INTRODUCTION
Lung cancer is the leading cause of cancer deaths worldwide. Non-small cell lung cancer (NSCLC) accounts for approximately 80% of lung cancers, and the majority are already unresectable and metastatic upon their initial diagnosis. Cytotoxic chemotherapies such as platinum-based regimens were once the primary therapeutic option for metastatic NSCLC, but their advancement has reached a plateau. Molecular-targeted therapies have been recently developed, and they have provided a remarkable benefit to patients harboring specific genetic alterations such as epidermal growth factor receptor (EGFR) gene mutations or anaplastic lymphoma kinase (ALK) gene fusions [1–3]. Efficacies of up-front EGFR- and ALK-tyrosine kinase inhibitors (TKIs) have been established for patients harboring these genetic alterations in prospective randomized phase III trials comparing platinum doublets, and the median progression-free survivals (PFSs) are approximately 12 months [4–5]. Despite an initial dramatic response, most patients receiving these TKIs finally acquire resistance. Therefore, further salvage therapeutic options are necessary after failure of these molecular-targeted therapies.
On the other hand, current advancement of immunotherapies is evolving. Among them, anti-programmed death-1 (PD-1)/PD-ligand 1 (PD-L1) antibodies have demonstrated their splendid efficacies in pretreated NSCLC. Anti-PD-1/PD-L1 antibodies, such as nivolumab, pembrolizumab, and atezolizumab have shown survival benefit in pretreated patients with NSCLC after failure of platinum doublet chemotherapies, in randomized phase III trials compared to docetaxel monotherapy [6–9]. Based on results of these trials, anti-PD-1/PD-L1 antibody monotherapies have become standard treatments for pretreated NSCLC.
In cases responding to such immunotherapies, durable response is expected over 1-2 years, much longer than common cytotoxic agents [6–9]. Unfortunately, the response rate and PFS of these immunotherapies are generally 10-20% and 2-3 months, respectively, and relatively many patients obtain no response and experience early progression. Notably, several studies demonstrated a possible poorer efficacy of anti-PD-1 antibodies for patients with EGFR mutations [7–9]. However, such immunotherapies are not always ineffective even in EGFR-mutant NSCLC. Practical predictive markers are necessary to select patients who benefit from anti-PD-1/PD-L1 antibody immunotherapies.
Several predictive markers for anti-PD-1/PD-L1 antibodies have been developed [10]. Among them, PD-L1 expression is the most widely investigated predictive marker for many types of cancers. Some studies for NSCLC have demonstrated correlations between PD-L1 expression and efficacies of anti-PD-1/PD-L1 antibodies [7–9, 11]. One of them has shown that nivolumab was associated with longer overall survival, longer PFS, and higher objective response rates than docetaxel in pretreated NSCLC at the prespecified PD-L1 expression levels of ≥1%, ≥5%, and ≥10% [7].
We thus hypothesized lower PD-L1 expression in EGFR-mutant NSCLC samples than in EGFR-wild type. The aim of this study was to investigate correlation between PD-L1 expression and EGFR mutation status in pretreated NSCLC.
RESULTS
Sample and patient profile
Flow chart of final investigated samples and patients is shown in Figure 1. All studied samples were collected between January 2010 and October 2015. In the first cohort, 117 rebiopsies to obtain histologic tissue samples were done in 87 patients with NSCLC. Eleven rebiopsies were unsuccessful and failed to obtain malignant tissue samples. Three ALK-fusion positive samples were excluded, and a total 103 EGFR-mutant and wild-type tissue samples of NSCLC were examined. Fifteen samples were unavailable, and 8 samples contained insufficient cancer cells to perform PD-L1 IHC. We registered 80 rebiopsied histologic samples of 63 EGFR-mutant and wild-type. In the second cohort, 17 surgical tissue samples were available in 17 NSCLC patients receiving surgery after neoadjuvant chemoradiation therapy. After exclusion of one ALK-fusion positive sample, 16 of these surgical tissue samples were registered. Finally, we combined pretreated rebiopsy samples and surgical tissue samples, and the number of final investigated samples was 96 (65 EGFR-mutant and 31 wild-type) in 77 (47 EGFR-mutant and 30 wild-type) NSCLC patients.
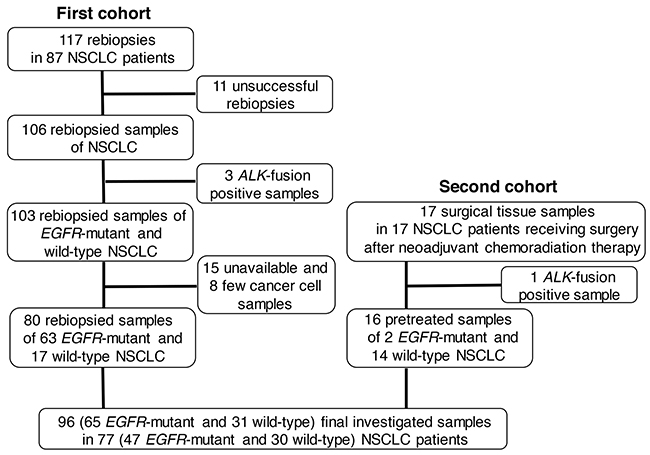
Figure 1: Flow chart of final investigated samples and patients. NSCLC, non-small cell lung cancer; ALK, anaplastic lymphoma kinase; EGFR, epidermal growth factor receptor.
Characteristics of final 77 patients investigated are shown in Table 1. Median age was 66 (range, 26-84). Approximately one-third of patients were never smoker. Most tumor histology was adenocarcinoma (63/77, 82%). Types of EGFR were: deletional mutation in exon 19 (20/77, 26%); L858R point mutation in exon 21 (25/77, 32%); L861Q point mutation in exon 21 (2/77, 3%), and wild-type (30/77, 39%). Radiotherapy before rebiopsy for sampled tissue was performed in 24 (31%) of patients. Median number of chemo-regimens before rebiopsy was 2 (range, 1–13). Approximately 90% of patients underwent cytotoxic chemotherapies. EGFR-TKIs were prescribed to 47 all EGFR-mutant and only 3 EGFR-wild-type patients. Rebiopsy was performed to lung lesions in approximately 80% of patients. Extra-lung lesions included: 8 lymph nodes (2 cervical, 2 supraclavicular, 2 axillary, 1 mediastinal, and 1 abdominal); 2 pleural; 2 liver; 2 rib; 1 muscle; and 1 adrenal metastases. Eleven (14%) patients underwent multiple rebiopsies. Median duration from rebiopsy/surgery to PD-L1 IHC evaluation (age of sample) was 21.3 (range, 3.5-71.1) months.
Table 1: Patient characteristics (n=77)
Characteristics |
Number (%) |
|---|---|
Age |
|
Median (range) |
66 (26-84) |
<70 |
48 (62%) |
70≤ |
29 (38%) |
Gender |
|
Male |
45 (58%) |
Female |
32 (42%) |
Smoking history |
|
Never |
22 (29%) |
Former |
27 (35%) |
Current |
28 (36%) |
Histology (initial rebiopsy) |
|
Adenocarcinoma |
63 (82%) |
Squamous/Large |
11/3 (18%) |
Types of EGFR mutation |
|
Exon 19 (deletion) |
20 (26%) |
Exon 21 (L858R) |
25 (32%) |
Exon 21 (L861Q) |
2 (3%) |
Wild-type |
30 (39%) |
Radiotherapy before rebiopsy for sampled tissue |
|
Irradiated |
25 (32%) |
Non-irradiated |
52 (68%) |
Number of chemo-regimens before rebiopsy |
|
Median (range) |
2 (1-13) |
Cytotoxic chemotherapy before rebiopsy |
|
Received |
67 (87%) |
None |
10 (13%) |
EGFR-TKIs before rebiopsy |
|
Prescribed |
50 (65%) |
None |
27 (35%) |
Rebiopsy site |
|
Lung |
61 (79%) |
Extra-lung |
16 (21%) |
Incidence of rebiopsy |
|
1 |
66 (86%) |
2/3/4/5 |
6/3/1/1 (14%) |
Age of sample (month) |
|
Median (range) |
21.3 (3.5-71.1) |
<12 months |
53 (69%) |
12 months≤ |
24 (31%) |
EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor.
Comparison of PD-L1 expression between EGFR-mutant and wild-type samples
Median H-score in EGFR-mutant samples (n=65) was 3 (range, 0-150), whereas EGFR-wild-type (n=31) was 8 (range, 0-134) (Wilcoxon, p=0.0075) (Figure 2). Using H-scores ≥1, ≥5, ≥10, ≥25, and ≥50 cut-offs, incidence of PD-L1+ in EGFR-mutant vs. EGFR-wild-type samples were 85% (55/65) vs. 94% (29/31) (p=0.2159), 42% (27/65) vs. 74% (23/31) (p=0.0027), 22% (14/65) vs. 48% (15/31) (p=0.0074), 5% (3/65) vs. 19% (6/31) (p=0.0205), and 3% (2/65) vs. 10% (3/31) (p=0.1735), respectively (Figure 3). Figure 4 shows PD-L1 expression of representative samples: A, EGFR-mutant (Del-19) (H-score: 0); B, EGFR-mutant (L858R) (H-score: 10); and C, EGFR wild-type (H-score: 134).
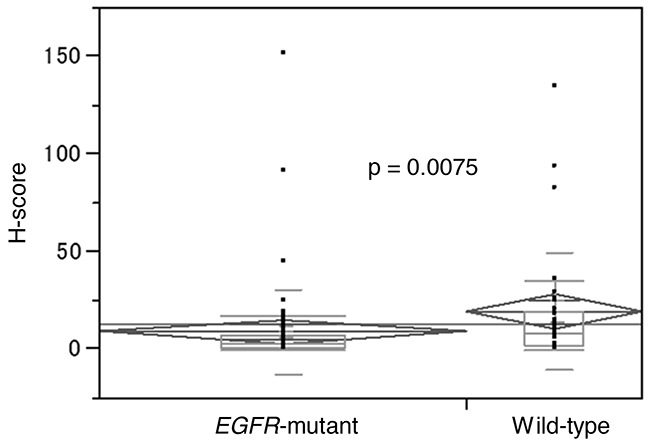
Figure 2: Comparison of H-scores between EGFR-mutant and wild-type samples using Wilcoxon rank sum test. EGFR, epidermal growth factor receptor.
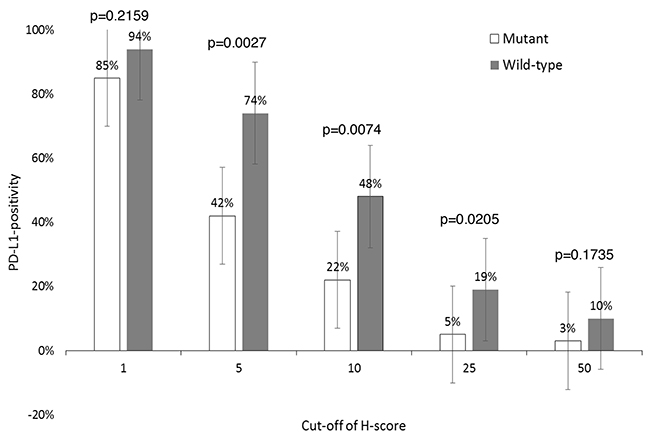
Figure 3: PD-L1-positivity according to EGFR status. PD-L1, programmed death-ligand 1; EGFR, epidermal growth factor receptor.
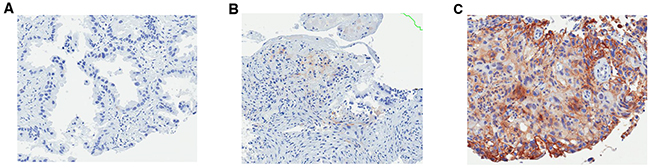
Figure 4: PD-L1 expression of representative samples: PD-L1, programmed death-ligand 1; EGFR, epidermal growth factor receptor. (A) EGFR-mutant (Del-19) (H-score: 0); (B) EGFR-mutant (L858R) (H-score: 10); (C) EGFR wild-type (H-score: 134).
Patient-oriented univariate and multivariate analyses for strong PD-L1+
Patient-oriented (n=77) univariate and multivariate analyses for strong PD-L1+ were performed at H-score ≥10 cut-off. In patients receiving multiple rebiopsy, first rebiopsy results were adopted in these analyses. Univariate analysis was performed on: age (<70 vs. 70≤); gender (male vs. female); smoking status (never vs. former vs. current); histology (adeno vs. non-adeno); EGFR mutation status (mutant vs. wild-type); radiation before rebiopsy for sampled tissue (irradiated vs. non-irradiated); cytotoxic chemotherapy before rebiopsy (received vs. none) rebiopsy site (lung vs. extra-lung); and age of sample (<12 months vs. 12 months≤). EGFR-TKIs before rebiopsy (prescribed vs. none) was eliminated because of strong confounding to EGFR mutation status. Univariate analysis found EGFR mutation status (p=0.0490) and age of sample (p=0.0226) as significant factors for strong PD-L1+ (Table 2). Results of multivariate analysis using the logistic regression model are shown in Table 3. We identified EGFR status as the only significant factor for strong PD-L1+ (odds ratio, 2.99; 95% confidence interval, 1.34-7.56; and p=0.0121).
Table 2: Univariate analysis for strong PD-L1+
Characteristics |
PD-L1-positivity (%) |
P-value |
|---|---|---|
Age |
||
<70 |
20/48 (41.7%) |
0.1440 |
| 70≤ |
7/29 (24.1%) |
|
Gender |
||
Male |
16/45 (35.6%) |
NS |
Female |
11/32 (34.4%) |
|
Smoking history |
||
Never |
7/22 (31.8%) |
NS |
Former |
10/27 (37.0%) |
|
Current |
10/28 (35.7%) |
|
Histology (initial rebiopsy) |
||
Adenocarcinoma |
23/63 (36.5%) |
0.7592 |
Squamous/Large |
4/14 (28.6%) |
|
Types of EGFR mutation |
||
Mutant |
14/47 (25.5%) |
0.0490 |
Wild-type |
15/30 (50.0%) |
|
Radiotherapy before rebiopsy for sampled tissue |
||
Irradiated |
9/25 (36.0%) |
NS |
Non-irradiated |
18/52 (34.6%) |
|
Cytotoxic chemotherapy before rebiopsy |
||
Received |
24/67 (35.8%) |
NS |
None |
3/10 (30.0%) |
|
Rebiopsy site |
||
Lung |
19/61 (31.2%) |
0.2384 |
Extra-lung |
8/16 (50.0%) |
|
Age of sample (month) |
||
<12 months |
14/53 (26.4%) |
0.0226 |
12 months≤ |
13/24 (54.2%) |
PD-L1, programmed death-ligand 1; EGFR, epidermal growth factor receptor; NS, not significant.
Table 3: Multivariate analysis for strong PD-L1+
Variable |
P-value |
Odds ratio (95% CI) |
|
|---|---|---|---|
Age |
(<70 vs. 70≤) |
0.1135 |
0.62 (0.33-1.10) |
Smoking history |
(Current vs. Former/Never) |
0.1063 |
1.81 (0.92-3.95) |
Histology |
(Adeno vs. Non-adeno) |
0.2268 |
0.64 (0.27-1.32) |
EGFR mutation status |
(Mutant vs. Wild-type) |
0.0121 |
2.99 (1.34-7.56) |
Radiation |
(Irradiated vs. Non-irradiated) |
0.1867 |
1.59 (0.83-3.33) |
Age of sample |
(<12 months vs. 12 months≤) |
0.2408 |
1.41 (0.77-2.51) |
PD-L1, programmed death-ligand 1; EGFR, epidermal growth factor receptor; CI, confidence interval.
PD-L1 expression in multiple rebiopsied cases
Eleven (14%) patients underwent multiple rebiopsies. H-scores of PD-L1 expression varied in all 11 cases receiving multiple rebiopsies (Table 4). PD-L1 expression increased in 7 (64%) patients, whereas decreased in 6 (55%) patients. Categories of positivity: negative (H-score=0); weak+ (1≤ H-score <5); moderate+ (5≤ H-score <10): strong+ (10≤ H-score) migrated in 10 (91%) of 11 patients.
Table 4: PD-L1 expression in multiple rebiopsied cases
Case |
EGFR |
Location |
H-score at 1st Rebiopsy |
H-score at 2nd Rebiopsy |
H-score at 3rd Rebiopsy |
H-score at 4th Rebiopsy |
|---|---|---|---|---|---|---|
#1 |
Mutant |
Primary |
0 |
0 |
0 |
3 |
Metastasis |
0 |
|||||
#2 |
Mutant |
Primary |
6 |
|||
Metastasis |
2 |
10 |
||||
#3 |
Mutant |
Primary |
2 |
18 |
||
Metastasis |
8 |
|||||
#4 |
Mutant |
Primary |
0 |
1 |
2 |
7 |
#5 |
Mutant |
Primary |
2 |
1 |
2 |
|
#6 |
Mutant |
Primary |
6 |
0 |
||
#7 |
Mutant |
Primary |
3 |
5 |
||
#8 |
Mutant |
Primary |
1 |
0 |
||
#9 |
Mutant |
Primary |
0 |
3 |
||
#10 |
Mutant |
Primary |
0 |
1 |
||
#11 |
Wild |
Primary |
19 |
7 |
PD-L1, programmed death-ligand 1; EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor.
DISCUSSION
We herein demonstrate a lower PD-L1 expression in EGFR-mutant NSCLC samples than in EGFR wild-type samples. Using H-scores ≥1, ≥5, and ≥10 cut-offs, incidences of PD-L1+ in EGFR-mutant samples was also less than in wild-type samples. These results suggest poorer efficacy of anti-PD-1/PD-L1 immunotherapies in EGFR-mutant than in wild-type. Gainor et al have reported that NSCLC harboring EGFR mutations or ALK-fusions were associated with low overall response rate to PD-1/PD-L1 inhibitors [12]. They have also showed lower PD-L1 expression of tissues in these driver oncogene-positive populations than in EGFR/ALK wild-type population. Our IHC results and their data both support subgroup analyses of clinical studies which showed poorer efficacy of anti-PD-1/PD-L1 immunotherapies in EGFR-mutant subgroup [7–9].
Meanwhile, some studies using surgical samples of chemo-naïve NSCLC have shown lower PD-L1 expression in EGFR-mutant NSCLC than in EGFR wild-type, as we have demonstrated [13, 14]. Others have exhibited higher PD-L1 expression in EGFR-mutant NSCLC than in EGFR wild-type [15–18]. This issue is still controversial and debatable. Notably, our study and several clinical studies have found a possible temporal heterogeneity of PD-L1 expression by therapeutic interventions, especially EGFR-TKIs [12, 19]. Preclinical studies have also shown that PD-L1 expression was reduced by EGFR-TKIs in NSCLC cell lines harboring EGFR activating mutations [20, 21]. Not chemo-naïve surgical samples, but pretreated histological samples (EGFR-mutant: after EGFR-TKI therapies) are more desirable for studies to investigate PD-L1 expression. Moreover, anti-PD-1 antibodies, nivolumab and pembrolizumab are approved only in pretreated patients with EGFR-mutant NSCLC. To properly investigate this issue, it is preferable to examine pretreated samples for best reflection of study results into clinical practice. Thus, our study focused on histological samples of pretreated NSCLC patients, which is more clinically valuable than studies using chemo-naïve surgical samples.
Our multivariate analysis identified EGFR status as the only significant factor for strong PD-L1+. Based on results of pivotal studies regarding nivolumab, PD-L1 expression was associated with clinical efficacies in non-squamous NSCLC population [7]. Although ascertaining PD-L1 expression could be beneficial for non-squamous NSCLC, PD-L1 IHC using 28-8 antibody is uncommon in current clinical practice. We can only utilize clinically available predictive markers. Smoking history and histology might have a predictive value [10, 13, 22], but our multivariate analysis did not reveal a statistical significance. A meta-analysis indicated PD-L1 expression was not associated with common clinicopathological characteristics such as smoking history and histology, except tumor differentiation [23]. Clinical practice demands a routine EGFR mutational analysis, which is performed in most cases. Therefore, EGFR status could be useful as a predictive marker of anti-PD-1/PD-L1 antibody therapies in pretreated patients with NSCLC.
Age of sample was identified as a significant factor for strong PD-L1+ in our univariate analysis, but multivariate analysis failed to confirm this result. Older samples revealed lesser prevalence of strong PD-L1+. This result implies a possible underestimation of PD-L1 expression in older samples. Several studies suggested that tissue processing and storage could alter the ability to detect PD-L1 in tumor samples [24]. The decreased prevalence may be caused by PD-L1 protein denaturation with formalin fixation and a loss in PD-L1 antigenicity. Age of sample could result in loss of detection of PD-L1 [25]. Based on these studies and our results, rebiopsied fresh samples may be better for PD-L1 IHC.
Issues of PD-L1 IHC contain not only tissue processing and storage but also interpretation of the test by pathologists. Reproducibility is another issue in PD-L1 IHC scoring. We adopted a digital pathological systematic procedure (Aperio). This system can digitally evaluate PD-L1 IHC scores, and demonstrated highly similar IHC staining results to visual evaluation by a pathologist [26]. Our pathologists also confirmed PD-L1 IHC score of each sample, and PD-L1 H-scores were similar between digital procedure and pathologists.
Our study includes several limitations. First is the selection of antibody for PD-L1 IHC. Our study adopted the 28-8 PD-L1 antibody. Four anti PD-L1 antibodies (28-8, 22c3, SP142, and SP263) are clinically used for PD-L1 IHC, but PD-L1 IHC is not globally standardized [27]. Each IHC antibody has been developed simultaneously with each anti-PD-1/PD-L1 therapeutic antibody (nivolumab, pembrolizumab, atezolizumab, and durvalumab). In order to translate basic data for clinical practice, one of these four antibodies for PD-L1 IHC should be used in clinical studies to investigate PD-L1 expression. At present, nivolumab is one of the most widely used immunotherapies in Japan, and thus the 28-8 antibody is an optimal antibody choice for PD-L1 IHC. Second is our cut-offs for PD-L1 positivity. We adopted H-score to evaluate both percentage and intensity, and defined H-scores ≥1 as PD-L1+, scores ≥5 as moderate PD-L1+, and scores ≥10 as strong PD-L1+, largely equivalent to PD-L1 expression ≥1%, ≥5%, and ≥10% cut-offs. Pivotal studies using nivolumab determined PD-L1 expression ≥1%, ≥5%, and ≥10% as their cut-offs [6, 7], and a study for non-squamous NSCLC has demonstrated significant correlation between PD-L1+ status and clinical efficacy of nivolumab [7]. Although optimal cut-off for PD-L1 positivity is yet to be determined, we are sure that our adopted cut-off is appropriate.
In conclusion, our study has demonstrated a significantly lower PD-L1 expression in EGFR-mutant NSCLC samples than in EGFR wild-type samples. As several studies have shown [7–9], efficacies of anti-PD-1/PD-L1 immunotherapies in EGFR-mutant population appear to be poorer than those in EGFR wild-type population. Priority of anti-PD-1/PD-L1 immunotherapies might be lower in EGFR-mutant population than in EGFR wild-type population. Our multiple rebiopsied cases suggested PD-L1 expression dynamism. Age of sample could affect PD-L1 expression, and rebiopsied fresh samples may be better for PD-L1 IHC. Further studies are warranted to investigate association between PD-L1 expression and EGFR mutation status.
MATERIALS AND METHODS
Samples and patients
We retrospectively screened electronic medical records of patients with NSCLC in our institute. Two sampling cohorts were adopted to collect histological samples in pretreated patients with NSCLC. The first was histological rebiopsied samples after several chemotherapies, regardless of driver oncogene alterations such as EGFR/ALK. The second was surgical tissue samples after neoadjuvant chemoradiation therapy. After exclusion of samples with ALK-fusion, we examined whether each sample contained sufficient cancer cells to perform PD-L1 immunohistochemistry (IHC). After confirmation of cancer cell sufficiency, PD-L1 IHC was carried out. Never smoker was defined as patients who had never smoked in their lifetime. Current smoker was categorized as those who had smoked within 1 year of the diagnosis. The rest were regarded as former smoker. The study was approved by the institutional review board, and complied with the Declaration of Helsinki.
EGFR mutational analysis
We isolated tumor DNA from each specimen, and analyzed EGFR mutations using highly sensitive assays: the peptide nucleic acid-locked nucleic acid PCR clamp method [28] or the cycleave method [29].
PD-L1 immunohistochemistry
Paraffin-embedded tumor tissue was sectioned at a thickness of 4 μm, and the sections were then pasted on coated glass slides for PD-L1 IHC. PD-L1 IHC was performed using the 28-8 antibody for tumor cell membrane staining. Slides were stained with Dako Autostainer Link48. Antigen retrieval was performed in Target Retrieval Solution Low pH. The primary antibody of PD-L1 (clone: 28-8) was diluted at 1:600, and incubated for 45 min at room temperature. The antibody was detected with Rabbit (LINKER) and EnVision FLEX/HRP. Digital image was captured using Aperio Scanscope AT Turbo slide scanner (Leica Biosystems, Vista, CA, USA) under 20x objective magnification. Scoring of PD-L1 was performed using digital image analysis software, namely Aperio membrane v9 and Aperio Genie Classifier. Score of PD-L1 was represented as H-score to evaluate both percentage and intensity. Semiquantitative H-score (maximum value of 300 corresponding to 100% of tumor cells positive for PD-L1 with an overall staining intensity score of 3) was determined by multiplying the percentage of stained cells by an intensity score (0, absent; 1,weak; 2, moderate; and 3, strong). Our pathologists also confirmed PD-L1 IHC score, and no significant difference was found in PD-L1 scores between digital procedure and pathologists. We defined H-scores ≥1 as PD-L1+, scores ≥5 as moderate PD-L1+, and scores ≥10 as strong PD-L1+.
Statistical analyses
To compare H-scores between EGFR-mutant and wild-type samples, we used the Wilcoxon rank sum test. Chi-square test was done to compare the incidence of PD-L1+. Multivariate analysis for strong PD-L1+ was performed using the logistic regression model. Final investigating variables were selected by backward elimination method. A P-value less than 0.05 was considered significant. The statistical analyses were performed using JMP 12 (SAS Institute, Inc., Cary, NC, USA).
ACKNOWLEDGMENTS
We thank David Martin for his writing support.
CONFLICTS OF INTEREST
Akito Hata received lecture fee from Chugai, AstraZeneca, Boeringer Ingelheim, and Eli Lilly. Nobuyuki Katakami received grants from AstraZeneca, Eisai, Ono, Kyowa Kirin, Shionogi, Daiichi-Sankyo, Taiho, Chugai, Eli Lilly, Boeringer Ingelheim, and Merck Serono, and payment for lectures from Dainippon Sumitomo, Chugai, Boeringer Ingelheim, AstraZeneca, Eli Lilly, Taiho, Janssen, Novartis, Pfizer, Ono, and Daiichi-Sankyo. The other authors declare no conflicts of interest.
GRANT SUPPORT
The study was partly supported by research funds from Foundation for Biomedical Research and Innovation.
REFERENCES
1. Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG, Louis DN, Christiani DC, Settleman J, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004; 350:2129–2139.
2. Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, Naoki K, Sasaki H, Fujii Y, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004; 304:1497–1500.
3. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, Fujiwara S, Watanabe H, Kurashina K, Hatanaka H, Bando M, Ohno S, Ishikawa Y, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. 2007;448:561–566.
4. Lee CK, Wu YL, Ding PN, Lord SJ, Inoue A, Zhou C, Mitsudomi T, Rosell R, Pavlakis N, Links M, Gebski V, Gralla RJ, Yang JC. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors versus chemotherapy in EGFR-mutant lung cancer: a meta-analysis. J Clin Oncol. 2015; 33:1958–1965.
5. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, et al, and PROFILE 1014 Investigators. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014; 371:2167–2177.
6. Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE, Holgado E, Waterhouse D, Ready N, Gainor J, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015; 373:123–135.
7. Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015; 373:1627–1639.
8. Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, Majem M, Fidler MJ, de Castro G Jr, et al. Pembrolizumab versus docetaxel for previously treated, PD-l1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016; 387:1540–1550.
9. Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols MC, Cortinovis DL, Leach J, Polikoff J, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2016 [Epub ahead of print].
10. Sacher AG, Gandhi L. Biomarkers for the clinical use of PD-1/PD-L1 inhibitors in non-small-cell lung cancer: A review. JAMA Oncol. 2016; 2:1217–22.
11. Fehrenbacher L, Spira A, Ballinger M, Kowanetz M, Vansteenkiste J, Mazieres J, Park K, Smith D, Artal-Cortes A, Lewanski C, Braiteh F, Waterkamp D, He P, et al, and POPLAR Study Group. Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (poplar): a multicentre, open-label, phase 2 randomised controlled trial. Lancet. 2016; 387:1837–1846.
12. Gainor JF, Shaw AT, Sequist LV, Fu X, Azzoli CG, Piotrowska Z, Huynh TG, Zhao L, Fulton L, Schultz KR, Howe E, Farago AF, Sullivan RJ, et al. EGFR mutations and alk rearrangements are associated with low response rates to PD-1 pathway blockade in non-small cell lung cancer (NSCLC): A retrospective analysis. Clin Cancer Res. 2016; 22:4585–4593.
13. Takada K, Okamoto T, Shoji F, Shimokawa M, Akamine T, Takamori S, Katsura M, Suzuki Y, Fujishita T, Toyokawa G, Morodomi Y, Okano S, Oda Y, et al. Clinical significance of PD-L1 protein expression in surgically resected primary lung adenocarcinoma. J Thorac Oncol. 2016; 11:1879–1890.
14. Ji M, Liu Y, Li Q, Li X, Ning Z, Zhao W, Shi H, Jiang J, Wu C. PD-1/PD-L1 expression in non-small-cell lung cancer and its correlation with EGFR/KRAS mutations. Cancer Biol Ther. 2016; 17:407–413.
15. Azuma K, Ota K, Kawahara A, Hattori S, Iwama E, Harada T, Matsumoto K, Takayama K, Takamori S, Kage M, Hoshino T, Nakanishi Y, Okamoto I. Association of PD-L1 overexpression with activating EGFR mutations in surgically resected nonsmall-cell lung cancer. Ann Oncol. 2014; 25:1935–1940.
16. Song Z, Yu X, Cheng G, Zhang Y. Programmed death-ligand 1 expression associated with molecular characteristics in surgically resected lung adenocarcinoma. J Transl Med. 2016; 14:188.
17. Tang Y, Fang W, Zhang Y, Hong S, Kang S, Yan Y, Chen N, Zhan J, He X, Qin T, Li G, Tang W, Peng P, Zhang L. The association between PD-L1 and EGFR status and the prognostic value of PD-L1 in advanced non-small cell lung cancer patients treated with EGFR-TKIs. Oncotarget. 2015; 6:14209–19. https://doi.org/10.18632/oncotarget.3694.
18. D’Incecco A, Andreozzi M, Ludovini V, Rossi E, Capodanno A, Landi L, Tibaldi C, Minuti G, Salvini J, Coppi E, Chella A, Fontanini G, Filice ME, et al. PD-1 and PD-L1 expression in molecularly selected non-small-cell lung cancer patients. Br J Cancer. 2015; 112:95–102.
19. Han JJ, Kim DW, Koh J, Keam B, Kim TM, Jeon YK, Lee SH, Chung DH, Heo DS. Change in PD-L1 expression after acquiring resistance to gefitinib in EGFR-mutant non-small-cell lung cancer. Clin Lung Cancer. 2016; 17:263–270.
20. Akbay EA, Koyama S, Carretero J, Altabef A, Tchaicha JH, Christensen CL, Mikse OR, Cherniack AD, Beauchamp EM, Pugh TJ, Wilkerson MD, Fecci PE, Butaney M, et al. Activation of the PD-1 pathway contributes to immune escape in EGFR-driven lung tumors. Cancer Discov. 2013; 3:1355–1363.
21. Chen N, Fang W, Zhan J, Hong S, Tang Y, Kang S, Zhang Y, He X, Zhou T, Qin T, Huang Y, Yi X, Zhang L. Upregulation of PD-L1 by EGFR activation mediates the immune escape in EGFR-driven nsclc: Implication for optional immune targeted therapy for NSCLC patients with EGFR mutation. J Thorac Oncol. 2015; 10:910–923.
22. Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, Miller ML, Rekhtman N, Moreira AL, et al. Cancer immunology. mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015; 348:124–128.
23. Pan ZK, Ye F, Wu X, An HX, Wu JX. Clinicopathological and prognostic significance of programmed cell death ligand1 (PD-L1) expression in patients with non-small cell lung cancer: a meta-analysis. J Thorac Dis. 2015; 7:462–470.
24. Thompson RH, Gillett MD, Cheville JC, Lohse CM, Dong H, Webster WS, Chen L, Zincke H, Blute ML, Leibovich BC, Kwon ED. Costimulatory molecule B7-H1 in primary and metastatic clear cell renal cell carcinoma. Cancer. 2005; 104:2084–2091.
25. Yu H, Boyle TA, Zhou C, Rimm DL, Hirsch FR. PD-L1 expression in lung cancer. J Thorac Oncol. 2016; 11:964–975.
26. Rizzardi AE, Johnson AT, Vogel RI, Pambuccian SE, Henriksen J, Skubitz AP, Metzger GJ, Schmechel SC, Kwak J, Stanford JL, Schmechel SC. Quantitative comparison of immunohistochemical staining measured by digital image analysis versus pathologist visual scoring. Diagn Pathol. 2012; 7:1596–1597.
27. Shukuya T, Carbone DP. Predictive markers for the efficacy of anti-PD-1/PD-L1 antibodies in lung cancer. J Thorac Oncol. 2016; 11:976–988.
28. Nagai Y, Miyazawa H, Huqun, Tanaka T, Udagawa K, Kato M, Fukuyama S, Yokote A, Kobayashi K, Kanazawa M, Hagiwara K. Genetic heterogeneity of the epidermal growth factor receptor in non-small cell lung cancer cell lines revealed by a rapid and sensitive detection system, the peptide nucleic acid-locked nucleic acid PCR clamp. Cancer Res. 2005; 65:7276–7282.
29. Kosaka T, Yatabe Y, Endoh H, Yoshida K, Hida T, Tsuboi M, Tada H, Kuwano H, Mitsudomi T. Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin Cancer Res. 2006; 12:5764–5769.

