INTRODUCTION
With the development of economy and society, the incidence of acute myocardial infarction (AIM) showed an upward trend year by year. World Health Organization (WHO) has predicted that by 2020, AMI would become one of the major causes of human death [1-2]. Although drug clinical trials on myocardial ischemic–reperfusion injury (IRI) have yielded encouraging results, the actual effect of clinical treatment is not satisfactory [3]. Therefore, development of new cardioprotective drugs against myocardial IRI remains the research hotspot.
It is universally acknowledged for us that the factors such as oxygen radicals, calcium overload, etc. could induce the pathological process of myocardial I/R injury. Oxidative stress is usually relative to the increased formation of reactive oxygen species [4, 5]. Oxygen derived free radicals are recognized to play a leading part in the genesis of tissue injury in ischemia and reperfusion of the heart [6-9]. It has been proved of benefit in many numerous researches that after ischemic insult resulting in reducing inflammatory responses during reperfusion [10, 11]. Toll-like receptor (TLR) is the important protein that is involved in the nonspecific immunity. By recognizing the pathogen associated molecular patterns, it could induce the cytokine to play the role of anti-infection [12]. By now, over 10 subtypes of TLR have been found in the human body. The different subtype can recognize the different receptor and all TLR ligands can act as the immune adjuvant. According to the previous researches, TLR-4 could activate many cytokine and promote the formation of active oxygen free radicals [13, 14]. The nuclear factor κB (NF-κB) pathway was involved in the tissue injury and stress reaction, while the myocardial ischemia reperfusion could activate the NF-kB pathway in the further process of oxidative stress and calcium overloaded [15, 16].
Ginkgo biloba which is one of the commonly used Chinese medicinal herbs has been be applied to ameliorate memory loss, and respiratory function for the thousand years in East China [17]. Recently, Ginkgo biloba leaves extracts (EGb 761) are amongst the top-selling phytomedicines in the world [18]. The beneficial pharmacologic effects and biological activities of scavenging blood free radical, anti-inflammation, anti-tumor and cardioprotective properties had been proposed to related with EGb 761 [19, 20]. Clinically, EGb 761 has been promoted for the prescribed cure of dementia, vaso-occlusion, and cochleovestibular disorders in Chinese clinic trails [21]. Beneficial actions of EGb 761 against ischemia/reperfusion injury in lung, brain and renal ischemia have also been described [22-24]. The relationship between EGb 761 and signal molecules in myocardial ischemia reperfusion has not been well elucidated, however.
In this study, the cardioprotective properties of EGb 761 were characterized and the evidence of cardioprotective effects which were partly mediated through TLR-4/NF-κB signaling pathways is provided.
RESULTS
UPLC-PDA-TOF/MS fingerprint of EGb 761
UPLC-PDA-TOF/MS analysis was used for the determination of the major constituents of EGb 761. Representative UPLC-MS chromatograms of EGb 761 are shown in Figure 1. The results of analysis demonstrated that the major constituents of EGb 761 were 1 (-)-epigallocatechin (2.21%), 2 (+)-catechin hydrate (0.85%), 3 (-)-epicatechin (1.01%), 4 quercetin 3-O-[6-O-(α-L-rhamnosyl)-β-D-glucoside] (3.45%), 5 quercetin 3-O-β-D-glucoside (6.44%), 6 bilobalide (5.44%), 7 quercetin 3-O-[4-O-(α-L-rhamnosyl)-β-D-glucoside] (3.21%), 8 ginkgolide C (5.98%), 9 quercetin 3-O-α-L-rhamnoside (4.32%), 10 ginkgolide B (1.01%), 11 ginkgolide A (3.99%), 12 luteolin (1.01%), 13 apigenin (2.01%), 14 kaempferol (0.57%), 15 isorhamnetin (0.41%), 16 bilobetin (0.20%), and 17 genkwanin (0.74%).
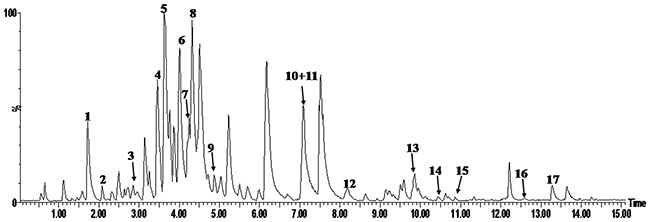
Figure 1: Representative UPLC- PDA -TOFMS chromatograms of EGb 761 in the negative ion mode.
Effect of EGb 761 on cardiac function
Pre-treatment with EGb 761 in significantly increased LVSP, +dP/dtma x and -dp/dtmax, and reduced the LVEDP compared to IR group. These results suggest that structure restoration in the ischemic heart (Figure 2) was translated into cardiac function improvements. Thus, EGb 761 could improve cardiac contractibility, attenuate hypercontraction and reduce diastolic pressure, which is believed to be a major reason for increased O2 demand.
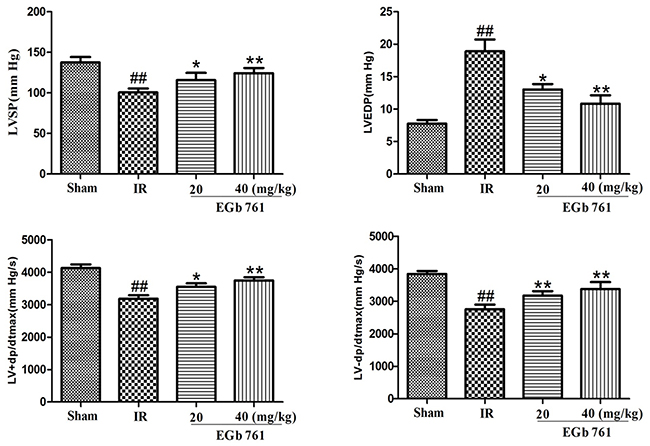
Figure 2: Pre-treatment with EGb 761 improve cardiac contractibility, attenuate hypercontraction and reduce diastolic pressure. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on the myocardium antioxidant enzymes
The myocardium antioxidant enzymes (Figure 3) demonstrated insignificant changes following the treatment of EGb 761. Compared with the sham group, IR rats demonstrated significant increase in MDA content, while that of antioxidant enzyme activities, such as SOD, CAT, GSH-Px and GR, were significantly decreased. Treatment of IR rats with EGb 761 enhanced the antioxidant enzymes activities.
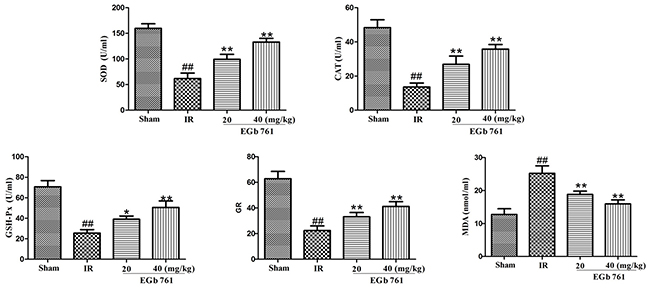
Figure 3: EGb 761 enhanced the antioxidant enzymes activities. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on release of AST, LDH, CK-MB
Results presented in Figure 4 Indicated that in IR group, the content levels of LDH, CK-MB and AST were remarkably higher than those in the sham group. In comparison with IR group, the levels of AST, CK-MB and LDH contents in the rats treated with EGb 761 were decreased.
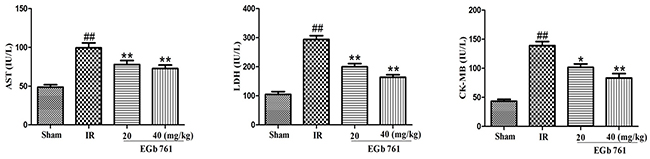
Figure 4: EGb 761 decreased the level of AST, CK-MB and LDH. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on Na+–K+–ATPase and Ca2+–ATPase activities
Results presented in Figure 5 showed us that in IR group the Na+–K+–ATPase and Ca2+–ATPase activities were remarkably lower compared with those in the sham group. Also, compared with IR group, the Na+–K+–ATPase and Ca2+–ATPase activities went up obviously in the rats treated with EGb 761.
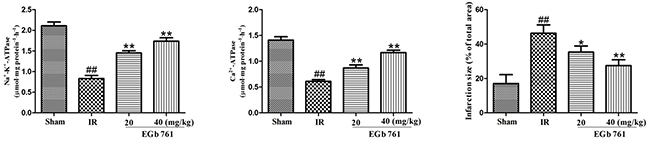
Figure 5: EGb 761 increased Na+–K+–ATPase and Ca2+–ATPase activities and percentage of infarct size. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on infarct size
As shown in Figure 5, the infarct size percentage in IR group was dramatically much higher than the sham group. The EGb 761 group with the administration of 20 or 40 mg/kg could decreased the percentage of infarct size significantly.
Effects of EGb 761 on pro-inflammatory cytokines
As shown in Figure 6, compared with the sham group, the levels of TNF-α, IL-6 and IL-1β in serum increased significantly in the IR rats group. However, the administrations of EGb 761 (20 and 40 mg/kg) had decreased levels of TNF-α, IL-6 and IL-1β.
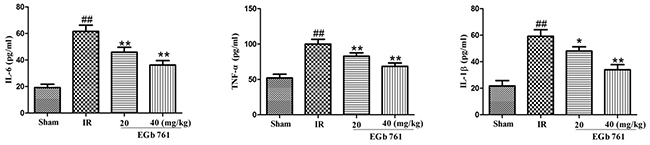
Figure 6: EGb 761 inhibited inflammatory cytokines. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on myocardial histology
Hematoxylin and eosin staining was adopted in the evaluating the protective effects of EGb 761. Some morphological damage were found in the MI/R group. However, EGb 761 with an administration of 20 and 40 mg/kg weakened the aforementioned morphological disorders (Figure 7).
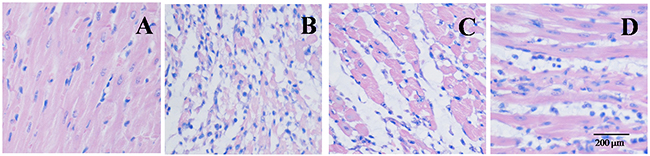
Figure 7: EGb 761 inhibited infiltration of inflammatory cells in Myocardial Histology. (A) sham group; (B) IR group; (C) IR + EGb 761 (20 mg/kg) group; (D) IR + EGb 761 (40 mg/kg) group. Scar bar: 200 μM (upper panel).
Effect of EGb 761 on Bax and Bcl-2 expressions
The expression of Bax protein notably increased in the myocardium tissue of I/R group. The expression of Bcl-2 protein obviously decreased by the contrast. The exchanges of MI/R in the Bax and Bcl-2 expressions were correspondingly induced by EGb 761 (20 and 40 mg/kg) administration (Figure 8).
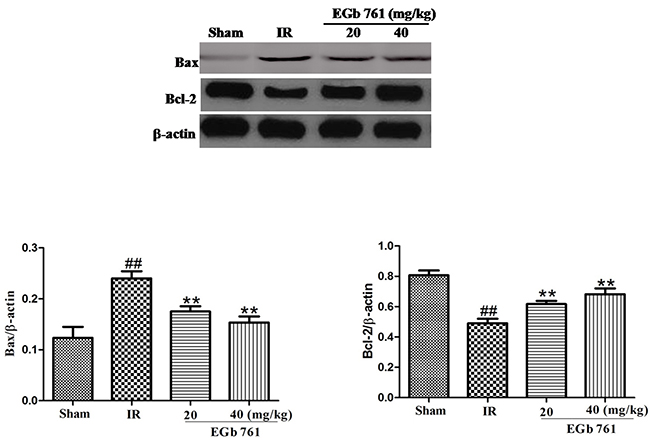
Figure 8: EGb 761 decreased Bax and increased Bcl-2 expressions. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
Effect of EGb 761 on TLR-4/NF-κB signaling pathway
In the present studys, the expression content of TLR4, p-TLR4, NF-κB p65 and p-NF-κB p65 were determined to find the effects of EGb 761 on TLR4 signaling pathway. Compared with the sham group rats, the expressions of p-TLR4 and p-NF-κB p65 both greatly raised a lot in I/R group. After administration of EGb 761 (20 and 40 mg/kg), the expressions of p-TLR4 and nuclear p-NF-κB p65 were reduced (Figure 9). These results indicated that the potential possessive reactions of EGb 761 on great I/R injury were on the basic of the inhibition of TLR4/NF-κB signaling pathway which may be a meaningful mechanism (Figure 9).
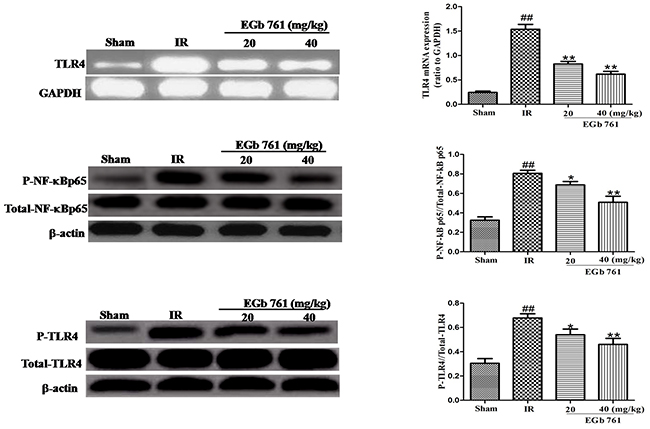
Figure 9: EGb 761 inhibited TLR-4/NF-κB-mediated inflammatory signalings in the myocardium tissue. Values were expressed as means ± SDs. ## P <0.01 compared with sham group. *P < 0.05 and ** P < 0.01 compared with IR group (n=10).
DISCUSSION
In normal, healthy cells could availably scavenge free radicals by plenty of antioxidants. Nonetheless, the unusual formation of reactive oxygen species dramatically results in breaking the balance with the expanded demand on the antioxidant defense system in pathological conditions which is similar to ischemic–reperfusion injury (IRI). With the accumulation of reactive oxygen species, the endogenous antioxidants are depleted at the same time such as SOD, CAT and GPx. It is of possibility to restrict the oxidative stress and reduce the induced tissue damage in order to prevent or alleviate disease progression by facilitating the balance with less oxidative stress. Present studies have shown that the ischemic–reperfusion injury was in connection with the accrued oxidative stress which was proved by the depleted myocardial endogenous antioxidants of SOD, GSH-Px, GR and CAT. On the other side, the reactive oxygen species can be scavenged precisely by EGb 761. Also, this is supported by the reduced level contains of AST, LDH and CK-MB in EGb 761 group.
Most members of interleukin-1 family are the pro-inflammatory cytokines, which could stimulate the inflammatory and autoimmune response [25]. The results of this study indicated that the expression of inflammatory factors in IR group was up-regulated, which was closely related to the injury mechanism of involved cardiac muscle. The major active calcium of Ca2+–ATPase protein transport takes the responsibility for keeping the maintenance of normal calcium levels intracellularly in many cell types. The Na+/K+–ATPase (sodiumpump) behaves as heterodimer protein which crucially retains cell homeostasis. The present studies have indicated that the observed Na+/K+–ATPase and Ca2+–ATPase with decreased activity may be explained as the cardiovascular damage index which is induced by IR. TLR4 which in the immune reaction response is one of the pattern recognition receptors not only takes participates but also makes I/R injury contributions to the inflammatory response. It mainly plays its role by mediating the MyD88 dependent and independent pathways. Through the MyD88 dependent pathway, it could activate the NF-kB, cause its transfer in the nucleus and activate the transcription of cytokine to release the related inflammatory cytokines [26]. The production of cytokine could also play the role of positive feedback to further activate the NF-kB. Meanwhile, there was the certain regulation of negative feedback. When the activation of NF-kB was inhibited, the mediation of TLR-4 was reduced and the expression was blocked as well [26]. The administration of EGb 761 with 20 and 40 mg/kg could reduce both the quantity of p-TLR4 and nuclear expression of p-NF-κB p65. It may be an important mechanism to inhibit the signaling pathway of TLR4/NF-κB to potentially on I/R injury exert the positive effects of EGb 761.
In summary, the observations in this study extended the vision in rat model was demonstrated of the protective effects of EGb 761 against I/R injury for the first time which was related with the downregulation of TLR4/NF-κB and inhibition of inflammatory response.
MATERIALS AND METHODS
Main reagents and kits
TNF-α, IL-6 and IL-1β enzyme-linked immunosorbent assay (ELISA) kits were provided by Nanjing KeyGEN Biotech. CO., Ltd. (Nanjing, China). CK, AST, LDH, MDA, SOD, CAT, GSH-Px and GR assay kits were produced by Jiancheng Bioengineering Institute (Nanjing, China). Antibodies against TLR4, NF-κB p65, B-cell lymphoma-2 (Bcl-2) and B-cell lymphoma-2 associated X (Bax) were purchased from Cell Signaling Technology (Beverly, MA, USA). TLR4 primers were obtained from TaKaRa Biotechnology Co. (Dalian, China). (The TLR4 forward primer: up, CTATCATCAGTGTAT CGGTG; down, CAGTCCTCATTCTGG CTC, the GAPDH forward primer: AATGCATCCTGCCACCAC CAACTGC, reverse primer: GGAGGCCATGTAGTAGG CCATGAGGTC). The Chinese medicine of EGb 761 which contained 6% of terpene trilactones, 24% of flavonol glycosides and less than 5 ppm of ginkgolic acids was purchased from Jiangsu Shenlong Pharmaceutical Co., 110722, Yancheng, China.
Animals
The Male Sprague-Dawley rats which were divided into four groups with 10 rats in each weighed 200–220 g and were provided by Shanghai Slac Laboratory Animal Co. LTD (Shanghai, China). The animals were fed individually under constant temperature (25 ± 1°C and humidity with a 12 h light/dark cycle and with a rodent standard diet with free access to water ad libitum. This study was performed in adherence with the National Institutes of Health Guidelines for the Use of Laboratory Animals.
Chemical analysis of EGb 761
The multi-components of the G. biloba extract were characterized by UPLC-PDA-TOF/MS. An ACQUITY UPLC™ BEH C18 (2.1 ×100 mm I.D., 1.7 μm, Waters, Milford, USA) column was used for the analyses. The mobile phase composed of A (acetonitrile) and B (0.1% formic acid, v/v) with a gradient elution: 0-15 min, 5-95% A.
Experimental protocol
All the male Sprague-Dawley rats were divided into four groups randomly: (1) Sham group dealt with saline vehicle but without I/R; (2) Ischemic reperfusion (IR) group given with saline alone; (3) I/R group given with EGb 761 of 20 mg/kg; (4) I/R group given with EGb 761 of 40 mg/kg. In the sham group and IR control group, the rats (10 rats in each) before IR operation were given saline by oral gavage for 15 d. In the two EGb 761 treatment groups, all the rats before IR operation were administered with EGb 761 (20 or 40 mg/kg) by oral gavage for 20 d. Rats were subjected to a coronary artery occlusion for 60 min followed by the reperfusion for 3 h. The snare encircling the coronary artery was used for occlusion by pulling up on the suture which was clamped with plastic tubes. Coronary artery reperfusion was restored by releasing the clamp. The sham groups were exposed to a time-matched normal perfusion without ischemia-reperfusion. For left ventricular (LV) pressure recordings, a water-filled small latex balloon by polythene cannula was inserted through left atrium into the left ventricle and was connected to the pressure transducer for the LV systolic and diastolic pressure measurements. The LV developed pressure (LVDP) was the difference between systolic and diastolic pressures. The LV end-diastolic pressure (LVEDP) was adjusted to 10mmHg at the beginning of the experiment and the LV pressures were differentiated to estimate the maximum rate of pressure development (+dP/dtmax) and the maximum rate of LV pressure decay (-dP/dtmax). At the end of reperfusion, blood samples were collected via carotid artery, then the left ventricle was obtained for determination of myocardial infarction size, and part of the anterior wall of the left ventricular myocardium near the cardiac apex was obtained for other analysis [27-29].
Evaluation of myocardial infarction and injury
After 60 min of myocardial ischemia and 180 min of reperfusion, hearts were immediately removed, cleaned of blood with physiological saline via the aorta, injected with 1% TTC (triphenyltetrazolium chloride) until they became red, and snap frozen in liquid nitrogen for 20 min. Subsequently, the hearts were cut into cross-sectional pieces and fixed in 10% formaldehyde for 12 h. The TTC-stained sections were photographed. The percentage of infarct size is determined by weight [28, 29].
Evaluation of antioxidant indices
SOD, CAT, GSH-Px, GR activities, MDA contents were used as indices of reactive oxygen species and membrane lipid peroxidation level. The content of MDA and activities of SOD, CAT, GSH-Px, GR were measured using commercial kits (JianCheng Bioengineering Institute, Nanjing, China) and analyzing with a spectrophotometer. Detailed manipulation process was performed according to the manufacturer’s instructions.
Determination of release of AST, LDH, CK-MB into serum
Myocardial cellular damage was evaluated by measuring serum AST, LDH, and CK-MB levels. Serum AST and LDH activities were measured spectrophotometrically, and serum CK-MB was quantified using a commercial ELISA kit according to the manufacturer’s instructions.
Determination of serum TNF-α, IL-6 and IL-1β
The levels of inflammatory cytokines, such as IL-6, IL-1β and TNF-α in the serum samples were quantified using specific ELISA kits for rat according to themanufacturers’ instructions.
Estimation of myocardial injury markers
Activities of Na+–K+–ATPase and Ca2+–ATPase from cardiac tissues were determined using commercially available standard kits by RT-9600 Semi-automatic Biochemical Analyzer. All measurements were performed according to the kits manufacturers’ instructions, respectively [28].
Histological examination of myocardium
Immediately after the sacrifice of the rats, the hearts were removed and fixed in 10% formalin solution. The heart tissue was processed for sectioning and staining by standard histological methods. Sections from the left ventricle were stained with hematoxylin and eosin (H&E) and examined by light microscopy at 200 × magnification.
Reverse transcription-polymerase chain reaction analysis
The sample amount of total RNA (2 μg) was used from each sample. RNA was reverse transcribed using avian myeloblastosis virus reverse transcriptase with random hexamers for 50 min at 42 °C. The complementary DNA was amplified under the following cycle conditions: 4 min at 94 °C for one cycle, 30 cycles at 94 °C for 50 s, 54 °C for 30 s and 72 °C for 1 min and lastly a final extension for 5 min at 72 °C. The polymerase chain reaction products were analyzed by 2% agarose gel stained with ethidium bromide and ultraviolet irradiation. The semiquantitative measure for atrial natriuretic peptide and brain natriuretic peptide mRNA was expressed as a ratio to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA.
Western blot analysis
Nuclear proteins were extracted from heart tissue using nuclear and cytoplasm extraction kit according to the manufacturer’s instructions. The protein concentration was determined by the Bradford method. After boiling the samples for 5 min, the protein samples were fractionated by sodium dodecyl sulfate polyacrylamide gel electropheresis (SDS-PAGE) (10–12% polyacrylamide gels) and transferred to polyvinylidene fluoride membrane (Millipore, Bedford, MA, USA). The membranes were blocked with milk powder at room temperature for 2 h. The membranes were incubated with primary antibodies of TLR4, Bcl-2, Bax and p65 at room temperature for 2 h, following primary antibody incubations, membranes were incubated with horseradish peroxidaselinked secondary antibodies (antirabbit, antimouse or antigoat IgG) mouse or antigoat IgG.
Statistical analysis
All values were expressed as the mean ± S.D. and analyzed by one-way analysis of variance (ANOVA) followed by Duncan's Multiple Range Test using SPSS version 13.0 software; a P-value of less than 0.05 was considered significant and P < 0.01 was considered to be statistically very significant.
ACKNOWLEDGMENTS AND FUNDING
This work was supported by the Jiangsu Provincial Medical Youth Talent (Grant No. QNRC2016715), Natural Science Foundation of Jiangsu Province (Grant No. BK20171068), Jiangsu Collaborative Innovation Center of Chinese Medicinal Resources Industrialization (Grant No. 012092002006-4) and 333 High-level Talents Training Project Funded by Jiangsu Province (Grant No. BRA2016387). We are also pleased to Ma Chun-hua for technical support from Cellular and Molecular Biology Center of China Pharmaceutical University.
CONFLICTS OF INTEREST
Authors declare that they have not any conflicts of interest.
REFERENCES
1. Zweier JL, Talukder MA. The role of oxidants and free radicals in reperfusion injury. Cardiovasc Res. 2006; 70: 181-90. https://doi.org/10.1016/j.cardiores.2006.02.025.
2. Roth E, Lantos J, Temes G, Varga G, Paroczai M, Karpati E. Cardioprotection during heart ischemia-reperfusion. Acta Chir Hung. 1997; 36: 306-9.
3. Zhao X, Fu W, Yuan W, Wu Z, Yang Z. A study of time window for oxidative stress associated with myocardial ischemia and reperfusion. Chin Med J (Engl). 1996; 109: 926-9.
4. Banerjee SK, Dinda AK, Manchanda SC, Maulik SK. Chronic garlic administration protects rat heart against oxidative stress induced by ischemic reperfusion injury. BMC Pharmacol. 2002; 2: 16.
5. Thompson-Gorman SL, Zweier JL. Evaluation of the role of xanthine oxidase in myocardial reperfusion injury. J Biol Chem. 1990; 265: 6656-63.
6. Visioli F, Keaney JF, Halliwell B. Antioxidants and cardiovascular disease; panaceas or tonics for tired sheep? Cardiovasc Res. 2000; 47: 409.
7. Zweier JL. Measurement of superoxide-derived free radicals in the reperfused heart. Evidence for a free radical mechanism of reperfusion injury. J Biol Chem. 1988; 263: 1353-7.
8. Bao W, Hu E, Tao L, Boyce R, Mirabile R, Thudium DT, Ma XL, Willette RN, Yue TL. Inhibition of Rho-kinase protects the heart against ischemia/reperfusion injury. Cardiovasc Res. 2004; 61: 548-58. https://doi.org/10.1016/j.cardiores.2003.12.004.
9. Sun J, Tan BK, Huang SH, Whiteman M, Zhu YZ. Effects of natural products on ischemic heart diseases and cardiovascular system. Acta Pharmacol Sin. 2002; 23: 1142-51.
10. Murphy JF. The global burden of disease. Ir Med J. 2013; 106: 4.
11. Mao SS, Hua R, Zhao XP, Qin X, Sun ZQ, Zhang Y, Wu YQ, Jia MX, Cao JL, Zhang YM. Exogenous administration of PACAP alleviates traumatic brain injury in rats through a mechanism involving the TLR4/MyD88/NF-kappaB pathway. J Neurotrauma. 2012; 29: 1941-59. https://doi.org/10.1089/neu.2011.2244.
12. Foley NM, Wang J, Redmond HP, Wang JH. Current knowledge and future directions of TLR and NOD signaling in sepsis. Mil Med Res. 2015; 2: 1. https://doi.org/10.1186/s40779-014-0029-7.
13. Li Y, Xie G, Li L, Jiang Z, Yue Z, Pan Z. The effect of TLR4/MyD88/NF-kappaB signaling pathway on proliferation and apoptosis in human nasopharyngeal carcinoma 5-8F cells induced by LPS. [Article in Chinese]. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2015; 29: 1012-5.
14. Dang YM, Huang G, Chen YR, Dang ZF, Chen C, Liu FL, Guo YF, Xie XD. Sulforaphane inhibits the proliferation of the BIU87 bladder cancer cell line via IGFBP-3 elevation. Asian Pac J Cancer Prev. 2014; 15: 1517-20.
15. Ozbek E, Cekmen M, Ilbey YO, Simsek A, Polat EC, Somay A. Atorvastatin prevents gentamicin-induced renal damage in rats through the inhibition of p38-MAPK and NF-kappaB pathways. Ren Fail. 2009; 31: 382-92.
16. Gao R, Brigstock DR. Activation of nuclear factor kappa B (NF-kappaB) by connective tissue growth factor (CCN2) is involved in sustaining the survival of primary rat hepatic stellate cells. Cell Commun Signal. 2005; 3: 14. https://doi.org/10.1186/1478-811x-3-14.
17. Huang CH, Yang ML, Tsai CH, Li YC, Lin YJ, Kuan YH. Ginkgo biloba leaves extract (EGb 761) attenuates lipopolysaccharide-induced acute lung injury via inhibition of oxidative stress and NF-kappaB-dependent matrix metalloproteinase-9 pathway. Phytomedicine. 2013; 20: 303-9. https://doi.org/10.1016/j.phymed.2012.11.004.
18. van Beek TA, Montoro P. Chemical analysis and quality control of Ginkgo biloba leaves, extracts, and phytopharmaceuticals. J Chromatogr A. 2009; 1216: 2002-32. https://doi.org/10.1016/j.chroma.2009.01.013.
19. Chan PC, Xia Q, Fu PP. Ginkgo biloba leave extract: biological, medicinal, and toxicological effects. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2007; 25: 211-44. https://doi.org/10.1080/10590500701569414.
20. Qiao ZY, Huang JH, Ma JW, Xu YW, Xie J, Liu HJ, Xiong SJ, Ge GH. Ginkgo biloba extract reducing myocardium cells apoptosis by regulating apoptotic related proteins expression in myocardium tissues. Mol Biol Rep. 2014; 41: 347-53. https://doi.org/10.1007/s11033-013-2868-9.
21. Diamond BJ, Shiflett SC, Feiwel N, Matheis RJ, Noskin O, Richards JA, Schoenberger NE. Ginkgo biloba extract: mechanisms and clinical indications. Arch Phys Med Rehabil. 2000; 81: 668-78.
22. Reynolds IJ, Hastings TG. Glutamate induces the production of reactive oxygen species in cultured forebrain neurons following NMDA receptor activation. J Neurosci. 1995; 15: 3318-27.
23. Aoshima H, Satoh T, Sakai N, Yamada M, Enokido Y, Ikeuchi T, Hatanaka H. Generation of free radicals during lipid hydroperoxide-triggered apoptosis in PC12h cells. Biochim Biophys Acta. 1997; 1345: 35-42.
24. Wang Y, Pei DS, Ji HX, Xing SH. Protective effect of a standardized Ginkgo extract (ginaton) on renal ischemia/reperfusion injury via suppressing the activation of JNK signal pathway. Phytomedicine. 2008; 15: 923-31. https://doi.org/10.1016/j.phymed.2008.09.003.
25. Castrillo A, de Las Heras B, Hortelano S, Rodriguez B, Villar A, Bosca L. Inhibition of the nuclear factor kappa B (NF-kappa B) pathway by tetracyclic kaurene diterpenes in macrophages. Specific effects on NF-kappa B-inducing kinase activity and on the coordinate activation of ERK and p38 MAPK. J Biol Chem. 2001; 276: 15854-60. https://doi.org/10.1074/jbc.M100010200.
26. Cheng Z, Taylor B, Ourthiague DR, Hoffmann A. Distinct single-cell signaling characteristics are conferred by the MyD88 and TRIF pathways during TLR4 activation. Sci Signal. 2015; 8: ra69. https://doi.org/10.1126/scisignal.aaa5208.
27. Li X, Jin Q, Yao Q, Xu B, Li Z, Tu C. Quercetin attenuates the activation of hepatic stellate cells and liver fibrosis in mice through modulation of HMGB1-TLR2/4-NF-kappaB signaling pathways. Toxicol Lett. 2016; 261: 1-12. https://doi.org/10.1016/j.toxlet.2016.09.002.
28. Liu H, Guo X, Chu Y, Lu S. Heart protective effects and mechanism of quercetin preconditioning on anti-myocardial ischemia reperfusion (IR) injuries in rats. Gene. 2014; 545: 149-55. https://doi.org/10.1016/j.gene.2014.04.043.
29. Lu M, Tang F, Zhang J, Luan A, Mei M, Xu C, Zhang S, Wang H, Maslov LN. Astragaloside IV attenuates injury caused by myocardial ischemia/reperfusion in rats via regulation of toll-like receptor 4/nuclear factor-kappaB signaling pathway. Phytother Res. 2015; 29: 599-606. https://doi.org/10.1002/ptr.5297.

