INTRODUCTION
Up to a third of patients with prostate cancer (PC) who undergo radical prostatectomy (RP) later develop a continuous rise of prostate specific antigen (PSA). A low but rising PSA level has been denoted biochemical recurrence (BCR). In this phase of recurrence, conventional imaging do not detect the sites of recurrence. Patients with BCR may be treated with salvage external beam radiotherapy (SRT) with or without androgen deprivation therapy (ADT) [1, 2]. Five treatments prolong survival for patients with metastatic castration-resistant (mCR) PC.
Most poor-risk PC express prostate membrane specific antigen (PSMA). Correspondingly, 68Ga-PSMA HBED-CC PET/CT detected cancer lesions for most of these patients [3]. 177Lu-PSMA-617 radioligand therapy (RLT) was effective for patients with PSMA-positive mCRPC [4]. Guidelines described the best practice for 68Ga-PSMA HBED-CC PET/CT and 177Lu-PSMA-617 RLT [5, 6]. However, 177Lu-PSMA-617 RLT is mainly used for patients with end-stage mCRPC who had failed with established systemic treatments.
We report a patient who had three episodes of recurrence with lymph node metastatic prostate cancer. The first two episodes had been treated with SRT and abiraterone. The third episode was treated with 177Lu-PSMA-617 RLT.
RESULTS
A 50-year-old patient was diagnosed with PC in May 2007. At diagnosis, he underwent RP and limited bilateral pelvic lymphadenectomy (pT3b, pN1 M0; Gleason score 9 (4+5)). He started androgen deprivation therapy (ADT) shortly after RP. Two years later, he had continuously rising PSA levels (Figure 1). With progression of PSA, PSA doubling time became reduced to four months. In 2011, an anti-1-amino-3-18F-fluorobutane-1-carboxylic acid (18F-FACBC) PET/CT showed one lesion in a pelvic lymph node. He was given SRT with a boost for the lesion. The boost was given with 2 Gy per fraction and a conventional fractionation scheme to a cumulative dose of 78 Gy. A follow-up PET/CT showed that the patient had achieved a partial remission [7]. Nadir PSA after SRT was 0.2 ng/ml. In 2013, he again had rising PSA. In 2014, a new restaging with 11C-choline PET/CT was carried out. The new PET/CT showed he had developed a new site in a retroperitoneal lymph node. Therefore his treatment was changed to abiraterone 150 mg a day and prednisone 5 mg twice daily. Three months later, the patient also underwent a second course of SRT with a cumulative dose of 60 Gy. The second SRT was targeting the new site. Again, the combined treatment transitorily reduced PSA. The new nadir PSA was 0.3 ng/ml.
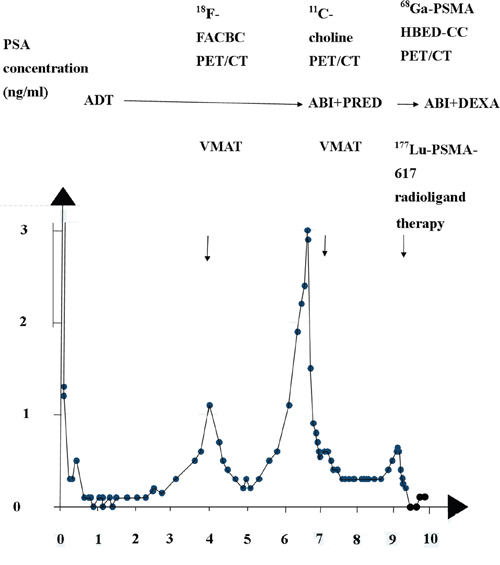
Figure 1: Three episodes of rising PSA after radical prostatectomy. The two first episodes were treated with volume modulated arc therapy (VMAT), and abiraterone and prednisone (ABI + PRED). The combination was later shifted to abiraterone and dexamethasone (ABI+DEXA). The third episode was treated with 177Lu-PSMA-617 RLT.
In 2016, the patient had a third episode of continuously rising PSA. A restaging 68Ga-PSMA HBED-CC PET/CT in July 2016 showed new pathologic uptakes in a mediastinal lymph node and several infradiaphragmal lymph nodes (Figure 2A). At that time, he had an ECOG performance status of 0, he had no pain, and had normal levels of thrombocytes. PSA was 0.6 ng/ml. However, the patient preferred to be treated with 177Lu-PSMA-617 RLT. He wanted to postpone chemotherapy with docetaxel. In July and September 2016, he was given two cycles of 177Lu-PSMA-617 RLT with a radioactivity of 6 gigabequerel (GBq) in both cycles [6]. The interval between the two cycles was eight weeks. Dosimetric analyses indicated a cumulative tumor dose of 37.7 Gy. The dose corresponded to a dose (EQD2) of 226 Gy. We calculated the EQD2 for external beam radiotherapy given with a standard fractionation of 2 Gy per fraction and with alfa/beta ratio for PC estimated as 1.5. The first cycle of 177Lu-PSMA-617 RLT caused acute nausea. But with use of anti-emetic drugs, the patient avoided nausea in connection with the second cycle. Otherwise the 177Lu-PSMA-617 RLT did not cause acute adverse effects.
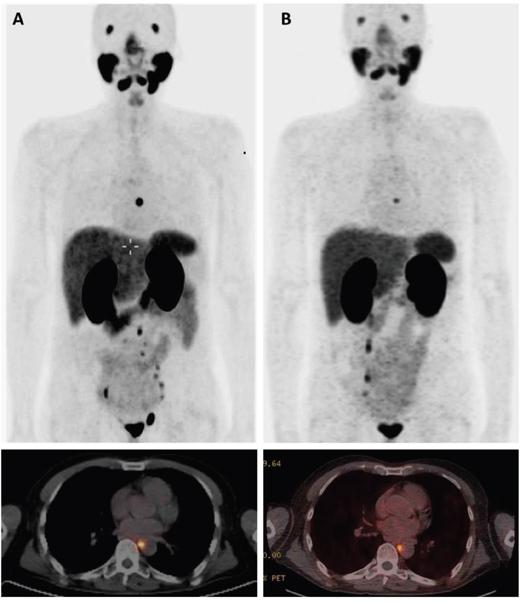
Figure 2: 68Ga-PSMA-HBED-CC PET/CT before and after treatment with 177Lu-PSMA-617 RLT. Panel A shows PET/CT before treatment July 2016 with the strongest lymph node uptake in a paraesophageal lymph node and uptakes in lymph nodes in the obturatory regions and sites in left internal and common iliac nodes. Panel B shows PET/CT after treatment November 2016 with uptake only in the mediastinal lymph node.
Follow-up 68Ga-PSMA HBED-CC PET/CT was carried out in November 2016 and February 2017 (Figure 2B). In the follow-up, the mediastinal lymph node showed a reduction of SUVmax from 21.6 to 8.6. All other pathologic sites had normalized. Total tumor volume had decreased from 18 cm3 to 1.5 cm3. From December 2016 to May 2017 PSA has been unmeasurable. In December 2016, the combination of abiraterone-and prednisone was switched to a combination of abiraterone 150 mg per day and dexamethasone 1 mg per day. In May and June 2017, he had a slight rise of PSA. Until June 2017, the patient had no chronic adverse effects.
DISCUSSION
We report treatment with 177Lu PSMA-617 RLT for a patient with a third episode of lymph node metastatic prostate cancer. The treatment had a positive balance between effects and adverse effects. The 177Lu-PSMA-617 RLT reduced pre-treatment PSA more than SRT and abiraterone. Also studies of patients with end-stage PC treated with 177Lu PSMA-617 RLT had reported a positive balance between effects an adverse effects [4].
Restaging with conventional imaging for the patient did not reveal sites of recurrence at the three episodes of continuously rising PSA Hence, restaging with conventional imaging would have concluded that the patient had biochemical recurrence. However, restaging with PET/CT with relevant radiotracers showed sites in abdominal and thoracic lymph nodes. The SRT was guided by the PET/CT findings. The clinical course after the guided SRT rules out that the PET/CT showed false-positive findings. Therefore the correct restaging for the patient at the time of the rising PSA levels was lymph node metastatic PC. Present guidelines recommend that clinicians include choline PET/CT in restaging of patients with rising PSA after RP.
According to guidelines, SRT should be started while PSA is <0.5ng/ml [2]. Previously choline PET/CT rarely gave positive findings while rising PSA levels were so low. That may have contributed to the patient had PSA levels > 1 ng/ml at the time of restaging PET/CTs. However today, 68Ga-PSMA HBED-CC PET/CT detects sites of recurrence for half of the patients who have rising PSA <0.5 ng/ml.
The 177Lu-PSMA RLT had given 48 mSv for each cycle according to dosimetric analyses in a previous study [8]. The radiation implies a class III risk of cancer according to the classification of the International Commission of Radiation Protection. We estimate the life-time risk for a 50-year old individual for developing cancer to be 25% without lutetium RLT. The estimated risk increased to 25.5% with the lutetium RLT.
Radiotherapy has a dose-response relation for PC [1]. The two previous series of SRT for the patient had been given with a total boost dose of 78 Gy and 60 Gy whereas 177Lu-PSMA RLT was given with a calculated E2Q dose of 226 Gy. Therefore 177Lu-PSMA-617 RLT reduced PSA more than the two previous courses of SRT. The previous SRT had limited effect despite the restaging had used the best available PET/CT and the SRT was guided by PET/CT. For the patient, the second and third recurrence of lymph node metastatic prostate cancer had started as micrometastases proximal to the radiation field of the two courses of SRT. The micrometastases were not detected by the PET/CTs. Anyway, an increasing number of studies report that SRT has been guided by 68Ga-PSMA-HBED-CC PET/CT [9–12].
In the third episode of rising PSA, the patient had four measurements with a progressive rise of PSA while he was treated only with abiraterone. Therefore abiraterone did not contribute to the fall of PSA after 177Lu-PSMA-617 RLT was started. The patient had the massive reduction of PSA to unmeasurable values in a period from December 2016 to May 2017. In December 2016, the combination of abiraterone and prednisone was switched to a combination of abiraterone and dexamethasone, Therefore dexamethasone did not contribute to the massive reduction of PSA after he was given 177Lu-PSMA-617 RLT.
A new study of patients with lymph node metastatic castration-resistant PC treated with 177Lu-PSMA-617 RLT is being planned. The study is submitted for the regional science-ethics committee of Southern Denmark. Also a systematic review of mCRPC is being planned that compares the therapeutic profile of third-line treatment and 177Lu-PSMA-617 RLT (Prospero database, CRD 42017067743).
In conclusion, our study is a good example of clinical efficacy with 177Lu-PSMA RLT for lymph node metastatic castration-resistant PC.
CONFLICTS OF INTEREST
The authors have stated that they have no conflicts of interest.
REFERENCES
1. Gandaglia G, Briganti A, Clarke N, Karnes RJ, Graefen M, Ost P, Zietman AL, Roach M 3rd. Adjuvant and Salvage Radiotherapy after Radical Prostatectomy in Prostate Cancer Patients. Eur Urol. 2017.
2. Ervandian M, Hoyer M, Petersen SE, Sengelov L, Hansen S, Holmberg M, Sveistrup J, Meidahl Petersen P, Borre M. Salvage radiation therapy following radical prostatectomy. A national Danish study. Acta Oncol. 2016; 55:598-603.
3. Perera M, Papa N, Christidis D, Wetherell D, Hofman MS, Murphy DG, Bolton D, Lawrentschuk N. Sensitivity, Specificity, and Predictors of Positive 68Ga-Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer: A Systematic Review and Meta-analysis. Eur Urol. 2016; 70:926-37.
4. Emmett L, Willowson K, Violet J, Shin J, Blanksby A, Lee J. Lutetium 177 PSMA radionuclide therapy for men with prostate cancer: a review of the current literature and discussion of practical aspects of therapy. J Med Radiat Sci. 2017; 64:52-60.
5. Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, Giesel F, Haberkorn U, Hope TA, Kopka K, Krause BJ, Mottaghy FM, Schöder H, et al. 68Ga-PSMA PET/CT: Joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017; 44:1014-24.
6. Fendler WP, Kratochwil C, Ahmadzadehfar H, Rahbar K, Baum RP, Schmidt M, Pfestroff A, Lützen U, Prasad V, Heinzel A, Heuschkel M, Ruf J, Bartenstein P, Krause BJ. [177Lu-PSMA-617 therapy, dosimetry and follow-up in patients with metastatic castration-resistant prostate cancer]. [Article in German]. Nuklearmedizin. 2016; 55:123-8.
7. von Eyben FE, Kangasmaki A, Kiljunen T, Joensuu T. Volumetric-modulated arc therapy for a pelvic lymph node metastasis from prostate cancer: a case report. Tumori. 2013; 99:e120-3.
8. Scarpa L, Buxbaum S, Kendler D, Fink K, Bektic J, Gruber L, Decristoforo C, Uprimny C, Lukas P, Horninger W, Virgolini I. The 68Ga/177Lu theragnostic concept in PSMA targeting of castration-resistant prostate cancer: correlation of SUVmax values and absorbed dose estimates. Eur J Nucl Med Mol Imaging. 2017; 44:788-800.
9. Henkenberens C, von Klot CA, Ross TL, Bengel FM, Wester HJ, Katja H, Christiansen H, Derlin T. 68Ga-PSMA Ligand PET/CT-based Radiotherapy for Lymph Node Relapse of Prostate Cancer After Primary Therapy Delays Initiation of Systemic Therapy. Anticancer Res. 2017; 37:1273-9.
10. Porres D, Pfister D, Thissen A, Kuru TH, Zugor V, Buettner R, Knuechel R, Verburg FA, Heidenreich A. The role of salvage extended lymph node dissection in patients with rising PSA and PET/CT scan detected nodal recurrence of prostate cancer. Prostate Cancer Prostatic Dis. 2017; 20:85-92.
11. Albisinni S, Artigas C, Aoun F, Biaou I, Grosman J, Gil T, Hawaux E, Limani K, Otte FX, Peltier A, Sideris S, Sirtaine N, Flamen P, van Velthoven R. Clinical impact of 68 Ga-prostate-specific membrane antigen (PSMA) positron emission tomography/computed tomography (PET/CT) in patients with prostate cancer with rising prostate-specific antigen after treatment with curative intent: preliminary analysis of a multidisciplinary approach. BJU Int. 2016.
12. Henkenberens C, von Klot CA, Ross TL, Bengel FM, Wester HJ, Merseburger AS, Vogel-Claussen J, Christiansen H, Derlin T. (68)Ga-PSMA ligand PET/CT-based radiotherapy in locally recurrent and recurrent oligometastatic prostate cancer : Early efficacy after primary therapy. Strahlenther Onkol. 2016; 192:431-9.

