INTRODUCTION
Radiation therapy is the primary treatment for patients with head and neck cancer, but it leads to the development of oral mucositis accompanied by tissue structure destruction and functional alteration. It should be noted that managing oral mucositis should be based on the pathobiology of the radiation-induced oral mucositis [1]. Among other symptoms, dry mouth and loss of taste can also occur during radiotherapy and occasionally persist for a long time after the therapy concludes [2]. It is known that irradiated oral mucosa, even after the complete healing of mucositis, become atrophic and susceptible to injury. Although we are responsible for managing the oral mucosa after radiotherapy, there is little information on the molecular basis for this vulnerability of irradiated oral mucosa.
In the initiation stage of radiation-induced oral mucositis, DNA damage and reactive oxygen species (ROS) release activate apoptotic factors such as p53 and the Bcl-2 family in the affected cells [3, 4]. Recently, Hif-1a seems to be a key transcription factor in activating apoptotic cell death in mucositis, [1, 5] and if Hif-1a plays a central role in the development of radiation-induced mucositis, it may also be implicated in the vulnerability of normal oral mucosa that are exposed to radiotherapy. However, only few reports have studied Hif-1a expression in oral mucosa after radiotherapy, with one report by Gruber et al. describing that Hif-1a played a critical role in the occurrence of radiation-induced oral mucositis [1, 6].
Recently, it has been reported that alpha lipoic acid (ALA) protects against radiation-induced normal tissue injury and dysfunction [7-9]. ALA is a strong antioxidant with high reactivity to free radicals, and it elevates tissue levels of glutathione [10]. ALA has been demonstrated to be effective in preventing pathological processes such as ischemia-reperfusion injury [11], diabetes [12], hypertension, and radiation injury [13].
In the present study, we aimed to investigate the protective effects of ALA on radiation-induced oral mucositis. We assumed that Hif-1a expression would be positively correlated with functional and structural tissue injury and apoptosis by radiation and that ALA would mitigate this injury.
RESULTS
ALA protected against radiation-induced oral mucosa injury
Figure 1A shows the typical damage to the mucosa with destruction of the squamous epithelium, the atrophic changes in the epithelium, the sterilization of mucosal stem cells, and the resulting edematous cellular morphology in the RT group, whereas the same cell types were less frequent in the ALA+RT group. We evaluated the histological changes by measuring the squamous epithelium height in the oral mucosa. At all time points, we found no significant differences between CON and ALA, but the height decreased significantly in RT compared with CON on days 4 and 7 (CON vs. RT, day 4: 180.127 μm vs. 49.088 μm; day 7: 203.277 μm vs. 0 μm, p < 0.05; Figure 1B). In particular, radiation resulted in a loss of the entire thickness of the epithelium with a small quantity of muscle tissue on day 7. The height decrease in ALA+RT was significantly lower than the decrease in RT on days 4 and 7 (RT vs. ALA+RT, day 4: 49.088 μm vs. 103.58 μm; day 7: 0 μm vs. 58.286 μm, p < 0.05; Figure 1B). On day 28 and 56, there is no significant difference between ALA+RT and RT groups.
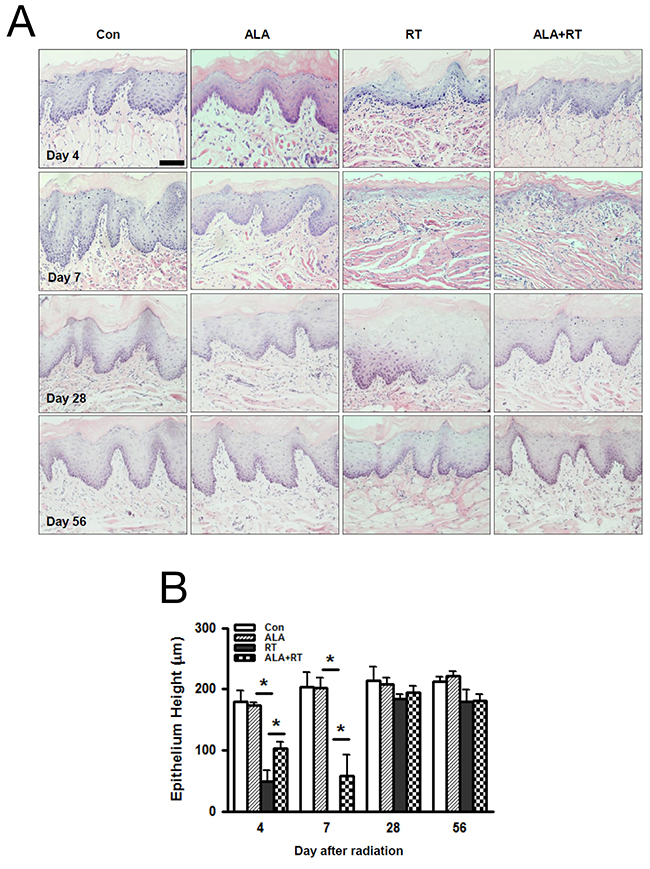
Figure 1: Histopathological changes in the oral mucosa 4, 7, 28, and 56 days after irradiation. ALA decreases pathological changes in the oral mucosa after irradiation. All tissues were sectioned and stained with H&E (A). Exposing the head and neck area to irradiation resulted in a shortened squamous epithelium, but this was decreased in ALA+RT (B). Con, control; ALA, ALA only; RT, irradiated; ALA+RT, ALA before irradiation. Scale bar, 100 μm.
ALA reduced Hif-1a and GLUT1 expression in the germinal and epithelial layers
Our results showed that Hif-1a expression was significantly increased in RT on days 4, 7, and 28, but it decreased significantly with ALA pretreatment; we found no significant differences between RT and ALA+RT on day 56. We did observe positive signals for Hif-1a in the germinal and epithelial layers of the squamous epithelium, and the signal was particularly abundant on day 4; on day 7, the expression was mainly in the germinal layer in ALA+RT (Figure 2B). Similar to Hif-1a, radiation also induced GLUT1 expression at all time points, and the expression levels peaked for the samples from day 28 in RT. ALA pretreatment significantly reduced expression levels on days 7, 28, and 56 (Figure 2A). GLUT1 levels clearly showed membrane-associated expression in the germinal layer of the squamous epithelium in RT at all time points. In particular, GLUT1 expression gradually increased in RT, peaked on day 28, and decreased on day 56, and ALA pretreatment attenuated this GLUT1 expression increase on days 4, 7, and 28; however, it was not significant on day 56 (Figure 2C).
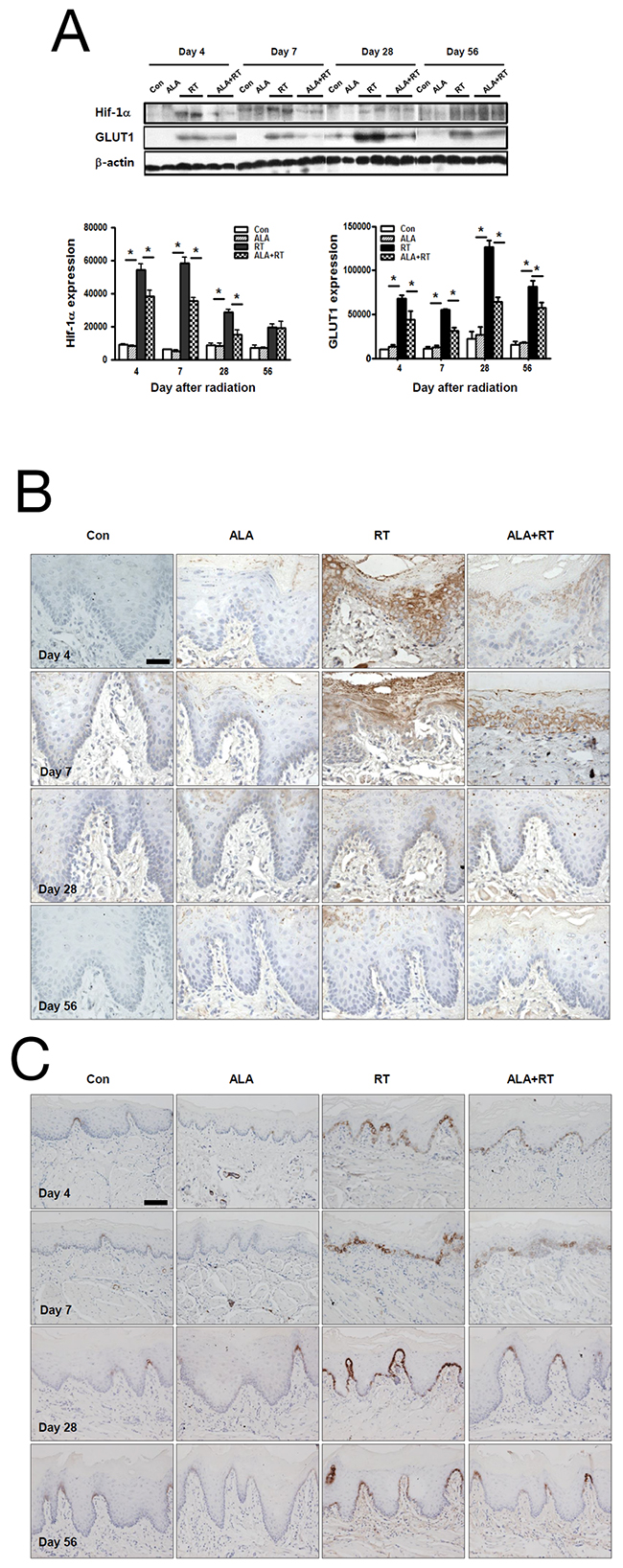
Figure 2: ALA decreases Hif-1a and GLUT1 expression in the oral mucosa. Western blotting was performed with anti-Hif-1a and anti-GLUT1 antibodies. Hif-1a and GLUT1 expression were induced in RT at days 4, 7, and 28 after irradiation, and this expression occurred on days 7, 28, and 56 in CON and ALA+RT. B-actin was used as the loading control (A). Strong Hif-1a–positive signals were detected in RT on days 4 and 7, and ALA pretreatment decreased the signals (B). GLUT1-positive signals were detected in RT on all days, and ALA pretreatment decreased the signals (C). Con, control; ALA, ALA only; RT, irradiated; ALA+RT, ALA before irradiation. Scale bar, 50 μm for Hif-1a and 100 μm for GLUT1.
ALA reduced protein expression of pro-apoptotic factors in the oral mucosa after radiation
We also investigated the apoptotic activity in the oral mucosa. Figure 3 shows that phosphorylated p53 and Bax protein expression were significantly increased in the radiation group on days 4 and 7 and day 4, respectively, but these expression increases were significantly dampened by ALA pretreatment. Phosphorylated p53 level showed no significant differences on day 28 and 56 between ALA+RT and RT groups. In contrast, ALA pretreatment significantly reduced Bax protein expression only on day 4 after the radiation treatment. We observed only negligible signals for phosphorylated p53 and Bax in CON and ALA at all time points.
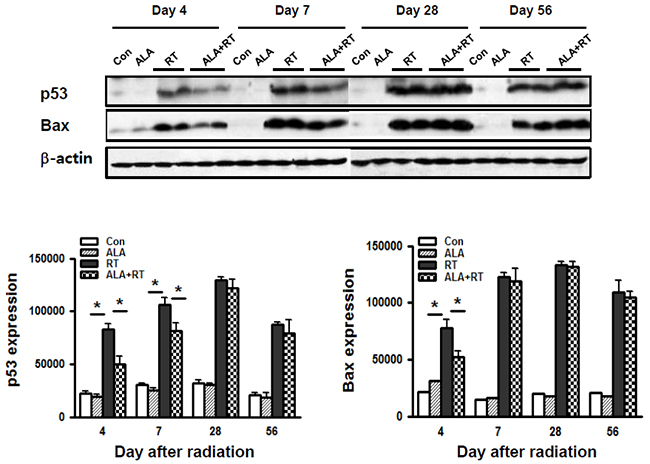
Figure 3: ALA decreased radiation-induced apoptotic signals in the oral mucosa. Western blotting was performed using anti-p-p53 and Bax. In RT, p53 expression was induced on days 4 and 7, and Bax expression was induced by day 4. β-actin was used as the loading control. Con, control; ALA, ALA only; RT, irradiated; ALA+RT, ALA before irradiation.
ALA decreased apoptosis of the squamous epithelium in the oral mucosa
Apoptosis is one of the major pathogenic aspects of mucositis, and the degree of apoptosis reflects the degree of mucositis. We assessed apoptosis levels using TUNEL staining, and at all time points, we found no significant differences between CON and ALA; we detected most of the positive signals at the germinal layer of the squamous epithelium and some at the functional epithelial layer (Figure 4A). In RT, we detected the TUNEL-positive signal in the new germinal layer on day 28, and this positive signal moved to the epithelial layer on day 56. In ALA+RT, we detected the TUNEL-positive signal in the germinal layer on day 7, and this signal also moved to the epithelium on day 28, followed by nearly disappearing on day 56 (Figure 4A). These TUNEL-positive signals increased significantly in RT at all time points; notably, the image from RT on day 7 shows that the squamous epithelium was appreciably injured by the radiation. ALA+RT showed a significant reduction in TUNEL-positive signals on day 7 compared to RT (Figure 4B).
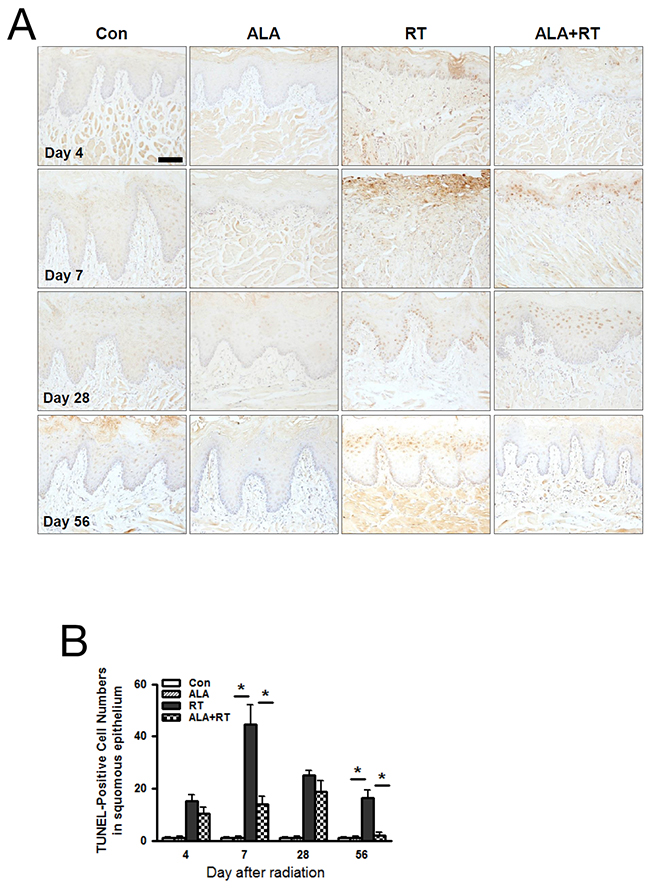
Figure 4: ALA decreased radiation-induced cell death in the squamous epithelium of the oral mucosa. Cell death was analyzed with the TUNEL assay (A). Microscopic image shows increased cell death in RT compared with Con and ALA+RT at 4 and 7 days after irradiation (B). Scale bar, 100 μm.
DISCUSSION
Apart from the targeted cancer cells, highly proliferative normal cells are the most affected by radiation therapy. The oral mucosa are particularly vulnerable to radiation because the mucosa consists of cells with highly self-renewing activity, and with this high turnover rate, the lining replaces itself every 7 to 14 days. Oral mucositis by radiation is an acute and transient adverse effect, and its onset from radiotherapy is a biologically complex process [17, 18]. Although radiation-induced oral mucositis spontaneously resolves 3–4 weeks after treatment, oral mucosa that are exposed to radiation appear to become atrophic and susceptible to injury even after they are completely re-epithelialized [18]. Cells in the oral mucosa that are harmed by radiation include squamous epithelial cells and submucosal stromal cells, which comprise fibroblasts and vascular endothelial cells. Radiation and radiation-induced ROS generation damage the DNA in the cell itself, lead to apoptosis or necrosis of squamous epithelial cells, and inhibit cell proliferation, which triggers the breakdown of the squamous cell layer [17, 18].
In the acute phase after radiation, inflammation by cytokines released from the basal epithelium and/or the adjacent connective tissue causes increased subepithelial vascularity and reduced epithelium thickness. These transformations are known to result in signs of local hypoxia [1, 6]. When cells are exposed to extreme or prolonged hypoxia such as with irradiation, Hif-1a does not protect cells from the adaptive mechanisms, but the cells can face Hif-1a-involved apoptosis [19]. Hypoxic conditions can induce apoptosis, but the role of Hif-1a in this process is still controversial; some studies reported that Hif-1a mediates hypoxia-induced cell death by activating the transcription of many pro-apoptotic genes such as NIX and NIP3, part of the pro-apoptotic Bcl-2 family [20, 21], and others report that Hif-1a-dependent p53 could mediate the induction of apoptosis in hypoxia [19, 22]. It was also shown that hypoxia could stabilize p53, including increased p53 phosphorylation [23-25]. Researchers have also detected a direct association between Hif-1a and p53 under hypoxic conditions [24, 26]. In hypoxia, p53 is stabilized, and p53 is a significant transcription factor in activating cell death-initiating genes including Bax and in arresting p21 growth in response to DNA damage or stress [21, 23, 25]. The findings from these previous studies are similar to our results. As shown in Figures 2A, 2B, and 3, increased Hif-1a expression correlated highly with increased p53 phosphorylation and Bax expression in RT on days 4 and 7 after radiation. As noted above, p53 mediated apoptosis in RT might be dependent on Hif-1 alpha. Our results indicate that head and neck radiotherapy leads to hypoxia of the oral mucosa, and this hypoxia increases p53 stabilization by increasing phosphorylated p53 levels, leading to increased pro-apoptotic Bax expression, and, finally, oral mucositis. However, ALA might be involved in ameliorating these expression levels in the acute phase. This result suggests that ALA might be involved in Hif-1a-p53-dependent apoptotic epithelial cell death in the oral mucosa after radiation. However, most studies have shown that elevated Hif-1a expression in tumor cells confers resistance to hypoxic exposure that ultimately contributes to tumor resistance to radiotherapy and chemotherapy [20-22]. Because HIF-1a plays an important role in protecting solid tumors against hypoxia by promoting angiogenesis, inducing growth factor expression, preventing apoptosis, and increasing anaerobic metabolism [27, 28]. In particular, HIF-1a can confer resistance to apoptosis under hypoxia by increasing GLUT-1 [29]. Interestingly, this coincides with our findings shown in Figures 2A and 2C. As mentioned above, radiation significantly increased Hif-1a expression, but ALA pretreatment reduced the expression. Radiation also significantly increased GLUT-1 expression, and ALA also reduced this expression at all time points. Excess radiation exposure can reduce the blood and nutrient supply to abnormal blood vessels in the submucosa, which can lead to epithelial cell death and damage to the basal layer. Most cells have defense mechanisms to protect themselves against insults, and the increased Hif-1a and GLUT-1 expression could be one of these mechanisms for protecting against radiation insult. Hif-1a-GLUT-1-dependent pathway might be a defense response to overcome Hif-1a-p53-dependent apoptotic epithelial cell death. However, ALA pretreatment might create a less hypoxic environment, which could lead to less Hif-1a-p53-dependent apoptotic machinery and less Hif-1a-GLUT-1-dependent adaptation.
Interestingly, ALA might be related to epithelial and basal layer regeneration. The TUNEL assay in Figure 4 shows increased cell death and a shortened epithelial layer in RT and ALA+RT compared with CON in the early period (days 4 and 7). However, in ALA+RT, the TUNEL-positive cells were detected only at the basal layer on day 7; these cells were moving toward the outer layer on day 28, and they had disappeared by day 56. This cell movement could have been caused by the regeneration of the epithelial layer. When the oral mucosa were exposed to radiation, the epithelial layer including the basal layer was destroyed, but the basal layer regenerated and the epithelial layer was generated from the basal layer; that is, the TUNEL-positive cells moved toward the outer layer over time. Thus, ALA+RT showed more advanced repair of the basal and epithelial layers than did RT, which suggests that ALA could be involved in the oral epithelial regeneration after radiation.
There were a few limitations to this study. First, the single dose of 18Gy that we used is not relevant for treating head and neck cancer in the clinic; because the doses we give patients are limited by the response of normal tissue, fractionated doses are more common and more beneficial in treatment. In addition, we were focused on the protective effect of ALA against radiation-induced oral injury, and thus, it was necessary to give the rats the high dose. Second, we should have verified that ALA did not promote cancer growth or prevent radiation-induced cancer cell death. Although studies have reported on ALA and cancer cell growth and death, the role of ALA in cancer cells is still controversial. We designed this work as a feasibility study of cancer cell growth and death and ALA treatment.
In conclusion, our results show that ALA could be used to ameliorate radiation-induced oral mucositis with head and neck cancer.
MATERIALS AND METHODS
Ethics statement
The Gyeongsang National University Institutional Animal Care & ethics committee specifically approved this study (GLA-120120-R0002).
Radiation exposure
We assigned male Sprague–Dawley rats (230–250 g; Koatech Inc., Peongtaek, Korea) to the following groups: control, n=12 (CON); irradiated, n=16 (RT); ALA administered before irradiation, n=16 (ALA+RT); and ALA administered alone, n=12 (ALA). We administered the ALA (100 mg/kg, i.p., Bukwang Pharmaceutical Co., Seoul, Korea) 24 h and 30 min before irradiation, and we chose the dose and frequency based on previous studies [14-16]. The neck area was evenly irradiated with 2 Gy/min (total dose, 18 Gy) using a photon 6-MV linear accelerator (21EX, Varian, Palo Alto, CA, USA). A 3-cm block of Lucite was positioned above the head and neck to provide adequate buildup and facilitate even radiation distribution. Each rat was exposed to a single dose of radiation and sacrificed 4, 7, 28, or 56 days after radiation.
Histopathology: height of the squamous epithelium
Tissues were fixed in 4% paraformaldehyde in 0.1 M PBS, embedded in paraffin, and cut into 5-μm sections. The sections were stained with hematoxylin and eosin (H&E) and Masson’s trichrome (Masson’s Trichrome Kit, Sigma Diagnostics, St. Louis, MO, USA). We measured the squamous epithelium height (from the top of the epithelium to the germinal layer) under light microscopy (×200) using a calibrated micrometer and NIS Elements BR3.2 software (Nikon, Tokyo, Japan). We measured and averaged 10 intact oral (especially buccal area) mucosa sections with squamous epithelium samples per rat for each sample.
TUNEL assay
We assessed the degree of apoptosis using the TUNEL assay (Roche, Indianapolis, IN, USA), conducting semi-quantitative analysis by counting the TUNEL-positive cells per field at 400× magnification; we randomly selected at least 10 areas in the squamous epithelium of each oral mucosa per slide for analysis, and we considered the mean number of brown cells in the selected fields to be the number of TUNEL-positive cells. A blind observer analyzed the signals in the 10 randomly selected fields using NIS Elements BR3.2.
Immunoblotting
The tissues were homogenized in a lysis buffer. Fifty micrograms of proteins were loaded on a sodium dodecyl sulfate-polyacrylamide gel. The blots were probed with primary antibodies to polyclonal anti-hypoxic inducible factor-1 alpha (Hif-1a (diluted 1:1000; ab2185, Abcam, Cambridge, MA, USA), anti-phosphorylated p53 (diluted 1:500; #9284, Cell Signaling, Danvers, MA, USA), anti-Bax (diluted 1:500; #2772, Cell Signaling), and monoclonal anti-glucosetransporter (GLUT1; diluted 1:1000; ab40084, Abcam) at 4°C overnight. The primary antibody was visualized using secondary antibodies with an enhanced chemiluminescence kit (Amersham Pharmacia Biotech, Piscataway, NJ, USA).
Immunohistochemistry
After deparaffinization, the sections were incubated with primary antibodies to polyclonal anti-Hif-1a (diluted 1:100; ab2185, Abcam), monoclonal anti-GLUT1 (diluted 1:200; ab40084, Abcam), monoclonal anti-Ki67 (diluted 1:50; ab16667, Abcam), monoclonal anti-PCNA (diluted 1:200; sc-56, Santa Cruz Biotechnology, Dallas, TX, USA), and monoclonal anti-8-hydroxydeoxyguanosine (diluted 1:500; ab62623, Abcam); with biotin-conjugated secondary IgG (diluted 1:200; Vector Laboratories, Burlingame, CA, USA); with the avidin-biotin-peroxidase complex (ABC Elite Kit, Vector Laboratories); and with diaminobenzidine tetrahydrochloride. We then visualized the sections under light microscopy and captured and analyzed the digital images.
Statistical analysis
We conducted all statistical analyses using SPSS (SPSS Inc., Chicago, IL, USA), and results are presented as mean ± standard error of the mean. We assessed the differences between groups using one-way analysis of variance followed by the Student–Newman–Keuls test, and we considered a P-value of less than 0.05 significant.
ACKNOWLEDGMENTS
This study was supported by the Basic Science Research Program, through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT, and Future Planning (NRF-2016R1A2B4013000).
This study was also sponsored by the Leading Foreign Research Institute Recruitment Program, through the Na¬tional Research Foundation of Korea (NRF), funded by the Minis-try of Education, Science and Technology (MEST) (2012K1A4A3053142).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest. The funding sponsors had no role in the design of the study; collection, analyses, or interpretation of data; writing of the manuscript; or in deciding to publish the results.
GRANT SUPPORT
No financial or material support has been received for this work; moreover, the authors declare no financial interests in companies or other entities that could have an interest in the information within this contribution.
REFERENCES
1. Gruber S, Hamedinger D, Bozsaky E, Schmidt M, Wolfram K, Haagen J, Habelt B, Puttrich M, Dörr W. Local hypoxia in oral mucosa (mouse) during daily fractionated irradiation - Effect of pentoxifylline. Radiother Oncol. 2015;116:404-8.
2. Vissink A, Jansma J, Spijkervet FK, Burlage FR, Coppes RP. Oral sequelae of head and neck radiotherapy. Crit Rev Oral Biol Med. 2003;14:199-212.
3. Dhanasekaran DN, Reddy EP. JNK signaling in apoptosis. Oncogene. 2008;27:6245-51.
4. Rodriguez-Rocha H, Garcia-Garcia A, Panayiotidis MI, Franco R. DNA damage and autophagy. Mutat Res. 2011;711:158-66.
5. Schmidinger M. Understanding and managing toxicities of vascular endothelial growth factor (VEGF) inhibitors. EJC Suppl. 2013;11:172-91.
6. Eltzschig HK, Carmeliet P. Hypoxia and inflammation. N Engl J Med. 2011;364:656-65.
7. Jeong BK, Song JH, Jeong H, Choi HS, Jung JH, Hahm JR, Woo SH, Jung MH, Choi BH, Kim JH, Kang KM. Effect of alpha-lipoic acid on radiation-induced small intestine injury in mice. Oncotarget. 2016;7:15105-17. http://doi.org/10.18632/oncotarget.7874.
8. Jung JH, Jung J, Kim SK, Woo SH, Kang KM, Jeong BK, Jung MH, Kim JH, Hahm JR. Alpha lipoic acid attenuates radiation-induced thyroid injury in rats. PLoS One. 2014;9:e112253.
9. Kim JH, Kim KM, Jung MH, Jung JH, Kang KM, Jeong BK, Kim JP, Park JJ, Woo SH. Protective effects of alpha lipoic acid on radiation-induced salivary gland injury in rats. Oncotarget. 2016;7:29143-53. http://doi.org/10.18632/oncotarget.8661.
10. Bilska A, Wlodek L. Lipoic acid - the drug of the future? Pharmacol Rep. 2005;57:570-7.
11. Sehirli O, Sener E, Cetinel S, Yuksel M, Gedik N, Sener G. Alpha-lipoic acid protects against renal ischaemia-reperfusion injury in rats. Clin Exp Pharmacol Physiol. 2008;35:249-55.
12. Hurdag C, Ozkara H, Citci S, Uyaner I, Demirci C. The effects of alpha-lipoic acid on nitric oxide synthetase dispersion in penile function in streptozotocin-induced diabetic rats. Int J Tissue React. 2005;27:145-50.
13. Demir U, Demir T, Ilhan N. The protective effect of alpha-lipoic acid against oxidative damage in rabbit conjunctiva and cornea exposed to ultraviolet radiation. Ophthalmologica. 2005;219:49-53.
14. Huh JW, Hong SB, Kim MJ, Lim CM, Koh Y. The Efficacy of alpha-lipoic Acid on the Endotoxin-induced Acute Lung Injury. Tuberc Respir Dis 2007;62:105-12. https://doi.org/10.4046/trd.2007.62.2.105.
15. Cremer DR, Rabeler R, Roberts A, Lynch B. Long-term safety of alpha-lipoic acid (ALA) consumption: A 2-year study. Regul Toxicol Pharmacol. 2006;46:193-201.
16. Wongmekiat O, Leelarungrayub D, Thamprasert K. Alpha-lipoic acid attenuates renal injury in rats with obstructive nephropathy. Biomed Res Int. 2013;2013:138719.
17. Sonis ST. The pathobiology of mucositis. Nat Rev Cancer. 2004;4:277-84.
18. Tetsuro Ikebe KY, Yasuo Takamune, Hideki Nakayama, Masanori Shinohara. Reduced expression of nuclear factor kB in oral mucosa undergoing preoperative chemoradiotherapy. Oral Sci Int. 2012;9:33-7.
19. Piret JP, Mottet D, Raes M, Michiels C. Is HIF-1alpha a pro- or an anti-apoptotic protein? Biochem Pharmacol. 2002;64:889-92.
20. Sowter HM, Ratcliffe PJ, Watson P, Greenberg AH, Harris AL. HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res. 2001;61:6669-73.
21. Zhang X, Kon T, Wang H, Li F, Huang Q, Rabbani ZN, Kirkpatrick JP, Vujaskovic Z, Dewhirst MW, Li CY. Enhancement of hypoxia-induced tumor cell death in vitro and radiation therapy in vivo by use of small interfering RNA targeted to hypoxia-inducible factor-1alpha. Cancer Res. 2004;64:8139-42.
22. Carmeliet P, Dor Y, Herbert JM, Fukumura D, Brusselmans K, Dewerchin M, Neeman M, Bono F, Abramovitch R, Maxwell P, Koch CJ, Ratcliffe P, Moons L, et al.. Role of HIF-1alpha in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature. 1998;394:485-90.
23. Graeber TG, Peterson JF, Tsai M, Monica K, Fornace AJ Jr, Giaccia AJ. Hypoxia induces accumulation of p53 protein, but activation of a G1-phase checkpoint by low-oxygen conditions is independent of p53 status. Mol Cell Biol. 1994;14:6264-77.
24. Woods DB, Vousden KH. Regulation of p53 function. Exp Cell Res. 2001;264:56-66.
25. Yun Z, Glazer PM. Tumor suppressor p53 stole the AKT in hypoxia. J Clin Invest. 2015;125:2264-6.
26. An WG, Kanekal M, Simon MC, Maltepe E, Blagosklonny MV, Neckers LM. Stabilization of wild-type p53 by hypoxia-inducible factor 1alpha. Nature. 1998;392:405-8.
27. Giaccia A, Siim BG, Johnson RS. HIF-1 as a target for drug development. Nat Rev Drug Discov. 2003;2:803-11.
28. Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721-32.
29. Kilic M, Kasperczyk H, Fulda S, Debatin KM. Role of hypoxia inducible factor-1 alpha in modulation of apoptosis resistance. Oncogene. 2007;26:2027-38.

