INTRODUCTION
The worldwide epidemic of obesity poses a major risk for the development of type 2 diabetes (T2D), cardiovascular disease (CVD), hypertension, stroke and certain types of cancer [1, 2]. In China, obesity rates have increased sharply over the past 20-30 years, particularly in childhood, rapidly climbing from 0.2% in 1985 to 8.1% in 2012 [3]. Both genetic predisposition and lifestyle factors are known to contribute to the risk of obesity [4]. To date, genome-wide association studies (GWAS) have identified numerical loci that contribute to body weight in adults and children of diverse ancestry [5-16]. However, the majority of these studies were conducted in populations of European ancestry whereas studies of pediatric obesity in Asians remain limited [11-14]. Moreover, recent reports of obesity-related loci enriched in brain-expressed genes such as FTO, MC4R, GNPDA2, and BDNF point to a neuronal influence on body weight regulation in adults [17]. We queried whether such an influence existed, or was even more pronounced in children. Therefore, we selected thirteen of the strongest BMI-related loci reported from a recent meta-analysis of GWAS in adult of East Asian-ancestry populations [12-14] to test their associations with obesity in a well-established cohort of Chinese school-aged children, with particularly focus on the single-nucleotide polymorphisms (SNPs) related to brain-expressed loci.
In parallel, recent studies have shown that obesity-related cadiometabolic risk can be mediated by the pleiotropic effects of adipokines, the secretory products that reflect endocrine function in adipose tissue [18], particularly by these adipokines which may function via the central nervous system (CNS), such as leptin, adiponectin, resistin, fibroblast growth factor 21 (FGF21) [19] and retinol binding protein 4 (RBP4) [20]. We therefore elected to investigate if these selected genetic variants are associates with a set of adipokines, as well as other cardiometabolic traits, in an attempt to gain novel insight in to their role in both obesity and cardiometabolic dysregulation.
RESULTS
Cohort characteristics
General characteristics of the study subjects are listed in Table 1. The study was conducted in 3,506 including normal weight (n = 1,626), overweight (n = 654), and obese (n = 1,226) unrelated individuals recruited through the BCAMS study. 51% of the participants were male, and the mean age was 12.4 ± 3.1 years. Obesity-related traits, blood pressures, triglyceride (TG), low density lipoprotein cholesterol (LDL-C), fasting insulin and homeostasis model assessment of insulin resistance (HOMA-IR) increased, while high density lipoprotein cholesterol (HDL-C) decreased, with increasing body mass index (BMI) (all P < 0.001). As expected, obese children exhibited highest leptin, resistin and RBP4 levels, and lowest adiponectin and FGF21 concentrations compared with the other two groups (all P < 0.001).
Table 1: Descriptive statistics of phenotypes used in the study
Phenotype |
Mean (SD, %) |
P |
|||
|---|---|---|---|---|---|
All |
Normal weight |
Overweight |
Obese |
||
Male/n (%) |
1787/3506 (51%) |
674/1626 (41.5%) |
328/654 (50.2%) |
785/1226(64%) |
< 0.001 |
Anthropometric parameters |
|||||
Age (yrs) |
12.4 ± 3.1 |
12.45 ± 3.16 |
13.29 ± 2.97 * |
11.80 ± 2.88 *+ |
< 0.001 |
Puberty stages |
2.68 ± 1.43 |
2.68 ± 1.36 |
3.16 ± 1.44 * |
2.41 ± 1.44 *+ |
< 0.001 |
BMI (kg/m2) |
21.9 ± 4.9 |
17.78 ± 2.43 |
23.41 ± 2.48* |
26.57 ± 3.66 *+ |
< 0.001 |
WC (cm) |
72.4 ± 13.1 |
62.24 ± 7.14 |
76.20 ± 7.79 * |
83.73 ± 10.84 *+ |
< 0.001 |
Percent body fat |
24. 4 ± 8.54 |
18.0 ± 5.64 |
27.8 ± 6.12* |
30.8 ± 6.66*+ |
< 0.001 |
UAC (cm) |
25.4 ± 4.94 |
21.7 ± 3.23 |
27.2 ± 3.05* |
29.3 ± 3.95*+ |
< 0.001 |
SBP (mmHg) |
108 ± 14 |
101± 12 |
111± 12* |
114± 13 *+ |
< 0.001 |
DBP (mmHg) |
68 ± 10 |
64± 10 |
69± 9 * |
72 ± 9 *+ |
< 0.001 |
Biochemical parameters |
|||||
TC (mmol/L) |
4.09 ± 0.82 |
4.12 ± 0.90 |
4.03 ± 0.76 * |
4.09 ± 0.74 * |
0.040 |
TG (mmol/L) |
0.93 (0.91-0.94) |
0.81 (0.80-0.83) |
0.95 (0.92-0.99) * |
1.08 (1.05-1.10) *+ |
< 0.001 |
LDL-C (mmol/L) |
2.55 ± 0.75 |
2.49 ± 0.84 |
2.53 ± 0.69 |
2.63 ± 0.66 *+ |
< 0.001 |
HDL-C (mmol/L) |
1.40 ± 0.32 |
1.53 ± 0.32 |
1.33 ± 0.29 * |
1.27 ± 0.26 *+ |
< 0.001 |
FBG (mmol/L) |
5.08 ± 0.55 |
5.00 ± 0.47 |
5.16 ± 0.79 * |
5.15 ± 0.48 * |
< 0.001 |
Fasting insulin (mU/L) |
8.19 (7.97-8.41) |
5.63 (5.43-5.83) |
9.43 (8.99-9.89) * |
12.05 (11.61-12.54) *+ |
< 0.001 |
HOMA-IR |
1.84 (1.80-1.90) |
1.25 (1.20-1.30) |
2.15 (2.05-2.26) * |
2.75 (2.64-2.87) *+ |
< 0.001 |
Adipokines |
|||||
Leptin (ng /ml) |
5.18 (4.93-5.41) |
2.05 (1.93-2.18) |
7.76 (7.26-8.40) * |
13.17 (12.59-13.77) *+ |
< 0.001 |
Adiponectin (ug/ml) |
5.41 (5.30-5.52) |
6.46 (6.28-6.66) |
4.89 (4.68-5.12) * |
4.57 (4.42-4.71) *+ |
< 0.001 |
Resistin (ng /ml) |
15.36 (15.06-15.63) |
14.82 (14.40-15.26) |
15.43 (14.75-16.08) |
16.01 (15.50-16.52) * |
< 0.001 |
FGF21 (pg/ml) |
619.8 (594.0-650.1) |
676.9 (635.0-730.4) |
628.3 (568.4-692.7) |
552.0 (513.0-595.9) * |
< 0.001 |
RBP4 (ug/ml) |
32.1 (31.7-32.5) |
29.75 (29.19-30.31) |
33.6 (32.7-34.5) * |
34.5(33.8-35.1) * |
< 0.001 |
Data were expressed as mean ± SD, n (%). Skewed distributions were expressed as geometric mean (95% confidence interval). One-way ANOVA where differences versus normal weight subjects are indicated as *P < 0.05, differences versus overweight individuals are indicated as + P < 0.05.
Abbreviation: BMI, body mass index; WC, waist circumference; UAC, upper arm circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; LDL-C, low density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; HOMA-IR, homeostasis model assessment for insulin resistance; FGF21, fibroblast growth factor 21 and RBP4, retinol binding protein 4.
Replication with childhood obesity
The allele frequencies of the SNPs and the results of genotypic tests are listed in Table 2. The minor allele frequencies of the twelve SNPs utilized ranged from 10% to 47%. Using the additive model, six SNPs revealed significant associations with obesity after multiple testing correction (Bonferroni corrected P < 0.004), with odds ratios (ORs) from 1.211 to 1.421, namely FTO-rs1558902 (P = 5.6×10−5), MC4R-rs2331841 (P = 4.4×10−4), GNPDA2-rs16858082(P = 3.4×10−4), PCSK1-rs261967 (P = 0.001), SEC16B-rs516636 (P = 0.004), and MAP2K5-rs4776970 (P = 0.004), while other two loci, ITIH4-rs2535633 (P = 0.012) and BDNF-rs2030323(P = 0.031), showed nominal association with obesity, with ORs 1.157 and 1.133, respectively. Similar trends were also evident when combining overweight and obesity status and comparing them with the normal weight group (normal weight vs. overweight & obesity).
Table 2: Associations of SNPs with childhood obesity
SNP |
Nearest gene |
a/A* |
MAF† |
P (HWE)‡ |
Normal vs Obesity |
Normal vs Overweight & Obesity |
BMI (kg/m2) |
||||
|---|---|---|---|---|---|---|---|---|---|---|---|
OR(95% CI) |
P § |
OR (95% CI) |
P § |
β (95% CI) |
P § |
||||||
rs1558902 |
FTO |
intron |
T/A |
0.10 |
0.02 |
1.421 (1.198-1.686) |
5.6E-5 |
1.405 (1.204-1.639) |
1.58E-5 |
0.831 (0.509-1.154) |
4.6E-7 |
rs2331841 |
MC4R |
near |
A/G |
0.22 |
0.82 |
1.267 (1.111-1.445) |
4.4E-4 |
1.212 (1.079-1.363) |
0.001 |
0.486 (0.238-0.735) |
1.3E-4 |
rs16858082 |
GNPDA2 |
near |
C/T |
0.34 |
0.56 |
1.239 (1.102-1.393) |
3.4E-4 |
1.220 (1.100-1.353) |
1.68E-4 |
0.384 (0.164-0.604) |
0.001 |
rs261967 |
PCSK1 |
intron |
A/C |
0.41 |
0.24 |
1.212 (1.081-1.359) |
0.001 |
1.164 (1.052-1.288) |
0.003 |
0.311 (0.095-0.528) |
0.005 |
rs516636 |
SEC16B |
near |
C/A |
0.20 |
0.38 |
1.221 (1.064-1.401) |
0.004 |
1.224 (1.082-1.384) |
0.001 |
0.371 (0.110-0.631) |
0.005 |
rs4776970 |
MAP2K5 |
intron |
T/A |
0.22 |
0.01 |
1.211 (1.063-1.379) |
0.004 |
1.207 (1.076-1.355) |
0.001 |
0.347 (0.101-0.594) |
0.006 |
rs2535633 |
ITIH4 |
intron |
C/G |
0.41 |
0.52 |
1.157 (1.033-1.295) |
0.012 |
1.122 (1.015-1.241) |
0.024 |
0.221 (0.006-0.436) |
0.044 |
rs2030323 |
BDNF |
intron |
T/G |
0.47 |
0.95 |
1.133 (1.011-1.270) |
0.031 |
1.117 (1.011-1.235) |
0.030 |
0.216 (0.004-0.428) |
0.046 |
rs6545814 |
ADCY3/RBJ |
intron |
A/G |
0.41 |
0.89 |
1.107 (0.989-1.240) |
0.078 |
1.073 (0.969-1.187) |
0.175 |
0.219 (0.002-0.435) |
0.047 |
rs652722 |
PAX6 |
intron |
T/C |
0.34 |
0.75 |
0.937 (0.832-1.056) |
0.287 |
0.936 (0.843-1.040) |
0.219 |
-0.118 (-0.343-0.107) |
0.303 |
rs12597579 |
GP2 |
intron |
T/C |
0.28 |
0.52 |
1.069 (0.942-1.213) |
0.303 |
1.049 (0.939-1.173) |
0.395 |
0.139 (-0.099-0.378) |
0.252 |
rs2237892 |
KCNQ1 |
intron |
T/C |
0.31 |
0.07 |
0.983 (0.872-1.109) |
0.783 |
0.982 (0.883-1.092) |
0.737 |
0.052 (-0.176-0.279) |
0.655 |
12 loci |
GPSall |
/ |
/ |
/ |
/ |
1.150 (1.108-1.194) |
2.4E-13 |
1.128 (1.091-1.166) |
9.1E-13 |
0.261 (0.193-0.330) |
9.4E-14 |
*a: non-effect; A: effect; † minor allele frequency; ‡P value for Hardy-Weinberg equilibrium test; OR/β (95% CI) and §P value for logistic/linear regression in the additive model were adjusted for gender, age, Tanner stage and residence.
Abbreviation: MC4R, melanocortin 4 receptor; FTO, fat mass and obesity associated; GNPDA2, glucosamine-6-phosphate deaminase 2; PCSK1, proprotein convertase subtilisin/kexin type 1; SEC16B, SEC16 homolog B, endoplasmic reticulum export factor; MAP2K5, mitogen-activated protein kinase 5; ITIH4, inter-alpha-trypsin inhibitor heavy chain family, member 4; BDNF, brain-derived neurotrophic factor; ADCY3/RBJ, adenylate cyclase 3; PAX6, paired box 6; GP2, glycoprotein 2; KCNQ1, potassium channel, voltage gated KQT-like subfamily Q, member 1 and GPSall, sum gene score of all 12 loci.
Note: Values in bold are significant at P < 0.05
Similarly, we found strong associations between BMI and variants in/near FTO (P = 4.6×10−7), MC4R (P = 1.3×10−4), and GNPDA2 (P = 0.001), and nominal association in/near SEC16B, PCSK1, MAP2K5, ITIH4, BDNF and ADCY3/RBJ (P < 0.05), with the effects ranging from 0.83 kg/m2 to 0.22 kg/m2 per allele (Table 2). Moreover, we found similar effects of BMI-related loci on other obesity-related anthropometric traits i.e. waist circumference (WC), percent body fat and upper arm circumference (UAC) (Table 5), which was largely consistent with the above observations with obesity risk. The genetic predisposition score consisting of all twelve adult BMI-associated variants (GPSall) was more significantly associated with BMI (P = 9.40×10−14), WC (P = 1.42×10−14), percent body fat (6.10×10−11) and UAC (3.50×10−12) than with any single SNP.
Biological mechanisms for obesity predisposing genes
In order to examine biological mechanisms for obesity predisposing genes, we selected the SNPs associated with BMI/obesity (at least one of these traits) from above analysis, plus all the 12 SNP-based gene score (GPSall) and tested their association with the 5 adipokines. As shown in Table 3, we found the risk alleles of FTO-rs1558902 (P = 0.002), MAP2K5-rs4776970 (P = 0.002) and MC4R-rs2331841 (P = 0.003) yielded significant association with increasing leptin levels at a Bonferroni-corrected threshold of P ≤ 0.004, while the risk alleles of GNPDA2-rs16858082 (P = 0.007), PCSK1-rs261967 (P = 0.007) and BDNF-rs2030323 (P = 0.027) yielded nominal association with leptin levels. In addition, the risk alleles of MC4R-rs2331841 (P = 0.020) and BDNF-rs2030323 (P = 0.032) displayed nominal association with decreased adiponectin levels, while PCSK1-rs261967 showed a nominal association with increased adiponectin levels (P = 0.038). The GPSall was more significantly associated with leptin levels (P = 9.70×10−10). However, neither GPSall nor any individual single SNP was associated with the levels of other adipokines.
Table 3: Associations of the selected SNPs and GPS with five adipokines
Gene |
SNP |
Leptin (ng/ml)# |
Adiponectin (ug/ml)# |
Resistin (ng/ml)# |
FGF21 (pg/ml)# |
RBP4 (ug/ml)# |
|
|---|---|---|---|---|---|---|---|
FTO |
rs1558902 |
β |
0.149 |
-0.027 |
0.019 |
0.022 |
0.004 |
P |
0.002 |
0.202 |
0.342 |
0.664 |
0.760 |
||
MC4R |
rs2331841 |
β |
0.110 |
-0.038 |
0.011 |
0.012 |
0.009 |
P |
0.003 |
0.020 |
0.454 |
0.764 |
0.340 |
||
GNPDA2 |
rs16858082 |
β |
0.089 |
-0.004 |
-0.005 |
-0.006 |
0.003 |
P |
0.007 |
0.800 |
0.721 |
0.815 |
0.736 |
||
SEC16B |
rs516636 |
β |
0.067 |
-0.025 |
0.013 |
-0.059 |
0.015 |
P |
0.088 |
0.153 |
0.402 |
0.150 |
0.157 |
||
PCSK1 |
rs261967 |
β |
0.087 |
0.030 |
0.012 |
-0.005 |
0.007 |
P |
0.007 |
0.038 |
0.350 |
0.882 |
0.423 |
||
MAP2K5 |
rs4776970 |
β |
0.114 |
-0.020 |
0.006 |
-0.012 |
0.014 |
P |
0.002 |
0.212 |
0.674 |
0.749 |
0.138 |
||
ITIH4 |
rs2535633 |
β |
0.050 |
-0.005 |
-0.022 |
-0.006 |
0.011 |
P |
0.118 |
0.719 |
0.094 |
0.868 |
0.185 |
||
BDNF |
rs2030323 |
β |
0.071 |
-0.030 |
0.005 |
-0.035 |
-0.007 |
P |
0.027 |
0.032 |
0.683 |
0.291 |
0.414 |
||
ADCY3/RBJ |
rs6545814 |
β |
0.050 |
-0.016 |
-0.013 |
-0.024 |
3.79E-4 |
P |
0.123 |
0.277 |
0.323 |
0.469 |
0.966 |
||
GPSall |
all 12 loci |
β |
0.062 |
-0.010 |
0.001 |
-0.02 |
0.004 |
P |
9.7E-10 |
0.022 |
0.807 |
0.064 |
0.128 |
||
Explained variation |
1.0% |
0.1% |
< 0.01% |
< 0.01% |
< 0.01% |
β means linear regression coefficients adjusted for gender, age, Tanner stage and residence.
Abbreviation: FGF21, fibroblast growth factor 21; RBP4, retinol binding protein 4; FTO, fat mass and obesity associated; MC4R, melanocortin 4 receptor; GNPDA2, glucosamine-6-phosphate deaminase 2; SEC16B, SEC16 homolog B, endoplasmic reticulum export factor; PCSK1, proprotein convertase subtilisin/kexin type 1; MAP2K5, mitogen-activated protein kinase 5; ITIH4, inter-alpha-trypsin inhibitor heavy chain family, member 4; BDNF, brain-derived neurotrophic factor ; ADCY3/RBJ, adenylate cyclase 3 and GPSall, sum gene score of all 12 loci.
# Skewed distributions were natural logarithmically (ln) transformed.
Note: Values in bold are significant at P < 0.004 after Bonferroni correction.
Next, we selected all the variants at the leptin-associated loci for further analysis. In the General Linear Model analyses (Table 4), after adjusting for age, sex, Tanner stage and residence, leptin levels were significantly increased in subjects with the minor alleles of PCSK1-rs261067, MC4R-rs2331841, FTO-rs1558902, MAP2K5-rs4776970 and GNPDA2-rs16858082, respectively (0.003 ≤ P ≤ 0.019), while there was a trend for an increase in subjects with the minor allele of BDNF-rs2030323 (P = 0.053), with effect sizes ranging from 0.2% to 0.4%. The GPS consisted of the six leptin-associated SNPs (GPSleptin) showed much stronger association with increased leptin levels (P = 2.8×10−6), with an increased effect size of 1.6%. Meanwhile, leptin levels in the top tertiles of GPSleptin (with 5-9 risk alleles) were increased by 31% compared to those in the lowest tertiles (with less than 3 risk alleles) (P < 0.001). In addition, GPSleptin was able to explain an estimated 1.5%, 1.3%, 1.2% and 1.2% of the total variance of BMI, WC, percent body fat, and UAC, respectively, thus the variations explained by GPSleptin, reaching or exceeding the cumulated effects of all twelve loci (i.e. GPS all), explaining 1.4 %, 1.3%, 1.2%, and 1.0% of the total variance, respectively (Table 5).
Table 4: Associations of selected loci with leptin levels
Gene |
Gene expression abundance in hypothalamus* |
SNP |
Effect size |
Leptin (ng/ml) [geometric mean (95% CI)] |
Padjusted1 |
Padjusted1+BMI |
||
|---|---|---|---|---|---|---|---|---|
aa |
aA |
AA |
||||||
PCSK1 |
32.4 (10.3) |
rs261967 |
0.4% |
4.6 (4.2-5.0) |
5.3 (5.0-5.7) |
5.2 (4.7-5.8) |
0.003 |
0.419 |
MC4R |
3.8 (3.45) |
rs2331841 |
0.3% |
4.7 (4.5-5.0) |
5.5 (5.1-5.8) |
5.6 (4.7-6.6) |
0.004 |
0.694 |
FTO |
543.45 (119.7) |
rs1558902 |
0.3% |
4.9 (4.6-5.1) |
5.7 (5.2-6.3) |
6.0 (4.1-8.7) |
0.008 |
0.216 |
MAP2K5 |
6.0 (5.6) |
rs4776970 |
0.3% |
4.8 (4.5-5.1) |
5.4 (5.0-5.8) |
6.0(5.2-7.0) |
0.013 |
0.151 |
GNPDA2 |
6.6 (6.65) |
rs16858082 |
0.2% |
4.7 (4.4-5.1) |
5.2 (4.9-5.6) |
5.4 (4.8-6.2) |
0.019 |
0.838 |
BDNF |
12.4 (5.9) |
rs2030323 |
0.2% |
4.8 (4.3-5.3) |
4.9 (4.6-5.2) |
5.5 (5.0-5.9) |
0.053 |
0.235 |
GPSleptin |
/ |
six leptin-related loci |
1.6% |
Tertile1 (0-3 risk alleles) (n = 1373) |
Tertile2 (4 risk alleles) (n = 828) |
Tertile3 (5-9 risk alleles) (n = 1130) |
2.8E-6 |
0.218 |
4.4 (4.1-4.8) |
5.1 (4.7-5.6) |
5.8 (5.4-6.3) |
||||||
Abbreviation: PCSK1, proprotein convertase subtilisin/kexin type 1; MC4R, melanocortin 4 receptor; FTO, fat mass and obesity associated; MAP2K5, mitogen-activated protein kinase 5; GNPDA2, glucosamine-6-phosphate deaminase 2; BDNF, brain-derived neurotrophic factor and GPSleptin, sum gene score of six leptin-related loci.
*Data were expressed as mean (median in all tissues and cells), from http://biogps.org.
Padjusted1 means the analysis of covariance in the general linear model after adjusting for gender, age, Tanner stage and residence.
Padjusted1+BMI: further adjusted for BMI.
Note: Values in bold are significant at P < 0.05.
Table 5: Associations of the 12 SNPs and GPSs with obesity-related and other cardio-metabolic traits
Gene |
SNP |
WC (cm) |
Percent body fat |
UAC (cm) |
SBP (mmHg) |
DBP (mmHg) |
TC (mmol/L) |
TG (mmol/L)# |
LDL-C (mmol/L) |
HDL-C (mmol/L) |
FBG (mmol/L) |
Insulin (mU/L)# |
HOMA-IR# |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
FTO |
rs1558902 |
β |
1.887 |
1.150 |
0.857 |
1.491 |
0.964 |
-0.020 |
0.007 |
-0.007 |
-0.011 |
-0.011 |
0.058 |
0.057 |
P |
6.3E-6 |
1.5E-4 |
2.5E-8 |
0.001 |
0.005 |
0.496 |
0.683 |
0.807 |
0.322 |
0.599 |
0.031 |
0.044 |
||
MC4R |
rs2331841 |
β |
1.183 |
0.847 |
0.482 |
0.588 |
0.300 |
-0.007 |
0.026 |
-0.001 |
-0.012 |
0.046 |
0.064 |
0.072 |
P |
2.3E-4 |
2.8E-4 |
4.7E-5 |
0.096 |
0.260 |
0.757 |
0.045 |
0.946 |
0.184 |
0.004 |
0.002 |
0.001 |
||
GNPDA2 |
rs16858082 |
β |
0.974 |
0.665 |
0.265 |
0.852 |
0.421 |
-0.048 |
0.005 |
-0.030 |
-0.021 |
0.017 |
0.056 |
0.059 |
P |
0.001 |
0.001 |
0.011 |
0.006 |
0.074 |
0.017 |
0.643 |
0.109 |
0.006 |
0.231 |
0.002 |
0.002 |
||
SEC16B |
rs516636 |
β |
0.972 |
0.493 |
0.279 |
0.575 |
0.148 |
0.003 |
0.015 |
0.008 |
-0.012 |
0.011 |
0.039 |
0.041 |
P |
0.004 |
0.044 |
0.025 |
0.121 |
0.595 |
0.884 |
0.268 |
0.715 |
0.180 |
0.518 |
0.073 |
0.068 |
||
PCSK1 |
rs261967 |
β |
0.790 |
0.339 |
0.287 |
0.406 |
0.508 |
-0.042 |
0.009 |
-0.024 |
-0.015 |
-0.014 |
0.042 |
0.041 |
P |
0.005 |
0.091 |
0.005 |
0.186 |
0.028 |
0.031 |
0.414 |
0.189 |
0.049 |
0.292 |
0.020 |
0.028 |
||
MAP2K5 |
rs4776970 |
β |
0.982 |
0.704 |
0.338 |
0.709 |
0.424 |
4.5E-4 |
0.030 |
0.001 |
-0.023 |
-0.015 |
0.035 |
0.032 |
P |
0.002 |
0.002 |
0.004 |
0.043 |
0.107 |
0.984 |
0.017 |
0.978 |
0.010 |
0.321 |
0.086 |
0.140 |
||
ITIH4 |
rs2535633 |
β |
0.586 |
0.430 |
0.165 |
0.266 |
0.109 |
0.033 |
0.019 |
0.037 |
-0.006 |
-0.001 |
0.036 |
0.035 |
P |
0.035 |
0.033 |
0.107 |
0.384 |
0.634 |
0.092 |
0.084 |
0.032 |
0.396 |
0.939 |
0.045 |
0.060 |
||
BDNF |
rs2030323 |
β |
0.386 |
0.343 |
0.191 |
-0.284 |
-0.262 |
0.016 |
0.006 |
0.018 |
-0.004 |
-0.004 |
0.029 |
0.027 |
P |
0.163 |
0.087 |
0.062 |
0.351 |
0.254 |
0.418 |
0.615 |
0.326 |
0.600 |
0.763 |
0,105 |
0.141 |
||
ADCY3/RBJ |
rs6545814 |
β |
0.599 |
0.367 |
0.177 |
0.385 |
0.395 |
-0.007 |
0.002 |
0.009 |
-0.008 |
-0.019 |
0.025 |
0.023 |
P |
0.032 |
0.070 |
0.086 |
0.211 |
0.088 |
0.728 |
0.865 |
0.625 |
0.290 |
0.160 |
0.157 |
0.226 |
||
KCNQ1 |
rs2237892 |
β |
0.271 |
0.135 |
0.048 |
-0.231 |
0.038 |
-0.041 |
0.020 |
-0.027 |
-0.028 |
0.016 |
0.008 |
0.011 |
P |
0.356 |
0.527 |
0.659 |
0.475 |
0.876 |
0.046 |
0.094 |
0.159 |
0.001 |
0.257 |
0.687 |
0.587 |
||
PAX6 |
rs652722 |
β |
-0.143 |
-0.044 |
-0.132 |
0.127 |
0.053 |
-0.016 |
-0.014 |
-0.010 |
-0.004 |
-0.017 |
-0.010 |
-0.013 |
P |
0.622 |
0.834 |
0.218 |
0.690 |
0.825 |
0.439 |
0.223 |
0.584 |
0.647 |
0.225 |
0.601 |
0.511 |
||
GP2 |
rs12597579 |
β |
0.474 |
0.164 |
0.119 |
0.168 |
0.183 |
0.016 |
0.010 |
0.009 |
-3.5E-4 |
0.002 |
-0.004 |
-0.003 |
P |
0.124 |
0.462 |
0.294 |
0.619 |
0.472 |
0.471 |
0.396 |
0.662 |
0.967 |
0.921 |
0.857 |
0.869 |
||
GPSall |
all 12 loci |
β |
0.683 |
0.418 |
0.226 |
0.397 |
0.26 |
-0.009 |
0.011 |
1.2E-4 |
-0.013 |
0.001 |
0.033 |
0.033 |
P |
1.4E-14 |
6.1E-11 |
3.5E-12 |
4.7E-5 |
4.1E-4 |
0.178 |
0.002 |
0.983 |
2.7E-7 |
0.735 |
7.3E-9 |
1.7E-8 |
||
EV |
1.3% |
1.2% |
1.0% |
0.4% |
0.3% |
< 0.01% |
0.3% |
< 0.01% |
0.8% |
< 0.01% |
0.9% |
0.8% |
||
GPSleptin |
six leptin-related loci |
β |
0.972 |
0.608 |
0.352 |
0.523 |
0.315 |
-0.017 |
0.013 |
-0.007 |
-0.015 |
0.004 |
0.046 |
0.047 |
P |
2.3E-13 |
3.4E-11 |
4.1E-14 |
1.8E-4 |
0.003 |
0.050 |
0.008 |
0.392 |
2.3E-5 |
0.529 |
1.2E-8 |
2.9E-8 |
||
EV |
1.3% |
1.2% |
1.2% |
0.4% |
0.3% |
< 0.01% |
0.2% |
< 0.01% |
0.6% |
< 0.01% |
1.0% |
1.0% |
β means linear regression coefficients adjusted for gender, age, Tanner stage and residence.
Abbreviation: BMI, body mass index; WC, waist circumference; UAC, upper arm circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; HOMA-IR, homeostasis model assessment for insulin resistance; EV, explained variation; FTO, fat mass and obesity associated; MC4R, melanocortin 4 receptor; GNPDA2, glucosamine-6-phosphate deaminase 2; SEC16B, SEC16 homolog B, endoplasmic reticulum export factor; PCSK1, proprotein convertase subtilisin/kexin type 1; MAP2K5, mitogen-activated protein kinase 5; ITIH4, inter-alpha-trypsin inhibitor heavy chain family, member 4; BDNF, brain-derived neurotrophic factor; ADCY3/RBJ, adenylate cyclase 3; KCNQ1, potassium channel, voltage gated KQT-like subfamily Q, member 1; PAX6, paired box 6; GP2, glycoprotein 2; GPSall, sum gene score of all 12 loci and GPSleptin, sum gene score of six leptin-related loci.
# Skewed distributions were natural logarithmically (ln) transformed.
Note: Values in bold are significant at P < 0.004 after Bonferroni correction.
Interestingly, as listed in Table 4, all the genes at these leptin-increasing loci were found to be expressed in CNS.
Best fitting model for the association between SNPs/gene score, leptin level and BMI
Since leptin was the only adipokine revealing Bonferroni-significant association with the SNPs, we therefore focused on the associations between leptin levels, leptin-related SNPs/GPSleptin and BMI to test which triangular association model fitted the best: mediation, pleiotropy or moderation.
First, we tested whether the associations between SNPs/gene score and leptin levels were independent of BMI. As shown in Table 4, these significant relationships were ablated after further adjustment for BMI, indicating that the associations of these obesity-related loci with leptin were likely not due to gene pleiotropy. Next, since all the genes at these leptin-increasing loci were found to be expressed in the CNS (Table 4), we tested if leptin plays a mediation role in genetic predisposition to polygenic obesity. Thus, leptin levels were added in to a multiple regression model to predict BMI from each of the selected six leptin-related SNPs plus their addictive score i.e. GPSleptin. As listed in Figure 1, the relationships were attenuated between FTO, MC4R, GNPDA2 and BMI (β from a model without leptin: 0.831, 0.486, and 0.384, respectively, see Table 2, and β from a model including leptin: 0.418, 0.188, and 0.170, respectively and βΔ 0.413, 0.298, and 0.214, respectively), and the relationships were lost between PCSK1, MAP2K5, BDNF and BMI (β from a model without leptin: 0.311, 0.347 and 0.216, respectively, β from a model including leptin: 0.086, 0.051 and 0.048, respectively, and β Δ 0.225, 0.296 and 0.168, respectively). Moreover, the Sobel test confirmed significant mediation of the association between this genetic risk and BMI by leptin levels (all P < 0.05). As expected, we also found stronger mediation effect of leptin on the relationship between gene scores consisting of all six leptin-related loci (GPSleptin) and BMI than any single SNP (P < 0.001) (Figure 1). The mediation effect of incremental leptin on the GPSleptin-BMI association was estimated at 63.8%, with a significant total indirect effect of 0.245.
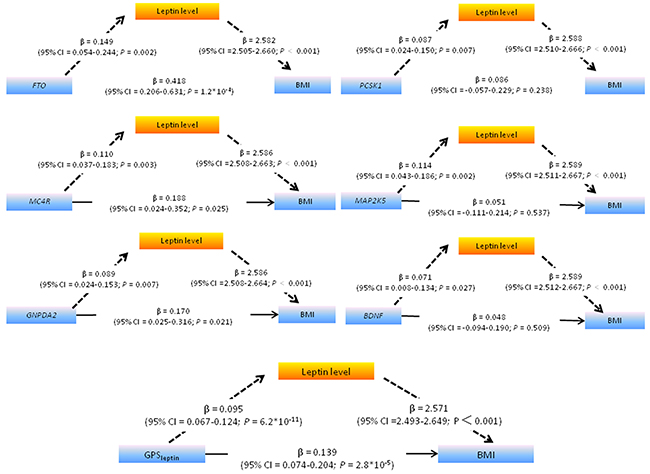
Figure 1: Path diagram showing that leptin significantly mediates the association between leptin-related SNPs /GPSleptin (including six leptin-related SNPs) and BMI. The path diagram shows the simple association between the six leptin-related loci FTO, MC4R, GNPDA2, PCSK1, MAP2K5, BDNF and GPSleptin and ln-leptin (β, 0.149, 0.110, 0.089, 0.087, 0.114, 0.071,and 0.095 respectively), the association between the FTO, MC4R, GNPDA2, PCSK1, MAP2K5, BDNF and GPSleptin and BMI adjusted for leptin (β, 0.418, 0.188, 0.170, 0.086, 0.051, 0.048, and 0.139, respectively), and the association between leptin and BMI adjusted for the individual SNP or GPSleptin (β, 2.582, 2.586, 2.586, 2.588, 2.589, 2.589 and 2.571, respectively). The simple association between the FTO, MC4R, GNPDA2, PCSK1, MAP2K5, GPSleptin and BMI (β, 0.831, 0.486, 0.384, 0.311, 0.347, 0.216 and 0.384, respectively) was significantly higher than the association between the individual SNP,GPSleptin and BMI adjusted for leptin (β Δ 0.413, 0.298, 0.214, 0.225, 0.296, 0.168 and 0.245, respectively), indicating that leptin mediated part of the association. The Sobel test confirmed that leptin significantly mediated the association between the SNPs and BMI (P = 0.002, P = 0.003, P = 0.007, P = 0.008, P = 0.003, P = 0.027 and P < 0.001, respectively). All the linear regression coefficients (β) were adjusted for gender, age, Tanner stage and residence.
Functional interactions between leptin, leptin receptor and genes associated with leptin and obesity
To further explore the interactions of the six leptin-increasing loci and LEP or LEPR, the functional interactions among these genes were predicted via the STRING database v10. As listed in Figure 2, the enrichment indicates that these genes as a group are at least partially biologically connected (clustering coefficient: 0.783). In addition, as expected, four of the six genes at these leptin-increasing loci (PCSK1, MC4R, FTO and BDNF) have interactions with LEP, while BDNF has an interaction with LEPR. Unlike those loci are parts or have well-established connections with the leptin pathway in literature, the interactions between the two loci in/near MAP2K5 and GNPDA2 and leptin were firstly found in our research.
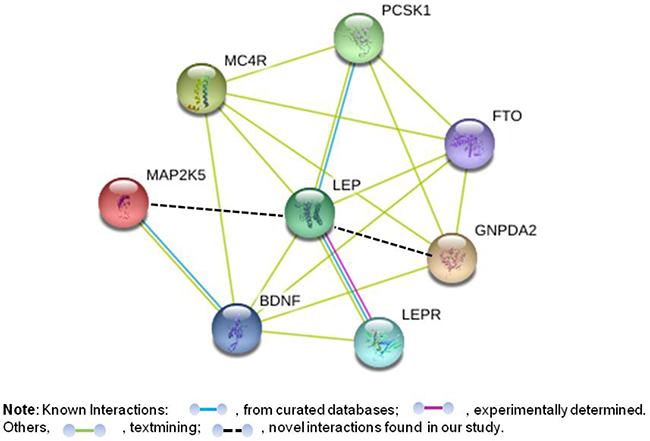
Figure 2: Possible interaction between the genes with at the six leptin-associated loci plus LEP and LEPR. This is a prediction of the interaction between the key proteins, including PCSK1, proprotein convertase subtilisin/kexin type 1; MC4R, melanocortin 4 receptor; FTO, fat mass and obesity associated; MAP2K5, mitogen-activated protein kinase 5; GNPDA2, glucosamine-6-phosphate deaminase 2; BDNF, brain-derived neurotrophic factor; LEP, leptin and LEPR, leptin receptor. The prediction was conducted by using a STRING interaction network.
Associations of obesity predisposing genes with cardio-metabolic traits
For other cardiometabolic traits (Table 5), the obesity predisposing locus, FTO-rs1558902 yielded significant association with systolic blood pressure (SBP) (P = 0.001), while the risk alleles of SNPs in/near GNPDA2, PCSK1 and MAP2K5 yielded nominal association with blood pressures (P < 0.05). In addition, the risk alleles of SNPs in/near GNPDA2, MAP2K5, PCSK1 and MC4R revealed association with serum lipid levels (0.006 ≤ P ≤ 0.049). Moreover, the risk allele of MC4R-rs2331841 showed significant association with glucose, insulin and HOMA-IR levels (0.001 ≤ P ≤ 0.004), while the risk alleles of SNPs in/near GNPDA2, PCSK1, FTO and ITIH4 were nominally associated with glucose/insulin traits (P < 0.05). Meanwhile, both GPSall and GPSleptin yield significant association with blood pressures, HDL-C, insulin and HOMA-IR at Bonferroni- corrected levels.
When further adjusted for BMI (Table 6), the obesity predisposing loci, GNPDA2-rs16858082 (P = 0.016) and PCSK1-rs261967 (P = 0.026) were still nominally associated with total cholesterol (TC) levels, and MC4R-rs2331841 remained correlated with fasting glucose (FBG) levels (P = 0.012). Interestingly, the adult obesity-related locus KCNQ1-rs2237892 which was not replicated the association with childhood obesity in our study yield significant association with HDL-C (P = 3.7×10−4) levels independent of BMI.
Table 6: Associations of the 12 SNPs and GPSs with obesity-related and other cardio-metabolic traits after adjusting for BMI
Gene |
SNP |
WC (cm) |
Percent body fat |
UAC (cm) |
SBP (mmHg) |
DBP (mmHg) |
TC (mmol/L) |
TG (mmol/L)# |
LDL-C (mmol/L) |
HDL-C (mmol/L) |
FBG (mmol/L) |
Insulin (mU/L)# |
HOMA-IR# |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
FTO |
rs1558902 |
β |
-0.084 |
-0.053 |
0.138 |
0.301 |
0.257 |
-0.023 |
-0.022 |
-0.024 |
0.012 |
-0.022 |
-0.019 |
-0.022 |
P |
0.561 |
0.746 |
0.017 |
0.451 |
0.419 |
0.444 |
0.165 |
0.380 |
0.241 |
0.273 |
0.407 |
0.359 |
||
MC4R |
rs2331841 |
β |
0.052 |
0.155 |
0.061 |
-0.096 |
-0.121 |
-0.009 |
0.010 |
-0.012 |
-0.002 |
0.040 |
0.021 |
0.028 |
P |
0.640 |
0.220 |
0.171 |
0.754 |
0.620 |
0.684 |
0.423 |
0.572 |
0.852 |
0.012 |
0.222 |
0.123 |
||
GNPDA2 |
rs16858082 |
β |
0.036 |
0.038 |
-0.066 |
0.306 |
0.103 |
-0.048 |
-0.008 |
-0.037 |
-0.010 |
0.012 |
0.023 |
0.025 |
P |
0.712 |
0.735 |
0.091 |
0.258 |
0.634 |
0.016 |
0.434 |
0.046 |
0.144 |
0.396 |
0.128 |
0.115 |
||
SEC16B |
rs516636 |
β |
0.081 |
-0.059 |
-0.043 |
0.067 |
-0.154 |
0.002 |
0.003 |
0.001 |
-0.002 |
0.005 |
0.007 |
0.009 |
P |
0.490 |
0.656 |
0.360 |
0.833 |
0.547 |
0.932 |
0.833 |
0.981 |
0.789 |
0.749 |
0.696 |
0.645 |
||
PCSK1 |
rs261967 |
β |
0.049 |
-0.135 |
0.018 |
-0.040 |
0.245 |
-0.044 |
-0.001 |
-0.031 |
-0.006 |
-0.018 |
0.013 |
0.012 |
P |
0.609 |
0.221 |
0.635 |
0.879 |
0.247 |
0.026 |
0.896 |
0.089 |
0.365 |
0.177 |
0.369 |
0.452 |
||
MAP2K5 |
rs4776970 |
β |
0.155 |
0.175 |
0.057 |
0.247 |
0.153 |
-4.0E-4 |
0.019 |
-0.006 |
-0.013 |
-0.020 |
0.005 |
0.001 |
P |
0.158 |
0.162 |
0.196 |
0.415 |
0.525 |
0.986 |
0.107 |
0.765 |
0.102 |
0.194 |
0.759 |
0.956 |
||
ITIH4 |
rs2535633 |
β |
0.028 |
0.098 |
-0.026 |
-0.058 |
-0.083 |
0.032 |
0.012 |
0.034 |
-1.7E-4 |
-0.004 |
0.015 |
0.014 |
P |
0.771 |
0.373 |
0.496 |
0.826 |
0.693 |
0.101 |
0.267 |
0.059 |
0.980 |
0.777 |
0.306 |
0.371 |
||
BDNF |
rs2030323 |
β |
-0.105 |
-0.04 |
0.007 |
-0.587 |
-0.434 |
0.016 |
-0.003 |
0.013 |
0.003 |
-0.008 |
0.009 |
0.007 |
P |
0.272 |
0.713 |
0.850 |
0.026 |
0.039 |
0.417 |
0.800 |
0.455 |
0.662 |
0.578 |
0.527 |
0.641 |
||
ADCY3/RBJ |
rs6545814 |
β |
0.121 |
0.07 |
-0.012 |
0.060 |
0.202 |
-0.007 |
-0.006 |
0.004 |
-0.002 |
-0.022 |
0.006 |
0.002 |
P |
0.211 |
0.528 |
0.762 |
0.822 |
0.341 |
0.707 |
0.568 |
0.815 |
0.811 |
0.109 |
0.704 |
0.876 |
||
KCNQ1 |
rs2237892 |
β |
0.114 |
0.028 |
0.003 |
-0.295 |
0.002 |
-0.042 |
0.018 |
-0.028 |
-0.026 |
0.015 |
0.003 |
0.006 |
P |
0.262 |
0.812 |
0.937 |
0.291 |
0.994 |
0.044 |
0.102 |
0.142 |
3.7E-4 |
0.291 |
0.852 |
0.735 |
||
PAX6 |
rs652722 |
β |
0.106 |
0.077 |
-0.028 |
0.283 |
0.155 |
-0.015 |
-0.010 |
-0.008 |
-0.007 |
-0.016 |
0.001 |
-0.002 |
P |
0.290 |
0.504 |
0.481 |
0.304 |
0.481 |
0.452 |
0.365 |
0.680 |
0.323 |
0.271 |
0.948 |
0.916 |
||
GP2 |
rs12597579 |
β |
0.119 |
-0.108 |
-0.003 |
-0.045 |
0.047 |
0.015 |
0.005 |
0.005 |
0.004 |
-2.8E-4 |
-0.017 |
-0.017 |
P |
0.264 |
0.375 |
0.948 |
0.878 |
0.839 |
0.492 |
0.645 |
0.794 |
0.619 |
0.985 |
0.315 |
0.330 |
||
GPSall |
all 12 loci |
β |
0.057 |
0.023 |
0.001 |
0.033 |
0.04 |
-0.01 |
0.002 |
-0.005 |
-0.005 |
-0.002 |
0.009 |
0.009 |
P |
0.065 |
0.512 |
0.939 |
0.702 |
0.562 |
0.139 |
0.540 |
0.389 |
0.019 |
0.634 |
0.050 |
0.072 |
||
GPSleptin |
six leptin- related loci |
β |
0.017 |
0.018 |
0.022 |
-0.024 |
-0.006 |
-0.019 |
1.7E-4 |
-0.015 |
-0.004 |
-0.001 |
0.012 |
0.012 |
P |
0.696 |
0.723 |
0.204 |
0.843 |
0.952 |
0.036 |
0.972 |
0.064 |
0.242 |
0.826 |
0.081 |
0.099 |
β means linear regression coefficients adjusted for gender, age, Tanner stage, residence and BMI.
Abbreviation: BMI, body mass index; WC, waist circumference; UAC, upper arm circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; HOMA-IR, homeostasis model assessment for insulin resistance; FTO, fat mass and obesity associated; MC4R, melanocortin 4 receptor; GNPDA2, glucosamine-6-phosphate deaminase 2; SEC16B, SEC16 homolog B, endoplasmic reticulum export factor; PCSK1, proprotein convertase subtilisin/kexin type 1; MAP2K5, mitogen-activated protein kinase 5; ITIH4, inter-alpha-trypsin inhibitor heavy chain family, member 4; BDNF, brain-derived neurotrophic factor; ADCY3/RBJ, adenylate cyclase 3; KCNQ1, potassium channel, voltage gated KQT-like subfamily Q, member 1; PAX6, paired box 6; GP2, glycoprotein 2; GPSall, sum gene score of all 12 loci and GPSleptin, sum gene score of six leptin-related loci.
# Skewed distributions were natural logarithmically (ln) transformed.
Note: Values in bold are significant at P < 0.004 after Bonferroni correction.
DISCUSSION
This is the first study to systematically investigate the associations of twelve obesity/BMI-related loci, which had been previously shown to be evident in East Asian adults [12-14], with adipokine profiles and risk factors for obesity, hypertension, dyslipidemia, and diabetes in a large cohort of Chinese school-aged children. We successfully replicated the association with childhood obesity for eight SNPs in/near FTO, MC4R, GNPDA2, PCSK1, SEC16B, MAP2K5, ITIH4 and BDNF, with odds ratio ranging from 1.13 to 1.42, implicating the transferability of susceptibility between Chinese and Europeans both in adulthood and childhood [12-14]. Notably, of these associated loci, six SNPs yielded significant or nominal association with leptin levels except for SEC16B and ITIH4. Moreover, the cumulated genetic scores i.e. GPSall and GPSleptin showed much stronger association with increasing leptin levels and metabolic traits. The overall variation in adiposity explained by the 6 leptin related loci and 12 BMI related loci were 1.5% and 1.4%, which is an improvement over the previously reported value of ~1.2% in East Asian adults including 22 BMI-associated loci [12]. For cardio-metabolic risk factors, the risk allele of KCNQ1-rs2237892 yield significant association with decreasing HDL-C levels and MC4R-rs2331841 showed association with increasing FBG levels independent of BMI. These cross-phenotype associations may either reflect pleiotropy, where a gene product influences multiple metabolic traits, and/or mediation effects, where one phenotype is causally related to a second phenotype [21].
One of the key novel findings of this study is the association of the BMI-increasing loci with increasing leptin levels. Leptin is a peptide hormone produced by adipose tissue which plays a role in regulating appetite, thermogenesis, lipid oxidation and insulin sensitivity, with each of these effects attenuated in obese individuals with leptin resistance, particularly in the CNS [19]. The associations between BMI-increasing loci and increasing leptin levels may partly explain the definitive identification of the specific mechanisms about these loci influence BMI and obesity. Firstly, six of these loci (in/near PCSK1, MC4R, FTO, MAP2K5, GNPDA2 and BDNF) showed a directionally concordant association with increasing leptin levels as well as increasing adiposity. Second, the predicted functional interactions among these genes and LEP/LEPR indicate that these genes as a group are at least partially biologically connected. In addition, mediation analysis revealed that leptin levels significantly mediated the association observed for genetic predisposition to adiposity. It should be noted that all the six leptin-increasing loci highlighted genes are expressed in and/or known to act in the brain (and several particularly so in the hypothalamus) [17, 22-24]. Therefore, these findings suggest that as is seen in monogenic forms of obesity, inherited variations in these leptin-increasing loci influence common obesity may through their effects in the CNS, particularly pointing to neuronal “leptin resistance”, thus, highlighting a possible neuronal influence on body weight regulation in children comparable to previous reports in adults [17].
In addition to leptin, we also found nominal association between adiponectin levels and three hypothalamic leptin–melanocortin pathway-related genes (MC4R, BDNF and PCSK1). Adiponectin is an insulin-sensitizer expressed in adipose tissue. A decrease in the circulating levels in obesity has been shown to contribute to the development of insulin resistance, T2D and metabolic syndrome [25]. Besides its peripheral actions, adiponectin has also been demonstrated to play an important role in the CNS: being present in the cerebral spinal fluid and enhancing AMP-activated protein kinase activity in the arcuate hypothalamus to stimulate food intake and decreases energy expenditure [26]. Thus, our findings suggest that the pathophysiology of obesity signaling in the CNS may not only influencing adiposity but also play an important role in the regulation of adiponectin levels.
We did not find any significant relationship between these BMI-increasing loci and other three important adipokines: resistin [19], FGF21 [19] and RBP4 [20], which can also function via the CNS. However, unlike adipokine leptin and adiponectin which are mainly produced by adipocytes, resistin is mainly secreted by mononuclear cells in humans, promoting both inflammation and IR [27]; FGF21 is primarily produced by liver and is associated with beneficial metabolic characteristics, including reduced body weight and hepatic steatosis, as well as improved insulin sensitivity and lipid/glucose profiles [28]; while RBP4 is secreted primarily by liver and adipocytes, playing an important role in the development of obesity and other metabolic abnormalities by inducing adipose tissue inflammation and promoted systemic IR [29]. Thus, the differing source of production may provide an explanation for the lack of association of the three adipokines with the BMI-related loci.
Among the six leptin-associated loci, three genes (MC4R, BDNF, and PCSK1) are known to be involved in the hypothalamic leptin–melanocortin pathway [9, 17, 30], thus it is not surprising that those three loci are found to be associated with obesity-related traits and leptin levels in our pediatric setting. Indeed, our findings suggest a potential effect of MC4R on glucose homeostasis beyond obesity. Given the fact that MC4R encodes the melanocortin-4 receptor in the leptin-melanocortin signaling pathway [17, 30], wherein leptin up-regulates anorexigenic neuropeptides such as alpha-melanocyte-stimulating hormone, which acts on the melanocortin-4 receptor to control energy intake and metabolic regulation, our findings, therefore, implicate that the role of MC4R in controlling glucose metabolism may also depend on leptin pathways.
FTO is the first GWAS-identified obesity-susceptibility locus; indeed, like many of the other replication efforts, FTO shows the strongest association with BMI in our pediatric cohort and accounts for the largest proportion of the variance (0.5%), which is higher than its estimated by East Asian adults (~0.15%) [14]. However, unlike the above-noted three genes with possible function through hypothalamic leptin–melanocortin pathway, the process by FTO impacts on obesity pathogenesis has remained elusive [17]. Given that available experimental evidence has shown that FTO modulates leptin receptor localization within neurons to control food intake and adiposity [31], our finding provides an additional insight into the linkage between FTO, leptin and adiposity, and obesity-related characters.
Notably, among the six leptin-associated loci, the two in/near MAP2K5 and GNPDA2, have not been reported before in this context. In consideration of the limited knowledge of the associations, particularly in the early years, our results highlight the need to further investigate the complex relationship between leptin and MAP2K5 and GNPDA2.
When compared the accumulated genetic effects by above six leptin-associated loci (GPSleptin) with all twelve selected loci (GPSall), GPSleptin showed equal or even more significant correlations with adiposity-traits than GPSall, thus, the six leptin-increasing SNPs may be used as a novel set of biomarkers to identify the risk of obesity. Since all the leptin-related loci were expressed and/or functioned in CNS, therefore, suggesting a possible central role of the six loci in the process of body weight regulation, and providing a potential therapeutic targets to prevent obesity.
Besides the six leptin-increasing SNPs, we also replicated the associations of other three loci in SEC16B, ITIH4 and ADCY3/RBJ with adiposity traits, but no relationship between the three loci and leptin levels was observed, suggesting the possible different mechanisms between these loci underlying the body weight regulation, and the specific mechanisms through which these loci affect BMI and obesity require further study.
In addition, despite our study being sufficiently powered, we failed to confirm the association with obesity for three novel identified adult BMI loci from East ancestry (KCNQ1-rs2237892, PAX6-rs652722 and GP2-rs12597579) in Chinese children [12, 14]. The reason for the results deviating from what is seen in adults may owe to the genes operating differently in childhood compared to adulthood [9]. Thus, separating genetic component that is exclusively associated with childhood obesity remains to be explored.
However, we found a strong connection between the risk allele of KCNQ1-rs2237892 and blood lipids, especially with the HDL-C levels. In line with our results, a study in a middle-aged Chinese Han population also found subjects with CC genotype in another locus rs2283228 in KCNQ1 (r2CEU = 0.867 with rs2237892) had lower levels of HDL-C [32]. Given that patients with obesity and T2D are more likely to have lower levels of HDL-C and large prospective studies have identified HDL-C as a strong, independent, inverse predictor of risk of CVD [33], the strong association of KCNQ1 variant with HDL-C in our pediatric population may provide a novel insight of the link between KCNQ1 and early risk in the pathogenesis of T2D and CVD.
MATERIALS AND METHODS
Population
Subjects were recruited via a cross-sectional population based survey: the Beijing Child and Adolescent Metabolic Syndrome (BCAMS) study [27]. This study evaluated the prevalence of obesity and related metabolic abnormalities (hypertension, hyperglycemia and dyslipidemia) among a representative sample of Beijing school-aged children and adolescents (n = 19593, ages 6 to18 years, 50% male) between April and October 2004. Within this cohort, 4500 subjects were identified as having one or more of the following disorders: Being overweight defined by BMI percentile, increased TC ≥ 5.2 (mmol/L), TG ≥ 1.7 (mmol/L) or FBG ≥ 5.6 (mmol/L) based on finger capillary blood tests. Age- and sex-specific BMI percentiles, according to the Working Group for Obesity in China, were used to define overweight (85th) and obesity (95th) [34]. Totally 3,506 subjects, including 1,024 normal controls, agreed to complete further medical examination, thus were included for this study. The BCAMS study was approved by the ethics committee of the Capital Institute of Pediatrics and all the methods were performed in accordance with relevant guidelines and regulations. Signed informed consents were obtained from all participants and/or their parents or guardians through all the study processes.
Phenotyping
Subjects’ height, waist circumference (WC), weight, percent body fat, upper arm circumference (UAC), systolic blood pressure and diastolic blood pressure (SBP and DBP) were measured by trained recruiters with standard methods. Height was measured to the nearest 0.1 cm using a portable stadiometer. WC was measured midway between the lowest rib and the top of the iliac crest. Weight and percent body fat was measured to the nearest 0.1 kg using a TANITA Body Composition Analyzer (Model TBF–300A). UAC was measured at the mid-point between the tip of acromion process and olecranon process of the right upper arm. Measurements of right arm SBP and DBP were performed 3 times at 10 minutes apart and the mean values of the latter two measurements were recorded. BMI was calculated as weight divided by height squared.
Venous blood samples were collected after an overnight (≥ 10 h) fast. The samples were centrifuged, and immediately frozen for future analysis of hormones. Serum lipids (enzymatic methods) and FBG (glucose oxidase method) were assayed using the Hitachi 7060 C automatic biochemistry analysis system. Insulin, leptin and adiponectin were measured by sandwich enzyme-linked immunosorbent assay (ELISA) developed in the Key Laboratory of Endocrinology, Peking Union Medical College Hospital. The insulin assay had an inter-assay coefficient of variation (CV) of < 9.0% and no cross-reactivity to proinsulin (< 0.05%) [34]. Insulin resistance index was calculated by homeostasis model assessment as [HOMA-IR= (fasting insulin IU/L) ×; (FBG mmol/L)/22.5].The intra-assay and inter-assay CVs for leptin were < 7.4% and < 9.3%, respectively and < 5.4% and < 8.5% for adiponectin, respectively [35, 36]. Serum resistin was measured using the ELISA kit by Phoenix Pharmaceuticals Inc. The intra-assay and inter-assay CVs of this assay were < 5.2 and < 10.1%, respectively [27]. RBP4 was measured by commercial ELISA kits (Dou set, R&D Systems, Minneapolis, MN, USA) with intra- and inter-assay CVs of 6.2% and 8.5%, respectively. FGF21 was measured by an ELISA kit (Phoenix Pharmaceuticals, Burlingame, CA, USA) with intra- and inter-assay CVs of < 6.0% and < 8.6%, respectively [37]. All samples were tested in duplicate and blinded.
SNP selection and genotyping
Thirteen SNPs were selected from GWAS reports of adult obesity in East Asian ancestry populations [12-14]. Among them, eight SNPs: FTO-rs1558902, MC4R-rs2331841, BDNF-rs2030323, ADCY3/RBJ-rs6545814, GIPR/QPCTL-rs11671664, MAP2K5-rs4776970, GNPDA2-rs16858082, and SEC16B-rs516636 were initially identified in European-ancestry populations which went on to satisfy the genome-wide significance threshold in East Asian-ancestry subjects [12-14], while the five other loci, KCNQ1-rs2237892, PCSK1-rs261967, PAX6-rs652722, GP2-rs12597579, and ITIH4-rs2535633 were first identified in adult BMI studies in East Asians [12, 14].
Genomic DNA was isolated from peripheral white blood cells using the QIAamp DNA Blood Midi Kits (Qiagen). All SNPs were genotyped on the Sequenom Mass Array iPLEX genotyping platform in BioMiao Biological Technology (Beijing) Co, Ltd. [38] Repeated control samples were present in each genotyping plate with the concordance rate being 100%. SNPs were excluded if they had genotyping efficiency less than 0.95, or a Hardy-Weinberg equilibrium P < 0.0042 (0.05/12). Except for GIPR/QPCTL-rs11671664, twelve SNPs were in Hardy-Weinberg equilibrium, thus incorporated in the present study.
Statistical analysis
Analyses were performed using Statistical Package for Social Sciences (SPSS) 19.0 for Windows. Hardy-Weinberg equilibrium was assessed using the chi-squared test in controls. By applying Bonferroni correction, a P-value below 0.0042 (0.05 divided by 12 SNPs) was considered significant, while a P-value between 0.05 and 0.0042 was considered nominally significant. All skewed distributions were naturally logarithmically transformed for analysis. Continuous data are presented as mean ± SD or geometric mean (95% CI). During the initial step, one-way ANOVA was utilized for continuous variables in relation to obesity. The adjustment for confounding factors was performed using the analysis of covariance in the general linear model (GLM). A score of 0, 1 or 2 was assigned to genotypes of associated SNPs according to the number of risk alleles in the additive model. To evaluate the combined effect of the SNPs on obesity and metabolic phenotypes, we generated two different genetic predisposition scores (GPSs) (GPSall: consisting of all twelve SNPs; GPSleptin: all leptin-related SNPs) by summing the number of risk alleles that each subject carried at each SNP. Logistic or linear regression analysis was then applied to examine the associations between each SNP or category of GPS tertile and obesity risk, or related phenotype, with adjustment for confounders. We obtained the pairwise distance and LD (r2) data from the International HapMap Project and derived the predicted associations among proteins using the publically available STRING database (http://string-db.org). The post-hoc power of the study was estimated using G*power software program [39]. The sample sizes revealed > 99% power to detect a significant association (α < 0.05), given an effect size index of 0.1 (corresponds to a “weak” gene effect). The Sobel test [40] was used to assess whether leptin significantly mediated the association between the leptin-related SNPs, gene score and BMI.
CONCLUSIONS
In summary, in our sample of Chinese children, we replicated eight adult East Asian obesity-related loci (in/near FTO, MC4R, GNPDA2, PCSK1, SEC16B, MAP2K5, ITIH4 and BDNF) in the same direction of effect as previous reports in adults. Moreover, the association of the harboring brain-expressed loci with leptin highlights a neuronal-related influence on body weight regulation in children. Interestingly, independent of BMI, MC4R-rs2331841 yielded an association with increased FBG levels and KCNQ1-rs2237892 with decreased HDL-C levels, indicating that the risk alleles at MC4R and KCNQ1 play a particular role in the regulation of glucose and lipid profiles besides their function in body weight control. The precise mechanism by which these loci act on BMI remains to be determined; as such, further studies are warranted to support this supposition and to give us more evidence.
Abbreviations
BMI, body mass index; WC, waist circumference; UAC, upper arm circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; HOMA-IR, homeostasis model assessment for insulin resistance; FGF21, fibroblast growth factor 21; RBP4, retinol binding protein 4; GPSall, sum gene score of all 12 loci and GPSleptin, sum gene score of six leptin-related loci.
Author contributions
JF analyzed the data and wrote the manuscript; SFG contributed to study design, data interpretation and reviewed/edited the manuscript; GL, LL, JY, HC, LH, QZ and NL contributed to data collection; XX and ML contributed to the data interpretation and reviewed the manuscript; JM was responsible for BCAMS; SG contributed to the design, data interpretation and edited the manuscript. ML was responsible for the biomarker study of BCAMS, and contributed to acquisition and interpretation of the data, and revised the manuscript. All authors read and approved the final manuscript.
Disclosure Summary
The authors have nothing to disclose.
Prior Presentation
Parts of this study were presented in abstract form at the American Diabetes Association’s 77th Scientific Sessions, June 9-13, 2017 in San Diego, California.
ACKNOWLEDGMENTS
We gratefully thank all the participants in the “Beijing Child and Adolescent Metabolic Syndrome (BCAMS) study”.
CONFLICTS OF INTEREST
The authors declare no conflict of interests regarding the publication of this paper.
FUNDING
This work was supported by key program of Beijing Municipal Science &Technology Commission (D111100000611001, D111100000611002); National Key Research program of China (2016YFC1304800); Beijing Science & Technology Star Program (2004A027); Novo Nordisk Union Diabetes Research Talent Fund (2011A002); National Key Program of Clinical Science (WBYZ2011-873) and Beijing Natural Science Foundation (7172169).
REFERENCES
1. Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC, Biryukov S, Abbafati C, Abera SF, Abraham JP, Abu-Rmeileh NM, Achoki T, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014; 384: 766-81.
2. Gong WJ, Zheng W, Xiao L, Tan LM, Song J, Li XP, Xiao D, Cui JJ, Li X, Zhou HH, Yin JY, Liu ZQ. Circulating resistin levels and obesity-related cancer risk: A meta-analysis. Oncotarget. 2016; 7: 57694-704. https://doi.org/10.18632/oncotarget.11034.
3. Song Y, Wang HJ, Ma J, Wang Z. Secular trends of obesity prevalence in urban Chinese children from 1985 to 2010: gender disparity. PLoS One. 2013; 8: e53069.
4. Shah S, Bonder MJ, Marioni RE, Zhu Z, McRae AF, Zhernakova A, Harris SE, Liewald D, Henders AK, Mendelson MM, Liu C, Joehanes R, Liang L, et al. Improving Phenotypic Prediction by Combining Genetic and Epigenetic Associations. Am J Hum Genet. 2015; 97: 75-85.
5. Speliotes EK, Willer CJ, Berndt SI, Monda KL, Thorleifsson G, Jackson AU, Lango Allen H, Lindgren CM, Luan J, Magi R, Randall JC, Vedantam S, Winkler TW, et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet. 2010; 42: 937-48.
6. Bradfield JP, Taal HR, Timpson NJ, Scherag A, Lecoeur C, Warrington NM, Hypponen E, Holst C, Valcarcel B, Thiering E, Salem RM, Schumacher FR, Cousminer DL, et al. A genome-wide association meta-analysis identifies new childhood obesity loci. Nat Genet. 2012; 44: 526-31.
7. Guo Y, Lanktree MB, Taylor KC, Hakonarson H, Lange LA, Keating BJ. Gene-centric meta-analyses of 108 912 individuals confirm known body mass index loci and reveal three novel signals. Hum Mol Genet. 2013; 22: 184-201.
8. Monda KL, Chen GK, Taylor KC, Palmer C, Edwards TL, Lange LA, Ng MC, Adeyemo AA, Allison MA, Bielak LF, Chen G, Graff M, Irvin MR, et al. A meta-analysis identifies new loci associated with body mass index in individuals of African ancestry. Nat Genet. 2013; 45: 690-6.
9. Chesi A, Grant SF. The Genetics of Pediatric Obesity. Trends Endocrinol Metab. 2015; 26: 711-21.
10. Felix JF, Bradfield JP, Monnereau C, van der Valk RJ, Stergiakouli E, Chesi A, Gaillard R, Feenstra B, Thiering E, Kreiner-Moller E, Mahajan A, Pitkanen N, Joro R, et al. Genome-wide association analysis identifies three new susceptibility loci for childhood body mass index. Hum Mol Genet. 2016; 25: 389-403.
11. Hong J, Shi J, Qi L, Cui B, Gu W, Zhang Y, Li L, Xu M, Wang L, Zhai Y, Miao L, Wang R, Bi Y, et al. Genetic susceptibility, birth weight and obesity risk in young Chinese. Int J Obes (Lond). 2013; 37: 673-7.
12. Wen W, Cho YS, Zheng W, Dorajoo R, Kato N, Qi L, Chen CH, Delahanty RJ, Okada Y, Tabara Y, Gu D, Zhu D, Haiman CA, et al. Meta-analysis identifies common variants associated with body mass index in east Asians. Nat Genet. 2012; 44: 307-11.
13. Okada Y, Kubo M, Ohmiya H, Takahashi A, Kumasaka N, Hosono N, Maeda S, Wen W, Dorajoo R, Go MJ, Zheng W, Kato N, Wu JY, et al. Common variants at CDKAL1 and KLF9 are associated with body mass index in east Asian populations. Nat Genet. 2012; 44: 302-6.
14. Wen W, Zheng W, Okada Y, Takeuchi F, Tabara Y, Hwang JY, Dorajoo R, Li H, Tsai FJ, Yang X, He J, Wu Y, He M, et al. Meta-analysis of genome-wide association studies in East Asian-ancestry populations identifies four new loci for body mass index. Hum Mol Genet. 2014; 23: 5492-504.
15. Warrington NM, Howe LD, Paternoster L, Kaakinen M, Herrala S, Huikari V, Wu YY, Kemp JP, Timpson NJ, St Pourcain B, Davey Smith G, Tilling K, Jarvelin MR, et al. A genome-wide association study of body mass index across early life and childhood. Int J Epidemiol. 2015; 44: 700-12.
16. Abadi A, Peralta-Romero J, Suarez F, Gomez-Zamudio J, Burguete-Garcia AI, Cruz M, Meyre D. Assessing the effects of 35 European-derived BMI-associated SNPs in Mexican children. Obesity (Silver Spring). 2016; 24: 1989-95.
17. Willer CJ, Speliotes EK, Loos RJ, Li S, Lindgren CM, Heid IM, Berndt SI, Elliott AL, Jackson AU, Lamina C, Lettre G, Lim N, Lyon HN, et al. Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nat Genet. 2009; 41: 25-34.
18. Bluher M, Mantzoros CS. From leptin to other adipokines in health and disease: facts and expectations at the beginning of the 21st century. Metabolism. 2015; 64: 131-45.
19. Parimisetty A, Dorsemans AC, Awada R, Ravanan P, Diotel N, Lefebvre d’Hellencourt C. Secret talk between adipose tissue and central nervous system via secreted factors-an emerging frontier in the neurodegenerative research. J Neuroinflammation. 2016; 13: 67.
20. Huang T, Tobias DK, Hruby A, Rifai N, Tworoger SS, Hu FB. An Increase in Dietary Quality Is Associated with Favorable Plasma Biomarkers of the Brain-Adipose Axis in Apparently Healthy US Women. J Nutr. 2016; 146: 1101-8.
21. Kilpelainen TO, Carli JF, Skowronski AA, Sun Q, Kriebel J, Feitosa MF, Hedman AK, Drong AW, Hayes JE, Zhao J, Pers TH, Schick U, Grarup N, et al. Genome-wide meta-analysis uncovers novel loci influencing circulating leptin levels. Nat Commun. 2016; 7: 10494.
22. Gerken T, Girard CA, Tung YC, Webby CJ, Saudek V, Hewitson KS, Yeo GS, McDonough MA, Cunliffe S, McNeill LA, Galvanovskis J, Rorsman P, Robins P, et al. The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase. Science. 2007; 318: 1469-72.
23. Ruiz-Narvaez EA, Haddad SA, Rosenberg L, Palmer JR. Birth weight modifies the association between central nervous system gene variation and adult body mass index. J Hum Genet. 2016; 61: 193-8.
24. Locke AE, Kahali B, Berndt SI, Justice AE, Pers TH, Day FR, Powell C, Vedantam S, Buchkovich ML, Yang J, Croteau-Chonka DC, Esko T, Fall T, et al. Genetic studies of body mass index yield new insights for obesity biology. Nature. 2015; 518: 197-206.
25. Fasshauer M, Blüher M. Adipokines in health and disease. Trends in pharmacological sciences. 2015; 36: 461-70.
26. Kadowaki T, Yamauchi T, Kubota N. The physiological and pathophysiological role of adiponectin and adiponectin receptors in the peripheral tissues and CNS. FEBS Lett. 2008; 582: 74-80.
27. Li M, Fisette A, Zhao XY, Deng JY, Mi J, Cianflone K. Serum resistin correlates with central obesity but weakly with insulin resistance in Chinese children and adolescents. Int J Obes (Lond). 2009; 33: 424-39.
28. Xu J, Lloyd DJ, Hale C, Stanislaus S, Chen M, Sivits G, Vonderfecht S, Hecht R, Li YS, Lindberg RA, Chen JL, Jung DY, Zhang Z, et al. Fibroblast growth factor 21 reverses hepatic steatosis, increases energy expenditure, and improves insulin sensitivity in diet-induced obese mice. Diabetes. 2009; 58: 250-9.
29. Lim S, Yoon JW, Choi SH, Park YJ, Lee JJ, Park JH, Lee SB, Kim KW, Lim JY, Kim YB, Park KS, Lee HK, Cho SI, et al. Combined impact of adiponectin and retinol-binding protein 4 on metabolic syndrome in elderly people: the Korean Longitudinal Study on Health and Aging. Obesity (Silver Spring, Md). 2010; 18: 826-32.
30. Yeo GS, Farooqi IS, Aminian S, Halsall DJ, Stanhope RG, O’Rahilly S. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat Genet. 1998; 20: 111-2.
31. Tung YC, Yeo GS, O’Rahilly S, Coll AP. Obesity and FTO: Changing Focus at a Complex Locus. Cell Metab. 2014; 20: 710-8.
32. Chen Z, Yin Q, Ma G, Qian Q. KCNQ1 gene polymorphisms are associated with lipid parameters in a Chinese Han population. Cardiovasc Diabetol. 2010; 9: 35.
33. Gordon T, Castelli WP, Hjortland MC, Kannel WB, Dawber TR. High density lipoprotein as a protective factor against coronary heart disease. The Framingham Study. Am J Med. 1977; 62: 707-14.
34. Group of China Obesity Task Force. Body mass index reference norm for screening overweight and obesity in Chinese children and adolescents. [Article in Chinese]. Zhonghua Liu Xing Bing Xue Za Zhi. 2004; 25: 97-102.
35. Li M, Yin JH, Zhang K, Wu CY. A highly sensitive enzyme-linked immunosorbent assay for measurement of leptin secretion in human adipocytes. [Article in Chinese]. Zhonghua Yi Xue Za Zhi. 2008; 88: 3293-7.
36. Li Q, Lu Y, Sun L, Yan J, Yan X, Fang L, Li M, Fan Z. Plasma adiponectin levels in relation to prognosis in patients with angiographic coronary artery disease. Metabolism. 2012; 61: 1803-8.
37. Xu L, Ping F, Yin J, Xiao X, Xiang H, Ballantyne CM, Wu H, Li M. Elevated plasma SPARC levels are associated with insulin resistance, dyslipidemia, and inflammation in gestational diabetes mellitus. PLoS One. 2013; 8: e81615.
38. Li L, Yin J, Cheng H, Wang Y, Gao S, Li M, Grant SF, Li C, Mi J, Li M. Identification of Genetic and Environmental Factors Predicting Metabolically Healthy Obesity in Children: Data From the BCAMS Study. J Clin Endocrinol Metab. 2016; 101: 1816-25.
39. Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007; 39: 175-91.
40. MacKinnon DP, Fairchild AJ, Fritz MS. Mediation analysis. Annu Rev Psychol. 2007; 58: 593-614.

