INTRODUCTION
Cytosolic free Ca2+ ([Ca2+]cyt) plays an essential role in a variety of mammalian cells through the regulation of many biological functions, including neurotransmitter release, muscle contraction, gene regulation, cell proliferation, and apoptosis [1]. Therefore, dysregulation of [Ca2+]cyt homeostasis may result in pathological changes in many systems. Under physiological conditions, the function of Ca2+ as a cellular messenger is based on the presence of a concentration gradient between intracellular Ca2+ ([Ca2+]i) and extracellular Ca2+ [2], although different cell types combine different types of Ca2+ signaling to accomplish their specific physiological functions. Differences in Ca2+ signatures, which are key factors that determine specific Ca2+-dependent cellular responses, depend on complex, spatiotemporal variations in [Ca2+]cyt. A major determinant of these variations is based on functionally distinct calcium channels and exchangers. Ca2+ release from intracellular stores is mediated by ryanodine receptor (RyR) and inositol triphosphate receptor (IP3R) channels. RyRs are activated by a rise in [Ca2+]i, i.e., Ca2+-induced Ca2+ release (CICR) [3].
[Ca2+]cyt also plays critical roles in the regulation of many biological functions in the digestive system [4, 5], such as nutrient digestion and absorption, epithelial ion transport and secretion, and gastrointestinal (GI) motility [6]. [Ca2+]i is also an important factor that can accelerate GI epithelial wound repair. Bicarbonate (HCO3−) secretion in the intestinal mucosa is the main protective mechanism for the intestinal mucosal barrier. The ability of the duodenal mucosa to secrete mucus and bicarbonate combined with epithelial cell proliferation and migration constitutes an important protective mechanism for the intestinal mucosa.
The cAMP and cGMP signaling pathways play important roles in the regulation of intestinal bicarbonate secretion, and the details of the underlying mechanisms are well understood. Several good reviews have discussed this topic. However, little is known about the role of Ca2+ signaling in this regulation. Our research team has been working in this field to provide solid evidence for an important regulatory role of Ca2+ signaling and the underlying mechanisms. Therefore, in this review, we attempt to systemically review the current status of knowledge of the roles of calcium signaling in the regulation of intestinal bicarbonate secretion and upper GI protection.
Intestinal HCO3− secretion in GI protection
Role of HCO3− in the intestinal epithelium
The proximal portion of the duodenal lumen often attains acidity approaching a pH of 2 [7–9], but the pH remains neutral in the vicinity of the epithelial cell surface [10]. The pH gradient affects the formation of bicarbonate and mucus by epithelial cells. The viscoelastic mucosal gel at the epithelial surface and bicarbonate secretion to the mucosal gel provide a pre-epithelial damage defense mechanism [11]. The mucosal gel consists of 0–5% mucins (glycoproteins) and > 90% water [12]. Glycoproteins are secreted through exocytosis at the epithelial cell surface and the Bruner glands. Water provides continuous coverage of the gel on the epithelial cell surface.
Epithelial cells in the gastrointestinal tract remain in close proximity to one another by closing the top of the spaces between cells. In contrast to gastric epithelial cells, duodenal epithelial cells are commonly referred to as a “leaky” epithelium due to the high penetration of ions between the cells via electrolyte passive transport. The GI epithelium covers the largest surface area in the body. Because they are exposed to the external environment, skin cells are subjected to potentially destructive factors of exogenous and endogenous origin. Duodenal mucosal bicarbonate secretion is recognized as the main defense mechanism for the intermittent expulsion of gastric hydrochloric acid. HCO3− is secreted in higher amounts by the duodenum than by areas such as the stomach and small intestine.
Duodenal mucosal HCO3− secretion
HCO3– is secreted by the duodenal mucosa in the presence of hydrochloric acid and pepsin and intermittently exits the gastric duodenum to protect people from the pulsed key role. One unique function of the duodenal epithelium in the small intestine is the secretion of bicarbonate with a higher velocity, which enables it to reach the more distant parts of the mucosa. HCO3− is secreted in response to major physiological stimuli, such as the presence of duodenal acid. Acid-induced HCO3−responsive medullary nerve pathways are involved in the release of vasoactive intestinal peptide (VIP), acetylcholine (ACh), and E-type prostaglandins (PGs) from mucosal epithelial cells [13]. VIP is a very potent peptide that stimulates intestinal HCO3− secretion, and infusion of the VIP peptide increases duodenal mucosal transport of HCO3− in all species [14–16]. Other mediators also stimulate duodenal bicarbonate transport, including cholecystokinin (CCK), pancreatic polypeptide, neurotensin, glucagon, pituitary adenylate cyclase activating peptide (PACAP), and angiotensin II [17–20]. Several reporter systems, including (iv) nitric oxide (NO) synthase (NOS), inhibit the addition of N-nitro-L-arginine methyl ester (L-NAME) to duodenal mucosal bicarbonate secretion [21–24].
Three major messenger system shave been implicated in the intracellular control of HCO3− transport: i) intracellular calcium-induced responses (muscarinic M3 receptor agonists and CCKA), ii) cyclic adenosine monophosphate-activated transport (prostaglandin EP3 receptor agonists with dopamine D1 receptor agonists) and iii) cyclic GMP activation transport (uroguanylin, guanylin, and heat-stable enterotoxin).
The duodenum has different acid-base transport mechanisms that possibly reflect the activation of a second messenger system. HCO3– and CO2 reach the epithelium via the blood, and HCO3− is imported to the basolateral membrane via Na+(n)-HCO3− cotransporters (NBCs). CO2 diffuses into enterocytes, and HCO3– is formed intracellularly by carbonic anhydrase during the conversion of CO2 + H2O to HCO3– + H+. Enterocytes export HCO3− at the apical membrane via a Cl–/HCO3– exchanger and an anion conductive pathway. The cystic fibrosis transmembrane conductance regulator (CFTR) has been suggested to function as a ubiquitous transmembrane channel for the transport of Cl - and HCO3– [25–27].
Ca2+ regulation of intestinal HCO3– secretion
Ca2+ signaling in intestinal epithelial cells
Most of the HCO3– secreted by epithelial cells is in response to multiple inputs from hormones, neurotransmitters, and autacoids that evoke cytoplasmic Ca2+ signaling.
The increase in the cytoplasmic IP3 concentration stimulates the IP3 receptor (IP3R) in the endoplasmic reticulum (ER) and the rapid release of free Ca2 + from the ER Ca2 + store to the cytoplasm. Among the three IP3R paralogues (IP3R1-IP3R3), IP3R2 and IP3R3 are the major isomers in epithelial cells [28].
ER Ca2+ release is frequently followed by the activation of store-operated channels (SOCs) in the plasma membrane, such as the Orai [29–31] and transient receptor potential cation (TRPC) channels [32–34]. In response to the depletion of Ca2+ stores, the ER Ca2+ sensor stromal interaction molecule 1 (STIM1) clusters with and activates these SOCs [35, 36]. In some epithelial sites, such as the duodenal mucosa, Ca2+ entry via the reverse mode of the Na+/Ca2+ exchanger (NCX) can play a role in the sustained increase in [Ca2+]i [37]. Finally, the increase in [Ca2+]i activates the sarco/endoplasmic Ca2+ ATPase (SERCA) and plasma membrane Ca2+ ATPase (PMCA) pumps to restore [Ca2+]i to basal levels [38].
Ca2+ signals in epithelial cells are highly polarized. This polarization is caused by the expression of the polylactic secretion receptor and Ca2+ signal transduction protein. For instance, polarized expression of Ca2+-signaling proteins, such as IP3Rs [39–41], SERCA and PMCA pumps [42, 43], TRPC [44] and Orai channels, and STIM1 [45, 46], has been demonstrated in epithelial cells.
Ca2+ signaling induces a physiological agonist concentration during Ca2+ oscillations that periodically results in recurrent Ca2+ signaling. The frequency and amplitude of the oscillations are determined by the intensity of the stimulus [47]. The direct binding of [Ca2+]cyt to the target transporter can regulate its function, as has been demonstrated in Ca2+ activation of Cl- channel (CaCC) [48]. Additionally, increasing [Ca2+]i can regulate the function of target Ca2+ signaling proteins, such as calmodulin and Ca2+-calmodulin-dependent protein kinase (CamKs). Finally, Ca2+ evokes receptor-mediated Ca2+ signaling agonists that can regulate membrane transporters through the production of by-products, such as diacylglycerol (DAG) and IP3. Protein kinase C (PKC) mediates DAG activation or IP3-induced release of the IP3-binding protein with IP3 (IRBIT) from IP3Rs, which has been shown to regulate the number of epithelial transporters [49, 50].
Ca2+ regulatory mechanisms of intestinal HCO3− secretion
Many studies have shown that epithelial HCO3–secretion plays an important role at all levels of the gastrointestinal tract from the esophagus to the colon (especially the pancreas) and that abnormal HCO3– secretion is associated with many diseases in these organs. Of the intestinal segments, laryngeal HCO3– secretion has been most extensively studied and defined in the duodenum. Active HCO3- secretion is the key to protecting the epithelial cell surface against the toxic and acidic gastric contents. (i) 5-Hydroxytryptamine and ATP: The main stimulus that causes duodenal mucosal HCO3– secretion is luminal acid, and exposure to luminal acid activates neurons to reflexively induce duodenal HCO3– secretion [11, 12, 51, 52]. (ii) Gas: The effects of the gas medium (NO, H2S, and CO) on the regulation of duodenal HCO3– secretion have been studied. Acidified mucosal release may occur systemically instead of locally, possibly through activation of capsaicin-sensitive neurons. Additionally, a large number of neurotrophic factors are not involved in laryngeal acid-induced HCO3– secretion. These factors include capsaicin-sensitive neurons, peptides, PGs, NO, CO, and H2S gaseous media. Notably, many of these regulators also induce intracellular Ca2+ signaling in duodenal mucosal epithelial cells either directly or indirectly. For example, NO and CO can lead to the production of prostaglandin E2 (PGE2) through cyclooxygenase and cGMP-mediated activation, which in turn suggests that Ca2+ signaling activates Gq-coupled prostaglandin EP3 receptors [53]. (iii) Hormones: The circulating hormone CCK is released in response to the duodenal and stomach contents through postprandial duodenal HCO3– secretion via strong induction of Ca2+ signaling [54]. Acid-sensitive ion channel functions represent duodenal epithelial cells and can be stimulated by acid-stimulated HCO3– through the Ca2+ signaling pathway [55]. (iv) CFTR: CFTR expression is essential for HCO3– secretion by most of the gastrointestinal epithelial tissue; in these epithelial cells, a large proportion of the transport of transgene material, including HCO3–, is mediated through the electrical diffusion pathway, suggesting that anion channels are involved in this process. CFTR anion channel activity and CFTR-dependent Cl-/HCO3– exchangers appear to play important roles in all forms of duodenal HCO3– secretion. CFTR mainly activates Ca2+ receptor agonists that can partially activate or enhance cAMP-mediated activation of CFTR through PKC-mediated phosphorylation or release from IP3R IRBIT through the signaling pathway. In fact, cytoplasmic Ca2+ signaling induces the activation of cholinergic receptors, purine receptors, Toll-like receptors, and gaseous media, which can induce CFTR activation in the duodenum through PGE2 production [56]. PGE activates the EP4 receptor in the duodenum, which eventually evokes cAMP signals and activates CFTR. Ca2+-induced activation of large conductance and intermediate conductance K+ channels [57] and basolateral nuclear biochemical activity [58] and the Ca2+-induced inhibition of apical NHE activity [59, 60] have been shown to contribute to HCO3– secretion in parts of the intestine. (v) Calcium-sensing receptor (CaSR): The CaSRs participate in the regulation of intestinal secretion and absorption of Ca2+, organic nutrients, and amino acids [61, 62]. The intestinal brush border expresses CaSRs, which help detect the presence of calcium in the cavity and modify the cross-side cells to absorb calcium in cooperation with the vitamin D system [63]. The presence of CaSRs provides a basic mechanism for the detection of Ca2+ by intestinal cells and their responses to Ca2+-related biological behavior, such as intestinal secretion and uptake. In addition to the lumen in the acid-induced mechanism, the lumen of the bacterial component can induce HCO3– secretion by the duodenal mucosa to arouse cytoplasmic Ca2+ signaling. (Figure 1)
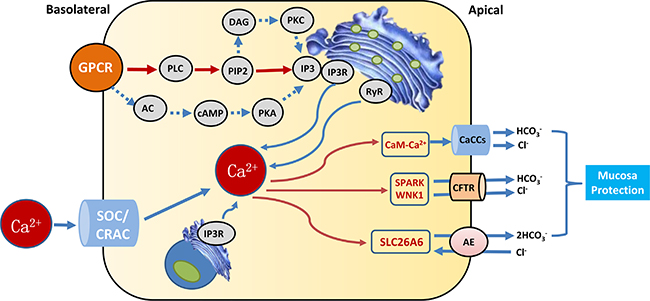
Figure 1: Calcium signaling that augment epithelial HCO3− secretion. G protein-coupled receptors (GPCRs), phospholipase C (PLC), phosphatidylinositol (4,5) bisphosphate(PIP2), inositol 1,4,5-trisphosphate (IP3), IP3 receptors (IP3Rs), adenylate cyclase(AC), cyclic adenosine monophosphate(cAMP), protein kinase A(PKA), diacylglycerol (DAG), protein kinase C(PKC), ryanodine receptor(RyR), Ca2+ release activated Ca2+ channel (CRAC), store-operated channels (SOCs), Ca2+-activated Cl− channel (CaCC), cystic fibrosis transmembrane conductance regulator (CFTR), with-nolysine kinase 1 (WNK1), STE20/SPS1-related proline/alanine-rich kinase (SPAK).
Ca2+ regulation of pancreatic HCO3− secretion
In the pancreas, acinar cells secrete initial fluids that are rich in digestive enzymes and Cl-. In humans and several other species, HCO3– concentrations in the pancreatic juice reach very high levels during stimulated secretion [64, 65]. Previous studies have shown that digestive enzymes secreted by receptor agonists and Ca2+ signaling play major roles in acinar cells, and those associated with cAMP signaling play a major role in HCO3– secretion from duct cells [38]. Apical Ca2+ signals are physiologically important as they activate Ca2+-sensitive Cl- channels, which are exclusively present in the apical membrane and are crucial for acinar fluid secretion. In pancreatic tissues, these functions act predominantly on muscarinic M3 receptors and the circulating hormone CCK, which act on CCK1 receptors. CCK also evokes Ca2+ spikes, albeit with a somewhat different pattern from that generated by ACh. Although all Ca2+ spikes can be blocked by IP3R or RyR antagonists irrespective of whether they are evoked by ACh or CCK, the action of ACh appears to be initiated by phospholipase C activation via IP3 generation. The higher pancreatic juice HCO3– concentration is transported by CFTR, which acts as a HCO3– channel. The activities of these transporters are directly or indirectly affected by Ca2+ signaling; thus, cytoplasmic Ca2+ and PKC can activate the CFTR-dependent Cl-/HCO3– exchange [66] and CFTR anion channel activity [50].
Epithelial restitution of GI protection
Role of epithelial restitution in the intestine
The small intestine is the main organ involved in the absorption and secretion nutrients by adjusting the flow of water and several ions (Na+, Cl-, K+, and Ca2+) to maintain the water and electrolyte balance. Therefore, the integrity of the intestinal mucosa is very important. Epithelial return is a highly regulated process that relies on energy and is involved in intracellular and extracellular signals and tissue repair via biomolecules. This repair process involves PGs, cytoskeletal rearrangement, ion transporters, and other cellular processes [67–70]. In response to acute interruption of gastric epithelial cells, cell migration is the first response towards the restoration of epithelial continuity and barrier function [71]. Recently, all gastrointestinal epithelial cells have been shown to secrete HCO3–, which plays an important role in protection against epithelial cell damage from the luminal contents, such as gastric acid, drugs, reactive oxygen species, bile acids, and bacterial products in the esophagus, stomach, and small intestine [1, 2, 72], trypsin, bile acids, and alcohols in the pancreatic ducts [4], and toxic bile acids in the biliary tract [73]. In the lower digestive tract, bicarbonate secretion not only protects the intestinal mucosal barrier but also is the most important mechanism regulating the acid/alkaline balance apart from kidney function. CFTR is obviously expressed in the gastrointestinal epithelium in most tissues, including the esophagus [6], small intestine [27], biliary tract [74, 75], and pancreatic duct [38], as well as the reproductive tract [76–79], which creates the necessary conditions for the airline HCO3– secretion.
Ca2+ signaling in epithelial restitution
Ca2+ is the second messenger of numerous cellular processes, including the effects of gastric acid/bicarbonate secretion, mucus secretion, and cell migration. We have shown that cytoplasmic Ca2+ mobilization in gastric epithelial cells occurs during the return of the small molecule signal to repair the central signal. However, extracellular Ca2+ is also mobilized in the upper part of wounded juxtamucosal lumen spaces [80], and evidence suggests that extracellular Ca2+ is the third messenger and thus also promotes restoration of the gastric epithelium. Intracellular and extracellular Ca2+ interactions are necessary for efficient gastric epithelial restitution. The human intestinal mucosal epithelia contains a Ca2+ sensing mechanism. In the early 1980s, changes in extracellular Ca2+ and modulation of the 1, 25-dihydroxyvitamin D3 levels were observed during regulated uptake and/or Ca2+ secretion in isolated rat colonic mucosal cells. Subsequently, there was a 30-year interval with virtually no in vivo studies exploring the role of [Ca2+]i in gastric epithelial cells.
The most important ion channels are Cl-/HCO3– exchangers, and in many epithelial tissues, including the pancreatic ducts, salivary gland ducts, and the duodenum, apical HCO3− secretion is frequently associated with Cl− absorption [81]. In humans and other mammals, which encode the SLC4 and SLC26 family gene products involved in Cl-/HCO3– exchange activity, recent evidence suggests that drug transporter SLC26 family members can mediate Cl-/HCO3– exchange. CaCCs can also mediate electro diffusive HCO3− transport in the apical epithelial membrane. Recently, members of the anoctamin family (ANO; also known as TMEM16), especially ANO1/TMEM16A and ANO2/TMEM16B, have been shown to function as CaCCs in the intestine, trachea, salivary glands, and olfactory organ [82–86]. Ca2+-induced activation of CaCCs has been suggested to contribute to HCO3− secretion in some epithelial tissues. CFTR is a cAMP-activated anion channel that is mutated in CF [87]. CFTR expression is a necessary condition for HCO3– secretion by most GI and airway epithelial cells [81]. Among these epithelial cells, a large part of the transgene material from HCO3– transportaccumulates through the electro diffusive pathway, suggesting that the anion channel is involved in this process.
The underlying mechanisms
Despite the exciting potential shown by the results discussed above, few reports have measured Ca2+ in the gastric epithelia. Intracellular loading of conventional acetoxymethyl ester Ca2+-sensitive fluorescent probes has been used to study this topic. In 1997, the gene encoding yellow cameleon (YC) protein was discovered; subsequently, cyan fluorescent protein (CFP) was developed, and yellow fluorescent protein (YFP) was associated with the M13 calmodulin-binding domain and calmodulin. YC transgenic mice have been created, which allows direct observation of [Ca2+] in real time [88].
Eitaro Aihara and Marshall H Montrose’s work and the work of others has shown that there is a pH microdomain adjacent to the surface of the epithelium that is altered in the presence of epithelial damage [80–89]. Based on these advances in our knowledge, the conceptual and experimental foundation for evaluating luminal Ca2+ microdomains has been solidified in recent years. These studies used two-photon confocal microscopy to investigate the gastric epithelial restitution model.
In the case of gastric mucosal protection, bicarbonate secretion is mediated by the EP1 receptor via a mechanism mediated by verapamil [13]. These data suggest that an increase in epithelium recovery in [Ca2+]i may mediate PGE2 activation via PLC/IP3 upstream of the EP1 receptor. Additionally, in vitro studies of gastric epithelial cells have reported that PGE2 is released by PLC inhibitors, suggesting that an increase in [Ca2+]i in response to damage enhances PGE2 production via the late maintenance cycle, which is expected to stimulate repair while maintaining high Ca2+ levels [90, 91]. Evidence from the use of inhibitors suggests that some of the Ca2+ influx important for cell migration occurs through voltage-gated Ca2+ channels in vivo [92]. Other Ca2+ channels, such as transient receptor potential (TRP) channels, may also regulate the Ca2+ influx. TRPC appears to serve as a store for the Ca2+ channels (SOC) in many cells, but the transnational radical subtype expression profiles of gastric epithelial cells are still unknown [93, 94]. Recently, TRPC has been shown to associate with Orai1 and STIM1 in several models [95–97]. However, due to lack of study of gastric epithelial cells or other areas of the gastrointestinal tract, the mechanism underlying the Ca2+ influx in gastric epithelial cells is unknown.
The key early observation was that the chelating activity of extracellular Ca2+ reduced the potential difference of the gastric mucosa. Recent reports have shown that Ca2+ release into the gastric gland can occur as part of the normal physiological functions of regulation. The extracellular Ca2+ gradient appears to be present in the various medial gastric lumen compartments, and this Ca2+source may at least have physiological effects that promote mucus and HCO3– secretion. Secretion from intact tissue is one component of the first line of gastric defense. Extracellular Ca2+ also plays a role in injured tissue. Increased luminal Ca2+ benefits epithelial repair and is dependent on [Ca2+]i increases, which most likely results from the active Ca2+ efflux from surviving epithelial cells as a result of epithelial cell repair.PMCA1 has been reported to be essential for the routine maintenance of intracellular Ca2+ homeostasis, whereas PMCA4 performs specialized physiological functions [98]. PMCA1 is reported to have an important effect on gastric restitution and the regulation of extracellular Ca2+ following injury [99]. Since the lateral cell membrane is exposed to light lesions in the gastric cavity and interruptions of epithelial continuity, enhanced permeability is the easiest way to predict the microenvironment that will allow observation of high Ca2+ concentrations at the site of injury.
CONCLUSIONS
In conventional signaling models, most physiological changes are triggered by intracellular second messengers, such as cAMP, cGMP, and Ca2+. Ca2+ is the most important signaling molecule involved in epithelial restitution; beyond HCO3– secretion, the promotion of intestinal epithelial restitution becomes the primary barrier against epithelial damage. We can apply these new perspectives to drug development; however, whether the activation of receptors that have recently been implicated in stimulating epithelial HCO3– secretion may be feasible and whether this approach will provide therapeutic benefits are unknown. The development of small molecules targeting CFTR, regulatory proteins, or stimulatory receptors suggest that such strategies may become available in the future.
CONFLICTS OF INTEREST
None.
REFERENCES
1. Johnson L, Road GF, Kaunitz J, Merchant JL, Said Hamid M, Wood J. Gastroduodenal Bicarbonate Secretion. 2012.
2. Kaji I, Akiba Y, Kaunitz JD. Digestive physiology of the pig symposium: involvement of gut chemosensing in the regulation of mucosal barrier function and defense mechanisms. J Anim Sci. 2013; 91:1957–62. https://doi.org/10.2527/jas.2012-5941.
3. Ambort JM, Gustafsson JK, Edmund A, Hansson GC. Perspectives on mucus properties and formation — lessons from the biochemical world. 2012.
4. Hegyi P, Maleth J, Venglovecz V, Rakonczay Z Jr. Pancreatic ductal bicarbonate secretion: challenge of the acinar Acid load. Front Physiol. 2011; 2:36. https://doi.org/10.3389/fphys.2011.00036.
5. Hohenester S, Wenniger LM, Paulusma CC, van Vliet SJ, Jefferson DM, Elferink RP, Beuers U. A biliary HCO3- umbrella constitutes a protective mechanism against bile acid-induced injury in human cholangiocytes. Hepatology. 2012; 55:173–83. https://doi.org/10.1002/hep.24691.
6. Abdulnour-Nakhoul S, Nakhoul HN, Kalliny MI, Gyftopoulos A, Rabon E, Doetjes R, Brown K, Nakhoul NL. Ion transport mechanisms linked to bicarbonate secretion in the esophageal submucosal glands. Am J Physiol Regul Integr Comp Physiol. 2011; 301:R83–96. https://doi.org/10.1152/ajpregu.00648.2010.
7. Rhodes J, Apsimon HT, Lawrie JH. pH of the contents of the duodenal bulb in relation to duodenal ulcer. Gut. 1966; 7:502–8.
8. Rune SJ, Viskum K. Duodenal pH values in normal controls and in patients with duodenal ulcer. Gut. 1969; 10:569–71.
9. Flemström KE. (1994). Gastric and duodenal mucosal secretion of bicarbonate.
10. Flemstrom G, Kivilaakso E. Demonstration of a pH gradient at the luminal surface of rat duodenum in vivo and its dependence on mucosal alkaline secretion. Gastroenterology. 1983; 84:787–94.
11. Flemstrom G, Isenberg JI. Gastroduodenal mucosal alkaline secretion and mucosal protection. News Physiol Sci. 2001; 16:23–8.
12. Allen A, Flemstrom G, Garner A, Kivilaakso E. Gastroduodenal mucosal protection. Physiol Rev. 1993; 73:823–57.
13. Takeuchi K, Yagi K, Kato S, Ukawa H. Roles of prostaglandin E-receptor subtypes in gastric and duodenal bicarbonate secretion in rats. Gastroenterology. 1997; 113:1553–9.
14. Flemstrom G, Kivilaakso E, Briden S, Nylander O, Jedstedt G. Gastroduodenal bicarbonate secretion in mucosal protection. Possible role of vasoactive intestinal peptide and opiates. Dig Dis Sci. 1985; 30:63S–8S.
15. Wolosin JD, Thomas FJ, Hogan DL, Koss MA, O'Dorisio TM, Isenberg JI. The effect of vasoactive intestinal peptide, secretin, and glucagon on human duodenal bicarbonate secretion. Scand J Gastroenterol. 1989; 24:151–7.
16. Glad H, Ainsworth MA, Svendsen P, Fahrenkrug J, Schaffalitzky de Muckadell OB. Effect of vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide on pancreatic, hepatic and duodenal mucosal bicarbonate secretion in the pig. Digestion. 2003; 67:56–66.
17. Konturek SJ, Bilski J, Tasler J, Laskiewicz J. Gut hormones in stimulation of gastroduodenal alkaline secretion in conscious dogs. Am J Physiol. 1985; 248:G687–91.
18. Wenzl E, Feil W, Starlinger M, Schiessel R. Alkaline secretion. A protective mechanism against acid injury in rabbit duodenum. Gastroenterology. 1987; 92:709–15.
19. Takeuchi K, Takehara K, Kato S, Yagi K. PACAPs stimulate duodenal bicarbonate secretion at PACAP receptors in the rat. Am J Physiol. 1997; 272:G646–53.
20. Takeuchi K, Yagi K, Sugamoto S, Furukawa O, Kawauchi S. Involvement of PACAP in acid-induced HCO3- response in rat duodenums. Pharmacol Res. 1998; 38:475–80.
21. Takeuchi K, Ohuchi T, Miyake H, Niki S, Okabe S. Effects of nitric oxide synthase inhibitors on duodenal alkaline secretion in anesthetized rats. Eur J Pharmacol. 1993; 231:135–8.
22. Hallgren A, Flemstrom G, Sababi M, Nylander O. Effects of nitric oxide inhibition on duodenal function in rat: involvement of neural mechanisms. Am J Physiol. 1995; 269:G246–54.
23. Sababi M, Nilsson E, Holm L. Mucus and alkali secretion in the rat duodenum: effects of indomethacin, N omega-nitro-L-arginine, and luminal acid. Gastroenterology. 1995; 109:1526–34.
24. Sababi M, Nylander O. Comparative study of the effects of nitric oxide synthase and cyclo-oxygenase inhibition on duodenal functions in rats anaesthetized with inactin, urethane or alpha-chloralose. Acta Physiol Scand. 1996; 158:45–52. https://doi.org/10.1046/j.1365-201X.1996.530287000.x.
25. Clarke LL, Harline MC. Dual role of CFTR in cAMP-stimulated HCO3- secretion across murine duodenum. Am J Physiol. 1998; 274:G718–26.
26. Hogan DL, Crombie DL, Isenberg JI, Svendsen P, Schaffalitzky de Muckadell OB, Ainsworth MA. CFTR mediates cAMP- and Ca2+-activated duodenal epithelial HCO3- secretion. Am J Physiol. 1997; 272:G872–8.
27. Seidler U, Blumenstein I, Kretz A, Viellard-Baron D, Rossmann H, Colledge WH, Evans M, Ratcliff R, Gregor M. A functional CFTR protein is required for mouse intestinal cAMP-, cGMP- and Ca(2+)-dependent HCO3- secretion. J Physiol. 1997; 505:411–23.
28. Futatsugi A, Nakamura T, Yamada MK, Ebisui E, Nakamura K, Uchida K, Kitaguchi T, Takahashi-Iwanaga H, Noda T, Aruga J, Mikoshiba K. IP3 receptor types 2 and 3 mediate exocrine secretion underlying energy metabolism. Science. 2005; 309:2232–4. https://doi.org/10.1126/science.1114110.
29. Zhang SL, Yeromin AV, Zhang XH, Yu Y, Safrina O, Penna A, Roos J, Stauderman KA, Cahalan MD. Genome-wide RNAi screen of Ca(2+) influx identifies genes that regulate Ca(2+) release-activated Ca(2+) channel activity. Proc Natl Acad Sci U S A. 2006; 103:9357–62. https://doi.org/10.1073/pnas.0603161103.
30. Vig M, Peinelt C, Beck A, Koomoa DL, Rabah D, Koblan-Huberson M, Kraft S, Turner H, Fleig A, Penner R, Kinet JP. CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science. 2006; 312:1220–3. https://doi.org/10.1126/science.1127883.
31. Feske S, Gwack Y, Prakriya M, Srikanth S, Puppel SH, Tanasa B, Hogan PG, Lewis RS, Daly M, Rao A. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature. 2006; 441:179–85. https://doi.org/10.1038/nature04702.
32. Worley PF, Zeng W, Huang GN, Yuan JP, Kim JY, Lee MG, Muallem S. TRPC channels as STIM1-regulated store-operated channels. Cell Calcium. 2007; 42:205–11. https://doi.org/10.1016/j.ceca.2007.03.004.
33. Yuan JP, Zeng W, Huang GN, Worley PF, Muallem S. STIM1 heteromultimerizes TRPC channels to determine their function as store-operated channels. Nat Cell Biol. 2007; 9:636–45. https://doi.org/10.1038/ncb1590.
34. Huang GN, Zeng W, Kim JY, Yuan JP, Han L, Muallem S, Worley PF. STIM1 carboxyl-terminus activates native SOC, I(crac) and TRPC1 channels. Nat Cell Biol. 2006; 8:1003–10. https://doi.org/10.1038/ncb1454.
35. Liou J, Kim ML, Heo WD, Jones JT, Myers JW, Ferrell JE Jr, Meyer T. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr Biol. 2005; 15:1235–41. https://doi.org/10.1016/j.cub.2005.05.055.
36. Roos J, DiGregorio PJ, Yeromin AV, Ohlsen K, Lioudyno M, Zhang S, Safrina O, Kozak JA, Wagner SL, Cahalan MD, Velicelebi G, Stauderman KA. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J Cell Biol. 2005; 169:435–45. https://doi.org/10.1083/jcb.200502019.
37. Dong H, Sellers ZM, Smith A, Chow JY, Barrett KE. Na(+)/Ca(2+) exchange regulates Ca(2+)-dependent duodenal mucosal ion transport and HCO(3)(-) secretion in mice. Am J Physiol Gastrointest Liver Physiol. 2005; 288:G457–65. https://doi.org/10.1152/ajpgi.00381.2004.
38. Lee MG, Ohana E, Park HW, Yang D, Muallem S. Molecular mechanism of pancreatic and salivary gland fluid and HCO3 secretion. Physiol Rev. 2012; 92:39–74. https://doi.org/10.1152/physrev.00011.2011.
39. Nathanson MH, Fallon MB, Padfield PJ, Maranto AR. Localization of the type 3 inositol 1,4,5-trisphosphate receptor in the Ca2+ wave trigger zone of pancreatic acinar cells. J Biol Chem. 1994; 269:4693–6.
40. Lee MG, Xu X, Zeng W, Diaz J, Wojcikiewicz RJ, Kuo TH, Wuytack F, Racymaekers L, Muallem S. Polarized expression of Ca2+ channels in pancreatic and salivary gland cells. Correlation with initiation and propagation of [Ca2+]i waves. J Biol Chem. 1997; 272:15765–70.
41. Wojcikiewicz RJ, Ernst SA, Yule DI. Secretagogues cause ubiquitination and down-regulation of inositol 1, 4,5-trisphosphate receptors in rat pancreatic acinar cells. Gastroenterology. 1999; 116:1194–201.
42. Zhao XS, Shin DM, Liu LH, Shull GE, Muallem S. Plasticity and adaptation of Ca2+ signaling and Ca2+-dependent exocytosis in SERCA2(+/-) mice. Embo j. 2001; 20:2680–9. https://doi.org/10.1093/emboj/20.11.2680.
43. Lee MG, Xu X, Zeng W, Diaz J, Kuo TH, Wuytack F, Racymaekers L, Muallem S. Polarized expression of Ca2+ pumps in pancreatic and salivary gland cells. Role in initiation and propagation of [Ca2+]i waves. J Biol Chem. 1997; 272:15771–6.
44. Kim JY, Zeng W, Kiselyov K, Yuan JP, Dehoff MH, Mikoshiba K, Worley PF, Muallem S. Homer 1 mediates store- and inositol 1,4,5-trisphosphate receptor-dependent translocation and retrieval of TRPC3 to the plasma membrane. J Biol Chem. 2006; 281:32540–9. https://doi.org/10.1074/jbc.M602496200.
45. Lur G, Haynes LP, Prior IA, Gerasimenko OV, Feske S, Petersen OH, Burgoyne RD, Tepikin AV. Ribosome-free terminals of rough ER allow formation of STIM1 puncta and segregation of STIM1 from IP(3) receptors. Curr Biol. 2009; 19:1648–53. https://doi.org/10.1016/j.cub.2009.07.072.
46. Hong JH, Li Q, Kim MS, Shin DM, Feske S, Birnbaumer L, Cheng KT, Ambudkar IS, Muallem S. Polarized but differential localization and recruitment of STIM1, Orai1 and TRPC channels in secretory cells. Traffic. 2011; 12:232–45. https://doi.org/10.1111/j.1600-0854.2010.01138.x.
47. Berridge MJ. Inositol trisphosphate and calcium oscillations. Biochem Soc Symp. 2007:1–7. https://doi.org/10.1042/bss0740001.
48. Park HW, Nam JH, Kim JY, Namkung W, Yoon JS, Lee JS, Kim KS, Venglovecz V, Gray MA, Kim KH, Lee MG. Dynamic regulation of CFTR bicarbonate permeability by [Cl-]i and its role in pancreatic bicarbonate secretion. Gastroenterology. 2010; 139:620–31. https://doi.org/10.1053/j.gastro.2010.04.004.
49. Park S, Shcheynikov N, Hong JH, Zheng C, Suh SH, Kawaai K, Ando H, Mizutani A, Abe T, Kiyonari H, Seki G, Yule D, Mikoshiba K, et al. Irbit mediates synergy between ca(2+) and cAMP signaling pathways during epithelial transport in mice. Gastroenterology. 2013; 145:232–41. https://doi.org/10.1053/j.gastro.2013.03.047.
50. Chappe V, Hinkson DA, Howell LD, Evagelidis A, Liao J, Chang XB, Riordan JR, Hanrahan JW. Stimulatory and inhibitory protein kinase C consensus sequences regulate the cystic fibrosis transmembrane conductance regulator. Proc Natl Acad Sci U S A. 2004; 101:390–5. https://doi.org/10.1073/pnas.0303411101.
51. Flemstrom G, Safsten B. Role of dopamine and other stimuli of mucosal bicarbonate secretion in duodenal protection. Dig Dis Sci. 1994; 39:1839–42.
52. Chew CS, Safsten B, Flemstrom G. Calcium signaling in cultured human and rat duodenal enterocytes. Am J Physiol. 1998; 275:G296–304.
53. Takeuchi K, Aihara E, Kimura M, Dogishi K, Hara T, Hayashi S. Gas mediators involved in modulating duodenal HCO3(-) secretion. Curr Med Chem. 2012; 19:43–54.
54. Sjoblom M, Lindqvist R, Bengtsson MW, Jedstedt G, Flemstrom G. Cholecystokinin but not ghrelin stimulates mucosal bicarbonate secretion in rat duodenum: independence of feeding status and cholinergic stimuli. Regul Pept. 2013; 183:46–53. https://doi.org/10.1016/j.regpep.2013.03.008.
55. Dong X, Ko KH, Chow J, Tuo B, Barrett KE, Dong H. Expression of acid-sensing ion channels in intestinal epithelial cells and their role in the regulation of duodenal mucosal bicarbonate secretion. Acta Physiol (Oxf). 2011; 201:97–107. https://doi.org/10.1111/j.1748-1716.2010.02207.x.
56. Mall M, Bleich M, Schurlein M, Kuhr J, Seydewitz HH, Brandis M, Greger R, Kunzelmann K. Cholinergic ion secretion in human colon requires coactivation by cAMP. Am J Physiol. 1998; 275:G1274–81.
57. Dong H, Smith A, Hovaida M, Chow JY. Role of Ca2+-activated K+ channels in duodenal mucosal ion transport and bicarbonate secretion. Am J Physiol Gastrointest Liver Physiol. 2006; 291:G1120–8. https://doi.org/10.1152/ajpgi.00566.2005.
58. Bachmann O, Reichelt D, Tuo B, Manns MP, Seidler U. Carbachol increases Na+-HCO3- cotransport activity in murine colonic crypts in a M3-, Ca2+/calmodulin-, and PKC-dependent manner. Am J Physiol Gastrointest Liver Physiol. 2006; 291:G650–7. https://doi.org/10.1152/ajpgi.00376.2005.
59. Cohen ME, Wesolek J, McCullen J, Rys-Sikora K, Pandol S, Rood RP, Sharp GW, Donowitz M. Carbachol- and elevated Ca(2+)-induced translocation of functionally active protein kinase C to the brush border of rabbit ileal Na+ absorbing cells. J Clin Invest. 1991; 88:855–63. https://doi.org/10.1172/jci115387.
60. Cohen ME, Reinlib L, Watson AJ, Gorelick F, Rys-Sikora K, Tse M, Rood RP, Czernik AJ, Sharp GW, Donowitz M. Rabbit ileal villus cell brush border Na+/H+ exchange is regulated by Ca2+/calmodulin-dependent protein kinase II, a brush border membrane protein. Proc Natl Acad Sci U S A. 1990; 87:8990–4.
61. Favus MJ, Kathpalia SC, Coe FL, Mond AE. Effects of diet calcium and 1,25-dihydroxyvitamin D3 on colon calcium active transport. Am J Physiol. 1980; 238:G75–8.
62. Favus MJ, Kathpalia SC, Coe FL. Kinetic characteristics of calcium absorption and secretion by rat colon. Am J Physiol. 1981; 240:G350–4.
63. Garg MK, Kalra S, Mahalle N. The intestinal calcistat: Determinant of clinical vitamin D deficiency. Indian J Endocrinol Metab. 2013; 17:780–3. https://doi.org/10.4103/2230-8210.117187.
64. Domschke S, Domschke W, Rosch W, Konturek SJ, Sprugel W, Mitznegg P, Wunsch E, Demling L. Inhibition by somatostatin of secretin-stimulated pancreatic secretion in man: a study with pure pancreatic juice. Scand J Gastroenterol. 1977; 12:59–63.
65. Beger HG, Buchler M, Kozarek R, Lerch M, Neoptolemos JP, Warshaw A, Whitcomb DC, Shiratori K. Pancreas: An Integrated Textbook of Basic Science, Medicine, and Surgery. 2008.
66. Namkung W, Lee JA, Ahn W, Han W, Kwon SW, Ahn DS, Kim KH, Lee MG. Ca2+ activates cystic fibrosis transmembrane conductance regulator- and Cl- -dependent HCO3 transport in pancreatic duct cells. J Biol Chem. 2003; 278:200–7. https://doi.org/10.1074/jbc.M207199200.
67. Starodub OT, Demitrack ES, Baumgartner HK, Montrose MH. Disruption of the Cox-1 gene slows repair of microscopic lesions in the mouse gastric epithelium. Am J Physiol Cell Physiol. 2008; 294:C223–32. https://doi.org/10.1152/ajpcell.00395.2006.
68. Xue L, Aihara E, Podolsky DK, Wang TC, Montrose MH. In vivo action of trefoil factor 2 (TFF2) to speed gastric repair is independent of cyclooxygenase. Gut. 2010; 59:1184–91. https://doi.org/10.1136/gut.2009.205625.
69. Cheng AM, Morrison SW, Yang DX, Hagen SJ. Energy dependence of restitution in the gastric mucosa. Am J Physiol Cell Physiol. 2001; 281:C430–8.
70. Critchlow J, Magee D, Ito S, Takeuchi K, Silen W. Requirements for restitution of the surface epithelium of frog stomach after mucosal injury. Gastroenterology. 1985; 88:237–49.
71. Ito S, Lacy ER, Rutten MJ, Critchlow J, Silen W. Rapid repair of injured gastric mucosa. Scand J Gastroenterol Suppl. 1984; 101:87–95.
72. Ambort D, Johansson ME, Gustafsson JK, Ermund A, Hansson GC. Perspectives on mucus properties and formation--lessons from the biochemical world. Cold Spring Harb Perspect Med. 2012; 2. https://doi.org/10.1101/cshperspect.a014159.
73. Hohenester S, Maillette de Buy Wenniger L, Jefferson DM, Oude Elferink RP, Beuers U. Biliary bicarbonate secretion constitutes a protective mechanism against bile acid-induced injury in man. Dig Dis. 2011; 29:62–5. https://doi.org/10.1159/000324687.
74. Zsembery A, Jessner W, Sitter G, Spirli C, Strazzabosco M, Graf J. Correction of CFTR malfunction and stimulation of Ca-activated Cl channels restore HCO3- secretion in cystic fibrosis bile ductular cells. Hepatology. 2002; 35:95–104. https://doi.org/10.1053/jhep.2002.30423.
75. Cohn JA, Strong TV, Picciotto MR, Nairn AC, Collins FS, Fitz JG. Localization of the cystic fibrosis transmembrane conductance regulator in human bile duct epithelial cells. Gastroenterology. 1993; 105:1857–64.
76. Bridges RJ. Mechanisms of bicarbonate secretion: lessons from the airways. Cold Spring Harb Perspect Med. 2012; 2. https://doi.org/10.1101/cshperspect.a015016.
77. Quinton PM. Cystic fibrosis: impaired bicarbonate secretion and mucoviscidosis. Lancet. 2008; 372:415–7. https://doi.org/10.1016/s0140-6736(08)61162-9.
78. Chan HC, Ruan YC, He Q, Chen MH, Chen H, Xu WM, Chen WY, Xie C, Zhang XH, Zhou Z. The cystic fibrosis transmembrane conductance regulator in reproductive health and disease. J Physiol. 2009; 587:2187–95. https://doi.org/10.1113/jphysiol.2008.164970.
79. Liu Y, Wang DK, Chen LM. The physiology of bicarbonate transporters in mammalian reproduction. Biol Reprod. 2012; 86:99. https://doi.org/10.1095/biolreprod.111.096826.
80. Phillipson M, Atuma C, Henriksnas J, Holm L. The importance of mucus layers and bicarbonate transport in preservation of gastric juxtamucosal pH. Am J Physiol Gastrointest Liver Physiol. 2002; 282:G211-9. https://doi.org/10.1152/ajpgi.00223.2001.
81. Seidler UE. Gastrointestinal HCO3- transport and epithelial protection in the gut: new techniques, transport pathways and regulatory pathways. Curr Opin Pharmacol. 2013; 13:900–8. https://doi.org/10.1016/j.coph.2013.10.001.
82. Yang YD, Cho H, Koo JY, Tak MH, Cho Y, Shim WS, Park SP, Lee J, Lee B, Kim BM, Raouf R, Shin YK, Oh U. TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature. 2008; 455:1210–5. https://doi.org/10.1038/nature07313.
83. Stephan AB, Shum EY, Hirsh S, Cygnar KD, Reisert J, Zhao H. ANO2 is the cilial calcium-activated chloride channel that may mediate olfactory amplification. Proc Natl Acad Sci U S A. 2009; 106:11776–81. https://doi.org/10.1073/pnas.0903304106.
84. Schroeder BC, Cheng T, Jan YN, Jan LY. Expression cloning of TMEM16A as a calcium-activated chloride channel subunit. Cell. 2008; 134:1019–29. https://doi.org/10.1016/j.cell.2008.09.003.
85. Romanenko VG, Catalan MA, Brown DA, Putzier I, Hartzell HC, Marmorstein AD, Gonzalez-Begne M, Rock JR, Harfe BD, Melvin JE. Tmem16A encodes the Ca2+-activated Cl- channel in mouse submandibular salivary gland acinar cells. J Biol Chem. 2010; 285:12990–3001. https://doi.org/10.1074/jbc.M109.068544.
86. Caputo A, Caci E, Ferrera L, Pedemonte N, Barsanti C, Sondo E, Pfeffer U, Ravazzolo R, Zegarra-Moran O, Galietta LJ. TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science. 2008; 322:590–4. https://doi.org/10.1126/science.1163518.
87. Kerem B, Rommens JM, Buchanan JA, Markiewicz D, Cox TK, Chakravarti A, Buchwald M, Tsui LC. Identification of the cystic fibrosis gene: genetic analysis. Science. 1989; 245:1073–80.
88. Tsien RY. Breeding molecules to spy on cells. Harvey Lect. 2003; 99:77–93.
89. Henriksnas J, Phillipson M, Petersson J, Engstrand L, Holm L. An in vivo model for gastric physiological and pathophysiological studies in the mouse. Acta Physiol Scand. 2005; 184:151–9. https://doi.org/10.1111/j.1365-201X.2005.01434.x.
90. Ota S, Hata Y, Terano A, Yoshiura K, Hiraishi H, Kawabe T, Mutoh H, Shiina S, Sugimoto T. Roles of Ca2+ and protein kinase C in regulation of prostaglandin E2 release by cultured rabbit gastric epithelial cells. Dig Dis Sci. 1993; 38:1426–34.
91. Sekiguchi F, Saito S, Takaoka K, Hayashi H, Nagataki M, Nagasawa K, Nishikawa H, Matsui H, Kawabata A. Mechanisms for prostaglandin E2 formation caused by proteinase-activated receptor-1 activation in rat gastric mucosal epithelial cells. Biochem Pharmacol. 2007; 73:103–14. https://doi.org/10.1016/j.bcp.2006.09.016.
92. Ranta-Knuuttila T, Kiviluoto T, Mustonen H, Puolakkainen P, Watanabe S, Sato N, Kivilaakso E. Migration of primary cultured rabbit gastric epithelial cells requires intact protein kinase C and Ca2+/calmodulin activity. Dig Dis Sci. 2002; 47:1008–14.
93. Cheng KT, Ong HL, Liu X, Ambudkar IS. Contribution and regulation of TRPC channels in store-operated Ca2+ entry. Curr Top Membr. 2013; 71:149–79. https://doi.org/10.1016/b978-0-12-407870-3.00007-x.
94. Rao JN, Platoshyn O, Golovina VA, Liu L, Zou T, Marasa BS, Turner DJ, Yuan JX, Wang JY. TRPC1 functions as a store-operated Ca2+ channel in intestinal epithelial cells and regulates early mucosal restitution after wounding. Am J Physiol Gastrointest Liver Physiol. 2006; 290:G782–92. https://doi.org/10.1152/ajpgi.00441.2005.
95. Ong HL, Cheng KT, Liu X, Bandyopadhyay BC, Paria BC, Soboloff J, Pani B, Gwack Y, Srikanth S, Singh BB, Gill DL, Ambudkar IS. Dynamic assembly of TRPC1-STIM1-Orai1 ternary complex is involved in store-operated calcium influx. Evidence for similarities in store-operated and calcium release-activated calcium channel components. J Biol Chem. 2007; 282:9105–16. https://doi.org/10.1074/jbc.M608942200.
96. Cheng KT, Liu X, Ong HL, Ambudkar IS. Functional requirement for Orai1 in store-operated TRPC1-STIM1 channels. J Biol Chem. 2008; 283:12935–40. https://doi.org/10.1074/jbc.C800008200.
97. Liao Y, Erxleben C, Abramowitz J, Flockerzi V, Zhu MX, Armstrong DL, Birnbaumer L. Functional interactions among Orai1, TRPCs, and STIM1 suggest a STIM-regulated heteromeric Orai/TRPC model for SOCE/Icrac channels. Proc Natl Acad Sci U S A. 2008; 105:2895–900. https://doi.org/10.1073/pnas.0712288105.
98. Prasad V, Okunade G, Liu L, Paul RJ, Shull GE. Distinct phenotypes among plasma membrane Ca2+-ATPase knockout mice. Ann N Y Acad Sci. 2007; 1099:276–86. https://doi.org/10.1196/annals.1387.029.
99. Aihara E, Hentz CL, Korman AM, Perry NP, Prasad V, Shull GE, Montrose MH. In vivo epithelial wound repair requires mobilization of endogenous intracellular and extracellular calcium. J Biol Chem. 2013; 288:33585–97. https://doi.org/10.1074/jbc.M113.48809.

