INTRODUCTION
Prostate cancer (PC) is the most frequently diagnosed non-cutaneous malignancy in males in the United States [1]. Androgen hormone/androgen receptor (AR) signaling are central to PC development, progression, and metastasis [2–6]. Therefore, androgen deprivation therapy (ADT) is the first line therapy for advanced PC. However, almost all patients who received ADT develop metastatic castration-resistant prostate cancer (CRPC) [7–13]. In addition, ADT has limited effects on long-term survival and side effects, making it undesirable for PC patients [14]. Recurrent cases are usually treated by cytotoxic chemotherapeutic agents, which have narrow therapeutic indices due to their high cytotoxic effects [13, 15–18]. Unfortunately, patients with metastatic CRPC do not respond well to cytotoxic agents and show high mortality rate. Therefore, finding a suitable target and a therapeutic agent with minimal or insignificant side effects, while also being effective on the recurrent cancer, would have major impact on the management of patients with CRPC.
Signal transducer and activator of transcription 3 (STAT3) and highly homologous isoforms STAT5A and STAT5B (STAT5A/B) are key components of the Janus tyrosine kinase (JAK)/STAT pathway [19]. Both STAT3 and STAT5 act as signaling proteins in the cytoplasm and transcription factors when they localize to the nucleus [19]. The STAT3 and STAT5 are activated by phosphorylation at a specific tyrosine (Y) residue such as phospho-Y705 in STAT3 (pY705-STAT3) and phospho-Y694 in STAT5A (pY694-STAT5A), followed by translocation into the nucleus where they bind cis-DNA and activate target genes to exert their biology [20–28]. Several growth factors such as epidermal growth factor receptor (EGFR), hepatocyte growth factor receptor (HGFR), and platelet-derived growth factor receptor (PDGFR) and non-receptor cytoplasmic tyrosine kinases such as Abelson leukemia protein and Src-related kinases have demonstrated to activate STAT3 [20–28]. Evidence suggest that STAT3 and STAT5 play critical roles in cancer progression, thus making themselves as therapeutic targets [29]. Activation of STAT3 (pY705-STAT3) and STAT5 (pY694-STAT5A) has been implicated in treatment-naïve as well as advanced PC [30–38]. Therefore, targeting of STAT3 and STAT5 in PC could in practice be of therapeutic significance. Expression of STAT3 and STAT5 has been demonstrated in PC cell lines [32, 35, 37] and PC tissues [30, 31, 33, 34, 36, 38]; however, the expression of STAT3 and STAT5A in metastatic CRPC has been rarely studied [31, 33, 38].
In this study, we evaluated the expression and possible role of STAT3/5A in CRPC. We observed a strong nuclear immunoreactivity for activated STAT3 (pY7705) and STAT5A (pY694) in CRPC cases compared with BPH. In addition, we found that up to 20% of CRPC clinical cases showed a frequent amplification of the STAT3 and STAT5A genes. Moreover, we showed that PC cells C4-2 and PC3 expressed low levels of STAT3 and STAT5A mRNA and were relatively resistant to pimozide [39] in comparison with 22Rv1 cells that express high levels of STAT3 and STAT5. These findings suggest that STAT3 and STAT5A may serve as a predictive biomarker for evaluating the responsiveness of therapies targeting the JAK/STAT pathway.
RESULTS
Demographics, clinical, and histopathologic parameters
CRPC cases used in the current study were diagnosed on transurethral resection of prostate for obstructive uropathy. All patients with CRPC were treated with ADT and developed CRPC. Ages ranged from 59 years to 95 years (median = 81 years and mean = 80.6 years) in the CRPC cases and 57 years to 86 years (median = 68 years and mean = 71 years) in the BPH category. Prostate tumor tissues analyzed were from patients who were treated ADT and developed resistance to it. The staining was performed on the primary tumors obtained by transurethral resection in all cases. Radical prostatectomy was not performed in any of patients with CRPC. 46% of cases (n=7) had metastatic disease. Additional therapies included chemotherapy (3 cases) and radiation therapy (2 cases). 46% (n=7) of cases displayed Gleason patterns 4 (primary) and 5 (secondary), 27% (n=4) of cases showed Gleason patterns 5 (primary) and 4 (secondary), and 27% (n=4) of cases exhibited Gleason patterns 5 (primary) and 5 (secondary). Evidence suggests that major proportions of prostate tumors exhibit characteristics of mixed neuroendocrine (NE) carcinoma-acinar adenocarcinoma, small cell carcinoma, and large cell carcinoma with history of hormonal therapy [40]. Therefore, NE markers performed in these cases were retrieved from the departmental archive and were studied. None of the cases analyzed in this study exhibited NE phenotype (small cell or large cell neuroendocrine carcinoma) and stained for NE markers (synaptophysin, chromogranin, and CD56). As reported previously, all tumors utilized in this study showed multifocal to diffuse and strong nuclear immunoreactivity for androgen receptor and NKX3.1, a homeobox-containing transcription factor that functions as a negative regulator of epithelial cell growth in the prostate [41, 42]. Its aberrant expression is associated with prostate tumor progression. A weak and sporadic expression of prostate specific antigen (PSA) was observed in only three (20%) of the tumors. Four of the metastatic cases were Gleason patterns 4 and 5 (one with 5 and 4, and two with 5 and 5). Three cases with Gleason patterns 4 and 5, and one case each with patterns 4 and 5, and 5 and 5 were treated with radiation therapy. Four cases with Gleason patterns 4 and 5 and one case with Gleason patterns 5 and 4, and 5 and 5 were treated with chemotherapy. The cases that were treated with both chemotherapy and radiation therapy were of Gleason patterns 4 and 5 (Table 1).
Table 1: Clinicopathological characteristics and immunoexpression for STAT3 and STAT5A in cohort of CRPC cases
Case |
Age (years) |
Specimen Type |
Gleason Score (Primary+ Secondary pattern) |
Prognostic Group Grade |
ADT |
Radical Prostate-ctomy |
CRPC |
Metastasis |
Radiation Therapy |
Chemo-therapy |
STAT3 Expression |
STAT5A Expression |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
1 |
59 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
3+ and strong |
3+ and strong |
2 |
69 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
No |
No |
Yes |
3+ and strong |
3+ and strong |
3 |
69 |
TURP |
9(5+4) |
5 |
Yes |
No |
Yes |
No |
No |
No |
2+ and strong |
2+ and strong |
4 |
70 |
TURP |
10(5+5) |
5 |
Yes |
No |
Yes |
No |
No |
No |
2+ and strong |
2+ and strong |
5 |
76 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
3+ and strong |
3+ and strong |
6 |
80 |
TURP |
9(5+4) |
5 |
Yes |
No |
Yes |
No |
No |
No |
2+ and strong |
2+ and strong |
7 |
81 |
TURP |
9(5+4) |
5 |
Yes |
No |
Yes |
No |
No |
Yes |
2+ and strong |
Negative |
8 |
81 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
Yes |
No |
No |
3+ and strong |
3+ and strong |
9 |
86 |
TURP |
10(5+5) |
5 |
Yes |
No |
Yes |
No |
No |
No |
Negative |
Negative |
10 |
86 |
TURP |
10(5+5) |
5 |
Yes |
No |
Yes |
Yes |
Yes |
No |
3+ and strong |
3+ and strong |
11 |
86 |
TURP |
9(5+4) |
5 |
Yes |
No |
Yes |
Yes |
Yes |
No |
2+ and strong |
3+ and strong |
12 |
88 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
No |
No |
No |
2+ and strong |
Negative |
13 |
91 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
No |
No |
No |
2+ and strong |
2+ and strong |
14 |
92 |
TURP |
9(4+5) |
5 |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
2+ and strong |
3+ and strong |
15 |
95 |
TURP |
10(5+5) |
5 |
Yes |
No |
Yes |
Yes |
No |
Yes |
3+ and strong |
3+ and strong |
Abbreviations: TURP: transurethral resection of prostate gland. ADT: Androgen deprivation therapy; CRPC: Castration-resistant prostate cancer
CRPC tissues exhibit strong nuclear immunoreactivity for STAT3 and STAT5A
To gain insight into the role of STAT3 and STAT5A in advanced PC, we evaluated the patterns of STAT3 and STAT5A protein expression in a cohort of CRPC (n=15) and BPH (n=15) cases by IHC (Figure 1, 2, and 3). As outlined in Table 2, pSTAT3 (Y705) and pSTAT5A (Y694) antibodies were used in IHC to assess the levels of activated, nuclear STAT3 and STAT5A proteins. Strong nuclear immunoreactivity for STAT3 and STAT5A were observed in 93% (n=14) and 80% (n=12) of CRPC cases, respectively, whereas only one (7%) BPH case showed strong STAT3 staining (Figure 1A, 1B, respectively). Focal and weak positivity for STAT3 and STAT5A were noted in 47% (n=7) and 67% (n=10) of BPH cases. None of the BPH cases showed strong STAT5A expression. Of 14 cases, which showed strong immunoreactivity for STAT3 in CRPC cases, diffuse staining pattern was observed in 6 cases and multifocal staining was observed in 8 cases (Figure 1A; Figure 2B, 2C). Diffuse and strong STAT5A expression was evident in 8 and 4 CRPC cases, respectively (Figure 1B; Figure 3B, 3C). None of CRPC cases showed weak staining. In contrast, 7 of 10 cases of BPH showed focal and weak staining for STAT3 (Figure 1A). Only one BPH case showed strong and diffuse staining for STAT3 (Figure 1A) and none showed strong STAT5A expression (Figure 1B). All metastatic cases were STAT3 (multifocal and strong, n=2 and diffuse and strong, n=5) and STAT5A positive (diffuse and strong) (Figure 2; Figure 3, respectively). The cases that were treated with both chemotherapy and radiation therapy exhibited diffuse and strong immunoreactivity for STAT5A in 3 cases and diffuse and strong and multifocal and strong immunoreactivity for STAT3 in 2 and 1 cases, respectively. All the cases treated with chemotherapy alone depicted diffuse and strong reaction for STAT3 (n=3) and diffuse or multifocal and strong (n=2) and no (n=1) reaction for STAT5A, respectively. One of the cancer cases treated with only radiation therapy showed diffuse and multifocal positivity for STAT3 and 2 cases that were treated with radiation therapy were diffuse and strongly positive for STAT5A (Table 1 to 5). The cases with weak and patchy or focal positivity for PSA exhibited diffuse and strong immunoreactivity for STAT3 and STAT5A.
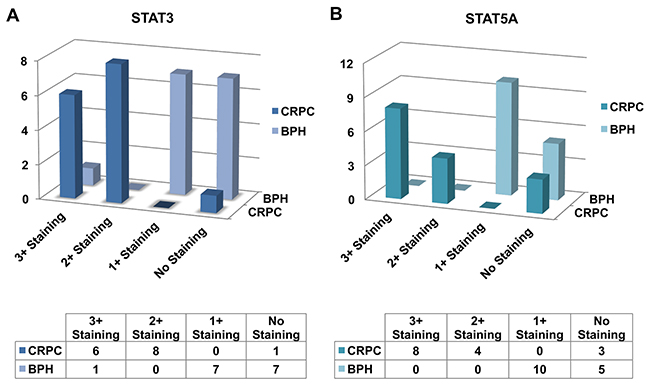
Figure 1: Bar diagram showing the expression patterns of STAT3 (A) and STAT5A (B) in castration-resistant prostate cancer (CRPC) and benign prostatic hyperplasia (BPH) clinical cases as analyzed by IHC.
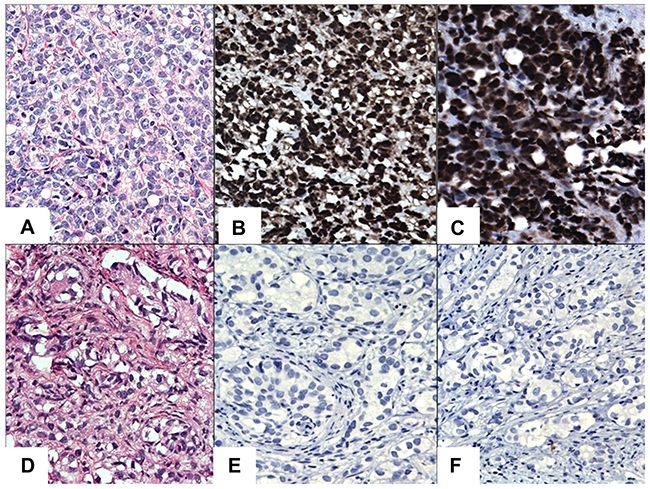
Figure 2: IHC staining of STAT3/5A in CRPC cases. (A) Hematoxylin and Eosin (H&E). (B) Diffuse and strong nuclear immunoreactivity for STAT3. (C) Diffuse and strong nuclear immunoreactivity for STAT5A. (D) A case of H&E staining in CRPC. (E) Negative staining for STAT3 in CRPC. (F) Negative staining for STAT5A in CRPC. Original magnification was x200. Micrographs are representation of multiple images.
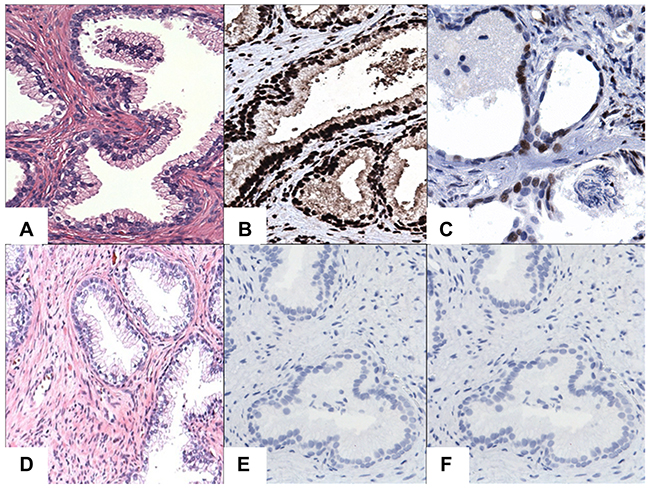
Figure 3: IHC staining of STAT3/5A in BPH cases. (A) Hematoxylin and Eosin (H&E) staining of BPH cases. (B) Diffuse and strong nuclear immunoreactivity for STAT3. (C) Diffuse and moderate to strong nuclear immunoreactivity for STAT5A. (D) A case of H&E staining in BPH. (E) Negative staining for STAT3 in BPH. (F) Negative staining for STAT5A in BPH. Original magnification was x200. Micrographs are representation of multiple images.
Table 2: Antibodies, instrument used, and antibody detection method were used in IHC
Antibodies |
Antibody Clone |
Catalog Number |
Vendor |
Dilution |
Antigen Retrieval Method |
Antibody Incubation Time |
Pretreatment |
Instrument |
Detection System |
|---|---|---|---|---|---|---|---|---|---|
STAT3 Rabbit Monoclonal |
STAT3-Y705(D3A7) |
9145 |
Cell Signaling, Beverly, MA |
1:50 |
High pH |
32 minutes |
CC1 standard-high pH |
Ventana Ultra |
Ultraview Universal DAB Detection Kit |
STAT5A Rabbit Polyclonal |
STAT5A-Y694 |
T3794 |
Epitomics, Burlingame, CA |
1:100 |
High pH |
32 minutes |
CC1 mild- high pH |
Ventana Ultra |
Optiview DAB IHC Detection Kit |
The CC1 (High PH) for the Ventana Ultra - Tris Based Buffer.
The ER1 (Low PH) for the Bond III instrument - Citrate based pH 6.0 solution.
The ER2 (High PH) for the Bond III instrument - EDTA based pH 9.0 solution.
Abbreviations: CC: Cell Conditioner 1; DAB:3,3diaminobenzidine; IHC: Immunohistochemistry.
Table 3: STAT3 and STAT5A expression patterns in CRPC and BPH cases
Categories |
Number of cases with focal positivity for STAT3 |
Number of cases with diffuse (>50% of the lesional cells) positivity for STAT5A |
Number of STAT3 negative cases |
Number of cases with focal positivity for STAT5A |
Number of cases with diffuse (>50% of the lesional cells) positivity for STAT5A |
Number of STAT5A negative cases |
|---|---|---|---|---|---|---|
CRPC (n=15) |
8 (Multifocal and strong) |
6 (Diffuse and strong) |
1 |
4 (Multifocal and strong) |
8 (Diffuse and strong) |
3 |
BPH (n=15) |
7 (Focal and weak) |
1(Diffuse and strong) |
7 |
10 (Focal and weak) |
None |
5 |
Abbreviations: CRPC: Castration-resistant prostate cancer; BPH: Benign prostatic hyperplasia.
Table 4: Overall results of immnuohistochemical expression among the cases (expressed as percentage of cases immunoreactive to the specific antibody) among CRPC and BPH cases
Categories |
STAT3 Positive Cases |
STAT5A Positive Cases |
|---|---|---|
CRPC (n=15) |
14(93.3%) |
12(80%) |
BPH (n=15) |
8(53.3%) |
10(66.6%) |
Abbreviations: CRPC: Castration-resistant prostate cancer; BPH: Benign prostatic hyperplasia.
Table 5: Immunohistochemical staining results for STAT3 and STAT5A represented in a semi-quantitative fashion as estimated percentage of tumor/lesional cells immunoreactive with the antibodies (0: no tumor/lesional cell staining, 1+: 1-25% of immunoreactive tumor/lesional cells, 2+: 26-50% of immunoreactive tumor/lesional cells, 3+: 51-100% of immunoreactive tumor/lesional cells) (expressed as number of cases)
Categories |
STAT3 expression |
STAT5A expression |
|---|---|---|
CRPC (n=15) |
3+ (Diffuse) staining, n=6 |
3+ (Diffuse) staining, n=8 |
BPH (n=15) |
3+ (Diffuse) staining, n=1 |
3+ (Diffuse) staining, n=0 |
Abbreviations: CRPC: Castration-resistant prostate cancer; BPH: Benign prostatic hyperplasia.
Increases in nuclear STAT3 and STAT5A staining in CRPC are statistically significant
CRPC cases showed significantly higher STAT3 nuclear positivity than BPH cases (Chi-square test, p=0.01). No significant differences were observed in focal and weak STAT5A expression between CRPC and BPH groups (Chi-square test, p=0.4). However, multifocal or diffuse and strong STAT3 (Chi-square test, p<0.0001) and STAT5A (Chi-square test, p<0.0001) expression was significantly higher in CRPC group than BPH group.
CRPC cells express the low levels of STAT3 and STAT5A transcripts
To complement the above clinical data, we performed a quantitative PCR analysis of STAT3 and STAT5A transcripts in PC cell lines LNCaP, C4-2, 22Rv1, and PC3. LNCaP is the AR-positive and castration-sensitive PC cell line [43]. C4-2 is the castration-resistant subline of LNCaP [44]. The AR-negative PC3 cell line is a bone metastatic model of human PC [45]. 22Rv1 is the castration-resistant subline of CW22R cells, and it was derived from human PC xenografts via serial circulation in castrated-mice [46]. The results of this analysis revealed that PC cells expressed varying levels of STAT3 and STAT5A transcripts (Figure 4A). LNCaP cells expressed the highest levels of STAT3 mRNA compared with C4-2, 22Rv1, and PC3. The levels of STAT3 mRNA were significantly (p<0.0002) lower in C4-2 and 22Rv1 than LNCaP cells. Although levels of STAT5A were slightly higher in C4-2 than LNCaP cells, the differences were not statistically (p>0.05) significant (Figure 4A). Surprisingly, however, 22Rv1 cells expressed the highest levels of STAT5A transcript relative to those of LNCaP, C4-2 and PC3. Unexpectedly, metastatic PC3 cells expressed the lowest levels of STAT3 and STAT5A transcripts compared with LNCaP, C4-2, and 22Rv1 (Figure 4A). In addition, we analyzed the PC public data set, which can be accessed through the www.cbioportal.org online platform, to see if metastatic CRPC cases exhibit altered patterns of the STAT3 and STAT5A genes. The results of our analysis showed that the STAT3 and STAT5A genes were amplified in up to 20% of metastatic CRPC clinical cases with the NE phenotype (Figure 4B). Here, we want to clarify that NE cells lack AR [40], and evidence suggested that the NE differentiation could be increased after ADT and in CRPC [15, 47].
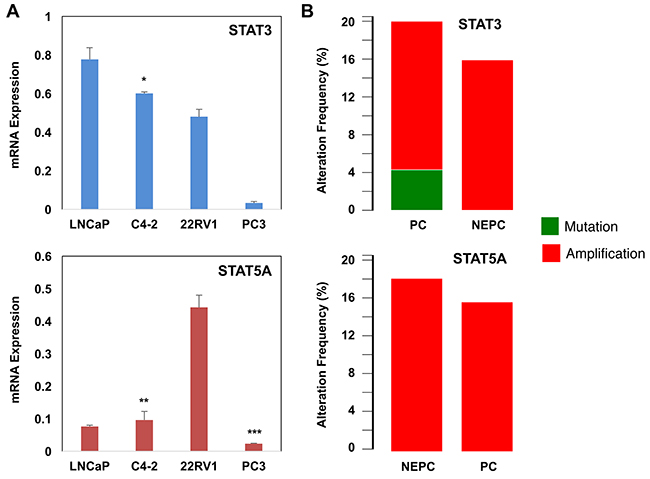
Figure 4: Alterations of STAT3/5A mRNA and genes in PC. (A) Quantitative RT-PCR analysis of STAT3 and STAT5A transcripts in LNCaP, C4-2, 22Rv1, and PC3 cells. *p<0.0002, **p>0.05 ***p<0.002. 24h after cell seeding, total RNA was isolated from cells that were grown in serum-fed conditions. (B) Alterations of the STAT3 and STAT5A genes in metastatic CRPC clinical cases. The data were accessed through the www.cbioportal.org online platform. Data (±SD) are representation of two independent experiments in triplicates.
CRPC cells confer resistance to pimozide
Pimozide was identified as an indirect inhibitor of STAT5 signaling and demonstrated to decrease the survival of chronic myelogenous leukemia cells that were resistant to the kinase inhibitors [39]. In that study, pimozide was demonstrated to inhibit STAT5 tyrosine phosphorylation, which coincided with inactivation of STAT5-dependent transcription [39]. To determine the effects of pimozide on PC cell growth and apoptosis, LNCaP, C4-2, and 22Rv1, and PC3 cells were exposed to varying doses of pimozide in cultures. The results showed that pimozide inhibited the growth of LNCaP, C4-2, 22Rv1, and PC3 cells in a dose-dependent manner (Figure 5A). Relative to mock control, 5 μM of pimozide was sufficient to suppress the growth of 22Rv1 cells significantly (p<0.02), whereas 10 μM of pimozide for LNCaP and 20 μM of pimozide for C4-2 and PC3 cells were required for a significant growth inhibition (p<0.002) under the same experimental conditions. In addition, we demonstrated that pimozide induced apoptosis in LNCaP and C4-2 cells in a dose-dependent manner (Figure 5B). We noted, however, that although 5 μM of pimozide was sufficient to significantly induce apoptosis in LNCaP cells (p<0.002), 20 μM of pimozide was necessary to achieve similar levels of apoptosis in C4-2 cells (p<0.007). Taken together, these observations indicate that CRPC cells C4-2 and PC3 confer resistance to pimozide, likely due to the low levels of STAT3/5A expression.
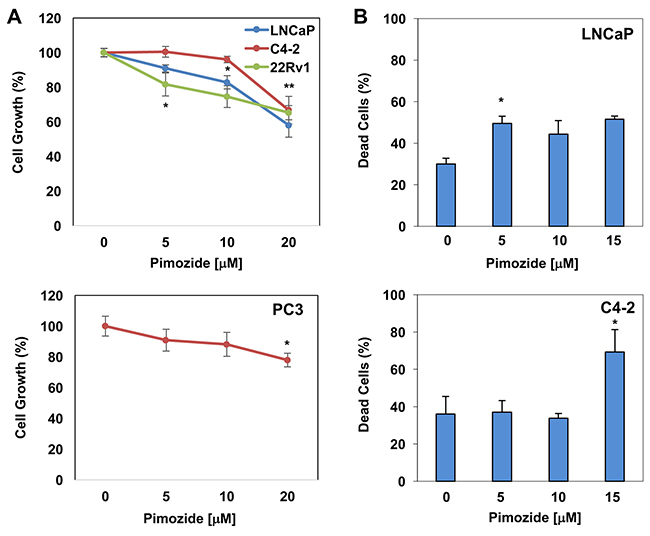
Figure 5: The effects of pimozide on PC cells growth and apoptosis in vitro. (A) The dose-dependent effects of pimozide on LNCaP (*p<0.001), C4-2 (*p>0.06; **p<0.002), 22Rv1 (*p<0.02), and PC3 (*p>0.15; **p<0.002) cell growth. (B) Dose-dependent effects of pimozide on LNCaP (*p<0.01) and C4-2 (*p<0.01) apoptosis. Cell proliferation by CCK-8 and apoptosis by PI staining were analyzed at 48h post pimozide treatment. Data (±SD) are representation of two independent experiments in triplicates.
DISCUSSION
Current therapeutic modalities available for patients with advanced PC include active surveillance, surgery [48], radiation therapy [48, 49], hormonal therapy [14, 18, 50], chemotherapy [18, 51, 52], and immunotherapy [53]. The clinical management of patients with PC depends on age, Gleason grade, prognostic grade group, overall health status of the patient, and the tumor stage. Currently, the mortality rate for recurrent PC is high, likely due to the lack of effective treatment. Therefore, effective pharmacotherapies designed to specifically target this lethal disease are much needed. Nevertheless, a better understanding of the cellular and molecular basis of CRPC is crucial for the identification of novel therapeutics. Evidence indicates the critical role of STAT3 and STAT5 in PC progression and metastasis. For example, activation of STAT3 and STAT5 has been implicated in treatment-naïve and advanced PC [30–38]. Don-Doncow et al. studied the protein and gene expression of STAT3 and IL6R in metastases from patients who died from CRPC [31]. The results of this study showed that 95% of metastatic cases expressed STAT3 and IL6R, as demonstrated by IHC. Interestingly, bone metastases showed significantly higher expression for both proteins compared to lymph node and visceral metastases. Similarly, STAT3 mRNA levels were significantly higher in bones than in lymph node and visceral metastases, whereas there was no significant difference in IL6R mRNA expression. In addition, using immunoblotting, immunofluorescence staining, and RT-PCR analysis, a study by Cocchiola et al. demonstrated that compared with the matched normal sections, prostate tumors with low Gleason scores (i.e. 6-7) exhibited a nuclear expression of pY705-STAT3 protein, whereas prostate tumors with high Gleason cases (i.e. 8–9) showed a cytoplasmic presence of pY705-STAT3 [30]. Likewise, evaluation of STAT3 protein in PC tissues by Western blotting showed a constitutively activated STAT3 (pY705) in all tumor grades compared to the matched normal sections.
Moreover, Singh et al. assessed on the expression and localization of STAT proteins by IHC in 150 formalin-fixed, paraffin-embedded human PC tissues with different Gleason scores [36]. A significantly strong STAT3 expression was seen in 68% PC cases as compared to only 12% of BPH cases (p<0.001). Of the cases with STAT3 expression, majority were of higher Gleason score (>7; 75%). 54% of the cases exhibited strong immunoreactivity for STAT5 as compared to only 13% of BPH controls (p<0.001), and 64% of the cases had a higher Gleason score (>7). A consistently intense nuclear staining of STAT proteins with high Gleason scores (>7) as compared to low scores (less than and equal to 7) was observed. Han et al. showed the usefulness of phosphorylated-STAT3 in detecting PC from negative biopsies [34]. STAT3 staining intensities in all samples (initial negative biopsies, cancer positive cores, and other negative cores from the same batch biopsies) of cancer patients was significantly higher than that of benign patients (p<0.001), with a high sensitivity (80.8%) and specificity (76.3%). Furthermore, Tam et al. studied the expression levels STAT pathway in the transition from a hormone-sensitive to hormone-refractory PC by assessing the expression levels of these IL-6R, JAK1, STAT3, pSTAT3 (Y705), and pSTAT3-Ser727 (S727) by IHC in 50 matched hormone-sensitive and hormone-refractory tumors pairs. An increase in expression of cytoplasmic IL-6 receptor, with the development of hormone-refractory PC was seen to be associated with reduced time to relapse, while an increase in expression of cytoplasmic pSTAT3 (Y705) was associated with reduced patient survival. In addition, those patients with high expression of cytoplasmic pSTAT3 (Y705) in their tumors had significantly shorter time to death from biochemical relapse and overall survival in comparison to those patients with low expression of cytoplasmic pY705-STAT3. Haddad et al. demonstrated amplification at the STAT5A/B gene locus in a significant fraction of clinical PC specimens [33]. The STAT5A/B gene amplification was more frequently found in PC with high histologic grades and in CRPC with distant metastases. Thus, the works presented herein suggest that targeting of STAT3 and STAT5 could in practice be of a therapeutic significance.
Here, we demonstrated that the levels of activated, nuclear STAT3 and STAT5A proteins were significantly elevated in metastatic CRPC compared with BPH. We recognize that although a limited number of CRPC clinical cases were analyzed, the result of our study is consistent with the findings in literatures [30, 31, 33, 34, 36, 38]. In addition, we demonstrated that human PC cell lines express differential levels of STAT3 and STAT5A transcripts. In general, compared with the STAT3, the expression of STAT5A is significantly low in LNCaP, C4-2, and PC3 cells, with an exception of 22Rv1 cell line, which expressed the highest levels of STAT3 mRNA. Moreover, our study revealed that metastatic PC3 PC cells expressed the lowest levels of STAT3 and STAT5A transcripts relative to that of LNCaP, C4-2, and 22Rv1. Furthermore, our analysis of the PC public data showed that the STAT3 and STAT5A gene locus were amplified in up to 20% of metastatic CRPC with the NE phenotype. Additionally, our data showed that C4-2 and PC3 cells expressing the low levels of STAT3/5A conferred resistance to pimozide in comparison with LNCaP and 22Rv1 cells under the same experimental conditions. Taken together, our results suggest that STAT3 and STAT5A can be utilized as a predictive biomarker to evaluate the efficacy of therapies targeting the JAK/STAT pathway.
Currently, there is no effective pharmacotherapy that prolongs the survival of patients with metastatic CRPC [11, 53–55]. To discover additional targeted agents, identification of the target population, or the subset of patients with druggable molecular abnormalities with minimal adverse effect, is necessary. The evaluation of both STAT3 and STAT5A expression in conjunction with morphology is important in therapeutic decision-making for patients who can benefit from the STAT inhibitor in pharmacotherapy. Here, we demonstrated that activated, nuclear expressions of STAT3 and STAT5A proteins was significantly high in metastatic CRPC cases. Nuclear STAT3 and STAT5A/B protein levels are increased in high-grade PC, CRPC, and distant metastases, and the high levels of nuclear STAT3 and STAT5A/B expression can predict early disease recurrence and PC-specific death in clinical PC. Based on the available data and our observation, STAT proteins represent a therapeutic target protein for advanced PC. Also, quantitative in situ analysis and STAT mRNA expression show that STAT3 and STAT5 gene amplification was associated with increased protein expression in PC. Functional studies showed that increased STAT3 and STAT5A/B copy numbers conferred growth advantage in PC cells in vitro and as xenograft tumors in vivo [31, 33, 38]. However, it is hard to estimate whether all STAT3/5 transcripts can fully translate into STAT3/5 protein, which ultimately drives the biology. Therefore, although intratumoral antigenic heterogeneity exists, we suggest that analysis of STAT3/5 protein expression may be a better way of predicting a therapeutic response.
In conclusion, our results demonstrate that STAT3/5A protein expression may potentially serve as a predictive biomarker of responsiveness to therapies targeting metastatic CRPC with NE characteristics. The JAK/STAT and AR pathways are functionally synergistic in the neoplastic cells and may involve the progression of PC [56–58]. Thus, the inhibition of the JAK/STAT signaling alone or in combination with the AR may lead to a novel treatment modality for patients with advanced PC. Further studies with a large cohort of metastatic CRPC patients with clinical follow-up information are warranted to substantiate the current findings and to evaluate whether there is a direct and positive correlation between the levels of activated STAT3/5A protein and induction of growth suppression and apoptosis in PC cell and animal models by pimozide. In addition, pimozide is a potent inhibitor of dopamine receptors (DRDs), particularly DRD2. Evidence suggests that activation of DRDs is linked to cancer with poor prognosis [59–63]. Therefore, future investigations warrant to establish whether the DRD and the JAK/STAT pathways mechanistically and functionally interact with each other to contribute to aggressive PC and that pimozide may disrupt the interactions, by which it suppresses PC cell growth.
MATERIALS AND METHODS
Tissue procurement
The Anatomic Pathology database was searched for CRPC cases from January 2008 to August 2011. All human subject research was conducted according to a protocol (#Pro00027882) approved by the Institutional Review Board (IRB). Fifteen cases of benign prostatic hyperplasia (BPH) were also included in this study as a control group. Demographics and related clinical details were recorded in each case. The clinical parameters including surgery, androgen ablation, chemotherapy, radiation therapy, androgen resistance, and metastasis were recorded. The hematoxylin and eosin (H&E) stained and relevant immunohistochemistry slides were reviewed to identify cases pertinent to this study.
Immunohistochemistry
Immunohistochemical (IHC) staining of STAT3 and STAT5A proteins was performed on four-micrometer thick formalin-fixed and paraffin embedded (FFPE) tissues sections after confirmation of the diagnosis on H&E-stained sections. The automated immunostainer was used to stain STAT3 and STAT5A proteins in FFPE sections (Ventana Benchmark Ultra System, Roche). Table 1 summarizes the antibody clones, antibody dilutions, commercial vendors, antigen retrieval methods, and incubation time for the primary antibodies, and the localization techniques utilized in this study. Appropriate positive and negative controls were performed with each antibody. Positive staining of STAT3 and STAT5A protein was defined as a nuclear brown staining pattern that can be easily observed at low-power magnification (<×40) in the neoplastic cells in cancer cases and epithelial cells lining of the prostatic glands in BPH cases. Scant fine granular background staining of cells, which cannot be seen at low-power magnification (<×40) or no staining at all, was considered negative. The status of the immune markers was assessed without knowledge of the Gleason patterns and other clinical parameters of the cases. The staining results were recorded in semi-quantitative fashion as an estimated percentage of immune reactive tumor cells (in cancer cases) and benign prostate tissue (in BPH cases). 0: no staining, 1+ (focal), <25% of immunoreactive tumor or benign glandular epithelium; 2+ (multifocal), 26% to 50% of immunoreactive tumor or benign glandular epithelium; 3+ (diffuse): 51% to 100% of immunoreactive tumor or benign glandular epithelium. The staining intensity was recorded as weak, moderate, and strong.
Cell lines and RNA isolation
PC cell lines LNCaP, C4-2, 22Rv1 and PC3 were grown in RPMI 1640 cell culture medium at 37°C in 5% CO2 incubator. The culture medium was supplemented with 10% fetal bovine serum (FBS) and 1% penicillin and streptomycin antibiotics (Thermo-Fisher Scientific). Total RNA from LNCaP, C4-2, 22Rv1, and PC3 cells that were grown at 70-80% confluency was isolated using RNA isolation kit according to manufacturer’s instruction (Thermo-Fisher Scientific).
Quantitative RT-PCR
Levels of STAT3 and STAT5A transcripts were analyzed by reverse transcriptase-quantitative polymerase chain reaction (RT-qPCR) using GoTag® 1-step RT-qPCR system (Promega). Primer pairs are for STAT3 (Fv: 5’-GGA GCA GAG ATG TGG GAA TG-3’; Rv: 5’-GTG ATA CAC CTC GGT CTC AAA G-3’) and for STAT5A (Fv: 5’-CGG TTT GAG TGA GGG TTT CT-3’; Rv: 5’- GTG GGC AAC AGC ATC ATA GA-3’). Ribosomal 18S RNA was used as an internal control in RT-qPCR reactions to normalize the expression of STAT3 and STAT5A transcripts.
Cell growth and apoptosis assays
LNCaP, C4-2, 22Rv1, and PC3 cells were seeded in 96 well plates (3x103 cells/well) in serum-fed growth conditions 24h before exposed to various concentrations (0, 5, 10, and 20 μM) of pimozide, an indirect inhibitor of STAT5 activity/phosphorylation. Pimozide was obtained from Sigma-Aldrich. Pimozide was prepared in DMSO in 20 (mM) stock concentration and stored in -20 °C. At 48h post treatment, cell proliferation or cell growth was assayed using a CellTiter 96 AQueous system (Promega) [64] or cell counting kit-8 (CCK-8) (Dojindo Molecular Technologies) in accordance with the manufacturer’s instruction. In a separate experiment and under the same growth and treatment conditions above, apoptosis induced by pimozide treatment was evaluated by Flow Cytometry using propidium iodide (PI) that stains apoptotic cells [65].
Statistical analysis
Statistical comparison between the expression of STAT3 and STAT5A was studied among both CRPC and BPH groups. In addition, comparisons were made between the patterns of expression (semi-quantitative estimation of staining) for both groups for each marker. Important clinical parameters including age of the patient, Gleason score, and metastasis were compared with STAT3 and STAT5A expression in tumor groups. Pearson Chi-square test, Fischer’s exact test, and Student t-test (2-tailed) were used. Student t-test (2-tailed) was used to determine the significance between the two groups. A p-value of ≤ 0.05 was considered statistically significant.
Abbreviations
PC: Prostate cancer, CRPC: Castration-resistant prostate cancer, AR: Androgen receptor, BPH: Benign prostatic hyperplasia, IRB: Institutional review board, IHC: Immunohistochemistry, STAT:Signal transducer and activator of transcription, JAK:Janus tyrosine kinase, FFPE:formalin-fixed and paraffin embedded, RT-qPCR: Reverse transcriptase-quantitative polymerase chain reaction, Fv: Forward, Rv: Reverse, PI: Propidium iodide.
Author’s contributions
Conception and design: S. Alkan, B. Cinar.
Development of methodology: S. Mohanty, K. Yagiz, B. Cinar.
Acquisition of data: S. Mohanty, K. Yagiz, B. Cinar.
Analysis and interpretation of data: S. Mohanty, K. Yagiz, B. Cinar. D. Pradhan, S. Alkan.
Writing and review/revision of the manuscript: S. Mohanty, B. Cinar, S. Alkan, D. Pradhan, D. Luthringer, M. Amin.
Administrative, technical, or material support: S. Alkan. B. Cinar.
Study supervision: S. Alkan, B. Cinar.
CONFLICTS OF INTEREST
The authors declare no competing conflicts of interest.
FUNDING
This study was supported in part by the NIH/NIMHD/RCMI Grant (5G12MD007590).
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017; 67:7-30.
2. Zhou Y, Bolton EC, Jones JO. Androgens and androgen receptor signaling in prostate tumorigenesis. J Mol Endocrinol. 2015; 54:R15-29.
3. Wang D, Tindall DJ. Androgen action during prostate carcinogenesis. Methods Mol Biol. 2011; 776:25-44.
4. Saylor PJ. Prostate cancer: The androgen receptor remains front and centre. Nat Rev Clin Oncol. 2013; 10:126-128.
5. Feldman BJ, Feldman D. The development of androgen-independent prostate cancer. Nat Rev Cancer. 2001; 1:34-45.
6. Cancer Genome Atlas Research Network. The Molecular Taxonomy of Primary Prostate Cancer. Cell. 2015; 163:1011-1025.
7. Yuan X, Cai C, Chen S, Chen S, Yu Z, Balk SP. Androgen receptor functions in castration-resistant prostate cancer and mechanisms of resistance to new agents targeting the androgen axis. Oncogene. 2014; 33:2815-2825.
8. Yin L, Hu Q. CYP17 inhibitors--abiraterone, C17,20-lyase inhibitors and multi-targeting agents. Nat Rev Urol. 2014; 11:32-42.
9. Watson PA, Arora VK, Sawyers CL. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat Rev Cancer. 2015; 15:701-711.
10. Lorente D, Mateo J, Zafeiriou Z, Smith AD, Sandhu S, Ferraldeschi R, de Bono JS. Switching and withdrawing hormonal agents for castration-resistant prostate cancer. Nat Rev Urol. 2015; 12:37-47.
11. Harris WP, Mostaghel EA, Nelson PS, Montgomery B. Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat Clin Pract Urol. 2009; 6:76-85.
12. Ciccarese C, Santoni M, Brunelli M, Buti S, Modena A, Nabissi M, Artibani W, Martignoni G, Montironi R, Tortora G, Massari F. AR-V7 and prostate cancer: The watershed for treatment selection? Cancer Treat Rev. 2016; 43:27-35.
13. Antonarakis ES, Lu C, Wang H, Luber B, Nakazawa M, Roeser JC, Chen Y, Mohammad TA, Chen Y, Fedor HL, Lotan TL, Zheng Q, De Marzo AM, et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med. 2014; 371:1028-1038.
14. Katzenwadel A, Wolf P. Androgen deprivation of prostate cancer: Leading to a therapeutic dead end. Cancer Lett. 2015; 367:12-17.
15. Beltran H, Tomlins S, Aparicio A, Arora V, Rickman D, Ayala G, Huang J, True L, Gleave ME, Soule H, Logothetis C, Rubin MA. Aggressive variants of castration-resistant prostate cancer. Clin Cancer Res. 2014; 20:2846-2850.
16. Attard G, Reid AH, Olmos D, de Bono JS. Antitumor activity with CYP17 blockade indicates that castration-resistant prostate cancer frequently remains hormone driven. Cancer Res. 2009; 69:4937-4940.
17. Ardiani A, Farsaci B, Rogers CJ, Protter A, Guo Z, King TH, Apelian D, Hodge JW. Combination therapy with a second-generation androgen receptor antagonist and a metastasis vaccine improves survival in a spontaneous prostate cancer model. Clin Cancer Res. 2013; 19:6205-6218.
18. Alva A, Hussain M. Intermittent androgen deprivation therapy in advanced prostate cancer. Curr Treat Options Oncol. 2014; 15:127-136.
19. Wagner KU, Schmidt JW. The two faces of Janus kinases and their respective STATs in mammary gland development and cancer. J Carcinog. 2011; 10:32.
20. Zhang X, Blaskovich MA, Forinash KD, Sebti SM. Withacnistin inhibits recruitment of STAT3 and STAT5 to growth factor and cytokine receptors and induces regression of breast tumours. Br J Cancer. 2014; 111:894-902.
21. Valgeirsdottir S, Paukku K, Silvennoinen O, Heldin CH, Claesson-Welsh L. Activation of Stat5 by platelet-derived growth factor (PDGF) is dependent on phosphorylation sites in PDGF beta-receptor juxtamembrane and kinase insert domains. Oncogene. 1998; 16:505-515.
22. Sato K, Nagao T, Iwasaki T, Nishihira Y, Fukami Y. Src-dependent phosphorylation of the EGF receptor Tyr-845 mediates Stat-p21waf1 pathway in A431 cells. Genes Cells. 2003; 8:995-1003.
23. Runge DM, Runge D, Foth H, Strom SC, Michalopoulos GK. STAT 1alpha/1beta, STAT 3 and STAT 5: expression and association with c-MET and EGF-receptor in long-term cultures of human hepatocytes. Biochem Biophys Res Commun. 1999; 265:376-381.
24. Neilson LM, Zhu J, Xie J, Malabarba MG, Sakamoto K, Wagner KU, Kirken RA, Rui H. Coactivation of janus tyrosine kinase (Jak)1 positively modulates prolactin-Jak2 signaling in breast cancer: recruitment of ERK and signal transducer and activator of transcription (Stat)3 and enhancement of Akt and Stat5a/b pathways. Mol Endocrinol. 2007; 21:2218-2232.
25. Kirken RA, Erwin RA, Wang L, Wang Y, Rui H, Farrar WL. Functional uncoupling of the Janus kinase 3-Stat5 pathway in malignant growth of human T cell leukemia virus type 1-transformed human T cells. J Immunol. 2000; 165:5097-5104.
26. Katsha A, Arras J, Soutto M, Belkhiri A, El-Rifai W. AURKA regulates JAK2-STAT3 activity in human gastric and esophageal cancers. Mol Oncol. 2014; 8:1419-1428.
27. Frost RA, Nystrom GJ, Lang CH. Regulation of IGF-I mRNA and signal transducers and activators of transcription-3 and -5 (Stat-3 and -5) by GH in C2C12 myoblasts. Endocrinology. 2002; 143:492-503.
28. Chang M, Kanwar N, Feng E, Siu A, Liu X, Ma D, Jongstra J. PIM kinase inhibitors downregulate STAT3(Tyr705) phosphorylation. Mol Cancer Ther. 2010; 9:2478-2487.
29. Gu L, Dagvadorj A, Lutz J, Leiby B, Bonuccelli G, Lisanti MP, Addya S, Fortina P, Dasgupta A, Hyslop T, Bubendorf L, Nevalainen MT. Transcription factor Stat3 stimulates metastatic behavior of human prostate cancer cells in vivo, whereas Stat5b has a preferential role in the promotion of prostate cancer cell viability and tumor growth. Am J Pathol. 2010; 176:1959-1972.
30. Cocchiola R, Romaniello D, Grillo C, Altieri F, Liberti M, Magliocca FM, Chichiarelli S, Marrocco I, Borgoni G, Perugia G, Eufemi M. Analysis of STAT3 post-translational modifications (PTMs) in human prostate cancer with different Gleason Score. Oncotarget. 2017; 8:42560-70. https://doi.org/10.18632/oncotarget.17245.
31. Don-Doncow N, Marginean F, Coleman I, Nelson PS, Ehrnstrom R, Krzyzanowska A, Morrissey C, Hellsten R, Bjartell A. Expression of STAT3 in Prostate Cancer Metastases. Eur Urol. 2017; 71:313-316.
32. Gu L, Liao Z, Hoang DT, Dagvadorj A, Gupta S, Blackmon S, Ellsworth E, Talati P, Leiby B, Zinda M, Lallas CD, Trabulsi EJ, McCue P, et al. Pharmacologic inhibition of Jak2-Stat5 signaling By Jak2 inhibitor AZD1480 potently suppresses growth of both primary and castrate-resistant prostate cancer. Clin Cancer Res. 2013; 19:5658-5674.
33. Haddad BR, Gu L, Mirtti T, Dagvadorj A, Vogiatzi P, Hoang DT, Bajaj R, Leiby B, Ellsworth E, Blackmon S, Ruiz C, Curtis M, Fortina P, et al. STAT5A/B gene locus undergoes amplification during human prostate cancer progression. Am J Pathol. 2013; 182:2264-2275.
34. Han G, Yu JY, Chen YD, Cao XL, Zhu J, Wang W, Wang XX, Zhang X, Yan JQ, Gao JP. The usefulness of phosphorylated-signal transduction and activators of transcription 3 in detecting prostate cancer from negative biopsies. Eur J Surg Oncol. 2012; 38:367-373.
35. Ni Z, Lou W, Lee SO, Dhir R, DeMiguel F, Grandis JR, Gao AC. Selective activation of members of the signal transducers and activators of transcription family in prostate carcinoma. J Urol. 2002; 167:1859-1862.
36. Singh N, Hussain S, Bharadwaj M, Kakkar N, Singh SK, Sobti RC. Overexpression of signal transducer and activator of transcription (STAT-3 and STAT-5) transcription factors and alteration of suppressor of cytokine signaling (SOCS-1) protein in prostate cancer. J Recept Signal Transduct Res. 2012; 32:321-327.
37. Singh RP, Raina K, Deep G, Chan D, Agarwal R. Silibinin suppresses growth of human prostate carcinoma PC-3 orthotopic xenograft via activation of extracellular signal-regulated kinase 1/2 and inhibition of signal transducers and activators of transcription signaling. Clin Cancer Res. 2009; 15:613-621.
38. Tam L, McGlynn LM, Traynor P, Mukherjee R, Bartlett JM, Edwards J. Expression levels of the JAK/STAT pathway in the transition from hormone-sensitive to hormone-refractory prostate cancer. Br J Cancer. 2007; 97:378-383.
39. Nelson EA, Walker SR, Weisberg E, Bar-Natan M, Barrett R, Gashin LB, Terrell S, Klitgaard JL, Santo L, Addorio MR, Ebert BL, Griffin JD, Frank DA. The STAT5 inhibitor pimozide decreases survival of chronic myelogenous leukemia cells resistant to kinase inhibitors. Blood. 2011; 117:3421-3429.
40. Epstein JI, Amin MB, Beltran H, Lotan TL, Mosquera JM, Reuter VE, Robinson BD, Troncoso P, Rubin MA. Proposed morphologic classification of prostate cancer with neuroendocrine differentiation. Am J Surg Pathol. 2014; 38:756-767.
41. Mohanty SK, Smith SC, Chang E, Luthringer DJ, Gown AM, Aron M, Amin MB. Evaluation of contemporary prostate and urothelial lineage biomarkers in a consecutive cohort of poorly differentiated bladder neck carcinomas. Am J Clin Pathol. 2014; 142:173-183.
42. Ylitalo EB, Thysell E, Jernberg E, Lundholm M, Crnalic S, Egevad L, Stattin P, Widmark A, Bergh A, Wikstrom P. Subgroups of Castration-resistant Prostate Cancer Bone Metastases Defined Through an Inverse Relationship Between Androgen Receptor Activity and Immune Response. Eur Urol. 2017; 71:776-787.
43. Horoszewicz JS, Leong SS, Kawinski E, Karr JP, Rosenthal H, Chu TM, Mirand EA, Murphy GP. LNCaP model of human prostatic carcinoma. Cancer Res. 1983; 43:1809-1818.
44. Thalmann GN, Anezinis PE, Chang SM, Zhau HE, Kim EE, Hopwood VL, Pathak S, von Eschenbach AC, Chung LW. Androgen-independent cancer progression and bone metastasis in the LNCaP model of human prostate cancer. Cancer Res. 1994; 54:2577-2581.
45. Kaighn ME, Narayan KS, Ohnuki Y, Lechner JF, Jones LW. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest Urol. 1979; 17:16-23.
46. Sramkoski RM, Pretlow TG 2nd, Giaconia JM, Pretlow TP, Schwartz S, Sy MS, Marengo SR, Rhim JS, Zhang D, Jacobberger JW. A new human prostate carcinoma cell line, 22Rv1. In Vitro Cell Dev Biol Anim. 1999; 35:403-409.
47. Beltran H, Prandi D, Mosquera JM, Benelli M, Puca L, Cyrta J, Marotz C, Giannopoulou E, Chakravarthi BV, Varambally S, Tomlins SA, Nanus DM, Tagawa ST, et al. Divergent clonal evolution of castration-resistant neuroendocrine prostate cancer. Nat Med. 2016; 22:298-305.
48. Stephenson AJ, Bolla M, Briganti A, Cozzarini C, Moul JW, Roach M 3rd, van Poppel H, Zietman A. Postoperative radiation therapy for pathologically advanced prostate cancer after radical prostatectomy. Eur Urol. 2012; 61:443-451.
49. Warde P, Mason M, Ding K, Kirkbride P, Brundage M, Cowan R, Gospodarowicz M, Sanders K, Kostashuk E, Swanson G, Barber J, Hiltz A, Parmar MK, et al, and NCIC CTG PR.3/MRC UK PR07 investigators. Combined androgen deprivation therapy and radiation therapy for locally advanced prostate cancer: a randomised, phase 3 trial. Lancet. 2011; 378:2104-2111.
50. Ferraldeschi R, Welti J, Luo J, Attard G, de Bono JS. Targeting the androgen receptor pathway in castration-resistant prostate cancer: progresses and prospects. Oncogene. 2015; 34:1745-1757.
51. Smith DC, Smith MR, Sweeney C, Elfiky AA, Logothetis C, Corn PG, Vogelzang NJ, Small EJ, Harzstark AL, Gordon MS, Vaishampayan UN, Haas NB, Spira AI, et al. Cabozantinib in patients with advanced prostate cancer: results of a phase II randomized discontinuation trial. J Clin Oncol. 2013; 31:412-419.
52. Quinn DI, Tangen CM, Hussain M, Lara PN Jr, Goldkorn A, Moinpour CM, Garzotto MG, Mack PC, Carducci MA, Monk JP, Twardowski PW, Van Veldhuizen PJ, Agarwal N, et al. Docetaxel and atrasentan versus docetaxel and placebo for men with advanced castration-resistant prostate cancer (SWOG S0421): a randomised phase 3 trial. Lancet Oncol. 2013; 14:893-900.
53. Clarke RA, Allen BJ. Next-generation therapy for residual prostate cancer. Immunotherapy. 2013; 5:1235-1241.
54. Santer FR, Erb HH, McNeill RV. Therapy escape mechanisms in the malignant prostate. Semin Cancer Biol. 2015; 35:133-144.
55. Robinson D, Van Allen EM, Wu YM, Schultz N, Lonigro RJ, Mosquera JM, Montgomery B, Taplin ME, Pritchard CC, Attard G, Beltran H, Abida W, Bradley RK, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015; 161:1215-1228.
56. Kroon P, Berry PA, Stower MJ, Rodrigues G, Mann VM, Simms M, Bhasin D, Chettiar S, Li C, Li PK, Maitland NJ, Collins AT. JAK-STAT blockade inhibits tumor initiation and clonogenic recovery of prostate cancer stem-like cells. Cancer Res. 2013; 73:5288-5298.
57. Kwon EM, Holt SK, Fu R, Kolb S, Williams G, Stanford JL, Ostrander EA. Androgen metabolism and JAK/STAT pathway genes and prostate cancer risk. Cancer Epidemiol. 2012; 36:347-353.
58. Gu L, Vogiatzi P, Puhr M, Dagvadorj A, Lutz J, Ryder A, Addya S, Fortina P, Cooper C, Leiby B, Dasgupta A, Hyslop T, Bubendorf L, et al. Stat5 promotes metastatic behavior of human prostate cancer cells in vitro and in vivo. Endocr Relat Cancer. 2010; 17:481-493.
59. Borcherding DC, Tong W, Hugo ER, Barnard DF, Fox S, LaSance K, Shaughnessy E, Ben-Jonathan N. Expression and therapeutic targeting of dopamine receptor-1 (D1R) in breast cancer. Oncogene. 2016; 35:3103-3113.
60. Hussein N, Amawi H, Karthikeyan C, Hall FS, Mittal R, Trivedi P, Ashby CR Jr, Tiwari AK. The dopamine D3 receptor antagonists PG01037, NGB2904, SB277011A, and U99194 reverse ABCG2 transporter-mediated drug resistance in cancer cell lines. Cancer Lett. 2017; 396:167-180.
61. Jandaghi P, Najafabadi HS, Bauer AS, Papadakis AI, Fassan M, Hall A, Monast A, von Knebel Doeberitz M, Neoptolemos JP, Costello E, Greenhalf W, Scarpa A, Sipos B, et al. Expression of DRD2 Is Increased in Human Pancreatic Ductal Adenocarcinoma and Inhibitors Slow Tumor Growth in Mice. Gastroenterology. 2016; 151:1218-1231.
62. Roy S, Lu K, Nayak MK, Bhuniya A, Ghosh T, Kundu S, Ghosh S, Baral R, Dasgupta PS, Basu S. Activation of D2 Dopamine Receptors in CD133+ve Cancer Stem Cells in Non-small Cell Lung Carcinoma Inhibits Proliferation, Clonogenic Ability, and Invasiveness of These Cells. J Biol Chem. 2017; 292:435-445.
63. Sachlos E, Risueño RM, Laronde S, Shapovalova Z, Lee JH, Russell J, Malig M, McNicol JD, Fiebig-Comyn A, Graham M, Levadoux-Martin M, Lee JB, Giacomelli AO, et al. Identification of drugs including a dopamine receptor antagonist that selectively target cancer stem cells. Cell. 2012; 149:1284-1297.
64. Cinar B, Collak FK, Lopez D, Akgul S, Mukhopadhyay NK, Kilicarslan M, Gioeli DG, Freeman MR. MST1 is a multifunctional caspase-independent inhibitor of androgenic signaling. Cancer Res. 2011; 71:4303-4313.
65. Kuser-Abali G, Alptekin A, Lewis M, Garraway IP, Cinar B. YAP1 and AR interactions contribute to the switch from androgen-dependent to castration-resistant growth in prostate cancer. Nat Commun. 2015; 6:8126.

