INTRODUCTION
Pediatric high grade gliomas (HGGs) are one of the most significant causes of morbidity and mortality among children due to their aggressive clinical behavior [1]. One of the driving forces behind the growth of HGG is thought to be a subpopulation of glioma stem cells (GSCs) [2]. The generation and maintenance of these tumorigenic cells is orchestrated in part by transcriptional and epigenetic changes [3], such as those induced by microRNAs (miRNAs).
MiRNAs are small non-coding RNAs (19-25 nucleotides long) and function as mRNA silencers and transcriptional regulators of gene expression. They have been identified to have important roles in tumor development and progression as oncomirs [3–5]. MiRNAs are also present in small (30-100 nm) sized membrane vesicles called exosomes [6, 7] which have an endocytic origin and are released by different cell types [8]. Exosomes have gained much attention since the discovery of their RNA content and the important role they have in cell to cell communication [9] as well as their possible role in tumor progression [10]. Previous studies have described miRNA expression in pediatric brain tumors [11, 12], differences between pediatric and adult gliomas [13], and in exosomes derived from adult glioblastoma (GBM) cell lines [14, 15]. However, nothing is known about the expression pattern of exosomal miRNAs, secreted by primary cell cultures originating from pediatric HGG patients. This is essential if we are to understand any potential role they have in the biology of HGG.
Here we used primary patient-derived GSCs to survey the expression of a large number of miRNAs – both exosome derived and cellular – in pediatric GSCs and compared these to normal neural fetal stem cells (NSCs) reference controls. We find that exosomes from GSCs altered the gene expression of recipient NSCs, suggesting that the exosomes could have a role in the tumor microenvironment influencing the properties of neighboring cells.
RESULTS
Characterization of exosomes released by glioma stem cells and neural stem cells
We used NSCs and patient-derived primary cell cultures from pediatric HGG (GSCs) grown in stem cell conditions. The GSCs have previously been thoroughly described [16]. Briefly, the cells express stem cell proteins, respond to differentiation cues and are tumor-initiating when orthotopically injected into mouse brains. The NSCs also express stem cell markers and have the capacity to differentiate (Supplementary Figure 1 and [17]). Extracellular vesicles released by the cells were isolated from the cell culture media (see Material and Methods). We characterized the exosomes based on their size and morphology using Transmission Electron Microscopy and Nanoparticle Tracking Analysis. The analyses confirmed the typical exosome shape and size (Figure 1A–1B) [18]. Presence of the exosomal surface markers CD81 and CD9 was indicated by Western blot. By contrast, Calnexin, an endoplasmic reticulum marker only present in cells, was absent (Figure 1C) [18].
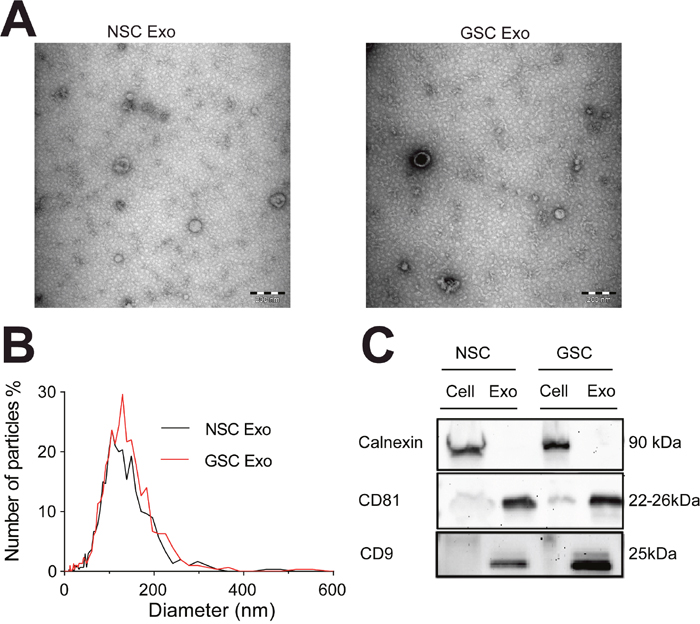
Figure 1: Characterization of exosomes isolated from neural and brain tumor stem cells. (A) Representative image from transmission electron microscopy of exosomes (Exo) (for each sample group), scale bars: 200 nm. (B) Determination of exosome size distribution by Nanoparticle Tracking Analysis and (C) characterization with Western blot of exosomal surface markers CD9 and CD81, and cell-specific marker Calnexin in cells and exosomes.
The miRNA expression pattern differs in cells and exosomes
We analyzed miRNA expression in cells and exosomes from six GSC and three NSC lines and detected, in total, 1954 miRNAs. Among these, 250 miRNAs were present in both sets of samples (in both cells and exosomes); 480 were present in all cell samples; 389 were detected in all of the exosome samples. The detected miRNAs in exosomes (exosomal miRNAs) clustered separately from those in cells (cellular miRNAs) based on their expression level (intensity) (Figure 2A). We detected 334 miRNAs that were present only in cells (cell-specific miRNAs) and 355 miRNAs that were present only in exosomes (exosome-specific miRNAs). Hence, both the quantity and nature of the miRNAs differed between cells and exosomes based on expression levels and the type of miRNA expressed.
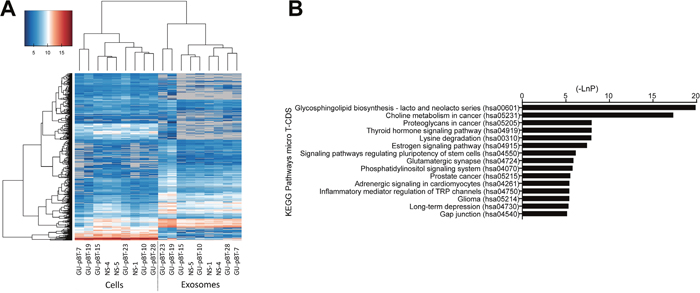
Figure 2: miRNA distribution and targeted pathways. (A) Hierarchical clustering with Euclidean distance of log2 miRNA expression in all samples. Undetected miRNAs are colored grey. The exosomes and cells cluster separately based on their miRNA intensity. (B) Top 15 targeted KEGG pathways of the differentially expressed miRNAs between Glioma stem cell (GSC) exosomes and normal neural stem cell (NSC) exosomes include several relevant cancer pathways.
Identification of differentially expressed miRNAs
We next focused on those miRNAs differentially expressed in the GSCs and NSCs. We detected 10 upregulated and 14 down-regulated miRNAs in the GSCs compared to the NSCs (Table 1, p<0.05). Reassuringly, the top up-regulated candidate was the glioma-associated miRNA-497-5p [19].
Table 1: Differentially expressed microRNAs in GSCs compared to NSCs
miRNA |
FCa |
p-valueb |
BHc |
|---|---|---|---|
Up-regulated |
|||
miR-497-5p |
11.76 |
2.70E-06 |
1.30E-03 |
miR-551b-3p |
3.04 |
5.92E-03 |
5.91E-01 |
miR-195-5p |
2.99 |
5.79E-03 |
5.91E-01 |
miR-9-3p |
2.68 |
2.08E-02 |
7.70E-01 |
miR-101-3p |
2.48 |
2.95E-02 |
8.27E-01 |
miR-505-5p |
2.26 |
4.90E-02 |
8.27E-01 |
miR-1199-5p |
2.15 |
3.96E-02 |
8.27E-01 |
miR-487a-3p |
2.09 |
2.67E-02 |
8.27E-01 |
miR-4783-5p |
2.05 |
4.85E-02 |
8.27E-01 |
miR-187-5p |
2.02 |
4.87E-02 |
8.27E-01 |
Down-regulated |
|||
miR-29a-3p |
2.20 |
4.77E-02 |
8.27E-01 |
miR-665 |
2.33 |
4.31E-02 |
8.27E-01 |
miR-4327 |
2.35 |
3.20E-02 |
8.27E-01 |
miR-3180 |
2.46 |
3.96E-02 |
8.27E-01 |
miR-4476 |
2.66 |
3.70E-02 |
8.27E-01 |
miR-3620-5p |
2.59 |
1.72E-02 |
7.40E-01 |
miR-4688 |
2.72 |
1.85E-02 |
7.40E-01 |
miR-221-3p |
5.61 |
1.84E-02 |
7.40E-01 |
miR-335-5p |
6.12 |
1.75E-02 |
7.40E-01 |
miR-3131 |
3.19 |
1.15E-02 |
6.90E-01 |
miR-6880-5p |
2.54 |
7.60E-03 |
5.91E-01 |
miR-4690-5p |
2.82 |
8.63E-03 |
5.91E-01 |
miR-4486 |
3.08 |
6.88E-03 |
5.91E-01 |
miR-8089 |
3.86 |
4.23E-03 |
5.91E-01 |
aFC-Fold change, b p-value <0.05 and c BH- Benjamini-Hochberg adjusted p-value.
We detected a larger number of differentially expressed miRNAs in the exosomes than in the cells; 37 miRNAs were up-regulated and five down-regulated in the GSC exosomes compared to the NSC exosomes (Table 2, p<0.05). Several of the upregulated miRNAs have not been identified previously in the context of GSC exosomes, such as miR-1290, miR-1246, miR-4299, miR-4732-5p, miR-6830-5p and miR-6165. We also detected many upregulated miRNAs that previously have been described to have a role in brain tumors such as miR-30c-1-3p [20], let-7a-5p [21], miR-24-3p [22] and miR-494-3p [23]. MiRNAs downregulated in the GSC exosomes included miR-4690-5p, miR-4443 and miR-198. We used qPCR to validate the miRNA array data, which confirmed the obtained results (Supplementary Figure 2). Enriched KEGG pathways targeted by the miRNAs that were differentially expressed between GSC and NSC exosomes included several cancer-related signaling pathways, such as Choline metabolism in cancer, Proteoglycans in cancer and Glioma (Figure 2B).
Table 2: Differentially expressed microRNAs in GSC exosomes compared to NSC exosomes
miRNA |
FCa |
p-valueb |
BHc |
|---|---|---|---|
Up-regulated |
|||
miR-4299 |
5.52 |
8.87E-05 |
1.38E-02 |
miR-4732-5p |
5.49 |
9.23E-05 |
1.38E-02 |
miR-6830-5p |
5.41 |
1.07E-04 |
1.38E-02 |
miR-7975 |
4.91 |
2.61E-04 |
2.34E-02 |
miR-1290 |
4.83 |
3.00E-04 |
2.34E-02 |
miR-1246 |
4.66 |
4.08E-04 |
2.65E-02 |
miR-3126-5p |
4.34 |
7.49E-04 |
4.16E-02 |
miR-6715b-5p |
4.27 |
8.64E-04 |
4.20E-02 |
miR-1273g-3p |
4.09 |
1.22E-03 |
4.70E-02 |
miR-8078 |
4.02 |
1.39E-03 |
4.70E-02 |
miR-4428 |
4.02 |
1.40E-03 |
4.70E-02 |
miR-6165 |
3.89 |
1.81E-03 |
4.70E-02 |
miR-4454 |
3.75 |
2.40E-03 |
5.81E-02 |
miR-5100 |
3.70 |
2.65E-03 |
5.81E-02 |
miR-30c-1-3p |
3.70 |
2.69E-03 |
5.81E-02 |
miR-652-5p |
3.56 |
3.54E-03 |
7.25E-02 |
miR-4531 |
3.52 |
3.82E-03 |
7.43E-02 |
miR-513b-5p |
3.30 |
6.06E-03 |
1.07E-01 |
miR-6778-5p |
3.29 |
6.19E-03 |
1.07E-01 |
miR-6851-5p |
3.28 |
6.31E-03 |
1.07E-01 |
miR-513a-5p |
3.19 |
7.68E-03 |
1.24E-01 |
miR-4286 |
3.16 |
8.19E-03 |
1.27E-01 |
let-7a-5p |
2.98 |
1.21E-02 |
1.74E-01 |
miR-5006-5p |
2.96 |
1.27E-02 |
1.77E-01 |
miR-3162-5p |
2.90 |
1.43E-02 |
1.86E-01 |
miR-4753-5p |
2.89 |
1.47E-02 |
1.86E-01 |
miR-711 |
2.88 |
1.51E-02 |
1.86E-01 |
miR-665 |
2.87 |
1.53E-02 |
1.86E-01 |
miR-24-3p |
2.83 |
1.69E-02 |
1.96E-01 |
miR-3714 |
2.82 |
1.72E-02 |
1.96E-01 |
miR-3122 |
2.75 |
2.00E-02 |
2.22E-01 |
miR-4649-5p |
2.61 |
2.78E-02 |
3.01E-01 |
miR-6893-5p |
2.57 |
3.03E-02 |
3.10E-01 |
miR-6870-5p |
2.56 |
3.09E-02 |
3.10E-01 |
miR-7977 |
2.55 |
3.17E-02 |
3.10E-01 |
miR-494-3p |
2.54 |
3.25E-02 |
3.10E-01 |
miR-4688 |
2.38 |
4.66E-02 |
4.32E-01 |
Down-regulated |
|||
miR-4690-5p |
3.91 |
1.75E-03 |
4.70E-02 |
miR-4443 |
3.91 |
1.74E-03 |
4.70E-02 |
miR-198 |
3.94 |
1.63E-03 |
4.70E-02 |
miR-3180 |
2.99 |
1.20E-02 |
1.74E-01 |
miR-6840-3p |
2.53 |
3.27E-02 |
3.10E-01 |
aFC-Fold change, b p-value <0.05 and c BH- Benjamini-Hochberg adjusted p-value.
In summary, we detected disease-associated pathways in the relevant miRNA expression differences between exosomes from GSCs and NSCs. Also, the exosome samples displayed a larger number of differentially expressed miRNAs between GSC and NSC than the cell samples, suggesting a specific active mechanism that is sorting miRNAs into exosomes and an important role of these vesicles in cell to cell communication.
Identification of differentially expressed miRNAs in exosomes compared to cells suggest a potential role of the exosomal miRNAs
Next, we compared the miRNA expression of the exosomes to the expression in their originating cells and detected 152 differentially expressed miRNAs between NSCs and their exosomes, and 196 differentially expressed miRNAs between GSCs and their exosomes. Most of these miRNAs were up-regulated in the exosomes (NSCs: 100, GSCs: 147) (Supplementary Tables 1-2).
To determine why some of the miRNAs had higher intensities in the exosomes compared to their originating cells, we searched for motifs in their sequence. We detected two often repeating patterns (Figure 3A) in the miRNAs from the GSC and NSC exosomes. One of the sequence patterns, GGAG, has previously been described [24] and termed ‘exo motif’ and the second motif was GGGGC. Figure 3B displays miRNAs overexpressed in GSC or NSC exosomes compared to their cell counterparts and that contains one or both of the identified motifs.
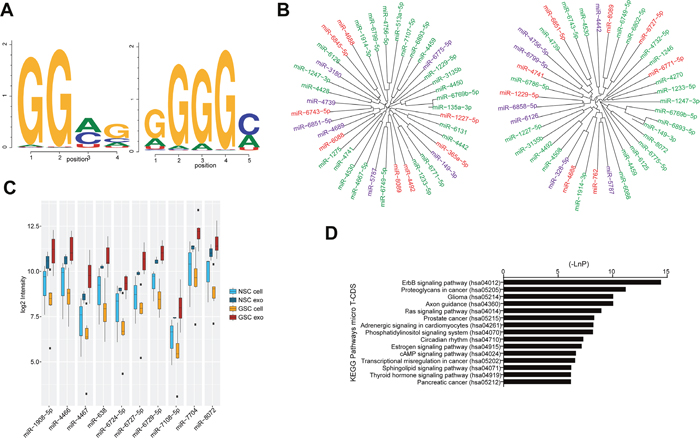
Figure 3: Exosomal miRNAs and their specific motifs. (A) The two frequently present motifs in miRNAs overexpressed in Glioma stem cells (GSC) exosomes compared to GSCs and in normal neural stem cell (NSC) exosomes compared to NSCs. (B) miRNAs overexpressed in NSC exosomes compared to NSCs (left) and miRNAs overexpressed in GSC exosomes compared to GSCs (right) whose sequences contain GGAG (green), GGGGC (red) or both GGAG and GGGGC (purple). (C) Top 10 ‘glioma exosome specific’ miRNAs: these miRNAs have significant differential expression in the GSC exosomes compared to their cells while there is no significant differential expression in NSC exosomes compared to their originating cells and (D) the top 15 targeted KEGG pathways by the ‘glioma exosome specific’ miRNAs.
Furthermore, we identified miRNAs that were differentially expressed between GSC exosomes and cells but not differentially expressed in NSC exosomes compared to their originating cells. This group of miRNAs, ‘glioma exosome specific’, was composed of 92 miRNAs, most of them (71) being up-regulated in the exosomes compared to their originating cells, for example miR-4467, miR-638 and miR-6727-5p (Figure 3C, Supplementary Table 3). In order to gain more information about the up-regulated ‘glioma exosome specific’ miRNAs, we searched for their targeted pathways using KEGG. Interestingly, most of the targeted pathways were cancer-, signaling- and glioma-related (Figure 3D). Based on these results, we hypothesize that loading of exosomal miRNAs are not random, but rather an effect of a well-defined mechanism.
Exosomes from glioma stem cells alter the gene expression of recipient cells
To determine if there is any potential functional role of exosomes secreted by GSCs, we aimed to test if the exosomes could affect normal recipient cells. We first verified, by confocal imaging, that exosomes can be taken up by NSCs (Supplementary Figure 3). Thereafter we treated cells with as follows, prior to gene expression analysis on TaqMan Low Density Array (TLDA) cards: 1) exosomes isolated from GSC (GU-pBT-28) media; 2) exosomes previously isolated from NSCs; and 3) no exosome treatment (Figure 4A). We profiled 192 genes that were predicted/validated target genes of the up-regulated GSC exosomal miRNAs or had a role in cell cycle, stemness, differentiation, glioma genesis, and neurogenesis.
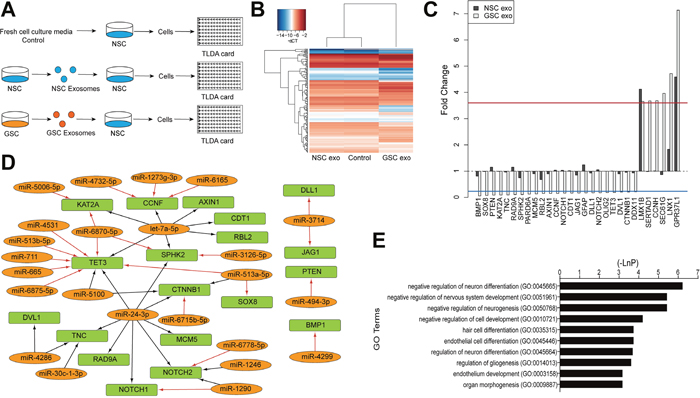
Figure 4: GSC exosomes effect on NSC gene expression. (A) Experimental set-up. (B) Hierarchical clustering of log2 intensities of genes measured by the TLDA cards in the three different experimental conditions, displaying a different profile of the GSC exosome treated cells. (C) Gene expression in NSC after GSC exosome treatment, where fold change (FC) less than the blue line or above the red line was defined as differentially expressed (Methods). (D) Up-regulated GSC exosomal miRNAs (orange) and their target (validated, Tarbase, arrow in black and predicted, microT-CDS, arrow in red) mRNAs (green) that were downregulated in NSC after GSC exosomal treatment and (E) top 10 biological processes affected by the differentially expressed genes in the GSC exosome treated NSCs.
We detected several gene expression changes induced by the GSC exosomes that were not induced by the NSC exosomes (Figure 4B). In total, 27 genes showed significant differential expression; 23 genes were down-regulated and four genes up-regulated (Figure 4C) in response to the GSC exosome treatment. Additionally, two up-regulated genes, LMX1B and GPR37L1, were also up-regulated in response to the treatment with NSC exosomes. Down-regulated transcripts included the tumor suppressor gene PTEN [25], stemness/differentiation/cell fate genes such as NOTCH1, NOTCH2, JAG1, DLL1, GFAP, and the cell cycle-related genes RAD9A, MCM5 and RBL2 (Figure 4C). The majority (19 of 23) of these down-regulated genes is predicted or validated mRNA targets for several of the up-regulated GSC exosomal miRNAs, which could indicate a direct effect (Figure 4D). For example, the GBM tumor suppressor gene TET3 [26] is likely downregulated by the delivered exosomal miR-24-3p, known to be up-regulated in brain tumors [22] and validated to target TET3 (Tarbase).
Up-regulated genes included the glioma related genes SEC61G and LNX1 and the cell cycle related SERTAD1 and CCNH (Figure 4C). The upregulation could be due to an indirect effect from the exosomal miRNAs or result from the mRNA content of the GSC exosomes.
Further, we examined which biological processes the altered genes are involved in and found relevant enriched GO terms such as nervous system development, neurogenesis or neuron differentiation (the genes measured by the TLDA cards was used as background for the enrichment analysis) (Figure 4E). In conclusion, these data suggest that GSC exosomal miRNAs altered the gene expression of the receiving NSCs in a manner that could affect several biological processes.
DISCUSSION
Previous studies have catalogued the miRNA expression profiles of pediatric GBM tumors [12, 13]. However, for a better understanding of these tumors’ biology it is crucial to study the cancer stem cells which have an important role in cancer development, progression and recurrence [2]. These cancer stem cells are thought to be regulated by several epigenetic mechanisms, with miRNAs being one of them [3]. MiRNAs are present in exosomes, vesicles that play an important role in cell communication. Hence, we here determined miRNA profiles of pediatric HGG-derived cancer stem cells and their exosomes. We further investigated their effect on NSCs.
We detected few differentially expressed cellular miRNAs between the GSCs and the NSCs, but a larger number in the exosome samples. These miRNAs are validated or predicted to be involved in relevant signaling pathways suggesting that they could have a role in the tumorigenic process. The loading of the miRNAs with a higher expression in exosomes compared to the cells therefore does not seem to be random, but rather suggestive of an active mechanism of sorting/loading, at least partly based on the recognition of different miRNA motifs that are favored for loading into the exosomes [24]. The specific mechanisms underlying this observation require further investigation.
We identified several exosomal miRNA which have previously been reported to have a role in cancer and stemness [27]. These included two evolutionary young miRNAs, miR-1246 and miR-1290, that have been described to have a role in neuronal differentiation [28]. The secretion of these miRNAs through exosomes could potentially be a result of the GSCs attempting to maintain their stemness by eliminating factors that induce differentiation. Alternatively, as the same miRNAs have been associated with cancer cell proliferation, stemness, invasion and chemoresistance in several tumor forms [29–33], one could speculate that the tumor cells mediate tumorigenic signals to cells in their microenvironment through the exosomes. We therefore investigated the potential of the exosomes to alter recipient cells, by profiling gene expression changes in NSCs resulting from GSC exosome treatment, and detected down-regulated expression of glioma-associated tumor suppressors PTEN [34] and TET3 [26]. Apart from inducing down-regulation of genes, the GSC exosomes also induced up-regulation of cancer-related genes such as SERTAD1 and SEC61G [35–37] which would support the hypothesis that exosomes released by tumor cells can promote tumorigenesis. These changes were not a direct effect of the up-regulated GSC exosomal miRNAs, but rather an indirect effect of the miRNAs or explained by other material in the exosomes. One limitation of our study is the relatively limited number of samples included. However pediatric cancer stem cells are not easily obtained and our unique cultures represent one of very few collections of well-characterized cells that are currently available [16]. Future studies will be needed to extend the broader significance of our observations. Moreover the specific contribution of each miRNA identified here remains to be explored.
In conclusion, the present study describes the miRNA expression pattern in pediatric HGG-derived stem cells and in the exosomes derived therefrom, as well as their potential role in affecting recipient cells. The study provides new insights into epigenetic regulation of cancer stem cells from pediatric brain tumors, as well as the possible roles of the exosomes in the regulation of normal cells in their microenvironment. These findings may act as a basis for further studies towards better diagnostics or prognostic biomarker development, as well as for deciphering the specific role of exosomes in cancer stem cell maintenance, regulation and progression.
MATERIALS AND METHODS
Patients and samples
The study was approved by the regional ethics committee (Dnr 604-12). Tumor samples were obtained after signed informed consent from the parents of children who underwent surgery at the Sahlgrenska University Hospital, Gothenburg, Sweden.
Cell cultures and exosome isolation
Established patient-derived primary cell cultures were grown in stem cell media (DMEM-F12 supplemented with B27 (Gibco), N2 (Gibco), EGF (20 ng/ml, Peprotech) and in some cases FGF-2 (20 ng/ml) on laminin (Sigma)-coated plastics as previously described [38]. Fresh media was added every 4 day. All established cell cultures were confirmed negative for mycoplasma contamination. The following cell lines were used: normal neural stem cells (NSCs): NS-1, NS-4, NS-5 and pediatric glioma stem cells (GSC): GU-pBT-7, GU-pBT-10, GU-pBT-15, GU-pBT-19, GU-pBT-23 and GU-pBT-28. The GSC lines are the same as in Wenger et al [16], described under different names, as follows: BPC-A7, BPC-B0, BPC-B5, BPC-B9, BPC-C3 and BPC-C8.
Conditioned medium was collected from each cell line and exosomes were purified by differential centrifugation [18]. In brief, the medium was centrifuged for 10 min at 300g to eliminate cell contamination. Supernatants were further centrifuged for 10 min at 2000g to eliminate apoptotic bodies, followed by ultracentrifugation with Ti70 fixed-angel rotor (Optima L-90 K Beckman Coulter). The supernatant was centrifuged at 28000x g (19400 rpm) in order to pellet microvesicles, followed by filtration of the supernatant through a 0.22 μm filter. Exosomes were pelleted by ultracentrifugation at 118000x g (40000 rpm) for 120 min. The exosome pellets were re-suspended in PBS.
Exosome characterization
Western blot
For Western blot analysis, proteins were extracted from isolated exosomes with Radio-Immunoprecipitation Assay (RIPA) Buffer (Sigma-Aldrich) and volumes, corresponding to 20 μg of proteins, were separated on 10% a SDS-PAGE gel. Samples were then transferred onto a nitrocellulose membrane (Bio-Rad laboratories, Hercules, CA, USA), which was subsequently blocked in 5% Non-Fat Dry Milk (Bio-Rad Laboratories). The membrane was incubated with primary antibodies against Calnexin (1:1000; clone H-70; Santa Cruz Biotechnology, Santa Cruz, CA, USA), CD9 (1:1000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) and CD81 (1:800; clone H-121; Santa Cruz Biotechnology, CA, USA) overnight at 4°C. Secondary antibody was ECL anti-rabbit IgG horseradish peroxidase-linked F(ab’)2 fragment (donkey-anti rabbit, 1:10000; GE Healthcare, UK). The membrane was visualized with ECL Prime Western Blotting Detection (GE Healthcare Life Sciences) and a VersaDoc 4000 MP (Bio-Rad Laboratories).
Nanoparticle tracking analysis
Size determination of exosomes was assayed using Zeta View (a Nanoparticle Tracking Analysis device: Particle Matrix, Germany).
Transmission electron microscopy
Exosomes (10μg) re-suspended in PBS were loaded onto Formvar/Carbon-coated grids (Ted Pella Inc., Redding, CA, USA) fixed in 2.5% glutaraldehyde, contrasted in 2% uranyl acetate and visualized with LEO 912AB Omega electron microscope (Carl Zeiss NTS, Jena, Germany) [39].
RNA isolation and microarray
Total RNA from exosomes and cells were extracted using QIAzol Lysis Reagent (Qiagen). Glycogen (Invitrogen) was added to increase the yield of RNA. Extracted RNA from cells was treated with TURBO DNase (Invitrogen) and enriched in small RNA fraction with RNA Clean & Concentrator™-5 (Zymo Research). RNA was quantified with Qubit RNA HS Assay Kit (Invitrogen).
MiRNA microarray analyses were carried out with 3D-Gene Human miRNA Oligo chip ver.21 (Toray Industries), which detects 2565 miRNA transcripts. The intensity of each miRNA was analyzed with the 3D-Gene Scanner 3000 (Toray) with auto gain, auto focus and auto analysis settings, according to the manufacturer’s instructions. The data normalization was performed on miRNA “spots” with background subtracted data. A normalization factor was calculated based on 25 divided with the median of the signal intensity of all background normalized data. Normalized data for each probe set and sample was calculated by multiplying background subtracted data with the normalization factor. The microarray data was analyzed with GenEx analysis software (MultiD Analyses).
Quantitative real-time PCR (qRT-PCR)
The relative quantification of selected differentially expressed miRNAs was performed by qRT-PCR with the miRCURY LNA™ Universal RT microRNA PCR, Starter Kit with validated primer sets (Exiqon, Denmark) using ABI 7500 FAST Real-Time PCR System. Extracted total RNA from exosomes and small RNA enriched from cell samples were used for reverse transcription (20 ng) by adding UniSp6 RNA Spike-in template according to the manufacturer protocol. Since no stable endogenous control for exosomal miRNA exists, we used UniSp6 for normalization. ΔCt values were transformed to relative quantities and log2 calculation was applied in order to compare the qRT-PCR data with the microarray data. Pearson’s Correlation was calculated between the qRT-PCR and microarray data for each sample group. All experiments were done in technical replicates.
Immunocytochemistry
NS-5 was stained with mouse monoclonal nestin, R&D MAB1259, 1:500; rabbit polyclonal SOX2, Abcam ab97959, 1:1000; mouse monoclonal GFAP, Sigma-Aldrich G3893 and incubated overnight at 4°C. When applicable, EdU was visualized according to the manufacturer’s instructions. Goat secondary antibody conjugated to Alexa dye, 1:1000 (Molecular Probes) was added for 1 h at room temperature and DAPI was used as a nuclear counterstain. Imaging of the cells was performed with the Operetta (Perkin Elmer). PKH67 Green Fluorescent Cell Linker for General Cell Membrane Labelling (Sigma-Aldrich) staining was used for exosomes for the confocal imaging (LSM 700 Carl Zeiss microscope) of the exosome up-take by cells.
Functional study
Total RNA was isolated from NSCs treated daily for 8 days with 1) 30μg/ml exosomes from GSCs (GU-pBT-28), 2) NSC exosomes or 3) only media (no exosomes). cDNA was synthesized using the High-Capacity RNA-to-cDNA Kit (Applied Biosystems) and 15-50 ng cDNA was loaded per port on TaqMan Custom Arrays (Applied Biosystems) examining 192 genes in duplicates. The genes were possible target genes of the up-regulated GSC exosomal miRNAs or have roles in cell cycle, stemness, differentiation, glioma genesis, and neurogenesis. The arrays were run on a Viia7 Real-Time PCR system (Applied Biosystems) according to the manufacturer’s instructions.
GAPDH was used as reference gene for ΔCT calculations, and hierarchical clustering was performed using Euclidean distance metric and average linkage. ΔΔCT-values were calculated between treatment 1) and 3) and treatment 2) and 3) (above), to estimate a Gaussian distribution. From this we estimated thresholds based on the mean ± 1.96*(standard deviation) to determine differentially expressed genes.
Statistical methods
Differential expression was analyzed with limma in R [40], for miRNAs detected with the microarray in all samples included in the two groups of each comparison. Hierarchical clustering on miRNA expression data was performed on miRNAs detected in all cell line samples and/or all exosome samples, using Euclidean distance metric and average linkage.
The search for significantly over-represented motifs was done on miRNAs identified as overexpressed in exosomes compared to cells for GSCs and/or NSCs using the online application Improbizer (https://users.soe.ucsc.edu/~kent/improbizer/improbizer.html). As background we used the miRNAs included on the arrays that were not identified as overexpressed.
Clustering of miRNA sequences was done with ClustalW, default settings, using the R package msa [41], and visualization was done using the R package ape [42].
Pathway analysis, Target gene prediction, network construction and GO analysis
Determination of targeted pathways based on Kyoto Encyclopedia of Genes and Genomes (KEGG pathways) for miRNAs was done using the DIANA TOOLS mirPath v3 software with the micro-T-CDS algorithm which predicts miRNA targets in CDS or 3’-UTR regions [43].
Based on Tarbase (validated miRNA targets) and microT-CDS (predicted miRNA targets) offered by the DIANA web tool, target networks were constructed for miRNAs and target genes with Cytoscape (Version 3.1.1) [44].
The involvement of target genes in biological processes was determined with the DAVID web tool using the GO BP_ALL settings [45, 46].
Abbreviations
GBM-glioblastoma multiforme; HGG-high grade gliomas; GSC-glioma stem cells; NSC-neural stem cells; Exo-exosomes; KEGG- Kyoto Encyclopedia of Genes and Genomes
Author contribution
Experiments were performed by A.T., T.R.L, A.W., H.C. and S.C.J. Data analysis was performed by T.K., A.T and H.C. Experimental design, interpretation of data and manuscript writing were performed by A.T., T.K. and H.C. H.C coordinated the study and scientific advice was given by H.C, J.L., A.D. and S.M.P. All authors revised and approved the final version of the manuscript.
ACKNOWLEDGMENTS
We would like to acknowledge The Center for Cellular Imaging of Gothenburg University for their assistance with the confocal imaging of the exosomal up-take by cells, TATAA Biocenter, Gothenburg, Sweden, for their technical assistance with the 3D-Gene Human miRNA array.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
FUNDING
This work was supported by the Swedish Cancer Society, the Swedish Children’s Cancer Society, the Swedish Research Council, the Swedish Society for Medical Research, the Wenner-Gren foundation, Wilhelm & Martina Lundgren, Assar Gabrielsson, The Royal Physiographic Society of Lund (The Nilsson-Ehle Endowments), Sahlgrenska Universitetssjukhusets fonder and a Marie Curie CIG from the EU’s Seventh Framework Programme (FP7).
REFERENCES
1. Fangusaro J. Pediatric high grade glioma: a review and update on tumor clinical characteristics and biology. Front Oncol. 2012; 2:105.
2. Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004; 432:396-401.
3. Muñoz P, Iliou MS, Esteller M. Epigenetic alterations involved in cancer stem cell reprogramming. Mol Oncol. 2012; 6:620-636.
4. Esquela-Kerscher A, Slack FJ. Oncomirs—microRNAs with a role in cancer. Nat Rev Cancer. 2006; 6:259-269.
5. Yang HW, Xing H, Johnson MD. A major role for microRNAs in glioblastoma cancer stem-like cells. Arch Pharm Res. 2015; 38:423-434.
6. Zhang J, Li S, Li L, Li M, Guo C, Yao J, Mi S. Exosome and exosomal microRNA: trafficking, sorting, and function. Genomics Proteomics Bioinformatics. 2015; 13:17-24.
7. Lunavat TR, Cheng L, Kim DK, Bhadury J, Jang SC, Lässer C, Sharples RA, López MD, Nilsson J, Gho YS. Small RNA deep sequencing discriminates subsets of extracellular vesicles released by melanoma cells–Evidence of unique microRNA cargos. RNA Biol. 2015; 12:810-823.
8. Zappulli V, Friis KP, Fitzpatrick Z, Maguire CA, Breakefield XO. Extracellular vesicles and intercellular communication within the nervous system. J Clin Invest. 2016; 126:1198-1207.
9. Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007; 9:654-659.
10. Sakha S, Muramatsu T, Ueda K, Inazawa J. Exosomal microRNA miR-1246 induces cell motility and invasion through the regulation of DENND2D in oral squamous cell carcinoma. Sci Rep. 2016; 6:38750.
11. Liu F, Xiong Y, Zhao Y, Tao L, Zhang Z, Zhang H, Liu Y, Feng G, Li B, He L. Identification of aberrant microRNA expression pattern in pediatric gliomas by microarray. Diagn Pathol. 2013; 8:1-10.
12. Birks DK, Barton VN, Donson AM, Handler MH, Vibhakar R, Foreman NK. Survey of MicroRNA expression in pediatric brain tumors. Pediatr Blood Cancer. 2011; 56:211-216.
13. Miele E, Buttarelli FR, Arcella A, Begalli F, Garg N, Silvano M, Po A, Baldi C, Carissimo G, Antonelli M. High-throughput microRNA profiling of pediatric high-grade gliomas. Neuro Oncol. 2013:not215.
14. Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Curry WT, Carter BS, Krichevsky AM, Breakefield XO. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008; 10:1470-1476.
15. Redzic JS, Ung TH, Graner MW. Glioblastoma extracellular vesicles: reservoirs of potential biomarkers. Pharmgenomics Pers Med. 2014; 7:65-77.
16. Wenger A, Larsson S, Danielsson A, Elbaek KJ, Kettunen P, Tisell M, Sabel M, Lannering B, Nordborg C, Schepke E, Carén H. Stem cell cultures derived from pediatric brain tumors accurately model the originating tumors. Oncotarget. 2017; 8:18626-18639. https://doi.org/10.18632/oncotarget.14826.
17. Carén H, Stricker SH, Bulstrode H, Gagrica S, Johnstone E, Bartlett TE, Feber A, Wilson G, Teschendorff AE, Bertone P, Beck S, Pollard SM. Glioblastoma stem cells respond to differentiation cues but fail to undergo commitment and terminal cell-cycle arrest. Stem Cell Rep. 2015; 5:829-842.
18. Théry C, Amigorena S, Raposo G, Clayton A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. 2006:3.22. 21-23.22. 29.
19. Lan J, Xue Y, Chen H, Zhao S, Wu Z, Fang J, Han C, Lou M. Hypoxia-induced miR-497 decreases glioma cell sensitivity to TMZ by inhibiting apoptosis. FEBS Lett. 2014; 588:3333-3339.
20. Quintavalle C, Donnarumma E, Iaboni M, Roscigno G, Garofalo M, Romano G, Fiore D, De Marinis P, Croce CM, Condorelli G. Effect of miR-21 and miR-30b/c on TRAIL-induced apoptosis in glioma cells. Oncogene. 2013; 32:4001-4008.
21. Li Y, Zhang X, Chen D, Ma C. Let-7a suppresses glioma cell proliferation and invasion through TGF-β/Smad3 signaling pathway by targeting HMGA2. Tumor Biol. 2016; 37:8107-8119.
22. Braoudaki M, Lambrou G, Giannikou K, Papadodima S, Lykoudi A, Stefanaki K, Sfakianos G, Kolialexi A, Tzortzatou-Stathopoulou F, Tzetis M. miR-15a and miR-24-1 as putative prognostic microRNA signatures for pediatric pilocytic astrocytomas and ependymomas. Tumor Biol. 2016; 37:9887-9897.
23. Li XT, Wang HZ, Wu ZW, Yang TQ, Zhao ZH, Chen GL, Xie XS, Li B, Wei YX, Huang YL, Zhou YX, Du ZW. miR-494-3p regulates cellular proliferation, invasion, migration, and apoptosis by PTEN/AKT signaling in human glioblastoma cells. Cell Mol Neurobiol. 2015; 35:679-687.
24. Villarroya-Beltri C, Gutiérrez-Vázquez C, Sánchez-Cabo F, Pérez-Hernández D, Vázquez J, Martin-Cofreces N, Martinez-Herrera DJ, Pascual-Montano A, Mittelbrunn M, Sánchez-Madrid F. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun. 2013; 4:2980.
25. Endersby R, Baker S. PTEN signaling in brain: neuropathology and tumorigenesis. Oncogene. 2008; 27:5416-5430.
26. Cui Q, Yang S, Ye P, Tian E, Sun G, Zhou J, Sun G, Liu X, Chen C, Murai K. Downregulation of TLX induces TET3 expression and inhibits glioblastoma stem cell self-renewal and tumorigenesis. Nature communications. 2016; 7:10637.
27. Hasegawa S, Eguchi H, Nagano H, Konno M, Tomimaru Y, Wada H, Hama N, Kawamoto K, Kobayashi S, Nishida N. MicroRNA-1246 expression associated with CCNG2-mediated chemoresistance and stemness in pancreatic cancer. Br J Cancer. 2014; 111:1572.
28. Yelamanchili SV, Morsey B, Harrison EB, Rennard D, Emanuel K, Thapa I, Bastola D, Fox H. The evolutionary young miR-1290 favors mitotic exit and differentiation of human neural progenitors through altering the cell cycle proteins. Cell Death Dis. 2014; 5:e982.
29. Kim G, An HJ, Lee MJ, Song JY, Jeong JY, Lee JH, Jeong HC. Hsa-miR-1246 and hsa-miR-1290 are associated with stemness and invasiveness of non-small cell lung cancer. Lung Cancer. 2016; 91:15-22.
30. Fu HL, Wu DP, Wang XF, Wang JG, Jiao F, Song LL, Xie H, Wen XY, Shan HS, Du YX, Zhao YP. Altered miRNA expression is associated with differentiation, invasion, and metastasis of esophageal squamous cell carcinoma (ESCC) in patients from Huaian, China. Cell Biochem Biophys. 2013; 67:657-668.
31. Sun Z, Meng C, Wang S, Zhou N, Guan M, Bai C, Lu S, Han Q, Zhao RC. MicroRNA-1246 enhances migration and invasion through CADM1 in hepatocellular carcinoma. BMC Cancer. 2014; 14:616.
32. Hasegawa S, Eguchi H, Nagano H, Konno M, Tomimaru Y, Wada H, Hama N, Kawamoto K, Kobayashi S, Nishida N, Koseki J, Nishimura T, Gotoh N, et al. MicroRNA-1246 expression associated with CCNG2-mediated chemoresistance and stemness in pancreatic cancer. Br J Cancer. 2014; 111:1572-1580.
33. Chen J, Yao D, Zhao S, He C, Ding N, Li L, Long F. MiR-1246 promotes SiHa cervical cancer cell proliferation, invasion, and migration through suppression of its target gene thrombospondin 2. Arch Gynecol Obstet. 2014; 290:725-732.
34. Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. 2008; 455:1061-1068.
35. Jung S, Li C, Jeong D, Lee S, Ohk J, Park M, Han S, Duan J, Kim C, Yang Y. Oncogenic function of p34SEI-1 via NEDD4–1–mediated PTEN ubiquitination/degradation and activation of the PI3K/AKT pathway. Int J Oncol. 2013; 43:1587-1595.
36. Lu Z, Zhou L, Killela P, Rasheed AB, Di C, Poe WE, McLendon RE, Bigner DD, Nicchitta C, Yan H. Glioblastoma proto-oncogene SEC61γ is required for tumor cell survival and response to endoplasmic reticulum stress. Cancer Res. 2009; 69:9105-9111.
37. You J, Liu J, Bao Y, Wang L, Yu Y, Wang L, Wu D, Liu C, Wang N, Wang F, Wang F, Xu L, Tian X, et al. SEI1 induces genomic instability by inhibiting DNA damage response in ovarian cancer. Cancer Lett. 2017; 385:271-279.
38. Pollard SM. in vitro expansion of fetal neural progenitors as adherent cell lines. Methods Mol Biol. 2013; 1059:13-24.
39. Lunavat TR, Jang SC, Nilsson L, Park HT, Repiska G, Lässer C, Nilsson JA, Gho YS, Lötvall J. RNAi delivery by exosome-mimetic nanovesicles–Implications for targeting c-Myc in cancer. Biomaterials. 2016; 102:231-238.
40. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47.
41. Bodenhofer U, Bonatesta E, Horejš-Kainrath C, Hochreiter S. msa: an R package for multiple sequence alignment. Bioinformatics. 2015; 31:3997-3999.
42. Popescu A-A, Huber KT, Paradis E. ape 3.0: new tools for distance-based phylogenetics and evolutionary analysis in R. Bioinformatics. 2012; 28:1536-1537.
43. Vlachos IS, Zagganas K, Paraskevopoulou MD, Georgakilas G, Karagkouni D, Vergoulis T, Dalamagas T, Hatzigeorgiou AG. DIANA-miRPath v3. 0: deciphering microRNA function with experimental support. Nucleic Acids Res. 2015; 43:W460-466.
44. Kohl M, Wiese S, Warscheid B. Cytoscape: software for visualization and analysis of biological networks. Methods Mol Biol. 2011; 696:291-303.
45. Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009; 4:44-57.
46. Huang DW, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009; 37:1-13.

