INTRODUCTION
Bone is a very common site of distant metastases in nasopharyngeal cancer patients [1–3]. Bone metastasis accounts for approximately 10–16% of all patients with nasopharyngeal cancer and 60–80% of patients with distant metastasis at initial diagnosis [1–3]. The selected treatment strategies, such as radiotherapy, chemotherapy, and targeted therapy is mainly dependent on the TNM staging. If bone metastasis is found, the treatment strategies may change significantly. The accurate assessment of bone metastasis is necessary for M staging and the selection of optimal treatment.
Various techniques of diagnostic imagings, such as bone scintigraphy (BS), positron emission tomography (PET), PET/computed tomography (CT), and magnetic resonance imaging are widely used for the assessment of bone metastasis. 99mTc-phosphonate BS is most widely used to assess bone metastasis for many decades because of its ability to evaluate the entire skeleton at a relatively low cost. BS relies on the osteoblastic response to bone destruction by tumor cells [4]. But the high false positive rate of BS may be caused by some benign processes (osteoarthritis, fractures, degenerative changes, etc) [5]. As a functional imaging modality,18FDG PET can detect potential tumor activity and facilitate earlier detection of bone metastatic lesions. The introduction of PET-CT has combined the functional imaging of PET with the anatomic imaging of CT into a single examination. Several studies have validated the potential value of 18FDG PET/PET-CT and PBS for the assessment of bone metastases in nasopharyngeal cancer [1–3, 6–8]. However, the findings of 18FDG PET/PET-CT and BS are variable or incongruent. Therefore, we undertook a meta-analysis to compare the diagnostic efficacy of 18FDG PET/PET-CT and PBS in detecting bone metastases of nasopharyngeal cancer patients.
RESULTS
Study selection and description
The flow chart of search for eligible studies was showed in Figure 1. After independent review, 7 articles dealing with the comparison of 18FDG PET/PET-CT and BS for detecting bone metastases of nasopharyngeal cancer patients were eligible for this meta-analysis. Of seven articles, one article [9] was excluded because the data was already reported in an included article [1]. Consequently, 6 articles [1–3, 6–8] were included in this meta-analysis (Figure 1). A total of 1238 patients were analyzed for the diagnostic efficacy of 18FDG PET/PET-CT and BS (Table 1). In five articles (83.3%), the study design was prospective.
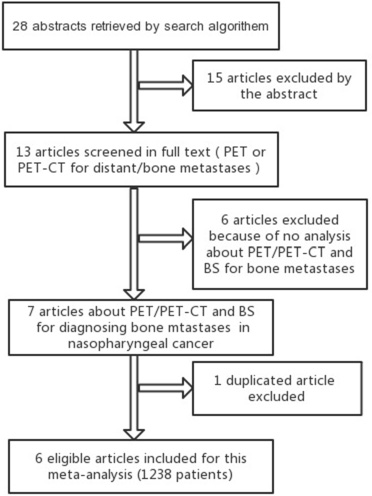
Figure 1: Shows the flow chart of search for eligible studies.
Table 1: The clinical characteristics of all included studies
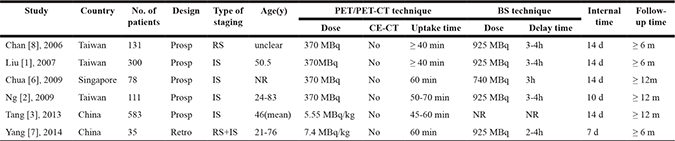
#IS = Initial Staging, RS = Restaging, Prosp = Prospective, Retro = Retrospective, NR = Not Reported, CE-CT = Contrast Enhanced-Computed Tomography.
Study quality
Quality assessment of all included studies is shown in Table 2. Overall, the quality of the included studies was satisfactory. For all six studies, the results of 18FDG PET/PET-CT and BS was interpreted without any knowledge of the reference standard. But the reference standard wasn’t executed without any knowledge of the results of 18FDG PET/PET-CT and BS in all included studies.
Table 2: QUADAS-2 results for all included studies
Studies |
Risk of bias |
Applicability concerns |
|||||
|---|---|---|---|---|---|---|---|
Patient selection |
Index test |
Reference standard |
Flow and timing |
Patient selection |
Index test |
Reference standard |
|
Chan [8], 2006 |
HR |
LR |
HR |
LR |
HR |
LR |
LR |
Liu [1], 2007 |
LR |
LR |
HR |
LR |
LR |
LR |
LR |
Chua [6], 2009 |
LR |
LR |
HR |
LR |
LR |
LR |
LR |
Ng [2], 2009 |
LR |
LR |
HR |
LR |
LR |
LR |
LR |
Tang [3], 2013 |
LR |
LR |
HR |
LR |
LR |
LR |
LR |
Yang [7], 2014 |
HR |
LR |
HR |
LR |
HR |
LR |
LR |
#HR = high risk, LR = high risk.
Accuracy of 18F-FDG PET/PET-CT and BS
All included studies
The forest plots of sensitivity and specificity for 18FDG PET/PET-CT and BS from all 6 studies (1238 patients) were shown in Figure 2A, 2B. When considering all 6 studies (1238 patients), the pooled sensitivity, specificity, DOR, PLR and NLR of 18FDG PET/PET-CT were 0.81 (95% confidence interval [CI] = 0.70 to 0.98), 0.99 (95% CI = 0.98 to 0.99), 312 (95% CI = 144 to 676), 58.6 (95% CI = 33.5 to 102.7) and 0.19 (95% CI = 0.11 to 0.31), respectively, and of BS were 0.39 (95% CI = 0.26 to 0.54), 0.98 (95% CI = 0.96 to 0.99), 32 (95% CI = 16 to 64), 19.9 (95% CI = 10.3 to 38.7) and 0.62 (95% CI = 0.49 to 0.78), respectively (Table 3).
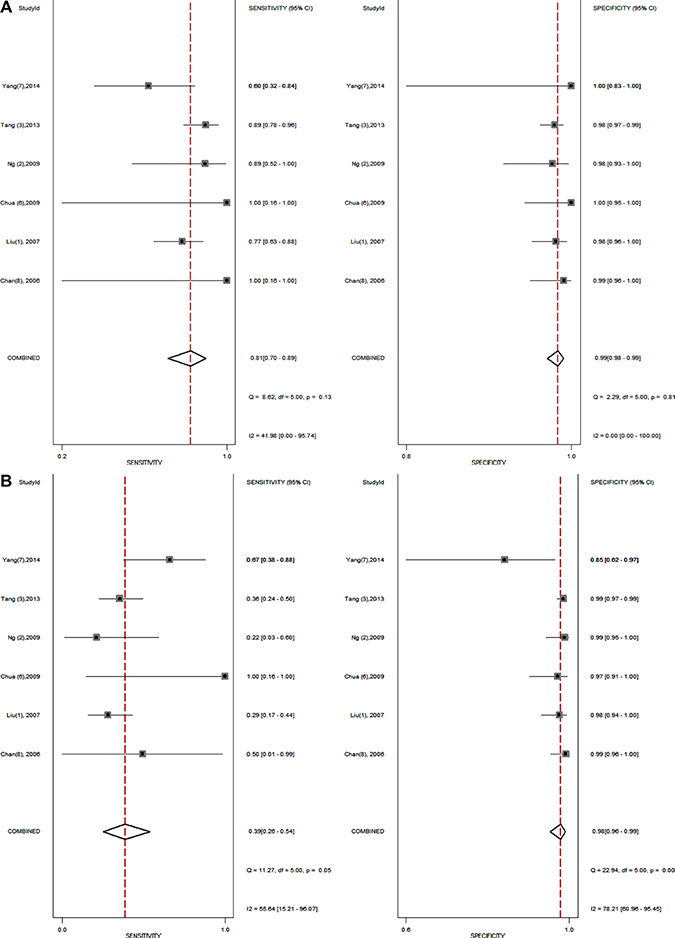
Figure 2: (A–B) shows the forest plot of sensitivity and specificity for 18FDG PET/PET-CT (A) and bone scintigraphy (B) from all 6 studies.
Table 3: Accuracy of 18FDG PET-CT and bone scan for detection of bone metastases in nasopharyngeal cancer patients
Imaging Modalites |
No. of Studies (No. of Patients) |
Sensitivity (95% CI) |
specificity(95% CI) |
DOR (95% CI) |
PLR (95% CI) |
NLR (95% CI) |
|---|---|---|---|---|---|---|
All Studies |
||||||
18FDG PET/PET-CT |
6 (1238) |
0.81 (0.70–0.89) |
0.99 (0.98–0.99) |
312 (144–676) |
58.6 (33.5–102.7) |
0.19 (0.11–0.31) |
Bone Scan |
6 (1238) |
0.39 (0.26–0.54) |
0.98 (0.96–0.99) |
32 (16–64) |
19.9 (10.3–38.7) |
0.62 (0.49–0.78) |
Prospective Studies |
||||||
18FDG PET/PET-CT |
5 (1203) |
0.85 (0.75–0.91) |
0.99 (0.98–0.99) |
373 (173–806) |
57.7 (35.1–94.8) |
0.15 (0.09–0.26) |
Bone Scan |
5 (1203) |
0.34 (0.26–0.43) |
0.98 (0.98–0.99) |
34 (18–66) |
23.2 (13.0–41.5) |
0.67 (0.59–0.77) |
PET-CT system |
||||||
18FDG PET-CT |
4 (807) |
0.83 (0.65–0.99) |
0.99 (0.97–0.99) |
351 (115–1077) |
61.6 (24.8–153.1) |
0.18 (0.08–0.38) |
Bone Scan |
4 (807) |
0.46 (0.28–0.65) |
0.98 (0.93–0.99) |
34 (14–78) |
18.7 (7.7–45.2) |
0.56 (0.39–0.79) |
Initial Staging |
||||||
18FDG PET/PET-CT |
4 (1072) |
0.84 (0.75–0.91) |
0.98 (0.97–0.99) |
342 (158–740) |
54.0 (32.3–90.3) |
0.16 (0.10–0.26) |
Bone Scan |
4 (1072) |
0.33 (0.25–0.42) |
0.98 (0.97–0.99) |
32 (16–62) |
21.5 (11.8–39.1) |
0.68 (0.59–0.77) |
Prospective studies
When considering 5 prospective studies (1203 patients), the pooled sensitivity, specificity, DOR, PLR and NLR of 18FDG PET/PET-CT were 0.85 (95% CI = 0.75 to 0.91), 0.99 (95% CI = 0.98 to 0.99), 373 (95% CI = 173 to 806), 57.7 (95% CI = 35.1 to 94.8) and 0.15 (95% CI = 0.09 to 0.26), respectively, and of BS were 0.34 (95% CI = 0.26 to 0.43), 0.98 (95% CI = 0.98 to 0.99), 34 (95% CI = 18 to 66), 23.2 (95% CI = 13.0 to 41.5) and 0.67 (95% CI = 0.59 to 0.77), respectively (Table 3).
PET-CT system
When considering 4 studies with PET-CT system (807 patients), the pooled sensitivity, specificity, DOR, PLR and NLR of 18FDG PET-CT were 0.83 (95% CI = 0.65 to 0.99), 0.99 (95% CI = 0.97 to 0.99), 351 (95% CI = 115 to 1077), 61.6 (95% CI = 24.8 to 153.1) and 0.18 (95% CI = 0.08 to 0.38), respectively, and of BS were 0.46 (95% CI = 0.28 to 0.65), 0.98 (95% CI = 0.93 to 0.99), 34 (95% CI = 14 to 78), 18.7 (95% CI = 7.7 to 45.2) and 0.56 (95% CI = 0.39 to 0.79), respectively (Table 3).
Initial staging
When considering 4 studies at initial staging (1072 patients), the pooled sensitivity, specificity, DOR, PLR and NLR of 18FDG PET/PET-CT were 0.84 (95% CI = 0.75 to 0.91), 0.98 (95% CI = 0.97 to 0.99), 342 (95% CI = 158 to 740), 54.0 (95% CI = 32.3 to 90.3) and 0.16 (95% CI = 0.10 to 0.26), respectively, and of BS were 0.33 (95% CI = 0.25 to 0.42), 0.98 (95% CI = 0.97 to 0.99), 32 (95% CI = 16 to 62), 21.5 (95% CI = 11.8 to 39.1) and 0.68 (95% CI = 0.59 to 0.77), respectively (Table 3).
SROC curves
The SROC curve presents a global summary of test performance, and shows the tradeoff between sensitivity and specificity. The SROC curves of 18FDG PET/PET-CT and BS from all 6 studies (1238 patients) were shown in Figure 3A, 3B. Overall weight area under the SROC curves for 18FDG PET/PET-CT and BS was 0.98 (95% CI = 0.97 to 0.99) and 0.84 (95% CI = 0.81 to 0.87).
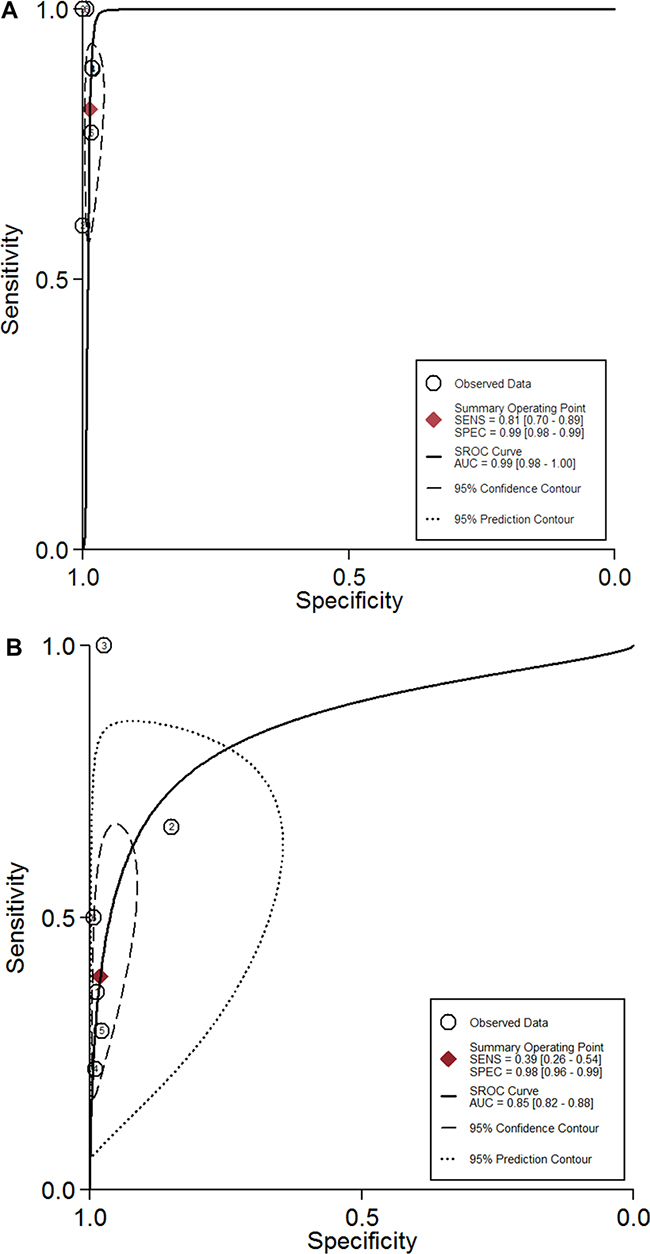
Figure 3: (A–B) shows the SROC curves of 18FDG PET/PET-CT (A) and bone scintigraphy (B) from all 6 studies.
DISCUSSION
In the past twenty years, 18FDG PET or PET-CT entered into clinical usage as a practical imaging technique for distant metastasis staging of nasopharyngeal cancer [1–3, 6–8]. The previous meta-analysis of 8 studies (770 patients) showed that 18FDG PET/PET-CT had a sensitivity and a specificity of 0.82 (95% CI = 0.72 to 0.89) and 0.97 (95% CI = 0.95 to 0.98), and conventional imaging procedures (chest radiography, abdominal ultrasonography, and bone scan) had a sensitivity and a specificity of 0.30 (95% CI = 0.19 to 0.44) and 0.97 (95% CI = 0.91 to 0.99) [10]. 18FDG PET/PET-CT is more sensitive than conventional imaging procedures for distant metastasis staging in patients with nasopharyngeal cancer. The skeleton is the most frequent distant-site, involving about 60–80% of nasopharyngeal cancer patients with distant metastasis [1–3]. 99mTc-phosphonate-based BS is widely used for detecting bone metastases in nasopharyngeal cancer patients. New imaging techniques such as PET or PET-CT can identify bone metastases at an earlier stage of metastatic tumor growth. But the advantages of 18FDG PET/PET-CT over BS are still variable. This meta-analysis showed that 18FDG PET/PET-CT was more sensitive than BS (0.81 vs 0.39). The use of PET/PET-CT may provide additional information to BS for diagnosing bone metastatic lesions in nasopharyngeal cancer patients.
The introduction of PET-CT can provide more anatomical details for the PET images. Several studies have demonstrated the diagnostic properties of 18FDG PET-CT and BS [2–3, 6–7]. However, the differences for diagnostic accuracy between PET/CT and BS were not clearly delineated. Our meta-analysis showed that 18FDG PET-CT was found to have higher sensitivity (0.83 vs 0.46) than BS. The scanner of PET-CT can take the place of BS as a first-line modality for diagnosing bone metastases in nasopharyngeal cancer patients.
Likelihood ratios are considered to be more meaningful for clinical practice. The values of >10 for PLR and < 0.1 for NLR indicate the high accuracy for diagnostic methods [11, 12]. The PLR values of for 18FDG PET/PET-CT and BS were 58.6 and 19.9, which were therefore high enough to diagnose bone metastases. But the NLR values for 18FDG PET/PET-CT and BS were 0.19 and 0.62, indicating that the negative results of 18FDG PET/PET-CT and BS couldn’t be used alone as a diagnostic tool to rule out bone metastatic lesions.
There were some limitations in this meta-analysis. First, the publication bias caused by positive results is a major concern, because many studies with nonsignificant or unfavorable results tend to be discarded. In this meta-analysis, publication bias was not performed because of the small number of included studies. Second, there was no consensus for the optimal execution of 18FDG PET/PET-CT and BS in all included studies. And the optimal sensitivities and specificities for 18FDG PET/PET-CT and BS are still unclear. Third, the included studies did not report sufficient information to separately evaluate the diagnostic value of 18FDG PET/PET-CT and BS in early-stage (N0-1) and advanced-stage (N2-3) patients with nasopharyngeal cancer. Fourth, the gold standard for confirmation of bone metastatic lesions, being histopathologic examination from biopsies, was not obtained from all the lesions in all included studies. However, clinical follow-up results from renewed diagnostic imaging were recorded as a gold standard when histologic confirmation was missing. Fifth, not all included studies had a prospective design. The retrospective studies may have some limitations because the possibility that the imaging interpreters might have known some outcomes of conventional imaging modalities before the interpretation of PET-CT cannot be excluded.
MATERIALS AND METHODS
Literature search and study selection
An extensive search was performed to identify relevant articles about the diagnostic efficacy of 18FDG PET/PET-CT and BS for detecting bone metastases in nasopharyngeal cancer patients. The MEDLINE and EMBASE databases (last update May 30, 2017) were searched with the following combination of search terms: positron emission tomography, PET, bone metastases, distant metastases, nasopharyngeal cancer, and nasopharyngeal carcinoma. We had no language restrictions for searching relevant studies. References of the retrieved articles were also screened for additional studies.
Studies were eligible for inclusion based on the following criteria: (1) both 18FDG PET/PET-CT and BS evaluated bone metastatic cancers in nasopharyngeal cancer patients; 2) histopathology and/or imaging follow-up data were used as the gold standard of diagnosis; (3) the studies were based on a per-patient analysis; and (4) when similar data appeared in more than one article, the article with the most details were chosen. (5) the studies with more than 20 patients were selected for inclusion. Studies were excluded based on the following criteria: (1) only PET/PET-CT or BS was performed; (2) absolute number of true-positive, false-positive, true-negative, and false-negative results were not provided; and (3) the studies were based on a per-lesion analysis.
Data extraction and quality assessment
Two reviewers (Xu.CH and Zhang.RM) independently extracted the relevant data from each article. And any difference was resolved by consensus. Data was extracted from the included studies, including authors, year of publication, study design, number of patient enrollment, technical characteristics of imaging modalities (PET/PET-CT or BS), and the reference standard. Totals of true positives, false positives, true negatives, and false negatives were also extracted from included studies.
We independently assessed the methodological quality of the included studies using the updated quality assessment tool ‘‘Quality Assessment of Diagnostic Accuracy Studies (QUADAS)-2” [13]. This revised tool allows for more transparent rating of bias and applicability of primary diagnostic accuracy studies. And it may be a considerable improvement over the original assessment tool.
Statistical analysis
All participants were classified as having positive or negative results of 18FDG PET/PET-CT and BS. We used the bivariate model to obtain weighted overall estimates of the sensitivities, specificities, diagnostic odds ratios (DORs), positive/negative likelihood ratios (PLRs/NLRs) as the main outcome measures, and to construct summary receiver operating characteristic (SROC) curves for 18FDG PET/PET-CT and BS, respectively [14–15].
All statistical analyses were performed using Stata 11.0 (Stata Corporation, College Station, TX).
CONCLUSIONS
Compared with BS, 18FDG PET/PET-CT has excellent diagnostic performance for the detection of bone metastases in nasopharyngeal cancer patients. 18FDG PET/PET-CT may enhance our diagnosis of bone metastases and provide more information for the optimal management of nasopharyngeal cancer patients.
REFERENCES
1. Liu FY, Lin CY, Chang JT, Ng SH, Chin SC, Wang HM, Liao CT, Chan SC, Yen TC. 18F-FDG PET can replace conventional work-up in primary M staging of nonkeratinizing nasopharyngeal carcinoma? J Nucl Med. 2007; 18:1614–1619.
2. Ng SH, Chan SC, Yen TC, Chang JT, Liao CT, Ko SF, Liu FY, Chin SC, Fan KH, Hsu CL. Staging of untreated nasopharyngeal carcinoma with PET/CT: comparison with conventional imaging work-up. Eur J Nucl Med Mol Imaging. 2009; 36:12–22.
3. Tang LQ, Chen QY, Fan W, Liu H, Zhang L, Guo L, Luo DH, Huang PY, Zhang X, Lin XP, Mo YX, Liu LZ, Mo HY, et al. Prospective study of tailoring whole-body dual-modality [18F] fluorodeoxyglucose positron emission tomography/computed tomography with plasma Epstein-Barr virus DNA for detecting distant metastasis in endemic nasopharyngeal carcinoma at initial staging. J Clin Oncol. 2013; 31:2861–2869.
4. Min JW, Um SW, Yim JJ, Yoo CG, Han SK, Shim YS, Kim YW. The role of whole-body FDG PET/CT, Tc 99m MDP bone scintigraphy, and serum alkaline phosphatase in detecting bone metastasis in patients with newly diagnosed lung cancer. J Korean Med Sci. 2009; 24:275–280.
5. Bezwoda WR, Lewis D, Livini N. Bone marrow involvement in anaplastic small cell lung cancer: diagnosis, hematologic features, and prognostic implications. Cancer. 1986; 58:1762–1765.
6. Chua ML, Ong SC, Wee JT, Ng DC, Gao F, Tan TW, Fong KW, Chua ET, Khoo JB, Low JS. Comparison of 4 modalities for distant metastasis staging in endemic nasopharyngeal carcinoma. Head Neck. 2009; 31:346–354.
7. Yang Z, Zhang Y, Shi W, Zhu B, Hu S, Yao Z, Pan H, Hu C, Zhang Y, Li D. Is 18F-FDG PET/CT more reliable than 99mTc-MDP planar bone scintigraphy in detecting bone metastasis in nasopharyngeal carcinoma? Ann Nucl Med. 2014; 28:411–416.
8. Chan SC, Yen TC, Ng SH, Lin CY, Wang HM, Liao CT, Fan KH, Chang JT. Differential roles of 18F-FDG PET in patients with locoregional advanced nasopharyngeal carcinoma after primary curative therapy: response evaluation and impact on management. J Nucl Med. 2006; 47:1147–1154.
9. Liu FY, Chang JT, Wang HM, Liao CT, Kang CJ, Ng SH, Chan SC, Yen TC. [18F]fluorodeoxyglucose positron emission tomography is more sensitive than skeletal scintigraphy for detecting bone metastasis in endemic nasopharyngeal carcinoma at initial staging. J Clin Oncol. 2006; 24:599–604.
10. Xu G, Li J, Zuo X, Li C. Comparison of whole body positron emission tomography (PET)/PET-computed tomography and conventional anatomic imaging for detecting distant malignancies in patients with head and neck cancer: a meta-analysis. Laryngoscope. 2012; 122:1974–1978.
11. Deeks JJ. Systematic reviews of evaluations of diagnostic and screening tests. In: Egger M, Smith GD, Altman DG, editors. Systematic reviews in health care: meta-analysis in context. London, UK: BMJ Publishing Group; 2001. p. 248–282.
12. Jaeschke R, Guyatt G, Lijmer J. Diagnostic tests. In: Guyatt G, Rennie D, editors. Users’ guides to the medical literature: a manual for evidence-based clinical practice. Chicago, IL: AMA Press; 2002. p. 121–140.
13. Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM. QUADAS-2 Group. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011; 155:529–536
14. Chu H, Cole SR. Bivariate meta-analysis of sensitivity and specificity with sparse data: a general linear mixed model approach. J Clin Epidemiol. 2006; 59:1331–1332.
15. Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH. Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol. 2005; 58:982–990.

