INTRODUCTION
Hepatocellular carcinoma (HCC) is approximately 90% of all cases of primary liver cancer, the 5th most common cancer worldwide and the third leading cause of cancer-related mortality [1, 2]. The main risk factors include hepatitis B and C virus infection, alcohol–related liver cirrhosis, non-alcoholic steatohepatitis and ingestion of aflatoxin B1 [3]. Patients with early-stage of HCC are amenable to therapies such as resection, liver transplantation or local ablation [4]. Nevertheless, less than 20% of patients are eligible for curative treatment including chemoembolization or sorafenib because of its late-stage, multiple comorbidities and hepatic dysfunction [5]. Also, even sorafenib has not been widely used because of its high cost and toxicity [6]. Thus, recently natural compunds are attractive for combination therapy or novel target therapy for HCC.
AMP-activated protein kinase (AMPK) is a key regulator of energy metabolism [7] by activating the synthesis of fatty acid, cholesterol, protein through phosphorylation of metabolic enzymes and ATP-generating processes, including glucose uptake [8]. A tumor suppressor liver kinase B1 (LKB1) is its upstream kinase that phosphorylates and activates AMPK signaling by encoding serine/threonine kinase [9, 10]. Also, many studies revealed that AMPK suppresses cell proliferation by the inhibition of cell cycle progression and regulation of mitosis [11, 12]. Thus, AMPK activation is a therapeutic target for the prevention and treatment of cancer by metformin and other anticancer agents.
Though many researchers have been trying to find magic bullets for cancer therapy for decades, recent attention moves toward to cancer prevention rather than treatment with no magic bullets for cancer fighting to date. Thus, it was well documented that several compounds from natural products have cancer preventive efficacies, such as curcumin [13–16], capsaicin [17, 18], resveratrol [19–22], ursolic acid [23], brazilin [24], isothiocyanates [25], tanshinone I [26], tanshinone IIa [27], coumestrol [28], ginkgetin [29], emodin [30] and auraptene [31–39].
In the same line, though labmertianic acid (LA) is known to have anti-obesity [40], stress-protective [41], anti-allergic [42] and neurotropic [43, 44] activities, its other anti-cancer studies have not been reported except our group’s report on anticancer effect of LA via androgen receptor(AR) ablation [45] inhibition until now. Thus, in the current study, the underlying apoptotic mechanism of LA was elucidated in association with reactive oxygen species(ROS) and liver kinase B1 (LKB1)/AMP activated protein kinase (AMPK)/acetyl-CoA carboxylase (ACC) signaling pathway in HCCs.
RESULTS
Cytotoxic effect of LA in human hepatocellular carcinoma (HCC) cells
The cytotoxicity of LA in HepG2 and SK-Hep1 and Hep3B hepatocellular carcinoma (HCC) cells and Chang hepatocytes was evaluated by using MTT assay. Cells were treated with indicated concentrations of LA (0, 10, 20, 40, 80 μM) for 24 h. As shown in Figure 1, LA significantly suppressed the viability of HepG2, SK-Hep1 and Hep3B cells in a concentration dependent fashion, but not Chang normal hepatocyte cells. However, we used noninvasive and lipidemic HepG2 cells and metastatic SK-Hep1 cells origianted from hepatoblastoma of white male rather than invasive Hep 3B cells originated from HCC of black male in next experiments [46], though the susceptibility of Hep 3 B cells to LA was almost similar to that of SK-Hep1 cells. Next, cell proliferation assay was conducted in HepG2 and SK-Hep1 cells using crystal violet staining. After exposure to LA for 24 h, 48 h and 72 h, LA significantly inhibited proliferation of two HCC cells in a concentration and time dependent manner (Figure 1C).
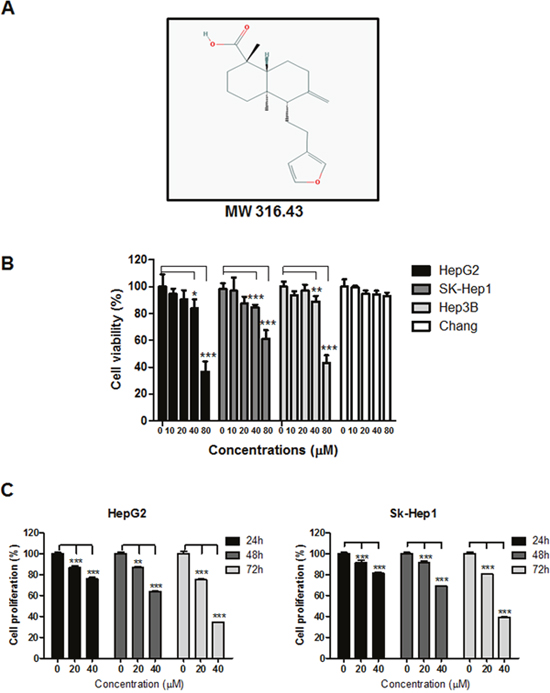
Figure 1: Effect of LA on cytotoxicity in HepG2, SK-Hep1, Hep3B and Chang cells. (A) Chemical structure of LA, (B) cytotoxic effect of LA in HCCs and Chang liver normal cells. Cells were seeded onto 96 well microplates and treated with various concentrations of LA (0, 10, 20, 40, 80 μM) for 24 h. Cell viability was evaluated by MTT assay. (C) Effect of LA on proliferation of HepG2 and SK-Hep1 cells. Cells were cultured with LA(0-40 μM) for 24h, 48h or 72 h and then stained with crystal violet. Data represent means ± SD. *, p<0.05, **, p<0.01 and ***, p<0.001 versus untreated control (n=3, one-way ANOVA, Tukey's test).
LA increased sub G1 population and regulated apoptosis related proteins in HCC cells
To test whether the cytotoxic effect of LA is due to apoptosis induction, cell cycle analysis and Western blotting were performed. LA increased sub-G1 in a dose-dependent fashion. Especially, LA at 40 μM elevated the sub-G1 population to 22.14% compared to untreated control (2.06%) (Figure 2A). Also, Western blotting showed that LA cleaved caspase-3, PARP and suppressed Bcl-2, Bcl-xL (Figure 2B) in HepG2 and SK-Hep1 cells. However, pan-caspase inhibitor Z-VAD-fmk reversed the cytotoxicity induced by LA in HepG2 cells (Figure 2C).
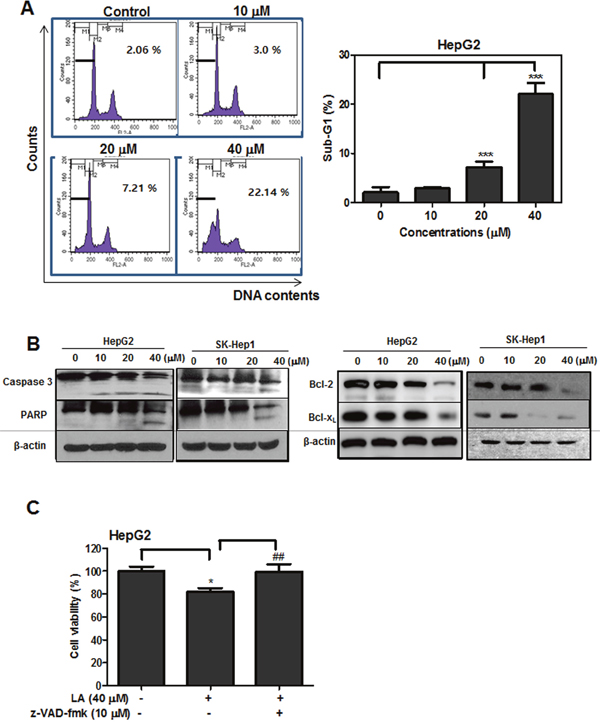
Figure 2: Effect of LA on sub-G1 population and apoptosis related proteins in hepatocellular carcinoma cells. (A) Effect of LA on sub-G1 population in HepG2 cells. HepG2 cells were exposed to LA (10, 20, 40 μM) for 24 h. After fixing in 75% ethanol, the cells were stained with propidium iodide and the cell cycle was analyzed by flow cytometry. Bar graphs showed quantification of sub-G1 cell population (%). Data represent means ± SD. **, p<0.01 versus untreated control. (B) Effect of LA on sub-G1 population and apoptosis related proteins in HCCs.HepG2 and SK-Hep1 cells were treated with various concentrations of LA for 24 h and subjected to Western blotting for caspase-3, PARP, Bcl-2, Bcl-xL and β-actin. (C) Effect of pancaspase inhbibtor Z-VAD-fmk on the viability of LA treated HepG2 cells. HepG2 cells were treated with 40 μM of LA with or without Z-VAD-fmk (10 μM). The cell viability was measured by MTT. Data represent means ± SD. *, p<0.05 and ***, p<0.001 versus untreated control. ##, p<0.01 versus LA (40 μM) treated group (n=3, one-way ANOVA, Tukey's test).
LA activated phosphorylation of LKB1/AMPK/ACC signaling in HCC cells
To confirm whether the apoptotic effect of LA is related to AMPK signaling, Western blotting was performed in LA treated HCC cells with antibodies of p-LKB1, p-AMPK and p-ACC. Here, phosphorylation of LKB1, AMPK and ACC was increased in a dose dependent manner in LA treated HepG2 and SK-Hep1 cells (Figure 3A). As shown in Figure 3B, the cytotoxic effect of LA was significantly reversed by AMPK inhibitor compound C treatment. In addition, Western blotting showed that LA suppressed the expression of PI3K, p-AKT, p-mTOR and COX-2 in HepG2 and SK-Hep1 cells (Figure 3C). Also, as shown in Figure 3D, LA activated phosphorylation of ERK and p38 and attenuated phosphorylation of JNK in HepG2 cells. In contrast, LA decreased phosphorylation of p38 and JNK and increased phosphorylation of ERK in SK-Hep1 cells.
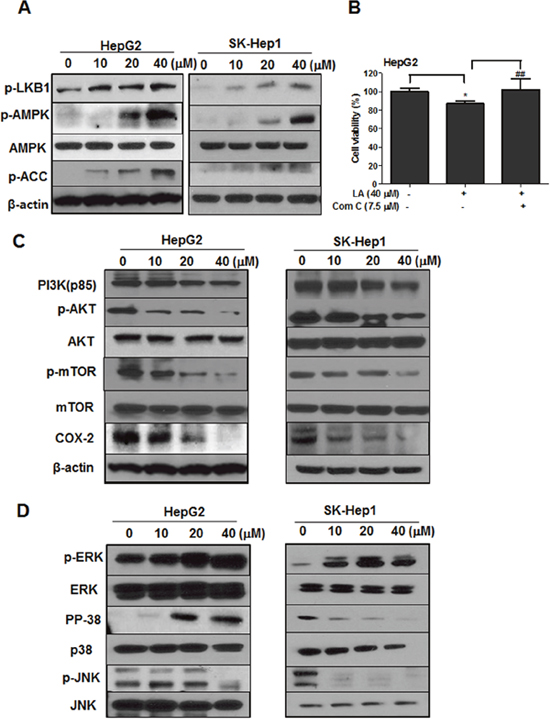
Figure 3: Effect of LA on AMPK and its related proteins in HCC cells. (A) HepG2 and SK-Hep1 cells were treated with indicated concentrations of LA for 24 h and the cells were subjected to Western blotting for p-LKB1, p-AMPK, AMPK, p-ACC, COX-2 and β-actin. (B) HepG2 cells were treated with 40 μM of LA with or without 7.5 μM of compound C for 24 h and MTT assay was conducted. Data represent means ± SD. *, p<0.05 versus untreated control. ##, p<0.01 versus LA (40 μM) only treated group (n=3, one-way ANOVA, Tukey's test). (C) HepG2 and SK-Hep1 cells were treated with various concentrations of LA and Western blotting was conducted with antibodies of PI3K, p-AKT, AKT, p-mTOR, mTOR, COX-2 and β-actin. (D) HepG2 and SK-Hep1 cells were treated with various concentrations of LA and Western blotting was performed with antibodies of p-ERK, ERK, p-p38, p38, p-JNK and JNK.
LA generated ROS production in HepG2 cells
To test whether ROS play a crictial role in LA induced apoptosis, DCFH-DA staining was used to measure the ROS generation by FACS Calibur. As shown in Figure 4, LA induced ROS production in HepG2 and SK-Hep1 cells in a time dependent manner compared to untreated control.
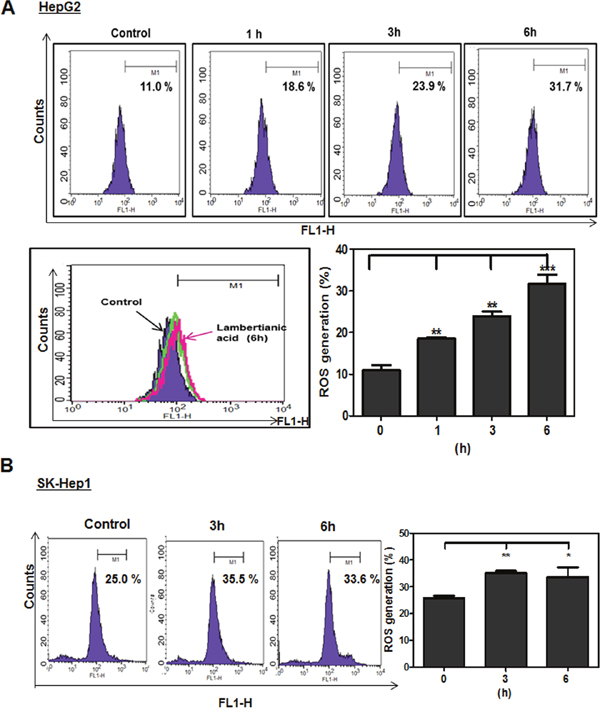
Figure 4: Effect of LA on ROS production in HepG2 and SK-Hep1 cells. HepG2 (A) and SK-Hep1 (B) cells were treated with LA (40 μM) for 1 h, 3 h, 6 h and then 5 μM DCFH-DA for 30 min at 37°C. Experiments were performed three times. Fluorescence intensity was measured by FACS Calibur. Bar graphs showed quantification of ROS generation (%). Data represent means ± SD. * p<0.05, **, p<0.01 and ***, p<0.001 versus untreated control (n=3, one-way ANOVA, Tukey's test).
ROS dependent AMPK activation is critically involved in LA induced apoptosis in HepG2 and SK-Hep1 cells
Given that ROS generation is related to AMPK mediated apoptosis [47, 48], cell cycle anaylsis and Western blotting were conducted with AMPK inhibitor compound C or ROS inhibitor NAC in LA treated HepG2 cells. The increased sub-G1 population by LA was significantly attenuated in HepG2 and SK-Hep1 cells by compound C and/or NAC (Figure 5A). Consistently, Cell apoptosis assay using Annexin-V/PI double staining revealed that LA increased the percentage of early apoptotic cells (Annexin V+/PI− staining: 30.1 %) for 24 h and late apoptotic or necrosis cells (Annexin V+/PI+ staining: 22.9%) for 48h. Conversely, increased early/late apoptosis by LA was significantly reversed by compound C or NAC in HepG2 cells (Figure 5B). Likewise, the increased phosphorylation of AMPK and ACC, PARP cleavage and decreased expression of Bcl-2 and Cox-2 by LA were reversed in HepG2 and SK-Hep1 cells by compound C (Figure 5C) or in HepG2 cells by NAC (Figure 5D).
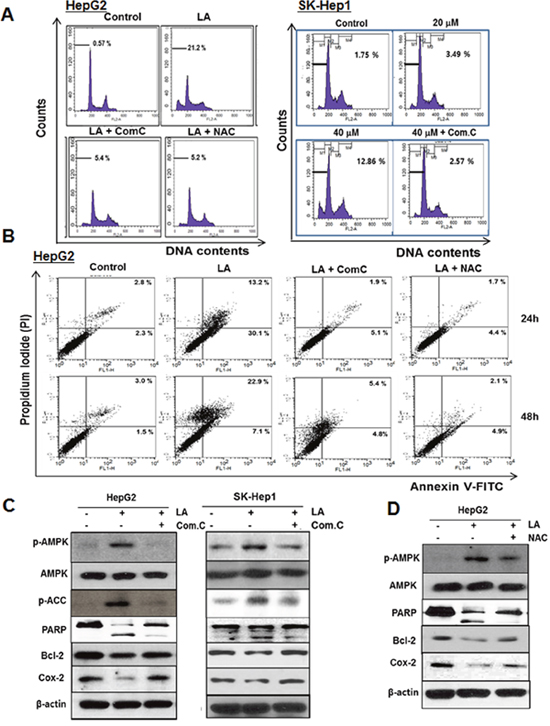
Figure 5: ROS and AMPK signalings mediate LA induced apoptosis in HCC cells. (A) HepG2 and SK-Hep1 cells were treated with LA (40 μM) in the absence and presence of compound C (7.5 μM) or NAC (5 mM). After fixing in 75% ethanol, the cells were stained with PI and the cell cycle was analyzed by flow cytometry. (B) HepG2 cells were treated with LA (40 μM) for 24 h and 48 h in the absence and presence of compound C (7.5 μM) or NAC (5 mM). The cells were stained using FITC-Annexin V/PI dye and early and late apoptotic portions were detected by flow cytometry. (C) HepG2 and SK-Hep1 cells were treated with various concentrations of LA (20, 40 μM) for 24 h with or without compound C. Then Western blotting was conducted in LA and/or compound C treated HCCs with antibodies of p-AMPK, AMPK, p-ACC, PARP, Bcl-2, COX-2 and β-actin. (D) HepG2 cells were exposed to LA (40 μM) in the presence or absence of NAC and subjected to Western blotting for p-AMPK, AMPK, PARP, Bcl-2, COX-2 and β-actin.
To further confirm the critical role of AMPK in LA induced apoptosis, AMPK α wild type and knockout (KO) mouse embryonic fibroblast (MEF) cells were used in this study. Herein LA increased sub-G1 population to 10.24% in AMPK α wild type MEF cells compard to untreated control (4.77%), while there were no significant changes of subG1 population in AMPK KO MEF cells (Figure 6A). Consitently, Western blotting showed that cleavages of PARP and caspase-3 and phosphorylation of AMPK and ACC were induced in AMPK α wild type MEF cells by LA, but not in AMPK KO MEF cells (Figure 6B). Of note, STRING database showed that Protein-Protein Interaction(PPI) scores between AMPK and other proteins such as ACC, LKB1, mTOR and AKT1 were found 0.998, 0.995, 0.981 and 0.480, respectively (Figure 6C). Next, immunoprecipitation (IP) was performed with lysates from HepG2 cells using anti-AMPK antibody and Western-blot analysis was also conducted to confirm the binding of AMPK and LKB1. Here IP revealed that AMPK was directly bound to LKB1 in LA treated HepG2 cells (Figure 6D).
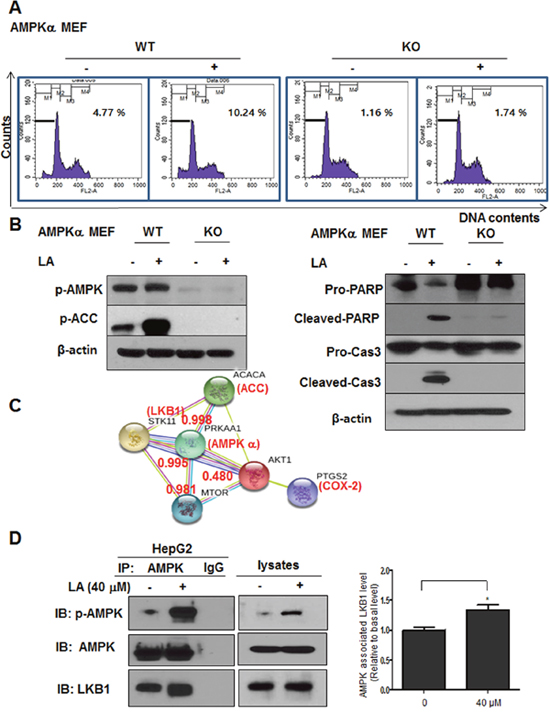
Figure 6: The critical role of AMPK in LA induced apoptosis in MEF cells and its interaction proteins. (A) AMPKα wild type and AMPK knockout MEF cells were treated with LA (40 μM) for 24 h. After fixing in 75% ethanol, the cells were stained with PI and the cell cycle was analyzed by flow cytometry. (B) AMPKα wild type and AMPK knockout MEF cells were treated with LA (40 μM) for 24 h and subjected to Western blotting with antibodies of p-AMPK, p-ACC, PARP, Cleaved-PARP, Caspase3, Cleaved-caspase3 and β-actin. (C) Interacting proteins with AMPK through PPI networks by STRING database. Red text (interaction score). Different color lines represent the interaction score between AMPK and its related proteins. (D) Effect of LA the binding between LKB1 and AMPK in HepG2 cells. Immunoprecipitation (IP) was performed with lysates from HepG2 cells using anti-AMPK antibody and Western blotting was performed to detect AMPK and LKB1 in whole cell lysates. Bar graphs show associated level between AMPK and LKB1 in LA treated HepG2 cells. *p<0.05 versus untreated control (Student’s t-test).
DISCUSSION
Hepatocellular carcinoma (HCC) as one of the most malignant human cancers [49] is known to be caused by risk factors including hepatitis B and C virus infection, alcohol abuse, non-alcoholic fatty liver disease, aflatoxins, diabetes, obesity, and genetic factors [50]. Nevertheless, the evident therapeutics for HCC still remain unclear so far [51, 52]. Hence, the development of novel therapies for HCC has been requested all over the world.
Apoptosis is well defined as a programmed cell death; there are several types of cell death including apoptosis, pyroptosis, necrosis, or autophagy [53–55]. It is characterized by cell shrinkage, nuclear condensation and fragmentation, loss of adhesion to extracellular matrix and membrane blebbing in the cells [56]. Nowadays, apoptosis induction is recognized as one of the important strategies for cancer prevention or treatment [57, 58].
In the current study, the apoptotic mechanism of LA was elucidated in HCCs. LA suppressed the viability of three HCC cells in a dose-dependent manner. To confirm the cytotoxicity of LA was due to apoptosis induction, cell cycle analysis was conducted. LA significantly increased sub-G1 population and the number of Annexin V/PI stained cells in HepG2 and SK-Hep1 cells, indicating that cytotoxicity of LA is induced by apoptotic effect of LA [59].
The cysteine-dependent aspartate-specific proteases as caspases have pivotal roles in apoptosis induction. Caspases consist of two groups; initiator caspases (caspase-2, -8, -9 and -10) and effector caspases (caspase-3 and -7) [60]. Once the process of intrinsic apoptosis pathway is initiated, cytochrome c is released from the mitochondria, which is a peripheral protein of the mitochondrial inner membrane [61]. The released cytochrome c attaches to apoptosis-activating factor-1 (Apaf-1) and procaspase-9 to form apoptosome in cytosol [62]. After procaspase-9 is activated by apoptosome, which in turn activates procaspase-3 to caspase-3 leading to apoptosis [60]. Here, LA cleaved PARP, caspase-3 and inhibited antiapoptotic proteins such as Bcl-2, Bcl-xL and also suppressed the expression of COX-2, PI3K, p-AKT and p-mTOR as survival pathway proteins in two HCC cells, implying LA induces apoptosis via inhibition of antiapoptotic and survival proteins. Also, the apoptotic effect of LA was blocked by pan-caspase inhibitor, Z-VAD-fmk, indicating that apoptosis induced by LA is mainly via caspase activation. Also, It was well documented that MAPKs including ERK, p38 and JNK regulate multicellular functions such as proliferation, differentiation, mitosis, gene expression, cell survival and apoptosis [63]. As shown in Figure 3D, LA activated phosphorylation of ERK and p38 and attenuated phosphorylation of JNK in HepG2 cells, while it decreased phosphorylation of p38 and JNK and increased phosphorylation of ERK in SK-Hep1 cells, implying cell specific effect of LA on MAPKs and further mechanistic study in the near future.
AMPK is a serine/threonine protein kinase, a key regulator of cellular metabolism and plays an important role in apoptosis [64, 65]. Several reports have revealed that AMPK can be a potential therapeutic target for cancer treatment [66, 67]. Here, LA induced phosphorylation of AMPK, its upstream LKB1 and downstream ACC and also decreased protein expression of COX-2 in HCC cells, indicating the important role of LKB1/AMPK/ACC signaling in LA induced anticancer effect, since AMPK related signaling is critically involved in survival and proliferation of cancer cells through Warburg effect during lipid metabolism including lipogenesis [68, 69].
It is well documented that the increased level of ROS is often observed during apoptosis induction in several cells [70]. ROS are ubiquitous among biological activities and excessive generation of ROS has been shown to induce damage in a variety of cancer cells via disruption of lipid membranes, proteins and DNA [71]. The current study revealed that LA increased ROS production in a time dependent manner. Conversely, the increase of sub G1 population and Annexin V/PI stained cells by LA was reversed by AMPK inhibitor compound C or ROS scavenger NAC in two HCC cells. Consistently, compound C blocked the ability of LA to induce phosphorylation of AMPK/ACC, PARP cleavage and decreased expression of Bcl-2 and COX-2 in two HCC cells. Also, NAC reversed phosphorylation of AMPK, PARP cleavage and decreased expression of Bcl-2 and COX-2 in HepG2 cells, demonstrating that LA generates ROS and subsequently induces AMPK phosphorylation, PARP cleavage and inhibits antiapoptotic proteins such as Bcl-2 and COX-2 leading to apoptosis in HCC cells (Figure 7).
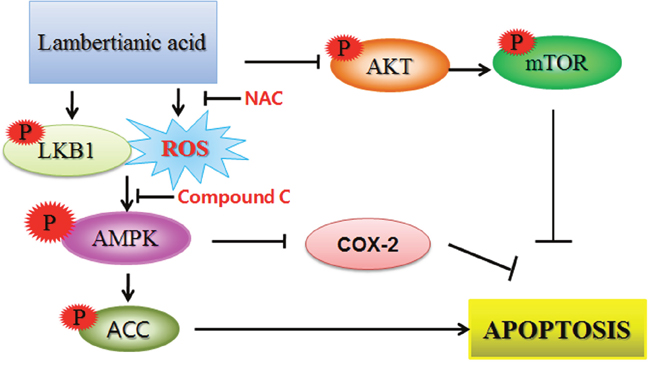
Figure 7: The schematic diagram for LA induced apoptosis via phosphorylation of LKB1/AMPK/ACC, ROS production and inhibition of p-AKT/mTOR signaling.
Notably, LA increased sub-G1 population only in AMPK α wild type MEF cells, but not in AMPK KO MEF cells. Similarly, LA induced cleavages of PARP and caspase-3 and phosphorylation of AMPK/ACC in AMPK α wild type MEF cells, but not in AMPK KO MEF cells, indicating the pivotal role of LKB1/AMPK/ACC signaling in LA induced apoptosis. Overall, our findings support scientific evidence that LA can be a potent chemopreventive candidate for HCC treatment via ROS dependent phosphorylation of LKB1/AMPK/ACC signaling.
MATERIALS AND METHODS
Isolation of LA
Pinus koraiensis leaves (3 kg) were pulverized, immersed in 50% MeOH (10 L) for 3 days and distilled to be concentrated for 10 h by using Rotary Evaporator(IKA Korea Limited, Seoul, Korea). Then the MeOH extracts were partitioned with EtOAc / distilled water (1:1) and the water layer was suspended and partitioned with n-butanol/distilled water. A part of EtOAc fraction was subjected to a celite column chromatography and eluted with CHCl3-MeOH (3:1) to yield 15 fractions. Among these fractions, a distinct and vivid red-purple spot from fr. 6 was isolated, purified and identified as lambetianic acid (LA) with over 98% purity based on spectroscopic analyses such as NMR, MS, and IR as well as the comparison of the data with those reported in the literature [72].
Cell culture
Human hepatocellular carcinoma(HCC) HepG2, Hep 3B,SK-Hep1 and normal hepatocytes Chang cells were obtained from American Type Culture Collection (ATCC). AMPK α WT and KO mouse embryonic fibroblast (MEF) cells were obtained from Dr. Joohuh Ha. HepG2, SK-Hep1, Chang and MEF cells were cultured in DMEM supplemented with 10% FBS and 1% antibiotic (Welgene, South Korea).
Cytotoxicity assay
The cytotoxicity of LA was measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. In brief, HepG2, SK-Hep1 and Chang cells (1×104 cells/well) were seeded onto 96-well culture plate and exposed to various concentrations of LA for 24 h. The cells were incubated with MTT (1 mg/mL) (Sigma Chemical) for 2 h and then treated with MTT lysis solution overnight. Optical density (OD) was measured using a microplate reader (Molecular Devices Co., USA) at 570 nm. Cell viability was calculated as a percentage of viable cells in LA treated group versus untreated control.
Crystal violet assay
For viability and proliferation, crystal violet assay was performed inHepG2 and SK-Hep1 cells. The cells (1×105 cells/well) were seeded onto 6-well culture plate and exposed to various concentrations of LA for 24 h, 48h and 72h. The cells were fixed (4% paraformaldehyde) and stained with crystal violet solution (40% ethanol, 60% PBS and 0.5% crystal violet). Fifteen min later, 1 ml of 10% acetic acid was added to each well, and the absorbance was read at 590 nm using a microplate reader (Molecular Devices Co., USA).
Cell cycle analysis
HepG2 and SK-Hep1 cells (2 × 105 cells/ml) were treated with LA (0, 10, 20 or 40 μM) for 24 h, washed twice with cold PBS and fixed in 75% ethanol at −20 °C for 24 h. The cells were incubated with RNase A (10 mg/ml) for 1 h at 37°C and stained with PI (50 μg/ml) for 30 min at room temperature in dark. The stained cells were analyzed for the DNA content by FACSCalibur (Becton Dickinson, Franklin Lakes, NJ, USA) using CellQuest Software.
Annexin V/propidium iodide apoptosis assay
Cell apoptosis assay was performed using the double-staining method of the Annexin-V/PI apoptosis detection kit (BD Pharmingen, Franklin Lakes, NJ, USA) according to the manufacturer's instructions. HepG2 cells (2 × 105 cells/ml) were treated with LA (0, 20 or 40 μM) for 24 h and 48 h. Cells were stained with Annexin V-FITC/PI dye and analyzed immediately by FACSCalibur (Becton Dickinson, Franklin Lakes, NJ, USA). Apoptotic cells were identified as either Annexin V+/PI− staining (early apoptosis) or Annexin V+/PI+ staining (late apoptosis or necrosis cells).
Measurement of ROS generation
2,7-Dichlorofluorescein diacetate (DCFH-DA) was used to measure the levels of ROS production. HepG2 and SK-Hep1 cells were treated with LA (40 μM) for 3 h, 6 h and then 5 μM DCFH-DA for 30 min at 37°C. Fluorescence intensity was measured by FACSCalibur (Becton Dickinson, Franklin Lakes, NJ, USA).
Western blotting and co-immunoprecipitation (co-IP)
HepG2 and SK-Hep1 cells (1 × 106 cells/ml) were treated with various concentrations of LA (0, 10, 20 or 40 μM) for 24 h, lyzed in lysis buffer (50 mM Tris–HCl, pH 7.4, 150 mM NaCl, 1% Triton X-100, 0.1% SDS, 1 mM EDTA, 1 mM Na3VO4, 1 mM NaF, and 1× protease inhibitor cocktail) on ice, and spun down at 14,000×g for 20 min at 4°C. The supernatants were collected and quantified for protein concentration by using RC DC protein assay kit (Bio-Rad, Hercules, CA, USA), The protein samples were separated on 4–12% NuPAGE Bis–Tris gels (Novex, Carlsbad, CA, USA) and transferred to a Hybond ECL transfer membrane for detection with antibodies for PARP, cleaved-PARP, Caspase-3, cleaved-Caspase-3, Bcl-2, Bcl-XL, phospho-AMPK, AMPK, phospho-ACC, phospho-LKB1, LKB1, PI3K, phospho-AKT, phospho-mTOR, COX-2 (Cell signaling Technology, Beverly, MA, USA) and β-actin (Sigma, St. Louis, MO, USA). For immunoprecipitation experiment, cell lysates were precleared with protein A/G-agarose beads and subsequently incubated for an 1 - 2 h with protein G/A beads covalently coupled with anti-AMPK antibody. Immune complexes were washed four times with cell extraction buffer. Eluted samples or whole cellular lysates were resolved by SDS-PAGE and proteins were detected by Western blotting using the indicated antibodies. Then densitometric analysis was performed using ImageJ software. Unless otherwise specified, actin protein was immunoblotted in order to normalize the quantity of sample protein.
Statistical analyses
Statistical analysis was performed by Graphpad Prism 5.0 software (GraphPad Software, San Diego, CA, USA). The statistical significance was determined by using one-way ANOVA and Tukey's test or Student's t-test. All data were expressed as means ± standard deviation (SD). Statistically significant difference (P<0.05).
Abbreviations
HCC, hepatocellular carcinoma; LA, lambertianic acid; AMPK, AMP-activated protein kinase; ACC, acetyl-CoA carboxylase; LKB1, liver kinase B1; COX-2, cyclooxygenase-2; mTOR, mammalian target of rapamycin; ROS, reactive oxygen species; NAC, N-acetyl-L-cysteine; MEF, mouse embryonic fibroblasts; MTT, 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide; OD, optical density; FBS, fetal bovine serum; SDS, sodium dodecylsulfate; PBS, phosphate buffered saline; SD, standard deviation; Caspase, cysteine aspartyl-specific protease; PARP, poly (ADP-ribose) polymerase; DAPI, 4’-6-diamidino-2-phenylindole; ECL, enhanced chemiluminescence; AKT, protein kinase B; FACS, fluorescence-activated cell sorting; Bcl-2, B-cell lymphoma 2; DNA, deoxyribonucleic acid; APAF-1, apoptosis-activating factor-1; MAPK, mitogen-activated protein kinase. PPI, protein-protein interaction.
ACKNOWLEDGMENTS
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (MEST) (2014R1A2A10052872).
CONFLICTS OF INTEREST
The authors declare no competing financial interests.
REFERENCES
1. de Oliveria Andrade LJ, D'Oliveira A, Melo RC, De Souza EC, Costa Silva CA, Parana R. Association between hepatitis C and hepatocellular carcinoma. J Glob Infect Dis. 2009; 1:33-37.
2. Bosch FX, Ribes J, Cleries R, Diaz M. Epidemiology of hepatocellular carcinoma. Clin Liver Dis. 2005; 9:191-211, v.
3. Bruix J, Gores GJ, Mazzaferro V. Hepatocellular carcinoma: clinical frontiers and perspectives. Gut. 2014; 63:844-855.
4. Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, Gores G. Hepatocellular carcinoma. Nat Rev Dis Primers. 2016; 2:16018.
5. Salem R, Gilbertsen M, Butt Z, Memon K, Vouche M, Hickey R, Baker T, Abecassis MM, Atassi R, Riaz A, Cella D, Burns JL, Ganger D, et al. Increased quality of life among hepatocellular carcinoma patients treated with radioembolization, compared with chemoembolization. Clin Gastroenterol Hepatol. 2013; 11:1358-1365. e1.
6. Zhong J, Dong X, Xiu P, Wang F, Liu J, Wei H, Xu Z, Liu F, Li T, Li J. Blocking autophagy enhances meloxicam lethality to hepatocellular carcinoma by promotion of endoplasmic reticulum stress. Cell Prolif. 2015; 48:691-704.
7. Xie Z, Dong Y, Scholz R, Neumann D, Zou MH. Phosphorylation of LKB1 at serine 428 by protein kinase C-zeta is required for metformin-enhanced activation of the AMP-activated protein kinase in endothelial cells. Circulation. 2008; 117:952-962.
8. Jiang YS, Lei JA, Feng F, Liang QM, Wang FR. Probucol suppresses human glioma cell proliferation in vitro via ROS production and LKB1-AMPK activation. Acta Pharmacol Sin. 2014; 35:1556-1565.
9. Hardie DG. The LKB1-AMPK pathway-friend or foe in cancer? Cancer Cell. 2013; 23:131-132.
10. Shackelford DB, Shaw RJ. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer. 2009; 9:563-575.
11. Song P, Wang S, He C, Liang B, Viollet B, Zou MH. AMPKalpha2 deletion exacerbates neointima formation by upregulating Skp2 in vascular smooth muscle cells. Circ Res. 2011; 109:1230-1239.
12. Fu YN, Xiao H, Ma XW, Jiang SY, Xu M, Zhang YY. Metformin attenuates pressure overload-induced cardiac hypertrophy via AMPK activation. Acta Pharmacol Sin. 2011; 32:879-887.
13. Cheng KW, Wong CC, Mattheolabakis G, Xie G, Huang L, Rigas B. Curcumin enhances the lung cancer chemopreventive efficacy of phospho-sulindac by improving its pharmacokinetics. Int J Oncol. 2013; 43:895-902.
14. Dujic J, Kippenberger S, Ramirez-Bosca A, Diaz-Alperi J, Bereiter-Hahn J, Kaufmann R, Bernd A, Hofmann M. Curcumin in combination with visible light inhibits tumor growth in a xenograft tumor model. Int J Cancer. 2009; 124:1422-1428.
15. Lee YK, Lee WS, Hwang JT, Kwon DY, Surh YJ, Park OJ. Curcumin exerts antidifferentiation effect through AMPKalpha-PPAR-gamma in 3T3-L1 adipocytes and antiproliferatory effect through AMPKalpha-COX-2 in cancer cells. J Agric Food Chem. 2009; 57:305-310.
16. Picone P, Nuzzo D, Caruana L, Messina E, Scafidi V, Di Carlo M. Curcumin induces apoptosis in human neuroblastoma cells via inhibition of AKT and Foxo3a nuclear translocation. Free Radic Res. 2014:1-12.
17. Jung DL, Lee SD, Choi IH, Na HS, Hong SU. Effects of electroacupuncture on capsaicin-induced model of atopic dermatitis in rats. J Dermatol Sci. 2014; 74:23-30.
18. Pramanik KC, Fofaria NM, Gupta P, Srivastava SK. CBP-mediated FOXO-1 acetylation inhibits pancreatic tumor growth by targeting SirT. Mol Cancer Ther. 2014; 13:687-698.
19. Alkhalaf M. Resveratrol-induced apoptosis is associated with activation of p53 and inhibition of protein translation in T47D human breast cancer cells. Pharmacology. 2007; 80:134-143.
20. Alkhalaf M. Resveratrol-induced growth inhibition in MDA-MB-231 breast cancer cells is associated with mitogen-activated protein kinase signaling and protein translation. Eur J Cancer Prev. 2007; 16:334-341.
21. Aluyen JK, Ton QN, Tran T, Yang AE, Gottlieb HB, Bellanger RA. Resveratrol: potential as anticancer agent. J Diet Suppl. 2012; 9:45-56.
22. Whitlock NC, Bahn JH, Lee SH, Eling TE, Baek SJ. Resveratrol-induced apoptosis is mediated by early growth response-1, Kruppel-like factor 4, and activating transcription factor 3. Cancer Prev Res (Phila). 2011; 4:116-127.
23. Song YH, Jeong SJ, Kwon HY, Kim B, Kim SH, Yoo DY. Ursolic acid from Oldenlandia diffusa induces apoptosis via activation of caspases and phosphorylation of glycogen synthase kinase 3 beta in SK-OV-3 ovarian cancer cells. Biol Pharm Bull. 2012; 35:1022-1028.
24. Kim B, Kim SH, Jeong SJ, Sohn EJ, Jung JH, Lee MH. Brazilin induces apoptosis and G2/M arrest via inactivation of histone deacetylase in multiple myeloma U266 cells. J Agric Food Chem. 2012; 60:9882-9889.
25. Gupta P, Kim B, Kim SH, Srivastava SK. Molecular targets of isothiocyanates in cancer: recent advances. Mol Nutr Food Res. 2014; 58:1685-1707.
26. Shin EA, Sohn EJ, Won G, Choi JU, Jeong M, Kim B, Kim MJ, Kim SH. Upregulation of microRNA135a-3p and death receptor 5 plays a critical role in Tanshinone I sensitized prostate cancer cells to TRAIL induced apoptosis. Oncotarget. 2014; 5:5624-5636. https://doi.org/10.18632/oncotarget.2152.
27. Yun SM, Jung JH, Jeong SJ, Sohn EJ, Kim B, Kim SH. Tanshinone IIA induces autophagic cell death via activation of AMPK and ERK and inhibition of mTOR and p70 S6K in KBM-5 leukemia cells. Phytother Res. 2013; 28:458-464.
28. Cho SY, Cho S, Park E, Kim B, Sohn EJ, Oh B, Lee EO, Lee HJ, Kim SH. Coumestrol suppresses hypoxia inducible factor 1alpha by inhibiting ROS mediated sphingosine kinase 1 in hypoxic PC-3 prostate cancer cells. Bioorg Med Chem Lett. 2014; 24:2560-2564.
29. You OH, Kim SH, Kim B, Sohn EJ, Lee HJ, Shim BS, Yun M, Kwon BM. Ginkgetin induces apoptosis via activation of caspase and inhibition of survival genes in PC-3 prostate cancer cells. Bioorg Med Chem Lett. 2013; 23:2692-2695.
30. Ha MK, Song YH, Jeong SJ, Lee HJ, Jung JH, Kim B, Song HS, Huh JE, Kim SH. Emodin inhibits proinflammatory responses and inactivates histone deacetylase 1 in hypoxic rheumatoid synoviocytes. Biol Pharm Bull. 2011; 34:1432-1437.
31. de Medina P, Genovese S, Paillasse MR, Mazaheri M, Caze-Subra S, Bystricky K, Curini M, Silvente-Poirot S, Epifano F, Poirot M. Auraptene is an inhibitor of cholesterol esterification and a modulator of estrogen receptors. Mol Pharmacol. 2010; 78:827-836.
32. Epifano F, Genovese S, Miller R, Majumdar AP. Auraptene and its effects on the re-emergence of colon cancer stem cells. Phytother Res. 2012; 27:784-786.
33. Epifano F, Molinaro G, Genovese S, Ngomba RT, Nicoletti F, Curini M. Neuroprotective effect of prenyloxycoumarins from edible vegetables. Neurosci Lett. 2008; 443:57-60.
34. Jun DY, Kim JS, Park HS, Han CR, Fang Z, Woo MH, Rhee IK, Kim YH. Apoptogenic activity of auraptene of Zanthoxylum schinifolium toward human acute leukemia Jurkat T cells is associated with ER stress-mediated caspase-8 activation that stimulates mitochondria-dependent or -independent caspase cascade. Carcinogenesis. 2007; 28:1303-1313.
35. Kawabata K, Murakami A, Ohigashi H. Auraptene decreases the activity of matrix metalloproteinases in dextran sulfate sodium-induced ulcerative colitis in ICR mice. Biosci Biotechnol Biochem. 2006; 70:3062-3065.
36. Murakami A, Kuki W, Takahashi Y, Yonei H, Nakamura Y, Ohto Y, Ohigashi H, Koshimizu K. Auraptene, a citrus coumarin, inhibits 12-O-tetradecanoylphorbol-13-acetate-induced tumor promotion in ICR mouse skin, possibly through suppression of superoxide generation in leukocytes. Jpn J Cancer Res. 1997; 88:443-452.
37. Sekiguchi H, Takabayashi F, Irie K, Murakami A. Auraptene attenuates gastritis via reduction of Helicobacter pylori colonization and pro-inflammatory mediator production in C57BL/6 mice. J Med Food. 2012; 15:658-663.
38. Soltani F, Mosaffa F, Iranshahi M, Karimi G, Malekaneh M, Haghighi F, Behravan J. Auraptene from Ferula szowitsiana protects human peripheral lymphocytes against oxidative stress. Phytother Res. 2010; 24:85-89.
39. Soltani F, Mosaffa F, Iranshahi M, Karimi G, Malekaneh M, Haghighi F, Behravan J. Auraptene from Ferula szowitsiana protects human peripheral lymphocytes against oxidative stress. Phytother Res. 2009; 24:85-89.
40. Lee MS, Cho SM, Lee MH, Lee EO, Kim SH, Lee HJ. Ethanol extract of Pinus koraiensis leaves containing lambertianic acid exerts anti-obesity and hypolipidemic effects by activating adenosine monophosphate-activated protein kinase (AMPK). BMC Complement Altern Med. 2016; 16:51.
41. Avgustinovich DF, Fomina MK, Sorokina IV, Tolstikova TG. Complex investigation of the effects of lambertianic acid amide in female mice under conditions of social discomfort. Bull Exp Biol Med. 2014; 157:583-587.
42. Chae HS, Chin YW. Anti-allergic effect of lambertianic acid from Thuja orientalis in mouse bone marrow-derived mast cells. Immunopharmacol Immunotoxicol. 2011; 34:250-255.
43. Tolstikova TG, Dolgikh MP, Tolstikov GA. Lambertian acid and its amino derivatives: a new group of perspective neurotropic agents. Dokl Biol Sci. 2000; 374:445-447.
44. Tolstikova TG, Voevoda TV, Dolgikh MP, Sorokina IV. [Neurotropic activity of lambertian acid and its amino derivatives]. [Article in Russian]. Eksp Klin Farmakol. 2002; 65:9-11.
45. Lee MS, Lee SO, Kim SH, Lee EO, Lee HJ. Anti-cancer effect of lambertianic acid by inhibiting the AR in LNCaP cells. Int J Mol Sci. 2016; 17.
46. Jung CW, Song TJ, Lee KO, Choi SB, Kim WB, Suh SO, Kim YC, Choi SY. Characterization of hepatocellular carcinoma cell lines based on cell adhesion molecules. Int J Mol Med. 2012; 29:1158-1164.
47. Wu SB, Wu YT, Wu TP, Wei YH. Role of AMPK-mediated adaptive responses in human cells with mitochondrial dysfunction to oxidative stress. Biochim Biophys Acta. 2014; 1840:1331-1344.
48. Sid B, Verrax J, Calderon PB. Role of AMPK activation in oxidative cell damage: implications for alcohol-induced liver disease. Biochem Pharmacol. 2013; 86:200-209.
49. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011; 365:1118-1127.
50. Thomas M. Molecular targeted therapy for hepatocellular carcinoma. J Gastroenterol. 2009; 44:136-141.
51. Yang JD, Roberts LR. Hepatocellular carcinoma: a global view. Nat Rev Gastroenterol Hepatol. 2010; 7:448-458.
52. Spangenberg HC, Thimme R, Blum HE. Targeted therapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2009; 6:423-432.
53. Fitzwalter BE, Thorburn A. Recent insights into cell death and autophagy. FEBS J. 2015; 282:4279-4288.
54. Green DR, Llambi F. Cell death signaling. Cold Spring Harb Perspect Biol. 2015; 7.
55. Thompson CB. Apoptosis in the pathogenesis and treatment of disease. Science. 1995; 267:1456-1462.
56. Portt L, Norman G, Clapp C, Greenwood M, Greenwood MT. Anti-apoptosis and cell survival: a review. Biochim Biophys Acta. 2011; 1813:238-259.
57. Kleinberg L, Davidson B. Cell survival and apoptosis-related molecules in cancer cells in effusions: a comprehensive review. Diagn Cytopathol. 2009; 37:613-624.
58. Ouyang L, Shi Z, Zhao S, Wang FT, Zhou TT, Liu B, Bao JK. Programmed cell death pathways in cancer: a review of apoptosis, autophagy and programmed necrosis. Cell Prolif. 2012; 45:487-498.
59. Han MH, Lee WS, Nagappan A, Kim HJ, Park C, Kim GY, Hong SH, Kim ND, Kim G, Ryu CH, Shin SC, Choi YH. Polyphenols from Korean prostrate spurge Euphorbia supina induce apoptosis through the Fas-associated extrinsic pathway and activation of ERK in human leukemic U937 cells. Oncol Rep. 2016; 36:99-107.
60. Kim B, Srivastava SK, Kim SH. Caspase-9 as a therapeutic target for treating cancer. Expert Opin Ther Targets. 2014; 19:113-127.
61. Garrido C, Galluzzi L, Brunet M, Puig PE, Didelot C, Kroemer G. Mechanisms of cytochrome c release from mitochondria. Cell Death Differ. 2006; 13:1423-1433.
62. Beesoo R, Neergheen-Bhujun V, Bhagooli R, Bahorun T. Apoptosis inducing lead compounds isolated from marine organisms of potential relevance in cancer treatment. Mutat Res. 2014; 768:84-97.
63. Burotto M, Chiou VL, Lee JM, Kohn EC. The MAPK pathway across different malignancies: a new perspective. Cancer. 2014; 120:3446-3456.
64. Qi D, Young LH. AMPK: energy sensor and survival mechanism in the ischemic heart. Trends Endocrinol Metab. 2015; 26:422-429.
65. Anwar MA, Kheir WA, Eid S, Fares J, Liu X, Eid AH, Eid AA. Colorectal and prostate cancer risk in diabetes: metformin, an actor behind the scene. J Cancer. 2014; 5:736-744.
66. Song X, Kim SY, Zhang L, Tang D, Bartlett DL, Kwon YT, Lee YJ. Role of AMP-activated protein kinase in cross-talk between apoptosis and autophagy in human colon cancer. Cell Death Dis. 2014; 5:e1504.
67. Rehman G, Shehzad A, Khan AL, Hamayun M. Role of AMP-activated protein kinase in cancer therapy. Arch Pharm (Weinheim). 2014; 347:457-468.
68. Faubert B, Vincent EE, Poffenberger MC, Jones RG. The AMP-activated protein kinase (AMPK) and cancer: many faces of a metabolic regulator. Cancer Lett. 2015; 356:165-170.
69. Luo J, Hong Y, Lu Y, Qiu S, Chaganty BK, Zhang L, Wang X, Li Q, Fan Z. Acetyl-CoA carboxylase rewires cancer metabolism to allow cancer cells to survive inhibition of the Warburg effect by cetuximab. Cancer Lett. 2017; 384:39-49.
70. Mates JM, Sanchez-Jimenez FM. Role of reactive oxygen species in apoptosis: implications for cancer therapy. Int J Biochem Cell Biol. 2000; 32:157-170.
71. Chvanov M, Huang W, Jin T, Wen L, Armstrong J, Elliot V, Alston B, Burdyga A, Criddle DN, Sutton R, Tepikin AV. Novel lipophilic probe for detecting near-membrane reactive oxygen species responses and its application for studies of pancreatic acinar cells: effects of pyocyanin and L-ornithine. Antioxid Redox Signal. 2014; 22:451-464.
72. Yang X, Ding Y, Sun ZH, Zhang DM. Studies on chemical constituents of Pinus armandii. Acta Pharm Sin. 2005; 40:435-437.

