INTRODUCTION
Historically, the role of radiotherapy (RT) for the treatment of hepatocellular carcinoma (HCC) has been limited because of the low tolerance of the whole liver to RT and the risk of radiation-induced liver disease (RILD) [1–3]. With the introduction of 3-dimensional conformal radiotherapy (3DCRT), however, the delivery of conformal partial liver RT allows for safe dose escalation as the liver parenchyma is arranged with a functionally parallel architecture [4, 5]. In addition, further development of radiotherapy techniques, including intensity-modulated radiotherapy (IMRT) and stereotactic body radiotherapy (SBRT), has expanded the indication of RT for the treatment of HCC from a palliative to a curative-intent aim [6–9]. The current practical guidelines for the treatment of HCC are summarized in Table 1 [10–17]. Although the use of RT has been limited in several guidelines, the recommendation of RT as a local treatment modality from recent versions of the National Comprehensive Cancer Network (NCCN) guidelines [17] and the practice guidelines from the Korean Liver Cancer Study Group and National Cancer Center Korea [11], is noteworthy changes. Especially, the NCCN guidelines suggest IMRT is helpful in selected HCC patients. However, there is insufficient evidence to support the routine use of RT as shown in Table 1. The level of evidence regarding RT use is only B-C. Therefore, further efforts are needed to maximize the efficacy and minimize the toxicity of RT. The solution might be provided by the application of IMRT, which is the most advanced RT technique currently available.
Table 1: Current practical guidelines for the management of hepatocellular carcinoma (HCC) around the world
Region |
Group |
Abbreviations |
Publishing year |
Comments about RT |
|---|---|---|---|---|
Asia |
Korean Liver Cancer Study Group and the National Cancer Center [11] |
KLCSG-NCC |
2014 |
· EBRT can be performed in HCC patients if liver functions are CP class A or superb B and the irradiated total liver volume receiving ≥ 30 Gy is ≤ 60% (B1a)). · EBRT can be considered for HCC patients ineligible for surgical resection, LT, RFA, PEIT, or TACE (C1). · EBRT can be considered for HCC patients who show incomplete response to TACE when the dose-volume criteria in Recommendation 1 are met (B2). · EBRT can be considered for HCC patients with PVTT when the dose-volume criteria in Recommendation 1 are met (C1). · EBRT is performed to alleviate symptoms caused by primary HCC or its metastases (B1). |
Japan Society of Hepatology [12] |
JSH |
2013 |
· 3DCRT can be considered for patients with PVTT or unresectable tumors who are contraindicated for other standard treatment methods because of complications or other reasons (C1). · There is insufficient scientific evidence to support the extension of survival duration by RT alone. However, survival duration is expected to increase in unresectable cases if RT is combined with TACE (C1). |
|
Asian Pacific Association for the Study of the Liver [13] |
APASL |
2010 |
None |
|
Europe |
European Association for the Study of the Liver and the European Organization for Research and Treatment of Cancer [14] |
EASL–EORTC |
2012 |
· 3DCRT is under investigation, and there is no evidence to support this therapeutic approach in the management of HCC (2C) |
European Society for Medical Oncology - European Society of Digestive Oncology [15] |
ESMO-ESDO |
2012 |
· EBRT can be used to control pain in patients with bone metastases [II, B]. |
|
USA |
American Association for the Study of Liver Disease [16] |
AASLD |
2010 |
· There are multiple other treatment modalities such as octreotide, interferon, EBRT, tamoxifen, or anti-androgenic therapy, but none have been shown to improve survival |
National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Hepatobiliary Cancers (Version 1.2016)] [17] |
NCCN |
2016 |
· All tumors irrespective the location may be amenable EBRT (SBRT, IMRT, or 3DCRT). · There is growing evidence for the usefulness of SBRT in the management of patients with HCC. SBRT can be considered as an alternative to the ablation/embolization techniques mentioned above or when there therapies have failed or are contraindicated. · Proton beam therapy may be appropriate in specific situations. · Palliative EBRT is appropriate for symptom control and/or prevention of complications from metastatic HCC lesions, such as bone or brain. |
RT: radiotherapy, EBRT: external beam radiotherapy, CP: Child Pugh, LT: liver transplantation, RFA: radiofrequency ablation, PEIT: percutaneous ethanol injection therapy, TACE: transarterial chemoembolization, PVTT: portal vein tumor thrombosis, 3DCRT: 3-dimensional conformal radiotherapy, SBRT; stereotactic body radiotherapy, IMRT: intensity-modulated radiotherapy.
a) the alphabet means the level of evidence and the number means the grade of recommendation. The definition of grading system in detail should be checked at each guideline.
Compared with previous treatment techniques such as 3DCRT, the features of IMRT are the inverse treatment planning process and the use of large number of treatment fields or subfields that fall within the conformal beam’s-eye view of the target [18]. Therefore, IMRT provides a high precision and an exquisitely conformal dose distribution using multiple beams, each with a non-uniform intensity profile [19, 20]. Conventional multileaf collimator (MLC)-mounted linear accelerators are equipped with the following three types of IMRT delivery systems that enable the creation of non-uniform intensity profiles: (1) step-and-shoot IMRT, in which small MLC-generated segments are used, and radiation is not delivered while the leaves move to create the next segment, (2) sliding window IMRT, in which modulated MLC velocity in multiple static radiation fields is used, and the radiation is delivered as the leaves are moving, and (3) volumetric modulated arc therapy (VMAT), which is a rotational form of IMRT whereby moving MLC and changing dose delivery rates occur during rotation [21]. On the other hand, helical tomotherapy is an independent rotational IMRT machine that has a binary MLC to quickly open and close apertures in front of the different beam elements in the fan beam. It uses a slow, continuous movement of the patient support system with a rapidly rotating X-ray source to make many rotations possible in a relatively short period of time using a helical dose delivery technique similar to the modern spiral CT scanner [18].
Currently, IMRT is postulated to be an attractive technique for the treatment of HCC, and is becoming more widely applied [22]. Although intrafractional and interfractional setup error from the effects of respiratory motions may introduce dosimetric errors, the application of 4-D CT, advanced immobilization system, use of gating and tracking techniques, and fractionation schedule could minimize physically treatment inaccuracy [23–27]. On the other hand, the clinical advantages and potential disadvantages of IMRT for treating HCC have not been fully established. This article reviews dosimetric and clinical studies using IMRT for the treatment of HCC. To compare the characteristics of IMRT techniques, we define the step-and-shoot and sliding window techniques as static IMRT (s-IMRT), the rotational IMRT using conventional MLCs as VMAT, and the rotational IMRT using helical tomotherapy as helical IMRT (h-IMRT).
DOSIMETRIC STUDIES
Target volume coverage
When the conformity index (CI) and homogeneity index (HI) were used to assess target volume coverage, Lee et al. [28] reported significantly improvement in both parameters with h-IMRT compared with s-IMRT and 3DCRT for 12 patients with advanced HCC. Chen et al. [29] showed that VMAT and s-IMRT achieved significantly better CI than 3DCRT. Additionally, VMAT showed significant improvement compared with s-IMRT and 3DCRT with regard to HI. To compare among IMRT techniques, Hsieh et al. [30] made h-IMRT and s-IMRT plans in nine HCC patients with portal vein tumor thrombosis (PVTT). CI values showed no significant difference between h-IMRT and s-IMRT, but HI values significantly improved with h-IMRT compared with s-IMRT. Park et al. [31] reported that the CI of VMAT was significantly better than that of s-IMRT. Another study showed no significant differences in CI values between h-IMRT and s-IMRT, however h-IMRT achieved significantly better HI values than s-IMRT [32].
Sparing of the liver
The liver sparing effect between 3DCRT and IMRT techniques is affected by tumor location and size. Lee et al. [28] analyzed the dosimetric parameters of normal liver based on the location of HCC lesion (right lobe vs. left lobe vs. both lobes). The high dose region, which included the volume of the normal liver receiving more than 40 Gy (V40), V50, and V60, was significantly smaller in h-IMRT than s-IMRT and 3DCRT. On the other hand, the low dose region (V20) and mean dose were significantly smaller in 3DCRT than h-IMRT and s-IMRT. These differences were meaningful when the tumor was present in a single lobe, either right or left, but disappeared when the tumor was located in both lobes. Cheng et al. [29] compared liver protection, based on tumor size in 20 patients with centrally located HCC on the right lobe. The mean dose of the normal liver was significantly higher using s-IMRT than 3DCRT and VMAT. The low dose region, including V5 and V10, was significantly smaller in 3DCRT, and the high dose region, including V20 and V30, was significantly smaller in VMAT. In larger HCC (> 8 cm), the mean dose was lower in 3DCRT than s-IMRT and VMAT, leading the authors to suggest that 3DCRT might be a more suitable technique for larger tumors (> 8 cm) located in the right lobe, in terms of minimizing the risk of RILD. This size limitation of IMRT to achieve a maximal liver sparing effect was supported by another study that compared s-IMRT and proton beam therapy in 10 HCC patients [33]. Using the Lyman-normal-tissue complication probability model to estimate the risk of RILD, the risk of RILD for s-IMRT dramatically increased between nominal diameters of 6.3–7.8 cm gross tumor volume.
Among the IMRT techniques, the mean and low dose region of the normal liver in h-IMRT and VMAT were higher than s-IMRT [28, 30, 31, 34]. To minimize this difference, one study performed h-IMRT re-planning [32]. Specifically to reduce V15 of the normal liver as much as possible, which was identified as the best dosimetric parameter for predicting RILD in their previous study, the mean dose of the total liver and V15 of the normal liver were still significantly higher in h-IMRT than s-IMRT [35]. Another method to improve liver sparing in h-IMRT is to apply a directional beam blocking technique, especially for left lobe lesions [28]. This technique reduces the normal liver dose similar to s-IMRT, although homogenous dose distribution within the tumor slightly deteriorates. In case of VMAT, the use of non-coplanar arcs provides superior liver protection than s-IMRT for HCC located in the left lobe [36]. Because of the asymmetric shape and eccentric location of the liver, non-coplanar arcs from the right cranial direction may focus more radiation on the left lobe, and threfore reduce the radiation dose to the normal liver.
Despite many dosimetric studies, it is unclear whether the increase of the low dose region in the normal liver using IMRT techniques, compared to 3DCRT, could increase the risk of RILD. From a radiobiology perspective, the liver is the classic example of a parallel architecture model [37]. However, although RILD mainly occurs if the partial volume damaged exceeds a threshold, the risk of RILD is also added by dose distribution of the functional reserve and subunit radiosensitivity [38, 39]. As a result, the increased risk of RILD associated with IMRT may be related to dose distribution throughout the liver and the patients’ functional reserve, rather than the maximum dose to a limited area [40]. This is supported by evidence from the lung, another typical example of a parallel architecture model, where an increase in radiation pneumonitis is seen when there is an increase to the low dose region during IMRT. An extra dose constraint for the low dose region (V5), in addition to V20 and the mean dose significantly reduced radiation pneumonitis after IMRT in locally advanced non-small cell lung cancer [41, 42]. Therefore, further studies are needed to evaluate the potential risk of RILD in the low dose region associated with IMRT in HCC patients.
Sparing of other organs at risk (OARs) except the liver
Kuo et al. [34] reported that mean doses of the stomach and left kidney were significantly lower using s-IMRT than 3DCRT in nine patients with unresectable HCC. However, there was no difference between 3DCRT and VMAT. Lee et al. [28] found that the best sparing of high dose regions in the stomach and small bowel (V40, V50, and V60) were achieved with h-IMRT, followed by s-IMRT, and 3DCRT without statistical significance. In HCCs located in both lobes, however, s-IMRT showed better sparing of the stomach than h-IMRT. For right-lobe HCC, irradiation dose to the left kidney was higher with h-IMRT because of the helical delivery nature. Park et al. [31] also suggested that VMAT tended to be more effective in sparing non-liver OARs than s-IMRT. For patients with primary HCC and PVTT, V35 of the duodenum in VMAT was significantly smaller than that in s-IMRT. For patients with primary HCC alone, V20 of the kidney was significantly smaller in VMAT than s-IMRT. The maximal point dose of the spinal cord was significantly lower in VMAT than s-IMRT regardless of tumor location.
The prevalence of gastric ulcer in cirrhotic patients was as high as 20%, compared to 2–4% in the general population because of underlying cirrhosis and portal hypertensive gastropathy (PHG) [43, 44]. The prevalence of gastroduodenal toxicity in HCC patients who received 3DCRT and underwent endoscopy was 30–50% [45–47]. Until now, we cannot determine whether the gastroduodenal toxicities developed as a consequence of RT or occurred spontaneously due to cirrhosis and PHG. Severe hematemesis by aggravation of pre-existing PHG was reported in HCC patients receiving 3DCRT regardless of radiation dose [48]. Considering that V25 or V35 of gastroduodenum were significant dosimetric parameters affecting severe gastroduodenal toxicity in two studies, the application of IMRT would reduce gastroduodenal toxicity in HCC patients by sparing the high dose regions of the gastroduodenum as mentioned above [45, 47].
The strengths and weakness of IMRT techniques are summarized in Table 2.
Table 2: The strengths and weakness of intensity-modulated radiotherapy (IMRT) techniques for hepatocelluar carcinoma
s-IMRT |
h-IMRT |
VMAT |
|
|---|---|---|---|
Strength |
Compared with 3DCRT - Improving target coverage - Sparing OARs |
- |
- |
Compared with h-IMRT and VMAT - Sparing the normal liver |
Compared with s-IMRT - Same or better homogeneous dose distribution within target - Sparing non-liver OARs |
Compared with s-IMRT - Same or better homogeneous dose distribution within target - Sparing non-liver OARs - Lower MUs - Shorter treatment time: reduction of intra-fractional movement; improvement of patient’s comfort; higher patient throughput |
|
Weakness |
Compared with 3DCRT - Higher MUs - Longer treatment time - Larger low dose region of OARs - Less sparing the normal liver in case of large tumor > 6–8 cm |
- |
- |
Compared with h-IMRT or VMAT - More dependent on the beam angle and the experience of the physicist |
Compared with s-IMRT - Larger low dose region of the normal liver (consider a directional block) |
Compared with s-IMRT - Larger low dose region of the normal liver (consider use of non-coplanar arc) - Limitation of non-coplanar arc: availability of only asymmetric partial arc; Decrease of advantage duo to increased treatment time by couch rotation and increased MUs |
s-IMRT: static IMRT using step-and-shoot technique and sliding window technique delivered by conventional multileaf collimator (MLC)-mounted linear accelerators, h-IMRT: helical IMRT using rotational dose delivery by binary MLC mounted helical tomotherapy, VMAT: volumetric modulated arc therapy using rotational dose delivery by conventional MLC mounted linear accelerators, 3DCRT: 3-dimensional conformal radiotherapy, OARs: organs at risk, MUs: monitor units.
CLINICAL STUDIES
3DCRT vs. IMRT
There were two clinical studies that compared 3DCRT to IMRT for the treatment of HCC, and both used h-IMRT technique (Table 3). Yoon et al. [49] retrospectively reviewed 187 patients with locally advanced HCC treated with RT. Sixty-five patients received h-IMRT (median fractional dose and total dose; 2.5 Gy and 50 Gy) and 122 patients received 3DCRT (median fractional dose and total dose; 1.8 Gy and 45 Gy). Baseline characteristics were not significantly different between the two groups. With median follow-up of 21 months, the local control rate (LCR) and overall survival (OS) rate at 3 years were significantly different according to RT techniques; the LCRs were 28% and 47% (P = 0.007) for 3DCRT and h-IMRT, respectively, and the OS rates were 14% and 33% (P < 0.001) for 3DCRT and h-IMRT, respectively. The use of h-IMRT was the significant parameter for OS in both univariate and multivariate analyses. On the other hand, RILD or severe toxicity ≥ grade 3 according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) was not different. The authors suggested that delivery of high doses without increased toxicity, as achieved with the use of h-IMRT led to improve LCR, leading to a longer OS. A recent study also reported the efficacy of h-IMRT [50]. They compared 3DCRT to h-IMRT in 118 advanced HCC patients with PVTT and/or inferior vena cava tumor thrombosis (IVCTT). Fifty-four patients received h-IMRT (median total dose; 60 Gy) and 64 patients received 3DCRT (median total dose; 54 Gy). The overall objective response rate (RR), including complete response and partial response, was 43% in the 3DCRT group and 70% in the h-IMRT group (P = 0.056). When the tumor thrombi were assessed, the objective RR was significantly different, being 47% after 3DCRT and 67% after h-IMRT (P = 0.031). With median follow-up of 11.8 months, the OS rate at 1 year was 36% in patients who received 3DCRT and 59% in patients who received h-IMRT (P = 0.005). Severe toxicity ≥ grade 3 as defined by the Radiation Therapy Oncology Group (RTOG) toxicity criteria was not different. The authors suggested that the better RR after h-IMRT, especially for the tumor thrombi, allowed more patients to receive further trans-arterial chemo-embolization (TACE) after RT, and both increased RR and additional treatments improved survival.
Table 3: Clinical studies compared 3-dimensional conformal radiotherapy (3DCRT) with helical intensity-modulated radiotherapy (h-IMRT) by helical tomotherapy for hepatocellular carcinoma
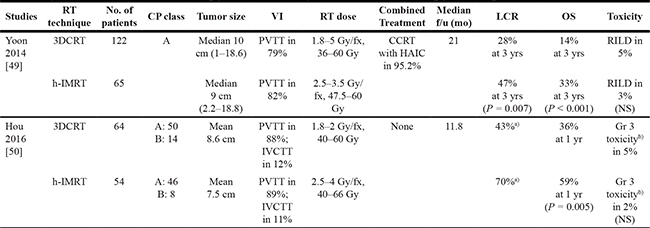
CP class: Child-Pugh class, VI: vascular invasion, LCR: local control rate, OS: overall survival rate, PVTT: portal vein tumor thrombosis, CCRT: concurrent chemo-radiotherapy, HAIC: hepatic arterial infusion chemotherapy, RILD: radiation-induced liver disease, IVCTT: inferior vena cava tumor thrombosis, NS: no significant.
a)means overall response rate.
b)is classified by the grading system of the Radiation Therapy Oncology Group.
IMRT studies
Several studies have reported treatment outcomes after IMRT for HCC, as shown in Table 4 [51–59]. The objective RR ranged from 43% to 74%, and the OS rate at 1 year ranged from 45% to 85%. The diversity of treatment outcomes among the IMRT studies may be mainly due to differences in baseline characteristics of the patients. Although all studies included patients with Child-Pugh (CP) class B and patients with PVTT, the proportion of these patients were different among the individual studies, and it has been well documented that they are important prognostic factors for survival [60–62]. Kang et al. [51] found that CP class and PVTT were significant parameters affecting RR after s-IMRT. And objective response was the only significant parameter affecting OS in a multivariate analysis. Kong et al. [55] found that CP class and PVTT were significant parameters for OS on multivariate analysis. Kim et al. [56] reported that CP class was a significant parameter that predicted the response of PVTT, and involvement of the main portal trunk was a significant parameter for an unfavorable OS. In terms of dose-response relationship, only one study showed that a higher biologically effective dose > 65.5 Gy10 contributed significantly to superior local control, both in univariate analysis and multivariate analyses [57]. On the other hand, other studies have not verified the association between RT dose and RR or survival [54–56].
Table 4: Clinical studies treated with intensity-modulated radiotherapy (IMRT) for hepatocellular carcinoma
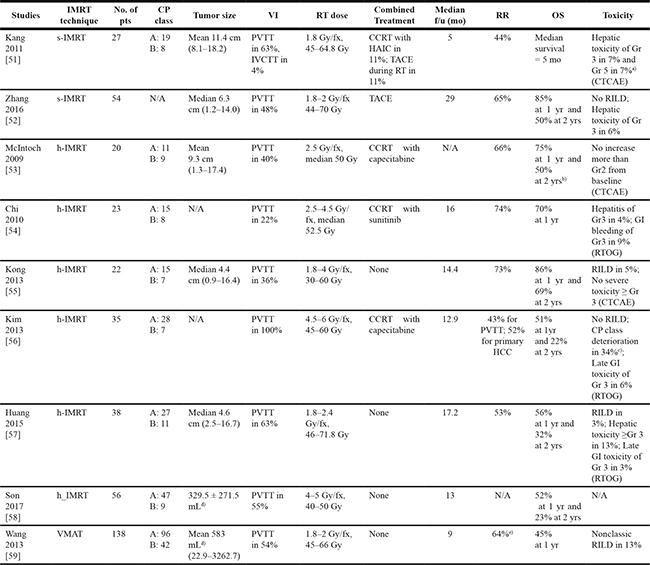
CP class: Child-Pugh class, VI: vascular invasion, RR; response rate including complete response and partial response, OS: overall survival rate, s-IMRT: static IMRT using step-and-shoot technique and sliding window technique delivered by conventional multileaf collimator (MLC)-mounted linear accelerators, h-IMRT: helical IMRT using rotational dose delivery by binary MLC mounted helical tomotherapy, VMAT: volumetric modulated arc therapy using rotational dose delivery by conventional MLC mounted linear accelerators, PVTT: portal vein tumor thrombosis, IVCTT: inferior vena cava tumor thrombosis, CCRT: concurrent chemo-radiotherapy, HAIC: hepatic arterial infusion chemotherapy, TACE: transarterial chemo-embolization, CTCAE: the National Cancer Institute Common Terminology Criteria for Adverse Events, N/A: not available, RTOG; the Radiation Therapy Oncology Group toxicity criteria, RILD: radiation-induced liver disease.
a)Fatal hepatic toxicity occurred only in patients who received combine treatment: 1 patient received TACE and 1 patient received HAIC.
b)was estimated in patients with CP A class.
c)12 patients (34%) experienced CP class deterioration: 2 patients experienced local tumor progression and 2 patients had the progression of distant metastases.
d)means planning target volume.
e)Percentage are relative to 109 patients with available follow-up image.
Table 4 also shows IMRT-related toxicity. Combined treatment tends to increase severe toxicity. Kang et al. [51] reported two fatal hepatic toxicities (grade 5 according to CTCAE). Both patients received combined treatments; one patient started s-IMRT 10 days after TACE, and the other patient received concurrent chemoradiotherapy (CCRT) with hepatic arterial infusion chemotherapy (HAIC). They recommended that special attention should be given to any other treatment given during or immediately after IMRT. One study concurrently used sunitinib during IMRT and reported the highest rate of gastrointestinal (GI) bleeding (9%) [54]. Sunitinib is a multityrosine kinase inhibitor, and one of the vascular endothelial growth factor inhibitors (VEGFIs). It is considered experimentally as a radiosensitizer under clinical setting [63]. Although the authors did not suggest a relationship between GI bleeding and the use of sunitinib, there have been some case reports about VEGFIs, including sunitinib, related to GI perforation [64]. Recently, a study reported that the use of VEGFIs ≤ 3 months after SBRT significantly increased severe GI toxicity [65]. Therefore, clinicians should remember the possibility of higher risk of GI toxicity when IMRT with a hypofractionation regimen is combined with VEGFIs.
Another important issue about IMRT-related toxicity is the application of various toxicity criteria among studies, especially for hepatic toxicity. This causes changes in the incidence of hepatic toxicity, and makes it difficult to compare studies. Studies listed in Table 4 applied different toxicity criteria, including RILD alone, CTCAE or RTOG criteria alone, both RILD and CTCAE or RTOG criteria, as well as RILD, the change of CP class, and RTOG criteria. Historically, hepatic toxicity after RT has been documented as RILD. CP class and CTCAE have been used together to grade the prognosis of RILD, or used independently to assess hepatic toxicity [66]. RILD is useful in comparing results to historical studies, but rarely occurs in an era whereby advanced technology like IMRT or SBRT are used. The change of CP class is directly associated with patients’ prognosis, but some problems are associated with this measurement. Both the degree of ascites and encephalopathy are subjective assessments evaluated by performing physical examination alone, and the parameters are categorized with arbitrary cut-off points [67]. CTCAE criteria are more consist, but the occurrence of toxicity is not always related to clinical significance [68]. The following recent experts’ recommendations should be considered; Studies should separately record the incidence of classic and non-classic RILD, CP class should be recorded as well as any change in status of CP class after treatment, and the use of CTCAE criteria is advisable to promote consistency of reporting [69].
New approaches
Because the GI organ is located near the liver, it is one of the important dose-limiting organs when RT is used to treat HCC. The classic method to minimize GI toxicity is to reduce target volume and decrease total dose using cone-down technique [59]. A more complex method is to use simultaneous integrated boost IMRT (SIB-IMRT), in which different doses are delivered to different targets at the same time. One study reported promising results of SIB-IMRT in HCC [70]. The following two dose-fractionation schemes were applied depending on the proximity of GI organ: (1) 41 patients in the low dose-fractionation group, with an internal target volume (ITV) < 1 cm from the GI organ who received 55 and 44 Gy in 22 fractions to planning target volume 1 (PTV1) and 2 (PTV2), and 12 patients in the high dose-fractionation (HD) group, with ITV ≥1 cm from the GI organ who received 66 and 55 Gy in 22 fractions to PTV1 and PTV2. There was no report of toxicity ≥ grade 3. Overall LCR and OS rates at 2 years were 67.3% and 54.7%, respectively. The HD group tended to show better objective RR (100% vs. 62%, P = 0.039), LCR at 2 years (85.7% vs. 59%, P = 0.119), and OS rate at 2 years (83.3% vs. 44.3%, P = 0.037). The authors suggested that the advantages of SIB-IMRT have been sustained in HCC although there is a risk of dose uncertainty resulting from liver motion during respiration.
Although surgical resection or transplantation are considered the standard treatments for HCC, less than 20% of patients are suitable for surgery, and recurrence rates can be as high as 25% per year after curative-intent surgery [71, 72]. To improve surgical outcome, several randomized trials investigated the role adjuvant treatment including systemic chemotherapy, HAIC, or TACE, but no adjuvant treatment has shown a therapeutic benefit, in terms of recurrence or OS after resection [73]. Recent technical advances in perioperative care have extended the surgical indications for HCC and patients with HCCs, that are adherent to the major vascular structures, are able to receive surgical resection. Recently, one study investigated the role of adjuvant IMRT in patients with HCCs close to major vessels [74]. In total, 181 patients were enrolled: 33 patients with narrow-margin (< 1 cm) after hepatectomy who received adjuvant s-IMRT (Group A), 83 patients with narrow-margin (< 1 cm) but who did not receive adjuvant RT (Group B), and 65 patients with wide-margin (≥ 1 cm) and who also did not receive adjuvant RT (Group C). Groups A and C showed significantly fewer intrahepatic marginal recurrences (P = 0.048) and diffuse recurrences (P = 0.018), and extrahepatic metastases (P = 0.038) than group B. The 3-year OS and disease-free survival rates were 89.1% and 64.2% in group A, 67.7% and 52.2% in group B, and 86.0% and 60.1% in group C. Based on this favorable outcome, the authors have embarked on an ongoing prospective phase II study with a larger patient cohort.
For locally advanced HCC, sorafenib, the multi-targeted tyrosine kinase inhibitor is recommended as the first-line treatment option from two phase III randomized trials [75–77]. Theoretically, a better treatment outcome is expected with a combination of IMRT and sorafenib for advanced HCC. One phase II study evaluated the efficacy and safety of IMRT with concurrent and sequential use of sorafenib for unresectable HCC with or without PVTT [78]. Total RT dose ranged from 40 Gy to 60 Gy (median: 50 Gy). Sorafenib was administered from the commencement of RT at a dose of 400 mg twice daily, and continued at the same dosage. They reported a promising 2-year OS rate of 32%, but patients experienced higher hepatic toxicity ≥ grade 3 than the above-mentioned studies. During IMRT, four patients (10%) experienced hepatic toxicity of grade 3. After IMRT, six patients (15%) developed hepatic toxicity ≥ grade 3, and three of them were fatal. They suggested that this combination should be used with caution. A phase I study of SBRT combined with sorafenib also reported high hepatic toxicity, including worsening of CP class in six patients (50%) with large-sized HCC [79]. Based on these studies, other sequences involving combination treatments with IMRT and sorafenib should be evaluated with the aim of reducing toxicity. There are inconsistent outcomes with pre-RT and post-RT use of sorafenib in preclinical studies. Li et al. [80] reported that sorafenib given 30 min before RT reduced the anti-proliferative effects of irradiation against HCC, whereas sorafenib given 24 hour after RT increased the anti-tumor effects against HCC in vitro. Yu et al. [81] found that there was inconsistent observation with pre-RT and post-RT use of sorafenib in two different cell lines. However, Chen et al. [82] suggested that pre-RT use of sorafenib could provide a better tumor growth inhibition than concurrent or post-RT use of sorafenib. Further studies are necessary to clarify the optimal sequence for use of this combination treatment.
CONCLUSIONS
As the most advanced current radiotherapy technology, IMRT provides a precise and conformal dose distribution by using multiple beams with non-uniform intensity profiles. IMRT is divided into s-IMRT, VMAT, and h-IMRT according to beam delivery methods. Generally, IMRT techniques show better conformity to target volume coverage than 3DCRT for the treatment of HCC. Among IMRT techniques, VMAT and h-IMRT achieve a more homogenous dose distribution within a tumor than s-IMRT. Although the high dose region of the normal liver is smaller in IMRT than 3DCRT, the low dose region is increased in IMRT, and this increase is remarkable in h-IMRT or VMAT. Until present times, it has been unclear whether the increased low dose regions with IMRT techniques could be a risk factor for developing RILD. Additional studies are needed to confirm the safety of IMRT for treating HCC. In contrast, the sparing effect of non-liver OARs is beneficial in h-IMRT or VMAT.
Because of the differences in dosimetric advantages based on IMRT techniques, the choice of optimal IMRT technique should be personalized to the location of HCC and OARs. In HCCs < 6–8 cm, IMRT would be considered to spare the liver. In large HCCs or advanced HCC with PVTT, IMRT would be considered to spare non-liver OARs, especially in reducing high dose regions of gastroduodenum. In the clinical setting, a few studies have demonstrated the therapeutic benefit of IMRT for HCC in comparison to 3DCRT. Some studies utilizing IMRT reported promising treatment results. A new approach using SIB-IMRT provides another means of reducing GI toxicity. Adjuvant IMRT after surgical resection could reduce recurrence and increase survival in selected patients. A combination of sorafenib and IMRT resulted in a promising OS. However, many studies had small sample sizes, were retrospective in design, and had relatively short-term follow up periods. To validate the treatment outcomes of IMRT and obtain definitive conclusions, additional prospective studies in larger study populations will be necessary in the future.
Author contributions
SHB and WIJ have contributed equally to this work. They searched and analyzed the data and drafted the manuscript. HCP designed and supervised the review. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
This research was supported by the Marine Biotechnology Program funded by Ministry of Oceans and Fisheries, Korea (20150220). This work was supported by the Soonchunhyang University Research Fund.
CONFLICT OF INTEREST
The authors have no conflicts of interest to disclose.
REFERENCES
1. Borgelt BB, Gelber R, Brady LW, Griffin T, Hendrickson FR. The palliation of hepatic metastases: results of the Radiation Therapy Oncology Group pilot study. Int J Radiat Oncol Biol Phys. 1981; 7:587–591.
2. Emami B, Lyman J, Brown A, Coia L, Goitein M, Munzenrider JE, Shank B, Solin LJ, Wesson M. Tolerance of normal tissue to therapeutic irradiation. Int J Radiat Oncol Biol Phys. 1991; 21:109–122.
3. Russell AH, Clyde C, Wasserman TH, Turner SS, Rotman M. Accelerated hyperfractionated hepatic irradiation in the management of patients with liver metastases: results of the RTOG dose escalating protocol. Int J Radiat Oncol Biol Phys. 1993; 27:117–123.
4. Ohri N, Dawson LA, Krishnan S, Seong J, Cheng JC, Sarin SK, Kinkhabwala M, Ahmed MM, Vikram B, Coleman CN, Guha C. Radiotherapy for Hepatocellular Carcinoma: New Indications and Directions for Future Study. J Natl Cancer Inst. 2016; 108.
5. Seol SW, Yu JI, Park HC, Lim DH, Oh D, Noh JM, Cho WK, Paik SW. Treatment outcome of hepatic re-irradiation in patients with hepatocellular carcinoma. Radiat Oncol J. 2015; 33:276–283.
6. Tanguturi SK, Wo JY, Zhu AX, Dawson LA, Hong TS. Radiation therapy for liver tumors: ready for inclusion in guidelines? Oncologist. 2014; 19:868–879.
7. Rim CH, Seong J. Application of radiotherapy for hepatocellular carcinoma in current clinical practice guidelines. Radiat Oncol J. 2016; 34:160–167.
8. Keane FK, Wo JY, Zhu AX, Hong TS. Liver-Directed Radiotherapy for Hepatocellular Carcinoma. Liver Cancer. 2016; 5:198–209.
9. Kang JK, Kim MS, Jang WI, Seo YS, Kim HJ, Cho CK, Yoo HJ, Paik EK, Cha YJ, Song HJ. The clinical utilization of radiation therapy in Korea between 2009 and 2013. Radiat Oncol J. 2016; 34:88–95.
10. Yu SJ. A concise review of updated guidelines regarding the management of hepatocellular carcinoma around the world: 2010–2016. Clin Mol Hepatol. 2016; 22:7–17.
11. Korean Liver Cancer Study Group (KLCSG), National Cancer Center Korea (NCC). 2014 KLCSG-NCC Korea Practice Guideline for the Management of Hepatocellular Carcinoma. Gut Liver. 2015; 9:267–317.
12. Kokudo N, Hasegawa K, Akahane M, Igaki H, Izumi N, Ichida T, Uemoto S, Kaneko S, Kawasaki S, Ku Y, Kudo M, Kubo S, Takayama T, et al. Evidence-based Clinical Practice Guidelines for Hepatocellular Carcinoma: The Japan Society of Hepatology 2013 update (3rd JSH-HCC Guidelines). Hepatol Res. 2015; 45.
13. Omata M, Lesmana LA, Tateishi R, Chen PJ, Lin SM, Yoshida H, Kudo M, Lee JM, Choi BI, Poon RT, Shiina S, Cheng AL, Jia JD, et al. Asian Pacific Association for the Study of the Liver consensus recommendations on hepatocellular carcinoma. Hepatol Int. 2010; 4:439–474.
14. European Association For The Study Of The Liver; European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2012; 56:908–943.
15. Verslype C, Rosmorduc O, Rougier P, ESMO Guidelines Working Group. Hepatocellular carcinoma: ESMO-ESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012; 23:vii41–48.
16. Bruix J, Sherman M, American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology. 2011; 53:1020–1022.
17. Network. NCC. NCCN Guidelines. Hepatobilliary Cancers, version 2, 2016. Available at: http://wwwnccnorg/professionals/physician_gls/f_guidelinesasp.
18. Galvin JM, De Neve W. Intensity modulating and other radiation therapy devices for dose painting. J Clin Oncol. 2007; 25:924–930.
19. Leibel SA, Fuks Z, Zelefsky MJ, Wolden SL, Rosenzweig KE, Alektiar KM, Hunt MA, Yorke ED, Hong LX, Amols HI, Burman CM, Jackson A, Mageras GS, et al. Intensity-modulated radiotherapy. Cancer J. 2002; 8:164–176.
20. Gutiontov SI, Shin EJ, Lok B, Lee NY, Cabanillas R. Intensity-modulated radiotherapy for head and neck surgeons. Head Neck. 2016; 38 Suppl 1:E2368–2373.
21. Nakamura K, Sasaki T, Ohga S, Yoshitake T, Terashima K, Asai K, Matsumoto K, Shioyama Y, Honda H. Recent advances in radiation oncology: intensity-modulated radiotherapy, a clinical perspective. Int J Clin Oncol. 2014; 19:564–569.
22. Fuss M, Salter BJ, Herman TS, Thomas CR Jr. External beam radiation therapy for hepatocellular carcinoma: potential of intensity-modulated and image-guided radiation therapy. Gastroenterology. 2004; 127:S206–217.
23. Hu Y, Zhou YK, Chen YX, Shi SM, Zeng ZC. 4D-CT scans reveal reduced magnitude of respiratory liver motion achieved by different abdominal compression plate positions in patients with intrahepatic tumors undergoing helical tomotherapy. Med Phys. 2016; 43:4335.
24. Hu Y, Zhou YK, Chen YX, Shi SM, Zeng ZC. Clinical benefits of new immobilization system for hypofractionated radiotherapy of intrahepatic hepatocellular carcinoma by helical tomotherapy. Med Dosim. 2017; 42:37–41.
25. Giraud P, Yorke E, Jiang S, Simon L, Rosenzweig K, Mageras G. Reduction of organ motion effects in IMRT and conformal 3D radiation delivery by using gating and tracking techniques. Cancer Radiother. 2006; 10:269–282.
26. Yu J, Choi JH, Ma SY, Jeung TS, Lim S. Comparison between audio-only and audiovisual biofeedback for regulating patients’ respiration during four-dimensional radiotherapy. Radiat Oncol J. 2015; 33:250–255.
27. Shin E, Han Y, Park HC, Sung Kim J, Hwan Ahn S, Suk Shin J, Gyu Ju S, Ho Choi D, Lee J. Cumulative dose on fractional delivery of tomotherapy to periodically moving organ: a phantom QA suggestion. Med Dosim. 2013; 38:359–365.
28. Lee IJ, Seong J, Koom WS, Kim YB, Jeon BC, Kim JH, Han KH. Selection of the optimal radiotherapy technique for locally advanced hepatocellular carcinoma. Jpn J Clin Oncol. 2011; 41:882–889.
29. Chen D, Wang R, Meng X, Liu T, Yan H, Feng R, Liu S, Jiang S, Xu X, Zhu K, Dou X. A comparison of liver protection among 3-D conformal radiotherapy, intensity-modulated radiotherapy and RapidArc for hepatocellular carcinoma. Radiat Oncol. 2014; 9:48.
30. Hsieh CH, Liu CY, Shueng PW, Chong NS, Chen CJ, Chen MJ, Lin CC, Wang TE, Lin SC, Tai HC, Tien HJ, Chen KH, Wang LY, et al. Comparison of coplanar and noncoplanar intensity-modulated radiation therapy and helical tomotherapy for hepatocellular carcinoma. Radiat Oncol. 2010; 5:40.
31. Park JM, Kim K, Chie EK, Choi CH, Ye SJ, Ha SW. RapidArc vs intensity-modulated radiation therapy for hepatocellular carcinoma: a comparative planning study. Br J Radiol. 2012; 85:e323–329.
32. Song JH, Son SH, Kay CS, Jang HS. Reducing the probability of radiation-induced hepatic toxicity by changing the treatment modality from helical tomotherapy to fixed-beam intensity-modulated radiotherapy. Oncotarget. 2015; 6:33952–33960. http://doi.org/10.18632/oncotarget.5581.
33. Toramatsu C, Katoh N, Shimizu S, Nihongi H, Matsuura T, Takao S, Miyamoto N, Suzuki R, Sutherland K, Kinoshita R, Onimaru R, Ishikawa M, Umegaki K, et al. What is the appropriate size criterion for proton radiotherapy for hepatocellular carcinoma? A dosimetric comparison of spot-scanning proton therapy versus intensity-modulated radiation therapy. Radiat Oncol. 2013; 8:48.
34. Kuo YC, Chiu YM, Shih WP, Yu HW, Chen CW, Wong PF, Lin WC, Hwang JJ. Volumetric intensity-modulated Arc (RapidArc) therapy for primary hepatocellular carcinoma: comparison with intensity-modulated radiotherapy and 3-D conformal radiotherapy. Radiat Oncol. 2011; 6:76.
35. Son SH, Kay CS, Song JH, Lee SW, Choi BO, Kang YN, Jang JW, Yoon SK, Jang HS. Dosimetric parameter predicting the deterioration of hepatic function after helical tomotherapy in patients with unresectable locally advanced hepatocellular carcinoma. Radiat Oncol. 2013; 8:11.
36. Tsai YC, Tsai CL, Hsu FM, Jian-Kuen W, Chien-Jang W, Cheng JC. Superior liver sparing by combined coplanar/noncoplanar volumetric-modulated arc therapy for hepatocellular carcinoma: a planning and feasibility study. Med Dosim. 2013; 38:366–371.
37. Withers HR, Taylor JM, Maciejewski B. Treatment volume and tissue tolerance. Int J Radiat Oncol Biol Phys. 1988; 14:751–759.
38. Jackson A, Ten Haken RK, Robertson JM, Kessler ML, Kutcher GJ, Lawrence TS. Analysis of clinical complication data for radiation hepatitis using a parallel architecture model. Int J Radiat Oncol Biol Phys. 1995; 31:883–891.
39. Dawson LA, Ten Haken RK. Partial volume tolerance of the liver to radiation. Semin Radiat Oncol. 2005; 15:279–283.
40. Gallicchio R, Nardelli A, Mainenti P, Nappi A, Capacchione D, Simeon V, Sirignano C, Abbruzzi F, Barbato F, Landriscina M, Storto G. Therapeutic Strategies in HCC: Radiation Modalities. Biomed Res Int. 2016; 2016:1295329.
41. Song CH, Pyo H, Moon SH, Kim TH, Kim DW, Cho KH. Treatment-related pneumonitis and acute esophagitis in non-small-cell lung cancer patients treated with chemotherapy and helical tomotherapy. Int J Radiat Oncol Biol Phys. 2010; 78:651–658.
42. Khalil AA, Hoffmann L, Moeller DS, Farr KP, Knap MM. New dose constraint reduces radiation-induced fatal pneumonitis in locally advanced non-small cell lung cancer patients treated with intensity-modulated radiotherapy. Acta Oncol. 2015; 54:1343–1349.
43. Chen LS, Lin HC, Hwang SJ, Lee FY, Hou MC, Lee SD. Prevalence of gastric ulcer in cirrhotic patients and its relation to portal hypertension. J Gastroenterol Hepatol. 1996; 11:59–64.
44. Akdamar K, Ertan A, Agrawal NM, McMahon FG, Ryan J. Upper gastrointestinal endoscopy in normal asymptomatic volunteers. Gastrointest Endosc. 1986; 32:78–80.
45. Kim H, Lim DH, Paik SW, Yoo BC, Koh KG, Lee JH, Choi MS, Park W, Park HC, Huh SJ, Choi DH, Ahn YC. Predictive factors of gastroduodenal toxicity in cirrhotic patients after three-dimensional conformal radiotherapy for hepatocellular carcinoma. Radiother Oncol. 2009; 93:302–306.
46. Chon YE, Seong J, Kim BK, Cha J, Kim SU, Park JY, Ahn SH, Han KH, Chon CY, Shin SK, Kim DY. Gastroduodenal complications after concurrent chemoradiation therapy in patients with hepatocellular carcinoma: endoscopic findings and risk factors. Int J Radiat Oncol Biol Phys. 2011; 81:1343–1351.
47. Yoon H, Oh D, Park HC, Kang SW, Han Y, Lim DH, Paik SW. Predictive factors for gastroduodenal toxicity based on endoscopy following radiotherapy in patients with hepatocellular carcinoma. Strahlenther Onkol. 2013; 189:541–546.
48. Bae BK, Kim JC. The response of thrombosis in the portal vein or hepatic vein in hepatocellular carcinoma to radiation therapy. Radiat Oncol J. 2016; 34:168–176.
49. Yoon HI, Lee IJ, Han KH, Seong J. Improved oncologic outcomes with image-guided intensity-modulated radiation therapy using helical tomotherapy in locally advanced hepatocellular carcinoma. J Cancer Res Clin Oncol. 2014; 140:1595–1605.
50. Hou JZ, Zeng ZC, Wang BL, Yang P, Zhang JY, Mo HF. High dose radiotherapy with image-guided hypo-IMRT for hepatocellular carcinoma with portal vein and/or inferior vena cava tumor thrombi is more feasible and efficacious than conventional 3D-CRT. Jpn J Clin Oncol. 2016; 46:357–362.
51. Kang MK, Kim MS, Kim SK, Ye GW, Lee HJ, Kim TN, Eun JR. High-dose radiotherapy with intensity-modulated radiation therapy for advanced hepatocellular carcinoma. Tumori. 2011; 97:724–731.
52. Zhang T, Zhao YT, Wang Z, Li CR, Jin J, Jia AY, Wang SL, Song YW, Liu YP, Ren H, Fang H, Bao H, Liu XF, et al. Efficacy and Safety of Intensity-Modulated Radiotherapy Following Transarterial Chemoembolization in Patients With Unresectable Hepatocellular Carcinoma. Medicine (Baltimore). 2016; 95:e3789.
53. McIntosh A, Hagspiel KD, Al-Osaimi AM, Northup P, Caldwell S, Berg C, Angle JF, Argo C, Weiss G, Rich TA. Accelerated treatment using intensity-modulated radiation therapy plus concurrent capecitabine for unresectable hepatocellular carcinoma. Cancer. 2009; 115:5117–5125.
54. Chi KH, Liao CS, Chang CC, Ko HL, Tsang YW, Yang KC, Mehta MP. Angiogenic blockade and radiotherapy in hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2010; 78:188–193.
55. Kong M, Hong SE, Choi WS, Choi J, Kim Y. Treatment outcomes of helical intensity-modulated radiotherapy for unresectable hepatocellular carcinoma. Gut Liver. 2013; 7:343–351.
56. Kim JY, Yoo EJ, Jang JW, Kwon JH, Kim KJ, Kay CS. Hypofractionated radiotheapy using helical tomotherapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis. Radiat Oncol. 2013; 8:15.
57. Huang CM, Huang MY, Tang JY, Chen SC, Wang LY, Lin ZY, Huang CJ. Feasibility and efficacy of helical tomotherapy in cirrhotic patients with unresectable hepatocellular carcinoma. World J Surg Oncol. 2015; 13:201.
58. Son SH, Park EY, Park HH, Kay CS, Jang HS. Pre-radiotherapy neutrophil-to-lymphocyte ratio as an independent prognostic factor in patients with locally advanced hepatocellular carcinoma treated with radiotherapy. Oncotarget. 2017; 8:16964–16971. http://doi.org/10.18632/oncotarget.15209.
59. Wang PM, Hsu WC, Chung NN, Chang FL, Fogliata A, Cozzi L. Radiotherapy with volumetric modulated arc therapy for hepatocellular carcinoma patients ineligible for surgery or ablative treatments. Strahlenther Onkol. 2013; 189:301–307.
60. Tandon P, Garcia-Tsao G. Prognostic indicators in hepatocellular carcinoma: a systematic review of 72 studies. Liver Int. 2009; 29:502–510.
61. Chan SL, Chong CC, Chan AW, Poon DM, Chok KS. Management of hepatocellular carcinoma with portal vein tumor thrombosis: Review and update at 2016. World J Gastroenterol. 2016; 22:7289–7300.
62. Cheng S, Yang J, Shen F, Zhou W, Wang Y, Cong W, Yang GS, Cheng H, Hu H, Gao C, Guo J, Li A, Meng Y, et al. Multidisciplinary management of hepatocellular carcinoma with portal vein tumor thrombus - Eastern Hepatobiliary Surgical Hospital consensus statement. Oncotarget. 2016; 7:40816–40829. http://doi.org/10.18632/oncotarget.8386.
63. Shannon AM, Williams KJ. Antiangiogenics and radiotherapy. J Pharm Pharmacol. 2008; 60:1029–1036.
64. Walraven M, Witteveen PO, Lolkema MP, van Hillegersberg R, Voest EE, Verheul HM. Antiangiogenic tyrosine kinase inhibition related gastrointestinal perforations: a case report and literature review. Angiogenesis. 2011; 14:135–141.
65. Barney BM, Markovic SN, Laack NN, Miller RC, Sarkaria JN, Macdonald OK, Bauer HJ, Olivier KR. Increased bowel toxicity in patients treated with a vascular endothelial growth factor inhibitor (VEGFI) after stereotactic body radiation therapy (SBRT). Int J Radiat Oncol Biol Phys. 2013; 87:73–80.
66. Guha C, Kavanagh BD. Hepatic radiation toxicity: avoidance and amelioration. Semin Radiat Oncol. 2011; 21:256–263.
67. Cholongitas E, Papatheodoridis GV, Vangeli M, Terreni N, Patch D, Burroughs AK. Systematic review: The model for end-stage liver disease--should it replace Child-Pugh’s classification for assessing prognosis in cirrhosis? Aliment Pharmacol Ther. 2005; 22:1079–1089.
68. Son SH, Choi BO, Ryu MR, Kang YN, Jang JS, Bae SH, Yoon SK, Choi IB, Kang KM, Jang HS. Stereotactic body radiotherapy for patients with unresectable primary hepatocellular carcinoma: dose-volumetric parameters predicting the hepatic complication. Int J Radiat Oncol Biol Phys. 2010; 78:1073–1080.
69. Pan CC, Kavanagh BD, Dawson LA, Li XA, Das SK, Miften M, Ten Haken RK. Radiation-associated liver injury. Int J Radiat Oncol Biol Phys. 2010; 76:S94–100.
70. Kim TH, Park JW, Kim YJ, Kim BH, Woo SM, Moon SH, Kim SS, Lee WJ, Kim DY, Kim CM. Simultaneous integrated boost-intensity modulated radiation therapy for inoperable hepatocellular carcinoma. Strahlenther Onkol. 2014; 190:882–890.
71. Schwarz RE, Smith DD. Trends in local therapy for hepatocellular carcinoma and survival outcomes in the US population. Am J Surg. 2008; 195:829–836.
72. Lai EC, Fan ST, Lo CM, Chu KM, Liu CL, Wong J. Hepatic resection for hepatocellular carcinoma. An audit of 343 patients. Ann Surg. 1995; 221:291–298.
73. Schwartz JD, Schwartz M, Mandeli J, Sung M. Neoadjuvant and adjuvant therapy for resectable hepatocellular carcinoma: review of the randomised clinical trials. Lancet Oncol. 2002; 3:593–603.
74. Wang WH, Wang Z, Wu JX, Zhang T, Rong WQ, Wang LM, Jin J, Wang SL, Song YW, Liu YP, Ren H, Fang H, Wang WQ, et al. Survival benefit with IMRT following narrow-margin hepatectomy in patients with hepatocellular carcinoma close to major vessels. Liver Int. 2015; 35:2603–2610.
75. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008; 359:378–390.
76. Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009; 10:25–34.
77. Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M, Talwalkar J, Gores GJ, Panel of Experts in HCCDCT. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008; 100:698–711.
78. Chen SW, Lin LC, Kuo YC, Liang JA, Kuo CC, Chiou JF. Phase 2 study of combined sorafenib and radiation therapy in patients with advanced hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2014; 88:1041–1047.
79. Brade AM, Ng S, Brierley J, Kim J, Dinniwell R, Ringash J, Wong RR, Cho C, Knox J, Dawson LA. Phase 1 Trial of Sorafenib and Stereotactic Body Radiation Therapy for Hepatocellular Carcinoma. Int J Radiat Oncol Biol Phys. 2016; 94:580–587.
80. Li Q, Hu Y, Xi M, He L, Zhao L, Liu M. Sorafenib modulates the radio sensitivity of hepatocellular carcinoma cells in vitro in a schedule-dependent manner. BMC Cancer. 2012; 12:485.
81. Yu W, Gu K, Yu Z, Yuan D, He M, Ma N, Lai S, Zhao J, Ren Z, Zhang X, Shao C, Jiang GL. Sorafenib potentiates irradiation effect in hepatocellular carcinoma in vitro and in vivo. Cancer Lett. 2013; 329:109–117.
82. Chen JC, Chuang HY, Hsu FT, Chen YC, Chien YC, Hwang JJ. Sorafenib pretreatment enhances radiotherapy through targeting MEK/ERK/NF-kappaB pathway in human hepatocellular carcinoma-bearing mouse model. Oncotarget. 2016; 7:85450–85463. http://doi.org/10.18632/oncotarget.13398.

