INTRODUCTION
Clonal heterogeneity of non-small cell lung cancer (NSCLC) has been well documented [1, 2] and contributes to the acquired resistance to targeted treatments [3-5]. Heterogeneity is a major challenge for the development of selective pharmacologic treatments. However, it remains unclear to which degree treatment outcome is affected by clonal evolution. There are multiple evidences that cells driving resistance in NSCLC are selected during treatment with EGFR-tyrosine kinase inhibitors (TKIs) [6]. Several mechanisms have been described, including the gatekeeper EGFR mutation p.T790M, MET amplification, and HER2 mutations [6-9]. The missense p.T790M is the substitution of a threonine (T) with a methionine (M) in codon 790; it is found in approximately 50-60% of NSCLCs after progression to EGFR-TKIs [10] and is the principal biomarker of resistance to EGFR-TKIs [7, 11, 12]. The p.T790M mutation is located within the gatekeeper residue of EGFR; it increases the affinity of the receptor to ATP [7] and strongly reduces the pharmacologic activity of first generation EGFR-TKIs. This view is supported by the observation that introduction of vectors carrying p.T790M into cells confers gefitinib resistance [13]. Approximately 0.32% to 78.95% of patients with NSCLC harboring EGFR activating mutations display p.T790M before administration of EGFR-TKI, although this percentage is variable according to test sensitivity [14]. As expected, subjects with a high p.T790M mutation burden had poorer clinical outcomes to EGFR-TKIs than patients with a low one [15]. It is therefore hypothesized that the selective pressure of EGFR-TKIs may select and favor the growth of p.T790M sub-clones, leading to acquired resistance.
Although data on pretreatment p.T790M [16] and at progression to first-line EGFR-TKI [11, 17-20] are available, the information on the relationship between p.T790M levels and activating EGFR mutation in patients progressing to first-generation EGFR-TKI are lacking. The availability of a minimally invasive approach based on the analysis of cell-free circulating tumor DNA in plasma (cftDNA) may represent a suitable approach to interrogate tumor heterogeneity by capturing DNA released from multiple tumor sites [4, 8] and may dynamically monitor the molecular evolution of drug resistance. Detection of p.T790M in combination with clinical signs of disease progression would represent a critical information to guide the administration of p.T790M-active drugs, including osimertinib that represents the new standard of care on this group of NSCLC patients [21, 22].
The aim of the present study was to document the relationship between plasma levels of activating EGFR mutations and p.T790M at the time of progression to first-line EGFR TKIs in patients with NSCLC and provide initial information on the dynamics of cftDNA changes upon treatment with osimertinib.
The results show that the amount of p.T790M can be substantially lower than EGFR activating mutations in drug resistance setting. Despite this, patients may respond well to osimertinib, thus a threshold value (i.e., copies/ml) of p.T790M is not required for successful treatment with p.T790M-selective drug.
RESULTS
A total of 49 patients were enrolled in this study; their characteristics are summarized in Table 1. The median PFS of the first-line EGFR-TKI treatment was 21 months (range 6-57 months). The activating mutations detected in cftDNA at the time of first-line EGFR-TKI progression were ex19del (42 subjects, 85.7%) and p.L858R (7 patients, 14.3%). The number of copies/mL of plasma of EGFR activating and p.T790M mutations are reported in Table 2; the median values of ex19del, p.L858R and p.T790M were 3,550 (range 130-3,390,000), 420 (range 170-671,000) and 500 copies/mL (range 80-194,000) respectively. The median value of the ratio p.T790M/EGFR activating mutations was 0.26 (range, 0.0004-0.9) showing, overall, a low to very low ratio of p.T790M vs. activating mutations in patients at the time of progression to first/second-line EGFR-TKI (p<0,0001, Figure 1).
Table 1: Characteristics of patients
Characteristic |
Statistics |
|---|---|
Nr. Patients |
49 |
Age (years) |
63 (42 - 81) |
Sex |
|
Male |
16 (32.6%) |
Female |
33 (67.3%) |
Smoking history |
|
Former |
20 (40.8%) |
Never |
29 (59.2%) |
Stage |
|
Stage IV |
33 (67.3%) |
Stage IIIb |
16 (32.5%) |
Prior TKIs |
|
Gefitinib |
31 (63.3%) |
Erlotinib |
14 (28.6%) |
Afatinib |
4 (8.2%) |
Table 2: Plasma levels of EGFR activating mutations and p.T790M
Patient |
Mutation |
Activating mutation (copies/ml) |
p.T790M (copies/ml) |
p.T790M/activating mutation ratio |
Lines of treatment |
PFS to EGFR-TKIs (months) |
|---|---|---|---|---|---|---|
1 |
ex19del |
333,000 |
194,000 |
0.58 |
1 |
16 |
2 |
ex19del |
3,400 |
300 |
0.09 |
1 |
15 |
3 |
ex19del |
1,200 |
190 |
0.16 |
1 |
35 |
4 |
ex19del |
5,900 |
900 |
0.15 |
2 |
25 |
5 |
ex19del |
160 |
80 |
0.5 |
1 |
12 |
6 |
ex19del |
3,200 |
800 |
0.25 |
1 |
13 |
7 |
ex19del |
570 |
500 |
0.87 |
2 |
27 |
8 |
ex19del |
310 |
190 |
0.61 |
2 |
20 |
9 |
ex19del |
1,500 |
270 |
0.18 |
1 |
12 |
10 |
ex19del |
13,000 |
700 |
0.05 |
1 |
17 |
11 |
ex19del |
420 |
80 |
0.19 |
2 |
23 |
12 |
ex19del |
6,400 |
410 |
0.06 |
1 |
19 |
13 |
ex19del |
410 |
80 |
0.19 |
2 |
11 |
14 |
ex19del |
2,500 |
1,000 |
0.4 |
1 |
32 |
15 |
ex19del |
700 |
180 |
0.26 |
3 |
57 |
16 |
ex19del |
3,900 |
700 |
0.18 |
1 |
11 |
17 |
ex19del |
32,600 |
14,500 |
0.44 |
2 |
19 |
18 |
ex19del |
150 |
90 |
0.6 |
3 |
41 |
19 |
ex19del |
6,900 |
2,100 |
0.3 |
1 |
23 |
20 |
ex19del |
275,000 |
47,200 |
0.17 |
1 |
28 |
21 |
ex19del |
54,000 |
4,000 |
0.07 |
1 |
7 |
22 |
ex19del |
82,000 |
56,400 |
0.69 |
2 |
22 |
23 |
ex19del |
32,100 |
5,800 |
0.18 |
1 |
13 |
24 |
ex19del |
5,700 |
1,100 |
0.19 |
2 |
48 |
25 |
ex19del |
130 |
110 |
0.85 |
1 |
17 |
26 |
ex19del |
1,000 |
280 |
0.28 |
2 |
32 |
27 |
ex19del |
44,200 |
11,000 |
0.25 |
1 |
30 |
28 |
ex19del |
250 |
160 |
0.64 |
1 |
19 |
29 |
ex19del |
270 |
140 |
0.52 |
2 |
28 |
30 |
ex19del |
800 |
480 |
0.6 |
2 |
36 |
31 |
ex19del |
3,390,000 |
5,200 |
0.0015 |
1 |
11 |
32 |
ex19del |
3,700 |
90 |
0.02 |
1 |
13 |
33 |
ex19del |
8,200 |
2,100 |
0.26 |
1 |
36 |
34 |
ex19del |
44,400 |
110 |
0.002 |
1 |
10 |
35 |
ex19del |
1,200 |
210 |
0.18 |
1 |
21 |
36 |
ex19del |
3,200 |
1,100 |
0.34 |
1 |
18 |
37 |
ex19del |
3,300 |
1,500 |
0.45 |
1 |
15 |
38 |
ex19del |
1,400 |
300 |
0.21 |
1 |
6 |
39 |
ex19del |
8,300 |
2,700 |
0.32 |
1 |
13 |
40 |
ex19del |
62,000 |
11,000 |
0.17 |
2 |
10 |
41 |
ex19del |
17,300 |
11,100 |
0.64 |
1 |
14 |
42 |
ex19del |
102,000 |
43,900 |
0.43 |
1 |
12 |
43 |
L858R |
4,120 |
620 |
0.15 |
2 |
17 |
44 |
L858R |
420 |
170 |
0.4 |
1 |
10 |
45 |
L858R |
400 |
90 |
0.23 |
2 |
10 |
46 |
L858R |
4,900 |
4,400 |
0.9 |
1 |
11 |
47 |
L858R |
170 |
80 |
0.48 |
1 |
18 |
48 |
L858R |
671,000 |
290 |
0.0004 |
1 |
13 |
49 |
L858R |
340 |
270 |
0.8 |
2 |
20 |
Median (range) |
3,400 (130-3,390,000) |
500 (80-194,000) |
0.26 (0.0004-0.9) |
17 (6-57) |
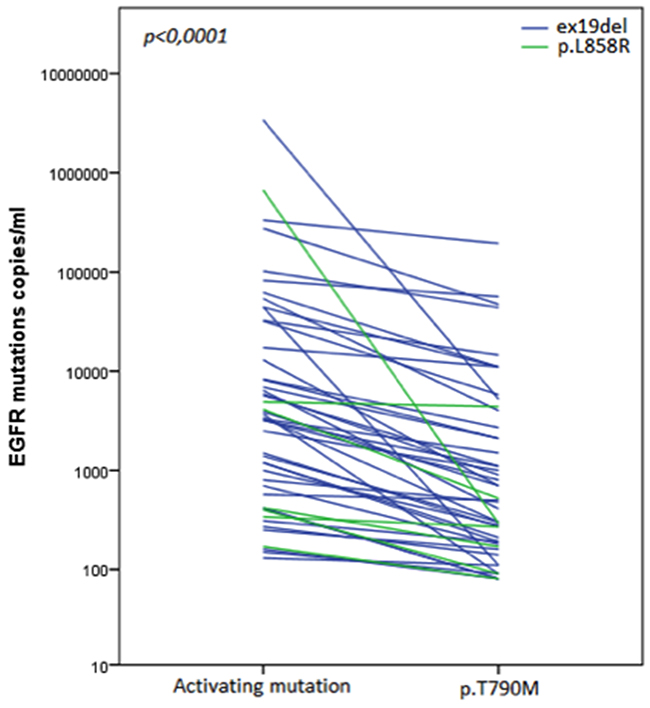
Figure 1: Plasma levels of EGFR activating mutations (ex19del and p. L858R) and p.T790M at the moment of disease progression to first/second-generation EGFR-TKI. The analysis showed the very low ratio of p.T790M vs. activating mutations in patients at the time of progression to EGFR-TKI.
The statistical analysis of PFS vs. the ratio of p.T790M/EGFR activating mutations showed no significant difference (p = 0.075; Spearman’s rank correlation coefficient = 0.256) suggesting that previous EGFR-TKI does not influence the ratio p.T790M/EGFR and that a high ratio is not required to obtain resistance to treatment (Figure 2). No differences in mutation amounts were seen between fast and slow progressing patients comparing months of PFS.
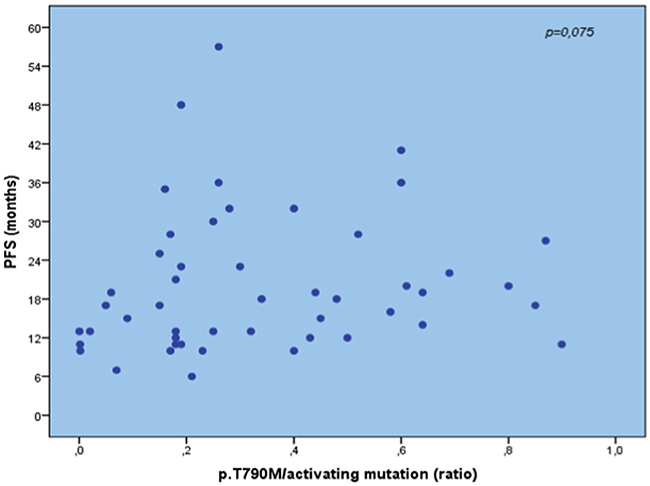
Figure 2: Absence of correlation between p. T790M/activating mutation ratio and PFS to first/second-generation EGFR-TKI, highlighting that a high ratio is not required to obtain resistance to treatment, but also very low amounts drive the resistance.
The amount of ex19del, p.L858R and p.T790M EGFR mutant copies in plasma was monitored during treatment with osimertinib in 5 patients (2 with complete response to osimertinib, 2 with partial response and 1 with stable disease at first-tumor evaluation after 12 weeks) in order to gather information about the dynamics of EGFR mutational pattern as a function of therapy and time. The amount of EGFR mutant clones in plasma decreased in parallel (ex19del and p.T790M or p.L858R and p.T790M), although the activity of osimertinib is higher on p.T790M than ex19del and p.L858R and it would be, therefore, expected a steeper decline of p.T790M than ex19del and p.L858R. In general, the reduction of both ex19del and p.T790M was more marked than in the single patient bearing both p.T790M and p.L858R, which persisted in plasma (Figure 3). The decrease of mutated alleles in plasma was maintained during response to treatment.
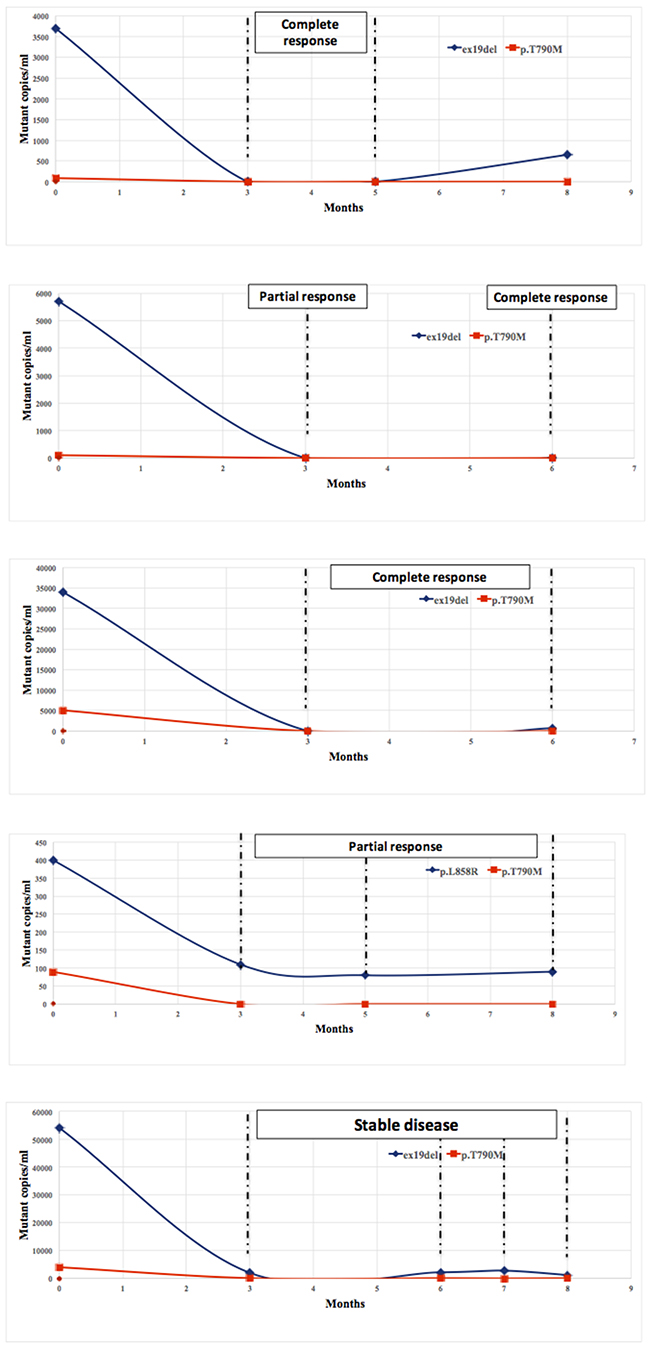
Figure 3: Decreasing of mutated EGFR alleles (ex19del, p. L858R, p.T790M) in plasma during treatment with osimertinib. The amount of EGFR mutant clones in plasma rapidly decreased in parallel and was maintained during treatment, accordingly to the tumor response.
DISCUSSION
The EGFR gatekeeper mutation p.T790M confers resistance to first- and second- generation EGFR-TKIs and prolongs the biologic condition of tumor addiction to EGFR transduction pathway in these tumors. Therefore, the development of new drugs with improved targeting capability of EGFR activating mutations and with extended spectrum to p.T790M is an active area of research [23, 24]. Osimertinib is the first drug approved for the treatment of p.T790M-positive NSCLC after failure of first/second generation EGFR TKIs [21, 22].
Detection of mutant alleles can be performed in tissue as well as plasma, and ddPCR is a sensitive technology suitable for cftDNA detection and analysis [25]. Indeed, our previous work demonstrated that ddPCR detection of p.T790M in plasma reached a meaningful sensitivity of 81.8% and a specificity of 85.7%; in addition, the overall concordance between plasma and tissue analysis was good and corresponded to 83.3% [8].
Although the detection of p.T790M in EGFR-mutant NSCLC patients can be performed on both plasma and tumor tissue, the evolution of diagnostic approaches towards minimally-invasive procedures such as cftDNA is desirable for a number of reasons, including a) the invasive character of a tissue biopsy, b) unreachable tumor sites or insufficient tissue obtained after biopsy [26] and c) limitation of tissue biopsy in capturing tumor heterogeneity due to the small amount of tissue collected and number of tumor sites sampled. Indeed, ESMO clinical practice guidelines for diagnosis, treatment and follow-up of patients with NSCLC reports that if the p.T790M in peripheral blood is observed, treatment with third-generation EGFR TKIs is justified, while it recommends rebiopsy if cftDNA is negative for p.T790M [27]. Timely analysis of biomarkers to guide treatment decision is crucial and any delay in obtaining molecular testing results can postpone treatment decisions and reduce effectiveness of therapy for patients with advanced NSCLC [28].
p.T790M can be detected in plasma at a median time of 2.2 months prior to disease progression and is a predictive factor of resistance and disease outcome [29]. The role of p.T790M may change, depending on disease stage/phase. In particular, patients positive for p.T790M before treatment with EGFR-TKI show significantly inferior PFS (8.9 vs. 12.1 months) and overall survival (19.3 vs. 31.9 months) compared with those without p.T790M [30]. On contrary, at the time of disease progression after first- and second- generation EGFR-TKI, the presence of EGFR p.T790M is a favourable prognostic marker independently from the treatment with osimertinib [11]. Patients with p.T790M had a significantly longer post-progression survival, while subjects without p.T790M more often progressed in a previously uninvolved organ system and exhibited a poorer performance status at time of progression [11].
p.T790M shows a complex biological behaviour, since p.T790M status in patients may change both temporally and spatially among tumor sites at least in part due to the selective pressure from EGFR-TKI [31]. Interestingly, p.T790M status of NSCLC varies after EGFR-TKI discontinuation and may change from positive to negative, thus justifying a potential re-challenge with EGFR-TKI [31]. It is not surprising that after further EGFR-TKI administration, p.T790M status may change from negative to positive again [31]. Previous work provided initial evidence of the different amounts of p.T790M vs. activating mutations in plasma samples in 9 patients [32]. In addition, a recent report suggested a possible clinical importance for detection of p.T790M at low levels in plasma samples [18]. Longitudinal cftDNA analysis revealed an increase in plasma EGFR-activating mutation, and p.T790M announced rociletinib resistance in some patients, whereas in others the activating mutation increased but p.T790M remained suppressed [20]. These findings demonstrate the role of tumor heterogeneity when drugs targeting a single resistance mechanism are given to patients [20]. Based on the results of the present study, it would be interesting to evaluate the effectiveness of combination regimens that also target p.T790–wild-type clones. The present work shows that patients progressing to first/second-generation EGFR-TKIs displayed a low amount of p.T790M compared to the EGFR activating mutations.
On the basis of the cftDNA data of the present work it can be speculated that either EGFR amplification could be a frequent finding in these patients, or that p.T790M is present in a minority of cells of the tumor cell population after TKI failure or both. Therefore, the intriguing questions are: 1) how frequent is EGFR amplification in these patients, 2) how p.T790M can drive tumor progression being so low within the tumor; 3) is there another mechanism of resistance in addition to p.T790M, and 4) is p.T790M able to synergize with activating EGFR mutation.
Our data indicate that, even in presence of low amount of p.T790M, tumors respond to the third-generation TKI osimertinib, which is mainly active against p.T790M. The drug shows higher activity against the EGFR double mutants ex19del/p.T790M and p.L858R/p.T790M with respect to the EGFR bearing only the activating mutation as demonstrated by in vitro experiments. In particular, mean IC50 values are 17 and 4 nM for the ex19del and the p.L858R, respectively and 13 and 5 nM for the double mutants ex19del+p.T790M and p.L858R+p.T790M, respectively [6]. These data are confirmed also in patients of the present study because by monitoring the amount of residual EGFR mutations during osimertinib treatment, it is clear that cell clones carrying the double mutant ex19del/p.T790M or p.L858R/p.T790M and clones with the ex19del alone drastically decrease in their amount, while cell clones carrying the p.L858R alone remain detectable because of the low potency of osimertinib against this mutation.
In patients monitored during osimertinib treatment, p.T790M reduction was marked and similar to the decline of plasma levels of EGFR activating mutations. The differences in concentration between them and the p.T790M may confirm that the tumor is heterogeneous, and it is composed by 1) wild-type clones, 2) cells carrying both the EGFR activating and p.T790M mutations, 3) clones with only the original EGFR activating mutation. A distinct population of cells with the p.T790M only is unlikely to be present.
In conclusion, the present work demonstrates the feasibility of detecting very low amounts of p.T790M in plasma by a sensitive analytical approach and that this mutation is associated with tumor progression – and response to osimertinib – even though it may be a minority with respect to ex19del and p.L858R activating mutations. Further studies are warranted to gain additional knowledge on the interaction between EGFR mutations in TKI-resistant NSCLC, and to determine the clinical consequences to be connected to cftDNA outcomes in plasma. However, nowadays, is often required a treatment adaptation based on pharmacogenetic data [33, 34], and these results provide strong evidence supporting the usefulness of cftDNA as a good predictive biomarker.
MATERIALS AND METHODS
The present study included 49 NSCLC patients carrying the p.T790M mutation in cftDNA. These patients were consecutively enrolled between June and October 2016 from a population of subjects with the following characteristics: 1) stage IIIb/IV disease carrying EGFR activating mutations (exon 19 deletions [ex19del] and/or exon 21 p.L858R) in tumor tissue at the time of initial diagnosis; 2) treatment with first- or second-generation EGFR-TKIs (gefitinib/erlotinib/afatinib), as per approved indication and 3) clinical and imaging evidence of disease progression, as per standard practice.
In order to detect EGFR ex19del, p.L858R and p.T790M mutations, one blood sample was taken in each patient at the time of disease progression; in 5 subjects (enrolled in ASTRIS Trial [35]), 2 to 4 additional blood drawings were obtained to monitor EGFR ex19del, p.L858R and p.T790M mutations during osimertinib administration.
Blood was sampled as per routine biochemistry testing, collected in EDTA tubes and centrifuged at 4°C for 10 min at 3000 rpm within two hours after drawing. Plasma samples (2 ml) were taken from material to be discarded and stored at −80°C until analysis. Circulating tumor DNA was extracted using the QIAmp Circulating Nucleic Acid Kit (Qiagen®, Valencia, CA, USA) following the manufacturer’s protocol. The ex19del, p.L858R and p.T790M alleles were examined using a digital droplet PCR (QX100™ Droplet Digital™ PCR System, BioRad®, Hercules, CA, USA) as previously described [8]. Droplets with a fluorescence intensity threshold higher than 3,000 were considered positive for the presence of mutations and results were given as copies of allele/ml.
This study was compliant with local ethical practices; in particular, patients received treatments as per approved drug label and molecular tests were performed on residual material from standard diagnostic procedures.
Statistical analysis
Progression-free survival (PFS) was calculated from the first day of first-line EGFR-TKI treatment to radiological evidence of disease progression according to RECIST criteria; deaths for other causes than disease progression were considered events [36]. PFS of patients receiving osimertinib was not considered for statistical analysis.
Before inferential statistics, an exploration phase was performed using box plots and scatter plots. We assessed three main quantitative factors: 1) concentration of the activating mutations (ex19del, p.L858R), 2) levels of p.T790M, and 3) their ratio (resistant/activating). In order to verify if the quantitative data were normally distributed we used the Kolmogorov–Smirnov and Shapiro–Wilks tests. A comparison between the three factors and PFS (all patients relapsed to first-line EGFR-TKI) was performed by a nonparametric correlation analysis (Spearman’s rank correlation coefficient), whereas Kruskal–Wallis and Mann–Whitney (two-tailed) tests were used to perform comparisons among the factor associated to the different treatment lines. Finally, the Wilcoxon test (two-tailed) has been performed to compare the number of copies/ml of plasmatic EGFR activating and p.T790M mutations. A p<0.05 was considered as statistically significant; all statistical analyses were performed using the SPSS version 24 software.
Author contributions
M. Del Re had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: R. Danesi, M. Del Re, I. Petrini, M. Tiseo. Acquisition of data: M. Del Re, E. Rofi, G. Restante, P. Bordi, I. Petrini, F. Mazzoni, L. Belluomini, C.M.J. Steendam. Analysis and interpretation of data: M. Del Re, P. Bordi, E. Rofi, G. Restante. Drafting of the manuscript: M. Del Re, R. Danesi Critical revision of the manuscript for important intellectual content: R. Danesi, van Schaik, E. Vasile, F. Di Costanzo, A. Falcone, A. Frassoldati, C.M.J. Steendam, A. Chella, M. Tiseo Statistical analysis: R. Morganti, M. Del Re Obtaining funding: R. Danesi. Administrative, technical, or material support: M. Del Re, R. Danesi Other (specify): None.
CONFLICTS OF INTEREST
Authors have nothing to disclose.
FUNDING
Fondazione Cassa Risparmio di Lucca, Lucca (Italy), Istituto Toscano Tumori, Firenze (Italy), and University of Pisa (Italy) to RD.
REFERENCES
1. Saber A, Hiltermann TJ, Kok K, Terpstra MM, de Lange K, Timens W, Groen HJ, van den Berg A. Mutation patterns in small cell and non-small cell lung cancer patients suggests a different level of heterogeneity between primary and metastatic tumors. Carcinogenesis. 2017; 38:144-151.
2. Kim EY, Cho EN, Park HS, Kim A, Hong JY, Lim S, Youn JP, Hwang SY, Chang YS. Genetic heterogeneity of actionable genes between primary and metastatic tumor in lung adenocarcinoma. BMC Cancer. 2016; 16:27.
3. Kim S, Kim TM, Kim DW, Go H, Keam B, Lee SH, Ku JL, Chung DH, Heo DS. Heterogeneity of genetic changes associated with acquired crizotinib resistance in ALK-rearranged lung cancer. J Thorac Oncol. 2013; 8:415-422.
4. Imamura F, Uchida J, Kukita Y, Kumagai T, Nishino K, Inoue T, Kimura M, Oba S, Kato K. Monitoring of treatment responses and clonal evolution of tumor cells by circulating tumor DNA of heterogeneous mutant EGFR genes in lung cancer. Lung Cancer. 2016; 94:68-73.
5. Suda K, Murakami I, Sakai K, Tomizawa K, Mizuuchi H, Sato K, Nishio K, Mitsudomi T. Heterogeneity in resistance mechanisms causes shorter duration of epidermal growth factor receptor kinase inhibitor treatment in lung cancer. Lung Cancer. 2016; 91:36-40.
6. Hirano T, Yasuda H, Tani T, Hamamoto J, Oashi A, Ishioka K, Arai D, Nukaga S, Miyawaki M, Kawada I, Naoki K, Costa DB, Kobayashi SS, et al. In vitro modeling to determine mutation specificity of EGFR tyrosine kinase inhibitors against clinically relevant EGFR mutants in non-small-cell lung cancer. Oncotarget. 2015; 6:38789-38803. https://doi.org/10.18632/oncotarget.5887.
7. Yun CH, Mengwasser KE, Toms AV, Woo MS, Greulich H, Wong KK, Meyerson M, Eck MJ. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc Natl Acad Sci U S A. 2008; 105:2070-2075.
8. Del Re M, Tiseo M, Bordi P, D’Incecco A, Camerini A, Petrini I, Lucchesi M, Inno A, Spada D, Vasile E, Citi V, Malpeli G, Testa E, et al. Contribution of KRAS mutations and c.2369C > T (p.T790M) EGFR to acquired resistance to EGFR-TKIs in EGFR mutant NSCLC: a study on circulating tumor DNA. Oncotarget. 2017; 8:13611-13619. https://doi.org/10.18632/oncotarget.6957.
9. Suda K, Onozato R, Yatabe Y, Mitsudomi T. EGFR T790M mutation: a double role in lung cancer cell survival? J Thorac Oncol. 2009; 4:1-4.
10. Arcila ME, Oxnard GR, Nafa K, Riely GJ, Solomon SB, Zakowski MF, Kris MG, Pao W, Miller VA, Ladanyi M. Rebiopsy of lung cancer patients with acquired resistance to EGFR inhibitors and enhanced detection of the T790M mutation using a locked nucleic acid-based assay. Clin Cancer Res. 2011; 17:1169-1180.
11. Oxnard GR, Arcila ME, Sima CS, Riely GJ, Chmielecki J, Kris MG, Pao W, Ladanyi M, Miller VA. Acquired resistance to EGFR tyrosine kinase inhibitors in EGFR-mutant lung cancer: distinct natural history of patients with tumors harboring the T790M mutation. Clin Cancer Res. 2011; 17:1616-1622.
12. Pao W, Miller VA, Politi KA, Riely GJ, Somwar R, Zakowski MF, Kris MG, Varmus H. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2005; 2:e73.
13. Jiang SX, Yamashita K, Yamamoto M, Piao CJ, Umezawa A, Saegusa M, Yoshida T, Katagiri M, Masuda N, Hayakawa K, Okayasu I. EGFR genetic heterogeneity of nonsmall cell lung cancers contributing to acquired gefitinib resistance. Int J Cancer. 2008; 123:2480-2486.
14. Chen LY, Molina-Vila MA, Ruan SY, Su KY, Liao WY, Yu KL, Ho CC, Shih JY, Yu CJ, Yang JC, Rosell R, Yang PC. Coexistence of EGFR T790M mutation and common activating mutations in pretreatment non-small cell lung cancer: a systematic review and meta-analysis. Lung Cancer. 2016; 94:46-53.
15. Lee Y, Lee GK, Lee YS, Zhang W, Hwang JA, Nam BH, Kim SH, Kim JH, Yun T, Han JY, Kim HT, Lee JS. Clinical outcome according to the level of preexisting epidermal growth factor receptor T790M mutation in patients with lung cancer harboring sensitive epidermal growth factor receptor mutations. Cancer. 2014; 120:2090-2098.
16. Inukai M, Toyooka S, Ito S, Asano H, Ichihara S, Soh J, Suehisa H, Ouchida M, Aoe K, Aoe M, Kiura K, Shimizu N, Date H. Presence of epidermal growth factor receptor gene T790M mutation as a minor clone in non-small cell lung cancer. Cancer Res. 2006; 66:7854-7858.
17. Chabon JJ, Simmons AD, Lovejoy AF, Esfahani MS, Newman AM, Haringsma HJ, Kurtz DM, Stehr H, Scherer F, Karlovich CA, Harding TC, Durkin KA, Otterson GA, et al. Circulating tumour DNA profiling reveals heterogeneity of EGFR inhibitor resistance mechanisms in lung cancer patients. Nat Commun. 2016; 7:11815.
18. Remon J, Caramella C, Jovelet C, Lacroix L, Lawson A, Smalley S, Howarth K, Gale D, Green E, Plagnol V, Rosenfeld N, Planchard D, Bluthgen MV, et al. Osimertinib benefit in EGFR-mutant NSCLC patients with T790M-mutation detected by circulating tumour DNA. Ann Oncol. 2017; 28:784-790.
19. Oxnard GR, Thress KS, Alden RS, Lawrance R, Paweletz CP, Cantarini M, Yang JC, Barrett JC, Janne PA. Association between plasma genotyping and outcomes of treatment with osimertinib (AZD9291) in advanced non-small-cell lung cancer. J Clin Oncol. 2016; 34:3375-3382.
20. Piotrowska Z, Niederst MJ, Karlovich CA, Wakelee HA, Neal JW, Mino-Kenudson M, Fulton L, Hata AN, Lockerman EL, Kalsy A, Digumarthy S, Muzikansky A, Raponi M, et al. Heterogeneity underlies the emergence of EGFRT790 wild-type clones following treatment of T790M-positive cancers with a third-generation EGFR inhibitor. Cancer Discov. 2015; 5:713-722.
21. Cross DA, Ashton SE, Ghiorghiu S, Eberlein C, Nebhan CA, Spitzler PJ, Orme JP, Finlay MR, Ward RA, Mellor MJ, Hughes G, Rahi A, Jacobs VN, et al. AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov. 2014; 4:1046-1061.
22. Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, Shepherd FA, He Y, Akamatsu H, Theelen WS, Lee CK, Sebastian M, Templeton A, et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. 2017; 376:629-640.
23. Liao BC, Lin CC, Lee JH, Yang JC. Update on recent preclinical and clinical studies of T790M mutant-specific irreversible epidermal growth factor receptor tyrosine kinase inhibitors. J Biomed Sci. 2016; 23:86.
24. Rho JK, Lee IY, Choi JY, Choi CM, Hur JY, Koh JS, Lee J, Suh BC, Song HJ, Salgaonkar P, Lee J, Lee J, Jung DS, et al. Superior efficacy and selectivity of novel small molecule kinase inhibitors of T790M mutant EGFR in preclinical models of lung cancer. Cancer Res. 2017; 77:1200-1211.
25. Wang W, Song Z, Zhang Y. A comparison of ddPCR and ARMS for detecting EGFR T790M status in ctDNA from advanced NSCLC patients with acquired EGFR-TKI resistance. Cancer Med. 2017; 6:154-162.
26. Lim C, Sung M, Shepherd FA, Nouriany N, Sawczak M, Paul T, Perera-Low N, Foster A, Zawisza D, Feld R, Liu G, Leighl NB. Patients with advanced non-small cell lung cancer: are research biopsies a barrier to participation in clinical trials? J Thorac Oncol. 2016; 11:79-84.
27. Novello S, Barlesi F, Califano R, Cufer T, Ekman S, Levra MG, Kerr K, Popat S, Reck M, Senan S, Simo GV, Vansteenkiste J, Peters S; ESMO Guidelines Committee. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016; 27:v1-v27.
28. Lim C, Tsao MS, Le LW, Shepherd FA, Feld R, Burkes RL, Liu G, Kamel-Reid S, Hwang D, Tanguay J, da Cunha Santos G, Leighl NB. Biomarker testing and time to treatment decision in patients with advanced nonsmall-cell lung cancer. Ann Oncol. 2015; 26:1415-1421.
29. Zheng D, Ye X, Zhang MZ, Sun Y, Wang JY, Ni J, Zhang HP, Zhang L, Luo J, Zhang J, Tang L, Su B, Chen G, et al. Plasma EGFR T790M ctDNA status is associated with clinical outcome in advanced NSCLC patients with acquired EGFR-TKI resistance. Sci Rep. 2016; 6:20913.
30. Wang Z, Chen R, Wang S, Zhong J, Wu M, Zhao J, Duan J, Zhuo M, An T, Wang Y, Bai H, Wang J. Quantification and dynamic monitoring of EGFR T790M in plasma cell-free DNA by digital PCR for prognosis of EGFR-TKI treatment in advanced NSCLC. PLoS One. 2014; 9:e110780.
31. Hata A, Katakami N, Yoshioka H, Kaji R, Masago K, Fujita S, Imai Y, Nishiyama A, Ishida T, Nishimura Y, Yatabe Y. Spatiotemporal T790M heterogeneity in individual patients with EGFR-mutant non-small-cell lung cancer after acquired resistance to EGFR-TKI. J Thorac Oncol. 2015; 10:1553-1559.
32. Sorensen BS, Wu L, Wei W, Tsai J, Weber B, Nexo E, Meldgaard P. Monitoring of epidermal growth factor receptor tyrosine kinase inhibitor-sensitizing and resistance mutations in the plasma DNA of patients with advanced non-small cell lung cancer during treatment with erlotinib. Cancer. 2014; 120:3896-3901.
33. Grech G, Zhan X, Yoo BC, Bubnov R, Hagan S, Danesi R, Vittadini G, Desiderio DM. EPMA position paper in cancer: current overview and future perspectives. EPMA J. 2015; 6:9.
34. Golubnitschaja O, Baban B, Boniolo G, Wang W, Bubnov R, Kapalla M, Krapfenbauer K, Mozaffari MS, Costigliola V. Medicine in the early twenty-first century: paradigm and anticipation - EPMA position paper 2016. EPMA J. 2016; 7:23.
35. De Marinis F Chul CB, Kim DW, Kim SW, Hochmair MJ, Metro G, Vansteenkiste JF, Vicente D, Solomon BJ, Cheema PK, Freitas HC, Provencio M, Chen YM, et al. ASTRIS: A real world treatment study of osimertinib in patients (pts) with EGFR T790M positive non-small cell lung cancer (NSCLC). J Clin Oncol. 2017; 35:9036.
36. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009; 45:228-247.

