INTRODUCTION
Epithelial ovarian cancer (EOC), the leading cause of gynecologic cancer-related mortality, is known to be a multifactorial disease involving genetic, environmental, and epigenetic factors [1, 2]. Recently, research has focused on aberrant variation in the expression of transcription factors and its importance in the development of this malignancy [3].
Homeobox (HOX) genes, defined by a highly conserved 183-base pair DNA sequence called the homeobox, encode homeoproteins that function as transcription factors in differentiation and proliferation processes during embryonic structural development [4]. Aberrant expression of these proteins has been associated with carcinogenesis and aggressiveness [5, 6]. This is consistent with the idea that expression levels of many genes involved in normal embryogenesis are aberrant in various malignancies [7].
Previous studies showed that dysregulation of several HOX genes (including aberrant expression of HOXA7, HOXA9, HOXC8, HOXB13, and HOXC6) was associated with adverse prognostic factors in EOC; however, the majority of this research was limited by small sample sizes and was performed only in vitro [8–14]. Several studies have shown potential roles for HOX antisense long non-coding RNAs in ovarian cancer aggressiveness [15, 16]. These results suggest that aberrant expression of HOX genes may be correlated with the process of ovarian cancer carcinogenesis.
We analyzed the mRNA expression level of all 39 HOX genes using data from The Cancer Genome Atlas (TCGA) database on epithelial ovarian cancer and explored correlations with clinical data, including survival outcomes. We also evaluated the prognostic value of HOX gene expression analysis.
RESULTS
In total, we included 630 ovarian cancer cases, which were all the patients who included in the TCGA ovarian cancer database. The median follow-up period was 34.0 months (range, 0.3–182.7 months). Among clinicopathological features, age at the time of diagnosis, advanced Federation of Gynecology and Obstetrics (FIGO) stage, suboptimal residual disease, and poor Eastern Cooperative Oncology Group (ECOG) performance status were found to be independent predictive factors for overall survival in multivariate Cox regression analysis (Table 1).
Table 1: Correlation between clinicopathological features and overall mortality
Variables |
Total n = 630 n (%) |
Multivariate analysis |
|
|---|---|---|---|
HR (95% CI) |
P |
||
Age |
|
|
|
<45 |
54 (8.6) |
1 (Reference) |
0.042 |
≥45 |
553 (87.8) |
1.512 (0.991-2.309) |
|
N/A |
23 (3.7) |
|
|
Stage |
|
|
|
I |
18 (2.9) |
1 (Reference) |
0.002 |
II |
33 (5.2) |
|
|
III |
462 (73.3) |
2.264 (1.272-4.029) |
|
IV |
89 (14.1) |
|
|
N/A |
28 (4.4) |
|
|
Histology |
|
|
|
Serous |
607 (96.3) |
|
|
N/A |
23 (3.7) |
|
|
Grade |
|
|
|
1, 2 |
85 (13.5) |
1 (Reference) |
0.253 |
3, 4 |
505 (80.2) |
1.189 (0.884-1.598) |
|
N/A |
40 (6.3) |
|
|
LN metastasis |
|
|
|
No |
85 (13.5) |
1 (Reference) |
0.125 |
Yes |
139 (22.1) |
1.387 (0.913-2.107) |
|
N/A |
406 (64.4) |
|
|
Residual disease |
|
|
|
≤ 1 cm |
387 (61.4) |
1 (Reference) |
<0.001 |
≤ 2 cm |
151 (24.0) |
1.548 (1.222-1.960) |
|
N/A |
92 (14.6) |
|
|
ECOG |
|
|
|
0, 1 |
123 (19.5) |
1 (Reference) |
0.002 |
2, 3 |
12 (1.9) |
3.317 (1.552-7.087) |
|
N/A |
495 (78.6) |
|
|
N/A, not available; ECOG, The Eastern Cooperative Oncology Group performance status; HR, hazard ratio; CI, confidence interval; LN, lymph node.
Table 2 shows correlations between clinicopathological features and mRNA expression levels of HOX family genes. When the cases were categorized into low and high mRNA expression groups based on cut-off values determined as the median for each gene, higher expression of HOXA1 (P = 0.011), A4 (P = 0.045), A5 (P = 0.01), A7 (P = 0.047), A10 (P = 0.009), A11 (P = 0.001), B13 (P = 0.01), C13 (P = 0.022), D1 (P = 0.004), and D3 (P = 0.011) was associated with advanced FIGO stage. Suboptimal residual disease after debulking surgery was significantly correlated with higher expression of HOXB9 (P = 0.002), B13 (P = 0.008), and C13 (P = 0.04). In addition, patients with high expression of HOXC6 (P = 0.018) and C11 (P = 0.049) were significantly more likely to have poor performance status (ECOG 2 or 3).
Table 2: Summary of the correlation between clinicopathological features and mRNA expression counts of homeobox (HOX) family genes
Age≥45 |
Stage III/IV |
Grade 3/4 |
LN metastasis |
Residual disease > 1 cm |
ECOG 2, 3 |
|---|---|---|---|---|---|
HOXA1 |
+ |
|
|
|
|
HOXA2 |
|
|
|
|
|
HOXA3 |
|
|
+ |
|
|
HOXA4 |
+ |
|
|
|
|
HOXA5 |
+ |
|
+ |
|
|
HOXA6 |
|
|
|
|
|
HOXA7 |
+ |
|
|
|
|
HOXA9 |
|
|
|
|
|
HOXA10 |
++ |
|
|
|
|
HOXA11 |
++ |
|
+ |
|
|
HOXA13 |
|
|
|
|
|
HOXB1 |
|
|
|
|
|
HOXB2 |
|
|
++ |
|
|
HOXB3 |
|
+ |
|
|
|
HOXB4 |
|
|
|
|
|
HOXB5 |
|
|
|
|
|
HOXB6 |
|
|
|
|
|
HOXB7 |
|
|
|
|
|
HOXB8 |
|
|
|
|
|
HOXB9 |
|
|
|
++ |
|
HOXB13 |
+ |
|
|
++ |
|
HOXC4 |
|
|
|
|
|
HOXC5 |
|
|
|
|
|
HOXC6 |
|
|
|
|
+ |
HOXC8 |
|
|
|
|
|
HOXC9 |
|
|
|
|
|
HOXC10 |
|
|
|
|
|
HOXC11 |
|
|
|
|
+ |
HOXC12 |
|
|
|
|
|
HOXC13 |
+ |
|
|
+ |
|
HOXD1 |
++ |
|
|
|
|
HOXD3 |
+ |
++ |
|
|
|
HOXD4 |
|
|
|
|
|
HOXD8 |
|
|
|
|
|
HOXD9 |
|
|
|
|
|
HOXD10 |
|
|
|
|
|
HOXD11 |
|
|
++ |
|
|
HOXD12 |
|
|
++ |
|
|
HOXD13 |
|
+ |
|
|
|
+, Correlation with P value < 0.05; ++, correlation with P value <0.01; LN, lymph node; ECOG, The Eastern Cooperative Oncology Group performance status.
Out of the total 630 patients, 584 patients (92.7%) had information on both prognostic value and microarray for the transcriptome analysis, so the prognosis of the 584 patients was analyzed. Kaplan-Meier survival analysis showed that overall survival (OS) was significantly shorter in patients with high rather than low expression levels of HOXA10 (P = 0.008, median OS: 36 vs. 32 months) and HOXB3 (P = 0.003, median OS: 34 vs. 32 months), and significantly longer in patients with high rather than low HOXC5 expression (P = 0.022, median OS: 33 vs. 35 months) (Figure 1). Multivariate Cox regression analysis of HOX gene expression also demonstrated that HOXA10 and HOXB3 were significant positive predictive factors [odds ratio (OR): 1.305 and 1.462, 95% confidence interval (CI): 1.052–1.619 and 1.174–1.822, respectively], while HOXC5 (OR: 0.713, 95% CI: 0.572–0.889) was a significant negative predictive factor for overall survival (Figure 2).
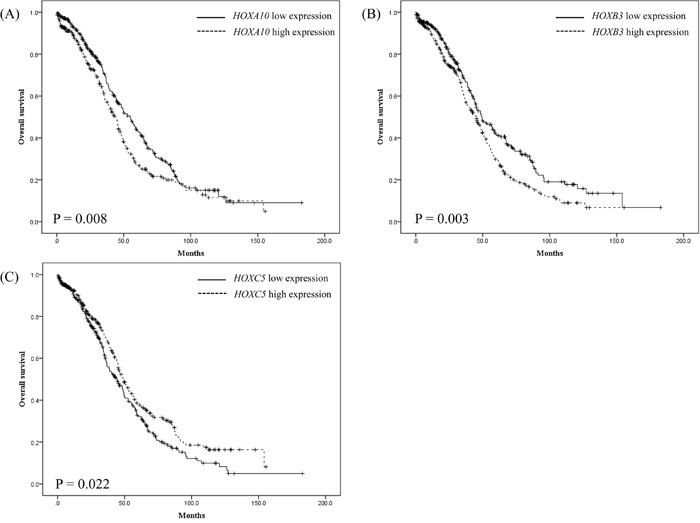
Figure 1: Kaplan-Meier survival analysis of 584 patients with ovarian cancer stratified by (A) HOXA10, (B) HOXB3, and (C) HOXC5 gene expression levels on microarray.
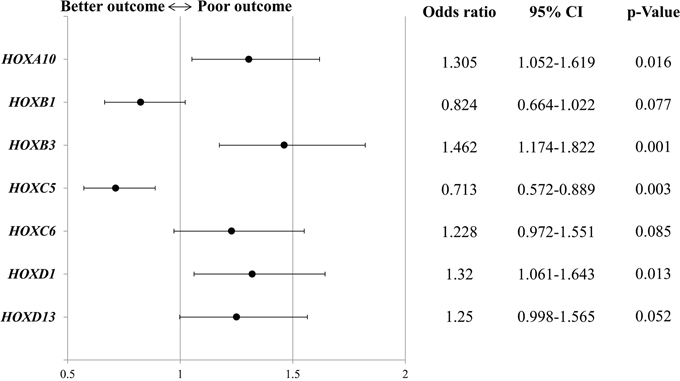
Figure 2: Multivariate analyses of homeobox (HOX) gene expression predicting overall survival. CI, confidence interval.
DISCUSSION
In this study, we demonstrated that increased mRNA expression of certain HOX genes was independently associated with multiple risk factors, including advanced FIGO stage, suboptimal residual disease, and poor performance status, that were found to influence survival outcomes in ovarian cancer using the TCGA database. Specifically, upregulation of HOXA10 and HOXB3 genes and downregulation of HOXC5 were significantly associated with unfavorable survival outcomes in patients with ovarian cancer.
Accumulating evidence indicates that HOX genes may be useful biomarkers to predict and evaluate responses to chemotherapy [17]. Our results were not completely consistent with those of previous in vitro research. In particular, the clinical relevance of HOXB3 and HOXC5 was not known until now. This might be explained by the fact that the majority of studies reported to date have been retrospective in nature or conducted in small sized patient cohorts. To the best of our knowledge, this is the first study of HOX genes performed using clinical data from TCGA database. Also, it is feasible to clinically classify each subtype to determine the prognosis based on the results of this study. Further functional studies are required to identify the specific mechanisms by which HOX gene dysregulation influences oncogenesis.
During embryogenesis, as well as in adult tissues, growth, differentiation, and organization of cell populations are tightly coordinated and controlled [18]. In contrast, cancer has been identified as aberrations of these basic processes [19]. The oncogerminative theory of carcinogenesis suggests that malignant transformation is due to the aberrant expression of embryogenesis-related genes [20]. According to this concept, carcinogenesis is a dynamic self-organizing process that is similar to the process of early embryo development. Additionally, the malignant transformation that arises from gene mutations, in combination with epigenetic dysregulation, may reprogram somatic cells into immortal cells that simulate germline cells; this is consistent with cancer stem cells [20].
A substantial body of evidence supports a crucial role for HOX genes as developmental genes during embryogenesis, and also supports the critical role of aberrant HOX gene expression in the development of various tumors, specifically in ovarian cancer [5–7, 17, 21, 22]. Further research is needed to elucidate all roles of HOX genes in ovarian cancer. A major limitation of this study is the short-term follow-up period, and the small number of overall mortalities during the period might weaken the clinical applicability of the current findings. A more comprehensive investigation is promising in the further analysis based on the regularly updated TCGA data in the future. Although this study proved the hypothesis that dysregulated expression of HOX genes, based on microarray data, was correlated with altered survival outcomes in EOC, further validation using a different modality, such as large-volume RNA sequencing or immunohistochemical assay, might also be needed to verify the current results.
Analyzing HOX gene expression levels in a cytologic or surgical specimen can facilitate the selection of patients with ovarian cancer who are expected to have a poor prognosis. Based on analytical results, we can prescribe more aggressive treatment or more frequent follow-up for cases with an expected high risk. Moreover, there are great unmet needs of patients with refractory ovarian cancer that recurs after treatment with standard therapy, which demands novel therapeutic agents. Drugs regulating HOX gene expression could potentially have a therapeutic effect by inducing inhibition of reprogramed oncogerminative cells, as was shown by in vitro experiments using a homeobox transcription factor inhibitor [23]. Recently, decitabine, a drug that induced CpG island hypomethylation of HOXA10 and HOXA11 genes, showed significant correlation with improved progression-free survival in an open-label, single-center, phase II clinical trial [24].
In conclusion, dysregulated expression of the HOXA10, HOXB3, and HOXC5 genes was significantly associated with favorable or unfavorable overall survival in a large ovarian cancer cohort from TCGA. Additionally, many of the HOX genes were correlated with clinical variables that were independent predictive factors for overall survival. Evaluation of HOX gene expression may be potentially valuable as a marker to predict prognosis in serous ovarian cancer.
MATERIALS AND METHODS
Data acquisition
We collected mRNA expression data for 39 HOX family genes and corresponding clinicopathological information from the TCGA data portal (https://tcga-data.nci.nih.gov/tcga/tcgaDownload.jsp). The patient personal information is anonymized and deidentified. According to the TCGA publication guidelines (http://cancergenome.nih.gov/publications/publicationguidelines), these mRNA sequencing data have no restrictions on publication, and no additional approval by an ethics committee was required to publish findings utilizing the data. The database compiled from microarray analysis performed using GeneChip Human Genome U133A 2.0 Array (Affymetrix) was analyzed in this study.
Patient information
We obtained the mRNA expression levels of 39 HOX family genes for each patient, along with their clinicopathologic features, including age at initial pathologic diagnosis, International FIGO stage, histologic subtype, lymph node metastasis, and overall survival (Supplementary Table 1).
Statistical analyses
We used the Fisher exact or χ2 tests for categorical variables, according to sample size. Linear regression was applied to assess associations between clinical variables and each HOX gene expression count. Medians of HOX gene expression were determined as the cut-off value for prediction of survival. Kaplan-Meier survival analyses and multivariate Cox regression analysis based on the calculated cut-off values were performed. SPSS version 23.0 (IBM Corp., Armonk, NY, USA) was used for data analysis.
Abbreviations
HOX, homeobox; TCGA, The Cancer Genome Atlas; EOC, epithelial ovarian cancer; OR, odds ratio; CI, confidence interval; FIGO, Federation of Gynecology and Obstetrics; N/A, not available; ECOG, The Eastern Cooperative Oncology Group performance status
Author contributions
Conception & Design of Study was done by KJE, HJK, JYL, EJN, SK, SWK and YTK. Data Collection was done by KJE. Data Analysis & Interpretation was done by KJE and YTK. Statistical Analysis was done by KJE. Manuscript Preparation was done by KJE, and YTK.
ACKNOWLEDGMENTS
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (grant numbers NRF-2015R1A2A2A01008162 and NRF-2015R1C1A2A01053516).
CONFLICTS OF INTEREST
The authors report no conflicts of interest.
FINANCIAL SUPPORT
None
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66: 7-30. https://doi.org/10.3322/caac.21332.
2. Rai A, Pradhan P, Nagraj J, Lohitesh K, Chowdhury R, Jalan S. Understanding cancer complexome using networks, spectral graph theory and multilayer framework. Sci Rep. 2017; 7: 41676. https://doi.org/10.1038/srep41676.
3. Wang LL, Xiu YL, Chen X, Sun KX, Chen S, Wu DD, Liu BL, Zhao Y. The transcription factor FOXA1 induces epithelial ovarian cancer tumorigenesis and progression. Tumour Biol. 2017; 39. https://doi.org/10.1177/1010428317706210.
4. Daftary GS, Taylor HS. Endocrine regulation of HOX genes. Endocr Rev. 2006; 27: 331-55. https://doi.org/10.1210/er.2005-0018.
5. Gehring WJ, Hiromi Y. Homeotic genes and the homeobox. Annu Rev Genet. 1986; 20: 147-73. https://doi.org/10.1146/annurev.ge.20.120186.001051.
6. Haria D, Naora H. Homeobox gene deregulation: impact on the hallmarks of cancer. Cancer Hallm. 2013; 1: 67-76. https://doi.org/10.1166/ch.2013.1007.
7. Abate-Shen C. Deregulated homeobox gene expression in cancer: cause or consequence? Nat Rev Cancer. 2002; 2: 777-85. https://doi.org/10.1038/nrc907.
8. Chong GO, Jeon HS, Han HS, Son JW, Lee YH, Hong DG, Park HJ, Lee YS, Cho YL. Overexpression of microRNA-196b accelerates invasiveness of cancer cells in recurrent epithelial ovarian cancer through regulation of homeobox A9. Cancer Genomics Proteomics. 2017; 14: 137-41. https://doi.org/10.21873/cgp.20026.
9. Lu S, Liu R, Su M, Wei Y, Yang S, He S, Wang X, Qiang F, Chen C, Zhao S, Qian L, Shao M, Mao G. Overexpression of HOXC8 is associated with poor prognosis in epithelial ovarian cancer. Reprod Sci. 2016; 23: 944-54. https://doi.org/10.1177/1933719115625845.
10. Yuan H, Kajiyama H, Ito S, Chen D, Shibata K, Hamaguchi M, Kikkawa F, Senga T. HOXB13, ALX4 induce SLUG expression for the promotion of EMT and cell invasion in ovarian cancer cells. Oncotarget. 2015; 6: 13359-70. https://doi.org/10.18632/oncotarget.3673.
11. Tait DL, Bahrani-Mostafavi Z, Vestal CG, Richardson C, Mostafavi MT. Downregulation of HOXC6 in serous ovarian cancer. Cancer Invest. 2015; 33: 303-11. https://doi.org/10.3109/07357907.2015.1041641.
12. Ko SY, Naora H. HOXA9 promotes homotypic and heterotypic cell interactions that facilitate ovarian cancer dissemination via its induction of P-cadherin. Mol Cancer. 2014; 13: 170. https://doi.org/10.1186/1476-4598-13-170.
13. Ko SY, Ladanyi A, Lengyel E, Naora H. Expression of the homeobox gene HOXA9 in ovarian cancer induces peritoneal macrophages to acquire an M2 tumor-promoting phenotype. Am J Pathol. 2014; 184: 271-81. https://doi.org/10.1016/j.ajpath.2013.09.017.
14. Naora H, Montz FJ, Chai CY, Roden RB. Aberrant expression of homeobox gene HOXA7 is associated with mullerian-like differentiation of epithelial ovarian tumors and the generation of a specific autologous antibody response. Proc Natl Acad Sci U S A. 2001; 98: 15209-14. https://doi.org/10.1073/pnas.011503998.
15. Yim GW, Kim HJ, Kim LK, Kim SW, Kim S, Nam EJ, Kim YT. Long non-coding RNA HOXA11 antisense promotes cell proliferation and invasion and predicts patient prognosis in serous ovarian cancer. Cancer Res Treat. 2017; 49: 656-68. https://doi.org/10.4143/crt.2016.263.
16. Richards EJ, Permuth-Wey J, Li Y, Chen YA, Coppola D, Reid BM, Lin HY, Teer JK, Berchuck A, Birrer MJ, Lawrenson K, Monteiro AN, Schildkraut JM, et al. A functional variant in HOXA11-AS, a novel long non-coding RNA, inhibits the oncogenic phenotype of epithelial ovarian cancer. Oncotarget. 2015; 6: 34745-57. https://doi.org/10.18632/oncotarget.5784.
17. Miller KR, Patel JN, Ganapathi MK, Tait DL, Ganapathi RN. Biological role and clinical implications of homeobox genes in serous epithelial ovarian cancer. Gynecol Oncol. 2016; 141: 608-15. https://doi.org/10.1016/j.ygyno.2016.03.004.
18. Vinnitsky VB. Oncogerminative hypothesis of tumor formation. Med Hypotheses. 1993; 40: 19-27. https://doi.org/10.1016/0306-9877(93)90191-R.
19. Negrini S, Gorgoulis VG, Halazonetis TD. Genomic instability--an evolving hallmark of cancer. Nat Rev Mol Cell Biol. 2010; 11: 220-8. https://doi.org/10.1038/nrm2858.
20. Vinnitsky V. The development of a malignant tumor is due to a desperate asexual self-cloning process in which cancer stem cells develop the ability to mimic the genetic program of germline cells. Intrinsically Disord Proteins. 2014; 2: e29997. https://doi.org/10.4161/idp.29997.
21. Kelly Z, Moller-Levet C, McGrath S, Butler-Manuel S, Kavitha Madhuri T, Kierzek AM, Pandha H, Morgan R, Michael A. The prognostic significance of specific HOX gene expression patterns in ovarian cancer. Int J Cancer. 2016; 139: 1608-17. https://doi.org/10.1002/ijc.30204.
22. Kar SP, Tyrer JP, Li Q, Lawrenson K, Aben KK, Anton-Culver H, Antonenkova N, Chenevix-Trench G, Baker H, Bandera EV, Bean YT, Beckmann MW, Berchuck A, et al. Network-based integration of GWAS and gene expression identifies a HOX-centric network associated with serous ovarian cancer risk. Cancer Epidemiol Biomarkers Prev. 2015; 24: 1574-84. https://doi.org/10.1158/1055-9965.epi-14-1270.
23. Burton LJ, Dougan J, Jones J, Smith BN, Randle D, Henderson V, Odero-Marah VA. Targeting the nuclear cathepsin L CCAAT displacement protein/cut homeobox transcription factor-epithelial mesenchymal transition pathway in prostate and breast cancer cells with the Z-FY-CHO inhibitor. Mol Cell Biol. 2017; 37. https://doi.org/10.1128/mcb.00297-16.
24. Matei D, Fang F, Shen C, Schilder J, Arnold A, Zeng Y, Berry WA, Huang T, Nephew KP. Epigenetic resensitization to platinum in ovarian cancer. Cancer Res. 2012; 72: 2197-205. https://doi.org/10.1158/0008-5472.can-11-3909.

