INTRODUCTION
Ependymomas are glial tumors that can arise anywhere in the neuroaxis and account for approximately 4% of all malignant tumors in the central nervous system (CNS) [1]. They are postulated to originate from radial glia cells in the subventricular zone [2] and result in the development of tumors in three major anatomic compartments: the supratentorial cranium, the infratentorial cranium, and the spine. Ependymomas, which are more common in pediatric patients than in adult patients, constitute 6–12% of all childhood CNS neoplasms (1). The anatomical location of ependymomas largely depends on patient age, with 90% of pediatric patients having cranial lesions [3] and 60% of adult patients having tumors in the spine [4, 5]. Although ependymal tumors from different locations are histologically indistinct, their clinical behaviors are highly variable [6, 7]. Recent studies have identified ependymoma molecular subtypes that contribute to observed differences in clinical outcome [3, 8, 9].
Regardless of their subtype or location, the cornerstone of treatment for all ependymomas is surgery. Gross total resection (GTR) is well established to be associated with improved patient survival [7, 10], and spinal ependymomas can often be cured with complete resection alone [3, 11]. The GTR of cranial tumors located near eloquent structures (e.g., the fourth ventricle, the brainstem) can be challenging, and the potential for devastating neurological outcomes may preclude GTR in such cases [11, 12]. In both adult and pediatric ependymoma patients, maximal safe resection followed by adjuvant focal radiation is the standard of care. However, the role of adjuvant chemotherapy in the treatment of ependymal tumors is less established, and its overall benefit is controversial [13, 14].
Owing to insufficient adjuvant treatment options, patients with recurrent ependymoma face a devastating clinical scenario and poor survival [15–17]. Ependymomas are notoriously chemoresistant, which greatly limits options for salvage therapy. Only a few studies have investigated the mechanisms governing this chemoresistance [18–20]. Although recent breakthroughs have improved our understanding of the genetic and molecular characteristics of ependymomas [3, 8, 9, 21], the mediators of chemoresistance in ependymomas remain largely unknown [20, 22]. The purpose of the present study was to determine the distribution of chemoresistance-related proteins in ependymoma. We performed multi-platform profiling on a cohort of these tumors and found ependymomas are enriched with proteins critical for DNA synthesis and repair. These findings suggest new potential treatment approaches and may help guide the stratification of ependymoma patients in clinical trials of chemotherapy.
RESULTS
Patient and tumor characteristics
Patient and tumor characteristics observed were consistent with those in previous reports [11, 23]. Of the 41 ependymomas included in our analysis (20 [49%]) from males and 21 [51%] from females), 33 (80%) were adult and 8 (20%) were pediatric. Most ependymomas (26 [63%]) were cranial; 15 (37%) were spinal. Among the adult ependymomas, 19 (58%) were cranial and 14 (42%) were spinal; whereas among pediatric ependymomas, 7 (88%) were cranial and only 1 (13%) was spinal. The patient population was equally dichotomized based on gender with 49% being male (n = 20). Cranial tumors were more frequent in female patients (16/21 [76%]) than in male patients (10/20 [50%]). Tumor location was significantly correlated with patient age. Overall, the median age at which cranial tumors occurred (28 years) was significantly lower than that at which spinal tumors occurred (47 years; p = 0.008).
Eight patients (19%) had grade I tumors, 11 (27%) had grade II tumors, and 22 (54%) had grade III tumors. Tumor grade and patient age were significantly associated (Figure 1), which is consistent with a previous study’s findings [24]. The median ages of patients with grade I, II, and III tumors (46, 38, and 29 years, respectively) differed significantly (p = 0.0213). There was also a relationship between tumor grade and location. Most grade I tumors were spinal (7/8 [88%]), whereas most grade II and III tumors were cranial (18/22 [82%]). All grade I spinal lesions were myxopapillary tumors. The percentages of cranial tumors that were grade I, II, or III (4% [1/26], 27% [7/26], and 69% [18/26], respectively) and those of spinal tumors that were grade I, II, or III (46% [7/15], 27% [4/15], and 27% [4/15], respectively) differed significantly (p = 0.0009).
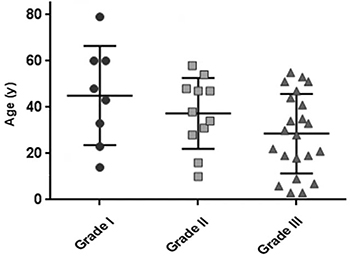
Figure 1: Scatterplot displaying the relationship between patient age and tumor grade.
Chemoresistance proteins are highly expressed in ependymomas
The rates of cancer-associated protein overexpression among ependymomas are given in Figure 2. Of the investigated proteins, PTEN was the most frequently expressed; specifically, 33 of 39 specimens (85%) showed PTEN overexpression. Five of 24 ependymomas (21%) had MGMT overexpression. Notably, ependymomas frequently overexpressed proteins implicated in DNA synthesis, transcription, and repair and/or drug resistance, including BCRP (5/7 [71%]), RRM1 (14/30 [47%]), ERCC1 (13/25 [48%]), TUBB3 (12/21 [57%]), TOPO1 (23/37 [62%]), and MRP1 (8/14 [43%]) [25–30]. Overall, 83% of the ependymomas overexpressed at least one DNA synthesis or repair protein or multi-drug resistance protein, and 80% overexpressed 2 or more.
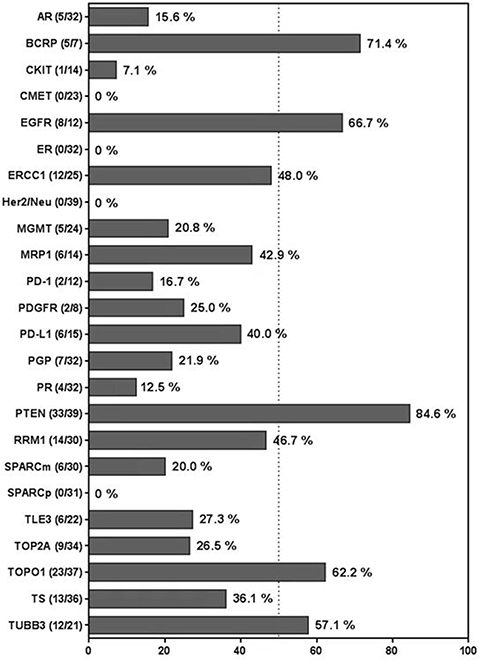
Figure 2: Bar graph displaying rates of cancer-associated protein overexpression among ependymomas.
Chemoresistance protein expression depends on ependymoma grade and location
The protein expression profiles of this ependymoma cohort by location, grade, and patient age are displayed in Figure 3. An analysis of the protein expression of 21 cranial and 15 spinal tumors revealed a significant association between TS expression and tumor location. The proportion of cranial ependymomas that overexpressed TS (57%) was significantly higher than that of spinal ependymomas that overexpressed the protein (15%; p = 0.0328) (Figure 3A). The proportion of cranial tumors that expressed EGFR and MRP1 was higher than that of spinal lesions; however, this difference was not statistically significant. In addition to its association with tumor location, TS overexpression was also correlated with tumor grade (Figure 3B). The frequency of TS overexpression among grade III tumors (65% [11/17]) was significantly higher than that among grade II tumors (18% [2/11]) and grade I tumors (0% [0/9]; p = 0.0009). Tumor grade was correlated with the overexpression of several additional proteins, including EGFR, which was overexpressed in 100%, 50%, and 0% of grade I, II, and III tumors, respectively (p = 0.0283); TOP2A, which was overexpressed in 47%, 10%, and 0% of grade I, II, and III tumors, respectively (p = 0.0092); and TUBB3, which was overexpressed in 80%, 50%, and 0% of grade I, II, and III tumors, respectively (p = 0.0157). Differences in protein expression between adult and pediatric cases did not differ significantly (Figure 3C).
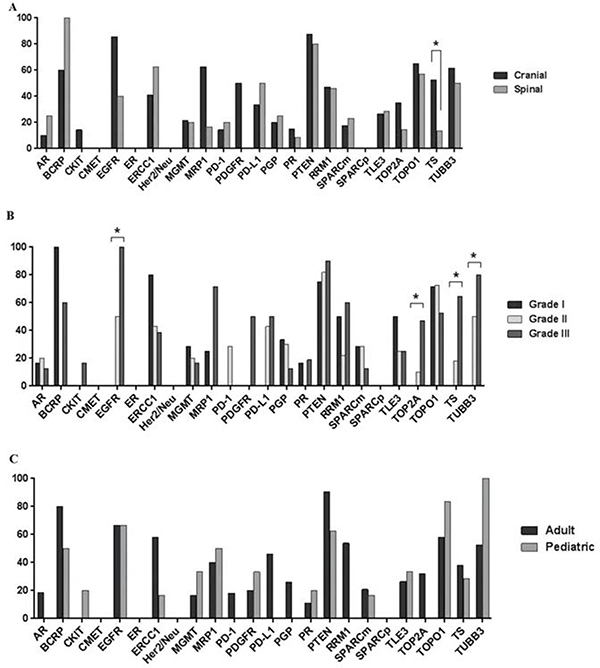
Figure 3: Bar graphs showing differential protein expression in ependymomas by (A) tumor location, (B) tumor grade, and (C) patient age. Asterisks indicate significant differences (p < 0.05)
Cranial and spinal ependymomas have differential expression of chemoresistance proteins
A subgroup analysis of cranial ependymomas revealed that the biomarker profiles of adult and pediatric tumors did not differ significantly (Figure 4A). However, tumor grade and TS expression were significantly correlated, with 69% of grade III tumors displaying TS overexpression compared with 0% of grade I lesions (p = 0.042) (Figure 4B). In a subgroup analysis of spinal ependymomas, high tumor grade was associated with increased TS and TOP2A expression (Figure 5). TS was exclusively overexpressed in grade III tumors, with no TS expression detected in grade I or II tumors (p = 0.0286). TOP2A expression was observed in 50% of grade III spinal tumors and 0% of grade II or I tumors (p = 0.0361).
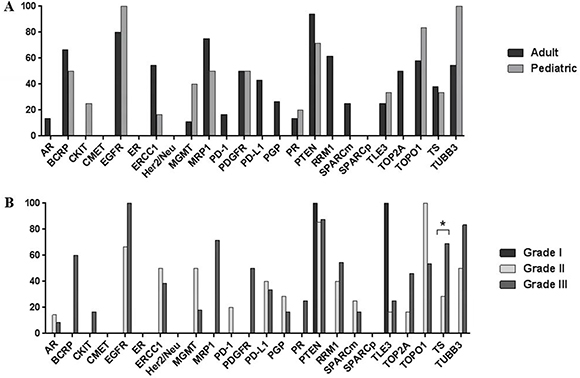
Figure 4: Bar graphs showing differential protein expression in cranial ependymomas by (A) patient age and (B) tumor grade. Asterisk indicates a significant difference (p < 0.05).
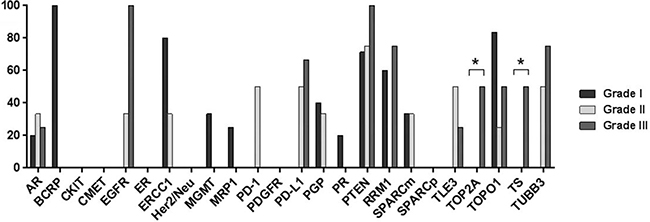
Figure 5: Bar graph showing differential protein expression in spinal ependymomas by tumor grade. Asterisks indicate significant differences (p < 0.05).
DISCUSSION
We found that proteins involved in DNA synthesis (e.g. RRM1), transcription (TOPO1), and repair (ERCC1) are enriched in ependymoma. As these proteins affect DNA metabolism, several are key mediators of chemoresistance in other solid malignancies [26, 27, 29]. Specifically, the expressions of RRM1, ERCC1, and TUBB3 are inversely correlated with tumor responses to anti-metabolites such as gemcitabine, platinum-based agents, and spindle poisons, respectively, which may help explain why these drugs have shown little efficacy in vivo [31] or in clinical studies [32, 33]. We also found transporter proteins such as BCRP and MRP1, which have roles in multidrug resistance, to be elevated in our ependymoma cohort. BCRP (also referred to as ABCG2) belongs to the ATP-binding cassette transporter family and reduces the intracellular concentrations of multiple chemotherapeutic agents, including doxorubicin, topotecan, and mitoxantrone [34]. Our findings are consistent with those of a smaller study that reported that the BCRP and MRP1 genes are expressed in ependymomas [35]. BCRP-mediated multi-drug resistance may be reversed by EGFR tyrosine kinase inhibitors, and several preclinical studies (utilizing breast, colon and small cell lung cancer cell lines) have demonstrated this phenomenon [36–38]. Thus, the combination of transporter inhibition plus chemotherapy may be a novel treatment paradigm for refractory ependymoma [39].
Our subgroup analyses of cranial tumors and of spinal tumors demonstrated that ependymomas at different locations have differential protein expression. For example, although ependymomas had an overall low expression rate of TS, a key enzyme in DNA biosynthesis and a known marker of cell proliferation and poor prognosis in other malignancies [40], cranial ependymomas had a significantly higher expression rate of TS than spinal ependymomas did. This was not unexpected as cranial lesions are known to display more aggressive clinical behavior. Low TS expression is correlated with response to 5-fluorouracil [41], which indicates that that 5-fluorouracil–based therapy might be efficacious in a select cohort of ependymoma patients. In addition, commercially available direct TS inhibitors such as pemetrexed have shown efficacy in other solid malignances [42] and have yielded favorable results in patients with non–small cell lung cancer brain metastases in phase II trials [43, 44] and may hold promise for ependymoma patients [45].
We also found that the expression of TS, TUBB3, and TOP2A were associated with tumor grade. These proteins are associated with aggressive behavior, poor patient prognosis, and chemoresistance in other malignancies [27, 40, 46], and our results indicate a similar pattern in ependymoma. Elevated TOP2A expression in ependymoma has not been reported previously; however, etoposide, a TOP2A inhibitor, has been used to treat recurrent ependymoma with very modest clinical results, likely because the agent has meager CNS penetration [47–49]. WP744 (berubicin), another TOP2A inhibitor able to penetrate the blood-brain barrier, has been tested in primary CNS malignancies with some success; however, additional studies of this agent in ependymoma may be warranted [50].
Even though this study is strengthened by a wide array of biomarkers analyzed, it is limited by the fact that it is commercial database, hence clinical data regarding patient outcome is not available. In addition, data regarding the molecular subtype (i.e. RELA and/or YAP1 fusion) of these ependymoma samples was unavailable for the majority of cases as physicians rarely requested this specific testing. Moreover, the effect of previous treatments on protein expression cannot be analyzed.
MATERIALS AND METHODS
Study population
We reviewed the Caris Life Sciences database and identified 41 ependymal tumors submitted to the company between 2009 and 2015 for multiplatform analysis (including mutational analysis, in situ hybridization, and immunohistochemistry) for biomarker profiling. WHO criteria were used to grade tumors. Because Caris Life Sciences maintains a commercial database, specimens are not clinically annotated; thus, outcome (e.g., survival) and prior treatment data were not available for the present study. Biomarkers analyzed varied by case owing to the preference of the ordering physician, tissue availability, and variation in technology over the study period. This study was exempt from Institutional Review Board approval as per 45 CFR 46.101(b), as the data analyzed were from an existing commercial repository and patients’ protected health information was de-identified.
Mutational analysis
Genomic DNA isolated from formalin-fixed, paraffin-embedded tumor tissue was sequenced with the Illumina MiSeq platform. The specific regions of 47 pan-cancer genes considered to be of interest and related to cancer genomics on the basis of current literature were amplified using the customized Illumina TruSeq Amplicon Cancer Hotspot panel. The hotspot regions of the following genes were sequenced: ABL1, AKT1, ALK, APC, ATM, BRAF, BRCA1, BRCA2, CDH1, cKIT, cMET, CSF1R, CTNNB1, EGFR, ERBB2, ERBB4, FBXW7, FGFR1, FGFR2, FLT3, GNA11, GNAS, GNAQ, HNF1A, HRAS, IDH1, JAK2, JAK3, KDR (VEGFR2), KRAS, MLH1, MPL, NOTCH1, NPM1, NRAS, PDGFRα, PIK3CA, PTEN, PTPN11, RB1, RET, SMAD4, SMARCB1, SMO, STK11, TP53, and VHL. All variants reported were detected with > 99% confidence based on the mutation frequency.
In situ hybridization
Fluorescence in situ hybridization (FISH) and chromogenic in situ hybridization were used to detect amplification of cMET (with the cMET/CEP7 probe and a Ventana kit) and HER2 (with the HER-2/CEP17 probe and INFORM HER-2 Dual ISH DNA Probe Cocktail). FISH was also used to detect EGFR amplification (with the EGFR/CEP7 probe). cMET was considered to be amplified if ≥ 5 copies per tumor cell were detected on average. A HER-2/CEP17 ratio of ≥ 2 was considered to indicate amplified HER2. EGFR amplification was defined by the presence of an EGFR/CEP7 ratio of ≥ 2 or ≥ 15 EGFR copies per cell in ≥ 10% of analyzed cells.
Immunohistochemistry
Formalin-fixed, paraffin-embedded tumor specimens were used for the immunohistochemical analysis. Antibodies against the following proteins were used: androgen receptor (AR), cMET, cKIT, epidermal growth factor receptor (EGFR), estrogen receptor (ER), excision repair cross-complementation group 1 (ERCC1), human epidermal growth factor receptor 2 (HER2), O(6)-methylguanine-methyltransferase (MGMT), p-glycoprotein (PGP), programmed cell death protein 1 (PD-1), programmed death-ligand 1 (PD-L1), platelet-derived growth factor receptor alpha (PDGFR), phosphatase and tensin homolog (PTEN), progesterone receptor (PR), ribonucleotide reductase M1 (RRM1), SPARC (monoclonal and polyclonal), thymidylate synthase (TS), topoisomerases 1 and 2 (TOPO1, TOP2A), transducin-like enhancer of split 3 (TLE3), and tubulin beta-3 chain (TUBB3). The primary antibody clones are listed in Supplementary Table 1. The conditions for staining were implemented using automated staining techniques and in accordance with the manufacturer’s instructions. Staining conditions were validated following the requirements of the Clinical Laboratory Improvement Amendments/Compliance Assistance Office and International Organization for Standardization. Immunohistochemical staining scores were based on staining percentage (0–100%) and intensity (0 = no staining; 1+ = weak staining; 2+ = moderate staining; 3+ = strong staining). Independent board-certified pathologists confirmed the results. PD-L1 staining was specific to membranous tumor cells, and PD-1 staining was specific to tumor-infiltrating lymphocytes.
Statistical analysis
Fisher’s exact test, Cochran-Mantel-Haenszel chi-squared test and Cochran Armitage test were used to assess differences in biomarker expression rates between groups. All analyses were exploratory, performed with R v3.3.1 with package DescTools v0.99.16. P values ≤0.05 were defined as significant.
CONCLUSIONS
Our findings demonstrate that chemoresistance-related proteins are markedly upregulated in ependymomas, and large-scale studies are needed to determine the extent to which this expression pattern is related to patient outcomes. However, our data suggest that a more tailored treatment approach based on biomarker expression may be warranted to better stratify ependymoma patients for specific therapies and clinical trials. Furthermore, an adjuvant treatment that combines conventional chemotherapy with drugs that inhibit DNA repair–related proteins or ATP-binding cassette transporter proteins may have enhanced clinical efficacy in appropriately selected patients.
Author contributions
Concept and design: Amy B. Heimberger, Joanne Xiu, David Spetzler, and Sherise D. Ferguson. Acquisition of data: Joanne Xiu and David Spetzler. Analysis and interpretation of data: Sherise D. Ferguson, Joanne Xiu, Amy B. Heimberger, Shouhao Zhou, Yuuri Hashimoto. Writing, review, and/or revision of the manuscript: Sherise D. Ferguson, Joanne Xiu, Santosh Kesari, Yuuri Hashimoto, Nader Sanai, John F. DeGroot, David Spetzler, Amy B. Heimberger contributed to writing and/or revision of this manuscript. Study supervision: Sherise D. Ferguson, Amy B. Heimberger. All authors reviewed the manuscript and approved the final version.
ACKNOWLEDGMENTS
We thank Audria Patrick for editorial assistance.
CONFLICTS OF INTEREST
Amy B. Heimberger serves on the Caris Life Sciences Scientific Advisory Board and is a stockholder in the company. Santosh Kesari serves as a consultant to Caris Life Sciences. Joanne Xiu is an employee of Caris Life Sciences. David is President and Chief Scientific Officer of Caris Life Science.
FUNDING
This study was supported by NIH grants P30CA16672, CA1208113 and by provost funds provided by Ethan Dmitrovsky.
REFERENCES
1. Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008–2012. Neuro Oncol. 2015; 17:iv1-iv62.
2. Taylor MD, Poppleton H, Fuller C, Su X, Liu Y, Jensen P, Magdaleno S, Dalton J, Calabrese C, Board J, Macdonald T, Rutka J, Guha A, et al. Radial glia cells are candidate stem cells of ependymoma. Cancer Cell. 2005; 8:323–35.
3. Pajtler KW, Witt H, Sill M, Jones DT, Hovestadt V, Kratochwil F, Wani K, Tatevossian R, Punchihewa C, Johann P, Reimand J, Warnatz HJ, Ryzhova M, et al. Molecular Classification of Ependymal Tumors across All CNS Compartments, Histopathological Grades, and Age Groups. Cancer Cell. 2015; 27:728–43.
4. Singh PK, Gutmann DH, Fuller CE, Newsham IF, Perry A. Differential involvement of protein 4.1 family members DAL-1 and NF2 in intracranial and intraspinal ependymomas. Mod Pathol. 2002; 15:526–31.
5. Zadnik PL, Gokaslan ZL, Burger PC, Bettegowda C. Spinal cord tumours: advances in genetics and their implications for treatment. Nat Rev Neurol. 2013; 9:257–66.
6. Korshunov A, Witt H, Hielscher T, Benner A, Remke M, Ryzhova M, Milde T, Bender S, Wittmann A, Schöttler A, Kulozik AE, Witt O, von Deimling A, et al. Molecular staging of intracranial ependymoma in children and adults. J Clin Oncol. 2010; 28:3182–90.
7. Merchant TE, Li C, Xiong X, Kun LE, Boop FA, Sanford RA. Conformal radiotherapy after surgery for paediatric ependymoma: a prospective study. Lancet Oncol. 2009; 10:258–66.
8. Ramaswamy V, Hielscher T, Mack SC, Lassaletta A, Lin T, Pajtler KW, Jones DT, Luu B, Cavalli FM, Aldape K, Remke M, Mynarek M, Rutkowski S, et al. Therapeutic Impact of Cytoreductive Surgery and Irradiation of Posterior Fossa Ependymoma in the Molecular Era: A Retrospective Multicohort Analysis. J Clin Oncol. 2016; 34:2468–77.
9. Witt H, Mack SC, Ryzhova M, Bender S, Sill M, Isserlin R, Benner A, Hielscher T, Milde T, Remke M, Jones DT, Northcott PA, Garzia L, et al. Delineation of two clinically and molecularly distinct subgroups of posterior fossa ependymoma. Cancer Cell. 2011; 20:143–57.
10. McLaughlin MP, Marcus RB Jr. Ependymoma: results, prognostic factors and treatment recommendations. Int J Radiat Oncol Biol Phys. 1998; 40:845–50.
11. McGuire CS, Sainani KL, Fisher PG. Incidence patterns for ependymoma: a surveillance, epidemiology, and end results study. J Neurosurg. 2009; 110:725–9.
12. Ferguson SD, Levine NB, Suki D, Tsung AJ, Lang FF, Sawaya R, Weinberg JS, McCutcheon IE. The surgical treatment of tumors of the fourth ventricle: a single-institution experience. J Neurosurg. 2017:1–13.
13. Grill J, Le Deley MC, Gambarelli D, Raquin MA, Couanet D, Pierre-Kahn A, Habrand JL, Doz F, Frappaz D, Gentet JC, Edan C, Chastagner P, Kalifa C. Postoperative chemotherapy without irradiation for ependymoma in children under 5 years of age: a multicenter trial of the French Society of Pediatric Oncology. J Clin Oncol. 2001; 19:1288–96.
14. Venkatramani R, Ji L, Lasky J, Haley K, Judkins A, Zhou S, Sposto R, Olshefski R, Garvin J, Tekautz T, Kennedy G, Rassekh SR, Moore T, et al. Outcome of infants and young children with newly diagnosed ependymoma treated on the “Head Start” III prospective clinical trial. J Neurooncol. 2013; 113:285–91.
15. Antony R, Wong KE, Patel M, Olch AJ, McComb G, Krieger M, Gilles F, Sposto R, Erdreich-Epstein A, Dhall G, Gardner S, Finlay JL. A retrospective analysis of recurrent intracranial ependymoma. Pediatr Blood Cancer. 2014; 61:1195–201.
16. Iqbal MS, Lewis J. An overview of the management of adult ependymomas with emphasis on relapsed disease. Clin Oncol (R Coll Radiol). 2013; 25:726–33.
17. Sowar K, Straessle J, Donson AM, Handler M, Foreman NK. Predicting which children are at risk for ependymoma relapse. J Neurooncol. 2006; 78:41–6.
18. Kim SK, Lim SY, Wang KC, Kim YY, Chi JG, Choi YL, Shin HJ, Cho BK. Overexpression of cyclooxygenase-2 in childhood ependymomas: role of COX-2 inhibitor in growth and multi-drug resistance in vitro. Oncol Rep. 2004; 12:403–9.
19. Korshunov A, Sycheva R, Timirgaz V, Golanov A. Prognostic value of immunoexpression of the chemoresistance-related proteins in ependymomas: an analysis of 76 cases. J Neurooncol. 1999; 45:219–27.
20. Phi JH, Choi SA, Kim SK, Wang KC, Lee JY, Kim DG. Overcoming Chemoresistance of Pediatric Ependymoma by Inhibition of STAT3 Signaling. Transl Oncol. 2015; 8:376–86.
21. Parker M, Mohankumar KM, Punchihewa C, Weinlich R, Dalton JD, Li Y, Lee R, Tatevossian RG, Phoenix TN, Thiruvenkatam R, White E, Tang B, Orisme W, et al. C11orf95-RELA fusions drive oncogenic NF-kappaB signalling in ependymoma. Nature. 2014; 506:451–5.
22. Chou PM, Barquin N, Gonzalez-Crussi F, Ridaura Sanz C, Tomita T, Reyes-Mugica M. Ependymomas in children express the multidrug resistance gene: immunohistochemical and molecular biologic study. Pediatr Pathol Lab Med. 1996; 16:551–61.
23. Amirian ES, Armstrong TS, Aldape KD, Gilbert MR, Scheurer ME. Predictors of survival among pediatric and adult ependymoma cases: a study using Surveillance, Epidemiology, and End Results data from 1973 to 2007. Neuroepidemiology. 2012; 39:116–24.
24. Koos B, Peetz-Dienhart S, Riesmeier B, Fruhwald MC, Hasselblatt M. O(6)-methylguanine-DNA methyltransferase (MGMT) promoter methylation is significantly less frequent in ependymal tumours as compared to malignant astrocytic gliomas. Neuropathol Appl Neurobiol. 2010; 36:356–8.
25. Abdallah EA, Fanelli MF, Souza ESV, Machado Netto MC, Gasparini Junior JL, Araujo DV, Ocea LM, Buim ME, Tariki MS, Alves Vda S, Piana de Andrade V, Dettino AL, Abdon Lopes de Mello C, et al. MRP1 expression in CTCs confers resistance to irinotecan-based chemotherapy in metastatic colorectal cancer. Int J Cancer. 2016; 139:890–8.
26. Davidson JD, Ma L, Flagella M, Geeganage S, Gelbert LM, Slapak CA. An increase in the expression of ribonucleotide reductase large subunit 1 is associated with gemcitabine resistance in non-small cell lung cancer cell lines. Cancer Res. 2004; 64:3761–6.
27. McCarroll JA, Sharbeen G, Liu J, Youkhana J, Goldstein D, McCarthy N, Limbri LF, Dischl D, Ceyhan GO, Erkan M, Johns AL, Biankin AV, Kavallaris M, et al. betaIII-tubulin: a novel mediator of chemoresistance and metastases in pancreatic cancer. Oncotarget. 2015; 6:2235–49.
28. Morfouace M, Cheepala S, Jackson S, Fukuda Y, Patel YT, Fatima S, Kawauchi D, Shelat AA, Stewart CF, Sorrentino BP, Schuetz JD, Roussel MF. ABCG2 Transporter Expression Impacts Group 3 Medulloblastoma Response to Chemotherapy. Cancer Res. 2015; 75:3879–89.
29. Ozcan MF, Dizdar O, Dincer N, Balci S, Guler G, Gok B, Pektas G, Seker MM, Aksoy S, Arslan C, Yalcin S, Balbay MD. Low ERCC1 expression is associated with prolonged survival in patients with bladder cancer receiving platinum-based neoadjuvant chemotherapy. Urol Oncol. 2013; 31:1709–15.
30. Pessina A. Topoisomerase I in multiple drug resistance. Cytotechnology. 1993; 12:127–35.
31. Atkinson JM, Shelat AA, Carcaboso AM, Kranenburg TA, Arnold LA, Boulos N, Wright K, Johnson RA, Poppleton H, Mohankumar KM, Féau C, Phoenix T, Gibson P, et al. An integrated in vitro and in vivo high-throughput screen identifies treatment leads for ependymoma. Cancer Cell. 2011; 20:384–99.
32. Brandes AA, Cavallo G, Reni M, Tosoni A, Nicolardi L, Scopece L, Franceschi E, Sotti G, Talacchi A, Turazzi S, Ermani M. A multicenter retrospective study of chemotherapy for recurrent intracranial ependymal tumors in adults by the Gruppo Italiano Cooperativo di Neuro-Oncologia. Cancer. 2005; 104:143–8.
33. Gramatzki D, Roth P, Felsberg J, Hofer S, Rushing EJ, Hentschel B, Westphal M, Krex D, Simon M, Schnell O, Wick W, Reifenberger G, Weller M. Chemotherapy for intracranial ependymoma in adults. BMC Cancer. 2016; 16:287.
34. Liang SC, Yang CY, Tseng JY, Wang HL, Tung CY, Liu HW, Chen CY, Yeh YC, Chou TY, Yang MH, Whang-Peng J, Lin CH. ABCG2 localizes to the nucleus and modulates CDH1 expression in lung cancer cells. Neoplasia. 2015; 17:265–78.
35. Ginguene C, Champier J, Maallem S, Strazielle N, Jouvet A, Fevre-Montange M, Ghersi-Egea JF. P-glycoprotein (ABCB1) and breast cancer resistance protein (ABCG2) localize in the microvessels forming the blood-tumor barrier in ependymomas. Brain Pathol. 2010; 20:926–35.
36. Dai CL, Tiwari AK, Wu CP, Su XD, Wang SR, Liu DG, Ashby CR Jr, Huang Y, Robey RW, Liang YJ, Chen LM, Shi CJ, Ambudkar SV, et al. Lapatinib (Tykerb, GW572016) reverses multidrug resistance in cancer cells by inhibiting the activity of ATP-binding cassette subfamily B member 1 and G member 2. Cancer Res. 2008; 68:7905–14.
37. Nakamura Y, Oka M, Soda H, Shiozawa K, Yoshikawa M, Itoh A, Ikegami Y, Tsurutani J, Nakatomi K, Kitazaki T, Doi S, Yoshida H, Kohno S. Gefitinib (“Iressa”, ZD1839), an epidermal growth factor receptor tyrosine kinase inhibitor, reverses breast cancer resistance protein/ABCG2-mediated drug resistance. Cancer Res. 2005; 65:1541–6.
38. Shi Z, Peng XX, Kim IW, Shukla S, Si QS, Robey RW, Bates SE, Shen T, Ashby CR Jr, Fu LW, Ambudkar SV, Chen ZS. Erlotinib (Tarceva, OSI-774) antagonizes ATP-binding cassette subfamily B member 1 and ATP-binding cassette subfamily G member 2-mediated drug resistance. Cancer Res. 2007; 67:11012–20.
39. Gilbert M, Wani K, Wu J, Omuro A, Lieberman F, Robins HI, Gerstner E, Wu J, Wen P, Mikkelsen T, Armstrong T, Aldape K. A PHASE II study of Lapatinib and dose dense Temozolomide (TMZ) for adults with recurrent ependymoma: a CERN clinical trial. Neuro Oncol. 2014;16: v13.
40. Koumarianou A, Tzeveleki I, Mekras D, Eleftheraki AG, Bobos M, Wirtz R, Fountzilas E, Valavanis C, Xanthakis I, Kalogeras KT, Basdanis G, Pentheroudakis G, Kotoula V. Prognostic markers in early-stage colorectal cancer: significance of TYMS mRNA expression. Anticancer Res. 2014; 34:4949–62.
41. Kakimoto M, Uetake H, Osanai T, Shirota Y, Takagi Y, Takeshita E, Toriya Y, Danenberg K, Danenberg PV, Sugihara K. Thymidylate synthase and dihydropyrimidine dehydrogenase gene expression in breast cancer predicts 5-FU sensitivity by a histocultural drug sensitivity test. Cancer Lett. 2005; 223:103–11.
42. Galvani E, Peters GJ, Giovannetti E. Thymidylate synthase inhibitors for non-small cell lung cancer. Expert Opin Investig Drugs. 2011; 20:1343–56.
43. Barlesi F, Gervais R, Lena H, Hureaux J, Berard H, Paillotin D, Bota S, Monnet I, Chajara A, Robinet G. Pemetrexed and cisplatin as first-line chemotherapy for advanced non-small-cell lung cancer (NSCLC) with asymptomatic inoperable brain metastases: a multicenter phase II trial (GFPC 07–01). Ann Oncol. 2011; 22:2466–70.
44. Dinglin XX, Huang Y, Liu H, Zeng YD, Hou X, Chen LK. Pemetrexed and cisplatin combination with concurrent whole brain radiotherapy in patients with brain metastases of lung adenocarcinoma: a single-arm phase II clinical trial. J Neurooncol. 2013; 112:461–6.
45. Kim L, Judy K, Farrell C, Andrews D. Pemetrexed: A potential new therapeutic option for the treatment of Thymidylate Synthase (TS) negative primary and systemic malignancies with CNS metastasis. Neuro Oncol 2016;18: vi118.
46. Wang J, Zhang J, Zhang L, Zhao L, Fan S, Yang Z, Gao F, Kong Y, Xiao GG, Wang Q. Expression of P-gp, MRP, LRP, GST-pi and TopoIIalpha and intrinsic resistance in human lung cancer cell lines. Oncol Rep. 2011; 26:1081–9.
47. Chamberlain MC. Recurrent intracranial ependymoma in children: salvage therapy with oral etoposide. Pediatr Neurol. 2001; 24:117–21.
48. Sandri A, Massimino M, Mastrodicasa L, Sardi N, Bertin D, Basso ME, Todisco L, Paglino A, Perilongo G, Genitori L, Valentini L, Ricardi U, Gandola L, et al. Treatment with oral etoposide for childhood recurrent ependymomas. J Pediatr Hematol Oncol. 2005; 27:486–90.
49. Sardi I, Sanzo M, Giordano F, Sandri A, Mussa F, Donati PA, Genitori L. Intracavitary chemotherapy (Gliadel) and oral low-dose etoposide for recurrent anaplastic ependymoma. Oncol Rep. 2008; 19:1219–23.
50. Conrad C, Madden T, Priebe W. An anthracycline analog, WP744, purposely designed to circumvent the blood-brain barrier appears to show activity in patients with recurrent high-grade gliomas in a Phase I study. Proceedings: 2015 SNO-SCIDOT Joint Conference. 2015.

