DJ, Okada Y, Kawahara A. Efficient identification of TALEN-mediated genome modifications using heteroduplex mobility assays. Genes Cells. 2013; 18:450-58. https://doi.org/10.1111/gtc.12050.
78. Chu L, Li J, Liu Y, Cheng CH. Gonadotropin Signaling in Zebrafish Ovary and Testis Development: Insights From Gene Knockout Study. Mol Endocrinol. 2015; 29:1743-58. https://doi.org/10.1210/me.2015-1126.
79. Yang H, Carmichael C, Varga ZM, Tiersch TR. Development of a simplified and standardized protocol with potential for high-throughput for sperm cryopreservation in zebrafish Danio rerio. Theriogenology. 2007; 68:128-36. https://doi.org/10.1016/j.theriogenology.2007.02.015.
80. Zhang Z, Lau SW, Zhang L, Ge W. Disruption of Zebrafish Follicle-Stimulating Hormone Receptor (fshr) But Not Luteinizing Hormone Receptor (lhcgr) Gene by TALEN Leads to Failed Follicle Activation in Females Followed by Sexual Reversal to Males. Endocrinology. 2015; 156:3747-62. https://doi.org/10.1210/en.2015-1039.
81. Leal MC, Cardoso ER, Nóbrega RH, Batlouni SR, Bogerd J, França LR, Schulz RW. Histological and stereological evaluation of zebrafish (Danio rerio) spermatogenesis with an emphasis on spermatogonial generations. Biol Reprod. 2009; 81:177-87. https://doi.org/10.1095/biolreprod.109.076299.
82. Pedroso GL, Hammes TO, Escobar TD, Fracasso LB, Forgiarini LF, da Silveira TR. Blood collection for biochemical analysis in adult zebrafish. J Vis Exp. 2012; 63:e3865.
83. Sun M, Li Z, Gui JF. Dynamic Distribution of Spindlin in Nucleoli, Nucleoplasm and Spindle From Primary Oocytes to Mature Eggs and its Critical Function for Oocyte-to-Embryo Transition in Gibel Carp. J Exp Zool A Ecol Genet Physiol. 2010; 313A: 461-73. https://doi.org/10.1002/jez.618.
84. Peng JX, Xie JL, Zhou L, Hong YH, Gui JF. Evolutionary conservation of Dazl genomic organization and its continuous and dynamic distribution throughout germline development in gynogenetic gibel carp. J Exp Zoolog B Mol Dev Evol. 2009; 312:855-71. https://doi.org/10.1002/jez.b.21301.
85. Chen S, Zhang H, Wang F, Zhang W, Peng G. nr0b1 (DAX1) mutation in zebrafish causes female-to-male sex reversal through abnormal gonadal proliferation and differentiation. Mol Cell Endocrinol. 2016; 433:105-16. https://doi.org/10.1016/j.mce.2016.06.005.
86. Pradhan A, Olsson PE. Juvenile ovary to testis transition in zebrafish involves inhibition of ptges. Biol Reprod. 2014; 91:33. https://doi.org/10.1095/biolreprod.114.119016.

