INTRODUCTION
Total joint arthroplasty is an established surgical technique that has been successfully used to relieve pain and improve the movement and quality of life of patients with severe joint diseases [1]. Today, total knee replacement (TKR) surgeries are performed worldwide, with excellent results. Artificial knee joints are comprised of cobalt-chromium-molybdenum (CoCrMo) alloys with femoral components articulated using polyethylene (PE) on the tibial surface [2]. Kretzer et al. [3] highlights that although the majority of wear arises at the sites of polyethylene, clinically significant amounts of metal wear also occur. In addition, CoCrMo and other metal alloys used in knee components have some further drawbacks. For example, there are concerns regarding potential metal ion release and subsequent osteolysis or allergenicity [4, 5]. In particular, metals used for implantation have a large elastic modulus (approximately 6–20 times greater than that of the surrounding bone) [5–8], causing impaired load force transmission at the implant-tissue interface, stress shielding, and peri-implant bone resorption [5, 9]. Polyether-ether-ketone (PEEK), which exhibits excellent wear-resistance characteristics, mechanical properties, lighter weight, low elastic modulus [5, 10], natural radiolucency, compatibility with magnetic resonance imaging [5, 11, 12], constitutes a potential alternative for metals in metal-on-polymer bearing surfaces. PEEK has become highly attractive for use as a biomaterial for trauma and orthopedic applications, as it has already been successfully employed for spinal surgery [13, 14]. In addition, a recent study revealed the potential of PEEK as a surface material for artificial joints with PE as the other articulating surface [2], and PEEK is used as a component of knee prosthesis (e.g. the bushings and flanges) in current knee products [e.g. the Enduro knee system (Aesculap, Germany)] [14].
Aseptic loosening and peri-prosthetic osteolysis comprise the major complications that limit the long-term survival of prosthetic joints [15]. The mechanisms underlying the development of osteolysis and aseptic loosening are known to influence the longevity of TKRs. Several animal studies have focused on the potential role of wear particles in bone resorption around implants [16]. For example, a study by Pearson et al. had demonstrated that CD4+ T-cells were activated in response to wear debris from CoCrMo alloys [17]. Previous animal studies have shown that metal hypersensitivity was mediated by T-cells in classical delayed-type hypersensitivity (DTH)-type responses (DTH type IV). The effector phase of a DTH response is initiated by a contact between sensitized T-cells and an antigen presented in a class-II major histocompatibility complex (MHC) by antigen-presenting cells (APCs). In this phase, T-cells, which are activated by antigens, are characterized as helper T-cells, which, in conjunction with APCs, can secrete a variety of cytokines that recruit and activate macrophages, monocytes, neutrophils, and other inflammatory cells [18, 19]. Previous studies have shown an increased proportion of CD8+ T-cells and a decreased ratio of CD4+ to CD8+ T-cells in the peripheral blood during revision arthroplasty, compared with the blood during primary arthroplasty [20]. Additionally, histological examinations revealed that metal wear induced a typical non-specific inflammatory response as well as a specific metal hypersensitivity immune reaction [21].
However, to assess the feasibility of its use as a potential alternative for metals in TKRs, the mechanisms by which the immune system reacts to PEEK particles need to be determined. Previous studies have made some headway in understanding the role of macrophages and the cytokine secretion profiles in response to PEEK debris. For example, Hallab et al. found that there were no significant differences between the effects of PEEK and PE particles on macrophage viability or proliferation [22]. In addition, subtle elevations in inflammatory cytokines indicated a mild persistence of responses to PEEK debris in an animal model [23]; however, it is also unknown whether the immune response to PEEK particles is stronger than that to metals. We hypothesized that, similar to CoCrMo particles, the proliferation reactivity (peripheral blood mononuclear cells [PBMCs] or local tissues) responses to challenges with PEEK particles would also be mediated by both macrophages and early T-cell activation. In this study, we aimed to determine the effect of particulate wear debris on the immune system, with a focus on T-lymphocytes (both CD4+ and CD8+ T-cells), and to identify the effects of two types of wear particles on cytotoxic and immunological reactions.
RESULTS
Wear particle characterization
The scanning electron microscope (SEM) micrographs showed that the particles had similar morphological characteristics; i.e., globular or granular shapes, at all size ranges (Figure 1A–1D). The mean particle diameter of the PEEK samples was 1.05 μm. The range was 0.25–46.47 μm, with 99% of the particles measuring less than 5 μm and 93% in the submicron range (Figure 1E, Table 1). The mean diameter of the CoCrMo particles was 3.16 μm. The range was 0.39–61.65 μm, with 90% of the particles measuring less than 5 μm and 50% in the submicron range (Figure 1E, Table 1). The area, perimeter, ellipse major axis, and ellipse minor axis measurements are shown in Table 1.
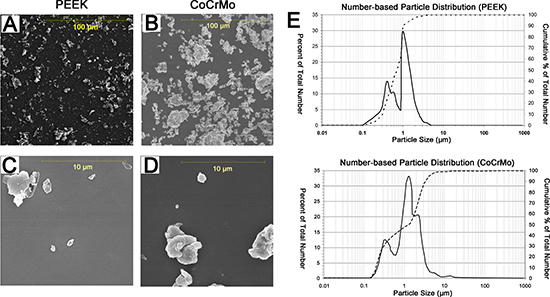
Figure 1: Scanning electron microscopy (SEM) images and size distribution (number-based) of wear debris from polyether-ether-ketone (PEEK) and cobalt-chromium-molybdenum (CoCrMo) groups. (A and C) SEM images of PEEK wear debris in two magnification fields (A. magnification 1000×, C. magnification 10,000×). (B and D) SEM images of CoCrMo wear debris in two magnification fields (B. magnification 1000×, D. magnification 10,000×). (E) Size distribution of PEEK and CoCrMo particles, as determined using SEM.
Table 1: SEM particle analysis of PEEK and CoCrMo particles
PEEK |
CoCrMo |
|
|---|---|---|
Particle size (μm) |
1.05 (0.70) |
3.16 (2.13) |
Aspect ratio |
1.83 (1.53) |
1.81 (1.59) |
Roundness |
0.61 (0.52) |
0.60 (0.51) |
Form factor |
0.67 (0.60) |
0.67 (0.60) |
Perimeter (μm) |
4.51 (2.12) |
5.96 (3.65) |
Parameter values are reported as means (medians).
Effects of wear particles on T-cell populations
The full gating strategies are shown in Supplementary Figure 1 (taking the identification of CD4+IL2+ T-cell populations induced by CoCrMo wear particles as an example). For CD4+ T-cell responses in human peripheral blood, there were no significant differences between the experimental groups (PEEK and CoCrMo) and the negative control group. Compared to the positive control, the experimental groups all showed decreased or similar responses. For example, PEEK debris showed lower responses than the positive control in CD4+IL2+ (p = 0.008) and CD4+IL10+ (p = 0.003) cells. There was no difference between PEEK and CoCrMo wear particles in CD4+ T-cell responses (Figure 2).
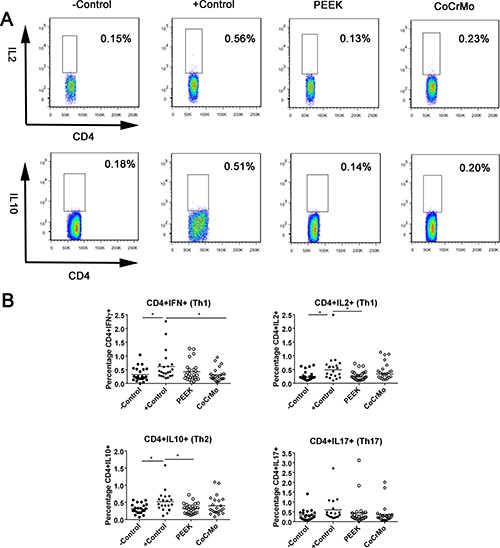
Figure 2: CD4+ T-cell populations identified by immunostaining and flow cytometry in human peripheral blood. (A) Raw data showing differences between PEEK and positive control groups in the induction of CD4+IL2+ and CD4+IL10+ T-cell responses. (B) Th1, Th2, and Th17 cells are shown in the control group or the groups challenged with PEEK, and CoCrMo particles. The −Control group was unchallenged, resuspended in RPMI 1640 medium; the +Control group was treated with human T-activator CD3/28. Each data point represents the response of an individual participant; the bar indicates the mean value. *P < 0.05.
For CD8+ T-cell responses in human peripheral blood, there were no significant differences between the experimental groups (PEEK and CoCrMo) and the negative control group, except for CoCrMo, which exhibited a higher CD8+IL10+ T-cell response than the negative control (p = 0.006; Figure 3). Compared to the positive control, the experimental groups all showed decreased or similar responses. PEEK debris showed lower responses than the positive control in CD8+IL2+ (p = 0.027; Figure 3) and CD8+IL10+ T-cell responses (p = 0.021; Figure 3). Clear differences were observed between PEEK and CoCrMo particles in the induction of CD8+IL2+ and CD8+IL10+ T-cell responses, with lower responses in PEEK debris (p = 0.023 and 0.036, respectively; Figure 3).
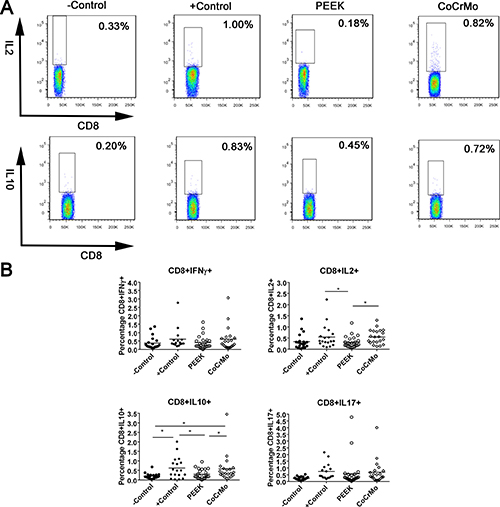
Figure 3: CD8+ T-cell populations identified by immunostaining and flow cytometry in human peripheral blood. (A) Raw data showing differences between PEEK and CoCrMo particles in the induction of CD8+IL2+ and CD8+IL10+ T-cell responses. (B) CD8+IFNγ+/IL-2+/IL-10+/IL-17+ cells in the control groups or the groups challenged with PEEK, and CoCrMo particles. The −Control group was unchallenged, resuspended in RPMI 1640 medium; the +Control group was treated with human T-activator CD3/28. Each data point represents the response of an individual subject; the bar indicates the mean value. *P < 0.05.
Histological analysis of the rat synovium
The detection of wear particles is shown in Figure 4A–4D. Multinucleated giant cells were concentrated in particle-containing areas (Figure 4B and 4D). In the immunohistochemical analysis, almost no CD4+ T-cells were found in the control or experimental groups (Figure 5). However, CD8+ T-cells were highly expressed in experimental groups, with comparable expressions in the PEEK and CoCrMo groups (Figure 5). Inflammatory factors (IL-6 and TNFα) showed increased expression in the CoCrMo groups (Figure 5).
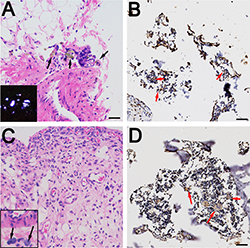
Figure 4: Histological examination of rat synovium tissues. (A and C) Hematoxylin and eosin staining of synovium (A for PEEK, C for CoCrMo), indicated by black arrows; wear particles (A) and inflammatory cells (C) can be observed in the necrotic tissues. Polarizing microscopy of PEEK particles is shown in the lower left quarter. (B and D) Immunohistochemical staining of synovium (B for PEEK, D for CoCrMo), indicated by red arrows; multinucleated giant cells and Langhans giant cells are concentrated at the sites of wear particles. Bar = 50 μm.
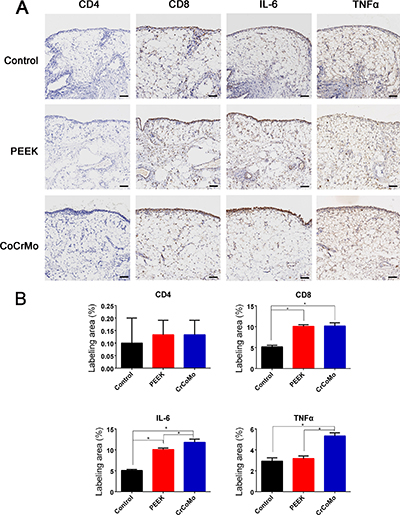
Figure 5: Immunohistochemical staining and analysis of synovium. (A) Immunohistochemical staining (CD4, CD8, IL-6, and TNFα) of synovium in three groups. (B) Semiquantitative analysis of the positive area. Bar = 100 μm. *P < 0.05.
Immunofluorescence labeling of the rat spleen
With respect to CD4+ T-cell responses in the rat spleen, there were no significant differences between the experimental groups (PEEK and CoCrMo) and the negative control group (Figure 6). In contrast, for CD8+ T-cell responses, an increased number of positive cells were found in the PEEK and CoCrMo groups compared to controls, with comparable responses between the two groups (Figure 7).
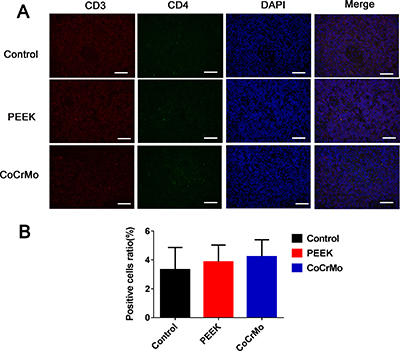
Figure 6: Immunofluorescence labeling of CD4+ T-cell responses. (A) Immunofluorescence labeling of CD4+ T-cells of the spleen in three groups. (B) Analysis of the ratio of positive cells. Bar = 50 μm.
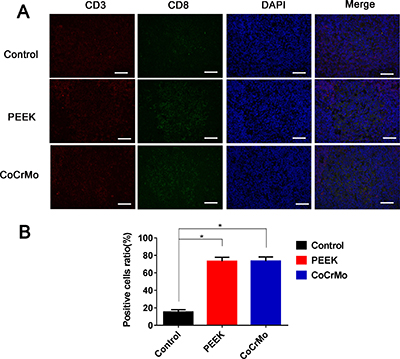
Figure 7: Immunofluorescence labeling of CD8+ T-cell responses. (A) Immunofluorescence labeling of CD8+ T-cells of the spleen in three groups. (B) Analysis of the ratio of positive cells. Bar = 50 μm. *P < 0.05.
DISCUSSION
In this study, we assessed the effect of particulate wear debris on T-cells. We also investigated the effects of two kinds of wear particles on cytotoxic and immunological reactions. Our findings demonstrated that PEEK and CoCrMo particles induced immune responses that were mainly mediated by CD8+ T-cells, and that PEEK wear particles induced fewer inflammatory reactions than CoCrMo particles, providing important insights into the immunological reactions caused by these common wear particles (Supplementary Figure 2).
The wear particles from bearing materials can affect the longevity of total joint arthroplasty, as debris can elicit biological responses and cause periprosthetic osteolysis, leading to loosening of the prosthesis [16]. Numerous factors are thought to affect the cellular response to wear particles including particle composition, dose, volume, size, and shape [24–26]. Particles in the phagocytosable size range (0.1–10 μm in diameter) are considered the most biologically reactive, particularly those with a mean size of 1 μm [27]. Green et al. [28] showed that 0.45- and 1.71-μm-sized PE particles were necessary to stimulate cytokine release and bone resorbing activities. In the present study, the size of the wear particles was in the range of 0.1–102 μm in diameter, with an average diameter of 1–3 μm, which was thus biologically phagocytic and phlogistic.
Lymphocyte activation requires the simultaneous delivery of an antigen-specific signal associated with MHC proteins and a “costimulatory” signal by APCs [29, 30]. When PBMCs treated with debris for over 72 h were initially processed by macrophages, downstream activation of T-cells was observed. Previous clinical and in vitro studies have shown that metal hypersensitivity is mediated by T-cells in DTH type-IV responses [19]. Previous studies have also examined the specific roles of Th1 and Th2 lymphocytes in osteolysis and aseptic loosening in TKRs [30]. In particular, the Th1 response is crucial for the activation of macrophages and cytotoxic T-lymphocytes, and is known to be involved in cell-mediated immune responses. In comparison, the Th2 response is the most effective activator of B-lymphocytes and is associated with humoral immunity [30, 31]. Th17 cells play an important role in the initiation of inflammation by recruiting neutrophils and macrophages to sites of infection or sites of tissue damage, in the case of sterile inflammation [17, 32]. However, in contrast with previous literature, in both in vitro and in vivo studies, we verified that the two types of particles induced relatively low levels of CD4+ T-cells, especially in synovial tissues and peripheral spleen. Similarly, Willert et al. [33] had reported that metal particle-induced responses were not characteristic of a delayed-type IV hypersensitivity reaction, but rather comprised aseptic lymphocyte-dominated vasculitis-associated lesions (ALVAL) or lymphocyte-dominated immunological answers (LYDIA).
In in vitro studies, CD8+ T-cells were also induced in relatively low levels, with PEEK particles showing lower responses in the induction of CD8+IL2+/IL10+ T-cells. However, in in vivo studies, CD8+ T-cells were more highly induced in local synovial tissues and peripheral organs than the control group. In accordance with this, previous studies have shown an increased proportion of CD8+ T-cells and a decreased ratio of CD4+ to CD8+ T-cells in the peripheral blood lymphocytes of patients with worn implants [20]. Furthermore, Hailer et al. [34] had reported a correlation between the percentage of CD8+ T-cells and the concentration of chromium and cobalt in the blood of patients. The mechanisms behind this phenomenon were unclear, and it was speculated that CD8+ T-cells were specific for class-I MHC molecules on foreign proteins [35, 36]. In the present study, the wear particles were able to induce apoptosis and/or direct necrosis of endothelial cells, causing the accumulation of macrophages and Langerhans cells (shown in Figure 4). These necrotic substances and APCs might activate the class-I MHC molecular pathway, resulting in elevated CD8+ T-cell response. The apoptosis of T-lymphocytes may also be responsible for imbalanced CD4+/CD8+ T-cell responses. For example, Stefan et al. [36] reported that the number of CD4+ cells decreased during strong apoptotic reactions, whereas CD8+ cells were affected to a much lesser degree. Thus, the high incidence of CD8+-related lymphocyte reactivity and the subsequent in vivo activation may contribute to the etiology of debris-induced osteolysis.
Previous studies have reported that metallic debris derived from alloy implants induced macrophage activation and triggered immune responses, releasing an array of pro-inflammatory mediators, including TNF-α, IL-1, IL-6 [37, 38], and colony-stimulating factors such as macrophage- and granulocyte-macrophage colony-stimulating factor [38, 39]. In this study, we examined the levels of inflammatory factors (IL-6 and TNFα) in the local tissues, and found that CoCrMo particles induced more inflammatory factors than PEEK particles, and that the inflammatory factors were induced not only by macrophage activation, but also by T-cell activation. This is consistent with a proposal by Bainbridge et al. [40, 41], that intracellular particles from the prosthesis, along with elevated costimulatory molecule expression, might promote T-cell inflammatory reactions in the prosthetic tissues.
There were several limitations to this study. First, the wear condition alone may not represent the nature of the wear in clinical cases; thus, further studies using wear particles isolated from tissues or validated joint replacement simulators are required, instead of manufactured particles. Second, although metal ions have been reported to be a theoretical source of genotoxicity [42], we did not investigate the immunogenicity of degradation products of CoCrMo or PEEK particles. Third, it remains unknown whether the complex environment of the peri-implant milieu ultimately mitigates or enhances cell susceptibility to the effects of particle exposure in vivo. Thus, the results of this study established a foundation for more sophisticated studies using mixed cell populations or direct in vivo osteolysis models.
In summary, relatively minor cytotoxic and immunological reactions were observed in vitro, whereas PEEK and CoCrMo particle-induced immune responses were mainly mediated by CD8+ T-cells, rather than CD4+ T-cells, in vivo. PEEK wear particles induced fewer inflammatory reactions than CoCrMo particles, suggesting that PEEK was suitable as a potential alternative for metals in TKRs in terms of the relative immunological reaction to PEEK particles. Overall, the results of this study shed light on the effects of wear particles from polymer and metal-based implants on immune responses and demonstrate that determining the mechanism of T-cell activation may be important for developing therapeutic tools against aseptic loosening induced by PEEK and CoCrMo particles. Future studies should focus on the specific mechanisms of CD4 and CD8 lymphocyte-mediated osteolysis.
MATERIALS AND METHODS
Participants
The study included 25 participants (12 men and 13 women; average age: 62.1 years [range, 40–71 years] and 65.7 years [range, 54–75 years], respectively). Patients with knee osteoarthritis were chosen, as only they would need knee prosthesis, and their blood would be exposed to wear debris. Patients with known inflammatory diseases and recent infections were excluded. All participants provided written informed consent, and ethical approval for the protocols was received from the Renji Hospital, Shanghai Jiaotong University School of Medicine (grant no. 2017-021).
Generation of wear particles
Commercially available particles (BioEngineering Solutions, Oak Park, IL, USA) were generated from non-sterilized bulk material of a PEEK tibial tray and an highly cross-linked polyethylene (HXLPE) insert (Zeniva PEEK ZA-500, Chirulen HXLPE 1020X; Jiangsu Okani Medical Technology Co., Ltd., Soochow, JS, China), using proprietary techniques involving custom cryo-milling and pulverization. The CoCrMo wear debris was a gift from 3D Systems, Inc. (Rock Hill, SC, USA). The debris were isolated in ethanol and progressively filtered through polycarbonate membranes. Endotoxins were removed using Pyroclean. The debris were washed thrice in 70% ethanol, dried from a stock solution of more than 2 mg/100 μL of ethanol, and then tested for endotoxins. The particles were subjected to ethylene oxide (EtO) sterilization. Particles were then resuspended in ethanol and re-dried in a vacuum oven under sterile conditions.
Characterization of wear particles
Particle size analysis was performed using an SEM (PSEMII Aspex, Pittsburg, PA, USA) with a liquid sample circulator and an ultrasound dispersal system. The system was flushed 10 times with fresh reverse-osmosis-deionized water (> 18 MOhm) and sterile-filtered (0.2-μm pores) to remove potential contaminants. The particle-DH2O solution was added to the liquid circulator at 1 mL/s, to obtain the required laser obscuration value of less than 3%, as indicated by the automated software. The measurement integration time was 15 s, with three repeat measurements for each sample. Next, 100–200 μL from the remaining 1–2 mL of enzyme/acid-processed sample was vacuum-dried on a polycarbonate membrane (0.1–0.01-μm pores, GTTP; Thermo Fisher Scientific, Waltham, MA, USA). All the membranes were dried in a desiccator at 22°C for 24 h, prior to SEM analysis.
The total number of particles, regardless of size, was counted in at least 15 random image fields on each filter. The sizes and shapes of the particles were assessed by image analysis of the micrographs (Scion Image/NIH Image analysis software). The minimum number of particles in each sample was 450. The software program, Scion Image, was used to find the following measurements for each particle on the image: area, perimeter, ellipse major axis, and ellipse minor axis (for defining the particle dimensional measurements).
Challenging PBMCs with debris
A total of 20 mL of peripheral blood, with approximately 5 × 106 PBMCs, was isolated from each participant and subjected to density gradient centrifugation. The PBMCs were separated into five groups (two control groups and three experimental groups), and wear debris was added at a concentration of 20 particles/PBMC [17]. The negative control was unchallenged, being resuspended in RPMI 1640 medium (Thermo Fisher Scientific), and the positive control group was treated with human T-activator CD3/28 (Thermo Fisher Scientific), which is known to activate human T-cells. The PBMCs were incubated at 37°C and 5% CO2 for 72 h, and then analyzed by flow cytometry as described below.
T-cell phenotype and cytokine secretion
The T-cell populations and cytokine secretion were analyzed by immunostaining and flow cytometry, using fluorochrome-conjugated antibodies against CD3 (PB), CD8 (PerCP), interleukin (IL)-10 (PeCy7), IL-17 (PE), IL-2 (APC), and interferon (IFN)-γ (FITC) to identify Th1, Th2, Th17, and CD8+ T-cells. An anti-CD4 antibody was not used; CD3 positive and CD8 negative cell populations were identified as CD4+ T-cells in flow cytometry analyses (Supplementary Figure 1). All the antibodies were obtained from eBioscience (Thermo Fisher Scientific). Fluorescence staining was detected using a CyAn ADP flow cytometer (Beckton Dickinson) and analyzed using GraphPad Prism 5 software (La Jolla, CA, USA).
Animals
All the animal procedures and experiments were approved by the Animal Ethical Committee of the Renji Hospital, Shanghai Jiaotong University, School of Medicine (Shanghai, China). We obtained 30 female Sprague-Dawley rats, weighing 200–250 g. The rats were kept in a surgical research institution for one week prior to particle injection. They were kept in groups of three per cage and allowed food and water ad libitum. They were randomly assigned to one of three treatment groups: control (n = 10), PEEK particles (n = 10), and CoCrMo particles (n = 10).
Surgical procedures
Briefly, the rats were anesthetized by intraperitoneal injection of ketamine (10 mg/kg). Each rat was immobilized with the knee joint in the maximally flexed position, and the right leg was shaved and depilated. The two types of particle suspensions were sonicated for at least 60 min to avoid particle aggregation prior to injection. Each particle suspension (0.1 mL; approximately 1.0 × 108 wear particles) was then injected into the right knee of the experimental groups under sterile conditions. PBS (0.1 mL) was injected into the right knee of the control group. After surgery, the rats were housed in ventilated rooms with access to water and food.
Sample preparation
The rats were sacrificed one week post-surgery. The synovial tissues in the knee joints and spleen tissues were harvested and fixed in 4% buffered formaldehyde for histomorphometric observation and immunofluorescence labeling. The labeled area in the immunohistochemistry procedure was analyzed using Image-pro plus 6.0 (Media Cybernetics, Inc., Rockville, MD, USA). A total of six fields, at 200× original magnification of each slice, were digitized and transferred to the Image-pro plus 6.0 software. The area covered by positive cells (brown color) was determined, and the brown-labeled area was then divided by the area of cells and multiplied by 100.
Immunohistochemistry
For each knee synovial membrane, two sections were stained immunohistochemically with each primary antibody (CD4, CD8, IL-6, and TNF-α [R&D Systems, Minneapolis, MN, USA]). Additionally, for one section of each knee, PBS was used instead of the primary antibody, serving as negative control. After staining, the two samples of each primary antibody were evaluated semiquantitatively with a light microscope using different magnifications (10×, 20×; Carl Zeiss MicroImaging GmbH, Oberkochen, Germany) [43].
Double immunofluorescence staining of CD4+ and CD8+ T-cells in the spleen
The primary antibodies comprised a mixture of two antibodies (CD3 [rabbit antibodies] +CD4/CD8 [mouse antibodies]). For secondary antibodies, mixtures of Alexa Fluor Cy3-conjugated goat-anti-mouse IgG and Alexa Fluor 488-conjugated goat-anti rabbit IgG were used. Cells that showed double staining in the immunofluorescence procedure were manually counted in six fields at 1000× original magnification. The number of double-stained cells was then divided by the total number of cells and multiplied by 100 [44].
Statistical analysis
Data were analyzed using one-way analysis of variance (ANOVA) with LSD post-hoc t-tests. Mann-Whitney U or Wilcoxon rank sum tests were used for unpaired and paired nonparametric data. Differences with P < 0.05 were considered statistically significant.
Abbreviations
PEEK: Polyether-ether-ketone; CoCrMo: Cobalt-chromium-molybdenum; TKR: Total knee replacement; PE: Polyethylene; MHC: Major histocompatibility complex; APCs: Antigen-presenting cells; PBMCs: Peripheral blood mononuclear cells; SEM: Scanning electron microscope.
Author contributions
Zhe Du designed and performed the experiments, analyzed the data, and wrote the manuscript. Shujun Wang, Bing Yue, Ying Wang, and You Wang provided technical support and discussed the experimental strategy. You Wang contributed to the conception, wrote the manuscript, and supervised experiments.
ACKNOWLEDGMENTS
We thank Dr. Hallab from BioEngineering Solutions Inc. for consultant service in the generations of wear particles.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
FUNDING
This study was supported by the National Key R&D Program of China (Grant No. 2016YFC1101802). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The views and opinions expressed in this article are those of the authors only.
REFERENCES
1. Bin Abd Razak HR, Tan CS, Chen YJ, Pang HN, Tay KJ, Chin PL, Chia SL, Lo NN, Yeo SJ. Age and Preoperative Knee Society Score Are Significant Predictors of Outcomes Among Asians Following Total Knee Arthroplasty. J Bone Joint Surg Am. 2016; 98:735–41.
2. East RH, Briscoe A, Unsworth A. Wear of PEEK-OPTIMA(R) and PEEK-OPTIMA(R)-Wear Performance articulating against highly cross-linked polyethylene. Proc Inst Mech Eng H. 2015; 229:187–93.
3. Kretzer JP, Reinders J, Sonntag R, Hagmann S, Streit M, Jeager S, Moradi B. Wear in total knee arthroplasty—just a question of polyethylene?: Metal ion release in total knee arthroplasty. Int Orthop. 2014; 38:335–40.
4. Niki Y, Matsumoto H, Otani T, Suda Y, Toyama Y. Metal ion concentrations in the joint fluid immediately after total knee arthroplasty. Mod Rheumatol. 2001; 11:192–6.
5. Sagomonyants KB, Jarman-Smith ML, Devine JN, Aronow MS, Gronowicz GA. The in vitro response of human osteoblasts to polyetheretherketone (PEEK) substrates compared to commercially pure titanium. Biomaterials. 2008; 29:1563–72.
6. Kitamura E, Stegaroiu R, Nomura S, Miyakawa O. Biomechanical aspects of marginal bone resorption around osseointegrated implants: considerations based on a three-dimensional finite element analysis. Clin Oral Implants Res. 2004; 15:401–12.
7. Rho JY, Ashman RB, Turner CH. Young’s modulus of trabecular and cortical bone material: ultrasonic and microtensile measurements. Journal of biomechanics. 1993; 26:111–9.
8. Isidor F. Influence of forces on peri-implant bone. Clin Oral Implants Res. 2006; 17:8–18.
9. Huiskes R, Weinans H, van Rietbergen B. The relationship between stress shielding and bone resorption around total hip stems and the effects of flexible materials. Clinical orthopaedics and related research. 1992; 124–34.
10. Hunter A, Archer CW, Walker PS, Blunn GW. Attachment and proliferation of osteoblasts and fibroblasts on biomaterials for orthopaedic use. Biomaterials. 1995; 16:287–95.
11. Wenz LM, Merritt K, Brown SA, Moet A, Steffee AD. In vitro biocompatibility of polyetheretherketone and polysulfone composites. J Biomed Mater Res. 1990; 24:207–15.
12. Katzer A, Marquardt H, Westendorf J, Wening JV, von Foerster G. Polyetheretherketone—cytotoxicity and mutagenicity in vitro. Biomaterials. 2002; 23:1749–59.
13. Ponnappan RK, Serhan H, Zarda B, Patel R, Albert T, Vaccaro AR. Biomechanical evaluation and comparison of polyetheretherketone rod system to traditional titanium rod fixation. Spine J. 2009; 9:263–7.
14. Paulus AC, Hasselt S, Jansson V, Giurea A, Neuhaus H, Grupp TM, Utzschneider S. Histopathological Analysis of PEEK Wear Particle Effects on the Synovial Tissue of Patients. BioMed research international. 2016; 2016:2198914.
15. Li B, Hu Y, Zhao Y, Cheng M, Qin H, Cheng T, Wang Q, Peng X, Zhang X. Curcumin Attenuates Titanium Particle-Induced Inflammation by Regulating Macrophage Polarization In Vitro and In Vivo. Front Immunol. 2017; 8:55.
16. Sundfeldt M, Carlsson LV, Johansson CB, Thomsen P, Gretzer C. Aseptic loosening, not only a question of wear: a review of different theories. Acta Orthop. 2006; 77:177–97.
17. Pearson MJ, Williams RL, Floyd H, Bodansky D, Grover LM, Davis ET, Lord JM. The effects of cobalt-chromium-molybdenum wear debris in vitro on serum cytokine profiles and T cell repertoire. Biomaterials. 2015; 67:232–9.
18. Thierse HJ, Moulon C, Allespach Y, Zimmermann B, Doetze A, Kuppig S, Wild D, Herberg F, Weltzien HU. Metal-protein complex-mediated transport and delivery of Ni2+ to TCR/MHC contact sites in nickel-specific human T cell activation. J Immunol. 2004; 172:1926–34.
19. Hallab NJ, Caicedo M, Epstein R, McAllister K, Jacobs JJ. In vitro reactivity to implant metals demonstrates a person-dependent association with both T-cell and B-cell activation. Journal of biomedical materials research Part A. 2010; 92:667–82.
20. Case CP, Langkamer VG, Lock RJ, Perry MJ, Palmer MR, Kemp AJ. Changes in the proportions of peripheral blood lymphocytes in patients with worn implants. J Bone Joint Surg Br. 2000; 82:748–54.
21. Campbell P, Ebramzadeh E, Nelson S, Takamura K, De Smet K, Amstutz HC. Histological features of pseudotumor-like tissues from metal-on-metal hips. Clinical orthopaedics and related research. 2010; 468:2321–7.
22. Hallab NJ, McAllister K, Brady M, Jarman-Smith M. Macrophage reactivity to different polymers demonstrates particle size- and material-specific reactivity: PEEK-OPTIMA((R)) particles versus UHMWPE particles in the submicron, micron, and 10 micron size ranges. Journal of biomedical materials research Part B, Applied biomaterials. 2012; 100:480–92.
23. Hallab NJ, Bao QB, Brown T. Assessment of epidural versus intradiscal biocompatibility of PEEK implant debris: an in vivo rabbit model. Eur Spine J. 2013; 22:2740–51.
24. Purdue PE, Koulouvaris P, Potter HG, Nestor BJ, Sculco TP. The cellular and molecular biology of periprosthetic osteolysis. Clinical orthopaedics and related research. 2007; 454:251–61.
25. Fisher J, McEwen HM, Tipper JL, Galvin AL, Ingram J, Kamali A, Stone MH, Ingham E. Wear, debris, and biologic activity of cross-linked polyethylene in the knee: benefits and potential concerns. Clinical orthopaedics and related research. 2004; :114–9.
26. Utzschneider S, Becker F, Grupp TM, Sievers B, Paulus A, Gottschalk O, Jansson V. Inflammatory response against different carbon fiber-reinforced PEEK wear particles compared with UHMWPE in vivo. Acta biomaterialia. 2010; 6:4296–4304.
27. Stratton-Powell AA, Pasko KM, Brockett CL, Tipper JL. The Biologic Response to Polyetheretherketone (PEEK) Wear Particles in Total Joint Replacement: A Systematic Review. Clinical orthopaedics and related research. 2016; 474:2394–404.
28. Green TR, Fisher J, Matthews JB, Stone MH, Ingham E. Effect of size and dose on bone resorption activity of macrophages by in vitro clinically relevant ultra high molecular weight polyethylene particles. J Biomed Mater Res. 2000; 53:490–7.
29. Harlan DM, Abe R, Lee KP, June CH. Potential roles of the B7 and CD28 receptor families in autoimmunity and immune evasion. Clin Immunol Immunopathol. 1995; 75:99–111.
30. Arora A, Song Y, Chun L, Huie P, Trindade M, Smith RL, Goodman S. The role of the TH1 and TH2 immune responses in loosening and osteolysis of cemented total hip replacements. Journal of biomedical materials research Part A. 2003; 64:693–7.
31. Wallace WA, Ramage EA, Lamb D, Howie SE. A type 2 (Th2-like) pattern of immune response predominates in the pulmonary interstitium of patients with cryptogenic fibrosing alveolitis (CFA). Clin Exp Immunol. 1995; 101:436–41.
32. Chen DY, Chen YM, Chen HH, Hsieh CW, Lin CC, Lan JL. Increasing levels of circulating Th17 cells and interleukin-17 in rheumatoid arthritis patients with an inadequate response to anti-TNF-alpha therapy. Arthritis Res Ther. 2011; 13:R126.
33. Willert HG, Buchhorn GH, Fayyazi A, Flury R, Windler M, Koster G, Lohmann CH. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. A clinical and histomorphological study. J Bone Joint Surg Am. 2005; 87:28–36.
34. Hailer NP, Blaheta RA, Dahlstrand H, Stark A. Elevation of circulating HLA DR(+) CD8(+) T-cells and correlation with chromium and cobalt concentrations 6 years after metal-on-metal hip arthroplasty. Acta Orthop. 2011; 82:6–12.
35. Gao GF, Jakobsen BK. Molecular interactions of coreceptor CD8 and MHC class I: the molecular basis for functional coordination with the T-cell receptor. Immunol Today. 2000; 21:630–6.
36. Landgraeber S, von Knoch M, Loer F, Brankamp J, Tsokos M, Grabellus F, Schmid KW, Totsch M. Association between apoptotis and CD4(+)/CD8(+) T-lymphocyte ratio in aseptic loosening after total hip replacement. Int J Biol Sci. 2009; 5:182–91.
37. Kaufman AM, Alabre CI, Rubash HE, Shanbhag AS. Human macrophage response to UHMWPE, TiAlV, CoCr, and alumina particles: analysis of multiple cytokines using protein arrays. Journal of biomedical materials research Part A. 2008; 84:464–74.
38. Potnis PA, Dutta DK, Wood SC. Toll-like receptor 4 signaling pathway mediates proinflammatory immune response to cobalt-alloy particles. Cell Immunol. 2013; 282:53–65.
39. Ingham E, Fisher J. The role of macrophages in osteolysis of total joint replacement. Biomaterials. 2005; 26:1271–86.
40. Bainbridge JA, Revell PA, Al-Saffar N. Costimulatory molecule expression following exposure to orthopaedic implants wear debris. J Biomed Mater Res. 2001; 54:328–34.
41. Goodman SB. Wear particles, periprosthetic osteolysis and the immune system. Biomaterials. 2007; 28:5044–8.
42. Figgitt M, Newson R, Leslie IJ, Fisher J, Ingham E, Case CP. The genotoxicity of physiological concentrations of chromium (Cr(III) and Cr(VI)) and cobalt (Co(II)): an in vitro study. Mutat Res. 2010; 688:53–61.
43. Xing J, Ma J, Tang X, Sheng X, Zhan W. Characterizations of CD4-1, CD4-2 and CD8beta T cell subpopulations in peripheral blood leucocytes, spleen and head kidney of Japanese flounder (Paralichthys olivaceus). Mol Immunol. 2017; 85:155–65.
44. Ferreira LG, Rosin FC, Correa L. Analysis of Interleukin 17A in periapical abscess and granuloma lesions. Braz Oral Res. 2016; 30.

