INTRODUCTION
Nasopharyngeal carcinoma (NPC) is primarily found in the nasopharynx. As one of the world regions with highest incidence rates for this disease, south China has a standardized incidence rate of 20–30/100 000 [1, 2]. Non-keratinizing carcinoma (WHO 2005 classification), especially the undifferentiated type of Non-keratinizing carcinoma [3], is the major pathological type in regions with high incidence, and has better sensitivity to chemoradiotherapy. Oncogenes and tumor suppressor genes are involved in tumorigenesis and development, while DNA repair genes are involved in drug resistance [4–6]. Expression level of these protein should be considered to predict treatment responses and prognoses in NPC patients besides pathological type, clinical staging, and chemoradiotherapy. Therefore, understanding the underlying intrinsic factors would help predict the prognosis and select optimal treatments.
The Ki67 protein is closely related to mitosis and reflects the proliferation activity of malignant tumors [7], while indicating the development and prognosis of various malignant tumors such as breast, lung, urinary tract, and cervical cancers [8–11]. As an independent prognostic indicator, it is conventionally applied in the molecular classification and treatment strategies of breast cancer [12]. Regarding head and neck cancer, high expression of Ki67 affects the treatment response of oral squamous cell carcinoma [13] with high risk of recurrence of early tongue cancer [14]. However, the significance of Ki67 expression in NPC is unclear, and conclusions from few small sample studies are different [15, 16].
ERCC1 is involved in the DNA nucleotide excision repair process; it identifies and resects the damaged DNA strand. High ERCCl expression is associated with resistance to platinum-based chemotherapy [17–19]; patients with low ERCC1 expression are more likely to obtain survival benefit [18, 19]. ERCC1 expression predicts efficacy in patients with non-small cell lung cancer or head and neck squamous cell carcinoma, who have received platinum-based concurrent chemoradiotherapy; meanwhile, patients with low ERCC1 expression often show better treatment response and survival [20, 21]. Meta-analysis has shown that ERCC1 expression is associated with the efficacy of platinum-based chemoradiotherapy for head and neck squamous cell carcinoma as well as prognosis of the Asian population; meanwhile, Asians with low ERCC1 expression show better OS and PFS [22–24]. Moreover, ERCC1 level shows a correlation with objective response rate (ORR) and OS in NPC [22]. However, NPC data are from a few small sample studies, and the association of ERCC1 expression with treatment response and prognosis in NPC needs to be confirmed by additional large sample studies.
In addition, whether combined Ki67 and ERCC1 can better assess prognosis and provide clinical guidance remains unclear. This study aimed to retrospectively assess the prognostic value of combined Ki67 and ERCC1 levels and survival in the major pathologic type (non-keratinizing carcinoma) in a high-incidence area of NPC.
RESULTS
Cases and follow-up status
A total of 334 cases with complete data were assessed. Medium age was 48 years (17–72 years); males accounted for 73.1%, and females for 26.9%. The differentiated and undifferentiated types of Non-keratinizing carcinoma accounted for 5.1% and 94.9%, respectively. I-II stage disease accounted for 12.6%, while III-IVb was found in 87.4% of cases. Radiotherapy was performed by IMRT. A total of 3.6%, 13.5%, 39.5%, and 43.4% of patients received radiotherapy alone, platinum-based concurrent radiochemotherapy, platinum-based induction chemotherapy and concurrent radiochemotherapy, and platinum-based concurrent radiochemotherapy and adjuvant combination chemotherapy, respectively (Table 1).
Table 1: Patient characteristics
Characteristics |
No. of patients (range) |
% |
|---|---|---|
Total number of patients |
334 |
100 |
Age (Years) |
48 (17–72) |
|
Gender |
||
Males |
244 |
73.1 |
Females |
90 |
26.9 |
pathological subtype of Non-keratinizing carcinoma (2005 WHO)* |
||
Differentiated type |
17 |
5.1 |
Undifferentiated type |
317 |
94.9 |
Clinical stage (AJCC 7th, 2010)# |
||
I |
3 |
0.9 |
II |
39 |
11.7 |
III |
160 |
47.9 |
IV a/b |
132 |
39.5 |
Primary tumor (T) stage |
||
T1 |
27 |
8.1 |
T2 |
55 |
16.5 |
T3 |
141 |
42.2 |
T4 |
111 |
33.2 |
Regional lymph nodes (N) stage |
||
N0 |
20 |
6.0 |
N1 |
116 |
34.7 |
N2 |
161 |
48.2 |
N3 |
37 |
11.1 |
Treatment received by patients |
||
Radiotherapy alone |
12 |
3.6 |
Concurrent chemoradiation |
45 |
13.5 |
Induction chemotherapy + Concurrent chemoradiation |
132 |
39.5 |
Concurrent chemoradiation + Adjuvant chemotherapy |
145 |
43.4 |
*According to the 2005 WHO histological classification and pathologic stage [3]
#According to the 2010 International Union against Cancer/American Joint Committee on Cancer (UICC/ AJCC) staging system [25] for NPC.
Medium follow-up was 48.7 months (5–72 months) and ended in December 2016. The 1-, 2-, 3-, and 4-year OS rates were 99.4%, 96.7%, 92.8%, and 91.3%, respectively; DFS rates were 94.3%, 82.3%, 79.3%, and 76.0%, respectively; DMFS rates were 95.5%, 87.7%, 84.4%, and 82.0%, respectively; LRFS rates were 98.5%, 94.6%, 93.1%, and 91.9%, respectively. The disease had progressed in 80 cases, including 50 cases with distant metastasis as the initial event, 17 with local recurrence (7 in primary lesion, 9 in regional lymph nodes, and 3 in both primary lesion and regional lymph nodes), 10 with distant metastasis combined with local recurrence, and 3 with a second primary tumor (1 each of tongue cancer, gastric cancer, and nasal sarcoma). There were 29 cases of death.
Expression levels of Ki67 and ERCC1 in nasopharyngeal carcinoma
Of the 334 patients, low-medium Ki67 expression accounted for 64.4% (low and medium levels represented 27.5% and 36.9%, respectively), while high expression was found in 35.6%. Low expression of ERCC1 was found in 64.7% of patients, and high in 35.3%.
The expression of Ki67 was not significantly correlated with gender (χ2 = 1.096, P = 0.179), pathological subtype of Non-keratinizing carcinoma (χ2 = 0.302, P = 0.395), and T staging (χ2 = 7.496, P = 0.058), but significantly associated with clinical staging (χ2 = 12.714, P = 0.005) and N staging (χ2 = 28.004, P =< 0.001). ERCC1 expression was not significantly associated with gender (χ2 = 1.588, P = 0.128), pathological subtype of Non-keratinizing carcinoma (χ2 = 0.910, P = 0.475), and N staging (χ2 = 0.205, P = 0.977), but significantly associated with clinical staging (χ2 = 14.57, P = 0.002) and T staging (χ2 = 15.47, P = 0.002) (Table 2).
Table 2: Associations of Ki67 and ERCC1 levels in nasopharyngeal carcinoma with clinical characteristics
Characteristics |
Ki67 n = 334 |
χ2value |
P |
ERCC1 n = 334 |
χ2value |
P |
||
|---|---|---|---|---|---|---|---|---|
Low-medium expression (%) |
High expression (%) |
Low expression (%) |
High expression (%) |
|||||
Total cases |
215 |
119 |
216 |
118 |
||||
Gender |
||||||||
Male |
153 (71.2%) |
91 (76.5%) |
1.096 |
0.179 |
147 (70.7%) |
97 (77.0%) |
1.588 |
0.128 |
Female |
62 (28.8%) |
28 (23.5 %) |
61 (29.3%) |
29 (23.0%) |
||||
Non-keratinizing carcinoma |
||||||||
Differentiated type |
12 (5.6%) |
5 (4.2%) |
0.302 |
0.395 |
10 (4.8%) |
7 (5.6%) |
0.910 |
0.475 |
Undifferentiated type |
203 (94.4%) |
114 (95.8%) |
198 (95.2%) |
119 (94.4%) |
||||
Clinical staging |
||||||||
Stage I |
3 (1.4%) |
0 (0.0%) |
12.714 |
0.005 |
2 (1.0%) |
1 (0.8%) |
14.576 |
0.002 |
Stage II |
33 (15.3%) |
6 (5.0%) |
35 (16.8%) |
4 (3.2%) |
||||
Stage III |
105 (48.8%) |
55 (46.2%) |
96 (46.2%) |
64 (50.8%) |
||||
Stage IV a/b |
74 (34.4%) |
58 (48.7%) |
75 (36.1%) |
57 (45.2%) |
||||
T staging |
||||||||
T1 |
21 (9.8%) |
6 (5.0%) |
7.496 |
0.058 |
18 (8.7%) |
9 (7.1%) |
15.074 |
0.002 |
T2 |
38 (17.7%) |
17 (14.3%) |
46 (22.1%) |
9 (7.1%) |
||||
T3 |
95 (44.2%) |
46 (38.7%) |
85 (40.9%) |
56 (44.4%) |
||||
T4 |
61 (28.4%) |
50 (42.0%) |
59 (28.4%) |
52 (41.3%) |
||||
N staging |
||||||||
N0 |
15 (7.0%) |
5 (4.2%) |
28.004 |
<0.001 |
12 (5.8%) |
8 (6.3%) |
0.205 |
0.977 |
N1 |
95 (44.2%) |
21 (17.6%) |
73 (35.1%) |
43 (34.1%) |
||||
N2 |
87 (40.5%) |
74 (62.2%) |
101 (48.6%) |
60 (47.6%) |
||||
N3 |
18 (8.4%) |
19 (16.0%) |
22 (10.6%) |
15 (11.9%) |
||||
Ki67 expression and nasopharyngeal carcinoma prognosis
OS, DFS, and DMFS in patients with high Ki67 expression were significantly lower than those with low-medium expression. 4-year OS rates in the low-medium and high expression groups were 94.9% and 84.9%, respectively (P = 0.001); 4-year DFS rates were 80.0% and 68.9%, respectively (P = 0.012); 4-year DMFS rates were 86.0% and 74.8%, respectively (P = 0.007). There was no statistically significant difference in LRFS between the high and low-medium Ki67expression groups: 4-year LRFS in the low-medium and high expression groups were 92.6% and 90.8%, respectively (P = 0.547) (Figure 1). However, OS, DFS, DMFS, and LRFS were similar in the low and medium Ki67 expression groups. 4-year OS rates in the low and medium expression groups were 95.7% and 94.3%, respectively (P = 0.449); 4-year DFS rates were 84% and 77.2%, respectively (P = 0.136); 4-year DMFS rates were 89.1% and 84.6%, respectively (P = 0.266); LRFS rates were 93.5% and 92.7%, respectively (P = 0.515). High Ki67 expression was correlated with adverse prognosis of OS (HR = 3.155, 95% CI 1.489–6.683, P = 0.003), DFS (HR = 1.734, 95% CI 1.119–2.695, P = 0.014), and DMFS (HR = 1.974, 95% CI 1.190–3.274, P = 0.008) in NPC, but did not significantly increase LRFS risk (HR = 1.266, 95%CI 0.587–2.728, P = 0.547). It means that high Ki67 expression could predict adverse prognosis of OS, DFS and DMFS.
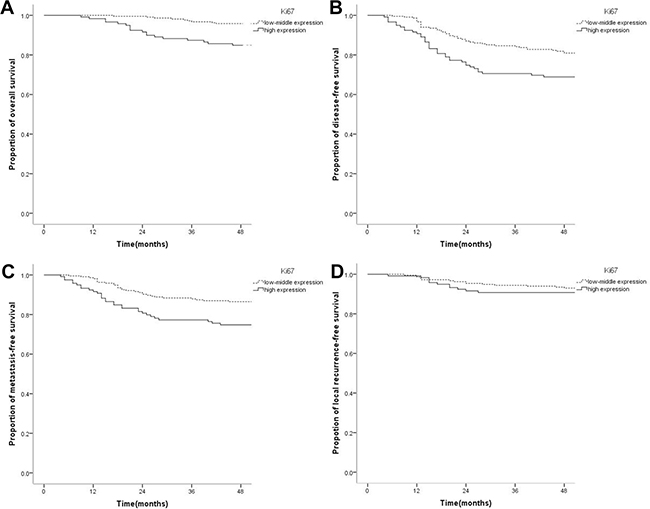
Figure 1: Kaplan-Meier survival curves of nasopharyngeal carcinoma patients with low-medium and high Ki67 levels. (A) The overall survival rate of patients with high Ki67 expression was significantly lower than that of the low-medium expression group (P = 0.001). (B) The disease-free survival rate of patients with high Ki67 level was significantly lower than that of the low-medium expression group (P = 0.012). (C) The distant metastasis-free survival rate of patients with high Ki67 expression was significantly lower than that of the low-medium expression group (P = 0.007). (D) The local recurrence-free survival rate was not significantly different between the high and low Ki67 expression groups (P = 0.547).
ERCC1 expression and nasopharyngeal carcinoma prognosis
Compared with the low expression group, OS, DFS, MDFS, and LRFS rates in the high ERCC1 expression group were decreased significantly. Specifically, 4-year OS in the low and high expression groups were 94.2% and 86.5%, respectively (P = 0.013); 4-year DFS rates were 83.2% and 64.3%, respectively (P < 0.001); 4-year DMFS rates were 86.5% and 74.6%, respectively (P = 0.005); 4-year LRFS rates were 95.2% and 86.5%, respectively (P = 0.005) (Figure 2). High ERCC1 expression was associated with adverse prognosis of, with risks of OS (HR = 2.409, 95%CI 1.147–5.058, P = 0.020), DFS (HR = 2.381, 95%CI 1.530–3.705, P < 0.001), DMFS (HR = 2.039, 95%CI 1.228–3.386, P = 0.006), and LRFS (HR = 2.909, 95%CI 1.332–6.354, P = 0.007) increased obviously.
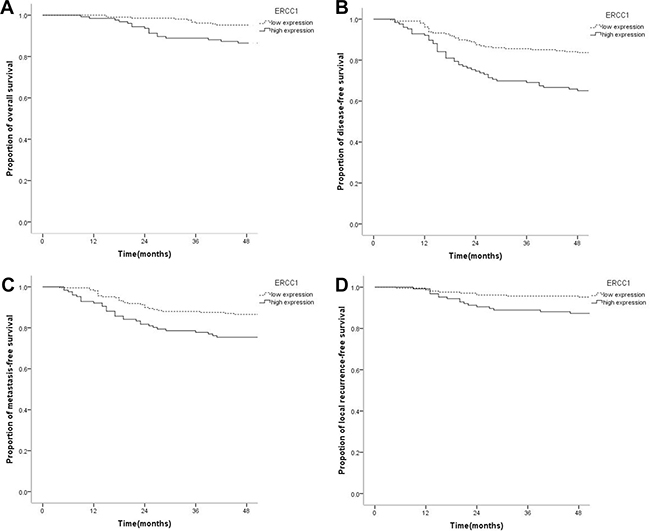
Figure 2: Kaplan-Meier survival curves of nasopharyngeal carcinoma patients with low ERCC1 level and those with high expression. (A) The overall survival rate of patients with high expression of ERCC1 was significantly lower than that of the low expression group (P = 0.013). (B) The disease-free survival rate of patients with high ERCC1 expression was significantly lower than that of the low expression group (P < 0.001). (C) The distant metastasis-free survival rate of patients with high ERCC1 amounts was significantly lower than that of the low expression group (P = 0.005). (D) The local recurrence-free survival rate of patients with high ERCC1 expression was significantly lower than that of the low expression group (P = 0.005).
Combined Ki67 and ERCC1 expression and survival of nasopharyngeal carcinoma
Of the 334 cases of NPC, the proportion with concurrent high Ki67 and ERCC1 expression was 13.5% (45/334); low-medium Ki67 expression combined with high ERCC1 expression was found in 24.0% (80/334). 21.6% (72/334) of patients had high Ki67 expression combined with low ERCC1 expression, while 41.0% (137/334) showed low-medium Ki67 and low ERCC1 amounts.
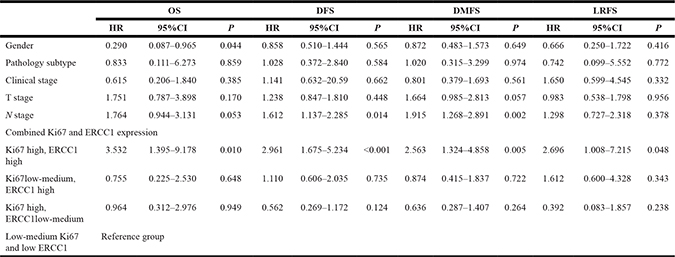
OS, DFS, DMFS, and LRFS rates in cases with concurrent high Ki67 and ERCC1 expression levels were significantly lower than those of other groups. Specifically, 4-year OS rates were 73.3%, 95.0%, 93.1% and 94.2%, respectively, in patients with concurrent high ERCC1 and Ki67 levels, low-medium Ki67 and high ERCC1 levels, high Ki67 and low ERCC1 amounts, and low-medium Ki67 and low ERCC1 amounts (P < 0.001); 4-year DFS rates were 42.2%, 77.5%, 86.1% and 81.0%, respectively (P < 0.001); 4-year DMFS rates were 55.6%, 86.3%, 87.5%, 85.4%, respectively (P < 0.001); 4-year LRFS rates were 80.0%, 90.0%, 97.2%, 94.2%, respectively (P = 0.005). There were no statistically significant differences in OS, DFS, DMFS, and LRFS rates among patients with low-medium Ki67 and high ERCC1 levels, high Ki67 and low ERCC1 amounts, and low-medium Ki67 and low ERCC1 levels (P = 0.878, P = 0.581, P = 0.416, P = 0.266) (Figure 3). Compared with the low-medium Ki67 and low ERCC1 group, patients with concurrent high Ki67 and ERCC1 levels showed significantly increased risks of OS (HR = 4.977, 95% CI 2.31–12.292, P < 0.001), DFS (HR = 4.178, 95% CI 2.421–7.212, P < 0.001), DMFS (HR = 3.722, 95% CI 2.028–7.015, P < 0.001), and LRFS (HR = 3.689, 95% CI 1.423–9.566, P = 0.007) in NPC, which were closely correlated with adverse prognosis; however, no statistically significant difference in the risk of survival prognosis was obtained in the low-medium Ki67 and high ERCC1 group, and patients with high Ki67 and low ERCC1 levels (P > 0.05) (Table 3). Multivariate analysis was performed by Cox risk proportion model, only concurrent high expression of Ki67 and ERCC1 increased the risk of every survival endpoint (P < 0.048) (Table 4). These results indicated that only concurrent high expression of Ki67 and ERCC1 was closely correlated with unfavorable prognosis.
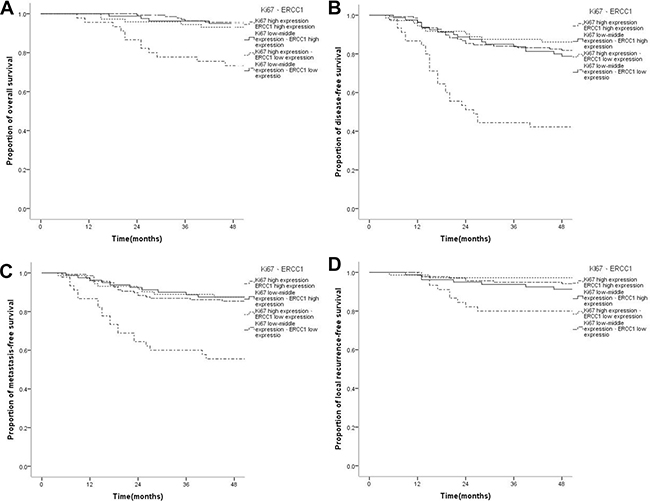
Figure 3: Kaplan-Meier survival curves of patients with nasopharyngeal carcinoma displaying different combinations of Ki67 and ERCC1 expressions. (A) The overall survival rate of patients with concurrent high ERCC1 and Ki67 levels was significantly lower than those of the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P < ss0.001); there were no statistically significant differences in overall survival rate among the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P = 0.878).(B) The disease-free survival rate of patients with concurrent high ERCC1 and Ki67 levels was significantly lower than those of the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P < 0.001); however, there were no statistically significant differences in disease-free survival among the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P = 0.581). (C) The distant metastasis-free survival rate of patients with concurrent high ERCC1 and Ki67 levels was significantly lower than those of the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P < 0.001); however, there were no statistically significant differences in the distant metastasis-free survival rate among the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P = 0.416). (D) The local recurrence-free survival rate of patients with concurrent high ERCC1 and Ki67 levels was significantly lower than those of the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P = 0.005); however, there were no statistically significant differences in local recurrence-free survival among the low-medium Ki67/high ERCC1, high Ki67/low ERCC1, and low-medium Ki67/low ERCC1 groups (P = 0.266).
Table 3: Survival risks compared with the low-medium Ki67 and low ERCC1 group
Variable |
HR |
95%CI |
P |
|
|---|---|---|---|---|
Lower |
Upper |
|||
OS |
||||
Ki67 high, ERCC1 high |
4.997 |
2.031 |
12.292 |
< 0.001 |
Ki67low-medium, ERCC1 high |
0.870 |
0.262 |
2.890 |
0.820 |
Ki67 high, ERCC1low-medium |
1.259 |
0.412 |
3.851 |
0.686 |
DFS |
||||
Ki67 high, ERCC1 high |
4.178 |
2.421 |
7.212 |
< 0.001 |
Ki67low-medium, ERCC1 high |
1.183 |
0.649 |
2.158 |
0.583 |
Ki67 high, ERCC1low-medium |
0.723 |
0.349 |
1.500 |
0.384 |
MDFS |
||||
Ki67 high, ERCC1 high |
3.722 |
2.028 |
7.015 |
< 0.001 |
Ki67low-medium, ERCC1 high |
0.933 |
0.447 |
1.946 |
0.853 |
Ki67 high, ERCC1low-medium |
0.853 |
0.388 |
1.874 |
0.692 |
LRFS |
||||
Ki67 high, ERCC1 high |
3.689 |
1.423 |
9.566 |
0.007 |
Ki67low-medium, ERCC1 high |
1.736 |
0.652 |
4.626 |
0.270 |
Ki67 high, ERCC1 low-medium |
0.473 |
0.101 |
2.229 |
0.344 |
Table 4: Multivariate analysis of the Cox risk ratio model for survival endpoints of combined Ki67 and ERCC1 expression in non-keratinizing nasopharyngeal carcinoma
DISCUSSION
The combination therapy with IMRT as the major therapy has dramatically improved the prognosis of NPC patients; metastasis is the main manifestation of failed treatment [26, 27]. In this study, 87.4% of patients had local advanced disease, and 4-year OS and LRFS rates were 91.3% and 91.9%, respectively, for a DMFS rate of 82.0%, achieving better overall survival and local control. Distant metastasis was the main cause of disease progression, consistent with previous reports [26, 27]. With inherent tumor parameters, some patients with the same clinical characteristics and treatment show different treatment responses and prognoses [26]. Therefore, understanding potential biological indicators and a more accurate prediction of NPC prognosis might contribute to individual treatment options.
It is convenient to detect Ki67 and ERCC1 expression levels in primary tumor tissues as biological prognostic indicators. In this study, patients with NPC expressed Ki67 and ERCC1 at various degrees. Ki67 expression can highly reflect the proliferative activity of malignant tumors [7]; however, its threshold for clinical significance remains unclear. The 14% threshold recommended by common sense remains controversial even in breast cancer [12]. The limited available small sample studies of NPC involving Ki67 expression have yielded discrepant conclusions due to different thresholds used. Gene et al. [15] evaluated 35 NPC cases, and found that Ki67 < 10% indicates longer survival (P = 0.03), but Ki67 expression was not significantly associated with T/N and clinical staging. Ma et al. [16] used 60% as a threshold for high Ki67 expression, and found that Ki67 expression is not significantly associated with clinical staging and survival prognosis. In this study, OS, DFS, DMFS, and LRFS rates between patients with Ki67 ≤ 25% (low expression) and those with Ki67 > 25%~50% (median expression) were not significantly different (P = 0.449, P = 0.136, P = 0.266, P = 0.515). For the high expression group (Ki67 > 50%), OS, DFS, and DMFS rates decreased significantly compared with the low-medium expression group (P = 0.001, P = 0.012, P = 0.007), with the corresponding risks of adverse prognosis increasing significantly (P = 0.003, P = 0.014, P = 0.008), corroborating the findings by Shi et al. [28], who used 50% as observation threshold. Due to sample size difference, the high expression rate of Ki67 in this study was 35.6%, which was lower than that reported by Shi et al. (54.5%, 30/55) [28]; meanwhile, Ki67 expression was significantly correlated with clinical staging (P = 0.005) and N staging (P < 0.001). Thus, selection of an appropriate threshold based on sample size is key to effectively investigate the predictive role of Ki67 in NPC; Ki67 > 50% used as a better observation threshold could reveal adverse prognosis in NPC. Observations with larger samples could help clarify the uncertainty of the association of Ki67 expression with T staging observed in this study (P = 0.058).
We also assessed ERCC1, whose high expression in locally advanced NPC was demonstrated in 14.5%-74% patients [22, 29–31]. As shown above, high expression of ERCC1 was found in 35.6% of all patients (including 12.8% of early cases). A meta-analysis [22–24] showed that high ERCC1 expression is related to adverse prognosis of OS in NPC (HR = 2.72, 95% CI 1.79–4.13, P < 0.001), but not significantly related to the clinical characteristics DFS, DMFS, and LRFS; however, sample sizes of individual studies were small, and conclusions were not consistent. Huang et al. [30] reported that 5-year DMFS of patients with high ERCC1 expression is significantly lower than that of the low expression group (51.3% and 73.5%, respectively, P = 0.037). Meanwhile, Zhang Z et al. [32] revealed that high ERCC1 expression is associated with higher local recurrence rate (29.4% vs. 12.5%, P = 0.041). However, Liang et al. [31] assessed 76 cases of NPC, and demonstrated that although ERCC1 expression is not significantly correlated with gender and N staging, it is significantly associated with T staging, clinical staging, and OS (P < 0.05), corroborating our findings. However, in this larger sample studies, OS, DFS, DMFS, and LRFS rates in patients with high ERCC1 expression were shown to decrease significantly, while the risk of adverse prognosis increases markedly, compared with those with low expression.
In this study, high expression of Ki67 or ERCC1 alone indicated adverse prognosis of NPC. However, combined analysis found no statistically significant differences in OS, DFS, DMFS, and LRFS rates as well as corresponding prognostic risks between the patients with Ki67 and ERCC1 cross-expression, and those with low-medium Ki67 expression combined with low ERCC1 levels (P > 0.05). On the one hand, tumor cells are in the proliferation period when Ki67 is highly expressed. Highly proliferative cells often are more sensitive to drugs and radiation, which results in better therapeutic response [32]. Koelbl et al. [33] reported a complete remission (CR) rate of preoperative chemoradiotherapy in OSCC patients with high Ki67 expression of 43.5%. This was significantly higher than values obtained in the low (26.1%) and medium (30.9%) expression groups. In this study, the high and low-medium Ki67 expression groups showed no statistically significant difference in LRFS of NPC (P = 0.547), and local recurrence risk did not increase significantly (HR = 1.266, 95% CI 0.587–2.728, P = 0.547). On the other hand, tumors with high ERCC1 expression have better ability of damage repair, which is associated with resistance to platinum radiotherapy. The therapeutic response of NPC is therefore worse [31, 34]. Sun et al. [34] assessed 118 cases of NPC who received radiation therapy alone, and found that the LRFS rate in patients with high ERCC1 expression is significantly lower than that of the low expression group (70% vs 86%, P = 0.046). Liang et al. [31] evaluated 76 NPC cases with concurrent chemoradiotherapy, and demonstrated that the response rate of patients with positive ERCC1 expression is significantly lower than that of the negative expression group (75% vs 97.7%, P = 0.008). Therefore, although NPC with high Ki67 and low ERCC1 levels has higher risk of cell proliferation, it also presents increased sensitivity to chemoradiotherapy, which can improve prognosis. However, in patients with low-medium Ki67 and high ERCC1 levels, although there is a certain degree of resistance to chemoradiotherapy, the risk of proliferation for the tumor itself is not high, which can yield relatively better prognosis. With both Ki67 and ERCC1 highly expressed, and the patients having both high risk of tumor proliferation and resistance to treatment, the risk of adverse prognosis may increase significantly.
In this study, 82.9% of patients received concurrent chemoradiotherapy and chemotherapy. Under sufficient treatment, compared with patients with low-medium Ki67 and low ERCC1 levels, OS, DFS, DMFS, and LRFS rates in patients with high Ki67 and low ERCC1 amounts did not decrease significantly, with the corresponding prognostic risk not statistically significant (P > 0.05). There were no statistically significant differences in survival indicators for patients with low-medium Ki67 and high ERCC1 levels after treatment (P > 0.05); indeed, concurrent high expression of Ki67 and ERCC1 was closely correlated with adverse prognosis (P < 0.007), and OS, DFS, DMFS, and LRFS rates decreased significantly (P < 0.005). Thus, combined Ki67 and ERCC1 can predict survival prognosis more effectively and guide treatment options. Systematic treatment of sufficient intensity can improve the prognosis of patients with high Ki67 levels combined with low ERCC1 expression, yielding better survival. However, patients with low-medium Ki67 expression had limited treatment sensitivity as low risk cases; therefore, they might be exempted from high-intensity treatment.
This study had some limitations. First, due to limited data in this retrospective analysis, we did not assess the efficacy of Ki67 and ERCC1 as well as chemoradiotherapy, or verify the impacts of Ki67 can ERCC1 on radiation and drug sensitivity. Insufficient follow-up time and small sample size were additional shortcomings. Longer follow-up and/or prospective studies with larger sample size would be helpful to further confirm the actual significance of Ki67 and ERCC1 expression in NPC. Moreover, combination analysis of Ki67 and ERCC1 could guide prognosis prediction and treatment options for NPC.
High expression levels of Ki67 and ERCC1 are associated with adverse prognosis of NPC. Combined analysis can be more effective in predicting survival prognosis. Ki67 and ERCC1 highly and concurrently expressed result in significantly decreased OS, DFS, DMFS, and LRFS rates, with increased risk of adverse prognosis, suggesting this combination can indicate adverse prognosis more sensitively; reasonable treatment may improve the survival of patients with cross-expression of Ki67 and ERCC1.
MATERIALS AND METHODS
Clinical data
362 NPC patients were selected from the Fourth Affiliated Hospital of Guangxi Medical University from January 2011 to December 2014. Inclusion criteria were: 1) no distant metastasis or previous treatment; 2) pathology of non-keratinizing carcinoma according to the 2005 WHO pathological classification [3]; 3) defined stage according to seventh edition of UICC/AJCC clinical staging standard [25]; 4) IMRT-based radiotherapy or Platinum-based chemoradiotherapy. The study was approved by the Ethics Committee of our Hospital, and informed consent was waived.
Immunohistochemistry
All tissue samples are gained from the nasopharyngeal biopsy by the nasopharyngeal lens in the diagnose and immunohistochemistry should be finished before the therapy.
Tissue sections were dewaxed and hydrated, and placed in 0.01 mol/L citrate buffer at pH 6.0 for high temperature and high pressure antigen retrieval. The sections were washed with PBS for 5 min, and 3% H2O2 was added for 10 min at room temperature. Next, the sections were washed with PBS for 5 min (three times) before addition of primary antibodies raised against Ki67 and ERCC1, respectively, for overnight incubation at 4°C. Subsequently, the sections were incubated with secondary antibodies for 15 min at room temperature. After washing with PBS as above, the DAB reagent was added for color development, and counterstaining was carried out by hematoxylin staining. All antibodies and kits were purchased from Beijing Zhongshan Biotechnology Co., Ltd (China).
Immunohistochemistry data interpretation
Independent assessments of the results for immunohistochemical staining of ERCC1 and Ki67 were performed by two blinded pathologists.
Ki67 positive cells were graded as: < 10%, grade 0; 10%~25%, grade 1 (+); > 25%~50%, grade 2 (++); > 50%, grade 3 (+++). Furthermore, ≤ 25% was considered low expression, >25%~50% medium level, and > 50% high expression.
ERCC1 positive cells were graded as: < 5%, grade 0; 5%~25%, grade 1; 26%~50%, grade 2; 5l%~75%, grade 3; > 75%, grade 4. Staining grades included: 0, no staining; 1, weakly positive staining; 2, medium staining; 3, strong staining.
Based on the sum of the two results: a total score of 0~1 represented no staining (-), 2~3 weakly positive (+), 4~5 medium positive (++), and 6~7 strongly positive (+++); meanwhile, (-~+) was considered low expression, and (++~+++) indicated high expression.
Statistical analysis
Statistical analyses were performed with SPSS19.0 (SPSS, USA). Categorical data were descriptively analyzed; the associations of Ki67 and ERCC1 expression levels with clinical characteristics were assessed by χ2 test. Overall survival (OS), disease-free survival (DFS), distant metastasis-free survival (DMFS) and local recurrence-free survival (LRFS) were assessed. The Kaplan-Meier method and log-rank test, as well as the cox risk ratio model were used to perform correlation analyses of survival and prognosis. Two sided P < 0.05 was considered statistically significant.
Abbreviations
overall survival (OS); disease-free survival (DFS); distant metastasis-free survival (DMFS); local recurrence-free survival (LRFS); Nasopharyngeal carcinoma (NPC); objective response rate (ORR).
Authors’ contributions
(1) Ying Lu, Haixin Huang and Rensheng Wang: Study conception and design; Ying Lu, Hui Yang and Sibei Wu: data acquisition, analysis, and interpretation.
(2) Ying Lu: Manuscript writing; Rensheng Wang, Haixin Huang and Min Kang: critical revision of for important intellectual content.
(3) Rensheng Wang: Final approval of the version to be submitted.
ACKNOWLEDGMENTS
We would like to thank the patients and staff of the Fourth Affiliated Hospital of Guangxi Medical University Oncology Department.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
FUNDING
This work was supported by the Guangxi Zhuang Autonomous Region Natural Science Foundation project (#2011GXZSFA018236), the Liuzhou City Science and technology research projects (#2016G020204), and the Department of Health of Guangxi Zhuang Autonomous Region Self-raised Funds Project (# Z2016167).
REFERENCES
1. Lang J, Gao L, Guo Y, Zhao C, Zhang C. Comprehensive treatment of squamous cell cancer of head and neck: Chinese expert consensus 2013. Future Oncol. 2014; 10:1635–1648.
2. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.
3. Thompson LD. Update on nasopharyngeal carcinoma. Head Neck Pathol. 2007; 1:81–86.
4. Qin HD, Shugart YY, Bei JX, Pan QH, Chen L, Feng QS, Chen LZ, Huang W, Liu JJ, Jorgensen TJ, Zeng YX, Jia WH. Comprehensive pathway-based association study of DNA repair gene variants and the risk of nasopharyngeal carcinoma. Cancer Res. 2011; 71:3000–3008.
5. Vicente-Duenas C, Romero-Camarero I, Cobaleda C, Sanchez-Garcia I. Function of oncogenes in cancer development: a changing paradigm. Embo j. 2013; 32:1502–1513.
6. Kumar RD, Searleman AC, Swamidass SJ, Griffith OL, Bose R. Statistically identifying tumor suppressors and oncogenes from pan-cancer genome-sequencing data. Bioinformatics. 2015; 31:3561–3568.
7. Hall PA, Levison DA, Woods AL, Yu CC, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R. Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: an index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol. 1990; 162:285–294.
8. Dowsett M, Nielsen TO, A’Hern R, Bartlett J, Coombes RC, Cuzick J, Ellis M, Henry NL, Hugh JC, Lively T, McShane L, Paik S, Penault-Llorca F, et al. Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working group. J Natl Cancer Inst. 2011; 103:1656–1664.
9. Martin B, Paesmans M, Mascaux C, Berghmans T, Lothaire P, Meert AP, Lafitte JJ, Sculier JP. Ki-67 expression and patients survival in lung cancer: systematic review of the literature with meta-analysis. Br J Cancer. 2004; 91:2018–2025.
10. Pascale M, Aversa C, Barbazza R, Marongiu B, Siracusano S, Stoffel F, Sulfaro S, Roggero E, Bonin S, Stanta G. The proliferation marker Ki67, but not neuroendocrine expression, is an independent factor in the prediction of prognosis of primary prostate cancer patients. Radiol Oncol. 2016; 50:313–320.
11. Piri R, Ghaffari A, Azami-Aghdash S, Ali-Akbar YP, Saleh P, Naghavi-Behzad M. Ki-67/MIB-1 as a Prognostic Marker in Cervical Cancer–a Systematic Review with Meta-Analysis. Asian Pac J Cancer Prev. 2015; 16:6997–7002.
12. Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thurlimann B, Senn HJ. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013; 24:2206–2223.
13. Xie S, Liu Y, Qiao X, Hua RX, Wang K, Shan XF, Cai ZG. What is the Prognostic Significance of Ki-67 Positivity in Oral Squamous Cell Carcinoma? J Cancer. 2016; 7:758–767.
14. Wangsa D, Ryott M, Avall-Lundqvist E, Petersson F, Elmberger G, Luo J, Ried T, Auer G, Munck-Wikland E. Ki-67 expression predicts locoregional recurrence in stage I oral tongue carcinoma. Br J Cancer. 2008; 99:1121–1128.
15. Genc E, Hosal AS, Gedikoglu G, Ozyar E, Sozeri B. Prognostic value of p53, proliferating cell nuclear antigen, and Ki-67 expression in undifferentiated nasopharyngeal carcinomas. Otolaryngol Head Neck Surg. 2000; 122:868–873.
16. Ma BB, Poon TC, To KF, Zee B, Mo FK, Chan CM, Ho S, Teo PM, Johnson PJ, Chan AT. Prognostic significance of tumor angiogenesis, Ki 67, p53 oncoprotein, epidermal growth factor receptor and HER2 receptor protein expression in undifferentiated nasopharyngeal carcinoma--a prospective study. Head Neck. 2003; 25:864–872.
17. Olaussen KA, Dunant A, Fouret P, Brambilla E, Andre F, Haddad V, Taranchon E, Filipits M, Pirker R, Popper HH, Stahel R, Sabatier L, Pignon JP, et al. DNA repair by ERCC1 in non-small-cell lung cancer and cisplatin-based adjuvant chemotherapy. N Engl J Med. 2006; 355:983–991.
18. Azuma K, Komohara Y, Sasada T, Terazaki Y, Ikeda J, Hoshino T, Itoh K, Yamada A, Aizawa H. Excision repair cross-complementation group 1 predicts progression-free and overall survival in non-small cell lung cancer patients treated with platinum-based chemotherapy. Cancer Sci. 2007; 98:1336–1343.
19. Li S, Wu J, Chen Y, Tang W, Peng Q, Deng Y, Xie L, Wang J, Huang S, Li R, Qin X, Zhao J. ERCC1 expression levels predict the outcome of platinum-based chemotherapies in advanced bladder cancer: a meta-analysis. Anticancer Drugs. 2014; 25:106–114.
20. Hwang IG, Ahn MJ, Park BB, Ahn YC, Han J, Lee S, Kim J, Shim YM, Ahn JS, Park K. ERCC1 expression as a prognostic marker in N2(+) nonsmall-cell lung cancer patients treated with platinum-based neoadjuvant concurrent chemoradiotherapy. Cancer. 2008; 113:1379–1386.
21. Jun HJ, Ahn MJ, Kim HS, Yi SY, Han J, Lee SK, Ahn YC, Jeong HS, Son YI, Baek JH, Park K. ERCC1 expression as a predictive marker of squamous cell carcinoma of the head and neck treated with cisplatin-based concurrent chemoradiation. Br J Cancer. 2008; 99:167–172.
22. Bisof V, Zajc Petranovic M, Rakusic Z, Samardzic KR, Juretic A. The prognostic and predictive value of excision repair cross-complementation group 1 (ERCC1) protein in 1288 patients with head and neck squamous cell carcinoma treated with platinum-based therapy: a meta-analysis. Eur Arch Otorhinolaryngol. 2016; 273:2305–2317.
23. Xuelei M, Jingwen H, Wei D, Hongyu Z, Jing Z, Changle S, Lei L. ERCC1 plays an important role in predicting survival outcomes and treatment response for patients with HNSCC: a meta-analysis. Oral Oncol. 2015; 51:483–492.
24. Gao Y, Liu D. The roles of excision repair cross-complementation group1 in objective response after cisplatin-based concurrent chemoradiotherapy and survival in head and neck cancers: a systematic review and meta-analysis. Oral Oncol. 2015; 51:570–577.
25. Edge SB, Byrd DR, Compton CC. (2010). AJCC cancer staging manual. (NY, USA: Springer).
26. Zhang MX, Li J, Shen GP, Zou X, Xu JJ, Jiang R, You R, Hua YJ, Sun Y, Ma J, Hong MH, Chen MY. Intensity-modulated radiotherapy prolongs the survival of patients with nasopharyngeal carcinoma compared with conventional two-dimensional radiotherapy: A 10-year experience with a large cohort and long follow-up. Eur J Cancer. 2015; 51:2587–2595.
27. Kang M, Long J, Li G, Yan H, Feng G, Liu M, Zhu J, Wang R. A new staging system for nasopharyngeal carcinoma based on intensity-modulated radiation therapy: results of a prospective multicentric clinical study. Oncotarget. 2016; 7:15252–15261. https://doi.org/10.18632/oncotarget.7553.
28. Shi S, Li X, You B, Shan Y, Cao X, You Y. High Expression of FGFR4 Enhances Tumor Growth and Metastasis in Nasopharyngeal Carcinoma. J Cancer. 2015; 6:1245–1254.
29. Shen C, Chen L, Fu J, Lin H. Expression of excision repair cross-complementation group 1 in locoregionally advanced nasopharyngeal carcinoma treated with cisplatin-based induction chemotherapy. J Cancer Res Ther. 2016; 12:72–75.
30. Huang PY, Li Y, Mai HQ, Luo RZ, Cai YC, Zhang L. Expression of ERCC1 predicts clinical outcome in locoregionally advanced nasopharyngeal carcinoma treated with cisplatin-based induction chemotherapy. Oral Oncol. 2012; 48:964–968.
31. Liang R, Lin Y, Liu ZH, Liao XL, Yuan CL, Liao SN, Li YQ. Correlation between ERCC1 expression and concurrent chemotherapy and radiotherapy in patients with locally advanced nasopharyngeal cancer. Genet Mol Res. 2015; 14:5804–5811.
32. Freudlsperger C, Freier K, Hoffmann J, Engel M. Ki-67 expression predicts radiosensitivity in oral squamous cell carcinoma. Int J Oral Maxillofac Surg. 2012; 41:965–969.
33. Koelbl O, Rosenwald A, Haberl M, Muller J, Reuther J, Flentje M. p53 and Ki-67 as predictive markers for radiosensitivity in squamous cell carcinoma of the oral cavity? an immunohistochemical and clinicopathologic study. Int J Radiat Oncol Biol Phys. 2001; 49:147–154.
34. Sun JM, Ahn MJ, Park MJ, Lee HY, Ahn JS, Lee S, Kang G, Han J, Son YI, Baek CH, Ahn YC, Park K. Expression of excision repair cross-complementation group 1 as predictive marker for nasopharyngeal cancer treated with concurrent chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2011; 80:655–660.

