INTRODUCTION
The morbidity and mortality of colorectal cancer is increasing year by year, which threatens human health severely. It is generally believed that colorectal cancer is associated with high fat, high carbohydrate and high protein diet [1, 2]. Obesity is also considered to be a risk factor for colorectal cancer [3, 4]. So it is necessary to investigate the relationship between dietary intervention and colorectal cancer.
Interactions between cancer metabolism and immunity are arousing wide concern recently. Preliminary data indicates that short-term fasting or ketogenic diet, both to induce physiological hungry state, can reverse immunosuppressive character of the tumor microenvironment through decreasing lactic acid or increasing ATP in the tumor microenvironment [5, 6]. Most of these studies focus on immune cells such as regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), or NK cells [5, 6]. Meanwhile however, few studies are reported about fasting and tumor-associated macrophages, which are among the most important immune components of tumor microenvironment.
Tumor-associated macrophages (TAMs) possess type M2 macrophage biology characteristics and play a critical role in immunity in the tumor microenvironment. It is generally accepted that TAMs can shield tumor immune surveillance and stimulate new blood vessel formation, induce proliferation of cancer cells, facilitate local invasion and distant metastasis of tumor cells [7, 8].
Here, using both in vitro and in vivo colon carcinoma models, we found that fasting could suppress M2 polarization of TAMs through inactivating JAK1/STAT3 in the tumor microenvironment and therefore inhibit tumor cell growth. This process involved the lower generation of extracellular adenosine in the tumor microenvironment through increased cancer cell autophagy and decrease of CD73, an ectoenzyme that degrades extracellular ATP into immunosuppressive adenosine. Our study highlighted the importance of fasting in inhibiting colorectal cancer growth through lowering generation of adenosine and suppressing M2-TAMs proliferation in the tumor microenvironment.
RESULTS
Fasting inhibits tumor growth both in vivo and in vitro
Mice colorectal cancer model was constructed in this study. Compared with mice in control group, the tumor growth of ADF group was suppressed (Figure 1A, 1B), without a reduction in body weight (Figure 1C). For in vitro experiments, CT26 cancer cells and RAW264.7 macrophage cells were cocultured in fasting or normal RPMI-1640 medium. We found that the proliferation of CT26 cells was suppressed in fasting medium (Figure 1D).
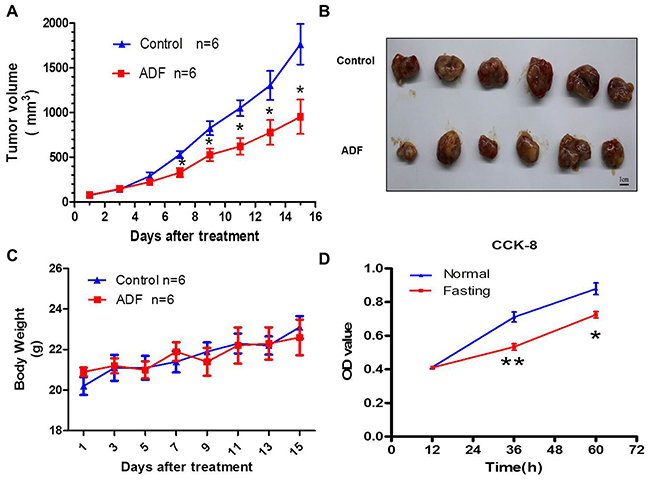
Figure 1: Fasting inhibits tumor growth both in vivo and in vitro. (A) Tumor growth curve of mice from Control and ADF groups for 2 weeks. *P<0.05. (B) Subcutaneously implanted tumors dissected from mice of Control and ADF groups. (C) Body weights of mice from Control and ADF groups. Data shows no significant difference between the two groups. (D) The proliferation of CT26 cells in normal or fasting coculture condition in vitro was tested by CCK8. *P<0.05, **P<0.01.
Fasting suppresses M2 polarization of TAMs both in vivo and in vitro
In order to find out whether fastinginhibits tumor growth through modulating TAMs, we tested the M2 polarization of TAMs. For in vivo experiments, we found that M2 polarization of TAMs was reduced in tumor tissues of ADF group (Figure 2A, 2B). For in vitro experiments, CT26 cancer cells and RAW264.7 macrophage cells were cocultured in fasting or normal RPMI-1640 medium for 24 h or 48 h. The results of flow cytometry showed that M2 polarization of RAW264.7 cells was reduced in fasting condition (Figure 2C, 2D).
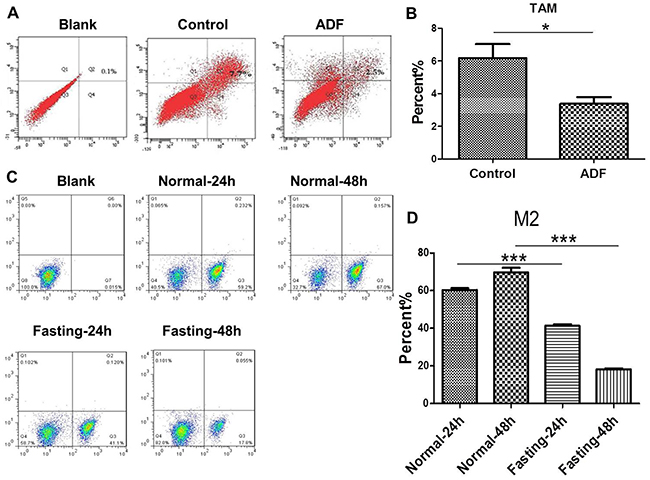
Figure 2: Fasting suppresses M2 polarization of TAMs both in vivo and in vitro. (A-B) The levels of M2 macrophages were detected by FCM (flow cytometry) in tumor tissues of mice from Control and ADF groups (ADF: alternate day fasting). *P<0.05. (C-D) The levels of M2 macrophages were detected by FCM (flow cytometry) in normal or fasting medium for 24 h or 48 h. ***P<0.001.
Fasting induces cancer cell autophagy, downregulates CD73 expression of cancer cells and suppresses adenosine generation in vitro
Previous studies have reported that autophagy of tumor cells can downregulate the expression of ectoenzymes such as CD39 and CD73 related to adenosine generation[9, 10]. Atg5 and LC3II/I were the indicators for the autophagy of cancer cells. In our study, the expression of Atg5, LC3II/I and CD73 in CT26 cells with fasting or normal medium was detected by qPCR or Western blotting. The results showed that fasting increased the expression of Atg5 and LC3II/I (Figure 3A, 3B, 3C) and decreased the expression of CD73 in CT26 cells (Figure 3D, 3E). Moreover, the generation of adenosine was reduced (Figure 3F).
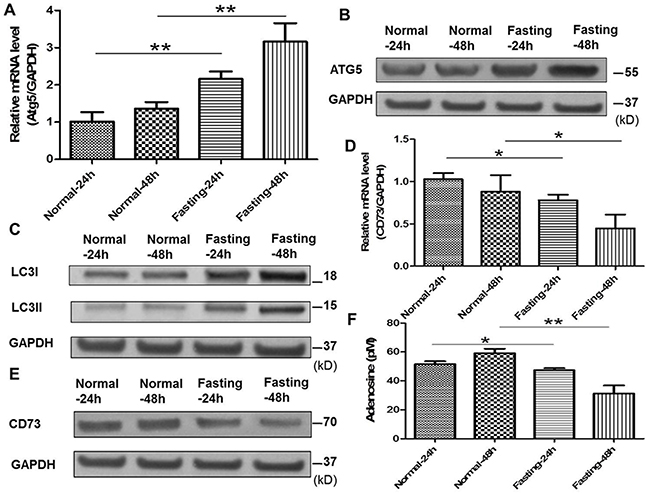
Figure 3: Fasting induces cancer cell autophagy, downregulates CD73 expression of cancer cells and suppresses adenosine generation in vitro. (A) The mRNA expression of Atg5 was examined by qPCR in normal or fasting medium for 24 h or 48 h. **P<0.01. (B) The expression of Atg5 was tested by Western blotting. (C) The expression of LC3I/II was tested by Western blotting. (D) The mRNA expression of CD73 was tested by qPCR. *P<0.05. (E) The expression of CD73 was determined by Western blotting. (F) The levels of adenosine were tested by ELISA. *P<0.05, **P<0.01.
Knockdown of Atg5 in cancer cells enhances adenosine’s effect on macrophages and tumor growth in vitro
To verify the role of autophagy in this study, autophagy-related Atg5 of CT26 cells was knocked down by Atg5 shRNA (Supplementary Figure 1) in fasting medium. We found that the expression of CD73 (Figure 4A, 4B) and the levels of extracellular adenosine (Figure 4C) were enhanced after knockdown of Atg5 in CT26 cells in the coculture system for 24 h. The M2 polarization of macrophages (Figure 4D, 4E) and the proliferation of CT26 cells were increased simultaneously (Figure 4F).
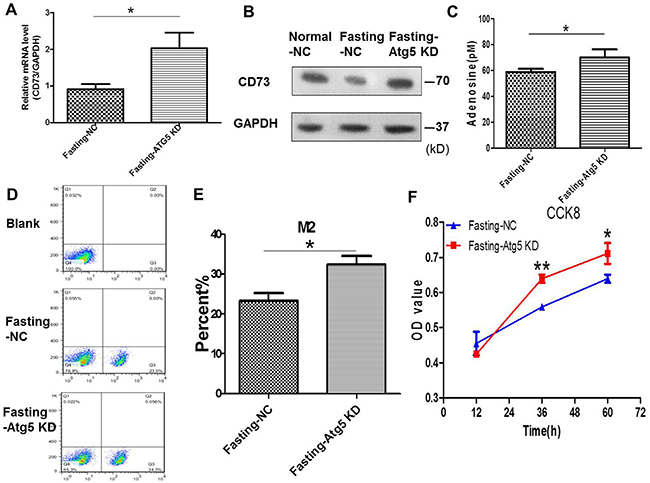
Figure 4: Knockdown of Atg5 in cancer cells enhances adenosine’s effect on macrophages and tumor growth in coculture condition. (A-F) CT26 cells were stably transfected with shAtg5 (Atg5 KD) or shNC (NC). (A-B) The CT26 expression of CD73 was detected by qPCR in fasting medium. *P<0.05. (C) The levels of extracellular adenosine were tested by ELISA. *P<0.05. (D-E) The levels of M2 macropages were detected by FCM (flow cytometry). *P<0.05. (F) The proliferation of CT26 cells was tested by CCK8. *P<0.05, **P<0.01.
Knockdown of CD73 in cancer cells suppresses adenosine’s effect on macrophages and tumor growth in vitro
We postulated that CD73 contributed to this progression by regulating adenosine generation. So in this part, CD73 of CT26 cells was knocked down by CD73 shRNA (Supplementary Figure 2). The results showed that the levels of extracellular adenosine were decreased after knockdown of CD73 in CT26 cells in the coculture system for 24 h. Accordingly, the polarization of M2 macrophages (Figure 5A, 5B, 5C) and the proliferation of CT26 cells were inhibitted (Figure 5D).
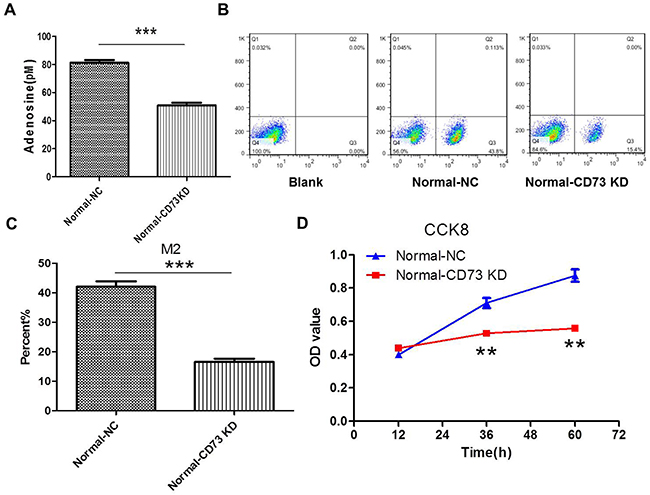
Figure 5: Knockdown of CD73 in cancer cells suppresses adenosine’s effect on macrophages and tumor growth in coculture condition. (A-D) CT26 cells were stably transfected with shCD73 (CD73 KD) or shNC(NC). (A) The levels of extracellular adenosine were tested by ELISA in normal medium. ***P<0.001. (B-C) The levels of M2 macropages were detected by FCM (flow cytometry). ***P<0.001. (D) The proliferation of CT26 cells was tested by CCK8. **P<0.01.
PSB1115 treatment downregulates adenosine’s effect on macrophages and tumor growth in vitro
It is reported that adenosine, as an immuno-suppressive endogenous purine nucleoside with high expression in the tumor microenvironment, can induce M2 polarization of TAMs [11, 12]. So in this part of study, the antagonist PSB1115 was used to block the binding of adenosine to its receptors of macrophages at a concentration of 1 μM. The M2 polarization of macrophages (Figure 6A, 6B) and the proliferation of CT26 cells (Figure 6C) were suppressed after PSB1115 treatment for 24 h in coculture system.
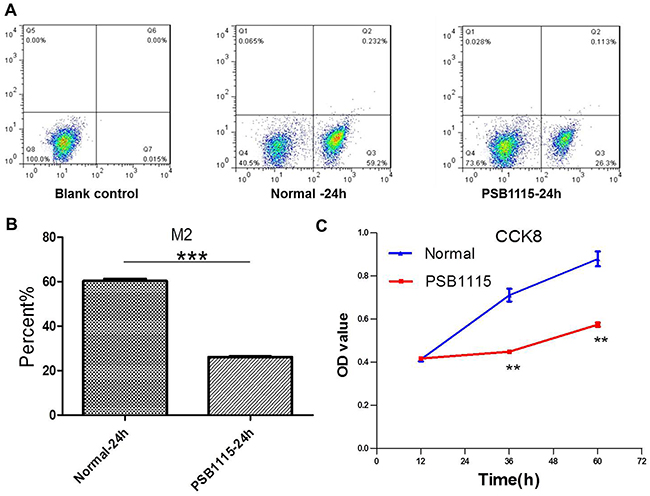
Figure 6: PSB1115 treatment downregulates adenosine’s effect on macrophages and tumor growth in coculture condition. (A-B) The levels of M2 macropages were detected by FCM (flow cytometry) in normal medium with or without PSB1115 treatment for 24 h. ***P<0.001. (C) The proliferation of CT26 cells was tested by CCK8. **P<0.01.
Fasting suppresses M2 polarization of macrophages through inactivating JAK1/STAT3 signal pathway
To find out the mechanism involved in the suppression of M2 polarization of TAMs by fasting, we investigated the expression of JAK1/STAT3 signal pathway in TAMs. For in vitro experiments, CT26 cells and RAW264.7 cells were cocultured for 24 h or 48 h. The results showed that the phosphorylation of JAK1 and STAT3 in RAW264.7 cells in fasting condition was suppressed (Figure 7A). Knockdown of CD73 in CT26 cells (Figure 7B) or PSB1115 treatment in normal coculture condition also inhibitted the phosphorylation of JAK1 and STAT3 in RAW264.7 cells. The inactivation of JAK1/STAT3 signal pathway of RAW264.7 cells was reversed after Atg5 of CT26 cells was knocked down in fasting coculture condition (Figure 7B). Knockdown of STAT3 in RAW264.7 cells in normal cocluture system (Supplementary Figure 3) suppressed M2 polarization of RAW264.7 cells and proliferation of CT26 cells (Figure 7C, 7D, 7E).
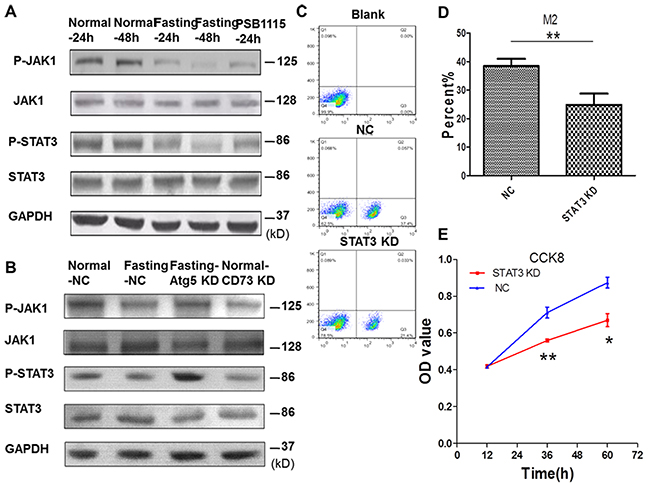
Figure 7: Fasting suppresses M2 polarization of macrophages through inactivating JAK1/STAT3 signal pathway in coculture condition. (C-E) Raw264.7 cells were stably transfected with shSTAT3 (STAT3 KD) or shNC (NC). (A) The expression of JAK1/STAT3 signal pathway of RAW264.7 cells was determined by Western blotting. (B) The expression of JAK1/STAT3 signal pathway of RAW264.7 cells was determined by Western blotting when Atg5 or CD73 of CT26 cells were knocked down. (C-D) The levels of M2 macropages were detected by FCM (flow cytometry). **P<0.01. (E) The proliferation of CT26 cells was tested by CCK8. *P<0.05, **P<0.01.
DISCUSSION
Under nutrition deprivation caused by fasting, tumor cells have higher levels of autophagy [13, 14]. It is believed that autophagy plays dual roles in the development of cancer by activating the immune response in some cases, and impairing the anti-tumor immunity in others. It is reported that in early oncogenesis, autophagy is often inhibitted. However, at advanced stage, autophagy is required for the progression of cancers [15, 16], suggesting that inducing autophagy in early stages of tumor progression may be an effective way to promote anti-tumor immunity. Of note, nutrition starvation is one of the most efficient ways to elicit autophagy in most cells [13, 17]. Moreover, both fasting and autophagy are reported to promote rejuvenation and delay senescence of hematopoietic stem cells so as to maintain the function of immune system [9, 18]. In our study, mice were subjected to alternate-day fasting when subcutaneous tumors formed. Without a reduction in body weight, the alternate-day fasting mice were as active and healthy as the AL fed mice and tumor sizes were reduced compared with control group. Our results showed that in this stage of CT26 tumor progression, fasting could promote anti-tumor immunity by inducing tumor autophagy. Our work provided a new evidence for the complex interactions among autophagy, tumor immunity and tumor progression.
It is reported that autophagy decreases expression of ectoenzymes such as CD39 and CD73 in lung cancer and reduces levels of adenosine [10, 14]. Here we found that autophagy in CT26 tumor cells decreased CD73 expression. As some reports suggest that HIF-1α could increase the expression of genes encoding CD39 and CD73 [13, 19], whether fasting-induced autophagy reduces the level of CD73 through decreasing HIF-1α needs further study.
Adenosine is well known to be an immuno-suppressive endogenous purine nucleoside with high expression in the tumor microenvironment [11, 12]. Macrophages are the major component within the tumor microenvironment (usually termed TAMs) participating both in the progression of tumor growth and suppression of antitumor immunity. TAMs exhibit a predominantly M2-like phenotype [20, 21]. Our work further verified the role of adenosine in suppressing anti-tumor immunity by linking it to fasting and TAMs.
Adenosine can bind to both A2A and A2B receptors, mainly A2B receptors [22, 23], on the membrane of TAMs to increase the M2 polarization. Our work used A2B receptor antagonist PSB1115 and found that A2B receptors were involved in adenosine’s effect on TAMs polarization. It is reported that A2B receptors worked at a high concentration of adenosine [24]. However, our study found that A2B receptor blockade could reduce M2 polarization of TAMs at a relatively low concentration of adenosine. Our further study will investigate the role of A2A receptors in this process.
The therapeutic potential of A2B receptor blockade has been reported [25–28]. To our knowledge, this is the first study demonstrating that selective blockade of A2B receptor by PSB1115 can be effective in delaying colon cancer growth by inhibiting TAMs accumulation and restoring antitumor immunity.
We also explored the relevant signal pathways associated with adenosine regulating of M2 polarization of TAMs. There are different signal pathways involved in polarization of macrophages such as MAPK/JNK, PI3K/AKT and JAK/STAT [29–31]. Here we found that JAK1/STAT3 signal pathway played an important role in M2 polarization of TAMs. We used PSB1115 to block adenosine from binding to its receptors on the surface of macrophages, and found that the phosphorylation of JAK1 and STAT3, M2 polarization in RAW264.7 cells were inhibitted, and the proliferation of CT26 cells was suppressed. Blockade of JAK1/STAT3 signal pathway by knocking down STAT3 significantly weakened M2 polarization of RAW264.7 macrophages induced by adenosine in coculture condition and suppressed proliferation of CT26 cells, which further confirmed the effect of TAMs on tumor growth. Although our previous results couldn’t exclude the possiblity of direct impact of Atg5 and CD73 knocking down or extracellular adenosine on the proliferation of CT26 cells, this part of study indicated that factors reducing M2 polarization of macrophages indeed could inhibit the growth of CT26 cells at least partially through altering tumor immunity related to TAMs.
Different methods of fasting have been reported to be good for tumor treatment. Many studies have focused on the combination of fasting and chemotherapy or radiotherapy [10, 32–34]. D’Aronzo et al. reported that 24 h starvation could enhance gemcitabine effect in a pancreatic cancer xenograft mouse model [35]. Our study indicates that alternate day fasting is also effective in delaying colon cancer growth, providing new approach for cancer prevention and cure. It is our later work to investigate the relationship between alternate day fasting and colon cancer in human. Moreover, whether fasting is efficient for other cancer types needs further investigation.
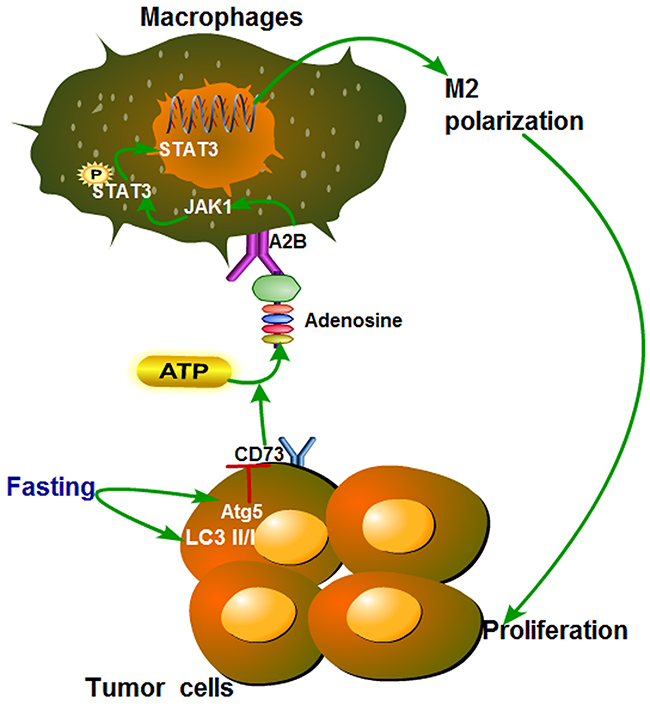
Figure 8: Proposed model of the effect of fasting on STAT3 pathway during polarization of tumor-associated macrophages. Fasting induces expression of autophagy-related Atg5 and LC3II/I of colon cancer, then downregulates the level of adenosine by suppressing CD73, which modulates M2 polarization of TAMs through inactivating JAK1/STAT3, and eventually inhibits tumor growth.
MATERIALS AND METHODS
Cell culture
Mouse CT26 cell lines purchased from the TypeCulture Collection of the Chinese Academy of Sciences (Shanghai, China) and RAW 264. 7 cell lines, a kind gift of Prof Miao (Fudan University Shanghai Cancer Center, China) were both grown in RPMI-1640 medium (BI, Israel) supplemented with 2g/l glucose, penicillin/streptomycin and 10% fetal bovine serum (FBS) as normal condition. All the cells were cultured at 37°C, in a 5% CO2 incubator. The cells were cocultured in a transwell system at a ratio of 1: 1 to simulate the tumor microenvironment.
In vitro starvation
CT26 cells and RAW264.7 cells were cocultured in RPMI-1640 with low glucose (0.5 g/l) and 1% FBS to achieve fasting condition as previously described [32]. Cells were washed twice with PBS before switching to fasting medium.
Atg5, CD73 and STAT3 knockdown in CT26 and RAW264.7 cells
The small hairpin RNA (shRNA) targeting mouse Atg5 (shAtg5, CACCGGACTGCAGAATGACAGATTTCAAGAGAATCTGTCATTCTGCAGTCCTTTTTTG), CD73(shCD73,CACCGCAGCCTGAAGTAGATAAACTTTCAAGAGAAGTTTATCTACTTCAGGCTGCTTTTTTG), STAT3(shSTAT3,CACCGGAGCAGCATCTTCAGGATGTTTCAAGAGAACATCCTGAAGATGCTGCTCCTTTTTTG) and a scrambled shRNA used as negative control (NC, Addgene) were synthesized and cloned into the pLenti-C-Myc-DDK (Origene, China) to generate the lentirival expression vectors. For lentivirus production, 1 μg of shRNA expression plasmid and 1 μg of helper plasmids (0.4 μg pMD2G and 0.6 μg psPAX2) were transfected into 293T cells (the TypeCulture Collection of the Chinese Academy of Sciences, China) with Effectene reagent (HilyMax, Japan). The supernatants were collected 48 h after transfection and cleared through a 0.45-μm filter. CT26 cells or RAW264.7 cells were infected with viral supernatants containing 4 μg/ml polybrene (Sigma, USA) for 24 h. After the transduction, the cells were selected with puromycin (VWR, USA) for 7 days for stable shRNA expression.
Cell proliferation assay
Cell viability was measured using cell counting kit-8 (CCK8) assay (Dojindo, Japan) according to the manufacture’s instructions. In short, CT26 was cocultured with macrophage cells as described above. Cells were grown for the indicated times. Then cells were transferred to 96-well plates at a concentration of 5×103 cells/well and 10 μL of CCK8 solution was added to each well and cells were further incubated for 2 h. The optical density (OD) values were measured using a microplate reader at 450 nm.
Mouse model and in vivo fasting
All animal studies and procedures were approved by the Medicine and Public Health Animal Care and Use Committee of Shanghai Medical College at Fudan University. Wild-type BALB/c mice (female, 6 weeks, 20–25 g) were purchased from Shanghai Sippr-BK laboratory animal Co. Ltd.. To establish a colon cancer bearing mouse model, 12 mice were injected subcutaneously with 100ul CT26 cells resuspended in PBS at a density of 2×106 cells/mL. When tumors were palpable 5-7 days after tumor cell inoculation, the animals were divided into 2 groups. “Control” mice (N=6) with no treatment which were kept under standard conditions for the whole duration of the study; “ADF” mice (N=6) that were subjected to alternate day fasting and ad libitum diet on non-fasting days with free access to water every day. Mice were individually housed in a clean new cage to avoid cannibalism or coprophagy. Tumor sizes, body weights and general behavior were monitored every other day. Tumor sizes were measured by a caliper and tumor volume was calculated using the following equation: tumor volume (mm3) = (length × width × width) × π/6, expressing length and width in mm.
Elisa
For in vitro experiments, the adenosine levels in supernatants of coculture system were measured with ELISA kit (Biovision, USA) according to the manufacturer’s instructions. The optical density at 450 nm each well was determined by PerkinElmermicroplate reader (Molecular Devices, USA). All experiments were performed in triplicates.
Western blotting
Cells were harvested and lysed with lysis buffer (Beyotime, China) containing protease inhibitors (PMSF, Dingguo, China). Cellular proteins were collected after centrifugation at 10 000 × g at 4°C, and boiled for 5 min together with loading buffer. Proteins were separated by 10% SDS-PAGE, and further transferred onto nitrocellulose membranes (Bio-Rad, USA). The membranes were incubated with primary antibodies (anti-Atg5, Santa Cruz, USA; anti-LC3I/II, Santa Cruz, USA; anti-CD73, Santa Cruz, USA; anti-JAK1, Santa Cruz, USA; anti-STAT3, Santa Cruz, USA; anti-P-JAK1, Cell Signaling Technology, USA; anti-P-STAT3, Cell Signaling Technology, USA) overnight at 4°C after blocking with 5% fat-free milk in TBS-T for 1 h at room temperature. Then the membranes were incubated with a HRP-conjugated secondary antibody for 1h at room temperature followed by three-time washing with TBS-T. Bands were detected with ELL by X-ray films (MidSci, St. Louis, MO, USA) after three-time washing with TBS-T.
qPCR
Total RNA was isolated from cells by Trizol® (Invitrogen, Carlsbad, CA). The cDNA was synthesized using iScript™ cDNA Synthesis Kit (BioRad, Hercules, CA, USA) according to the manufacturer’s instructions. Quantitative real-time PCR (qPCR) reactions were performed to detect the expression of Atg5, CD73 and STAT3 with the iTaq™ SYBR Green Supermix (Bio-Rad, Hercules, CA) and StepOnePlus™ Real-Time PCR System (Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s protocol. The qPCR primers used in this study can be found in Supplementary Table 1.
Flow cytometry
For in vivo experiments, tumor tissues were prepared as previously described [36–38], in brief, tumor tissues were mechanically dissociated using scalpels and after additional enzymatic dissociation with 1 mg/mL collagenases for 40 min, the fragments were filtrated through a 70 μm cell strainer. Cell suspensions were used for flow cytometry analysis. The expression of M2 macrophage biomarkers CD206 (Cell Signaling Technology, USA) and F4/80 (BD Biosciences, USA) was detected by flow cytometric analysis. For in vitro experiments, the expression of M2 macrophage biomarker CD206 was detected by flow cytometric analysis. In brief, cells were stained with anti-CD206 (Cell Signaling Technology, USA) for 30 min at room temperature according to the manufacturer’s instructions. Then cells were collected and analyzed. M2 polarization of macrophages was assessed by the percentage of positively stained cells. Flow cytometric data was acquired using a FACSAria™ flowcytometer (BD Biosciences, USA) and analyzed with FlowJo software version 7. 6. 1 (flowjo. com). All experiments were repeated in triplicates.
Statistical analysis
All results were presented as mean ± SD (standard deviation). Statistical analysis was performed by t-test or one-way ANOVA with GraphPad Prism Version 5 (GraphPad Software, San Diego, CA). In all cases, difference was consideredstatistically significant at P < 0.05.
CONCLUSIONS
To our knowledge, our work is the first to uncover the link between fasting and TAMs in tumor microenvironment. In summary, this study showed that fasting could induce autophagy of colon cancer cells and then downregulate the level of adenosine, which increased M2 polarization of TAMs through inactivating JAK1/STAT3, and eventually inhibitted tumor growth by promoting antitumor immunity (Figure 8) in vitro and in vivo.
Abbreviations
Tregs, regulatory T cells; MDSCs, myeloid-derived suppressor cells; TAMs, Tumor-associated macrophages; JAK1, Janus Kinase 1; STAT3, Signal Transducer and Activator of Transcription 3; Atg5, Autophagy Related 5; LC3, Microtubule Associated Protein 1 Light Chain 3; HIF-1α, Hypoxia Inducible Factor 1 Alpha; MAPK, Mitogen-Activated Protein Kinase; JNK, c-Jun N-terminal kinase; PI3K, Phosphatidylinositol 3-Kinase; AKT, Protein Kinase B.
Author contributions
Changhong Miao and Duan Ma supervised experiments, Pengfei Sun and Huihui Wang designed experiments, Pengfei Sun, Huihui Wang and Meilin Weng performed experiments and analyzed data, Pengfei Sun, Huihui Wang, Xiangyuan Chen and Qichao Wu wrote the manuscript, Wankun Chen, Zhirong Sun and Minmin Zhu advised on experimental design and provided critical feedback, all authors reviewed the manuscript.
ACKNOWLEDGMENTS
We would like to thank Professor Jin Zhang for critical feedback in designing and performing the experiments.
CONFLICTS OF INTEREST
There are no conflicts of interest to disclose.
FUNDING
This work was is supported by the National Natural Science Foundation of China (No. 81372101).
REFERENCES
1. Levin B. Nutrition and colorectal cancer. Cancer. 1992; 70:1723-1726.
2. Van Blarigan EL, Meyerhardt JA. Role of physical activity and diet after colorectal cancer diagnosis. J Clin Oncol. 2015; 33:1825-1834.
3. Bardou M, Barkun AN, Martel M. Obesity and colorectal cancer. Gut. 2013; 62:933-947.
4. Martinez-Useros J, Garcia-Foncillas J. Obesity and colorectal cancer: molecular features of adipose tissue. J Transl Med. 2016; 14:21.
5. Husain Z, Huang Y, Seth P, Sukhatme VP. Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol. 2013; 191:1486-1495.
6. Pietrocola F, Pol J, Vacchelli E, Rao S, Enot DP, Baracco EE, Levesque S, Castoldi F, Jacquelot N, Yamazaki T. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell. 2016; 30:147-160.
7. Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010; 141:39-51.
8. Hao NB, Lü MH, Fan YH, Cao YL, Zhang ZR, Yang SM. Macrophages in tumor microenvironments and the progression of tumors. Clin Dev Immunol. 2012; 2012:948098.
9. Mendelsohn AR, Larrick JW. Prolonged fasting/refeeding promotes hematopoietic stem cell regeneration and rejuvenation. Rejuvenation Res. 2014; 17:385-389.
10. Pietrocola F, Pol J, Vacchelli E, Rao S, Enot DP, Baracco EE, Levesque S, Castoldi F, Jacquelot N, Yamazaki T, Senovilla L, Marino G, Aranda F, et al. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell. 2016; 30:147-160.
11. Ohta A. A metabolic immune checkpoint: adenosine in tumor microenvironment. Front Immunol. 2016; 7:109.
12. Morello S, Pinto A, Blandizzi C, Antonioli L. Myeloid cells in the tumor microenvironment: role of adenosine. Oncoimmunology. 2016; 5:e1108515.
13. van Niekerk G, Hattingh SM, Engelbrecht AM. Enhanced therapeutic efficacy in cancer patients by short-term fasting: the autophagy connection. Front Oncol. 2016; 6:242.
14. Lashinger LM, O’Flanagan CH, Dunlap SM, Rasmussen AJ, Sweeney S, Guo JY, Lodi A, Tiziani S, White E, Hursting SD. Starving cancer from the outside and inside: separate and combined effects of calorie restriction and autophagy inhibition on Ras-driven tumors. Cancer Metab. 2016; 4:18.
15. Rao S, Tortola L, Perlot T, Wirnsberger G, Novatchkova M, Nitsch R, Sykacek P, Frank L, Schramek D, Komnenovic V, Sigl V, Aumayr K, Schmauss G, et al. A dual role for autophagy in a murine model of lung cancer. Nat Commun. 2014; 5:3056.
16. White E. Deconvoluting the context-dependent role for autophagy in cancer. Nat Rev Cancer. 2012; 12:401-410.
17. Mizushima N, Yamamoto A, Matsui M, Yoshimori T, Ohsumi Y. In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker. Mol Biol Cell. 2004; 15:1101-1111.
18. Ho TT, Warr MR, Adelman ER, Lansinger OM, Flach J, Verovskaya EV, Figueroa ME, Passegue E. Autophagy maintains the metabolism and function of young and old stem cells. Nature. 2017; 543:205-210.
19. Hatfield SM, Sitkovsky M. A2A adenosine receptor antagonists to weaken the hypoxia-HIF-1alpha driven immunosuppression and improve immunotherapies of cancer. Curr Opin Pharmacol. 2016; 29:90-96.
20. Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002; 23:549-555.
21. Sica A, Schioppa T, Mantovani A, Allavena P. Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer. 2006; 42:717-727.
22. Csoka B, Selmeczy Z, Koscso B, Nemeth ZH, Pacher P, Murray PJ, Kepka-Lenhart D, Morris SM Jr, Gause WC, Leibovich SJ, Hasko G. Adenosine promotes alternative macrophage activation via A2A and A2B receptors. FASEB J. 2012; 26:376-386.
23. Koscso B, Csoka B, Kokai E, Nemeth ZH, Pacher P, Virag L, Leibovich SJ, Hasko G. Adenosine augments IL-10-induced STAT3 signaling in M2c macrophages. J Leukoc Biol. 2013; 94:1309-1315.
24. Gessi S, Merighi S, Sacchetto V, Simioni C, Borea PA. Adenosine receptors and cancer. Biochim Biophys Acta. 2011; 1808:1400-1412.
25. Ma DF, Kondo T, Nakazawa T, Niu DF, Mochizuki K, Kawasaki T, Yamane T, Katoh R. Hypoxia-inducible adenosine A2B receptor modulates proliferation of colon carcinoma cells. Hum Pathol. 2010; 41:1550-1557.
26. Wei Q, Costanzi S, Balasubramanian R, Gao ZG, Jacobson KA. A2B adenosine receptor blockade inhibits growth of prostate cancer cells. Purinergic Signal. 2013; 9:271-280.
27. Iannone R, Miele L, Maiolino P, Pinto A, Morello S. Blockade of A2b adenosine receptor reduces tumor growth and immune suppression mediated by myeloid-derived suppressor cells in a mouse model of melanoma. Neoplasia. 2013; 15:1400-1409.
28. Cekic C, Sag D, Li Y, Theodorescu D, Strieter RM, Linden J. Adenosine A2B receptor blockade slows growth of bladder and breast tumors. J Immunol. 2012; 188:198-205.
29. Kang K, Reilly SM, Karabacak V, Gangl MR, Fitzgerald K, Hatano B, Lee CH. Adipocyte-derived Th2 cytokines and myeloid PPARdelta regulate macrophage polarization and insulin sensitivity. Cell Metab. 2008; 7:485-495.
30. Arranz A, Doxaki C, Vergadi E, Torre YM, Vaporidi K, Lagoudaki ED, Ieronymaki E, Androulidaki A, Venihaki M, Margioris AN. Akt1 and Akt2 protein kinases differentially contribute to macrophage polarization. Proc Natl Acad Sci U S A. 2012; 109:9517-9522.
31. Chen W, Xu Y, Zhong J, Wang H, Weng M, Cheng Q, Wu Q, Sun Z, Jiang H, Zhu M, Ren Y, Xu P, Chen J, Miao C. MFHAS1 promotes colorectal cancer progress by regulating polarization of tumor-associated macrophages via STAT6 signaling pathway. Oncotarget. 2016; 7:78726-78735. https://doi.org/10.18632/oncotarget.12807.
32. Bianchi G, Martella R, Ravera S, Marini C, Capitanio S, Orengo A, Emionite L, Lavarello C, Amaro A, Petretto A, Pfeffer U, Sambuceti G, Pistoia V, et al. Fasting induces anti-Warburg effect that increases respiration but reduces ATP-synthesis to promote apoptosis in colon cancer models. Oncotarget. 2015; 6:11806-11819. https://doi.org/10.18632/oncotarget.368810.
33. Safdie F, Brandhorst S, Wei M, Wang W, Lee C, Hwang S, Conti PS, Chen TC, Longo VD. Fasting enhances the response of glioma to chemo- and radiotherapy. PLoS One. 2012; 7:e44603.
34. Lee C, Raffaghello L, Brandhorst S, Safdie FM, Bianchi G, Martin-Montalvo A, Pistoia V, Wei M, Hwang S, Merlino A, Emionite L, de Cabo R, Longo VD. Fasting cycles retard growth of tumors and sensitize a range of cancer cell types to chemotherapy. Sci Transl Med. 2012; 4:124ra127.
35. D’Aronzo M, Vinciguerra M, Mazza T, Panebianco C, Saracino C, Pereira SP, Graziano P, Pazienza V. Fasting cycles potentiate the efficacy of gemcitabine treatment in in vitro and in vivo pancreatic cancer models. Oncotarget. 2015; 6:18545-18557. https://doi.org/10.18632/oncotarget.418610.
36. Leelatian N, Doxie DB, Greenplate AR, Mobley BC, Lehman JM, Sinnaeve J, Kauffmann RM, Werkhaven JA, Mistry AM, Weaver KD, Thompson RC, Massion PP, Hooks MA, et al. Single cell analysis of human tissues and solid tumors with mass cytometry. Cytometry B Clin Cytom. 2017; 92:68-78.
37. Luo J, Yin X, Sanchez A, Tripathy D, Martinez J, Grammas P. Purification of endothelial cells from rat brain. Methods Mol Biol. 2014; 1135:357-364.
38. Bianchi F, Maioli M, Leonardi E, Olivi E, Pasquinelli G, Valente S, Mendez AJ, Ricordi C, Raffaini M, Tremolada C, Ventura C. A new nonenzymatic method and device to obtain a fat tissue derivative highly enriched in pericyte-like elements by mild mechanical forces from human lipoaspirates. Cell Transplant. 2013; 22:2063-2077.

