INTRODUCTION
Pancreatic cancer is a highly fatal malignancy for which the incidence is increasing and mortality closely parallels the incidence [1, 2]. Surgical resection is regarded as the only potentially curative treatment for pancreatic cancer [3]. However, more than half of patients are not feasible for surgical resection at first diagnosis and there are no other effective treatments [4]. Advances in the improved survival are slow for pancreatic cancer, for which the 5-year relative survival is about 8% [2]. Thus, it is important to identify modifiable risk factors for pancreatic cancer incidence and mortality.
Vitamin D is a fat-soluble vitamin that correlates with calcium, phosphate, and bone metabolism [5]. Plasma 25-hydroxyvitamin D [25(OH)D], the precursor of the physiologically active form of vitamin D, is considered the best biomarker of vitamin D status because plasma 25(OH)D levels reflects both vitamin D intake from diet and synthesis from ultraviolet-B (UVB) exposure [6, 7]. In recent years, the role of vitamin D and its analogues in carcinogenesis has drawn more and more attention. Indeed, several studies have shown that vitamin D intake or plasma 25(OH)D levels are associated with the risk and mortality of breast cancer and colorectal cancer [8–11]. And for pancreatic cancer, experimental evidence demonstrates that vitamin D and its analogues may inhibit pancreatic cancer cell proliferation, induce differentiation, and promote apoptosis [12–16]. However, for clinical studies, the results on the association between vitamin D status, including vitamin D intake or plasma 25(OH)D levels, and pancreatic cancer risk and mortality are still inconsistent.
Thus, the purposes of this study are to assess whether vitamin D intake or plasma 25(OH)D levels is associated with pancreatic cancer risk and mortality through meta-analysis approach.
RESULTS
Study selection
There were 853 studies identified from literature search, and 635 studies were excluded after reviewing titles and abstracts. The remaining 218 studies were evaluated based on full-text review, and 206 studies were excluded based on eligible criteria. Finally, twelve studies were included (Figure 1) [17–28].
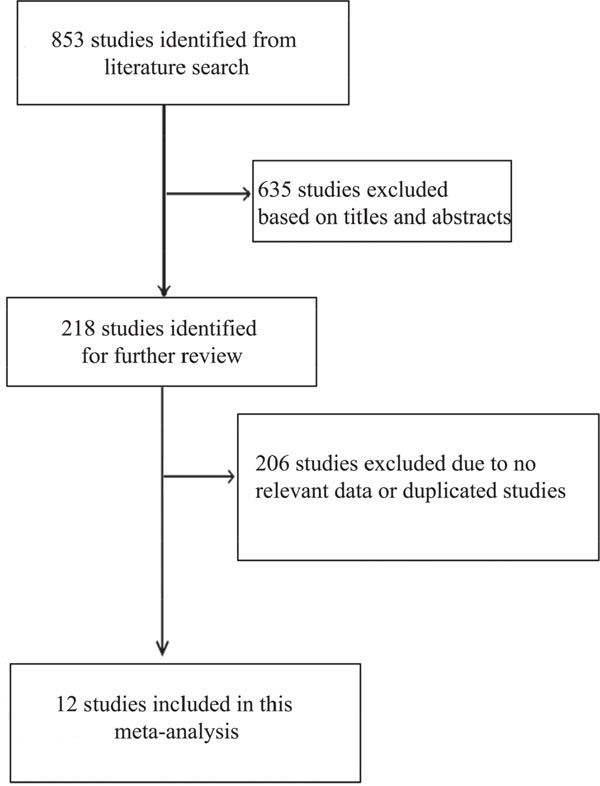
Figure 1: Literature search and study selection.
Study characteristics
Five studies on pancreatic cancer mortality in relation to plasma 25(OH)D levels were published between 2013 and 2016, and involved a total of 1613 patients [19, 21, 22, 25, 28] (Table 1). Of these five studies, four were conducted in the United States and one in Germany. Five studies on pancreatic cancer risk in relation to plasma 25(OH)D levels were published between 2010 and 2015, and involved a total of 17385 patients [17, 18, 23, 24, 27] (Table 2). Of these five studies, four were conducted in the United States and one in Denmark. Two studies on pancreatic cancer risk in relation to vitamin D intake were published between 2014 and 2015, and were conducted in the United States, involving a total of 874170 patients [20, 26] (Table 3). The baseline characteristics and quality of the included studies were shown in Tables 1-3.
Table 1: The main characteristics of included studies on plasma 25(OH)D levels and pancreatic cancer mortality
Article |
Country & year |
Study type |
Study name |
Sample (M/F) |
Age (years) |
Tumor feature |
BMI |
Baseline 25(OH)D (ng/mL) |
Study quality |
|---|---|---|---|---|---|---|---|---|---|
Yuan |
USA 2016 |
Cohort |
Health Professionals Follow-up Study, Nurses’ Health Study, Physicians’ Health Study, Women’s Health Initiative and Women’s Health Study |
493(148/345) |
Mean:71.2; SD:8.3 |
Localized:65; |
BMI<25.0:216; |
Mean:24.6; SD:9.84 |
7 |
McGovern |
USA 2016 |
Cohort |
Five Cancer Treatment Centers of America hospitals |
627(342/285) |
Mean:57;SD:7.7; range:31-82 |
I-II:83;III:86;IV:454 |
BMI<18.5:26; |
Mean: |
6 |
Haas |
Germany 2015 |
Cohort |
German Pancreatic Cancer Center (Comprehensive Cancer Center) |
59(33/26) |
Median:69; range:39-84 |
Locally advanced:11; |
NR |
Median:19.9; |
6 |
Van Loon |
USA 2014 |
Cohort |
Cancer and Leukemia Group B 80303 |
256(136/120) |
Median:64; range:35-84 |
Locally advanced:217; |
BMI<25:126; |
Median:21.7; |
7 |
Cho |
USA 2013 |
Cohort |
Siteman Cancer Center in St. Louis |
178(96/82) |
<50y:17; ≥50y:161 |
I+II:64;III+IV:114 |
BMI <25:68; |
NR |
7 |
25(OH)D: 25-hydroxyvitamin D; BMI: body mass index; M1: metastatic disease; M/F: male/female; NR: not reported; SD: standard deviation; USA: the United States of America.
Table 2: The main characteristics of included studies on plasma 25(OH)D levels and pancreatic cancer risk
Article |
Country |
Study design |
Sample (M/F) |
Age (years) |
Follow up (years) |
Baseline 25(OH)D (ng/mL) |
BMI |
Adjusted variables |
Study quality |
|---|---|---|---|---|---|---|---|---|---|
Piper 2015 |
USA |
Nested CCS |
882(549/ |
Median: 65; IDR:57-71 |
Follow-up to 15.1 |
Case:24.36(IDR |
BMI<25:306; |
Age, race-ethnicity, sex, and date of blood draw, smoking status and diabetes |
7 |
Ananthakris-hnan 2014 |
USA |
Cohorts |
2809 (1097/1712) |
Median:46; IQR:32-60 |
Median:11 |
Median:26; |
NR |
Age, sex, race, measurement season, follow-up duration, immunosuppression use and IBD type |
6 |
Afzal 2013 |
Denmark |
Cohorts |
9791 (4358/5431) |
<5ng/ml:59(IQR50-65); 5-9.9:58(IQR49-65); |
Median:21; |
Median:16.4 |
<5ng/ml:25 (22-29); |
Age, sex, pack-years, BMI, alcohol consumption, leisure time, physical activity and education duration |
7 |
Wolpin 2012 |
USA |
Nested CCS |
1618 (518/1100) |
Mean:62.5;SD:8.5 |
Median:14.3 |
Mean:25.8; SD:9.28 |
Mean:25.9; SD:4.5 |
Age, cohort, BMI, smoking status, history of diabetes mellitus, multivitamin use and month of blood draw. |
7 |
Stolzenberg-Solomon 2010 |
USA |
Nested CCS |
2285 (1521/764) |
Median: |
Median:6.5; |
Range: |
BMI<25:876; |
Age, race/ethnicity, sex, cohort, date of blood draw, BMI, smoking and diabetes status. |
7 |
Abbreviations, 25(OH)D: 25-hydroxyvitamin D; BMI: body mass index; CCS: case-control study; IDR: interdecile range; IQR: interquartile range; M/F: male/female; NR: not reported; SD: standard deviation; USA: the United States of America.
Table 3: The main characteristics of included studies on vitamin D intake and pancreatic cancer risk
Article |
Country |
Year |
Study design |
Age (years) |
Smoking |
BMI |
Ethnicity (white/black) |
Sample (M/F) |
Adjusted variables |
Study quality |
|---|---|---|---|---|---|---|---|---|---|---|
Waterhouse |
USA |
2015 |
Case-control study |
Median:65 |
Ever:6525; never:4948 |
BMI<25:4491; |
9159/408 |
11490(6165/5325) |
Age, sex, smoking status, total daily energy intake and other cohort-specific confounders (alcohol intake, BMI, history of diabetes, history of pancreatitis, family history of pancreatic cancer, education, race, total calcium intake, dietary calcium intake, total retinol intake, dietary retinol intake, total vitamin A intake, study centre and extent of proxy use) |
7 |
Genkinger |
USA |
2014 |
Cohorts |
Range:15-107; |
NR |
NR |
NR |
862680 |
Smoking habits, personal history of diabetes, alcohol intake, BMI, energy intake, age in years and year of questionnaire return |
6 |
Abbreviations, BMI: body mass index; M/F: male/female; NR: not reported; USA: the United States of America.
Association between plasma 25(OH)D levels and pancreatic cancer mortality
Five studies assessed the association between plasma 25(OH)D levels and pancreatic cancer mortality [19, 21, 22, 25, 28]. Our results indicated that the high versus the low plasma 25(OH)D levels was significantly associated with reduced pancreatic cancer mortality without significant heterogeneity (HR=0.81, 95% CI=0.68–0.96, I2=43.0%; Figure 2), and the result of Begg’s and Egger’s tests showed no evidence of publication bias (PBegg's = 1.00, PEgger's = 0.80, Figure 3). Moreover, we performed a subgroup analysis including only advanced disease, and the result indicated that high plasma 25(OH)D levels tended toward a favorable survival for locally advanced or metastatic disease, although statistical significance was not reached (HR=0.76, 95% CI=0.55–1.04, I2=51.4%). Of those five studies, only two studies provided the HR on progression-free survival (PFS) [22, 25], and the pooled HR indicated no association between plasma 25(OH)D levels and PFS for pancreatic cancer (HR=1.06, 95% CI=0.84–1.33, I2=12.9%).
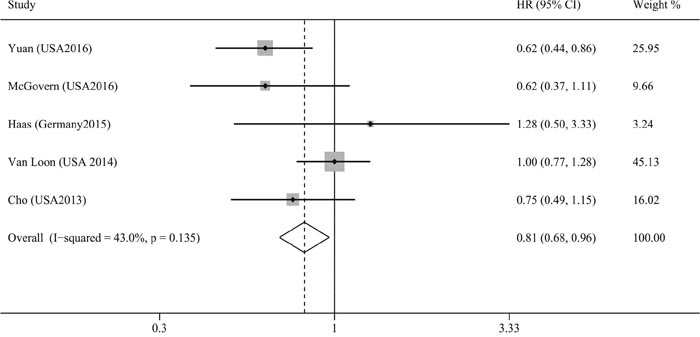
Figure 2: Meta-analysis of the association between plasma 25(OH)D levels and pancreatic cancer mortality.
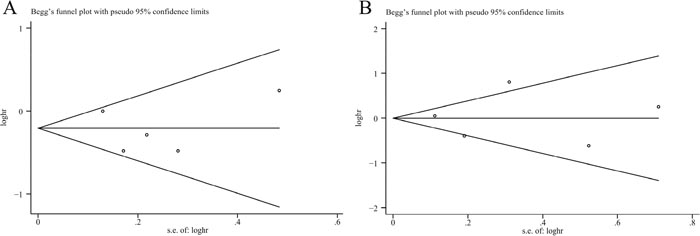
Figure 3: Funnel plots assessing publication bias for plasma 25(OH)D levels and pancreatic cancer mortality (A) and pancreatic cancer risk (B).
Association between plasma 25(OH)D levels and pancreatic cancer risk
There were five studies assessing the association between plasma 25(OH)D levels and pancreatic cancer risk [17, 18, 23, 24, 27]. And comparing with low plasma 25(OH)D levels, high plasma 25(OH)D levels could not reduce pancreatic cancer risk (RR=1.02, 95% CI=0.66–1.57, I2=68.9%; Figure 4), without publication bias (PBegg's = 1.00, PEgger's = 0.94, Figure 3). The sensitivity analysis indicated that the result was not obviously affected by any single study (Supplementary Figure 1). And the Galbraith plot showed that the study by Stolzenberg et al. [24] may contribute substantial heterogeneity (Supplementary Figure 2). Exclusion of the study by Stolzenberg et al. [24] could obtain similar result, with reduced heterogeneity (RR=0.93, 95% CI=0.77–1.11, I2=44.9%). Stratifying by sex, smoking status, ethnicity, and time between plasma collection and diagnosis, these subgroup analyses obtained similar results, indicating no association between plasma 25(OH)D levels and pancreatic cancer risk.
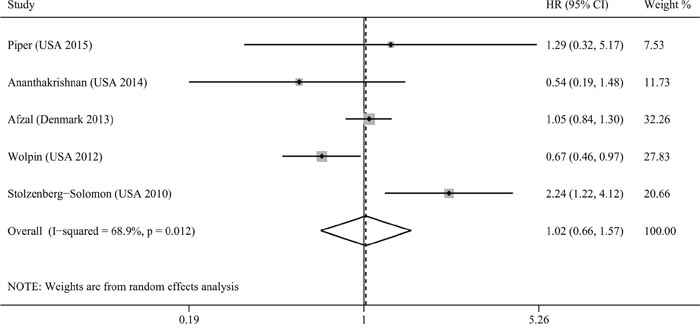
Figure 4: Meta-analysis of the association between plasma 25(OH)D levels and pancreatic cancer risk.
Association between vitamin D intake and pancreatic cancer risk
Two studies assessed the association between vitamin D intake and pancreatic cancer risk [20, 26]. Our result indicated that total vitamin D intake was not associated with pancreatic cancer risk (RR=1.11, 95% CI=0.67–1.86, I2=68.7%; Figure 5). Subgroup analysis based on dietary vitamin D intake was performed, and similar result was obtained (RR=1.16, 95% CI=0.85–1.59, I2=58.2%). However, our study could not assess the impact of supplementary vitamin D intake on pancreatic cancer risk because of the limited number of included studies.
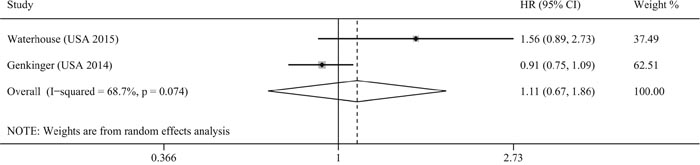
Figure 5: Meta-analysis of the association between vitamin D intake and pancreatic cancer risk.
DISCUSSION
Recent studies have shown that vitamin D status could reduce the incidence and improve the prognosis for numerous solid cancer risks [8-10, 29, 30]. However, there were no general agreements on the association between vitamin D status and pancreatic cancer risk as well as mortality.
Our studies included twelve eligible studies to evaluate the association between vitamin D status and pancreatic cancer risk and mortality. The results indicated that high plasma 25(OH)D levels were significantly associated with reduced pancreatic cancer mortality for pancreatic cancer, but not with PFS. And patients with high plasma 25(OH)D levels had a 19% lower risk for mortality (HR=0.81, 95% CI=0.68–0.96). However, our results indicated that high vitamin D intake or plasma 25(OH)D levels could not reduce the incidence of pancreatic cancer.
Our results were consistent with the 25(OH)D levels-cancer survival relationship reported from several studies, which have shown a favorable effect of vitamin D on cancer survival. Grant et al. reported that lower plasma 25(OH)D levels among African Americans than white Americans may explain many of the cancer survival disparities after consideration of socioeconomic status, stage at time of diagnosis, and treatment [31]. Gaksch et al. analyzed 26916 individuals and the results showed that individuals with low plasma 25(OH)D levels had unfavorable cancer mortality and plasma 25(OH)D levels may affect cancer mortality in a nonlinear relationship [32, 33]. A population-based study by Tretli et al. also showed that higher plasma 25(OH)D levels were positively associated with the survival for cancers of the breast, colon, lung, and lymphoma [34].
Although the intrinsic molecular mechanisms on vitamin D-cancer survival relationship remained unclear, there were some plausible mechanisms. 25(OH)D could be converted to 1,25-dihydroxyvitamin D [1,25(OH)2D], the active form of vitamin D, by 25(OH)D-1α-hydroxylase in pancreatic cell and 1,25(OH)2D bind to the vitamin D receptor (VDR) [35–37]. Then the 1,25(OH)2D-VDR complex promoted transcription of target genes involved in inhibition of proliferation and angiogenesis and induction of apoptosis and differentiation by interacting with vitamin D response elements (VDRE) [12, 38–40]. Moreover, some genes that lacked VDRE in their promoter regions were also transcriptionally affected by 1,25(OH)2D, which suggested that 1,25(OH)2D may have VDR-independent pathway for the antitumor effects [40]. And several studies have reported that 1,25(OH)2D could suppress the expression of epidermal growth factor and insulin-like growth factor 1 and promote the expression of transformation growth factor-β, which was an inhibitory growth factor, through VDR-independent pathway [41, 42]. In addition, pancreatic islet cells expressed 25(OH)D-1α-hydroxylase and VDR [43]. Thus, 25(OH)D may be linked to endocrine pancreatic function and inhibit pancreatic cancer development through regulation of insulin synthesis, binding, and responsiveness [14, 44–46]. Further studies are needed to elucidate the precise antitumor mechanisms, including VDR-dependent and VDR-independent pathways induced by 1,25(OH)2D.
Considering the antitumor activity of 1,25(OH)2D against pancreatic cancer, several studies were performed to evaluate the therapeutic effect of 1,25(OH)2D when used in combination with chemotherapy. A phase II study assessing calcitriol-enhanced docetaxel regimen showed that high-dose calcitriol with docetaxel may have activity in incurable pancreatic cancer, with a modest increase in time to progression when compared to single docetaxel regimen [47]. Barreto et al. also reported that the perioperative vitamin D use was safe in patients with operable pancreatic cancer [48]. Similar result was observed in prostate cancer, and a double-blind randomized phase II study by Beer et al. demonstrated that high-dose calcitriol plus docetaxel could produce 58% prostate-specific antigen decline (>50% confirmed 4weeks later) and was associated with improved overall survival (HR=0.6, 95% CI=0.45–0.97) [49]. The reasons for the synergistic effect of 1,25(OH)2D and chemotherapy were unclear. In addition to the above-mentioned mechanisms, vitamin D could markedly reduce inflammation and fibrosis in pancreatic tumor stroma and then induced stromal remodeling and increased intratumoral chemotherapy drug compared to chemotherapy alone because the VDR expressed in pancreatic tumor stroma acted as a master transcriptional regulator of pancreatic stellate cells to reprise the quiescent state [50]. Thus, expression of VDR in cancer cell and stroma cell may be a therapeutic target for pancreatic cancer. Despite the fact that 1,25(OH)2D was the biologically active form of vitamin D and directly exerted biological effects in tissue, the clinical use of 1,25(OH)2D may be impeded by the lethal side effects of hypercalcemia and hypercalciuria [40]. One strategy was using synthetic 1,25(OH)2D analogs in an effort to avoid those side effects. And numerous studies have shown that 1,25(OH)2D analogs exhibited comparable antitumor actions [51, 52]. Another strategy was administration of inactive prohormone 25(OH)D. Further studies are needed to evaluate the clinical benefit of vitamin D and its analogs in pancreatic cancer patients and to develop novel vitamin D analogs with better antitumor effects and lower side effects.
Although in vitro experimental studies have noted the protective effect of vitamin D and pancreatic cancer [53], both our pooled result and the included studies indicated null protective associations between vitamin D intake and pancreatic cancer risk. The possible explanation was that vitamin D intake was a small fraction of total vitamin D from all sources and meat intake, as an important source of vitamin D intake, was generally not considered in studies [54]. Indeed, studies have shown that vitamin D intake could not appropriately reflect internal vitamin D status because internal vitamin D primarily produced from endogenous synthesis from sunlight exposure [6, 55], and there was no sufficient evidence to support a significantly linear association between vitamin D intake and plasma 25(OH)D levels. Thus, it may be understandable that vitamin D intake was not a good indicator for pancreatic cancer risk. In terms of plasma 25(OH)D levels, several epidemiological studies have shown inconsistent and conflicted results. Wolpin et al. conducted a study assessing the chemopreventive effect of plasma 25(OH)D levels and demonstrated that higher plasma levels of 25(OH)D were associated with a lower risk of pancreatic cancer, consistent with their previous study [27, 56]. In contrast, the Prostate, Lung, Colorectal and Ovarian Screening Trial cohort showed an insignificant association between plasma 25(OH)D levels and pancreatic cancer risk [23, 57], and the Copenhagen City Heart Study also showed a similar result [17]. Surprisingly, however, a prospective nested case-control study in the Alpha-Tocopherol, Beta-arotene Cancer Prevention study of male Finnish smokers showed that high plasma 25(OH)D levels could increase the risk of pancreatic cancer (OR=2.92, 95% CI=1.56–5.48) [58]. The potential hypothesis for why male Finnish smokers with high plasma 25(OH)D levels had an increased pancreatic cancer risk may be that the interrelation between plasma 25(OH)D levels and pancreatic cancer was different for smokers and nonsmokers [59]. Studies suggested that cigarette smoking has repeatedly been associated with reductions in plasma levels of 25(OH)D [60, 61]. And the effects of smoking on vitamin D status may be involved in the carcinogenic effects of tobacco smoking [62]. Large studies are required to determine whether the associations between plasma 25(OH)D levels, vitamin D intake and pancreatic cancer risk are different among groups with different risk profiles.
Vitamin D was primarily derived from UVB radiation of precursors in skin [63], and thus there have been several UVB indices used in epidemiological studies on pancreatic cancer, including solar UVB dose, solar UV dose, solar radiation dose, and latitude [64]. Several studies evaluated the UVB-vitamin D-cancer hypothesis based on geographical variation of pancreatic cancer incidence and/or mortality [63, 65–67]. Grant et al. and Tran et al. showed that solar UVB or UV radiation was inversely associated with pancreatic cancer risk [63, 67]. Kinoshita et al. reported that high solar radiation could reduce pancreatic cancer risk [65]. Similarly, Neale et al. found a significant association between latitude, UV radiation and pancreatic cancer mortality, with a 1.5% decrease in mortality for every 10-kJ/m2 increase in yearly UV radiation [66]. Studies suggested that the inverse association between UVB radiation and pancreatic cancer risk and mortality may be mainly mediated by production of vitamin D. However, our present result did not found a significant association between plasma 25(OH)D levels and pancreatic cancer risk. Thus, further studies are needed to explore not only the association between UVB radiation and plasma 25(OH)D levels but also whether there are other pathways contribute to the protective effect of UVB radiation.
Several limitations of our meta-analysis should be considered. First, high plasma 25(OH)D levels may be correlated to favorable health status impacting survival. Non-differential misclassifications of vitamin D status may exist among some included studies, and some inherent confounding factors could not be solved perfectly in included studies. Thus, an underestimation of risk estimates of vitamin D on pancreatic cancer may exist in this study. Second, the included studies on pancreatic cancer risk used a single measure of plasma 25(OH)D levels and had long median follow-up period, ranging from 6.5 to 21 years. Thus, there was obvious concern that the studies with long follow-up period may lead to errors because a single 25(OH)D levels may be inappropriate for long-term vitamin D status and lose predictive ability for pancreatic cancer risk over time. Indeed, several studies have investigated the effect of follow-up period on the association between plasma 25(OH)D levels and cancer incidence and consistently showed that the association decreased with longer follow-up period [68–70]. Therefore, multiple 25(OH)D level measurements would be helpful and yield more accurate results, perhaps every 2 years and in different seasons. Further studies are needed to accurately evaluate the associations between plasma 25(OH)D levels and pancreatic cancer risk through multiple 25(OH)D level measurements during follow-up period. Third, significant heterogeneity existed among the studies, especially in the analysis on plasma 25(OH)D levels and pancreatic cancer risk, and could not be eliminated or explained completely because of differences in patient characteristics (i.e., age, sex and race) or treatment strategies. And we addressed the heterogeneity by using a relatively conservative random-effects model and Galbraith plot analysis. Therefore, the antitumor and therapeutic effect of vitamin D status should be confirmed with further large-scale multicenter studies with homogeneous patients. In addition, only two studies were included in the analysis evaluating vitamin D intake and pancreatic cancer risk; thus, the limited number of studies may impact the statistical power and limit interpretation of the results.
In conclusion, our results indicate that high plasma 25(OH)D levels was significantly associated with favorable survival for pancreatic cancer patients. No significant associations were observed between vitamin D intake or plasma 25(OH)D levels and pancreatic cancer risk. It is should be noted that most studies were confined to USA, which may impact the generalization of present results for pancreatic cancer worldwide. Further large-scale multicenter studies are required to assess the protective effect of vitamin D for pancreatic cancer patients worldwide.
MATERIALS AND METHODS
Literature search
A literature search for relevant studies was conducted using PubMed and Embase databases. Besides, a manual search for potential studies was also conducted using the reference lists of reviews and relevant studies. The following search terms were used in the database in a free manner and “MeSH Terms”: “vitamin D”, “25-hydroxy vitamin D”, “25-hydroxyvitamin D”, “25 hydroxy vitamin D”, “25 hydroxyvitamin D”, “1,25-dihydroxy vitamin D”, “1,25-dihydroxyvitamin D”, “1,25 dihydroxy vitamin D”, “1,25 dihydroxyvitamin D”, “25-hydroxyvitamin D3 1-alpha-hydroxylase”, “cholecalciferol”, “calcidiol”, “calcifediol”, “calcitriol”, “hydroxycholecalciferol”, “ergocalciferol”, “pancreatic cancer”, “pancreatic tumor”, “pancreatic neoplasm”, “pancreatic carcinoma”, “pancreas cancer”, “pancreas tumor”, “pancreas neoplasm”, and “pancreas carcinoma”. The detailed search strategy was shown in Supplementary File 1
Eligibility criteria
Studies that met the following eligible criteria were included: (1) the exposure of interest was vitamin D intake, or plasma 25(OH)D levels; (2) the outcome of interest was pancreatic cancer incidence or mortality; (3) the outcome measures [odds risk (OR), risk ratio (RR) or hazard ratio (HR)] and corresponding 95% confidence interval (CI) could be extracted; and (4) the study design was cohort study or case–control study with at least one of the outcome measures of interest. If several duplicated studies were published from the same population, only the most recent study was included and the data that only reported in excluded duplicated studies were also extracted.
Data extraction and quality assessment
Two authors reviewed the studies and extracted data independently. The following data was extracted: first author, publication year and country, study design, sample size, number of case, age and sex of patient, smoking status, tumor characteristics, outcome measures with corresponding 95% CIs, and adjusted variables. Outcome measures with the greatest degree of adjustment for potential confounders were extracted. The quality of the included studies was assessed using Newcastle–Ottawa Scale (NOS) criteria [71]. Any disagreements were resolved by discussion.
Statistical analysis
RR was used as a measure to evaluate the association between exposure and pancreatic cancer risk. OR in a case-control study was used as RR because the pancreatic cancer incidence was sufficiently rare and the OR were close to the RR [72]. HR was used as a measure to evaluate the association between plasma 25(OH)D levels and pancreatic cancer mortality. If HRs and 95% CIs were not reported directly, the values were recalculated from the available data [73]. The overall analysis was performed by evaluating all the relevant studies. Simultaneously, subgroup analysis was performed stratified by sex, tumor characteristics, smoking status, or ethnicity.
The heterogeneity among studies was evaluated using Cochran Q test and the I2 statistic [74]. A fixed-effects model was used if there was not significant heterogeneity among studies; otherwise, the random-effects model was used. Galbraith plot was used to explore the potential sources of heterogeneity. Publication bias was evaluated using Begg’s and Egger’s tests [75, 76]. A sensitivity analysis was performed to evaluate the stability of result and influence of a single study using the leave-one-out approach.
All statistical analyses were performed using Stata software 12.0 (Stata Corporation, College Station, TX, USA). A two-sided P value <0.05 was considered statistically significant.
Abbreviations
1,25(OH)2D: 1,25-dihydroxyvitamin D; 25(OH)D: 25-hydroxyvitamin D; CI: confidence interval; HR: hazard ratio; PFS: progression-free survival; RR: risk ratio; VDR: vitamin D receptor; VDRE: vitamin D response element.
Author contributions
X.Z., X.Z.H. and Z.N.W. were responsible for conception and design of the study. X.Z. and X.Z.H. did the studies selection, data extraction, and the writing of report. X.Z. did the statistical analyses. W.J.C. and J.W. provided statistical expertise. Y.C. and C.C.W. contributed to the literature search, acquisition of data, table and figures. X.Z.H. and W.J.C. provided draft of the article. X.Z., X.Z.H. and Z.N.W. provided clinical expertise and interpretation of data. All authors reviewed the final manuscript.
ACKNOWLEDGMENTS
We thank the department of Chemotherapy and Radiotherapy of Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University, and department of Surgical Oncology of First Hospital of China Medical University for technical assistance. The corresponding author had full access to all the data and analyses.
CONFLICTS OF INTEREST
All authors declare that they have no competing interests.
FUNDING
None.
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87-108.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017; 67:7-30.
3. Kamisawa T, Wood LD, Itoi T, Takaori K. Pancreatic cancer. Lancet. 2016; 388:73-85.
4. Gillen S, Schuster T, Meyer Zum Buschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010; 7:e1000267.
5. Rosen CJ. Clinical practice. Vitamin D insufficiency. N Engl J Med. 2011; 364:248-254.
6. Zerwekh JE. Blood biomarkers of vitamin D status. Am J Clin Nutr. 2008; 87:1087S-1091S.
7. Bulathsinghala P, Syrigos KN, Saif MW. Role of vitamin d in the prevention of pancreatic cancer. J Nutr Metab. 2010; 2010:721365.
8. Maalmi H, Ordonez-Mena JM, Schottker B, Brenner H. Serum 25-hydroxyvitamin D levels and survival in colorectal and breast cancer patients: systematic review and meta-analysis of prospective cohort studies. Eur J Cancer. 2014; 50:1510-1521.
9. Kim Y, Je Y. Vitamin D intake, blood 25(OH)D levels, and breast cancer risk or mortality: a meta-analysis. Br J Cancer. 2014; 110:2772-2784.
10. Ma Y, Zhang P, Wang F, Yang J, Liu Z, Qin H. Association between vitamin D and risk of colorectal cancer: a systematic review of prospective studies. J Clin Oncol. 2011; 29:3775-3782.
11. Moukayed M, Grant WB. The roles of UVB and vitamin D in reducing risk of cancer incidence and mortality: a review of the epidemiology, clinical trials, and mechanisms. Rev Endocr Metab Disord. 2017; 18:167-182.
12. Colston KW, James SY, Ofori-Kuragu EA, Binderup L, Grant AG. Vitamin D receptors and anti-proliferative effects of vitamin D derivatives in human pancreatic carcinoma cells in vivo and in vitro. Br J Cancer. 1997; 76:1017-1020.
13. Ohlsson B, Albrechtsson E, Axelson J. Vitamins A and D but not E and K decreased the cell number in human pancreatic cancer cell lines. Scand J Gastroenterol. 2004; 39:882-885.
14. Stolzenberg-Solomon RZ. Vitamin D and pancreatic cancer. Ann Epidemiol. 2009; 19:89-95.
15. Schwartz GG, Eads D, Rao A, Cramer SD, Willingham MC, Chen TC, Jamieson DP, Wang L, Burnstein KL, Holick MF, Koumenis C. Pancreatic cancer cells express 25-hydroxyvitamin D-1 alpha-hydroxylase and their proliferation is inhibited by the prohormone 25-hydroxyvitamin D3. Carcinogenesis. 2004; 25:1015-1026.
16. Persons KS, Eddy VJ, Chadid S, Deoliveira R, Saha AK, Ray R. Anti-growth effect of 1,25-dihydroxyvitamin D3-3-bromoacetate alone or in combination with 5-amino-imidazole-4-carboxamide-1-beta-4-ribofuranoside in pancreatic cancer cells. Anticancer Res. 2010; 30:1875-1880.
17. Afzal S, Bojesen SE, Nordestgaard BG. (2013). Low plasma 25-hydroxyvitamin D and risk of tobacco-related cancer. (United States: American Association for Clinical Chemistry Inc). pp. 771-780.
18. Ananthakrishnan AN, Cheng S, Cai T, Cagan A, Gainer VS, Szolovits P, Shaw SY, Churchill S, Karlson EW, Murphy SN, Kohane I, Liao KP. (2014). Association between reduced plasma 25-hydroxy vitamin D and increased risk of cancer in patients with inflammatory bowel diseases. (United States: W.B. Saunders), pp. 821-827.
19. Cho M, Peddi PF, Ding K, Chen L, Thomas D, Wang J, Lockhart AC, Tan B, Wang-Gillam A. Vitamin D deficiency and prognostics among patients with pancreatic adenocarcinoma. J Transl Med. 2013; 11:206.
20. Genkinger JM, Wang M, Li R, Albanes D, Anderson KE, Bernstein L, van den Brandt PA, English DR, Freudenheim JL, Fuchs CS, Gapstur SM, Giles GG, Goldbohm RA, et al. Dairy products and pancreatic cancer risk: a pooled analysis of 14 cohort studies. Ann Oncol. 2014; 25:1106-1115.
21. Haas M, Kern C, Kruger S, Michl M, Modest DP, Giessen C, Schulz C, von Einem JC, Ormanns S, Laubender RP, Holdenrieder S, Heinemann V, Boeck S. Assessing novel prognostic serum biomarkers in advanced pancreatic cancer: the role of CYFRA 21-1, serum amyloid A, haptoglobin, and 25-OH vitamin D3. Tumour Biol. 2015; 36:2631-2640.
22. McGovern EM, Lewis ME, Niesley ML, Huynh N, Hoag JB. Retrospective analysis of the influence of 25-hydroxyvitamin D on disease progression and survival in pancreatic cancer. Nutr J. 2016; 15:17.
23. Piper MR, Freedman DM, Robien K, Kopp W, Rager H, Horst RL, Stolzenberg-Solomon RZ. Vitamin D-binding protein and pancreatic cancer: a nested case-control study. Am J Clin Nutr. 2015; 101:1206-1215.
24. Stolzenberg-Solomon RZ, Jacobs EJ, Arslan AA, Qi D, Patel AV, Helzlsouer KJ, Weinstein SJ, McCullough ML, Purdue MP, Shu XO, Snyder K, Virtamo J, Wilkins LR, et al. Circulating 25-hydroxyvitamin D, risk of pancreatic cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010; 172:81-93.
25. Van Loon K, Owzar K, Jiang C, Kindler HL, Mulcahy MF, Niedzwiecki D, O'Reilly EM, Fuchs C, Innocenti F, Venook AP. 25-Hydroxyvitamin D levels and survival in advanced pancreatic cancer: findings from CALGB 80303 (Alliance). J Natl Cancer Inst. 2014; 106.
26. Waterhouse M, Risch HA, Bosetti C, Anderson KE, Petersen GM, Bamlet WR, Cotterchio M, Cleary SP, Ibiebele TI, La Vecchia C, Skinner HG, Strayer L, Bracci PM, et al. Vitamin D and pancreatic cancer: a pooled analysis from the Pancreatic Cancer Case-Control Consortium. Ann Oncol. 2015; 26:1776-1783.
27. Wolpin BM, Ng K, Bao Y, Kraft P, Stampfer MJ, Michaud DS, Ma J, Buring JE, Sesso HD, Lee IM, Rifai N, Cochrane BB, Wactawski-Wende J, et al. Plasma 25-hydroxyvitamin D and risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2012; 21:82-91.
28. Yuan C, Qian ZR, Babic A, Morales-Oyarvide V, Rubinson DA, Kraft P, Ng K, Bao Y, Giovannucci EL, Ogino S, Stampfer MJ, Gaziano JM, Sesso HD, et al. Prediagnostic plasma 25-hydroxyvitamin D and pancreatic cancer survival. J Clin Oncol. 2016; 34:2899-2905.
29. Chen GC, Zhang ZL, Wan Z, Wang L, Weber P, Eggersdorfer M, Qin LQ, Zhang W. Circulating 25-hydroxyvitamin D and risk of lung cancer: a dose-response meta-analysis. Cancer Causes Control. 2015; 26:1719-1728.
30. Liao Y, Huang JL, Qiu MX, Ma ZW. Impact of serum vitamin D level on risk of bladder cancer: a systemic review and meta-analysis. Tumour Biol. 2015; 36:1567-1572.
31. Grant WB, Peiris AN. Differences in vitamin D status may account for unexplained disparities in cancer survival rates between African and white Americans. Dermatoendocrinol. 2012; 4:85-94.
32. Gaksch M, Jorde R, Grimnes G, Joakimsen R, Schirmer H, Wilsgaard T, Mathiesen EB, Njolstad I, Lochen ML, Marz W, Kleber ME, Tomaschitz A, Grubler M, et al. Vitamin D and mortality: individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS One. 2017; 12:e0170791.
33. Grant WB, Karras SN, Bischoff-Ferrari HA, Annweiler C, Boucher BJ, Juzeniene A, Garland CF, Holick MF. Do studies reporting 'U'-shaped serum 25-hydroxyvitamin D-health outcome relationships reflect adverse effects? Dermatoendocrinol. 2016; 8:e1187349.
34. Tretli S, Schwartz GG, Torjesen PA, Robsahm TE. Serum levels of 25-hydroxyvitamin D and survival in Norwegian patients with cancer of breast, colon, lung, and lymphoma: a population-based study. Cancer Causes Control. 2012; 23:363-370.
35. Mathieu C, Adorini L. The coming of age of 1,25-dihydroxyvitamin D(3) analogs as immunomodulatory agents. Trends Mol Med. 2002; 8:174-179.
36. Iqbal S, Naseem I. Pancreatic cancer control: is vitamin D the answer? Eur J Cancer Prev. 2016; 25:188-195.
37. Barreto SG, Neale RE. Vitamin D and pancreatic cancer. Cancer Lett. 2015; 368:1-6.
38. Kawa S, Yoshizawa K, Tokoo M, Imai H, Oguchi H, Kiyosawa K, Homma T, Nikaido T, Furihata K. Inhibitory effect of 220-oxa-1,25-dihydroxyvitamin D3 on the proliferation of pancreatic cancer cell lines. Gastroenterology. 1996; 110:1605-1613.
39. Pettersson F, Colston KW, Dalgleish AG. Differential and antagonistic effects of 9-cis-retinoic acid and vitamin D analogues on pancreatic cancer cells in vitro. Br J Cancer. 2000; 83:239-245.
40. Chiang KC, Chen TC. Vitamin D for the prevention and treatment of pancreatic cancer. World J Gastroenterol. 2009; 15:3349-3354.
41. Cordero JB, Cozzolino M, Lu Y, Vidal M, Slatopolsky E, Stahl PD, Barbieri MA, Dusso A. 1,25-Dihydroxyvitamin D down-regulates cell membrane growth- and nuclear growth-promoting signals by the epidermal growth factor receptor. J Biol Chem. 2002; 277:38965-38971.
42. Wu Y, Craig TA, Lutz WH, Kumar R. Identification of 1 alpha,25-dihydroxyvitamin D3 response elements in the human transforming growth factor beta 2 gene. Biochemistry. 1999; 38:2654-2660.
43. Mathieu C, Gysemans C, Giulietti A, Bouillon R. Vitamin D and diabetes. Diabetologia. 2005; 48:1247-1257.
44. Maestro B, Molero S, Bajo S, Davila N, Calle C. Transcriptional activation of the human insulin receptor gene by 1,25-dihydroxyvitamin D(3). Cell Biochem Funct. 2002; 20:227-232.
45. Hennig R, Ding XZ, Adrian TE. On the role of the islets of Langerhans in pancreatic cancer. Histol Histopathol. 2004; 19:999-1011.
46. Skinner HG. Vitamin D for the treatment and prevention of pancreatic cancer. Cancer Biol Ther. 2008; 7:437-439.
47. Blanke CD, Beer TM, Todd K, Mori M, Stone M, Lopez C. Phase II study of calcitriol-enhanced docetaxel in patients with previously untreated metastatic or locally advanced pancreatic cancer. Invest New Drugs. 2009; 27:374-378.
48. Barreto SG, Ramadwar MR, Shukla PJ, Shrikhande SV. Vitamin D3 in operable periampullary and pancreatic cancer: perioperative outcomes in a pilot study assessing safety. Pancreas. 2008; 36:315-317.
49. Beer TM, Ryan CW, Venner PM, Petrylak DP, Chatta GS, Ruether JD, Redfern CH, Fehrenbacher L, Saleh MN, Waterhouse DM, Carducci MA, Vicario D, Dreicer R, et al. Double-blinded randomized study of high-dose calcitriol plus docetaxel compared with placebo plus docetaxel in androgen-independent prostate cancer: a report from the ASCENT Investigators. J Clin Oncol. 2007; 25:669-674.
50. Sherman MH, Yu RT, Engle DD, Ding N, Atkins AR, Tiriac H, Collisson EA, Connor F, Van Dyke T, Kozlov S, Martin P, Tseng TW, Dawson DW, et al. Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell. 2014; 159:80-93.
51. Kawa S, Nikaido T, Aoki Y, Zhai Y, Kumagai T, Furihata K, Fujii S, Kiyosawa K. Vitamin D analogues up-regulate p21 and p27 during growth inhibition of pancreatic cancer cell lines. Br J Cancer. 1997; 76:884-889.
52. Schwartz GG, Eads D, Naczki C, Northrup S, Chen T, Koumenis C. 19-nor-1 alpha,25-dihydroxyvitamin D2 (paricalcitol) inhibits the proliferation of human pancreatic cancer cells in vitro and in vivo. Cancer Biol Ther. 2008; 7:430-436.
53. Davis-Yadley AH, Malafa MP. Vitamins in pancreatic cancer: a review of underlying mechanisms and future applications. Adv Nutr. 2015; 6:774-802.
54. Crowe FL, Steur M, Allen NE, Appleby PN, Travis RC, Key TJ. Plasma concentrations of 25-hydroxyvitamin D in meat eaters, fish eaters, vegetarians and vegans: results from the EPIC-Oxford study. Public Health Nutr. 2011; 14:340-346.
55. Chiang KC, Yeh CN, Chen TC. Vitamin d and pancreatic cancer-an update. Cancers (Basel). 2011; 3:213-226.
56. Bao Y, Ng K, Wolpin BM, Michaud DS, Giovannucci E, Fuchs CS. Predicted vitamin D status and pancreatic cancer risk in two prospective cohort studies. Br J Cancer. 2010; 102:1422-1427.
57. Stolzenberg-Solomon RZ, Hayes RB, Horst RL, Anderson KE, Hollis BW, Silverman DT. Serum vitamin D and risk of pancreatic cancer in the prostate, lung, colorectal, and ovarian screening trial. Cancer Res. 2009; 69:1439-1447.
58. Stolzenberg-Solomon RZ, Vieth R, Azad A, Pietinen P, Taylor PR, Virtamo J, Albanes D. A prospective nested case-control study of vitamin D status and pancreatic cancer risk in male smokers. Cancer Res. 2006; 66:10213-10219.
59. Grant WB. Critique of the U-shaped serum 25-hydroxyvitamin D level-disease response relation. Dermatoendocrinol. 2009; 1:289-293.
60. Need AG, Kemp A, Giles N, Morris HA, Horowitz M, Nordin BE. Relationships between intestinal calcium absorption, serum vitamin D metabolites and smoking in postmenopausal women. Osteoporos Int. 2002; 13:83-88.
61. Chapurlat RD, Ewing SK, Bauer DC, Cummings SR. Influence of smoking on the antiosteoporotic efficacy of raloxifene. J Clin Endocrinol Metab. 2001; 86:4178-4182.
62. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Tobacco smoke and involuntary smoking. IARC Monogr Eval Carcinog Risks Hum. 2004; 83:1-1438.
63. Tran B, Whiteman DC, Webb PM, Fritschi L, Fawcett J, Risch HA, Lucas R, Pandeya N, Schulte A, Neale RE; Queensland Pancreatic Cancer Study Group. Association between ultraviolet radiation, skin sun sensitivity and risk of pancreatic cancer. Cancer Epidemiol. 2013; 37:886-892.
64. Moukayed M, Grant WB. Molecular link between vitamin D and cancer prevention. Nutrients. 2013; 5:3993-4021.
65. Kinoshita S, Wagatsuma Y, Okada M. Geographical distribution for malignant neoplasm of the pancreas in relation to selected climatic factors in Japan. Int J Health Geogr. 2007; 6:34.
66. Neale RE, Youlden DR, Krnjacki L, Kimlin MG, van der Pols JC. Latitude variation in pancreatic cancer mortality in Australia. Pancreas. 2009; 38:387-390.
67. Grant WB. Role of solar UVB irradiance and smoking in cancer as inferred from cancer incidence rates by occupation in Nordic countries. Dermatoendocrinol. 2012; 4:203-211.
68. Grant WB. Effect of follow-up time on the relation between prediagnostic serum 25-hydroxyvitamin D and all-cause mortality rate. Dermatoendocrinol. 2012; 4:198-202.
69. Grant WB. Effect of interval between serum draw and follow-up period on relative risk of cancer incidence with respect to 25-hydroxyvitamin D level: implications for meta-analyses and setting vitamin D guidelines. Dermatoendocrinol. 2011; 3:199-204.
70. Grant WB. 25-hydroxyvitamin D and breast cancer, colorectal cancer, and colorectal adenomas: case-control versus nested case-control studies. Anticancer Res. 2015; 35:1153-1160.
71. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010; 25:603-605.
72. Zhang J, Yu KF. What's the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998; 280:1690-1691.
73. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007; 8:16.
74. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557-560.
75. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ (Clinical research ed). 1997; 315:629-634.
76. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088-1101.

