INTRODUCTION
Hypoxia is a common feature of solid tumors. Despite its heterogeneity, it is tightly associated with tumor tissue survival and glucose metabolic pathways activity [1]. Hypoxia promotes angiogenesis, cancer invasiveness and resistance to radiotherapy and chemotherapy [2]. Furthermore, metastasis and prognosis have a close relationship with hypoxia [3]. Hypoxia can be classified into acute or chronic type. Acute hypoxia occurs when the microvasculatures surrounding tumor tissues close up, which is usually transient. And when the closed blood vessels reopen, the hypoxic cells are reoxygenated. In contrast, chronic hypoxia occurs when the tumor cells are distant from functional capillaries. It is typically irreversible or long-term. Chronic hypoxic cells may become necrotic or reoxygenated, or convert into more aggressive cell types. Several processes cause chronic hypoxia in cancer cells [4]. First, the increased oxygen demand from abnormal tumor cell proliferation may result in insufficient blood supply from the microvasculatures surrounding the tumor. Second, due to the gradual enlargement of the tumor, the cancer cells distance themselves from functional blood vessels. Finally, defects in tumor angiogenesis and abnormalities of microvascular function may cause a unstable oxygen environment [5]. Tumor hypoxia is spatially and temporally heterogeneous [6, 7]. Noninvasive methods for visualizing hypoxic imaging probes are necessary to detect the distribution of hypoxic cancer cells in the body. 18F-Fluoromisonidazole (18F-FMISO) is the most extensively used radiolabeled imaging tracer for hypoxia among several others. In this review, we introduce 18F-FMISO imaging with current hypoxia detection methods and applications in tumor hypoxia. Then discuss its advantages and disadvantages when compared with other hypoxia radiolabeled imaging tracers. Finally, we explore the potential development prospects of this imaging technology.
SYNTHESIS OF 18F-FMISO AND BIODISTRIBUTION
FMISO has the chemical structure of 1-(2’nitro-1’-imidazolyl)-3-fluoro-2-propranol and when radiolabeled with fluorine-18, it becomes 18F-FMISO (Figure 1). 18F-FMISO is typically synthesized from a commercially available precursor as previously described [8] and purified by preparative high-pressure liquid chromatography. The final product has purity between 95% and 99%, specific activity between 1 and 3 Ci/mol, and pH between 5 and 8. The misonidazole biological half-life is 50 minutes. 18F-FMISO is metabolized by the liver and then excreted by the kidney and bladder. The lowest activity is noted in the blood, spleen, heart, lung, muscle, bone, and brain [9]. The biodistribution data in human subjects with lung cancer is demonstrated in Figure 2 [10].
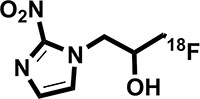
Figure 1: Chemical structure of 18F-FMISO.
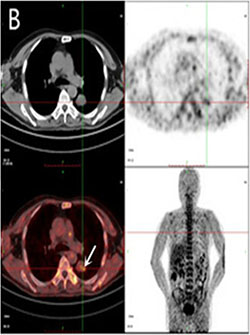
Figure 2: PET imaging. Major organs and regions uptake at 2 h after injection of 18F-FMISO (B) in small cell lung cancer patient. Arrows point to tumor. (Wei Y, Zhao W, Huang Y, Yu Q, Zhu S, et al. (2016). A Comparative Study of Noninvasive Hypoxia Imaging with 18F-Fluoroerythronitroimidazole and 18F-Fluoromisonidazole PET/CT in Patients with Lung Cancer. PLOS ONE 11(6): e0157606. https://doi.org/10.1371/journal.pone.0157606).
HYPOXIA DETECTION METHODS
Polarographic oxygen electrodes, which measure tissue oxygen pressure directly, are considered to be the gold standard for detecting tumor hypoxia. Oxygen partial pressure (PO2) of normal subcutaneous tissue is approximately 40–60 mmHg. The median PO2 of tumor tissue is about 10 mmHg. Hypoxia is defined as PO2 less than 10mmHg [11]. It is also possible to measure hypoxia indirectly in tissue sections by assessing protein expression using immunohistochemical methods such as endogenous cell markers and exogenous nitroimidazole-based assays [12]. Hypoxia inducible factor 1 (HIF-1, consisting of HIF-1α and HIF-1β) is an endogenous cell marker that regulates many genes, including vascular endothelial growth factor (VEGF) [13]. HIF-1 plays a central role in tumor progression. Exogenous hypoxic agents are mostly imidazole compounds such as pimonidazole and EF-5. Imaging methods of assessing for tumor hypoxia include MRI and PET.
Positron emission tomography (PET) is a widespread imaging technology that uses radioactive tracers to noninvasively detect tumors and monitor cancer therapy effectiveness [14]. 18F-FMISO is a nitroimidazole compound, which is labeled with radionuclides. After nitroimidazole enters the cell via the blood flow, it can occur redox reactions under the action of xanthine oxidase. In normal cells which are not hypoxia, reducted nitro group can be oxidized into the original substance by O2. While in hypoxic cells, reducted nitro group can’t be oxidized. It will stably combine with some cellular components permanently. 18F-FMISO can be detected by PET that has been widely used for tumor hypoxia imaging (Table 1). This is the first hypoxia PET tracer used in clinical settings, and currently there are some new tracers available, most derived from the family of 2-nitroimidazoles.
Table 1: Summary of studies using 18F-FMISO PET in tumor hypoxia
Tumor |
Time points |
Conclusion |
Reference |
|---|---|---|---|
HNSCC |
120 min |
Summarizing the 18F-FMISO uptake represents a global value for macroscopic tumor parts. As a noninvasive measurement this method seems highly feasible to evaluate the state of oxygenation in subjacent tumors. |
[15] |
STS |
1.5 h, 2.5 h, 3.5 h |
18F-FMISO PET in our setting seemed not to be feasible for the detection of tumour hypoxia in human soft tissue tumors. |
[16] |
OSCC |
4 h |
18F-FMISO uptake in the primary site of OSCC indicates a hypoxic environment with HIF-1α expression. |
[19] |
Glioma |
120 min |
The IRS of HIF-1α in the tumour did not correlate with the SUVtumor of 18F-FMISO in either newly diagnosed or recurrent glioma. |
[23] |
Glioma |
120 min |
Preoperative 18F-FMISO uptake is significantly correlated with the expression of VEGF in the tumour and might be used as a biomarker of antiangiogenic treatment in newly diagnosed malignant gliomas. |
[23] |
HNSCC |
4 h |
The values for 18F-FMISO PET uptake and hypoxic volume in head and neck tumors between the 2 18F-FMISO scans were highly reproducible. |
[31] |
RCC |
2 h |
Although 18F-FMISO scans showed significant uptake in other solid tumors, there was only mild 18F-FMISO uptake in the present RCCs. The invasive measurements indicated that there was hypoxia in RCC, but the median PO2 did not fall below 9.5 mmHg. |
[32] |
HNSCC |
162 min |
Of 13 patients, 6 had well-correlated intratumor distributions of 18F-FMISO suggestive of chronic hypoxia. |
[33] |
FaDu and CAL-33 xenografted tumors |
2 h |
Whether the Wang model can be used to predict radiation response after serial 18F-FMISO PET imaging, needs to be confirmed in experimental and clinical studies. |
[34] |
Glioma |
4 h |
18F-FMISO PET may distinguish glioblastoma multiforme from lower grade gliomas. |
[37] |
Glioma |
120 min |
18F-FMISO PET is a potential tracer in the assessment of noninvasive tumor grading in newly diagnosed gliomas. |
[38] |
HNSCC |
2 h |
Hypoxia-targeted radiotherapy dose painting for head and neck cancer using FMISO PET is technically feasible, increases the TCP without increasing the NTCP, and increases the UTCP. 18F-FMISO PET is superior to uniform dose escalation. |
[45] |
C6 and 9L glioblastomas rat tumor model |
dynamic 18F-FMISO PET (for a total of 2 hours) |
This has implications for improved patient selection, particularly in clinical trials, for treatment with hypoxia-activated cytotoxic prodrugs, such as evofosfamide. |
[49] |
HNSCC |
2 h |
Hypoxia on 18F-FMISO PET imaging, in patients receiving a nontirapazamine-containing chemoradiotherapy regimen, is associated with a high risk of LRF. |
[50] |
A549 peritoneal micrometastases model |
2 h |
Microscopic non-small cell lung cancer metastases are severely hypoxic. 18F-FMISO PET is capable to image hypoxia noninvasively not only in macroscopic tumors but also in micrometastases growing in mice. 18F-misonidazole may be a promising agent to detect the burden of micrometastatic diseases. |
[54] |
glioblastoma multiforme |
120 to 140 min |
This type of imaging could be integrated into new treatment strategies to target hypoxia more aggressively in glioblastoma multiforme and could be applied to assess the treatment outcomes. |
[22] |
ER-α-positive breast cancer |
2 and 4 h |
18F-FMISO PET/CT can be used to predict primary endocrine resistance in ER-positive breast cancer. |
[56] |
mRCC |
2–4 h |
Sunitinib reduced hypoxia in initially hypoxic RECIST target metastases but did not induce significant hypoxia in nonhypoxic RECIST target metastases. Patients with initially hypoxic targets have shorter PFS than others. |
[59] |
HNSCC, head and neck squamous cell carcinoma; STS, soft tissue sarcomas; OSCC, oral squamous cell carcinoma; IRS, immunoreactivity score; RCC, renal cell carcinoma; TCP, tumor control probability; NTCP, normal tissue complication probability; UTCP, uncomplicated tumor control probability; LRF, locoregional failure; ER+, positive estrogen receptor; PFS, progression-free survival; RECIST, response evaluation criteria in solid tumors.
18F-FMISO PET vs. polarographic oxygen electrodes
Although polarographic oxygen electrodes are commonly used to measure hypoxia, this method presents several disadvantages. For instance, besides being invasive, polarographic oxygen electrodes can fail to detect the whole regions because of the known heterogeneity in tumor. In addition, the results of these measurements cannot be used for radiotherapy treatment planning. Gagel et al. reported there was a significant correlation between tumor-to-muscle ratio of 18F-FMISO and parameters of hypoxic fraction in head and neck cancer [15]. This indicated 18F-FMISO uptake represented a noninvasive measurement for macroscopic tumor hypoxia and seemed highly feasible to evaluate the state of oxygenation in tumors. To the opposite, a few studies showed no correlation between polarographic measurements and 18F-FMISO. Bentzen et al suggested 18F-FMISO PET seemed not to be feasible for the detection of tumor hypoxia in human soft tissue sarcomas [16]. Neither did it reflect the extent of hypoxia as determined with the oxygen electrode measurements. Mortensen et al analyzed the relationship between 18F-FMISO PET and Eppendorf electrode measurements by use of a virtual voxel model [17]. However, no correlation was observed, and in generally, tumors were more hypoxic based on Eppendorf measurements as compared to 18F-FMISO PET. One of possible explanations for the contrast results is polarographic electrodes can´t discriminate between hypoxia and necrosis, as both regions present low oxygen partial pressure. It may over estimate hypoxia. This may explain the diverging results of 18F-FMISO PET hypoxia imaging and polarographic measurements.
18F-FMISO PET vs. immunohistochemical findings
Endogenous cell markers and exogenous nitroimidazole-based assays have similar disadvantages with polarographic oxygen electrodes. One of the main early cellular events evoked by hypoxia is induction of HIF-1. HIF-1 induces the transcription of a wide variety of genes, including glycolysis, angiogenesis, survival pathways and invasion [18]. There are a few studies exploring the correlation between 18F-FMISO uptake and immunohistochemical expressions. Sato et al. demonstrated 18F-FMISO PET uptake was correlated with HIF-1α expression in oral squamous cell carcinoma [19]. Similarly, Norikane et al found the values of 18F-FMISO hypoxic volume showed a weak correlation with HIF-1α obtained on immunohistochemical examinations in head and neck cancer [20]. But in clinical gliomas researches, none of studies was able to show a correlation between 18F-FMISO uptake and expression of HIF-1α [21–23]. Except for the small sample size, there are several possibilities to explain the discrepancy between 18F-FMISO uptake and HIF-1α expression. For one thing, even though hypoxia is the main factor in activating HIF-1α, many non-hypoxic stimuli are also very easy to turn on this transcription factor, such as growth factors, cytokines, etc [24]. For another, HIF-1α expression may depend on the degree and duration of intermittent hypoxia in tumor [23]. Although hypoxia can increase HIF-1α protein expression, HIF-1α has a short half-life under normoxic conditions and the tissue becomes anoxic between resection and embedding [25, 26].
HIF-1 is thought to be key for up-regulating factors such as VEGF. Most of 18F-FMISO analysis of individual whole tumor volumes suggests no association between VEGF expression and hypoxia [27]. Heterogeneity in VEGF expression in individual tumors is likely a reflection of similar heterogeneity in tumor hypoxia. And VEGF expression in response to hypoxia is a local phenomenon rather than generalized in a tumor [27]. In recent years, Kawai et al investigated whether hypoxia detected by 18F-FMISO PET could accurately reflect biomarkers or prognostic factors treated by bevacizumab which is used as anti-angiogenic treatment [23]. The results revealed a weak correlation between the immunoreactivity score of VEGF and 18F-FMISO maximum standardized uptake value (SUVmax) in newly diagnosed glioma brain tumors, but not in recurrent gliomas.
18F-FMISO PET vs. BOLD fMRI
BOLD fMRI (blood oxygenation-level dependent functional magnetic resonance imaging) technology utilizes the impact of the oxyhemoglobin and deoxyhemoglobin ratio changes onto the magnetic resonance signals to indirectly infer the state of PO2. Compared with 18F-FMISO imaging, it does not require to inject a compound. And, BOLD has a good temporal and spatial resolution, that can improve quality and contrast of images. Research has shown that hypoxia detection using BOLD fMRI shows a good correlation with PO2 inside tumors [28]. But BOLD fMRI is a semi-quantitative method, and affected by respiratory that is not suitable for respiratory system, such as lung cancer.
APPLICATION OF 18F-FMISO PET
In addition to 18F-FMISO with other detection methods studies, there have been many settings using 18F-FMISO as a noninvasive examinations. Most of them are clinical settings. As a clinical agent, it has a good reproducibility so that can detect hypoxia, guide therapy, manage therapeutic efficacy and estimate prognosis. And also it is used in some preclinical cases to help further clinical studies.
Detecting hypoxia
According to the research, the accuracy of malignant tumor detection in the lung using traditional 18F-FDG PET is more than 90% [29]. However, a significant proportion will be false positive results because 18F-FDG is a metabolic imaging tracer. Glycolysis is increased not only in tumor, but also in other diseases including granuloma, sarcoidosis, tuberculosis and inflammation [30]. So, it is difficult to distinguish atelectasis and local infections in lung tumors using 18F-FDG. Maybe the use of 18F-FMISO alone or in combination with 18F-FDG can improve the accuracy of diagnosis, especially in lung cancer. Furthermore, it has a good reproducibility. Okamoto et al addressed the reproducibility of 18F-FMISO PET uptake to detect tumor hypoxia by calculating SUVmax, tumor/blood ratio and tumor/muscle ratio at two different time points in head and neck squamous cell carcinoma (HNSCC) patients [31]. The results confirmed the important role of 18F-FMISO in precisely and reproducibly in detecting tumor hypoxia. Although 18F-FMISO scans showed significant uptake in sarcomas, gliomas, head and neck tumors, there was only mild uptake in renal cell carcinoma (RCC). Lawrentschuk et al studied 7 of 17 patients with clear RCC [32]. The difference between tumor uptake and normal uptake in the contralateral kidney was not statistically significant. Except for the limited cases on hypoxia studies in RCC, one possible reason may be RCC was not as hypoxic a tumor as suspected. The application of 18F-FMISO in RCC is still to be determined.
Chronic and acute hypoxia are caused by oxygen diffusion and perfusion limitation, respectively. It is important to estimate the fraction of hypoxia in solid tumors because it plays an important role in tumor aggression, progression and metastasis. A study by Nehmeh et al attempted to detect areas of chronic hypoxia in HNSCC by comparing 18F-FDG scans taken at day 0 with 18F-FMISO scans at two different time points (day 1 and 4) [33]. The outline of the tumors was set with 18F-FDG as the threshold, and the outline of the hypoxia areas was set by 18F-FMISO in tumor/blood ratio ≥1.2 as the threshold. In 6 of 13 patients, the hypoxia areas detected by 18F-FMISO overlapped well, suggesting they corresponded to regions of chronic hypoxia. However, the data cannot identify regions of acute hypoxia. In a study by Maftei et al, serial 18F-FMISO PET/CT imaging was used in combination with immunofluorescence stainings (pimonidazole, CD31 and Hoechst 33342) of cryosections to assess the fraction of acute hypoxia in HNSCC tumors [34]. But no correlation between the results obtained by each method was found. Warren et al presented a computational model to simulate the effects of temporally-incoherent cyclic variations in perfusion on 18F-FMISO contrast [35]. Results suggested that the signal observed at late time points did not specifically relate to ‘chronic’ or ‘acute’ hypoxia. But it was representative of the time-averaged oxygenation during the imaging study.
Grading
Glioma is one of the most common types of malignant primary brain tumors. Glioma has a variety of different grades, however, biopsies are not reliable for grading due to sampling errors resulting from tumor tissue heterogeneity. Errors in glioma grading effect treatment strategy and prognosis and can therefore have serious consequences. Since 18F-FMISO PET was first used in clinical glioma imaging by Valk et al, its advantages were immediately recognized and subsequently 18F-FMISO PET was introduced into glioma researches [36]. Necrotic tissues are not observed in grade III or lower grade gliomas. 18F-FMISO cannot detect necrosis, but necrosis even microscopic may be surrounded by a hypoxic area. That is sufficiently large to be identified by 18F-FMISO PET. Hirata et al found that 18F-FMISO uptake occurred only in glioblastoma multiforme (grade IV), and there was no significant uptake in lower gliomas (below grade IV) [37]. In this study, the sensitivity of both 18F-FMISO and 18F-FDG was 100%, but the specificity of the former was significantly higher (100%) than that of the latter (66%). Consistent with these findings, Cher et al and Yamamoto et al reported that grade IV glioma accumulated more 18F-FMISO than lower grade gliomas [21, 38]. Others have discussed these results and emphasized the importance of the imaging time for grading gliomas: 4 hours of imaging showed only the hypoxic zone; but 2 hours of imaging could reflect changes in blood flow with or without hypoxia [39, 40]. These studies show that 18F-FMISO PET may be useful not only for diagnosis, but also for grading and estimating prognosis of glioma.
Guiding dose painting
HNSCC accounts for about 5% of all malignancies. Radiotherapy is currently one of the most effective treatments for HNSCC [41, 42]. However, radiation resistance is an inevitable problem of radiotherapy. Multiple experiments demonstrate that hypoxia and radiation resistance are associated [43, 44]. Chang et al showed that 18F-FMISO PET technology was suitable for dose painting in HNSCC [45]. This radiotherapy approach not only increased tumor control probability, but also increased uncomplicated tumor control probability. Thus it revealed to be a superior method to traditional dose escalation radiotherapy. The authors forecasted that the clinical application of this method would improve the results of radiotherapy and promote the development of individual doses prescriptions. Furthermore, dose painting will be more robust based on dynamic PET analysis described by Thorwarth [39, 46]. However, Cui et al and Ljungkvist et al reported that hypoxic cancer cells in vivo and in vitro culture had different lifespan, and the turnover of hypoxic tumor cells may relate to the therapeutic response and outcome in patients [7, 47]. That may indicate dose painting would be unhelpful. So, there need to do more experiments to verify the usefulness of dose painting with 18F-FMISO PET technology.
Guiding hypoxic targeted therapy
At present, there has developed a series of hypoxia targeted drugs such as Evofosfamide, Banoxantrone and tirapazamine. They belong to bioreductive prodrugs and are reduced by intracellular oxidoreductases in an oxygensensitive manner to form cytotoxins in order to disrupt the DNA replication fork [48]. Stokes et al showed that C6 tumors exhibited more hypoxia and were less perfused than 9L tumors by using 18F-FMISO imaging [49]. On the basis of these differences in their tumor hypoxic burden, treatment with Evofosfamide resulted in 4 and 2 folds decreases in tumor growth rates of C6 and 9L tumors, respectively. This had implications for improved patient selection in clinical trials. Rischin et al provided the first clinical evidence that hypoxia on 18F-FMISO imaging, in patients receiving a nontirapazamine-containing chemoradiotherapy regimen, was associated with a high risk of locoregional failure [50].
Several of hypoxia targeted drugs has been used in clinical studies, especially in larger primary tumors. But most of them, such as Evofosfamide failed in phase III of soft tissue sarcoma and pancreatic cancer. Primary tumor growth accounts for few cancer-related deaths, for its outcome is necrosis or metastasis. Cancer mortality is mostly due to the development of metastatic disease. Hypoxia is a significant microenvironmental factor to induce invasion and metastasis. It suggests to be a master regulator role in metastasis. For each step of the metastasis process, from the initial epithelial mesenchymal transition to the ultimate organotropic colonization, is associated with hypoxia/HIF-1α-regulated target genes [51, 52]. Metastatic disease especially micrometastasis is highly hypoxia. Thus, in recent years, cancer researches have begun to focus on the changes in the tumor microenvironment of micrometastasis [53]. Huang et al compared the uptake of 18F-FMISO with the hypoxia-staining marker pimonidazole in subcutaneous xenograft and peritoneal metastases generated by non-small cell lung cancer cell lines (A549 and HTB177), and found significant overlap between the two markers [6]. Micrometastases were too small for detection. In another study, the same authors showed that, in addition to the normal uptake in the liver, bladder and intestines, 18F-FMISO accumulated strongly in the left peritoneal wall in a A549 peritoneal micrometastases model [54]. Hematoxylin and eosin staining confirmed that this region of severe hypoxia corresponded to small metastases. Furthermore, positive pimonidazole staining in ascites cells collected from the ascites fluid of the peritoneal cavity further demonstrated that 18F-FMISO PET could detect small metastases. Therefore, hypoxic targeted therapy may be more effective in metastasis with the help of 18F-FMISO PET.
Management of therapeutic efficacy
18F-FMISO PET is suitable for management of therapeutic efficacy, as it can reflect the degree of cell reoxidation. For instance, by comparing 18F-FMISO uptake before and after radiotherapy with chemotherapy, it was possible to determine the best conditions for killing cancer cells with these treatments [55]. Furthermore, Spence et al showed that the volume of the hypoxic tumor and the maximum level of hypoxia assessed by 18F-FMISO PET before radiotherapy, correlated negatively with the progression time and survival [22]. Thus, this imaging technology is a promising new approach for clinical diagnosis, prognosis estimation and evaluation of therapy efficacy. About 70% of breast cancers belong to the hormone-dependent type, which is characterized by the overexpression of hormone receptors [56]. Although endocrine therapy is an effective treatment for positive estrogen receptor breast cancer, about 30% of these tumors develop either primary or acquired resistance [56–58]. To assess the feasibility of using 18F-FMISO to monitor therapy resistance, Cheng et al compared PET/CT scans of positive estrogen receptor patients performed before and after treatment. 18F-FDG uptake showed no significant correlation with the results of the treatment, but a positive correlation was found with 18F-FMISO uptake. Furthermore, when tumor/blood ratio ≥ 1.2, 18F-FMISO uptake was a predictor of endocrine resistance in 88% of the tumors [56]. Thus, 18F-FMISO imaging could significantly improve breast cancer treatment. To investigate the possible prognostic value of initial tumor hypoxia or its changes under sunitinib therapy, Hugonnet et al evaluated initial tumor hypoxia in metastatic renal cell carcinoma and its changes after sunitinib treatment using 18F-FMISO PET/CT [59]. They found patients with initially hypoxic targets had shorter progression-free survival than others. But the prognostic value of 18F-FMISO PET/CT performed earlier after initiation of treatment, with dynamic acquisition, needed to be assessed in further studies.
In all, imaging with 18F-FMISO presents many advantages: (i) it is a noninvasive and comprehensive method to assess hypoxia; (ii) 18F-FMISO is specifically retained in hypoxia tissues; (iii) it has a good reproducibility; (iv) it can be used for guiding therapy. On the other hand, due to its high radioactive concentration in liver, kidney and intestine, 18F-FMISO is not suitable for abdominal tumor imaging. Furthermore, when compared with 18F-FDG, 18F-FMISO produces images of inferior quality and contrast. Moreover, although 18F-FMISO imaging can continuously measure hypoxia, it requires at least one day of interval between scans. It influences monitoring of temporal heterogeneity.
OTHER HYPOXIA IMAGING AGENTS
To overcome the limitations of 18F-FMISO, new researches and improved derivatives are in constant development. For instance, small changes in the structure of 18F-FMISO make the hydrophilic 18F-FETNIM (18F-Fluoroerythronitroimidazole). The main advantage of 18F-FETNIM when compared to similar compounds is its higher hydrophilicity and relatively lower neurotoxicity, in addition to a lower peripheral metabolic rate [60] .The radiochemical purity of 18F-FETNIM can typically reach 99%. This tracer is also easier to prepare and less costly than 18F-FMISO. However, despite these advantages, more research is necessary to determine its clinical prospects [61].
18F-FAZA (18F-Fluoroazomycinarabinoside) is developed in recent years and currently being tested in hypoxic PET imaging studies. It is thought that 18F-FAZA accumulates in hypoxic cells through a similar mechanism as imidazole derivatives. 18F-FAZA has stronger affinity to the tumor hypoxic areas than other tracers and it is removed faster in blood and muscle tissue [62]. The typical radiochemical yielding of this tracer is 20.7 ± 3.5% at the end of the reaction [63]. Furthermore, the precursor can be labeled in various manners, including by iodinated counterparts (123I/124I-IAZA) [64].
18F-HX4 (18F-3-fluoro-2-(4-((2-nitro-1H-imidazol-1-yl)methyl)-1H-1, 2, 3-triazol-1-yl)-propan-1-ol) has higher sensitivity, specificity, faster clearance, and shorter injection-imaging time compared with 18F-FMISO in a pilot clinical study of 12 patients with head and neck cancer [65]. Biodistribution and dosimetry studies in healthy monkeys and humans showed that the highest up take of 18F-HX4 was found in the bladder, kidney, liver and gastrointestinal tract uptake was relatively low [66]. Repeated hypoxia PET scans with 18F-HX4 provided reproducible and spatially stable results in patients with head and neck cancer and patients with lung cancer [67]. 18F-HX4 PET imaging can be used to assess the hypoxic status of tumors and has the potential to aid hypoxia-targeted treatments.
18F-EF series is mainly included EF1, EF3,and EF5 which are from etanidazoles compounds. The combination with hypoxic cells is more specific, while the toxicity decreases significantly. However, in a comparative PET imaging study, no superiority of 18F-EF3 (18F-(2-2-nitroimidazol-1-yl)-N-(3,3,3-trifluoropropyl)-acetamide) to 18F-FMISO for the evaluation of hypoxia [68]. Other chemical class of hypoxia radiotracer currently under scrutiny in preclinical and clinical studies is typically labeled by 60Cu, 61Cu, 62Cu, and 64Cu, for example, 64Cu-ATSM (64Cu- diacetyl-bis (N4-methylthiosemicarbazone)). This compound shows high variability in tumor uptake between different tumor types in vitro [69]. However, 64Cu-ATSM is not valid in all types of tumor such as fibrosarcoma. Further studies are needed to define retention mechanisms for this PET marker [70].
CONCLUSIONS
This review begins by explaining the concept of hypoxia and introducing the most widely used hypoxic imaging agent: 18F-FMISO. A comprehensive discussion of 18F-FMISO applications for hypoxia imaging is presented. 18F-FMISO shows great promise as a clinical hypoxia-imaging agent, however, it also presents certain limitations. Obviously, a method is difficult to meet all the needs of hypoxia imaging. Therefore, it need a combination of multiple imaging methods to explore tumor hypoxia in deeply. Future studies should therefore focus on improving the properties of 18F-FMISO and trying to combine this tracer with other hypoxia imaging methods in order to increase sensitivity and specificity of hypoxia detection. Thus, 18F-FMISO imaging currently offers vast potential for development to ultimately provide a comprehensive description of tumor hypoxic states and hence better guide clinical assessment and therapy design.
Abbreviations
18F-FMISO PET, 18F-Fluoromisonidazole positron emission tomography; PO2, oxygen partial pressure; HIF-1, hypoxia inducible factor 1; VEGF, vascular endothelial growth factor; SUVmax, maximum standardized uptake value; BOLD fMRI, blood oxygenation-level dependent functional magnetic resonance imaging; HNSCC, head and neck squamous cell carcinoma; RCC, renal cell carcinoma; 18F-FETNIM, 18F-Fluoroerythronitroimidazole; 18F-FAZA, 18F-Fluoroazomycinarabinoside; 18F-HX4, 18F-3-fluoro-2-(4-((2-nitro-1H-imidazol-1-yl)methyl)-1H-1, 2, 3-triazol-1-yl)-propan-1-ol; 18F-EF3, 18F-(2-2-nitroimidazol-1-yl)-N-(3,3,3-trifluoropropyl)-acetamide; 64Cu-ATSM, 64Cu-diacetyl-bis (N4-methylthiosemicarbazone).
ACKNOWLEDGMENTS
We thank Mr. Yang Liu for technical assistant.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
FUNDING
The authors’ work was supported, in part, by the National Basic Research Program of China (2015CB931800), National Natural Science Foundation of China (81627901, 31210103913, 81471724, 81101088, 81130028), Heilongjiang Province Foundation for Returned Overseas Chinese Scholars, and the Key Laboratory of Molecular Imaging Foundation (College of Heilongjiang Province).
REFERENCES
1. Guillaumond F, Leca J, Olivares O, Lavaut MN, Vidal N, Berthezene P, Dusetti NJ, Loncle C, Calvo E, Turrini O, Iovanna JL, Tomasini R, Vasseur S. Strengthened glycolysis under hypoxia supports tumor symbiosis and hexosamine biosynthesis in pancreatic adenocarcinoma. Proc Natl Acad Sci U S A. 2013; 110:3919–24. https://doi.org/10.1073/pnas.1219555110.
2. Ren Y, Hao P, Dutta B, Cheow ES, Sim KH, Gan CS, Lim SK, Sze SK. Hypoxia modulates A431 cellular pathways association to tumor radioresistance and enhanced migration revealed by comprehensive proteomic and functional studies. Mol Cell Proteomics. 2013; 12:485–98. https://doi.org/10.1074/mcp.M112.018325.
3. Semenza GL. Hypoxia and cancer. Cancer Metastasis Rev. 2007; 26:223–4. https://doi.org/10.1007/s10555-007-9058-y.
4. Cardenas-Navia LI, Richardson RA, Dewhirst MW. Targeting the molecular effects of a hypoxic tumor microenvironment. Front Biosci. 2007; 12:4061–78.
5. Carmeliet P. VEGF as a key mediator of angiogenesis in cancer. Oncology. 2005; 69:4–10. https://doi.org/10.1159/000088478.
6. Huang T, Civelek AC, Li J, Jiang H, Ng CK, Postel GC, Shen B, Li XF. Tumor microenvironment-dependent 18F-FDG, 18F-fluorothymidine, and 18F-misonidazole uptake: a pilot study in mouse models of human non-small cell lung cancer. J Nucl Med. 2012; 53:1262–8. https://doi.org/10.2967/jnumed.111.098087.
7. Cui YL, Wang X, Li XF. (18)F-fluoromisonidazole PET reveals spatial and temporal heterogeneity of hypoxia in mouse models of human non-small-cell lung cancer. Future Oncol. 2015; 11:2841–9. https://doi.org/10.2217/fon.15.205.
8. Lim JL, Berridge MS. An efficient radiosynthesis of [18F]fluoromisonidazole. Appl Radiat Isot. 1993; 44:1085–91.
9. Lee ST, Scott AM. Hypoxia positron emission tomography imaging with 18f-fluoromisonidazole. Semin Nucl Med. 2007; 37:451–61. https://doi.org/10.1053/j.semnuclmed.2007.07.001.
10. Wei Y, Zhao W, Huang Y, Yu Q, Zhu S, Wang S, Zhao S, Hu X, Yu J, Yuan S. A Comparative Study of Noninvasive Hypoxia Imaging with 18F-Fluoroerythronitroimidazole and 18F-Fluoromisonidazole PET/CT in Patients with Lung Cancer. PLoS One. 2016; 11:e0157606. https://doi.org/10.1371/journal.pone.0157606.
11. Wood KA, Wong WL, Saunders MI. [(64)Cu]diacetyl-bis(N(4)-methyl-thiosemicarbazone) - a radiotracer for tumor hypoxia. Nucl Med Biol. 2008; 35:393–400. https://doi.org/10.1016/j.nucmedbio.2008.02.002.
12. Ljungkvist AS, Bussink J, Kaanders JH, van der Kogel AJ. Dynamics of tumor hypoxia measured with bioreductive hypoxic cell markers. Radiat Res. 2007; 167:127–45.
13. Kizaka-Kondoh S, Konse-Nagasawa H. Significance of nitroimidazole compounds and hypoxia-inducible factor-1 for imaging tumor hypoxia. Cancer Sci. 2009; 100:1366–73. https://doi.org/10.1111/j.1349-7006.2009.01195.x.
14. Cyran CC, Paprottka PM, Eisenblatter M, Clevert DA, Rist C, Nikolaou K, Lauber K, Wenz F, Hausmann D, Reiser MF, Belka C, Niyazi M. Visualization, imaging and new preclinical diagnostics in radiation oncology. Radiat Oncol. 2014; 9:3. https://doi.org/10.1186/1748-717X-9-3.
15. Gagel B, Reinartz P, Dimartino E, Zimny M, Pinkawa M, Maneschi P, Stanzel S, Hamacher K, Coenen HH, Westhofen M, Bull U, Eble MJ. pO(2) Polarography versus positron emission tomography ([(18)F] fluoromisonidazole, [(18)F]-2-fluoro-2'-deoxyglucose). An appraisal of radiotherapeutically relevant hypoxia. Strahlenther Onkol. 2004; 180:616–22. https://doi.org/10.1007/s00066-004-1229-y.
16. Bentzen L, Keiding S, Nordsmark M, Falborg L, Hansen SB, Keller J, Nielsen OS, Overgaard J. Tumour oxygenation assessed by 18F-fluoromisonidazole PET and polarographic needle electrodes in human soft tissue tumours. Radiother Oncol. 2003; 67:339–44.
17. Mortensen LS, Buus S, Nordsmark M, Bentzen L, Munk OL, Keiding S, Overgaard J. Identifying hypoxia in human tumors: A correlation study between 18F-FMISO PET and the Eppendorf oxygen-sensitive electrode. Acta Oncol. 2010; 49:934–40. https://doi.org/10.3109/0284186X.2010.516274.
18. Kaur B, Khwaja FW, Severson EA, Matheny SL, Brat DJ, Van Meir EG. Hypoxia and the hypoxia-inducible-factor pathway in glioma growth and angiogenesis. Neuro Oncol. 2005; 7:134–53. https://doi.org/10.1215/S1152851704001115.
19. Sato J, Kitagawa Y, Yamazaki Y, Hata H, Okamoto S, Shiga T, Shindoh M, Kuge Y, Tamaki N. 18F-fluoromisonidazole PET uptake is correlated with hypoxia-inducible factor-1alpha expression in oral squamous cell carcinoma. J Nucl Med. 2013; 54:1060–5. https://doi.org/10.2967/jnumed.112.114355.
20. Norikane T, Yamamoto Y, Maeda Y, Kudomi N, Matsunaga T, Haba R, Iwasaki A, Hoshikawa H, Nishiyama Y. Correlation of (18)F-fluoromisonidazole PET findings with HIF-1alpha and p53 expressions in head and neck cancer: comparison with (18)F-FDG PET. Nucl Med Commun. 2014; 35:30–5. https://doi.org/10.1097/MNM.0000000000000010.
21. Cher LM, Murone C, Lawrentschuk N, Ramdave S, Papenfuss A, Hannah A, O'Keefe GJ, Sachinidis JI, Berlangieri SU, Fabinyi G, Scott AM. Correlation of hypoxic cell fraction and angiogenesis with glucose metabolic rate in gliomas using 18F-fluoromisonidazole, 18F-FDG PET, and immunohistochemical studies. J Nucl Med. 2006; 47:410–8.
22. Spence AM, Muzi M, Swanson KR, O'Sullivan F, Rockhill JK, Rajendran JG, Adamsen TC, Link JM, Swanson PE, Yagle KJ, Rostomily RC, Silbergeld DL, Krohn KA. Regional hypoxia in glioblastoma multiforme quantified with [18F]fluoromisonidazole positron emission tomography before radiotherapy: correlation with time to progression and survival. Clin Cancer Res. 2008; 14:2623–30. https://doi.org/10.1158/1078-0432.CCR-07-4995.
23. Kawai N, Lin W, Cao WD, Ogawa D, Miyake K, Haba R, Maeda Y, Yamamoto Y, Nishiyama Y, Tamiya T. Correlation between (1)(8)F-fluoromisonidazole PET and expression of HIF-1alpha and VEGF in newly diagnosed and recurrent malignant gliomas. Eur J Nucl Med Mol Imaging. 2014; 41:1870–8. https://doi.org/10.1007/s00259-014-2776-9.
24. Dery MA, Michaud MD, Richard DE. Hypoxia-inducible factor 1: regulation by hypoxic and non-hypoxic activators. Int J Biochem Cell Biol. 2005; 37:535–40. https://doi.org/10.1016/j.biocel.2004.08.012.
25. Jewell UR, Kvietikova I, Scheid A, Bauer C, Wenger RH, Gassmann M. Induction of HIF-1alpha in response to hypoxia is instantaneous. FASEB J. 2001; 15:1312–4.
26. Stroka DM, Burkhardt T, Desbaillets I, Wenger RH, Neil DA, Bauer C, Gassmann M, Candinas D. HIF-1 is expressed in normoxic tissue and displays an organ-specific regulation under systemic hypoxia. FASEB J. 2001; 15:2445–53. https://doi.org/10.1096/fj.01-0125com.
27. Rajendran JG, Wilson DC, Conrad EU, Peterson LM, Bruckner JD, Rasey JS, Chin LK, Hofstrand PD, Grierson JR, Eary JF, Krohn KA. [(18)F]FMISO and [(18)F]FDG PET imaging in soft tissue sarcomas: correlation of hypoxia, metabolism and VEGF expression. Eur J Nucl Med Mol Imaging. 2003; 30:695–704. https://doi.org/10.1007/s00259-002-1096-7.
28. Baudelet C, Gallez B. How does blood oxygen level-dependent (BOLD) contrast correlate with oxygen partial pressure (pO2) inside tumors? Magn Reson Med. 2002; 48:980–6. https://doi.org/10.1002/mrm.10318.
29. Bar-Shalom R, Kagna O, Israel O, Guralnik L. Noninvasive diagnosis of solitary pulmonary lesions in cancer patients based on 2-fluoro-2-deoxy-D-glucose avidity on positron emission tomography/computed tomography. Cancer. 2008; 113:3213–21. https://doi.org/10.1002/cncr.23928.
30. Divgi CR. Molecular imaging of pulmonary cancer and inflammation. Proc Am Thorac Soc. 2009; 6:464–8. https://doi.org/10.1513/pats.200902-005AW.
31. Okamoto S, Shiga T, Yasuda K, Ito YM, Magota K, Kasai K, Kuge Y, Shirato H, Tamaki N. High reproducibility of tumor hypoxia evaluated by 18F-fluoromisonidazole PET for head and neck cancer. J Nucl Med. 2013; 54:201–7. https://doi.org/10.2967/jnumed.112.109330.
32. Lawrentschuk N, Poon AM, Foo SS, Putra LG, Murone C, Davis ID, Bolton DM, Scott AM. Assessing regional hypoxia in human renal tumours using 18F-fluoromisonidazole positron emission tomography. BJU Int. 2005; 96:540–6. https://doi.org/10.1111/j.1464-410X.2005.05681.x.
33. Nehmeh SA, Lee NY, Schroder H, Squire O, Zanzonico PB, Erdi YE, Greco C, Mageras G, Pham HS, Larson SM, Ling CC, Humm JL. Reproducibility of intratumor distribution of (18)F-fluoromisonidazole in head and neck cancer. Int J Radiat Oncol Biol Phys. 2008; 70:235–42. https://doi.org/10.1016/j.ijrobp.2007.08.036.
34. Maftei CA, Shi K, Bayer C, Astner ST, Vaupel P. Comparison of (immuno-)fluorescence data with serial [(1)(8)F]Fmiso PET/CT imaging for assessment of chronic and acute hypoxia in head and neck cancers. Radiother Oncol. 2011; 99:412–7. https://doi.org/10.1016/j.radonc.2011.05.079.
35. Warren DR, Partridge M. The role of necrosis, acute hypoxia and chronic hypoxia in 18F-FMISO PET image contrast: a computational modelling study. Phys Med Biol. 2016; 61: 8596–624. https://doi.org/10.1088/1361-6560/61/24/8596.
36. Valk PE, Mathis CA, Prados MD, Gilbert JC, Budinger TF. Hypoxia in human gliomas: demonstration by PET with fluorine-18-fluoromisonidazole. J Nucl Med. 1992; 33:2133–7.
37. Hirata K, Terasaka S, Shiga T, Hattori N, Magota K, Kobayashi H, Yamaguchi S, Houkin K, Tanaka S, Kuge Y, Tamaki N. (1)(8)F-Fluoromisonidazole positron emission tomography may differentiate glioblastoma multiforme from less malignant gliomas. Eur J Nucl Med Mol Imaging. 2012; 39:760–70. https://doi.org/10.1007/s00259-011-2037-0.
38. Yamamoto Y, Maeda Y, Kawai N, Kudomi N, Aga F, Ono Y, Nishiyama Y. Hypoxia assessed by 18F-fluoromisonidazole positron emission tomography in newly diagnosed gliomas. Nucl Med Commun. 2012; 33:621–5. https://doi.org/10.1097/MNM.0b013e3283529984.
39. Thorwarth D, Eschmann SM, Paulsen F, Alber M. A kinetic model for dynamic [18F]-Fmiso PET data to analyse tumour hypoxia. Phys Med Biol. 2005; 50:2209–24. https://doi.org/10.1088/0031-9155/50/10/002.
40. Kobayashi H, Hirata K, Yamaguchi S, Terasaka S, Shiga T, Houkin K. Usefulness of FMISO-PET for glioma analysis. Neurol Med Chir (Tokyo). 2013; 53:773–8.
41. Sadick M, Schoenberg SO, Hoermann K, Sadick H. Current oncologic concepts and emerging techniques for imaging of head and neck squamous cell cancer. GMS Curr Top Otorhinolaryngol Head Neck Surg. 2012; 11:Doc08. https://doi.org/10.3205/cto000090.
42. Parvathaneni U, Laramore GE, Liao JJ. Technical advances and pitfalls in head and neck radiotherapy. J Oncol. 2012; 2012:597467. https://doi.org/10.1155/2012/597467.
43. Grosso S, Doyen J, Parks SK, Bertero T, Paye A, Cardinaud B, Gounon P, Lacas-Gervais S, Noel A, Pouyssegur J, Barbry P, Mazure NM, Mari B. MiR-210 promotes a hypoxic phenotype and increases radioresistance in human lung cancer cell lines. Cell Death Dis. 2013; 4:e544. https://doi.org/10.1038/cddis.2013.71.
44. Overgaard J. Hypoxic radiosensitization: adored and ignored. J Clin Oncol. 2007; 25:4066–74. https://doi.org/10.1200/JCO.2007.12.7878.
45. Chang JH, Wada M, Anderson NJ, Lim Joon D, Lee ST, Gong SJ, Gunawardana DH, Sachinidis J, O'Keefe G, Gan HK, Khoo V, Scott AM. Hypoxia-targeted radiotherapy dose painting for head and neck cancer using (18)F-FMISO PET: a biological modeling study. Acta Oncol. 2013; 52:1723–9. https://doi.org/10.3109/0284186X.2012.759273.
46. Thorwarth D, Eschmann SM, Paulsen F, Alber M. A model of reoxygenation dynamics of head-and-neck tumors based on serial 18F-fluoromisonidazole positron emission tomography investigations. Int J Radiat Oncol Biol Phys. 2007; 68:515–21. https://doi.org/10.1016/j.ijrobp.2006.12.037.
47. Ljungkvist AS, Bussink J, Kaanders JH, Rijken PF, Begg AC, Raleigh JA, van der Kogel AJ. Hypoxic cell turnover in different solid tumor lines. Int J Radiat Oncol Biol Phys. 2005; 62:1157–68. https://doi.org/10.1016/j.ijrobp.2005.03.049.
48. Guise CP, Mowday AM, Ashoorzadeh A, Yuan R, Lin WH, Wu DH, Smaill JB, Patterson AV, Ding K. Bioreductive prodrugs as cancer therapeutics: targeting tumor hypoxia. Chin J Cancer. 2014; 33:80–6. https://doi.org/10.5732/cjc.012.10285.
49. Stokes AM, Hart CP, Quarles CC. Hypoxia Imaging With PET Correlates With Antitumor Activity of the Hypoxia-Activated Prodrug Evofosfamide (TH-302) in Rodent Glioma Models. Tomography. 2016; 2:229–37. https://doi.org/10.18383/j.tom.2016.00259.
50. Rischin D, Hicks RJ, Fisher R, Binns D, Corry J, Porceddu S, Peters LJ. Prognostic significance of [18F]-misonidazole positron emission tomography-detected tumor hypoxia in patients with advanced head and neck cancer randomly assigned to chemoradiation with or without tirapazamine: a substudy of Trans-Tasman Radiation Oncology Group Study 98.02. J Clin Oncol. 2006; 24:2098–104. https://doi.org/10.1200/JCO.2005.05.2878.
51. Lu X, Kang Y. Hypoxia and hypoxia-inducible factors: master regulators of metastasis. Clin Cancer Res. 2010; 16:5928–35. https://doi.org/10.1158/1078-0432.CCR-10-1360.
52. Tsai YP, Wu KJ. Hypoxia-regulated target genes implicated in tumor metastasis. J Biomed Sci. 2012; 19:102. https://doi.org/10.1186/1423-0127-19-102.
53. Li XF, Ma Y, Jiang H. Understanding hypoxia microenvironment of micrometastases. Journal of Solid Tumors. 2012; 2.
54. Huang T, Civelek AC, Zheng H, Ng CK, Duan X, Li J, Postel GC, Shen B, Li XF. (18)F-misonidazole PET imaging of hypoxia in micrometastases and macroscopic xenografts of human non-small cell lung cancer: a correlation with autoradiography and histological findings. Am J Nucl Med Mol Imaging. 2013; 3:142–53.
55. Narita T, Aoyama H, Hirata K, Onodera S, Shiga T, Kobayashi H, Murata J, Terasaka S, Tanaka S, Houkin K. Reoxygenation of glioblastoma multiforme treated with fractionated radiotherapy concomitant with temozolomide: changes defined by 18F-fluoromisonidazole positron emission tomography: two case reports. Jpn J Clin Oncol. 2012; 42:120–3. https://doi.org/10.1093/jjco/hyr181.
56. Cheng J, Lei L, Xu J, Sun Y, Zhang Y, Wang X, Pan L, Shao Z, Liu G. 18F-fluoromisonidazole PET/CT: a potential tool for predicting primary endocrine therapy resistance in breast cancer. J Nucl Med. 2013; 54:333–40. https://doi.org/10.2967/jnumed.112.111963.
57. Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, Apffelstaedt J, Smith R, Sleeboom HP, Janicke F, Pluzanska A, Dank M, Becquart D, et al. Superior efficacy of letrozole versus tamoxifen as first-line therapy for postmenopausal women with advanced breast cancer: results of a phase III study of the International Letrozole Breast Cancer Group. J Clin Oncol. 2001; 19:2596–606.
58. Ellis MJ, Coop A, Singh B, Tao Y, Llombart-Cussac A, Janicke F, Mauriac L, Quebe-Fehling E, Chaudri-Ross HA, Evans DB, Miller WR. Letrozole inhibits tumor proliferation more effectively than tamoxifen independent of HER1/2 expression status. Cancer Res. 2003; 63:6523–31.
59. Hugonnet F, Fournier L, Medioni J, Smadja C, Hindie E, Huchet V, Itti E, Cuenod CA, Chatellier G, Oudard S, Faraggi M. Metastatic renal cell carcinoma: relationship between initial metastasis hypoxia, change after 1 month's sunitinib, and therapeutic response: an 18F-fluoromisonidazole PET/CT study. J Nucl Med. 2011; 52:1048–55. https://doi.org/10.2967/jnumed.110.084517.
60. Gronroos T, Eskola O, Lehtio K, Minn H, Marjamaki P, Bergman J, Haaparanta M, Forsback S, Solin O. Pharmacokinetics of [18F]FETNIM: a potential marker for PET. J Nucl Med. 2001; 42:1397–404.
61. Lehtio K, Oikonen V, Nyman S, Gronroos T, Roivainen A, Eskola O, Minn H. Quantifying tumour hypoxia with fluorine-18 fluoroerythronitroimidazole ([18F]FETNIM) and PET using the tumour to plasma ratio. Eur J Nucl Med Mol Imaging. 2003; 30:101–8. https://doi.org/10.1007/s00259-002-1016-x.
62. Piert M, Machulla HJ, Picchio M, Reischl G, Ziegler S, Kumar P, Wester HJ, Beck R, McEwan AJ, Wiebe LI, Schwaiger M. Hypoxia-specific tumor imaging with 18F-fluoroazomycin arabinoside. J Nucl Med. 2005; 46:106–13.
63. Kurihara H, Honda N, Kono Y, Arai Y. Radiolabelled agents for PET imaging of tumor hypoxia. Curr Med Chem. 2012; 19:3282–9.
64. Carlin S, Humm JL. PET of hypoxia: current and future perspectives. J Nucl Med. 2012; 53:1171–4. https://doi.org/10.2967/jnumed.111.099770.
65. Chen L, Zhang Z, Kolb HC, Walsh JC, Zhang J, Guan Y. (1)(8)F-HX4 hypoxia imaging with PET/CT in head and neck cancer: a comparison with (1)(8)F-FMISO. Nucl Med Commun. 2012; 33:1096–102. https://doi.org/10.1097/MNM.0b013e3283571016.
66. Doss M, Zhang JJ, Belanger MJ, Stubbs JB, Hostetler ED, Alpaugh K, Kolb HC, Yu JQ. Biodistribution and radiation dosimetry of the hypoxia marker 18F-HX4 in monkeys and humans determined by using whole-body PET/CT. Nucl Med Commun. 2010; 31:1016–24. https://doi.org/10.1097/MNM.0b013e3283407950.
67. Zegers CM, van Elmpt W, Szardenings K, Kolb H, Waxman A, Subramaniam RM, Moon DH, Brunetti JC, Srinivas SM, Lambin P, Chien D. Repeatability of hypoxia PET imaging using [(1)(8)F]HX4 in lung and head and neck cancer patients: a prospective multicenter trial. Eur J Nucl Med Mol Imaging. 2015; 42:1840–9. https://doi.org/10.1007/s00259-015-3100-z.
68. Dubois L, Landuyt W, Cloetens L, Bol A, Bormans G, Haustermans K, Labar D, Nuyts J, Gregoire V, Mortelmans L. [18F]EF3 is not superior to [18F]FMISO for PET-based hypoxia evaluation as measured in a rat rhabdomyosarcoma tumour model. Eur J Nucl Med Mol Imaging. 2009; 36:209–18. https://doi.org/10.1007/s00259-008-0907-x.
69. Colombie M, Gouard S, Frindel M, Vidal A, Cherel M, Kraeber-Bodere F, Rousseau C, Bourgeois M. Focus on the Controversial Aspects of (64)Cu-ATSM in Tumoral Hypoxia Mapping by PET Imaging. Front Med (Lausanne). 2015; 2:58. https://doi.org/10.3389/fmed.2015.00058.
70. Yuan H, Schroeder T, Bowsher JE, Hedlund LW, Wong T, Dewhirst MW. Intertumoral differences in hypoxia selectivity of the PET imaging agent 64Cu(II)-diacetyl-bis(N4-methylthiosemicarbazone). J Nucl Med. 2006; 47:989–98.

