INTRODUCTION
Embryo implantation is a prerequisite for a successful pregnancy in mammals [1, 2]. Implantation failure accounts for the majority of pregnancy losses in humans [3] and aberrant implantation is believed to result in severe outcomes at later pregnancy stages including placental insufficiency, preeclampsia and preterm labour [1]. However, due to its complexity and technical limitations especially in humans, embryo implantation remains a poorly understood process.
Prostaglandins (PGs) are a group of bioactive lipid compounds long known to be required for embryo implantation. Deletion of enzymes for PGs synthesis including phospholipase A2 and cyclooxygenase-2 (COX-2) results in implantation failure in mice [4, 5]. In humans, exposure to non-steroid anti-inflammatory drugs that block PGs synthesis during pregnancy significantly increases the risk of miscarriage [6]. Also, impaired endometrial PGs synthesis was observed in women with repeatedly implantation failure in IVF trials [7]. Prostaglandin E2 (PGE2), mainly produced by COX-2-driven synthesis, is considered one of the most important PGs to initiate decidualization [5, 8], an endometrial stromal differentiation process required for embryo implantation [9]. We have previously demonstrated that the epithelial sodium channel (ENaC) in the endometrial epithelial cells can be activated by embryo-derived protease, which subsequently triggers a sequence of events in endometrial epithelial cells, including Ca2+ increase, phosphorylation of CREB (Ca2+/cAMP responsive element binding protein), downregulation of miR101 and miR199a, upregulation of COX-2 and eventually PGE2 production and release to the stroma, leading to decidualization and embryo implantation [10, 11]. However, it should be noted that the permeability of plasma membrane to PGE2 is low due to its negative charges [12, 13] and how PGE2 is released from the endometrial epithelial cells for induction of stromal decidualization required for embryo implantation remains unclear.
Multi-drug resistance protein 4 (MRP4), a member of the ATP-binding cassette (ABC) transporter family, is known to mediate the efflux of both exogenous drugs and endogenous molecules including PGs [14–17]. It has been reported that upregulation/overexpression of MRP4 in colorectal cancer tissues and cell lines is correlated to high extracellular level of PGE2, suggesting its capacity in transporting PGE2 [18]. Indeed, MRP4 has been reported to transport PGE2 in the bovine endometrium [19] and that MRP4 knockout mice showed reduced litter size [20]. Given the importance of ENaC-regulated COX-2/PGE2 signaling in embryo implantation and the PGE2-transporting capacity of MRP4, we hypothesized that MRP4 might be involved in mediating the ENaC-dependent signaling required for embryo implantation. We undertook the present study to test this possibility in vitro and in vivo.
RESULTS
Upregulation of MRP4 by ENaC activation during embryo implantation
We first examined the expression of MRP4 in mouse uterus at day 2 to 6 post mating and found that MRP4 was abundantly upregulated in midnight of day 4, when the implantation is initiated, and day 5, when decidualization begins in mice (Figure 1A&1B). This expression pattern of MRP4 is consistent with that previously reported for ENaC during embryo implantation [10], and thus we further examined whether the expression of MRP4 could possibly be altered by ENaC activation during embryo implantation. We used trypsin, an embryo-released protease known to activate ENaC, to treat human endometrial epithelial cells. As shown in Figure 1C&1D, activation of ENaC by trypsin (20 μg/ml, 15 mins) induced a significant increase of MRP4 at mRNA level in the cells, which could be abolished by pretreatment with amiloride (10 μM, 24 hours), a selective ENaC blocker, suggesting that MRP4 transcription could be activated by ENaC activation during embryo implantation.
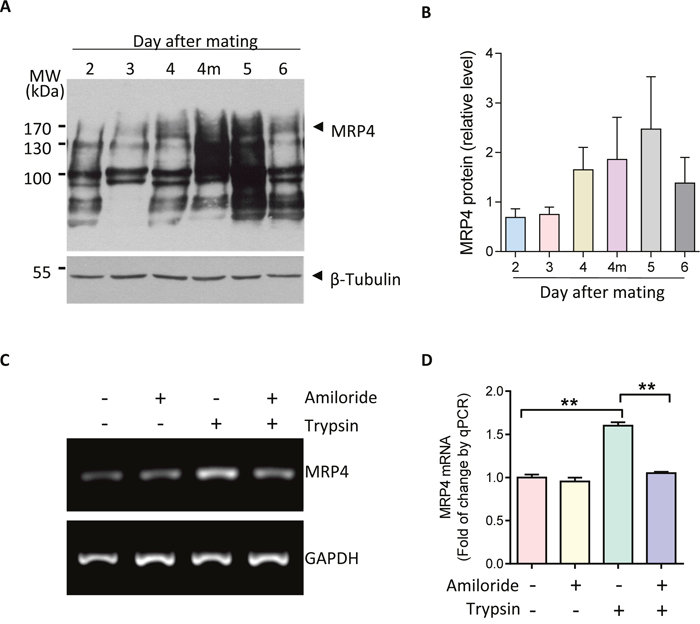
Figure 1: Upregulation MRP4 by ENaC activation during embryo implantation. (A-B) Western blots (A) with quantification (B) for MRP4 in mouse uterus during the peri-implantation period from days 2-6 post mating (4m: midnight of day 4) (n = 3). (C-D) Conventional (C) and quantitative (D) PCR of MRP4 in human endometrial epithelial cells in response to trypsin (20 μg/ml, ENaC activator) in the absence or presence of amiloride (10 μM, ENaC blocker) (n = 3, ** P < 0.01).
MRP4 mediates ENaC activation-triggered epithelial PGE2 release into stroma at embryo implantation
Given the reported capacity of MRP4 in transporting PGE2 uniquely among the ABC transporters [21], we next asked whether MRP4 could mediate the epithelial release of PGE2 triggered by ENaC activation during embryo implantation. We treated the human endometrial epithelial cells with MK-571 (10 μM, 24 hours), a selective inhibitor of MRP4 transporting function, and found that the concentration of PGE2 in the incubating medium as detected by ELISA, was significantly reduced as compared to the controls treated with PBS (Figure 2A). Moreover, activation of ENaC by trypsin (20 μg/ml, 15 mins) induced abundant increases in PGE2 release into the medium, which was also significantly blocked by MK-571 (10 μM), suggesting a role of MRP4 in mediating PGE2 release from endometrial epithelial cells upon ENaC activation at embryo implantation (Figure 2A). It should be noted that epithelial PGE2 release into the stroma is a particularly important signal leading to stromal decidualization required for embryo implantation. Using immunofluorescence labeling, we found that MRP4 expression was specifically localized to the basolateral membrane of the endometrial epithelial cells, the interface between the epithelium and the stroma (Figure 2B). The polarized expression pattern of MRP4 and its capacity in PGE2 transport suggested an important role of MRP4 in transporting PGE2 from epithelial cells to the stromal cells to induce decidualization required for embryo implantation.
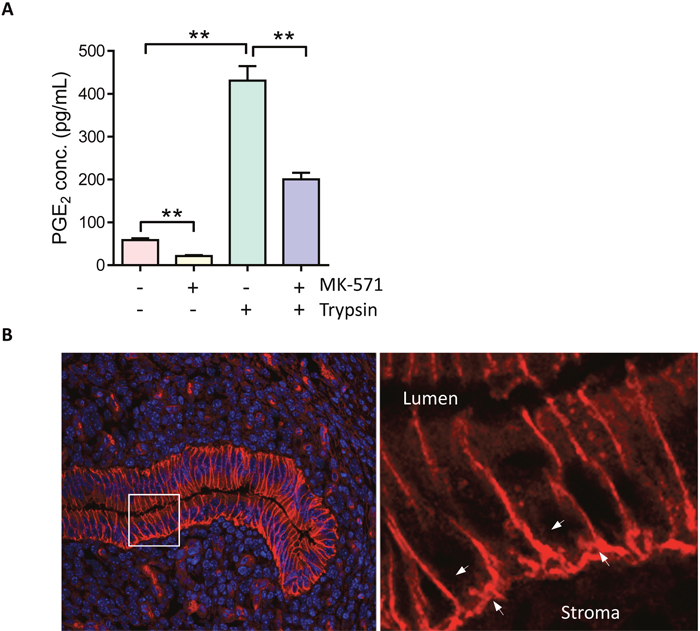
Figure 2: MPR4 mediates ENaC activation-triggered PGE2 release. (A) ELISA detection of PGE2 levels in medium incubated with human endometrial epithelial cells with or without trypsin (20 μg/ml) and MK-571 (10 μM, MRP4 blocker) (n = 3, ** P < 0.01). (B) Immunofluorescence labeling (Red) for MRP4 in mouse uterus at day 4 post mating. Arrows: basolateral membrane of the endometrial epithelial cells. Lu: lumen. Nuclei were visualized by DAPI staining. Scale bar = 5 μm.
Involvement of MRP4 in ENaC-activated CREB/COX-2 signaling and embryo implantation gene expression
Embryo implantation is a complicated process involving a series of critical marker genes, including COX-2, Claudin4 [22], HoxA10 [23], Lif [24] and PPARg [25]. We explored the involvement of MRP4 in embryo implantation by examing the effect of its inhibitor MK-571 on uterine expression of these implantation related genes. To inhibit MRP4, MK-571 was intrauterinally injected in pregnant mice on day 3 (prior to implantation). Uterine expression of genes was examined 24 hour later on day 4. The results showed that COX-2, Claudin4 and Lif were significantly reduced when MRP4 was inhibited by intrauterine injection with MK-571 (0.5 mM) as compared to the control group Figure 3A). Since COX-2 is a key enzyme in PGE2 synthesis and its transcription is known to be activated by ENaC-dependent phosphorylation of CREB during embryo implantation [10, 26], we next examined whether MRP4 may be involved in CREB phosphorylation by ENaC activation in endometrial epithelial cells. As shown in Figure 3B&3C, the level of phosphorylated CREB was significantly increased after treatment with trypsin, which was abolished by the treatment with MK-571 (10 μM) compared with that of the control group. Thus, these results suggest the involvement of MRP4 in ENaC-dependent CREB/COX-2 signaling during embryo implantation.
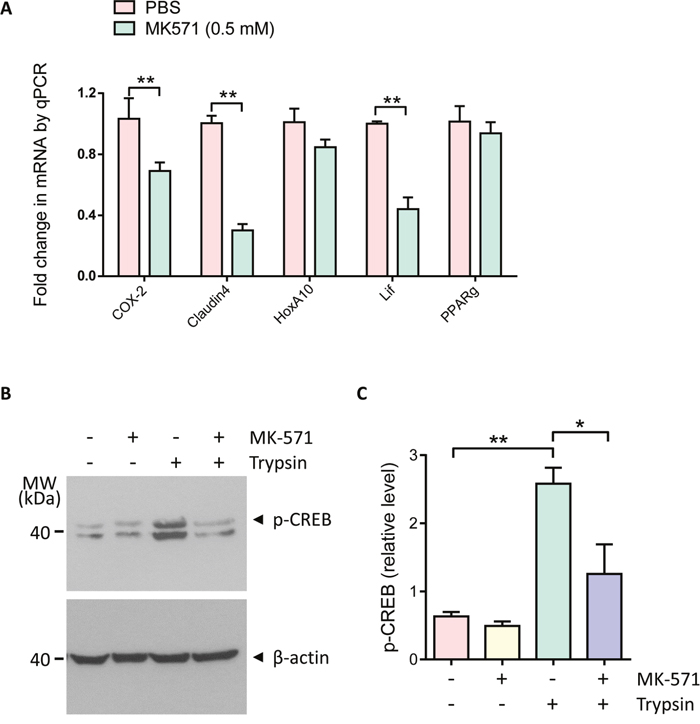
Figure 3: Involvement of MRP4 in regulation of embryo implantation gene expression and CREB/COX-2 signaling. (A) Quantitative PCR of COX-2, Claudin4, HoxA10, Lif and PPARg in mouse uteri 24 hours after intrauterine injection with MK-571 (0.5 mM, day 3 post mating) (n = 4, ** P < 0.01). (B-C) Western blots (B) with quantification (C) for phosphorylated CREB (p-CREB) in human endometrial epithelial cells after incubated with MK-571 (10 μM) in the absence or presence of trypsin (20 μg/ml) (n = 3, * P < 0.05, ** P < 0.01).
Inhibition of MRP4 reduces implantation rate in mice
To confirm that PGE2 transport function of MRP4 indeed plays a role in embryo implantation, we counted the implantation rate on day 7, when the implanted sites are visible, after intrauterine injection of MK-571 on day 3 in the mouse in vivo model. The results showed that MK-571 (0.1 - 5 mM) dose dependently decreased the number of implanted embryos in the uterine horns compared to the vehicle treated control horns, as counted on day 7 (Figure 4A&4B). The results therefore suggested essential involvement of MRP4 in embryo implantation.
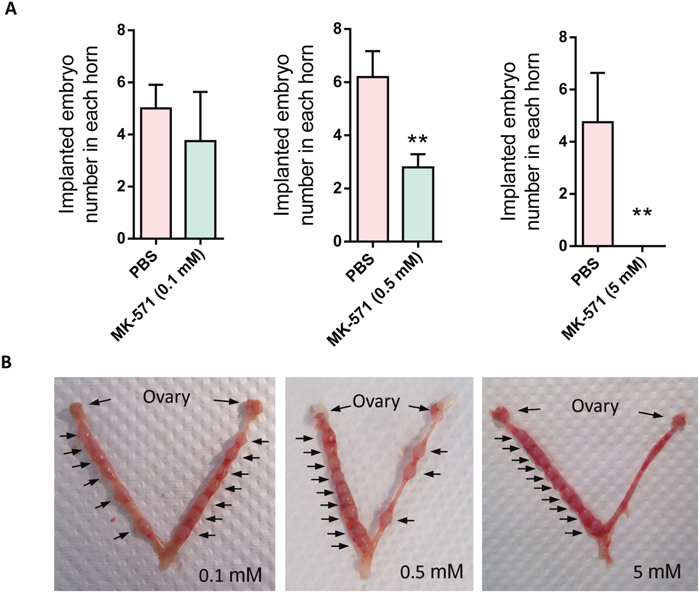
Figure 4: Effect of MRP4 inhibition on implantation rate in mice. (A-B) Effect of intrauterine injection of MK-571 (0.1-5 mM, day 3) on implantation rate in mice with representative photographs (B) showing implantation sites (arrows) in the control uterine horns (left) and the MK-571 treated ones (right) (n = 3 - 5, ** P < 0.01 compared to Ctrl).
DISCUSSION
The present study has identified a previously unexplored physiological role of MRP4 in mediating ENaC activation-triggered PGE2 production and release required for embryo implantation.
Despite the well-recognized role of PGE2 in embryo implantation, the study on the transport of PGE2 from uterine epithelium to stroma during embryo implantation is scarce. The present study has provided evidence suggesting that MRP4 is an essential mediator for the efflux or release of PGE2 from endometrial epithelial cells to the stroma required for embryo implantation. First, the expression of MRP4 in endometrial epithelial cells is upregulated during implantation and by ENaC activation induced by embryo-derived proteases, suggesting its responsiveness to embryo-derived signal. Second, MRP4 mediates epithelial PGE2 release triggered by ENaC activation. Importantly, the expression of MRP4 in the endometrial epithelial cells is polarized and predominately localized to the basolateral membrane, indicating the direction of PGE2 efflux toward the stroma beneath the epithelium. Interestingly, another known PG transporter (PGT), which only conducts the influx of PGE2 [27], has been observed to be strongly expressed in subluminal stroma cells at implantation sites in mice [28], suggesting an uptake of PGE2 in stroma cells at implantation. The differential expression pattern of these PGE2 transporters is consistent with a signal (PGE2) transfer from the epithelium to the stroma essential required for embryo implantation.
The present results also suggest the involvement of MRP4 in ENaC-dependent CREB phosphorylation. As we previously demonstrated, phosphorylated CREB may contribute to enhancement of PGE2 production in endometrial epithelial cells in two folds. First, it directly triggers the transcription of COX-2, a major enzyme for PGE2 synthesis in cells [10]. Second, it represses the expression of two COX-2 inhibiting microRNAs miR101 and miR199a and indirectly promote COX-2 expression [11]. More, other studies have suggested that CREB also represses 15-prostaglandin dehydrogenase (15-PDGH), which catalyzes the inactivation of PGE2 [29–31]. Therefore, the presently observed MRP4-dependent CREB phosphorylation may be another important mechanism regulating PGE2 production in the endometrial epithelial cells during embryo implantation. Of note, in addition to COX-2/PGE2, CREB seems to interact with other implantation genes such as Lif [32] and PPARg [33] in other systems, suggesting further role(s) of MRP4-regulated CREB in embryo implantation. A question remained how MRP4 contributes to CREB phosphorylation upon ENaC activation. We previously showed that the CREB phosphorylation is largely promoted by Ca2+ increase triggered by ENaC activation and depolarization [10]. Of note, CREB is responsive to both Ca2+ and cAMP. It has been reported that CREB phosphorylation can be evoked by PGE2 receptor E2 (EP2) activation and its coupled PKA signaling [34, 35]. Therefore, it is possible that MRP4-mediated PGE2 release may in turn promote CREB phosphorylation in epithelial cells. In this regard, MRP4, may not only mediate PGE2 release, but also contribute to a positive feedback that further enhances PGE2 production. However, the detailed underlying mechanism awaits further investigation.
The upregulation of MRP4 during embryo implantation, its responsiveness to ENaC activation by embryo-derived signal as well as its capacity in mediating PGE2 transport and promoting PGE2 production suggest an essential role of MRP4 in embryo implantation. This is confirmed by the present data obtained in the in vivo embryo implantation model showing that blockage of MRP4 function results in downregulation of implantation genes and implantation rate in mice. The present finding is consistent with the previous report showing a smaller little size in MRP4 knockout mice [20]. Of interest, the expression of MRP4 was reported in human eutopic and ectopic endometrial tissue and elevated in peritoneal endometriosis, which could be attenuated by the anti-inflammatory lipid lipoxin A(4) [36]. On the other hand, lipoxin A(4) was observed to block embryo implantation in mice [37]. In light of the present finding, the effect of lipoxinA(4) on embryo implantation could be due to its effect on downregulation of MRP4.
Like other members of MRP subfamily, MRP4 confers resistance to a wide variety of chemotherapeutic drugs [38, 39]. High levels of this gene are strongly predictive of poor outcome in cancers, including neuroblastoma, colorectal cancer and esophageal squamous cell carcinoma [18, 40, 41]. In addition, MRP4 has been shown to have remarkable ability to modulate cellular signaling processes besides its versatile efflux transport function as a potential therapeutic target in cancer treatment [17, 42]. It should be noted that there are striking similarities between the behavior of invasive blastocyst and that of cancer cells, such as migration, invasion and angiogenesis [43]. Indeed, the present study demonstrates a role of MRP4 in regulating ENaC-dependent CREB/COX-2/PGE2 signaling in embryo implantation. Similar finding has also been reported in esophageal squamous cell carcinoma [38]. Knockdown of MRP4 could attenuate the expression of p-CREB and COX-2, affecting PGE2 synthesis [41]; however, whether it involves ENaC was not investigated. Of note, the potential role of ENaC in the development and progression of multiple cancers has recently been recognized [44–47]. Therefore, the capacity of MRP4 in regulating ENaC-dependent signaling pathways during embryo implantation as demonstrated presently may provide new insights into the understanding of MRP4/ENaC related signaling in cancers as well. Given the multiple roles of both ENaC and CREB/COX-2/PGE2 signaling pathway in other physiological or pathological events, the role of MRP4 in regulation of ENaC-dependent CREB/COX-2/PGE2 signaling pathway may have far-reaching beyond embryo implantation.
MATERIALS AND METHODS
Mice and intrauterine injection
ICR mice were obtained from the Laboratory Animal Service Centre of the Chinese University of Hong Kong. All animal experiments were conducted in accordance with the university guidelines on animal experimentation, and approval by the Animal Ethics Committee of the Chinese University of Hong Kong was obtained for all related procedures. The day a vaginal plug was found after mating was identified as day 1. The intrauterine injection surgery under general anethesia was performed on day 3 after mating as previously reported [10]. MK-571 or vehicle control was injected into the lumen of each uterine horn close to the uterine-oviduct junction toward the uterine lumen. On day 7 after mating, the mice were killed by CO2 asphyxiation and implanted embryo numbers were counted. Uteri were collected for further analyses.
Cell culture
The human endometrial epithelial cell line was provided as a gift from Dr. Douglas A. Kniss from the Laboratory of Perinatal Research at the Ohio State University. The cells were cultured in DMEM supplemented with 10% fetal bovine serum (v/v) and 1% penicillin-streptomycin (v/v) in 5% CO2 incubators at 37°C.
RNA extraction and real-time PCR
Total RNA of cells were extracted using TRIzol reagent (Invitrogen Life Technologies) according to manufacturer’s instructions. 1 μg total RNA was applied on reverse transcription reaction using M-MLV reverse transcriptase (Promega) according to the manufacturer’s instructions. SYBR Green Master Mix (Tli RNase H Plus, Takara) was added to each PCR reaction along with cDNA and primers in a total volume of 10 μl. The primer sequences are listed in Table 1. Quantitative PCR were carried out in triplicate on a 96-well plate using an 7500 Fast Real-time PCR system (Applied Biosystems). The transcriptional expression of target genes were indicated with average CT value of GAPDH and calculated using the ΔΔCT method.
Table 1: Primers used for RT-PCR
Gene |
Primer |
Sequence |
Product size |
|---|---|---|---|
Mouse |
F |
GGCGCAGTTTATGTTGTCTG |
130 bp |
COX-2 |
R |
CAGCACTTCACCCATCAGTT |
|
Mouse |
F |
CCCTCATCAGTCACTCAGCA |
140 bp |
Claudin4 |
R |
AGCAAACGTCCACTGTCCTT |
|
Mouse |
F |
GTGTAAGGGCAGCGTTTCTT |
121 bp |
HoxA10 |
R |
CAGCCCCTTCAGAAAACAGT |
|
Mouse |
F |
AGCAGCAGTAAGGGCACAAT |
117 bp |
Lif |
R |
CCCCATTTGAGCATGAACTT |
|
Mouse |
F |
TGTCGGTTTCAGAAGTGCCTTG |
121 bp |
PPARg |
R |
TTCAGCTGGTCGATATCACTGGAG |
Immunoblotting
The cells was lysed in ice-cold RIPA lysis buffer (50 mM Tris-Cl, pH 7.5, 150 mM NaCl, 1% NP-40, 0.5% DOC, 0.1% SDS) with protease and phosphatase inhibitor cocktail (catalog #78443, Thermo Scientific) for 30 min on ice. Supernatant was colleceted after centrifugation at 14,000 rpm for 30 min at 4°C. Equal amounts of protein were resolved by SDS-polyacrylamide gel electrophoresis and electroblotted onto equilibrated nitrocellulose membrane. After blocking with 5% milk, the membranes were immunodetected for target proteins. Antibodies against MRP4 (1:100, Abcam, ab15602); phospho-CREB (1:1000, Cell Signaling, 9198), β-tubulin (1:2000, Santa Cruz, sc-9104) and β-actin (1:5000, Sigma, A1978). The protein bands were detected with HRP-conjugated antibodies and visualized by the enhanced chemiluminescence (ECL) assay (GE Healthcare) following manufacturer’s instructions. Signals were quantified by Imagine J software and defined as the ratio of target protein relative to internal loading control.
PGE2 ELISA
The human endometrial epithelial cells were grown in 24-well plates for 24 h. 1 % FBS in DMEM culture medium was used for 8 h to synchronize the cells before the experiment. After cell starvation, FBS-free DMEM medium was used for all the treatments and cell-free supernatant with PGE2 content was collected and measured using an EIA kit (Cayman Chemical, 514010).
Tissue fixation and cryosectioning
Uterus tissue from day 4 of pregnancy mice after intrauterine injection were harvested and fixed by immersion in 4% paraformaldehyde overnight. After three times washed in PBS, uterus were cryoprotected in 30% sucrose in PBS at 4°C for 24 hours, mounted in OCT embedding media (Tissue-Tek, 4583, Sakura) and frozen at -80°C. Cryosection were cut to a thickness of 5 μm using a cryostats (Shandon Cryotome, Thermo Scientific) and placed onto Superfrost/Plus microscope slides (Fisher Scientific).
Immunofluorescence
Sections were rehydrated in PBS for 5 mins, boiled in a microwave oven with citrate buffer (pH 6.0) for 20 mins and cooled down to room temperature and treated with 1% SDS in PBS for 4 mins. Sections were blocked with 1% bovine serum albumin (BSA) in PBS for 15 mins, incubated with primary antibody (MRP4, 1:20, Abcam, ab15602) overnight at 4°C and followed by incubation with fluorochrome-conjugated secondary antibody (invitrogen) for 1 h at room temperature. DAPI was used to stain cell nuclei. Images were acquired with a confocal microscope (Zeiss, Germany).
Statistical analysis
The results are shown as mean ± SEM. Differences in measured variables between two-group comparison were assessed by using Student’s unpaired t-test. One-way ANOVA for multiple comparisons was applied for comparing more than two groups. A Value of P < 0.05 was considered to be statistically significant.
ACKNOWLEDGMENTS
The study was supported in part by National Major Basic Research Program of China (2013CB967401 and 2013CB967404) and National Natural Science Foundation of China (No. 81471460 to Y.C.R.), (No. 81671860 to F.Z.), (No. 81370709 to H.C.C) and (No. 81571390 to Y.W).
CONFLICTS OF INTEREST
The authors declare that there are no conflicts of interest.
REFERENCES
1. Cha J, Sun X, Dey SK. Mechanisms of implantation: strategies for successful pregnancy. Nat Med. 2012; 18:1754-67. doi: 10.1038/nm.3012.
2. Ruan YC, Chen H, Chan HC. Ion channels in the endometrium: regulation of endometrial receptivity and embryo implantation. Hum Reprod Update. 2014; 20:517-29. doi: 10.1093/humupd/dmu006.
3. Norwitz ER, Schust DJ, Fisher SJ. Implantation and the survival of early pregnancy. N Engl J Med. 2001; 345:1400-8. doi: 10.1056/NEJMra000763.
4. Bonventre JV, Huang Z, Taheri MR, O'Leary E, Li E, Moskowitz MA, Sapirstein A. Reduced fertility and postischaemic brain injury in mice deficient in cytosolic phospholipase A2. Nature. 1997; 390:622-5. doi: 10.1038/37635.
5. Lim H, Paria BC, Das SK, Dinchuk JE, Langenbach R, Trzaskos JM, Dey SK. Multiple female reproductive failures in cyclooxygenase 2-deficient mice. Cell. 1997; 91:197-208.
6. Li DK, Liu L, Odouli R. Exposure to non-steroidal anti-inflammatory drugs during pregnancy and risk of miscarriage: population based cohort study. BMJ. 2003; 327:368. doi: 10.1136/bmj.327.7411.368.
7. Achache H, Tsafrir A, Prus D, Reich R, Revel A. Defective endometrial prostaglandin synthesis identified in patients with repeated implantation failure undergoing in vitro fertilization. Fertil Steril. 2010; 94:1271-8. doi: 10.1016/j.fertnstert.2009.07.1668.
8. Ruan YC, Zhou W, Chan HC. Regulation of smooth muscle contraction by the epithelium: role of prostaglandins. Physiology (Bethesda). 2011; 26:156-70. doi: 10.1152/physiol.00036.2010.
9. Dimitriadis E, Stoikos C, Baca M, Fairlie WD, McCoubrie JE, Salamonsen LA. Relaxin and prostaglandin E(2) regulate interleukin 11 during human endometrial stromal cell decidualization. J Clin Endocrinol Metab. 2005; 90:3458-65. doi: 10.1210/jc.2004-1014.
10. Ruan YC, Guo JH, Liu X, Zhang R, Tsang LL, Dong JD, Chen H, Yu MK, Jiang X, Zhang XH, Fok KL, Chung YW, Huang H, et al. Activation of the epithelial Na+ channel triggers prostaglandin E(2) release and production required for embryo implantation. Nat Med. 2012; 18:1112-7. doi: 10.1038/nm.2771.
11. Sun X, Ruan YC, Guo J, Chen H, Tsang LL, Zhang X, Jiang X, Chan HC. Regulation of miR-101/miR-199a-3p by the epithelial sodium channel during embryo implantation: involvement of CREB phosphorylation. Reproduction. 2014; 148:559-68. doi: 10.1530/REP-14-0386.
12. Schuster VL. Prostaglandin transport. Prostaglandins Other Lipid Mediat. 2002; 68-69:633-47.
13. Schuster VL. Molecular mechanisms of prostaglandin transport. Annu Rev Physiol. 1998; 60:221-42. doi: 10.1146/annurev.physiol.60.1.221.
14. Chen ZS, Lee K, Walther S, Raftogianis RB, Kuwano M, Zeng H, Kruh GD. Analysis of methotrexate and folate transport by multidrug resistance protein 4 (ABCC4): MRP4 is a component of the methotrexate efflux system. Cancer Res. 2002; 62:3144-50.
15. Chen ZS, Lee K, Kruh GD. Transport of cyclic nucleotides and estradiol 17-beta-D-glucuronide by multidrug resistance protein 4. Resistance to 6-mercaptopurine and 6-thioguanine. J Biol Chem. 2001; 276:33747-54. doi: 10.1074/jbc.M104833200.
16. Schuetz JD, Connelly MC, Sun D, Paibir SG, Flynn PM, Srinivas RV, Kumar A, Fridland A. MRP4: a previously unidentified factor in resistance to nucleoside-based antiviral drugs. Nat Med. 1999; 5:1048-51. doi: 10.1038/12487.
17. Russel FG, Koenderink JB, Masereeuw R. Multidrug resistance protein 4 (MRP4/ABCC4): a versatile efflux transporter for drugs and signalling molecules. Trends Pharmacol Sci. 2008; 29:200-7. doi: 10.1016/j.tips.2008.01.006.
18. Pereira C, Queiros S, Galaghar A, Sousa H, Pimentel-Nunes P, Brandao C, Moreira-Dias L, Medeiros R, Dinis-Ribeiro M. Genetic variability in key genes in prostaglandin E2 pathway (COX-2, HPGD, ABCC4 and SLCO2A1) and their involvement in colorectal cancer development. PLoS One. 2014; 9:e92000. doi: 10.1371/journal.pone.0092000.
19. Lacroix-Pepin N, Danyod G, Krishnaswamy N, Mondal S, Rong PM, Chapdelaine P, Fortier MA. The multidrug resistance-associated protein 4 (MRP4) appears as a functional carrier of prostaglandins regulated by oxytocin in the bovine endometrium. Endocrinology. 2011; 152:4993-5004. doi: 10.1210/en.2011-1406.
20. Morgan JA, Cheepala SB, Wang Y, Neale G, Adachi M, Nachagari D, Leggas M, Zhao W, Boyd K, Venkataramanan R, Schuetz JD. Deregulated hepatic metabolism exacerbates impaired testosterone production in Mrp4-deficient mice. J Biol Chem. 2012; 287:14456-66. doi: 10.1074/jbc.M111.319681.
21. Reid G, Wielinga P, Zelcer N, van der Heijden I, Kuil A, de Haas M, Wijnholds J, Borst P. The human multidrug resistance protein MRP4 functions as a prostaglandin efflux transporter and is inhibited by nonsteroidal antiinflammatory drugs. Proc Natl Acad Sci U S A. 2003; 100:9244-9. doi: 10.1073/pnas.1033060100.
22. Moriwaki K, Tsukita S, Furuse M. Tight junctions containing claudin 4 and 6 are essential for blastocyst formation in preimplantation mouse embryos. Dev Biol. 2007; 312:509-22. doi: 10.1016/j.ydbio.2007.09.049.
23. Daftary GS, Taylor HS. Pleiotropic effects of Hoxa10 on the functional development of peri-implantation endometrium. Mol Reprod Dev. 2004; 67:8-14. doi: 10.1002/mrd.20013.
24. Cheng JG, Chen JR, Hernandez L, Alvord WG, Stewart CL. Dual control of LIF expression and LIF receptor function regulate Stat3 activation at the onset of uterine receptivity and embryo implantation. Proc Natl Acad Sci U S A. 2001; 98:8680-5. doi: 10.1073/pnas.151180898.
25. Cammas L, Reinaud P, Bordas N, Dubois O, Germain G, Charpigny G. Developmental regulation of prostacyclin synthase and prostacyclin receptors in the ovine uterus and conceptus during the peri-implantation period. Reproduction. 2006; 131:917-27. doi: 10.1530/rep.1.00799.
26. Fang L, Chang HM, Cheng JC, Leung PC, Sun YP. Nitric oxide and cGMP induce COX-2 expression and PGE2 production in human granulosa cells through CREB signaling pathway. J Clin Endocrinol Metab. 2015; 100:E262-9. doi: 10.1210/jc.2014-2886.
27. Kanai N, Lu R, Satriano JA, Bao Y, Wolkoff AW, Schuster VL. Identification and characterization of a prostaglandin transporter. Science. 1995; 268:866-9.
28. Gao F, Lei W, Diao HL, Hu SJ, Luan LM, Yang ZM. Differential expression and regulation of prostaglandin transporter and metabolic enzymes in mouse uterus during blastocyst implantation. Fertil Steril. 2007; 88:1256-65. doi: 10.1016/j.fertnstert.2007.02.054.
29. Pino MS, Nawrocki ST, Cognetti F, Abruzzese JL, Xiong HQ, McConkey DJ. Prostaglandin E2 drives cyclooxygenase-2 expression via cyclic AMP response element activation in human pancreatic cancer cells. Cancer Biol Ther. 2005; 4:1263-9.
30. Tsatsanis C, Androulidaki A, Venihaki M, Margioris AN. Signalling networks regulating cyclooxygenase-2. Int J Biochem Cell Biol. 2006; 38:1654-61. doi: 10.1016/j.biocel.2006.03.021.
31. Castro-Sanchez L, Agra N, Llorente Izquierdo C, Motino O, Casado M, Bosca L, Martin-Sanz P. Regulation of 15-hydroxyprostaglandin dehydrogenase expression in hepatocellular carcinoma. Int J Biochem Cell Biol. 2013; 45:2501-11. doi: 10.1016/j.biocel.2013.08.005.
32. Boeuf H, Merienne K, Jacquot S, Duval D, Zeniou M, Hauss C, Reinhardt B, Huss-Garcia Y, Dierich A, Frank DA, Hanauer A, Kedinger C. The ribosomal S6 kinases, cAMP-responsive element-binding, and STAT3 proteins are regulated by different leukemia inhibitory factor signaling pathways in mouse embryonic stem cells. J Biol Chem. 2001; 276:46204-11. doi: 10.1074/jbc.M106718200.
33. Herzig S, Hedrick S, Morantte I, Koo SH, Galimi F, Montminy M. CREB controls hepatic lipid metabolism through nuclear hormone receptor PPAR-gamma. Nature. 2003; 426:190-3. doi: 10.1038/nature02110.
34. Fujino H, Salvi S, Regan JW. Differential regulation of phosphorylation of the cAMP response element-binding protein after activation of EP2 and EP4 prostanoid receptors by prostaglandin E2. Mol Pharmacol. 2005; 68:251-9. doi: 10.1124/mol.105.011833.
35. Ansari KM, Sung YM, He G, Fischer SM. Prostaglandin receptor EP2 is responsible for cyclooxygenase-2 induction by prostaglandin E2 in mouse skin. Carcinogenesis. 2007; 28:2063-8. doi: 10.1093/carcin/bgm011.
36. Gori I, Rodriguez Y, Pellegrini C, Achtari C, Hornung D, Chardonnens E, Wunder D, Fiche M, Canny GO. Augmented epithelial multidrug resistance-associated protein 4 expression in peritoneal endometriosis: regulation by lipoxin A(4). Fertil Steril. 2013; 99:1965-73 e2. doi: 10.1016/j.fertnstert.2013.01.146.
37. Xiong J, Zeng P, Cheng X, Miao S, Wu L, Zhou S, Wu P, Ye D. Lipoxin A4 blocks embryo implantation by controlling estrogen receptor alpha activity. Reproduction. 2013; 145:411-20. doi: 10.1530/REP-12-0469.
38. Szakacs G, Paterson JK, Ludwig JA, Booth-Genthe C, Gottesman MM. Targeting multidrug resistance in cancer. Nat Rev Drug Discov. 2006; 5:219-34. doi: 10.1038/nrd1984.
39. Chen ZS, Tiwari AK. Multidrug resistance proteins (MRPs/ABCCs) in cancer chemotherapy and genetic diseases. FEBS J. 2011; 278:3226-45. doi: 10.1111/j.1742-4658.2011.08235.x.
40. Norris MD, Smith J, Tanabe K, Tobin P, Flemming C, Scheffer GL, Wielinga P, Cohn SL, London WB, Marshall GM, Allen JD, Haber M. Expression of multidrug transporter MRP4/ABCC4 is a marker of poor prognosis in neuroblastoma and confers resistance to irinotecan in vitro. Mol Cancer Ther. 2005; 4:547-53. doi: 10.1158/1535-7163.MCT-04-0161.
41. Sun Y, Shi N, Lu H, Zhang J, Ma Y, Qiao Y, Mao Y, Jia K, Han L, Liu F, Li H, Lin Z, Li X, et al. ABCC4 copy number variation is associated with susceptibility to esophageal squamous cell carcinoma. Carcinogenesis. 2014; 35:1941-50. doi: 10.1093/carcin/bgu043.
42. Fletcher JI, Haber M, Henderson MJ, Norris MD. ABC transporters in cancer: more than just drug efflux pumps. Nat Rev Cancer. 2010; 10:147-56. doi: 10.1038/nrc2789.
43. Murray MJ, Lessey BA. Embryo implantation and tumor metastasis: common pathways of invasion and angiogenesis. Semin Reprod Endocrinol. 1999; 17:275-90. doi: 10.1055/s-2007-1016235.
44. Ousingsawat J, Spitzner M, Schreiber R, Kunzelmann K. Upregulation of colonic ion channels in APC (Min/+) mice. Pflugers Arch. 2008; 456:847-55. doi: 10.1007/s00424-008-0451-3.
45. Bondarava M, Li T, Endl E, Wehner F. α-ENaC is a functional element of the hypertonicity-induced cation channel in HepG2 cells and it mediates proliferation. Pflugers Arch. 2009; 458:675-87. doi: 10.1007/s00424-009-0649-z.
46. Amara S, Ivy MT, Myles EL, Tiriveedhi V. Sodium channel gammaENaC mediates IL-17 synergized high salt induced inflammatory stress in breast cancer cells. Cell Immunol. 2016; 302:1-10. doi: 10.1016/j.cellimm.2015.12.007.
47. Xu S, Liu C, Ma Y, Ji HL, Li X. Potential roles of amiloride-sensitive sodium channels in cancer development. Biomed Res Int. 2016; 2016:2190216. doi: 10.1155/2016/2190216.

