INTRODUCTION
Advance in diagnosis and treatment of colorectal cancer (CRC) is not well translated into a favorable clinical outcome, and the disease-free survival of CRC remains poor [1]. A critical challenge in management of colorectal cancer is metastasis and relapse of the disease [2].
Cancer stem cells (CSCs) are a group of tumor cells with stem cell characteristics of self-renewal, infinite proliferation, and potential of multi-directional differentiation [3, 4]. The CSCs account for a very minor population of cancer, but are closely related to tumor metastasis, drug resistance and recurrence after primary treatment [5]. Traditional drugs and radiotherapy may make solid tumors relieved, but not kill CSCs. These CSCs may enter dormant status after treatment intervention and then become a source of cancer recurrence [6]. To date, CSCs are a great concern in treatment response and prognosis of various cancers, including colorectal cancer.
COLORECTAL CANCER STEM CELLS
CSCs were described first in acute myeloid leukemia (AML), but soon evidenced in solid tumors. The CSCs arise by gene mutations or deregulation of genetic programs in normal stem/progenitor cells [7]. In CRC, cells with epithelial characteristics, i.e., EpCAM high/CD44+ are isolated and these cells show stem cell-like properties, such as tumorigenesis, invasion and metastasis, indicating the epithelial source of CSCs [8]. In general, colorectal CSCs are mainly derived from differentiated intestinal cells or intestinal stem cells (ISCs) through gains of genetic alterations that are sufficient to induce malignant transformation [9].
Colorectal CSCs share the major biological characteristics of stem cells from other solid tumors [6], including 1) self-renewal and multi-directional differentiation potential, 2) abnormal activation of proliferating signaling pathways, such as Wnt, Notch and Hedgehog, 3) high tumorigenicity, and 4) strong drug and/or radiation resistance [10]. The colorectal CSCs also share many features of normal intestinal stem cells, such as infinite division, telomerase activity and organ-specific differentiation [11].
Cancer stem cells preferentially demonstrate persistent activation of multiple signal transduction pathways for stemness maintenance and self-renewal [12, 13]. The abnormal signaling pathways that have been well addressed in colorectal CSCs, including Wnt/β-catenin, Notch, TGF-β and Hedgehog (Figure 1) [14–16]. The Wnt/β-catenin pathway is particularly important in stemness maintenance and drug resistance of colorectal CSCs [17]. Wnt/β-catenin signaling is activated through binding of Wnt ligands with Frizzled receptor complex, and β-catenin, a transcription co-regulator, is a key effector of this Wnt/β-catenin signaling [18–20]. Whether Wnt/β-catenin signaling functions depends on the level and cellular location of β-catenin, and GSK-3β, a multifunctional kinase located in the regulatory APC/Axin/GSK-3β complex, is a negative regulator of β-catenin. This GSK-3β phosphorylates β-catenin and drives ubiquitination and proteasomal degradation through the β-TrCP/Skp pathway. Activation of the Wnt/receptor complex displaces GSK-3β from APC/Axin and thus stabilizes β-catenin. Accumulated β-catenin translocates into the nucleus, binds to LEF/TCF transcription factors, and drives target gene expression. In colorectal CSCs, the mutations that prevent formation of the APC/Axin/GSK-3β destruction complex lead to accumulation and nuclear translocation of β-catenin, activating the target genes involved in stemness and differentiation [21].
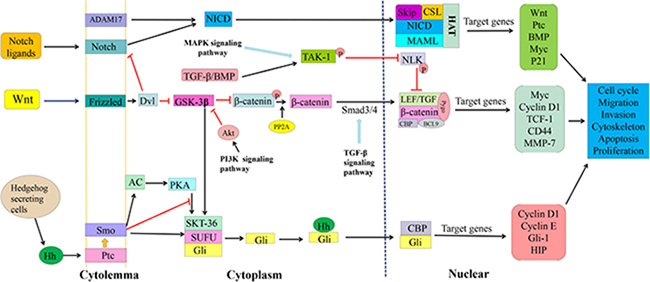
Figure 1: Wnt/β-catenin, Notch and Hedgehog signaling pathways and cross-talks in CSC cells. Wnt/β-catenin signaling is transmitted through Frizzled (FZD) receptor and inhibits disheveled (Dvl) and then glycogen synthase kinase-3β (GSK-3β), thereby stabilizing β–catenin. Accumulated β–catenin translocates into the nucleus where it binds to TCF/LEF transcription factors and form a complex of TCF/LEF/β-catenin/Pygo/CBP/BCL-9, regulating expression of target genes. Wnt/β-catenin signaling cross-talks with Notch, MAPK and TGF-β signaling. In Notch signaling, binding of Notch ligands to the receptor results in two proteolytic cleavages to release NICD. The released NICD then translocates into the nucleus where it interacts with the transcription factors, forming the complex of Skip/CSL/NICD/MAML/HAT to activate expression of Wnt, Ptc, BMP, Myc, and P21. Notch signaling cross-talks with the PI3K cascade. In Hedgehog signaling, Hh ligand secreted by Hedgehog secretory cells binds to PTCH1 (Ptc) and generates activated Gli/CBP that translocates into the nucleus and induce the expression of target genes, such as Cyclin D1, Cyclin E, Gli-1 and HIP. These main survival pathways and cross-talks among themselves and with other signaling pathways, i.e., TGF-β, MAPK and PI3K constitute a complex regulatory network for survival and proliferation of cancer stem cells.
Colorectal CSCs are identified via a group of surface markers. The main colorectal CSC markers documented are CD44, CD133, CD166, Lgr5, ALDH1 and EpCAM [8, 22]. Other more universal CSC markers include Nanog, Sox2, Oct-4, CD51, CD24, CD26 and CD29 [8, 22–29]. In addition to being a surface marker, these molecules are biologically functional. Table 1 summarizes the colorectal CSC markers and their cellular function identified thus far. Introduced below are several important colorectal CSC markers.
Table 1: Surface markers of colorectal cancer stem cells
Name of Marker |
Function |
References |
|---|---|---|
CD44 |
Cell surface glycoprotein involved in malignant progression, cell adhesion and migration, less sensitive to apoptosis signals and more resistance to therapies |
|
CD24 |
Cell adhesion |
|
CD133 |
Cell transmembrane glycoprotein involved in regulation of stemness, associated with cancer local recurrence and survival |
|
CD166 |
Cell adhesion molecule involved in neuronal extension, embryonic hematopoiesis and embryonic angiogenesis, associated with the development of adenoma to carcinoma |
|
Lgr-5 |
Expressed in intestinal stem cells and a downstream target of Wnt pathway, related to tumorigenesis, 5-fluorouracil resistance and recurrence of colorectal cancer |
|
EpCAM |
Cell adhesion molecule involved in Cadherin-Catenin and Wnt pathway, associated to lymph node metastasis, vascular invasion and distant metastasis |
|
ALDH1 |
Associated with poor differentiation, metastasis and drug resistance, correlated with advanced-stage of colorectal cancer |
|
Oct 4 |
Regulation of stemness, negatively correlated with cancer death, lymphatic invasion and lymph node metastasis |
|
SOX-2 |
Regulation of stemness, correlated with recurrence and lower disease-free survival |
[118] |
Nanog |
Transcriptional regulator, self-renewal |
|
CD29 |
Cell adhesion molecule regulating differentiation and self-renewal, involved in embryogenesis, hemostasis, tissue repair, immune response and cancer metastases |
|
CD26 |
Cell adhesion molecule involved in migration and invasion |
[28] |
CD51 |
Associated with the EMT of colorectal cancer cells, sphere formation, cell motility and tumor formation |
[26] |
CD44
CD44 is a hyaluronic acid receptor encoded by CD44 gene. It is a transmembrane glycoprotein that regulates cell-cell interaction, cell adhesion and migration [30]. CD44+ colon cancer cells display aggressive proliferation, high colony formation, insensitivity to apoptosis and resistance to chemo- and radiotherapies when compared to CD44 negative cells [22]. Knockdown of CD44 with short hairpin RNA (shRNA) leads to decrease of cell proliferation, migration and invasion, but to inhibition of apoptosis [31]. In the CD44-silenced HCT116 colon cancer cells, Bax was increased while Bcl-2 and Bcl-xL were downregulated, leading to cleavages of caspase-3, caspase-9 and PARP [31]. Therefore, this CD44 marker may be a potential therapeutic target of colorectal cancer.
CD133
CD133 (also named AC133 or prominin-1) is a transmembrane glycoprotein expressed in hematopoietic cells, endothelial cells and neuroepithelial cells [32]. CD133+ colorectal cancer cells show stem cell characteristics, such as self-renewal and multi-directional differentiation potential [8]. CD133 is considered a specific marker of primary colorectal CSCs and the CD133 expression is associated with colorectal cancer cell differentiation and tumor size [33]. CD133+ colorectal cancer cells are also resistant to radio- and chemotherapy [34, 35]. However, controversial results were reported, where the CD133- cells may be more aggressive [36, 37].
CD166
CD166 is a adhesion molecule of leucocytes, participating in intercellular interaction and cell adhesion with extracellular matrix [38]. In colorectal cancer, CD166 expression is correlated with the disease pathogenesis, being an early event in colon carcinogenesis [39]. However, CD166 expression was associated with a smaller size of the primary tumor in a meta-analysis [40]. As a CSC marker, therefore, CD166 needs to be considered together with other markers, such as CD44, CD24, CD29 and CD26 [8].
Lgr5
Leucine-rich repeat-containing G protein-coupled receptor 5 (Lgr5) belongs to the family of G protein-coupled receptors, which contains 17 leucine-rich repeats and a transmembrane domain of α-helix. Lgr-5 is a marker of normal intestinal stem cells [41] and also colorectal CSCs [42]. Lgr-5 plays an active role in pathogenesis of colorectal cancer [43], and the Lgr5 expression is closely related to tumorigenesis, 5-fluorouracil resistance and recurrence of colorectal cancer [43, 44]. In stage IV colorectal cancer, high Lgr5 expression is associated with poor prognosis [45]. Therefore, Lgr5 is considered an indicator of poor prognosis and a potential target of colorectal cancer.
ALDH1
Aldehyde dehydrogenase isoform 1 (ALDH1) catalyzes conversion of aldehyde to carboxylic acid. ALDH1 is often used as a surrogate marker of CSCs and non-CSCs in different cancers [46]. In colorectal cancer, ALDH1 serves as a CSC marker, and a high level of ALDH1 is associated with poor differentiation and high metastasis. ALDH1 is also a mediator of drug resistance in colorectal CSCs [47], and in rectal cancer, preoperative radiochemotherapy induces expression of ALDH1 [48]. However, opposite observations were reported in which loss of ALDH1 expression was correlated with advanced stage of colorectal cancer [49]. Further study may be needed to define its role in development and progression of colorectal cancer.
EpCAM
EpCAM is a 40 kDa single transmembrane protein encoded by tumor-associated calcium signal transducer-1 (TACSTD-1) gene [50]. EpCAM is involved in regulation of intercellular adhesion-mediated signaling transduction, cell migration, proliferation and differentiation [50]. As an epithelial adhesion molecule, EpCAM plays a role in carcinogenesis of epithelial cells by activating expression of proto-oncogenes c-myc and cyclin A/E [51]. In addition, EpCAM blocks antigen presentation of dendritic cells, driving escaping of the CD4+ T-cell dependent immune surveillance [52]. EpCAM can also potentiate the canonical WNT/β-catenin signaling cascade through intra-membrane proteolysis and subsequent nuclear translocation of its intracellular C-terminal domain, leading to cross-talk with the Notch, Hedgehog and TGFβ/BMP signaling cascades [53]. This constitutes the stem cell signaling network and regulates expression of other functional CSC markers [54–56]
COLORECTAL CSCS IN PROGRESSION OF COLORECTAL CANCER
CSC markers function in cell proliferation, metastasis and radio-/chemoresistance; and multiple cell proliferating cascades are activated in CSCs. Therefore, CSCs are critical to cancer progression and prognosis.
Colorectal CSCs and cancer metastasis
Distant metastasis of cancer cells is a complex process, including shedding and invading of primary cancer cells into circulatory system, migration and penetration of the vessel endothelial cell layer, tissue invasion, cell proliferation and angiogenesis [57]. Colorectal CSCs are “seed” cells for invasion and metastasis of colorectal cancer to distant organs, due to 1) infinite division, 2) plasticity to better adapt a new external microenvironment different from the primary tumor site, and 3) heterogeneity derived from asymmetric division, which produces a variety of heterogeneous tumor cells for the new microenvironmental selection. This also explains the heterogeneity of tumor cells in metastatic masses.
Migrating cancer stem cells and colorectal cancer metastasis
Not all CSCs in primary lesions are metastatic, and metastatic tumors are produced from a specific subpopulation of CSCs, named migrating cancer stem cells (MCSCs) [58, 59]. Brabletz’s and Oskarsson’s groups classified colorectal CSCs into two subgroups, i.e., stationary cancer stem cells (SCSCs) and MCSCs [60, 61]. The SCSCs exist in the epithelial tissues and are active even in benign precursor lesions. SCSCs mainly contribute to proliferation of the tumor mass in situ and remain in the differentiated area throughout tumor progression; they cannot disseminate [60]. The MCSCs lead to the rapidly invasive growth and dissemination of tumor cells [62]. MCSCs divide asymmetrically and generate differentiating cancer cells to start new proliferation and differentiation locally; the MCSCs then migrate a short distance and undergo a new asymmetric division to enlarge the primary tumor [63, 64]. SCSCs and other tumor cells can be transited into MCSCs in primary or metastatic tumor mass [65].
More interestingly, organ-specific metastases of cancer may be initiated by different MCSCs that have organ-unique characteristics. For example, CD110+ colorectal MCSCs are prone to colorectal-liver metastases (CRLM), but the colorectal MCSCs with a high level of CDCP1 are easier to colorectal-pulmonary metastases (CRPM) [11]. Nevertheless, specific surface markers of MCSCs are still under identification and further efforts are needed to accurately distinguish MCSCs and SCSCs. Furthermore, the CSCs may gradually evolve into MCSCs through epithelial mesenchymal transition (EMT) after formation of metastatic foci in distant organs [66].
EMT, CSCs and metastasis of colorectal cancer cells
Epithelial mesenchymal transition (EMT) is characterized by loss of epithelial morphology and markers but gains of mesenchymal features and markers. EMT is a basic process of organ development during the embryonic development [67]. Cancer cells that undergo EMT acquire stemness [68]. Indeed, non-CSCs acquire CSC-like characteristics, capacity of seeding tumors and surface markers through EMT [69]. The colorectal cancer cells that undergo EMT exhibit properties of EMT and CSCs, such as high expression of Snail, Lgr5, CD133, CD44 and EpCAM [70–73].
Signaling pathways involved in EMT, e.g., TGF-β, Wnt and Notch, also play roles in CSCs [74–76]. For instance, TGF-β1 induces expression of EMT markers (such as Slug, Twist1, β-catenin and N-cadherin) and also upregulates CSC markers (e.g., Oct4, Sox2, Nanog and Klf4) in colorectal cancer. Snail and Nanog signaling promotes EMT and acquisition of stemness in colorectal cancer cells, such as self-renewal, tumorigenicity, metastasis and drug resistance [77, 78]. The colorectal cancer cells with a high level of Nanog show stem cell properties and high expression of Slug, a driver of EMT through the IGF/STAT3/NANOG/Slug cascade.
CSCs and EMT processes interact at molecular levels [70]. CSC marker CD51 is co-localized with type I TGF-β receptor (TβRI) and type II TGF-β receptor (TβRII) and enhances the TGF-β dependent accumulation of p-Smad2/3 in the nucleus, which upregulates EMT-related genes, such as PAI1, MMP9 and Snail, and promotes sphere formation, cell motility and tumor formation [26]. Therefore, it is speculated that metastasis of colorectal cancer is due to the EMT of colorectal CSCs, leading to loss of epithelial characteristics and acquisition of mesenchymal phenotypes. This process offers colorectal CSCs the ability of migration and invasion through degradation of extracellular matrix and infiltration into distant organs [79].
Tumor microenvironment, colorectal CSCs and cancer metastasis
Microenvironment of stem cells is a physiological environment to maintain their biological features; aberrations of microenvironment can induce normal stem cells into cancer stem cells. The CSC microenvironment is complex, in which there are cytokines and molecules that promote development of CSCs and there are also factors that prevent CSCs (Figure 2). The pro-CSC cytokines, i.e., hepatocyte growth factor (HGF), prostaglandin E2 (PGE2), bone morphogenetic protein (BMP) and interleukins produced by the tumor microenvironment, increase the CSC pool [58]. For example, MFG-E8 secreted by tumor-associated macrophages maintains self-renewal of colorectal CSCs through the STAT3/Sonic Hedgehog signaling pathway; knockdown of MFG-E8 in the tumor-associated macrophages significantly inhibited tumorigenicity of CSCs in immunodeficient mice [80]. Oppositely, anti-CSC molecules decrease CSC number by forcing sequential differentiation into precursors [18]. Traditional chemotherapeutic agents are less effective in the presence of a pro-tumor microenvironment, but therapeutic agents that target CSC self-renewal or survival may be active.
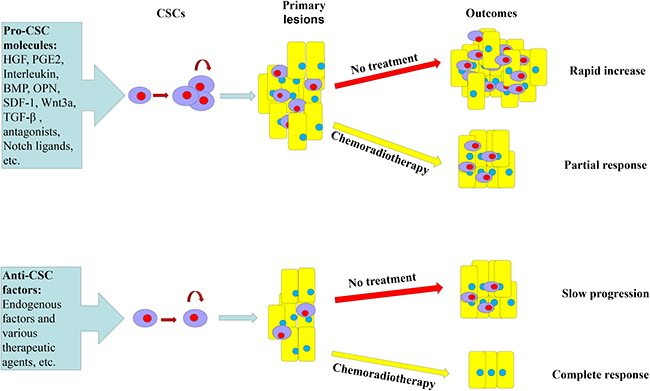
Figure 2: Microenvironmental molecules of colorectal CSCs. Microenvironmental molecules of CSCs include two groups: Pro-cancer stem cells (Pro-CSC) molecules and anti-cancer stem cells (anti-CSC) molecules. The Pro-CSC molecules in the tumor microenvironment promote proliferation of CSC while anti-CSC factors promote CSC differentiation, lowering down CSC number. Chemoradiotherapy is scarcely effective in the presence of a Pro-CSC tumor microenvironment and therapeutic molecules that target CSC self-renewal or survival may kill CSCs. HGF, hepatocyte growth factor; PGE2, prostaglandin E2; OPN, osteopontin; SDF1, stromal-cell-derived factor 1; BMP, bone morphogenetic protein; IL, interleukin; and TGF-β, transforming growth factor beta.
Distribution of cancer cells or cancer stem cells to specific organs is preferentially mediated by signals derived from the microenvironment, including oxygen gradient, chemokine receptors and cyclooxygenases [81–83]. Liver is a main target organ of colorectal cancer metastasis. In addition of the anatomical reason, chemokine receptor 4 (CXCR4) is an important factor involved in colorectal-liver metastases. CXCR4 is a specific receptor of stromal cell-derived factor 1 (SDF-1); the SDF-1 is highly expressed in the liver, which promotes the circulating CXCR4+ colorectal cancer cells to move into the liver [84]. TGF-β signaling pathway in hepatic stellate cells (HSCs) interacts with platelet-derived growth factor receptor alpha (PDGF-α), mediating proliferation and migration of CRC cells [85]. Thus HSCs with PDGF-α expression in the liver may form a microenvironment for colorectal-liver cancer metastasis. In addition, bone mesenchymal stem cells (MSCs) that migrate into the microenvironment of tumors in situ can differentiate into CRC-associated fibroblasts and promote cell invasion in the primary tumor site and metastasis to distant organs [86, 87]. Hence, CSCs have strong ability of migration and invasion, but the microenvironment provides specific biochemical factors for their metastasis to indicated tissues/organs with a specific microenvironment.
Colorectal CSCs and cancer resistance to treatment
Traditional treatments for CRC include surgery, radiotherapy and chemotherapy. Resistance of CRC to radiotherapy and chemotherapy is a major cause of treatment failure and cancer death. Radiation or chemotherapeutic drugs may effectively kill more differentiated no-CSCs in a mass, but have limited effects on CSCs. In fact, colorectal CSCs are widely resistant to radio- and chemotherapy, being a key factor of treatment resistance and cancer recurrence [88–91]. The discovery of colorectal CSCs highlights the intratumoral heterogeneity [92].
Exact mechanisms of colorectal CSC resistance to radiation and chemotherapeutic drugs remain to be fully understood, but several potential descriptions have been discussed [93]. First, CSCs may not enter the proliferating cycle, but be quiescent in the G0 phase and thus be resistant to radio- and chemotherapy [94]. Second, CSCs have enhanced ability of DNA damage repair and thus are resistant to DNA-damaging radiation and agents [95]. Third, CSCs express high levels of anti-apoptotic proteins, including Bcl-2 family members and apoptotic inhibitors, and thus are resistant to apoptosis [96]. Finally, CSCs express high levels of ABC transporters and p-glycoproteins that pump chemotherapeutic drugs out of cells [97]. Recently, galectin-3 (Gal3) is identified as a novel protein mediating resistance to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) through blockade of intracellular trafficking of death receptors [98]. The Gal3 expression is associated with increased sphere-forming ability (SFA), ALDH activity and tumorigenesis, and thus Gal3-positive colorectal CSCs are resistant to chemotherapy regimens, such as FOLFOX (5-fluorouracil, oxaliplatin and leucovorin) and FOLFIRI (5-fluorouracil, irinotecan and leucovorin) [98].
PER3 is a protein that is negatively involved in colorectal CSC drug resistance. The PER3 expression in colorectal CSCs and drug resistant HCT-116 cells is low, and induction of PER3 leads to inhibition of self-renewal and sensitivity to 5-fluorouracil of colorectal CSCs [24]. This works through suppression of β-catenin expression. Aberrant activation of Wnt/β-catenin signaling promotes proliferation of colorectal CSCs by upregulation of c-Myc and cyclin D1 [99]. PER3 inhibits β-catenin expression and thus leads to inhibition of self-renewal and sensitivity of CSCs to 5-fluorouracil. In addition, interleukin-6 (IL-6) stimulates stemness of colorectal CSCs and induces resistance to 5-fluorouracil through activation of Notch-3 signaling pathway [100, 101]. Anti-IL-6 therapy can reduce the expression of Oct-4, Klf4, Bmi-1, Lgr5 and Notch-3 and increase cell sensitivity to chemotherapeutic drugs [102].
Radiotherapy is a major means of CRC treatment, which kills cancer cells through DNA damage by ionizing radiation and resultant reactive oxygen species (ROS). Colorectal CSCs demonstrate unique characteristics, such as upregulated anti-apoptotic proteins, enhanced DNA damage repair and dormancy/slow cell cycle kinetics, and thus are radioresistant [103, 104]. To date, the described mechanisms that cause radioresistance of colorectal CSCs include enhanced DNA damage repair, decreased cell cycle activity, high ROS inhibitors, resistance to apoptosis and activation of survival pathways, e.g., Notch, c-Jun N-terminal kinase and protein kinase C δ signaling pathways [89, 105–107]. Radiation resistance of colorectal CSCs is also related to adaptive response induced by ionizing radiation and microenvironmental changes, such as cytokines, nitric oxide and oxygen contents [94]. In addtion, radiation can induce non-stem cancer cells to obtain the phenotypes and function of CSCs, leading to resistance to radiotherapy [108]. The radiotherapy may also induce cancer cells to undergo EMT, thus resulting in gain of stemness and radioresistance [109, 110]. Inhibition of NF-κB signaling blocks radiation-induced stemness, and inactivation of Notch signaling inhibits EMT via downregulation of Snail, enhancing sensitivity to radiotherapy [89, 111].
Colorectal CSCs and cancer recurrence
Traditional radiotherapy and chemotherapy can reduce the number of tumor cells and tumor volume, achieving remission of CRC on gross pathology, but the recurrence is high if the entire tumor is not surgically removed. Colorectal CSCs may be a key factor that promotes CRC recurrence. A principle limitation of current chemo-/radiotherapy is that it only eliminates more differentiated cancer cells, but not CSCs [112]. The CSCs tolerate or escape the destruction of chemotherapeutic agent and radiotherapy and survive. Due to their strong tumorigenicity, a small portion of survived CSCs in the quiescent status could re-enter into cell cycle for continuous proliferation by stimulation of appropriate signals in the microenvironment, leading to tumor recurrence. For instance, CD133+ colorectal cancer cells with high expression of ACBG2 and OCT-4 contribute to colorectal cancer recurrence [113].
CONCLUSIONS
High mortality of CRC is ascribed to metastasis, treatment resistance and recurrence. Understanding of colorectal CSCs has opened a window to cure this cancer. Through dedication of cancer research scientists in the past decades, knowledge of colorectal CSCs has been enriched in terms of their origins, biomarkers, signaling transduction and biological functions in tumor metastasis, treatment resistance and relapse (Figure 3). This provides a novel platform for development of new treatment modes to overcome the shortage of traditional therapies and achieve the purpose of curing cancer. However, due to lack of highly specific markers and complex biological characteristics of colorectal CSCs, effectively targeted therapies remain to be explored. Continuing efforts of scientists are needed for accurate and effective treatment targeted to CSCs in CRC.
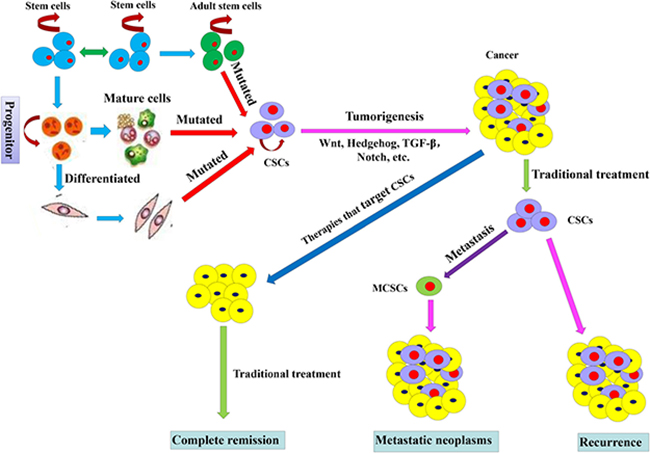
Figure 3: Origins and effects of colorectal CSCs on treatment. CSCs with sustained self-renewal, persistent proliferation and tumor initiation originate from mutated adult stem cells, mature cells and differentiated cells. CSCs play a role of pro-tumorigenesis due to the active Wnt, Hedgehog, TGF- beta and Notch signaling pathways. Traditional treatment cannot kill all CSCs, and the survival CSCs lead to the cancer relapse and form migrating cancer stem cells (MCSCs) which form metastatic neoplasms in the distant organs. Primary cancer that accepts therapies targeting CSCs can achieve complete remission.
Abbreviations
ABC transporter, ATP binding cassette transporter; ALDH1, aldehyde dehydrogenase isoform 1; BMP, bone morphogenetic protein; CSC, cancer stem cells; CCSC, circulating cancer stem cells; CRC, colorectal cancer; CRLM, colorectal-liver metastases; CRPM, colorectal-pulmonary metastases; CXCR4, chemokine receptor 4; EMT, epithelial mesenchymal transition; HGF, hepatocyte growth factor; HSC, hepatic stellate cells; ISC, intestinal stem cells; Lgr5, leucine-rich repeat-containing G protein-coupled receptor 5; MCSC, metastatic migrating cancer stem cells; MSC, mesenchymal stem cells; PGE2, prostaglandin E2; PDGF-α, platelet-derived growth factor receptor alpha; ROS, reactive oxygen species; SDF-1, stromal cell-derived factor; SFA, sphere-forming ability; SSCS, stationary cancer stem cells; TACSTD-1, tumor-associated calcium signal transducer-1; TCSC, tissue committed stem cells; TIC, tumor initiating cells; TRAIL, tumor necrosis factor-related apoptosis-inducing ligand.
Author contributions
YZ and LX prepared the draft. DC and QL revised and finalized the manuscript. Other authors enlisted helped with literature search and drafting.
CONFLICTS OF INTEREST
Authors declare no conflicts of interest.
FUNDING
This work was supported in part by grants from the National Natural Science Foundation of China (81402006 to ZY, 81472595 to LQ and 81472465 to DC) and the Natural Science Foundation of Hunan Province (2015JJ2094 to LQ).
REFERENCES
1. Ozgul Ozdemir RB, Ozdemir AT, Oltulu F, Kurt K, Yigitturk G, Kirmaz C. A comparison of cancer stem cell markers and nonclassical major histocompatibility complex antigens in colorectal tumor and noncancerous tissues. Ann Diagn Pathol. 2016; 25:60–3. https://doi.org/10.1016/j.anndiagpath.2016.09.012.
2. Zhu B, Zhou L, Yu L, Wu S, Song W, Gong X, Wang D. Evaluation of the correlation of vasculogenic mimicry, ALDH1, KAI1 and microvessel density in the prediction of metastasis and prognosis in colorectal carcinoma. BMC Surg. 2017; 17:47. https://doi.org/10.1186/s12893-017-0246-6.
3. Melo FS, Kurtova AV, Harnoss JM, Kljavin N, Hoeck JD, Hung J, Anderson JE, Storm EE, Modrusan Z, Koeppen H, Dijkgraaf GJ, Piskol R, de Sauvage FJ. A distinct role for Lgr5+ stem cells in primary and metastatic colon cancer. Nature. 2017; 543:676–80. https://doi.org/10.1038/nature21713.
4. Bu Y, Cao D. The origin of cancer stem cells. Front Biosci (Schol Ed). 2012; 4:819–30.
5. Shen Y, Cao D. Hepatocellular carcinoma stem cells: origins and roles in hepatocarcinogenesis and disease progression. Front Biosci (Elite Ed). 2012; 4:1157–69.
6. Hardin H, Zhang R, Helein H, Buehler D, Guo Z, Lloyd RV. The evolving concept of cancer stem-like cells in thyroid cancer and other solid tumors. Lab Invest. 2017; 97:1142–1151. https://doi.org/10.1038/labinvest.2017.41.
7. Lau EY, Ho NP, Lee TK. Cancer Stem Cells and Their Microenvironment: Biology and Therapeutic Implications. Stem Cells Int. 2017; 2017:3714190. https://doi.org/10.1155/2017/3714190.
8. Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW, Hoey T, Gurney A, Huang EH, Simeone DM, Shelton AA, Parmiani G, Castelli C, et al. Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci U S A. 2007; 104:10158–63. https://doi.org/10.1073/pnas.0703478104.
9. Basu S, Haase G, Ben-Ze’ev A. Wnt signaling in cancer stem cells and colon cancer metastasis. F1000Res. 2016; 5. https://doi.org/10.12688/f1000research.7579.1.
10. Cleophas MC, Joosten LA, Stamp LK, Dalbeth N, Woodward OM, Merriman TR. ABCG2 polymorphisms in gout: insights into disease susceptibility and treatment approaches. Pharmgenomics Pers Med. 2017; 10:129–42. https://doi.org/10.2147/PGPM.S105854.
11. Gao W, Chen L, Ma Z, Du Z, Zhao Z, Hu Z, Li Q. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology. 2013; 145:636–46 e5. https://doi.org/10.1053/j.gastro.2013.05.049.
12. de Sousa E Melo F, Vermeulen L. Wnt Signaling in Cancer Stem Cell Biology. Cancers (Basel). 2016; 8. https://doi.org/10.3390/cancers8070060.
13. Dorritie KA, Redner RL, Johnson DE. STAT transcription factors in normal and cancer stem cells. Adv Biol Regul. 2014; 56:30–44. https://doi.org/10.1016/j.jbior.2014.05.004.
14. Jaiswal AS, Marlow BP, Gupta N, Narayan S. Beta-catenin-mediated transactivation and cell-cell adhesion pathways are important in curcumin (diferuylmethane)-induced growth arrest and apoptosis in colon cancer cells. Oncogene. 2002; 21:8414–27. https://doi.org/10.1038/sj.onc.1205947.
15. Elamin MH, Shinwari Z, Hendrayani SF, Al-Hindi H, Al-Shail E, Khafaga Y, Al-Kofide A, Aboussekhra A. Curcumin inhibits the Sonic Hedgehog signaling pathway and triggers apoptosis in medulloblastoma cells. Mol Carcinog. 2010; 49:302–14. https://doi.org/10.1002/mc.20604.
16. Dandawate P, Padhye S, Ahmad A, Sarkar FH. Novel strategies targeting cancer stem cells through phytochemicals and their analogs. Drug Deliv Transl Res. 2013; 3:165–82. https://doi.org/10.1007/s13346-012-0079-x.
17. Takebe N, Harris PJ, Warren RQ, Ivy SP. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat Rev Clin Oncol. 2011; 8:97–106. https://doi.org/10.1038/nrclinonc.2010.196.
18. Todaro M, Gaggianesi M, Catalano V, Benfante A, Iovino F, Biffoni M, Apuzzo T, Sperduti I, Volpe S, Cocorullo G, Gulotta G, Dieli F, De Maria R, et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell. 2014; 14:342–56. https://doi.org/10.1016/j.stem.2014.01.009.
19. de Sousa E Melo F, Colak S, Buikhuisen J, Koster J, Cameron K, de Jong JH, Tuynman JB, Prasetyanti PR, Fessler E, van den Bergh SP, Rodermond H, Dekker E, van der Loos CM, et al. Methylation of cancer-stem-cell-associated Wnt target genes predicts poor prognosis in colorectal cancer patients. Cell Stem Cell. 2011; 9:476–85. https://doi.org/10.1016/j.stem.2011.10.008.
20. Li J, Yu B, Deng P, Cheng Y, Yu Y, Kevork K, Ramadoss S, Ding X, Li X, Wang CY. KDM3 epigenetically controls tumorigenic potentials of human colorectal cancer stem cells through Wnt/beta-catenin signalling. Nat Commun. 2017; 8:15146. https://doi.org/10.1038/ncomms15146.
21. Vadde R, Vemula S, Jinka R, Merchant N, Bramhachari PV, Nagaraju GP. Role of hypoxia-inducible factors (HIF) in the maintenance of stemness and malignancy of colorectal cancer. Crit Rev Oncol Hematol. 2017; 113:22–7. https://doi.org/10.1016/j.critrevonc.2017.02.025.
22. Wang JY, Chang CC, Chiang CC, Chen WM, Hung SC. Silibinin suppresses the maintenance of colorectal cancer stem-like cells by inhibiting PP2A/AKT/mTOR pathways. J Cell Biochem. 2012; 113:1733–43. https://doi.org/10.1002/jcb.24043.
23. Lopez-Arribillaga E, Rodilla V, Pellegrinet L, Guiu J, Iglesias M, Roman AC, Gutarra S, Gonzalez S, Munoz-Canoves P, Fernandez-Salguero P, Radtke F, Bigas A, Espinosa L. Bmi1 regulates murine intestinal stem cell proliferation and self-renewal downstream of Notch. Development. 2015; 142:41–50. https://doi.org/10.1242/dev.107714.
24. Zhang F, Sun H, Zhang S, Yang X, Zhang G, Su T. Overexpression of PER3 inhibits self-renewal capability and chemoresistance of colorectal cancer stem-like cells via inhibition of notch and beta-catenin signaling. Oncol Res. 2016; 25:709–719. https://doi.org/10.3727/096504016X14772331883976.
25. Rodda DJ, Chew JL, Lim LH, Loh YH, Wang B, Ng HH, Robson P. Transcriptional regulation of nanog by OCT4 and SOX2. J Biol Chem. 2005; 280:24731–7. https://doi.org/10.1074/jbc.M502573200.
26. Wang J, Zhang B, Wu H, Cai J, Sui X, Wang Y, Li H, Qiu Y, Wang T, Chen Z, Zhu Q, Xia H, Song W, et al. CD51 correlates with the TGF-beta pathway and is a functional marker for colorectal cancer stem cells. Oncogene. 2017; 36:1351–63. https://doi.org/10.1038/onc.2016.299.
27. Weichert W, Denkert C, Burkhardt M, Gansukh T, Bellach J, Altevogt P, Dietel M, Kristiansen G. Cytoplasmic CD24 expression in colorectal cancer independently correlates with shortened patient survival. Clin Cancer Res. 2005; 11:6574–81. https://doi.org/10.1158/1078-0432.CCR-05-0606.
28. Pang R, Law WL, Chu AC, Poon JT, Lam CS, Chow AK, Ng L, Cheung LW, Lan XR, Lan HY, Tan VP, Yau TC, Poon RT, et al. A subpopulation of CD26+ cancer stem cells with metastatic capacity in human colorectal cancer. Cell Stem Cell. 2010; 6:603–15. https://doi.org/10.1016/j.stem.2010.04.001.
29. Fujimoto K, Beauchamp RD, Whitehead RH. Identification and isolation of candidate human colonic clonogenic cells based on cell surface integrin expression. Gastroenterology. 2002; 123:1941–8. https://doi.org/10.1053/gast.2002.37065.
30. Spring FA, Dalchau R, Daniels GL, Mallinson G, Judson PA, Parsons SF, Fabre JW, Anstee DJ. The Ina and Inb blood group antigens are located on a glycoprotein of 80,000 MW (the CDw44 glycoprotein) whose expression is influenced by the In(Lu) gene. Immunology. 1988; 64:37–43.
31. Lee SY, Kim KA, Kim CH, Kim YJ, Lee JH, Kim HR. CD44-shRNA recombinant adenovirus inhibits cell proliferation, invasion, and migration, and promotes apoptosis in HCT116 colon cancer cells. Int J Oncol. 2017; 50:329–36. https://doi.org/10.3892/ijo.2016.3801.
32. Li YF, Xiao B, Tu SF, Wang YY, Zhang XL. Cultivation and identification of colon cancer stem cell-derived spheres from the Colo205 cell line. Braz J Med Biol Res. 2012; 45:197–204.
33. Kazama S, Kishikawa J, Kiyomatsu T, Kawai K, Nozawa H, Ishihara S, Watanabe T. Expression of the stem cell marker CD133 is related to tumor development in colorectal carcinogenesis. Asian J Surg. 2017 Feb 10. https://doi.org/10.1016/j.asjsur.2016.12.002. [Epub ahead of print].
34. Todaro M, Francipane MG, Medema JP, Stassi G. Colon cancer stem cells: promise of targeted therapy. Gastroenterology. 2010; 138:2151–62. https://doi.org/10.1053/j.gastro.2009.12.063.
35. Chen T, Zhang Y, Guo WH, Meng MB, Mo XM, Lu Y. Effects of heterochromatin in colorectal cancer stem cells on radiosensitivity. Chin J Cancer. 2010; 29:270–6.
36. O’Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007; 445:106–10. https://doi.org/10.1038/nature05372.
37. Shmelkov SV, Butler JM, Hooper AT, Hormigo A, Kushner J, Milde T, St Clair R, Baljevic M, White I, Jin DK, Chadburn A, Murphy AJ, Valenzuela DM, et al. CD133 expression is not restricted to stem cells, and both CD133+ and CD133- metastatic colon cancer cells initiate tumors. J Clin Invest. 2008; 118:2111–20. https://doi.org/10.1172/JCI34401.
38. Swart GW. Activated leukocyte cell adhesion molecule (CD166/ALCAM): developmental and mechanistic aspects of cell clustering and cell migration. Eur J Cell Biol. 2002; 81:313–21. https://doi.org/10.1078/0171-9335-00256.
39. Tachezy M, Zander H, Gebauer F, Marx A, Kaifi JT, Izbicki JR, Bockhorn M. Activated leukocyte cell adhesion molecule (CD166)—its prognostic power for colorectal cancer patients. J Surg Res. 2012; 177:e15–20. https://doi.org/10.1016/j.jss.2012.02.013.
40. Ni C, Zhang Z, Zhu X, Liu Y, Qu D, Wu P, Huang J, Xu AX. Prognostic value of CD166 expression in cancers of the digestive system: a systematic review and meta-analysis. PLoS One. 2013; 8:e70958. https://doi.org/10.1371/journal.pone.0070958.
41. Beumer J, Clevers H. Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Development. 2016; 143:3639–49. https://doi.org/10.1242/dev.133132.
42. He S, Zhou H, Zhu X, Hu S, Fei M, Wan D, Gu W, Yang X, Shi D, Zhou J, Zhou J, Zhu Z, Wang L, et al. Expression of Lgr5, a marker of intestinal stem cells, in colorectal cancer and its clinicopathological significance. Biomed Pharmacother. 2014; 68:507–13. https://doi.org/10.1016/j.biopha.2014.03.016.
43. Uchida H, Yamazaki K, Fukuma M, Yamada T, Hayashida T, Hasegawa H, Kitajima M, Kitagawa Y, Sakamoto M. Overexpression of leucine-rich repeat-containing G protein-coupled receptor 5 in colorectal cancer. Cancer Sci. 2010; 101:1731–7. https://doi.org/10.1111/j.1349-7006.2010.01571.x.
44. Hsu HC, Liu YS, Tseng KC, Hsu CL, Liang Y, Yang TS, Chen JS, Tang RP, Chen SJ, Chen HC. Overexpression of Lgr5 correlates with resistance to 5-FU-based chemotherapy in colorectal cancer. Int J Colorectal Dis. 2013; 28:1535–46. https://doi.org/10.1007/s00384-013-1721-x.
45. Wu W, Cao J, Ji Z, Wang J, Jiang T, Ding H. Co-expression of Lgr5 and CXCR4 characterizes cancer stem-like cells of colorectal cancer. Oncotarget. 2016; 7:81144–55. https://doi.org/10.18632/oncotarget.13214.
46. Crabb DW, Matsumoto M, Chang D, You M. Overview of the role of alcohol dehydrogenase and aldehyde dehydrogenase and their variants in the genesis of alcohol-related pathology. Proc Nutr Soc. 2004; 63:49–63.
47. Rassouli FB, Matin MM, Saeinasab M. Cancer stem cells in human digestive tract malignancies. Tumour Biol. 2016; 37:7–21. https://doi.org/10.1007/s13277-015-4155-y.
48. Deng Y, Zhou J, Fang L, Cai Y, Ke J, Xie X, Huang Y, Huang M, Wang J. ALDH1 is an independent prognostic factor for patients with stages II-III rectal cancer after receiving radiochemotherapy. Br J Cancer. 2014; 110:430–4. https://doi.org/10.1038/bjc.2013.767.
49. Hessman CJ, Bubbers EJ, Billingsley KG, Herzig DO, Wong MH. Loss of expression of the cancer stem cell marker aldehyde dehydrogenase 1 correlates with advanced-stage colorectal cancer. Am J Surg. 2012; 203:649–53. https://doi.org/10.1016/j.amjsurg.2012.01.003.
50. Trzpis M, McLaughlin PM, de Leij LM, Harmsen MC. Epithelial cell adhesion molecule: more than a carcinoma marker and adhesion molecule. Am J Pathol. 2007; 171:386–95. https://doi.org/10.2353/ajpath.2007.070152.
51. Maetzel D, Denzel S, Mack B, Canis M, Went P, Benk M, Kieu C, Papior P, Baeuerle PA, Munz M, Gires O. Nuclear signalling by tumour-associated antigen EpCAM. Nat Cell Biol. 2009; 11:162–71. https://doi.org/10.1038/ncb1824.
52. Zhou Y, Wang B, Wu J, Zhang C, Zhou Y, Yang X, Zhou J, Guo W, Fan J. Association of preoperative EpCAM Circulating Tumor Cells and peripheral Treg cell levels with early recurrence of hepatocellular carcinoma following radical hepatic resection. BMC Cancer. 2016; 16:506. https://doi.org/10.1186/s12885-016-2526-4.
53. Mani SK, Zhang H, Diab A, Pascuzzi PE, Lefrancois L, Fares N, Bancel B, Merle P, Andrisani O. EpCAM-regulated intramembrane proteolysis induces a cancer stem cell-like gene signature in hepatitis B virus-infected hepatocytes. J Hepatol. 2016; 65:888–98. https://doi.org/10.1016/j.jhep.2016.05.022.
54. Katoh M, Katoh M. WNT signaling pathway and stem cell signaling network. Clin Cancer Res. 2007; 13:4042–5. https://doi.org/10.1158/1078-0432.CCR-06-2316.
55. Katoh M, Nakagama H. FGF receptors: cancer biology and therapeutics. Med Res Rev. 2014; 34:280–300. https://doi.org/10.1002/med.21288.
56. Lamb R, Bonuccelli G, Ozsvari B, Peiris-Pages M, Fiorillo M, Smith DL, Bevilacqua G, Mazzanti CM, McDonnell LA, Naccarato AG, Chiu M, Wynne L, Martinez-Outschoorn UE, et al. Mitochondrial mass, a new metabolic biomarker for stem-like cancer cells: Understanding WNT/FGF-driven anabolic signaling. Oncotarget. 2015; 6:30453–71. https://doi.org/10.18632/oncotarget.5852.
57. Luzzi KJ, MacDonald IC, Schmidt EE, Kerkvliet N, Morris VL, Chambers AF, Groom AC. Multistep nature of metastatic inefficiency: dormancy of solitary cells after successful extravasation and limited survival of early micrometastases. Am J Pathol. 1998; 153:865–73.
58. Zeuner A, Todaro M, Stassi G, De Maria R. Colorectal cancer stem cells: from the crypt to the clinic. Cell Stem Cell. 2014; 15:692–705. https://doi.org/10.1016/j.stem.2014.11.012.
59. Sun X, Liu X. Cancer metastasis: enactment of the script for human reproductive drama. Cancer Cell Int. 2017; 17:51. https://doi.org/10.1186/s12935-017-0421-y.
60. Brabletz T, Jung A, Spaderna S, Hlubek F, Kirchner T. Opinion: migrating cancer stem cells - an integrated concept of malignant tumour progression. Nat Rev Cancer. 2005; 5:744–9. https://doi.org/10.1038/nrc1694.
61. Oskarsson T, Batlle E, Massague J. Metastatic stem cells: sources, niches, and vital pathways. Cell Stem Cell. 2014; 14:306–21. https://doi.org/10.1016/j.stem.2014.02.002.
62. Mukherjee S, Manna A, Bhattacharjee P, Mazumdar M, Saha S, Chakraborty S, Guha D, Adhikary A, Jana D, Gorain M, Mukherjee SA, Kundu GC, Sarkar DK, et al. Non-migratory tumorigenic intrinsic cancer stem cells ensure breast cancer metastasis by generation of CXCR4(+) migrating cancer stem cells. Oncogene. 2016; 35:4937–48. https://doi.org/10.1038/onc.2016.26.
63. Giroux V, Lento AA, Islam M, Pitarresi JR, Kharbanda A, Hamilton KE, Whelan KA, Long A, Rhoades B, Tang Q, Nakagawa H, Lengner CJ, Bass AJ, et al. Long-lived keratin 15+ esophageal progenitor cells contribute to homeostasis and regeneration. J Clin Invest. 2017; 127:2378–91. https://doi.org/10.1172/JCI88941.
64. Adamski V, Schmitt AD, Fluh C, Synowitz M, Hattermann K, Held-Feindt J. Isolation and Characterization of Fast-Migrating Human Glioma Cells in the Progression of Malignant Gliomas. Oncol Res. 2017; 25:341–53. https://doi.org/10.3727/096504016X14737243054982.
65. Liao WT, Ye YP, Deng YJ, Bian XW, Ding YQ. Metastatic cancer stem cells: from the concept to therapeutics. Am J Stem Cells. 2014; 3:46–62.
66. Tang DG. Understanding cancer stem cell heterogeneity and plasticity. Cell Res. 2012; 22:457–72. https://doi.org/10.1038/cr.2012.13.
67. Caramel J, Papadogeorgakis E, Hill L, Browne GJ, Richard G, Wierinckx A, Saldanha G, Osborne J, Hutchinson P, Tse G, Lachuer J, Puisieux A, Pringle JH, et al. A switch in the expression of embryonic EMT-inducers drives the development of malignant melanoma. Cancer Cell. 2013; 24:466–80. https://doi.org/10.1016/j.ccr.2013.08.018.
68. Zlobec I, Lugli A. Invasive front of colorectal cancer: dynamic interface of pro-/anti-tumor factors. World J Gastroenterol. 2009; 15:5898–906.
69. Chaffer CL, Brueckmann I, Scheel C, Kaestli AJ, Wiggins PA, Rodrigues LO, Brooks M, Reinhardt F, Su Y, Polyak K, Arendt LM, Kuperwasser C, Bierie B, et al. Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc Natl Acad Sci U S A. 2011; 108:7950–5. https://doi.org/10.1073/pnas.1102454108.
70. Shibue T, Weinberg RA. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat Rev Clin Oncol. 2017; 14:611–629. https://doi.org/10.1038/nrclinonc.2017.44.
71. Stavropoulou V, Kaspar S, Brault L, Sanders MA, Juge S, Morettini S, Tzankov A, Iacovino M, Lau IJ, Milne TA, Royo H, Kyba M, Valk PJ, et al. MLL-AF9 Expression in Hematopoietic Stem Cells Drives a Highly Invasive AML Expressing EMT-Related Genes Linked to Poor Outcome. Cancer Cell. 2016; 30:43–58. https://doi.org/10.1016/j.ccell.2016.05.011.
72. Puisieux A, Brabletz T, Caramel J. Oncogenic roles of EMT-inducing transcription factors. Nat Cell Biol. 2014; 16:488–94. https://doi.org/10.1038/ncb2976.
73. Brabletz T. EMT and MET in metastasis: where are the cancer stem cells? Cancer Cell. 2012; 22:699–701. https://doi.org/10.1016/j.ccr.2012.11.009.
74. Thien A, Prentzell MT, Holzwarth B, Klasener K, Kuper I, Boehlke C, Sonntag AG, Ruf S, Maerz L, Nitschke R, Grellscheid SN, Reth M, Walz G, et al. TSC1 activates TGF-beta-Smad2/3 signaling in growth arrest and epithelial-to-mesenchymal transition. Dev Cell. 2015; 32:617–30. https://doi.org/10.1016/j.devcel.2015.01.026.
75. Derynck R, Muthusamy BP, Saeteurn KY. Signaling pathway cooperation in TGF-beta-induced epithelial-mesenchymal transition. Curr Opin Cell Biol. 2014; 31:56–66. https://doi.org/10.1016/j.ceb.2014.09.001.
76. Yuan JH, Yang F, Wang F, Ma JZ, Guo YJ, Tao QF, Liu F, Pan W, Wang TT, Zhou CC, Wang SB, Wang YZ, Yang Y, et al. A long noncoding RNA activated by TGF-beta promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell. 2014; 25:666–81. https://doi.org/10.1016/j.ccr.2014.03.010.
77. Yao C, Su L, Shan J, Zhu C, Liu L, Liu C, Xu Y, Yang Z, Bian X, Shao J, Li J, Lai M, Shen J, et al. IGF/STAT3/NANOG/Slug Signaling Axis Simultaneously Controls Epithelial-Mesenchymal Transition and Stemness Maintenance in Colorectal Cancer. Stem Cells. 2016; 34:820–31. https://doi.org/10.1002/stem.2320.
78. Lee SY, Jeong EK, Ju MK, Jeon HM, Kim MY, Kim CH, Park HG, Han SI, Kang HS. Induction of metastasis, cancer stem cell phenotype, and oncogenic metabolism in cancer cells by ionizing radiation. Mol Cancer. 2017; 16:10. https://doi.org/10.1186/s12943-016-0577-4.
79. Meng Q, Wu W, Pei T, Li L, Tang X, Sun H. Novel markers for circulating tumor stem cells in colorectal carcinoma. Am J Transl Res. 2016; 8:4233–41.
80. Jinushi M, Chiba S, Yoshiyama H, Masutomi K, Kinoshita I, Dosaka-Akita H, Yagita H, Takaoka A, Tahara H. Tumor-associated macrophages regulate tumorigenicity and anticancer drug responses of cancer stem/initiating cells. Proc Natl Acad Sci U S A. 2011; 108:12425–30. https://doi.org/10.1073/pnas.1106645108.
81. Wuidart A, Ousset M, Rulands S, Simons BD, Van Keymeulen A, Blanpain C. Quantitative lineage tracing strategies to resolve multipotency in tissue-specific stem cells. Genes Dev. 2016; 30:1261–77. https://doi.org/10.1101/gad.280057.116.
82. Parsons BL, McKim KL, Myers MB. Variation in organ-specific PIK3CA and KRAS mutant levels in normal human tissues correlates with mutation prevalence in corresponding carcinomas. Environ Mol Mutagen. 2017; 58:466–476. https://doi.org/10.1002/em.22110.
83. Zheng F, Zhang Z, Flamini V, Jiang WG, Cui Y. The Axis of CXCR4/SDF-1 Plays a Role in Colon Cancer Cell Adhesion Through Regulation of the AKT and IGF1R Signalling Pathways. Anticancer Res. 2017; 37:4361–9. https://doi.org/10.21873/anticanres.11830.
84. Yamada S, Shimada M, Utsunomiya T, Morine Y, Imura S, Ikemoto T, Mori H, Arakawa Y, Kanamoto M, Iwahashi S, Saito Y. CXC receptor 4 and stromal cell-derived factor 1 in primary tumors and liver metastases of colorectal cancer. J Surg Res. 2014; 187:107–12. https://doi.org/10.1016/j.jss.2013.10.030.
85. Liu C, Li J, Xiang X, Guo L, Tu K, Liu Q, Shah VH, Kang N. PDGF receptor-alpha promotes TGF-beta signaling in hepatic stellate cells via transcriptional and posttranscriptional regulation of TGF-beta receptors. Am J Physiol Gastrointest Liver Physiol. 2014; 307:G749–59. https://doi.org/10.1152/ajpgi.00138.2014.
86. Shinagawa K, Kitadai Y, Tanaka M, Sumida T, Onoyama M, Ohnishi M, Ohara E, Higashi Y, Tanaka S, Yasui W, Chayama K. Stroma-directed imatinib therapy impairs the tumor-promoting effect of bone marrow-derived mesenchymal stem cells in an orthotopic transplantation model of colon cancer. Int J Cancer. 2013; 132:813–23. https://doi.org/10.1002/ijc.27735.
87. Shinagawa K, Kitadai Y, Tanaka M, Sumida T, Kodama M, Higashi Y, Tanaka S, Yasui W, Chayama K. Mesenchymal stem cells enhance growth and metastasis of colon cancer. Int J Cancer. 2010; 127:2323–33. https://doi.org/10.1002/ijc.25440.
88. Toden S, Tran HM, Tovar-Camargo OA, Okugawa Y, Goel A. Epigallocatechin-3-gallate targets cancer stem-like cells and enhances 5-fluorouracil chemosensitivity in colorectal cancer. Oncotarget. 2016; 7:16158–71. https://doi.org/10.18632/oncotarget.7567.
89. Li F, Zhou K, Gao L, Zhang B, Li W, Yan W, Song X, Yu H, Wang S, Yu N, Jiang Q. Radiation induces the generation of cancer stem cells: A novel mechanism for cancer radioresistance. Oncol Lett. 2016; 12:3059–65. https://doi.org/10.3892/ol.2016.5124.
90. Su YK, Shih PH, Lee WH, Bamodu OA, Wu ATH, Huang CC, Tzeng YM, Hsiao M, Yeh CT, Lin CM. Antrodia cinnamomea sensitizes radio-/chemo-therapy of cancer stem-like cells by modulating microRNA expression. J Ethnopharmacol. 2017; 207:47–56. https://doi.org/10.1016/j.jep.2017.06.004.
91. He YC, Zhou FL, Shen Y, Liao DF, Cao D. Apoptotic death of cancer stem cells for cancer therapy. Int J Mol Sci. 2014; 15:8335–51. https://doi.org/10.3390/ijms15058335.
92. Izumi D, Ishimoto T, Miyake K, Eto T, Arima K, Kiyozumi Y, Uchihara T, Kurashige J, Iwatsuki M, Baba Y, Sakamoto Y, Miyamoto Y, Yoshida N, et al. Colorectal Cancer Stem Cells Acquire Chemoresistance Through the Upregulation of F-Box/WD Repeat-Containing Protein 7 and the Consequent Degradation of c-Myc. Stem Cells. 2017; 35:2027–36. https://doi.org/10.1002/stem.2668.
93. Lu LL, Chen XH, Zhang G, Liu ZC, Wu N, Wang H, Qi YF, Wang HS, Cai SH, Du J. CCL21 Facilitates Chemoresistance and Cancer Stem Cell-Like Properties of Colorectal Cancer Cells through AKT/GSK-3beta/Snail Signals. Oxid Med Cell Longev. 2016; 2016:5874127. https://doi.org/10.1155/2016/5874127.
94. Huang TH, Wu SY, Huang YJ, Wei PL, Wu AT, Chao TY. The identification and validation of Trichosstatin A as a potential inhibitor of colon tumorigenesis and colon cancer stem-like cells. Am J Cancer Res. 2017; 7:1227–37.
95. Singh P, O’Connell M, Shubhashish S. Epigenetic regulation of human DCLK-1 gene during colon-carcinogenesis: clinical and mechanistic implications. Stem Cell Investig. 2016; 3:51. https://doi.org/10.21037/sci.2016.09.07.
96. Kangwan N, Kim YJ, Han YM, Jeong M, Park JM, Go EJ, Hahm KB. Sonic hedgehog inhibitors prevent colitis-associated cancer via orchestrated mechanisms of IL-6/gp130 inhibition, 15-PGDH induction, Bcl-2 abrogation, and tumorsphere inhibition. Oncotarget. 2016; 7:7667–82. https://doi.org/10.18632/oncotarget.6765.
97. Butler SJ, Richardson L, Farias N, Morrison J, Coomber BL. Characterization of cancer stem cell drug resistance in the human colorectal cancer cell lines HCT116 and SW480. Biochem Biophys Res Commun. 2017; 490:29–35. https://doi.org/10.1016/j.bbrc.2017.05.176.
98. Ilmer M, Mazurek N, Byrd JC, Ramirez K, Hafley M, Alt E, Vykoukal J, Bresalier RS. Cell surface galectin-3 defines a subset of chemoresistant gastrointestinal tumor-initiating cancer cells with heightened stem cell characteristics. Cell Death Dis. 2016; 7:e2337. https://doi.org/10.1038/cddis.2016.239.
99. Zhang T, Wang K, Zhang J, Wang X, Chen Z, Ni C, Qiu F, Huang J. Huaier aqueous extract inhibits colorectal cancer stem cell growth partially via downregulation of the Wnt/beta-catenin pathway. Oncol Lett. 2013; 5:1171–6. https://doi.org/10.3892/ol.2013.1145.
100. Ishihara K, Hirano T. IL-6 in autoimmune disease and chronic inflammatory proliferative disease. Cytokine Growth Factor Rev. 2002; 13:357–68.
101. Ying J, Tsujii M, Kondo J, Hayashi Y, Kato M, Akasaka T, Inoue T, Shiraishi E, Inoue T, Hiyama S, Tsujii Y, Maekawa A, Kawai S, et al. The effectiveness of an anti-human IL-6 receptor monoclonal antibody combined with chemotherapy to target colon cancer stem-like cells. Int J Oncol. 2015; 46:1551–9. https://doi.org/10.3892/ijo.2015.2851.
102. Kozlowska AK, Tseng HC, Kaur K, Topchyan P, Inagaki A, Bui VT, Kasahara N, Cacalano N, Jewett A. Resistance to cytotoxicity and sustained release of interleukin-6 and interleukin-8 in the presence of decreased interferon-gamma after differentiation of glioblastoma by human natural killer cells. Cancer Immunol Immunother. 2016; 65:1085–97. https://doi.org/10.1007/s00262-016-1866-x.
103. Rycaj K, Tang DG. Cancer stem cells and radioresistance. Int J Radiat Biol. 2014; 90:615–21. https://doi.org/10.3109/09553002.2014.892227.
104. Alison MR, Lin WR, Lim SM, Nicholson LJ. Cancer stem cells: in the line of fire. Cancer Treat Rev. 2012; 38:589–98. https://doi.org/10.1016/j.ctrv.2012.03.003.
105. Lagadec C, Vlashi E, Alhiyari Y, Phillips TM, Bochkur Dratver M, Pajonk F. Radiation-induced Notch signaling in breast cancer stem cells. Int J Radiat Oncol Biol Phys. 2013; 87:609–18. https://doi.org/10.1016/j.ijrobp.2013.06.2064.
106. Yoon CH, Kim MJ, Kim RK, Lim EJ, Choi KS, An S, Hwang SG, Kang SG, Suh Y, Park MJ, Lee SJ. c-Jun N-terminal kinase has a pivotal role in the maintenance of self-renewal and tumorigenicity in glioma stem-like cells. Oncogene. 2012; 31:4655–66. https://doi.org/10.1038/onc.2011.634.
107. Kim MJ, Kim RK, Yoon CH, An S, Hwang SG, Suh Y, Park MJ, Chung HY, Kim IG, Lee SJ. Importance of PKCdelta signaling in fractionated-radiation-induced expansion of glioma-initiating cells and resistance to cancer treatment. J Cell Sci. 2011; 124:3084–94. https://doi.org/10.1242/jcs.080119.
108. Ghisolfi L, Keates AC, Hu X, Lee DK, Li CJ. Ionizing radiation induces stemness in cancer cells. PLoS One. 2012; 7:e43628. https://doi.org/10.1371/journal.pone.0043628.
109. Chang L, Graham PH, Hao J, Bucci J, Cozzi PJ, Kearsley JH, Li Y. Emerging roles of radioresistance in prostate cancer metastasis and radiation therapy. Cancer Metastasis Rev. 2014; 33:469–96. https://doi.org/10.1007/s10555-014-9493-5.
110. Bastos LG, de Marcondes PG, de-Freitas-Junior JC, Leve F, Mencalha AL, de Souza WF, de Araujo WM, Tanaka MN, Abdelhay ES, Morgado-Diaz JA. Progeny from irradiated colorectal cancer cells acquire an EMT-like phenotype and activate Wnt/beta-catenin pathway. J Cell Biochem. 2014; 115:2175–87. https://doi.org/10.1002/jcb.24896.
111. Leong KG, Niessen K, Kulic I, Raouf A, Eaves C, Pollet I, Karsan A. Jagged1-mediated Notch activation induces epithelial-to-mesenchymal transition through Slug-induced repression of E-cadherin. J Exp Med. 2007; 204:2935–48. https://doi.org/10.1084/jem.20071082.
112. Vermeulen L, de Sousa e Melo F, Richel DJ, Medema JP. The developing cancer stem-cell model: clinical challenges and opportunities. Lancet Oncol. 2012; 13:e83–9.
113. Munakata K, Uemura M, Tanaka S, Kawai K, Kitahara T, Miyo M, Kano Y, Nishikawa S, Fukusumi T, Takahashi Y, Hata T, Nishimura J, Takemasa I, et al. Cancer Stem-like Properties in Colorectal Cancer Cells with Low Proteasome Activity. Clin Cancer Res. 2016; 22:5277–86. https://doi.org/10.1158/1078-0432.CCR-15-1945.
114. Lee CC, Kuo WH, Lin CK, Gao HW, Tsai WC. Discrimination of CD44 and Oct3/4 Expression in Pancreatic Adenocarcinoma from Benign Pancreatic Ducts in Small Biopsy Specimens. Chin J Physiol. 2017; 60:81–8. https://doi.org/10.4077/CJP.2017.BAF446.
115. Seo KJ, Kim M, Kim J. Prognostic implications of adhesion molecule expression in colorectal cancer. Int J Clin Exp Pathol. 2015; 8:4148–57.
116. Mirzaei H, Salehi H, Sahebkar A, Avan A, Jaafari MR, Namdar A, Rezaei A, Mirzaei HR. Deciphering biological characteristics of tumorigenic subpopulations in human colorectal cancer reveals cellular plasticity. J Res Med Sci. 2016; 21:64. https://doi.org/10.4103/1735-1995.187355.
117. Wahab SM, Islam F, Gopalan V, Lam AK. The Identifications and Clinical Implications of Cancer Stem Cells in Colorectal Cancer. Clin Colorectal Cancer. 2017; 16:93–102. https://doi.org/10.1016/j.clcc.2017.01.011.
118. Matsuoka J, Yashiro M, Sakurai K, Kubo N, Tanaka H, Muguruma K, Sawada T, Ohira M, Hirakawa K. Role of the stemness factors sox2, oct3/4, and nanog in gastric carcinoma. J Surg Res. 2012; 174:130–5. https://doi.org/10.1016/j.jss.2010.11.903.
119. Xu F, Dai C, Zhang R, Zhao Y, Peng S, Jia C. Nanog: a potential biomarker for liver metastasis of colorectal cancer. Dig Dis Sci. 2012; 57:2340–6. https://doi.org/10.1007/s10620-012-2182-8.
120. Zou J, Yu XF, Bao ZJ, Dong J. Proteome of human colon cancer stem cells: a comparative analysis. World J Gastroenterol. 2011; 17:1276–85. https://doi.org/10.3748/wjg.v17.i10.1276.
121. Vermeulen L, Todaro M, de Sousa Mello F, Sprick MR, Kemper K, Perez Alea M, Richel DJ, Stassi G, Medema JP. Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proc Natl Acad Sci U S A. 2008; 105:13427–32. https://doi.org/10.1073/pnas.0805706105.
122. Hynes RO. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992; 69:11–25.

